Abstract
Bariatric surgery (BS) effectively manages severe obesity, but postoperative dietary restrictions often lead to nutrient deficiencies, particularly in the first year. This study evaluated dietary intake and serum nutrient status at 6 and 12 months post-BS in 133 patients (sleeve gastrectomy/gastric bypass, 2022–2023). Nutritional assessments were conducted using 24-hour dietary recall, comparing results with the Guidelines for Medical Nutritional Treatment of Overweight/Obesity in China (2021). Total energy intake increased significantly from 6 to 12 months (116.2 ± 433.1 kcal/d, P = 0.02). Carbohydrate contribution rose by 3.0 ± 13.6 energy percentage (en%, P = 0.01), while fat (-1.6 ± 8.9 en%, P = 0.04) and protein (-1.4 ± 6.3 en%, P = 0.01) contributions decreased. Mean protein intake remained below recommendations at both time points (53.5 ± 22.1 g/d vs. 56.4 ± 19.9 g/d), with only 59.4% (6 months) and 53.4% (12 months) meeting criteria. Mean change in consumption from 3 to 6 months, reflecting significant increases, was observed for cereal (24.7 ± 81.1 g/d, P = 0.001), vegetables (57.7 ± 159.8 g/d, P < 0.001) and vegetable oil (2.7 ± 5.9 g/d, P < 0.001). The most common deficiencies in postoperative serum nutrients were those of 25 hydroxyvitamin D (91.7%), ferritin (27.1%), albumin (14.3%) and hemoglobin (12.8%) at 12 months after surgery. Regression analysis showed that gender, surgical type, fat intake and protein powder use were independent predictors of postoperative protein intake. In conclusion, patients demonstrated inadequate nutrient intake and persistent serum deficiencies during the year following surgery. Close monitoring of dietary adherence remains critical throughout the postoperative phase.
Similar content being viewed by others
Introduction
Bariatric surgery (BS) is broadly recognized as the most effective approach for addressing morbid obesity, providing substantial weight reduction, resolution of related comorbidities, and significant enhancements in overall well-being1,2,3. Although bariatric surgery is highly effective for weight reduction, it induces considerable alterations in the anatomy and physiology of the gastrointestinal tract. These changes can disrupt crucial processes related to nutrient intake, digestion and absorption4. Postoperative factors such as gastrointestinal symptoms, food intolerance, as well as alterations in appetite, taste and smell can contribute to insufficient dietary consumption, potentially leading to nutritional complications like anemia, vitamin deficiency, and protein malnutrition5,6. Nutritional deficiencies, especially involving iron, folate, vitamin B12 and vitamin D, are commonly observed in this patient population7. Some researchers have suggested that multivitamin supplementation alone may be inadequate for preventing nutrient deficiency8,9, thereby making personalized dietary counseling essential to enhance nutritional status following BS.
BS has become a critical therapeutic approach for obesity in China, as the prevalence of moderate to severe obesity is rising at an alarming rate10. However, research on the postoperative nutritional status and dietary patterns of Chinese patients remains very limited, with most studies still focusing on Western populations. It has been reported that energy intake typically decreases by approximately 45%–60% within 6 months11,12,13,14. This reduction may not only result from smaller portion sizes, but also reflect alterations in the nutrient profile of the diet12,15,16. Enhancing dietary quality is crucial for sustaining nutritional well-being and realizing long-term advantages from the surgery. However, achieving better diet quality remains challenging, as poor dietary habits are commonly observed in this population9,11,15,17,18,19. Only a subset of studies has reported favorable changes in dietary pattern post-BS20,21. The conclusions regarding postoperative dietary changes are inconsistent across different time points. Cultural, dietary, and genetic differences between Chinese and Western populations further highlight the need to understand specific nutritional challenges facing Chinese post-surgical patients.
This study aims to evaluate the dietary and nutritional changes in Chinese patients at 6 and 12 months following BS. The findings will provide valuable insights into the nutritional needs of Chinese bariatric surgery patients and inform the development of targeted nutritional support strategies to optimize their long-term health and well-being.
Results
Anthropometric changes
Table 1 shows the characteristics and anthropometric parameters of the population (n = 133). The cohort had a mean age of 32.9 ± 8.5 years, comprised of 62.4% females, with 55.6% being married and 52.6% having received education beyond 12 years. A total of 17.2% reported a history of smoking, while 68.4% underwent SG. Significant further reductions in body weight (82.0 ± 18.8 kg to 76.0 ± 16.3 kg, P < 0.001), BMI (28.8 ± 5.3 kg/m2 to 26.8 ± 4.6 kg/m2, P < 0.001) and body fat percentage (33.63 ± 8.2% to 30.01 ± 8.6%, P < 0.001) were observed from 6 to 12 months post-surgery. Correspondingly, the percentage of fat-free mass increased from 66.4 ± 8.2% to 69.9 ± 8.6% (P < 0.001). Additionally, significant improvements were recorded in waist-to-hip ratio (0.9 ± 0.1 to 0.8 ± 0.1, P < 0.001) and visceral fat area (133.6 ± 52.5 cm2 to 107.2 ± 45.2 cm2, P < 0.001). Excess weight loss (%) progressively increased from 76.6 ± 27.7% at 6 months to 89.3 ± 27.9% at 12 months (P < 0.001).
Changes in nutritional status
Table 2 presents the pre- and postoperative serum nutrient status of patients. Between 6 and 12 months postoperatively, albumin levels decreased from 44.3 ± 3.4 g/L to 42.9 ± 3.2 g/L (P < 0.05), with deficiency rates rising from 3.8% to 14.3% (P < 0.01). Hemoglobin also declined from 136.9 ± 15.8 g/L to 132.1 ± 18.0 g/L (P < 0.05), with deficiencies increasing from 5.3% to 12.8% (P < 0.01). Ferritin levels dropped from 138.9 ± 142.0 ng/mL to 107.2 ± 118.1 ng/mL (P < 0.05), with deficiency rates rising from 18.0 to 27.1% (P < 0.01). Vitamin D deficiency persisted at high levels, with rates of 94.7% at 6 months and 91.7% at 12 months. The PNI showed a slight reduction from 55.4 ± 5.1 to 54.2 ± 4.8 (P < 0.05). There was no notable difference in the levels of folate, vitamin B12, or 25 (OH) D at the two points. Moreover, there were significant differences in the mean concentrations of serum nutritional indices (albumin, hemoglobin, folate, 25 (OH) D and PNI) in patients before and after surgery (P < 0.05).
Postoperative dietary intake and nutritional adequacy
Overall, as shown in Table 3, the mean intake of energy, carbohydrates, fat, and protein in postoperative patients decreased significantly compared with baseline. Regarding postoperative dietary intake changes, total energy intake increased from 993.8 ± 325.6 kcal at 6 months to 1101.0 ± 290.5 kcal at 12 months (P = 0.02). Carbohydrate intake rose from 106.8 ± 51.9 g to 125.7 ± 46.2 g (P = 0.02), with the carbohydrate energy percentage increasing from 41.8 ± 11.7% to 44.8 ± 8.0% (P = 0.01). Fat intake increased from 37.2 ± 11.4 g to 40.4 ± 9.6 g (P = 0.01), although the percentage of energy from fat decreased from 34.7 ± 7.7% to 33.1 ± 5.2% (P = 0.04). Protein intake remained below the recommended level of 60 g (53.5 ± 22.1 g to 56.4 ± 19.9 g, P = 0.2), and the protein energy percentage decreased from 21.5 ± 5.1% to 20.1 ± 4.2% (P = 0.01). Furthermore, the proportion of protein powder used declined from 66.9% to 48.1% (P = 0.002), accompanied by a reduction in the amount of protein powder (15.4 ± 13.6 g to 10.7 ± 14.3 g, P = 0.007).
Regarding carbohydrates, the percentage of patients meeting the recommended range increased significantly from 6 to 12 month post-surgery (P < 0.05), while those below the threshold concurrently declined (P < 0.05). For fat, the proportion of patients with intake above the recommended threshold remained high, with 57.1% at 6 months and 58.6% at 12 months(P > 0.05). In contrast, protein intake showed a declining trend in the percentage of patients meeting the recommended standards (59.4% to 53.4%, P > 0.05), with an increase in those below the threshold by 12 months (Fig. 1).
Percentages of individuals with nutrient intake meeting, below and above the recommended standards at 6 and 12 months post-operation. Abbreviation: T6 or T12, 6 or 12 months post-operation. Different colors represent the proportion of postoperative patients whose nutritional energy supply ratio was within the recommended range (green), lower than (orange) or higher than (red) the recommended threshold. Significant difference between time points: *P < 0.05.
Table 4 and Fig. 2 present the consumption of food groups according to the the Chinese Nutritional Intervention Specification of Calorie-restricted Diet. Overall, compared with baseline, the intake of cereals, vegetables and vegetable oils in postoperative patients decreased significantly (P < 0.05), while their intake of meat, eggs and dairy products increased from pre-operation (P < 0.05). Regarding changes in food groups intake at 6 and 12 months postoperatively, significant increases were observed in the consumption of cereals (85.9 ± 64.3 g to 110.6 ± 55.8 g, P = 0.001). The intakes of vegetables and vegetable oils also increased significantly (57.7 ± 159.8 g, P < 0.001; 2.7 ± 5.9 g, P < 0.001). Meanwhile, the proportion of participants meeting the recommendation for vegetables rose from 17.3% to 28.6% (P < 0.05), while the percentage adhering to the guidelines for vegetable oils fell from 97.7 to 85.0% (P < 0.05). The intakes of meat and soybeans showed a slight upward trend (P > 0.05), but the consumption of eggs, low-fat milk, fruits and nuts exhibited a declining trend (P > 0.05). This bar chart also demonstrated that at both time points, the adherence to the recommendations for low-fat milk and soybeans was relatively low (< 10%). A relatively high percentage of participants (over 80%) followed the recommendations for consuming fruits, nuts, and vegetable oil.
Adherence to the Chinese dietary guidelines at 6 and 12 months after BS. Abbreviation: T6 or T12, 6 or 12 months post-operation. Different colors represent the proportion of postoperative patients whose dietary intake was within (green) or outside (red) the recommended range. Significant difference between time points: *P < 0.05.
After adjusting for relevant covariates, we found that male patients and those who underwent GB had a lower risk of protein intake inadequacy at 6 months post-surgery; similar results were not observed at 12 months post-surgery. Additionally, at both 6 and 12 months post-surgery, increased fat intake and protein powder consumption were positively associated with adequate protein intake (Table 5).
Discussion
The objective of this research was to evaluate dietary intake adequacy and nutritional changes in the Chinese population at 6 and 12 months following BS. Our results revealed a high prevalence of deficiencies in vitamin D, ferritin, hemoglobin and albumin, while the average protein intake remained low. Average protein intake was below the 60 g/d target, with over 50% of patients exceeding recommended fat energy proportions at both points. From 6 to 12 months post-surgery, although patients increased their intake of cereals and vegetables, adherence to the recommended intakes of low-fat milk and soy-based foods was extremely low. These findings underscore the necessity for targeted nutritional interventions to address these deficiencies and enhance overall dietary quality.
BS is closely linked to an elevated risk of deficiencies in vitamins, minerals and trace elements, rendering patients more susceptible to conditions such as anemia, osteoporosis and protein malnutrition5,6,22. In the present study, albumin, hemoglobin, and ferritin levels declined significantly from 6 to 12 months postoperatively, coupled with rising deficiency rates for these nutrients. Hypoalbuminemia affected 14.3% of participants at 1 year post-surgery, consistent with previous reports of 3–18% prevalence in postoperative populations23. Moreover, we found patients exhibited a high prevalence of hemoglobin and ferritin deficiencies, with anemia rates of 5.3% and 12.8% at 6 and 12 months after surgery, respectively, and ferritin deficiency rates of 18.0% and 27.1% at the same time points. A systematic review reported that anemia risk doubled 12 months post-surgery24. Many patients’ experience of red meat intolerance led to the limitation of their heme iron intake. Malabsorption of iron and vitamin B12 also likely contributes to these deficiencies25,26. The popularity of tea consumption in China27 could further exacerbate iron malabsorption. Notably, nearly all participants had vitamin D deficiency in this study, consistent with prior reports of 24–85% prevalence rates26. Population differences, regional variations, and disparities in the use of nutritional supplements may be significant factors contributing to the differences in deficiency rates. The PNI, a composite score integrating serum albumin and lymphocyte count28, demonstrated a biphasic trend in our cohort. PNI transiently increased from the baseline (53.4 ± 4.6) to 55.4 ± 5.1 at 6 months postoperatively, and then declined to 54.2 ± 4.8 at 12 months. This trend paralleled the changes in albumin levels, suggesting that protein malnutrition- potentially exacerbated by inadequate dietary protein intake (e.g., the average postoperative protein intake failing to meet recommended levels) or malabsorption -might contribute to the long-term deterioration of PNI. Notably, despite mean PNI remaining above the traditional threshold, the rising prevalence of PNI deficiency (from 1.5% at 6 months to 3.8% at 12 months) signals underlying nutritional vulnerability, and such subtle changes may reflect impaired immune function. Future studies should correlate the trajectory of PNI changes with clinical outcomes (e.g., wound dehiscence, infection rates) to validate its predictive value in this population. It is noteworthy that nutritional deficiencies are common in Chinese bariatric candidates, with vitamin D deficiency being the most serious (76.9–78.8%), followed by albumin (8.5–13.4%), folate (0–20%) and vitamin B12 (0–5%). The prevalence of iron deficiency and anemia was 0–5% and 0–4% in males, versus 0.7–15% and 3.7–15% in females29,30,31,32. Deficiencies of nutrients before surgery could be predictors for deficiencies 1 year after surgery29. Thus, routine evaluation of preoperative nutritional abnormalities could contribute to postoperative nutrient balance.
The majority of studies showed a reduction in energy intake following surgery, as well as inadequate intakes of protein and an excessive intake of fats15. Heusschen et al33. documented pre-surgical energy intake (1877 kcal/d) and macronutrient distribution (41.0% carbohydrates, 37.3% fat, 18.2% protein) in 107 bariatric patients. At 6 months post-surgery, energy significantly decreased by 512 kcal/d with protein proportion rising to 19.3%. Our cohort exhibited an average reduction in energy intake of approximately 1000 kcal postoperatively, with the carbohydrate energy contribution ratio decreasing significantly compared with preoperative levels, while those of fat and protein increased. Besides, total energy and carbohydrate intake percentages rose modestly from the 6th to the 12th month post-surgery, while fat and protein energy contributions declined correspondingly. Regarding the ideal post-surgical diet, some researchers suggest that carbohydrate, fat, and protein intakes should account for 40–45%, ≤ 35% and 20–25% of total energy intake, respectively15. Our recommendation for a high-protein dietary pattern is based on the Chinese Guidelines for Medical Nutrition Therapy in Overweight/Obesity (2021) and the Nutritional Intervention Specification of Calorie-restricted Diet (T/CNSS 016–2022). This pattern suggests energy contributions from carbohydrates, fats and proteins at 40–60%, 20–30% and 20–30% respectively, and has been demonstrated to significantly enhance satiety and improve dietary compliance for sustained weight loss efficacy34,35,36. The traditional Chinese diet is predominantly rice- and wheat-based37, possibly leading to poor adherence to the relatively low-carbohydrate, high-protein diet post-surgery. Notably, the energy contribution from fat exceeded the recommendation (20–30%) at both 6 and 12 months post-surgery, and more than half of the individuals exceeded the recommended fat intake. In line with this, a majority of studies observed an excessive intake of fat following surgery15. The research conducted by Kapoor et al. also revealed that avoidance of high-fat foods at ad libitum buffets was prevalent but not universal38. Furthermore, Chinese people believe that meat broth is more nutritious, and consuming fatty meat-based soups post-surgery tends to lead to higher fat intake. Besides, traditional Chinese cuisine commonly employs high-temperature stir-frying and deep-frying methods, which involve the use of substantial amounts of oil. Moreover, only 59.4% and 53.4% of the participants meet the recommended protein energy percentage at the 6th and 12th months. Protein intake was below the recommended goal of ≥ 60 g/d39 at both intervals, which was in line with previous research11,13,14,33,40,41. Sufficient protein intake is critical for maintaining fat free mass, preventing alopecia, supporting wound healing, and reducing edema risk41. Additionally, protein may enhance satiety and be a crucial factor in sustaining weight loss following surgery12. Hence, protein supplementation or dietary enrichment may assist in reaching the minimum recommended protein intake and enhance the quality and quantity of postoperative weight loss.
Evidence showed critical changes in food group consumption from baseline to 6 months post-surgery33. Marked deteriorations occurred in fiber-rich plant foods, with vegetables and whole grains declining substantially, while fruit persistently remained below recommended levels. Among animal-derived proteins, dairy consumption showed a modest increase. Plant-based proteins exhibited no improvement, with legumes and nuts persistently inadequate. Similar changes were also observed in our study, where dietary changes from pre- to post-surgery were primarily characterized by insufficient fiber intake and inadequate protein sources. Suboptimal cereal intake post-surgery may stem from food intolerance to rice and pasta15,42,43, compounded by prioritizing protein over grains to restrict total energy intake, as recommended in postoperative guidelines. Prior research has documented low fiber intake in bariatric patients9,11,33,43,44,45, which was associated with constipation—a prevalent postoperative complication43,44. Consistent with these findings, we found that at 6 and 12 months post-surgery, the average intake of cereals, soy products, vegetables and fruits remained below recommended levels, all of which were significant sources of dietary fiber. Regarding animal-derived protein sources, apart from meat, the average consumption of eggs and dairy products remained below recommended levels during both periods. This may partly explain why the patients’ average protein intake could not meet the goal of 60 g/d. Approximately 70–100% of the Asian adult population is lactase nonpersistent46, and this high prevalence of lactose intolerance may correlate with insufficient dairy intake among Chinese people. Soy and nut consumption remained below recommended levels, suggesting that patients could benefit from increasing plant-based protein intake. Moreover, our study revealed that 66.9% of patients reported using protein powder supplementation at 6 months postoperatively and further decreased at 12 months (48.1%). Schollenberger et al. demonstrated that protein supplementation may enhance body composition by preserving lean body mass and promoting fat loss47. Consequently, the use of protein supplements serves as a practical way to reach the recommended daily protein intake. It was also noted that dietary fat intake was positively associated with adequate protein intake. We advocate for a high-protein diet, which inherently contains higher fat due to the inclusion of animal-based sources (e.g., meat, dairy, eggs) and plant-based alternatives (e.g., nuts, seeds). Additionally, cooking methods for these foods often involve added fats. Males and patients who underwent GB had a lower risk of inadequate protein intake, but this phenomenon was only observed at 6 months post-surgery. In clinical practice, we observed that males preferred meat, protein powder, and other high-protein foods in the early postoperative period and showed better tolerance to such foods than females. Additionally, long-term dietary behaviors and lifestyle habits gradually converged, which may have led to no significant intergroup differences (e.g., gender and surgical type) in the risk of inadequate protein intake. However, the above deductions warrant further investigation in future studies. To sum up, our research reflected the changes in eating pattern and persistent gaps in meeting nutritional guidelines, possibly due to dietary preferences, intolerance to specific types of food, food restriction, and discomfort with eating42,48.
Indeed, patients need to adapt to reduced gastric volume and declined food tolerance, particularly in the initial postoperative year. According to studies with a 1-year follow-up period, postoperative serum nutrient status of patients was similar in SG and GB groups. These patients were advised to take multivitamin/mineral supplements routinely after surgery29,49. In our study, the incidence of anemia and ferritin deficiency after GB was relatively higher than that in the SG group, with no significant differences observed in the remaining nutritional indices (Table S3). We further analyzed differences in nutritional indices between patients who took multivitamin tablets postoperatively and those who did not, and no statistically significant differences were observed (Table S4). Regular nutritional follow-up and dietary consultation are crucial components of postoperative management, as short-term dietary intake changes correlate with long-term weight outcomes12,45,50. Furthermore, prioritizing dietary quality is critical for optimizing weight outcomes. Masood et al.51 proposed that weight regain following BS may not primarily result from overeating but rather from poor food choices. Numerous studies have also identified a correlation between poor dietary quality and postoperative weight regain during the long-term follow-up period15,50,52,53. Overall, poor diet quality is commonly reported among post-BS patients15,17,52,53,54,55. This underscored that inadequate nutritional education and guidance may lead to unhealthy eating and lifestyle habits, thereby increasing the risk of weight regain and undermining the efficacy of BS.
An advantage of this study was its focus on nutrient composition through analysis of food categories rather than single nutrients, providing a more comprehensive representation of overall diet. Additionally, we evaluated changes in dietary pattern within the first year after BS based on Chinese dietary guidelines. Given that the majority of postoperative nutrition-related research has focused on the Western population, this evaluation offered valuable insights for the development of personalized nutrition plans tailored to the Chinese population. Nevertheless, several limitations must be acknowledged. First, our study lacked a non-surgical comparison group and randomized intervention, so we could not definitively attribute the observed changes in nutritional status solely to surgery versus general lifestyle modifications. Second, the observational design of this study restricted the capacity to determine causal relationships between variables. Third, the 24-h dietary recall may not accurately represent usual dietary intake, potentially leading to an underestimation of infrequently consumed foods. Underestimation of energy intake represented a prevailing bias in nutritional studies, especially among overweight or obese populations56,57,58. Still, it can be assumed that postoperative measurements were affected by comparable biases, and the extent of changes appeared to maintain consistency over time. Finally, this study involves a single-center cohort of Chinese patients. Given the international variations in cultural dietary habits and supplement practices, the results may not directly extrapolate to non-Chinese populations or multicenter settings.
Conclusions
In the first year following BS, inadequate nutrient intake and deficiencies were commonly observed among patients. There were still gaps in the dietary patterns of patients after surgery in meeting the nutritional guidelines. Understanding short-term changes in dietary pattern post-surgery could help address common dietary challenges. Despite dietary differences between China and Western countries, the postoperative nutrient requirement following bariatric surgery are essentially consistent and primarily based on the American Society for Metabolic and Bariatric Surgery (ASMBS) guidelines. Dietitians should refer to guidelines and develop nutrition plans tailored to the unique dietary needs of the Chinese population. Emphasizing the intake of eggs, dairy products, and plant-based protein sources such as soy products and nuts could enhance absolute protein intake, whereas increasing consumption of whole grains, vegetables and fruits should be targeted to compensate for fiber deficiency. Future investigations need to focus on identifying underlying factors influencing postoperative dietary changes and their consequences. Long-term studies should further determine whether nutrient intake patterns and deficiency states persist, deteriorate or improve over time, given the dynamic evolution of eating behavior.
Methods
Study design and sample
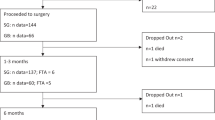
Patients who underwent either sleeve gastrectomy (SG) or gastric bypass (GB) between the years 2022 and 2023 were selected for evaluation. Participants were included if they met the following criteria: (1) aged 18–65 years, (2) a referral for follow-up at Nanjing Drum Tower Hospital. Exclusion criteria included: (1) Incomplete medical records or dietary recall data at baseline or during follow-up, (2) suffering from an eating disorder or severe intolerance to food intake, (3) severe gastrointestinal complications related to surgery, (4) chronic diseases (i.e., severe liver and renal dysfunction, tumors), (5) pregnancy during follow-up. Finally, 133 individuals were enrolled, and the sample size was comparable to that of previous research9.
Patients were arranged to attend follow-up visits with a multidisciplinary team that included a surgeon, an endocrinologist, a dietitian, an exercise medicine physician, and a psychologist. These follow-up appointments were scheduled at intervals of 1, 3, 6 and 12 months post-surgery. In general, lifelong supplementation with a multivitamin preparation (Centrum®, 1–2 tablets/d) was recommended. Calcium (Caltrate®) and vitamin D preparations (Rocaltrol®) were adjusted according to their serum levels. Iron (Sulifei®) and vitamin B12 supplementation (Methycobal®) depended on ferritin and anemia status (see detailed chemical composition in Table S5). The adherence rates to nutritional supplement use at 6 and 12 months postoperatively were as follows: multivitamin tablets, 84.9% vs 73.6%; vitamin D preparations, 74.4% vs 54.8%; calcium supplements, 82.7% vs 71.4%; iron supplements, 12.7% vs 16.5%.
The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of Nanjing Drum Tower Hospital (ethics code: 2024-201-02, date of approval 7 May 2024). Verbal informed consent was obtained from the participants. Verbal consent was obtained rather than written because conducting dietary surveys, nutritional guidance, and anthropometric measurements during follow-up was routine responsibility of physicians.
Demographic information and measurements
Data on socio-demographic factors such as age, gender, educational level and marital status, along with health-related details like the type of surgery and smoking habits, were collected from electronic patient records. Anthropometric data including body weight, height, waist and hip circumferences were collected before surgery and at the 6th and 12th months following BS. Body mass index (BMI) and the excess weight loss (%) were calculated, with ideal weight defined as BMI of 24 kg/m2. Additionally, the body composition analyzer InBody 770 (Biospace, Seoul, Korea) was employed to assess weight, body fat, fat free mass, and visceral fat area.
Dietary assessment
During the follow-up period, individualized dietary guidance was provided to each patient by the same dietitian. The dietary progression adhered to a staged approach: Week 1: A clear liquid diet was recommended; Weeks 2–3: Transition to a full liquid diet; Weeks 4–12: Advance to a semi-liquid diet, emphasizing soft, bland, and easily digestible foods; Starting from month 3: Gradual transition to solid soft foods, ultimately resuming a low-fat regular diet.
The dietitian was responsible for providing dietary consultations to ensure patients adhered to the nutritional regimen: 1. It is recommended to consume more than 1500 ml/d; 2. The target protein intake is 60–120 g/d or 1.5–2.0 g/kg of ideal body weight to maintain muscle mass; 3. Avoid drinking water or soup during meals, with fluids consumed between meals or 30–45 min afterward; 4. Consume 3–6 meals daily, chewing thoroughly before swallowing. each of the three main meals should last approximately 30 min; 5. Avoid hyperosmolar food or liquid (e.g., desserts, sugary beverages) to prevent dumping syndrome. Additionally, limit the intake of stimulants such as strong tea, spicy foods and alcohol, as well as high-fat foods like fried items, meat broths, and animal skin; 6. Consume nutritional supplements, such as multivitamins, vitamin D and calcium, on a regular basis.
The dietitian monitored patients’ dietary practices and evaluated adherence through either phone calls or face-to-face consultations during clinic visits. Compliance with dietary consumption was evaluated via phone interviews or by scheduling meetings with the study dietitian for counseling. Data on dietary intake were gathered through a 24-h dietary recall or food diaries following surgery. In addition to the scheduled outpatient clinic follow-up visits at specific time points (e.g., 6th and 12th month postoperatively), patients were regularly requested to report their dietary intake through online communications, food photography check-ins and telephone interviews during each follow-up interval. Instead, we employed a comprehensive assessment integrating data from multiple recall days to improve the reliability of dietary assessment. This continuous data collection strategy was designed to capture day-to-day dietary variations and minimize recall bias. Participants needed to record the types, quantities, and cooking methods of foods consumed the previous day using a form, verbal reporting or photographs. The dietitian converted the patients’ raw dietary records into standardized portions using the food exchange list (Table S1) and calculated the total energy intake as well as the proportion of macronutrients.
Evaluation of diet quality and nutritional status
The macronutrient composition was assessed based total energy, carbohydrates, fats and proteins. The appropriateness of dietary types and quantities was evaluated with reference to the Guidelines for medical nutritional treatment of overweight/obesity in China (2021) and the Nutritional Intervention Specification of Calorie-restricted Diet (T/CNSS 016-2022). Specifically, we designed an energy-restricted high-protein diet for patients 6 to 12 months post-surgery (Table S2). The energy contribution ratios of carbohydrates, fat, and protein were set at 40–60%, 20–30% and 20–30%, respectively. The diet primarily comprised these categories of food: grains and tubers, lean meats, eggs, low-fat dairy products, soy-based foods, vegetables, fruits, nuts, vegetable oils and salt. This research did not consider the intake of vitamin and mineral supplements, as its main objective was to analyze the nutritional quality of the dietary intake.
At both the 6-month and 12-month intervals post-surgery, the plasma levels of albumin, hemoglobin, ferritin, folate, vitamin B12 and 25-hydroxyvitamin D (25 (OH) D) were assessed in patients, as these micronutrients are frequently found at reduced levels in this population. Levels of vitamin B12 and folate was carried out utilizing chemiluminescent immunoassay, whereas 25 (OH) D concentrations were determined through enzyme immunoassay techniques. Ferritin levels were quantified using a human ferritin enzyme immunoassay method, and albumin concentration was performed employing bromocresol green. The calculation for the Prognostic Nutritional Index (PNI) was executed as follows: serum albumin (g/L) plus five times the total count of peripheral blood lymphocytes (× 10^9/L). Hypoalbuminemia was defined as serum levels < 35 g/L. Anaemia was characterized by a hemoglobin level < 120 g/L in men and < 110 g/L in women. Iron deficiency was defined as a ferritin level < 30 ng/mL. Folate deficiency and vitamin B12 deficiency were defined as serum levels < 4.0 ng/mL and < 203 pg/mL, respectively. Vitamin D deficiency was marked by a 25 (OH) D level of < 20 ng/mL.
Statistical analysis
Quantitative variables were reported as mean ± standard deviation (SD), and categorical data were described using percentages and frequencies. To assess the normality of the data, both histograms and the Kolmogorov–Smirnov test were utilized. Differences in anthropometric indices, dietary intake and serological indicators between 6 and 12 months post-surgery were analyzed using a paired t-test or a paired chi-square test. A reduction was represented by negative values. To assess longitudinal changes from the baseline to postoperative time points, we used repeated-measures analysis of variance (ANOVA) with Bonferroni post hoc correction. Logistic regression analysis identified predictors of protein intake adequacy after surgery. Statistical analyses were conducted utilizing SPSS version 26 (IBM Corp., Chicago, USA), with P values less than 0.05 deemed statistically significant.
Data availability
All data generated and analyzed in this study are included in this article. Further inquiries are available upon request from the corresponding authors.
References
Slomski, A. Weight loss is still substantial a decade after bariatric surgery. JAMA 328, 415. https://doi.org/10.1001/jama.2022.12728 (2022).
Konttinen, H., Sjoholm, K., Carlsson, L. M. S., Peltonen, M. & Svensson, P. A. Fifteen-year changes in health-related quality of life after bariatric surgery and non-surgical obesity treatment. Int. J. Obes. (Lond) 48, 1447–1456. https://doi.org/10.1038/s41366-024-01572-w (2024).
Miras, A. D. & le Roux, C. W. Metabolic surgery versus conventional therapy in type 2 diabetes. Lancet 397, 256–257. https://doi.org/10.1016/S0140-6736(21)00129-X (2021).
Steenackers, N. et al. Adaptations in gastrointestinal physiology after sleeve gastrectomy and Roux-en-Y gastric bypass. Lancet Gastroenterol. Hepatol. 6, 225–237. https://doi.org/10.1016/S2468-1253(20)30302-2 (2021).
Lupoli, R. et al. Bariatric surgery and long-term nutritional issues. World J. Diabetes 8, 464–474. https://doi.org/10.4239/wjd.v8.i11.464 (2017).
Osland, E., Powlesland, H., Guthrie, T., Lewis, C. A. & Memon, M. A. Micronutrient management following bariatric surgery: The role of the dietitian in the postoperative period. Ann. Transl. Med. 8, S9. https://doi.org/10.21037/atm.2019.06.04 (2020).
Gasmi, A. et al. Micronutrients deficiences in patients after bariatric surgery. Eur. J. Nutr. 61, 55–67. https://doi.org/10.1007/s00394-021-02619-8 (2022).
Gasteyger, C., Suter, M., Gaillard, R. C. & Giusti, V. Nutritional deficiencies after Roux-en-Y gastric bypass for morbid obesity often cannot be prevented by standard multivitamin supplementation. Am. J. Clin. Nutr. 87, 1128–1133. https://doi.org/10.1093/ajcn/87.5.1128 (2008).
Ziadlou, M. et al. Dietary macro- and micro-nutrients intake adequacy at 6th and 12th month post-bariatric surgery. BMC. Surg. 20, 232. https://doi.org/10.1186/s12893-020-00880-y (2020).
Pan, X. F., Wang, L. & Pan, A. Epidemiology and determinants of obesity in China. Lancet Diabetes Endocrinol. 9, 373–392. https://doi.org/10.1016/S2213-8587(21)00045-0 (2021).
Farias, G. et al. Impact of dietary patterns according to NOVA food groups: 2 y after Roux-en-Y gastric bypass surgery. Nutrition 74, 110746. https://doi.org/10.1016/j.nut.2020.110746 (2020).
Kanerva, N., Larsson, I., Peltonen, M., Lindroos, A. K. & Carlsson, L. M. Changes in total energy intake and macronutrient composition after bariatric surgery predict long-term weight outcome: findings from the Swedish Obese Subjects (SOS) study. Am. J. Clin. Nutr. 106, 136–145. https://doi.org/10.3945/ajcn.116.149112 (2017).
Raatz, S. K. et al. Reported nutrient intake over 7 years after Roux-en-Y gastric bypass in the Longitudinal Assessment of Bariatric Surgery-3 (LABS-3) psychosocial study. Surg. Obes. Relat. Dis. 16, 1022–1029. https://doi.org/10.1016/j.soard.2020.04.007 (2020).
Sherf Dagan, S. et al. Do bariatric patients follow dietary and lifestyle recommendations during the first postoperative year?. Obes. Surg. 27, 2258–2271. https://doi.org/10.1007/s11695-017-2633-6 (2017).
Zarshenas, N., Tapsell, L. C., Neale, E. P., Batterham, M. & Talbot, M. L. The relationship between bariatric surgery and diet quality: A systematic review. Obes. Surg. 30, 1768–1792. https://doi.org/10.1007/s11695-020-04392-9 (2020).
Nielsen, M. S. et al. Factors associated with favorable changes in food preferences after bariatric surgery. Obes. Surg. 31, 3514–3524. https://doi.org/10.1007/s11695-021-05374-1 (2021).
Hong, Y. R. et al. Assessment of physical activity and healthy eating behaviors among US adults receiving bariatric surgery. JAMA Netw. Open 5, e2217380. https://doi.org/10.1001/jamanetworkopen.2022.17380 (2022).
Marques da Silva, M. et al. Nutritional guidance, monitoring, and supplementation before and after bariatric surgery—Are we doing this correctly?. Nutr. Hosp. 38, 478–487. https://doi.org/10.20960/nh.03221 (2021).
Schiavo, L. et al. Long-term results of the mediterranean diet after sleeve gastrectomy. Obes. Surg. 30, 3792–3802. https://doi.org/10.1007/s11695-020-04695-x (2020).
Ullrich, J., Ernst, B., Wilms, B., Thurnheer, M. & Schultes, B. Roux-en Y gastric bypass surgery reduces hedonic hunger and improves dietary habits in severely obese subjects. Obes. Surg. 23, 50–55. https://doi.org/10.1007/s11695-012-0754-5 (2013).
Miller, G. D., Norris, A. & Fernandez, A. Changes in nutrients and food groups intake following laparoscopic Roux-en-Y gastric bypass (RYGB). Obes. Surg. 24, 1926–1932. https://doi.org/10.1007/s11695-014-1259-1 (2014).
Gasmi, A. et al. Dietary supplements and bariatric surgery. Crit. Rev. Food. Sci. Nutr. 63, 7477–7488. https://doi.org/10.1080/10408398.2022.2046542 (2023).
Thibault, R. & Pichard, C. Overview on nutritional issues in bariatric surgery. Curr Opin Clin Nutr. Metab. Care. 19, 484–490. https://doi.org/10.1097/MCO.0000000000000325 (2016).
Weng, T. C., Chang, C. H., Dong, Y. H., Chang, Y. C. & Chuang, L. M. Anaemia and related nutrient deficiencies after Roux-en-Y gastric bypass surgery: A systematic review and meta-analysis. BMJ Open 5, e006964. https://doi.org/10.1136/bmjopen-2014-006964 (2015).
Mischler, R. A. et al. Influence of diet and supplements on iron status after gastric bypass surgery. Surg. Obes. Relat. Dis. 12, 651–658. https://doi.org/10.1016/j.soard.2015.09.007 (2016).
Dogan, K. et al. Long-term nutritional status in patients following Roux-en-Y gastric bypass surgery. Clin. Nutr. 37, 612–617. https://doi.org/10.1016/j.clnu.2017.01.022 (2018).
Zhao, Y. et al. The association between tea consumption and blood pressure in the adult population in Southwest China. BMC Public Health 23, 476. https://doi.org/10.1186/s12889-023-15315-5 (2023).
Chen, G. et al. Prognostic nutritional index (PNI) and risk of non-alcoholic fatty liver disease and advanced liver fibrosis in US adults: Evidence from NHANES 2017–2020. Heliyon 10, e25660. https://doi.org/10.1016/j.heliyon.2024.e25660 (2024).
Guan, B., Yang, J., Chen, Y., Yang, W. & Wang, C. Nutritional deficiencies in Chinese patients undergoing gastric bypass and sleeve gastrectomy: Prevalence and predictors. Obes. Surg. 28, 2727–2736. https://doi.org/10.1007/s11695-018-3225-9 (2018).
Sun, L., Wang, C., Sun, W. & Wang, C. A pilot study of nutritional status prior to bariatric surgery in South China. Front. Nutr. 8, 697695. https://doi.org/10.3389/fnut.2021.697695 (2021).
Zhang, C. et al. Nutritional status in Chinese patients with obesity following sleeve gastrectomy/Roux-en-Y gastric bypass: A retrospective multicenter cohort study. Nutrients 14, 1932. https://doi.org/10.3390/nu14091932 (2022).
Zhang, C. et al. Anaemia and related nutritional deficiencies in Chinese patients with obesity, 12 months following laparoscopic sleeve gastrectomy. Diabetes Metab. Syndr. Obes. 14, 1575–1587. https://doi.org/10.2147/DMSO.S303320 (2021).
Heusschen, L. et al. Changes in nutrient composition and diet quality in the first 6 months following bariatric surgery: An observational cohort study. J. Hum. Nutr. Diet 37, 365–376. https://doi.org/10.1111/jhn.13258 (2024).
Dinu, M. et al. Effects of popular diets on anthropometric and cardiometabolic parameters: An umbrella review of meta-analyses of randomized controlled trials. Adv. Nutr. 11, 815–833. https://doi.org/10.1093/advances/nmaa006 (2020).
Santesso, N. et al. Effects of higher-versus lower-protein diets on health outcomes: A systematic review and meta-analysis. Eur. J. Clin. Nutr. 66, 780–788. https://doi.org/10.1038/ejcn.2012.37 (2012).
Johnston, B. C. et al. Comparison of weight loss among named diet programs in overweight and obese adults: A meta-analysis. JAMA 312, 923–933. https://doi.org/10.1001/jama.2014.10397 (2014).
Liu, X. & Reid, R. E. B. The prehistoric roots of Chinese cuisines: Mapping staple food systems of China, 6000 BC-220 AD. PLoS ONE 15, e0240930. https://doi.org/10.1371/journal.pone.0240930 (2020).
Kapoor, N. et al. A comparison of total food intake at a personalised buffet in people with obesity, before and 24 months after Roux-en-Y-Gastric Bypass Surgery. Nutrients 13, 3873. https://doi.org/10.3390/nu13113873 (2021).
Mechanick, J. I. et al. Clinical Practice Guidelines for the Perioperative Nutrition, Metabolic, and Nonsurgical Support of Patients Undergoing Bariatric Procedures—2019 Update: Cosponsored by American Association of Clinical Endocrinologists/American College of Endocrinology, The Obesity Society, American Society for Metabolic and Bariatric Surgery, Obesity Medicine Association, and American Society of Anesthesiologists. Obesity (Silver Spring) 28, O1–O58. https://doi.org/10.1002/oby.22719 (2020).
Ito, M. K. et al. Effect of protein intake on the protein status and lean mass of post-bariatric surgery patients: A systematic review. Obes. Surg. 27, 502–512. https://doi.org/10.1007/s11695-016-2453-0 (2017).
Steenackers, N., Gesquiere, I. & Matthys, C. The relevance of dietary protein after bariatric surgery: What do we know?. Curr. Opin. Clin. Nutr. Metab. Care. 21, 58–63. https://doi.org/10.1097/MCO.0000000000000437 (2018).
Boerlage, T. C., van de Laar, A. W., Westerlaken, S., Gerdes, V. E. & Brandjes, D. P. Gastrointestinal symptoms and food intolerance 2 years after laparoscopic Roux-en-Y gastric bypass for morbid obesity. Br. J. Surg. 104, 393–400. https://doi.org/10.1002/bjs.10419 (2017).
Grosse, C. S. & Cope, V. C. Dietary fibre intake and bowel habits after bariatric surgery: A structured literature review. Obes. Surg. 29, 2247–2254. https://doi.org/10.1007/s11695-019-03837-0 (2019).
Afshar, S. et al. The effects of bariatric procedures on bowel habit. Obes. Surg. 26, 2348–2354. https://doi.org/10.1007/s11695-016-2100-9 (2016).
Norouzian Ostad, A. et al. Macro- and micro-nutrient intake adequacy in gastric bypass patients after 24 months: A cross-sectional study. Clin. Nutr. Res. 10, 341–352. https://doi.org/10.7762/cnr.2021.10.4.341 (2021).
Misselwitz, B., Butter, M., Verbeke, K. & Fox, M. R. Update on lactose malabsorption and intolerance: pathogenesis, diagnosis and clinical management. Gut 68, 2080–2091. https://doi.org/10.1136/gutjnl-2019-318404 (2019).
Schollenberger, A. E. et al. Impact of protein supplementation after bariatric surgery: A randomized controlled double-blind pilot study. Nutrition 32, 186–192. https://doi.org/10.1016/j.nut.2015.08.005 (2016).
Maxim, M. et al. Dietary habits, obesity, and bariatric surgery: A review of impact and interventions. Nutrients 17, 474. https://doi.org/10.3390/nu17030474 (2025).
Hosseini-Esfahani, F. et al. Nutrient Intake and deficiency of patients 1 year after bariatric surgery: Tehran obesity treatment study (TOTS). J. Gastrointest Surg. 25, 911–918. https://doi.org/10.1007/s11605-020-04530-9 (2021).
Nymo, S. et al. Diet and physical activity are associated with suboptimal weight loss and weight regain 10–15 years after Roux-en-Y gastric bypass: A cross-sectional study. Obes. Res. Clin. Pract. 16, 163–169. https://doi.org/10.1016/j.orcp.2022.03.006 (2022).
Masood, A. et al. Dietary and lifestyle factors serve as predictors of successful weight loss maintenance postbariatric surgery. J. Obes. 2019, 7295978. https://doi.org/10.1155/2019/7295978 (2019).
da Silva, F. B., Gomes, D. L. & de Carvalho, K. M. Poor diet quality and postoperative time are independent risk factors for weight regain after Roux-en-Y gastric bypass. Nutrition 32, 1250–1253. https://doi.org/10.1016/j.nut.2016.01.018 (2016).
Freire, R. H. et al. Food quality, physical activity, and nutritional follow-up as determinant of weight regain after Roux-en-Y gastric bypass. Nutrition 28, 53–58. https://doi.org/10.1016/j.nut.2011.01.011 (2012).
Harbury, C., Collins, C. E. & Callister, R. Diet quality is lower among adults with a BMI >/=40 kgm(-2) or a history of weight loss surgery. Obes. Res. Clin. Pract. 13, 197–204. https://doi.org/10.1016/j.orcp.2018.10.003 (2019).
Harbury, C. M., Verbruggen, E. E., Callister, R. & Collins, C. E. What do individuals with morbid obesity report as a usual dietary intake? A narrative review of available evidence. Clin. Nutr. ESPEN. 13, e15–e22. https://doi.org/10.1016/j.clnesp.2016.02.001 (2016).
Redpath, T. L. et al. Methodological issues in assessing change in dietary intake and appetite following gastric bypass surgery: A systematic review. Obes. Rev. 22, e13202. https://doi.org/10.1111/obr.13202 (2021).
Poslusna, K. et al. Misreporting of energy and micronutrient intake estimated by food records and 24 hour recalls, control and adjustment methods in practice. Br. J. Nutr 101 Suppl 2, S73–S85. https://doi.org/10.1017/S0007114509990602 (2009).
Trijsburg, L. et al. BMI was found to be a consistent determinant related to misreporting of energy, protein and potassium intake using self-report and duplicate portion methods. Public Health Nutr. 20, 598–607. https://doi.org/10.1017/S1368980016002743 (2017).
Acknowledgements
We thank all the patients who participated in this study for their time and cooperation. We also extend our sincere thanks to the members of the multidisciplinary team for their invaluable assistance throughout this study.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Contributions
Y.L., X.S (Xitai Sun). and L.D. conceptualized the study. Y.L., X.S (Xiaodong Shan) ., X.K., X.C (Xuehui Chu). and L.D. contributed to data collection. Y.L. performed data analysis and wrote the manuscript. Y.L., X.C (Xiaotian Chen)., X.S (Xitai Sun). and L.D. critically reviewed the manuscript. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, Y., Shan, X., Kang, X. et al. Changes in dietary nutrient intakes at 6 and 12 months following bariatric surgery in a Chinese observational cohort. Sci Rep 15, 33998 (2025). https://doi.org/10.1038/s41598-025-13350-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-13350-3