Abstract
Global warming is driving contraction of species’ ranges through migration and mortality at their warm edge. However, for most species more subtle, sub-lethal changes in performance will be a more ubiquitous response to the Anthropocene. It has been suggested that reduction in body size will be a universal response to warming for cold-water species. Here we tested this hypothesis for two dominant kelp species in the northern and southern hemispheres, respectively. We tested if populations from cool and warm environments would be morphologically distinct, with warm-water populations displaying structural features indicative of sub-optimal conditions (smaller sizes). We found empirical evidence consistent with size reduction of kelp stipes, blades, and biomass of associated epiphytes from cool to warm water in both hemispheres. These changes are ecologically significant because they affect how kelps engineer their local environment, the three-dimensional habitat they create, and the associated communities they support. Reduced size of cold-water habitat forming species such as kelps may be a sublethal effect of warming that could have widespread but previously overlooked effects on the structure of ecosystems and the services that they provide.
Similar content being viewed by others
Introduction
Warming has been a pervasive change to the global environment over recent decades1. Organisms on land and in the sea have responded with substantial changes in their distribution as conditions for persistence have changed at their range margins, either allowing expansion or driving contraction2,3,4. Research on the ecological effects of warming has had a strong focus on range dynamics and shifts at species’ range-margins (e.g.5,6,7,8). However, range-margins usually represent a relatively small proportion of a species’ range and along most of a species distribution global warming is unlikely to be severe enough to limit persistence. Rather, throughout the warmer half of most species’ ranges, conditions will only shift to become less optimal, not lethal (e.g.9,10). Consequently, sublethal effects of warming are likely to be much more prevalent than lethal effects. While not as dramatic as lethal effects, which can lead to sweeping ecosystem reconfiguration11,12,13 the overall effects of these more subtle, sublethal changes could be substantial because of their pervasiveness.
Environmental conditions impose strong limitations on the shape, size and function of plants and animals14,15. This is particularly true for attached organisms which lack mobility, and therefore have to endure the circumstances that prevail where they live. While most species can survive across a broad range of environmental conditions, performance such as growth, reproduction and metabolism generally peaks within a narrow range of optimal conditions and declines outside of these16,17,18. Within a species, large body size is often considered a sign of fitness and most species attain their biggest sizes under optimal conditions19,20,21 (but see22,23. In contrast, persistence at suboptimal conditions is metabolically costly and leaves less available energy for growth and development24,25. For cool-water species, this leads to the expectation that individuals in warmer waters, where conditions are suboptimal, will be smaller than those in cooler waters.
Kelp forests are the largest vegetated coastal ecosystem26,27. Laminarian kelps are generally considered cool-water species that perform best in temperate, nutrient rich conditions28,29,30. Current and future ocean warming is therefore anticipated to become increasingly stressful and have negative consequences for many kelps, including species of Laminaria31,32,33. Many of the ecological functions of kelps, and therefore also the ecosystem services they provide, are intimately linked to their size, architecture and structural complexity34,35,36. For example, by creating a 3-dimensional structure, kelps provide a physical habitat for myriads of species living within and around the interstitial spaces created by holdfasts, fronds and epiphytes37,38,39,40. Habitat availability, therefore, depends to a large extent on the size of the kelp, which create the foundation for these highly diverse and productive ecosystems, providing valuable ecosystem services to humans41.
The temperature mediated reduction in body size of vertebrates and invertebrates is well-documented14 and holds for many species (i.e., Bergmann’s Rule). In contrast, fewer studies demonstrate a similar effect in marine plants, including macroalgae. Some examples do however exist. Seagrass (Zostera marina) shoots are largest in the middle of their latitudinal range, suggesting body size peaks where conditions are optimal21. In Japan, Serisawa et al.42 found consistently smaller kelp sporophytes (Ecklonia cava) in warm regions compared to cooler regions, and in eastern Australia Mabin et al.43 showed that both fully grown and microscopic kelp sporophytes (E. radiata) were smaller in warmer water. Morphological variation due to temperature differences between sites has also been documented in sugar kelp (Saccharina latissima) along the coasts of New York State44 and in Atlantic Canada45. In Europe, kelp (Laminaria hyperborea) showed reduced size and lower rates of primary production towards the warmer limit of their range46. Such structural readjustments can accompany physiological adaptation to sub-optimal conditions47. In a warming climate, the ecological consequences of shrinking body size could be pervasive and substantial—especially when habitat-forming species such as kelps are affected—because of the implied reduction in the total standing biomass (e.g.48) and volume of habitable 3-dimensional structure (e.g.49).
Here we investigated morphological differences between Laminaria hyperborea and L. pallida populations growing under cool and warm conditions within their respective ranges. In the northern hemisphere, Laminaria hyperborea is a dominant kelp in the northeast Atlantic, occurring between Portugal and western Russia50. In the southern hemisphere, L. pallida is a common kelp species along the west coast of southern Africa51 (Fig. 1). These species are structurally very similar with a single rigid stipe supporting one digitated blade (Fig. S1).
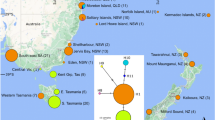
(A) Geographic ranges of Laminaria hyperborea113 and L. pallida114 with blue and red arrows indicating approximate location of cool and warm sites, respectively. Mean annual sea surface temperature (SST) at the study areas in (B) Europe and (C) South Africa, at cool (blue) and warm (red) sites (Bio-ORACLE from 2000 to 2014 data from Bio-ORACLE110. Due to upwelling and local heating and circulation close to the coast, SSTs can deviate substantially from subsurface temperature measurements (provided for each region in Table 1), which better reflect the temperatures experienced by the kelps. All comeponents of this figure were constructed by the authors using R using the mapdata package (A language and Environment for Statiscal Computing, R Core Team, R Foundation for Statiscal Computing, Vienna, Austria, 2017, https://www.R-project.org version 2.2–6, https://CRAN.R-project.org/package=mapdata) and Microsoft 365 Powerpoint version 2502.
Our aim was to assess if there would be systematic differences in plant morphology between warm and cool regions, indicative of changes that could affect the structure and ecological function of kelp forests in a warmer future. Specifically, we tested the hypothesis that L. hyperborea and L. pallida from cool and warm environments would be morphologically distinct, with warm-water populations displaying structural features indicative of sub-optimal conditions. In testing this hypothesis, we identified which morphological characters contributed to differences between species and environments, and therefore also obtained an indication of the degree to which patterns of structural and ecological function might be transferable between L. hyperborea and L. pallida kelp forests.
RESULTS
Environmental conditions
Kelps were measured in four regions, a cool and a warm region in both the northern and southern hemisphere (Table 1, S1). In all four regions, bottom temperatures varied 4–7 °C seasonally, and, on average, the regions in South Africa were 4.1 °C warmer than the European regions (Fig. 1; Table 1) and within both hemispheres temperatures in cool regions were, on average, ~ 4 °C lower than in warm regions. Light levels were somewhat higher in South Africa compared to Norway, but within both regions the differences between cool and warm sites were minimal, as were differences in currents among regions and sites (Table 1). Nutrient concentrations (particularly nitrate) were higher in Africa compared to Europe, but were similar among cool and warm sites in each region. Conversely, C:N ratios were lower for Laminaria hyperborea compared to L. pallida, indicating higher relative nitrogen content in L. hyperborea tissue. C: N ratios were overlapping at cool and warm sites in both regions, and while maximum values were higher at warm sites, perhaps suggesting occasional N limitation, average values were similar at cool and warm sites (Table 1).
Kelp morphology
Laminaria hyperborea and L. pallida were morphologically distinct (P < 0.001), but both species exhibited consistent morphological differences between warm and cool regions (P = 0.004) over and above substantial random variation among sites (P < 0.001, Fig. 2, Table S2). Overall, the most pronounced differences between warm and cool sites involved the vertical structure of the kelp forest and the shape of the blades, with differences in stipe lengths, blade widths and number of digits contributing the most to morphological differences influenced by climate (Table 2). Kelps were taller and had significantly thicker stipes in cool compared to warm regions (Fig. 3). There was no difference in blade length between warm and cool regions, and thallus weight was similar for L. hyperborea in warm and cool regions, and significantly smaller for L. pallida at warm compared to cool regions (Fig. 3). Kelps had much lower total epiphyte loads in warm compared to cool regions, with no epiphytes at warm regions in Africa and 4.3 times higher total epiphyte biomass at cool compared to warm regions in Europe (Fig. 3). This was also the case for relative epiphyte load, where on average cool and warm Laminaria hyperborea and L. pallida individuals had 1.1 vs. 0.4 and 0.2 vs. 0 g epiphytes cm−1 stipe, respectively.
Principal Coordinates Analysis (PCO) using the Euclidian distances to visualise the group differences between morphology of Laminaria hyperborea (triangles) and L. pallida (circles) growing in cool (blue) and warm (red) regions.
Boxplots of individual morphological characters for kelp species (Laminaria hyperborea vs. L. pallida) growing in different climates (cool vs. warm). Boxes show median, 25th and 75th quantiles, error bars show 90th and 10th quantiles, and points show outliers.
DISCUSSION
Bergmann’s rule - the tendency for individuals within a species to be smaller in warmer and larger in cooler environments, respectively - has garnered increasing interest because it predicts that a reduction in body size will be a prevalent, or even universal, response to global warming for many species52. Our study showed how structurally similar kelps in the northern and southern hemispheres both were smaller in warm compared to cool water areas. This pattern was evident across a suite of morphological measures, including stipe and blade dimensions, tissue thickness, biomass and epiphyte weight. These findings are consistent with both geographic patterns of smaller body sizes in warmer climates (i.e., Bergmann’s rule)3,4,46,53 and predicted size reductions of organisms with climate warming, such as those already seen in some populations of birds, fish, marine invertebrates, beetles and salamanders52,54,55,56.
General physiological mechanisms might explain the smaller size of kelps in warmer waters. Thermal sensitivity of growth rates and cell size has been linked to a negative relationship between development temperature and size in crustaceans57 and various plant species are predicted to grow slower and be smaller as temperatures rise with climate change58,59. As on land, where these relationships depend strongly on water availability and nutrients60 in the ocean warmer water tends to hold less nutrients so kelps may grow slower.
The relationship between morphology and fitness can be complex. In general, however, kelps are more likely to reach a large body size when conditions are optimal. For example, Laminaria hyperborea grow the tallest stipes near the middle of their distribution range46. The thermal tolerance of both kelp species suggests they are in suboptimal conditions at warm sites and in more optimal conditions at cool sites. L. hyperborea has a wide thermal range, with optimal growth between 5 and 15 °C61. Ecological niche models for L. hyperborea shows survival in areas where temperatures are above − 0.2 °C, and do not exceed 20.9 °C in the summer or 17.7 °C in the winter62. According to these thermal windows, our warm sites in France, where temperatures range between 8.7 and 16.3 °C, are less optimal than our cooler sites in Norway, where temperatures range between 3.7 and 14.9 °C. For L. pallida, the upper and lower survival temperatures are 22–24 °C and 0 °C respectively, with optimal growth occurring between 10 and 15 °C63. Temperatures at our warm sites were therefore suboptimal (mean 16.4 °C; maximum 19.8 °C) compared to our cool sites (mean 12 °C; maximum 15.8 °C). In this context it is important to note that the size reduction with warming is only expected in the warm half of a species’ distribution (e.g.52). In the Arctic, reduced sea ice cover and warming temperatures may have the opposite effect by increasing the growth of kelps, which could lead to larger sizes and altered morphologies at cool range margins64,65.
Broad-scale mensurative experiments that compare ecological conditions across different extant climates, can be a powerful way to gain insight into how future climates might affect species and communities66. However, inferring the underlying drivers of observed differences can be challenging in comparative studies such as ours, because many potentially important factors often covary in space and time. In the northern hemisphere cool and warm sites were located at different latitudes with varying day lengths. Although variation in day length can drive strong seasonal patterns of growth65 there is little evidence linking seasonal changes in day length to plant size per se. Importantly, in northern Norway annual growth of L. hyperborea individuals within the same age class was lower in Finnmark (~ 71oN) than Vega (~ 65oN)67 and an experiment on L. hyperborea also from Finnmark showed no differences in growth under increasing photoperiod68. Both of these observations are opposite to expected if longer days were driving larger individuals in cooler northern populations. In contrast, L. hyperborea was also found to be smaller at warmer than cooler sites across ~ 10o latitude in the UK69. In the southern hemisphere, different nutrient regimes and currents associated with upwelling on the Atlantic coast of South Africa could have affected the growth of L. pallida. Nutrient concentrations and nitrogen content in kelp tissues were however similar between the warm and cool regions in the southern hemisphere, suggesting that, at the time of sampling, there were no gross differences in nutrient availability underpinning differences in plant size. Nutrients may be more important for species like Macrocystis pyrifera that have little to no nutrient reserves70,71 compared to Laminaria spp., which can tolerate long periods of low nutrients72. Yet, short-term differences in nutrient regimes do occur in the upwelling zone, and cannot be discounted entirely. Nevertheless, the consistent response of kelp size across temperature gradients covarying with latitude (northern hemisphere) and upwelling regimes (southern hemisphere) suggest that neither day length nor nutrient conditions alone can account for the differences in kelp sizes.
Regardless of the exact mechanisms, declining size of kelps will likely have broad implications for forest and seascape structure. Kelp canopies create distinct environmental conditions by dampening water movement and reducing light levels73,74,75,76 which supports diverse understory communities of smaller, shade-tolerant algae and other species34,77. The smaller lamina sizes documented in warmer regions in this study would affect how these species ‘engineer’ the local environment such as increasing the light levels beneath the canopy and possibly altering the shaded understory communities78. Shorter stipes should make it easier for blades to reach and scour the sea floor during storms, which can modify community interactions in the understory79,80. Shorter stipes will also reduce the vertical habitat structure formed by kelps, creating less space for associated species to use for shelter and habitat38,81. This reduced canopy height also has implications for wave dampening effects of kelp, which may protect coasts from erosion82.
Kelps are highly productive species that provide food for coastal food webs through direct consumption by grazers83,84,85 or through detrital pathways86,87,88. For L. hyperborea, there is a strong positive relationship between total standing biomass and annual production46,89,90. Although it is unclear if a similar relationship holds for Laminaria pallida, it is highly likely that reduced standing biomass at warmer sites means that less kelp-carbon is produced and available for coastal food webs. Lower kelp biomass also means less carbon is produced, stored and available for sequestration, effectively shrinking an important potential carbon sink46,91.
Perhaps the most prominent pattern we observed from cool to warm sites was a large decrease in epiphyte biomass, particularly in the southern hemisphere. This could be related to the temperature tolerance of the epiphytes themselves, or be a direct consequence of the taller stipes, which provide more space for colonization. Epiphytes create important microhabitats that support high faunal abundance and diversity92,93. The small invertebrates associated with the epiphytes are food for fish and other species, and as a result the epiphyte biomass is tightly linked to secondary productivity93,94,95. Epiphyte abundance can also be indicative of habitat stability, suggesting kelps in cool areas live longer than kelps in warm areas96,97.
In conclusion, we provide evidence in support of an expected trend of shrinking organism size with warming. These temperature driven contractions in morphology of habitat forming species could be important sublethal impacts of warming with effects on ecosystem structure and the services that these habitats provide. Collapse and range contractions of kelp forests and other canopy-forming macroalgae in response to warming oceans have been predicted31,32,98,99 and observed on many coasts globally11,100,101,102,103,104. Yet, subtle effects of warming within the ranges of kelp are less understood105. These changes should be more pervasive and may be key to understanding how kelp forests respond to future climate warming. It is also unclear whether the morphological patterns we show here are the result of genetic adaptation to local climate or phenotypic plasticity, which has important implications for the rates of ecosystem change106,107. Species-specific responses to climate are varied and will depend on a complex interplay between multiple species and environmental changes. Although we are far from unravelling the complex interactions between environmental and ecological controls on macroalgal size, growth and communities that are required to predict general responses to warming108 our results suggest that warming could lead to a contraction in kelp size at currently cool sites. We need to understand why these organisms are smaller and what these patterns will mean for biodiversity and human societies that rely on these ecosystems.
Methods
This study compared kelps from Europe (Norway and France; Laminaria hyperborea) and southern Africa (Cape of Good Hope; L. pallida, Fig. 1). In Europe, cool and warm regions were separated along a latitudinal temperature gradient whereas in Africa, cool and warm regions were separated by the Cape Peninsula (Fig. 1, Table S1). Four accessible sites (at least 1 km apart) were sampled within each warm and cool region. This level of spatial replication was included to account for variation in site characteristics (e.g., wave exposure, water clarity, topography, herbivores). All samples were collected in spring from fully subtidal populations with a depth range across all sites of 5–15 m depth with no depth differences among temperature regimes or species (2-way ANOVA, p > 0.32) (Table S1).
Temperature data were obtained for each region for the period 2013–2014 where kelps were sampled. In France temperatures were recorded by an onset HOBO pendant logger at 10 m depth at a Roscoff site. In Norway temperatures were recorded by a CTD probe at 10 m on 15 separate days over the same period at the Bud hydrographic station, ca. 20 km northeast of Finnøy (http://www.imr.no/forskning/forskningsdata/stasjoner/view?station=Bud). In South Africa temperature data were recorded by Star Oddi Starmon mini temperature loggers at 8 m depth at the Bordjiesrif (warm) and Kommitjie (cool) sites, respectively. For comparison and analysis, temperatures from all regions were extracted for the same 15 dates as the measurements in Norway. Sub-surface temperatures were analysed by 2-way analysis of variance by 9,999 permutations of the residuals under a full model109. To visualize the temperature conditions over the range of both kelp species, mean sea surface temperatures for the larger study area were obtained from Bio-ORACLE from 2000 to 2014 (Fig. 1). Nutrient conditions at sites were compared using phosphate and nitrate concentrations obtained from Bio-ORACLE from 2000 to 2014110,111. We compared the long-term average, and the long-term average of the minimum and maximum concentrations of each measure per year. To further investigate these measures of available nutrients we also compared C: N ratios from kelp samples obtained from each of the four regions (Table 1). These C: N data (measured by mass spectrometry) were collected as part of other concurrent research programs in each of the four regions, and, as a result, were only available for 4 sites, one cool and one warm in each region.
At each site, 10–13 kelps were harvested by a SCUBA diver haphazardly collecting the largest individuals in sight within a 15–20 min search, targeting areas with no visible signs of kelp harvesting. Consequently, the sampled kelps represented a random subset of the largest part of the population. This sampling strategy was employed because the largest thalli are likely to reflect the maximum realised growth potential at the site and as such accounts for the many other environmental and biological factors that could act and interact to affect the mean size of the kelps (e.g., shading, tattering, herbivory and competition). Moreover, this sampling strategy also minimises potential biases from sampling insufficient numbers to describe the true mean.
Eight morphological measurements were made on all kelp thalli (Fig. S1). Total thallus wet mass (g), stipe length (cm), stipe diameter (mm), lamina length (cm), lamina width (cm), lamina thickness (mm) and number of digits. Finally, epibionts were scraped from the stipes and weighed wet (g). At these sites, the stipe epibionts were dominated by epiphytes (epiphytic seaweeds, see also112).
All data analyses were done in PRIMER (version 7). Multivariate analysis of variance by permutation, PERMANOVA109 was used to test for morphological differences between Laminaria hyperborea and L. pallida (Species, fixed factor) growing under cool and warm conditions (Climate, fixed factor) at four sites per combination (random factor nested in species and climate). Data were log(x + 1) transformed and normalised to down-weigh extreme values and to put all measures on a common scale, respectively, before 9,999 unrestricted permutations of the raw data. A similarity percentage (SIMPER) analysis was done to identify the contribution of each morphological variable to differences between Climate and Species. A Principal Coordinates Analysis (PCO) was applied using Euclidian distances in order to visualise the group differences.
Data availability
All data is available from the corresponding author on request.
References
IPCC. AR6 Synthesis Report. (2023).
Blackburn, T. M. & Hawkins, B. A. Bergmann’s rule and the mammal fauna of Northern North America. Ecography 27, 715–724. https://doi.org/10.1111/j.0906-7590.2004.03999.x (2004).
Ramirez, L., Diniz-Filho, J. A. F. & Hawkins, B. A. Partitioning phylogenetic and adaptive components of the geographical body-size pattern of new world birds. Glob. Ecol. Biogeogr. 17, 100–110. https://doi.org/10.1111/j.1466-8238.2007.00346.x (2008).
Diniz-Filho, J. A. F., Bini, L. M., Rodríguez, M. Á., Rangel, T. F. L. V. B. & Hawkins, B. A. Seeing the forest for the trees: partitioning ecological and phylogenetic components of bergmann’s rule in European carnivora. Ecography 30, 598–608. https://doi.org/10.1111/j.0906-7590.2007.04988.x (2007).
Angert, A. L. et al. Do species’ traits predict recent shifts at expanding range edges? Ecol. Lett. 14, 677–689. https://doi.org/10.1111/j.1461-0248.2011.01620.x (2011).
Sunday, J. M. et al. Species traits and climate velocity explain geographic range shifts in an ocean-warming hotspot. Ecol. Lett. 18, 944–953. https://doi.org/10.1111/ele.12474 (2015).
Pecl, G. T. et al. Biodiversity redistribution under climate change: Impacts on ecosystems and human well-being. Science https://doi.org/10.1126/science.aai9214 (2017).
Pinsky, M. L., Selden, R. L. & Kitchel, Z. J. Climate-Driven shifts in marine species ranges: scaling from organisms to communities. Annual Rev. Mar. Sci. 12, 153–179. https://doi.org/10.1146/annurev-marine-010419-010916 (2020).
Bennett, S., Duarte Carlos, M., Marbà, N. & Wernberg, T. Integrating within-species variation in thermal physiology into climate change ecology. Philosophical Trans. Royal Soc. B: Biol. Sci. 374, 20180550. https://doi.org/10.1098/rstb.2018.0550 (2019).
Brown, J. H. On the relationship between abundance and distribution of species. Am. Nat. 124, 255–279 (1984).
Wernberg, T. et al. Climate-driven regime shift of a temperate marine ecosystem. Science 353, 169–172. https://doi.org/10.1126/science.aad8745 (2016).
Filbee-Dexter, K., Feehan, C. J. & Scheibling, R. E. Large-scale degradation of a Kelp ecosystem in an ocean warming hotspot. Mar. Ecol. Prog. Ser. 543, 141–152 (2016).
Harris, R. M. B. et al. Biological responses to the press and pulse of climate trends and extreme events. Nat. Clim. Change. 8, 579–587. https://doi.org/10.1038/s41558-018-0187-9 (2018).
Gardner, J. L., Peters, A., Kearney, M. R., Joseph, L. & Heinsohn, R. Declining body size: a third universal response to warming? Trends Ecol. Evol. 26, 285–291. https://doi.org/10.1016/j.tree.2011.03.005 (2011).
Koehl, M. A. R. When does morphology matter? Ann. Rev. Ecol. Syst. 27, 501–542 (1996).
Pörtner, H. O. Oxygen- and capacity-limitation of thermal tolerance: a matrix for integrating climate-related stressor effects in marine ecosystems. J. Exp. Biol. 213, 881–893. https://doi.org/10.1242/jeb.037523 (2010).
Kordas, R. L., Harley, C. D. G. & O’Connor, M. I. Community ecology in a warming world: the influence of temperature on interspecific interactions in marine systems. J. Exp. Mar. Biol. Ecol. 400, 218–226. https://doi.org/10.1016/j.jembe.2011.02.029 (2011).
Lefort, S. et al. Spatial and body-size dependent response of marine pelagic communities to projected global climate change. Glob. Change Biol. 21, 154–164. https://doi.org/10.1111/gcb.12679 (2015).
Blanckenhorn, W. U. The evolution of body size: what keeps organisms small?? Q. Rev. Biol. 75, 385–407 (2000).
Zuo, W., Moses, M. E., West, G. B., Hou, C. & Brown, J. H. A general model for effects of temperature on ectotherm ontogenetic growth and development. Proceedings: Biological Sciences 279, 1840–1846 (2012).
Ruesink, J. L. et al. Form–function relationships in a marine foundation species depend on scale: a shoot to global perspective from a distributed ecological experiment. Oikos 127, 364–374. https://doi.org/10.1111/oik.04270 (2018).
Pincheira-Donoso, D., Hodgson, D. J. & Tregenza, T. The evolution of body size under environmental gradients in ectotherms: why should bergmann’s rule apply to lizards? BMC Evol. Biol. 8, 68. https://doi.org/10.1186/1471-2148-8-68 (2008).
Teplitsky, C. & Millien, V. Climate warming and bergmann’s rule through time: is there any evidence? Evol. Appl. 7, 156–168. https://doi.org/10.1111/eva.12129 (2014).
Brown, G. P. & Weatherhead, P. J. Thermal ecology and sexual size dimorphism in northern water snakes, Nerodia sipedon. Ecological Monographs https://doi.org/10.1890/0012-9615 (2000).
Pörtner, H. O. & Farrell, A. P. Physiology and climate change. Science 322, 690–692. https://doi.org/10.1126/science.1163156 (2008).
Duarte, C. et al. Global Estimates of the extent and production of macroalgal forests. Global Ecology and Biogeography In press (2022).
Filbee-Dexter, K. et al. Leveraging the blue economy to transform marine forest restoration. J. Phycol. 58, 198–207. https://doi.org/10.1111/jpy.13239 (2022). https://doi.org:.
Wernberg, T., Krumhansl, K., Filbee-Dexter, K. & Pedersen, M. in World Seas: An Environmental Evaluation Vol. III: Ecological Issues and Environmental Impacts (ed C. Sheppard) 57–78 (Elsevier, 2019).
Lüning, K. Seaweeds. Their environment, biogeography and ecophysiology (John Wiley and Sons, NY., 1990).
Pessarrodona, A. et al. Global seaweed productivity. Science Advances In press (2022).
Martínez, B. et al. Distribution models predict large contractions of habitat-forming seaweeds in response to ocean warming. Divers. Distrib. 24, 1350–1366. https://doi.org/10.1111/ddi.12767 (2018).
Wilson, K. L., Skinner, M. A. & Lotze, H. K. Projected 21st-century distribution of canopy-forming seaweeds in the Northwest Atlantic with climate change. Divers. Distrib. 25, 582–602. https://doi.org/10.1111/ddi.12897 (2019).
Assis, J. et al. Major shifts at the range edge of marine forests: the combined effects of climate changes and limited dispersal. Sci. Rep. 7, 44348. https://doi.org/10.1038/srep44348 (2017).
Wernberg, T. & Filbee-Dexter, K. Missing the marine forest for the trees. Mar. Ecol. Prog. Ser. 612, 209–215 (2019).
Teagle, H. & Smale, D. A. Climate-driven substitution of habitat-forming species leads to reduced biodiversity within a temperate marine community. Divers. Distrib. 24, 1367–1380. https://doi.org/10.1111/ddi.12775 (2018).
Pessarrodona, A., Foggo, A. & Smale, D. A. Can ecosystem functioning be maintained despite climate-driven shifts in species composition? Insights from novel marine forests. J. Ecol. 107, 91–104. https://doi.org/10.1111/1365-2745.13053 (2019).
Christie, H., Norderhaug, K. & Fredriksen, S. Macrophytes as habitat for fauna. Mar. Ecol. Prog. Ser. 396, 221–233. https://doi.org/10.3354/meps08351 (2009).
Teagle, H., Hawkins, S. J., Moore, P. J. & Smale, D. A. The role of Kelp species as biogenic habitat formers in coastal marine ecosystems. J. Exp. Mar. Biol. Ecol. 492, 81–98. https://doi.org/10.1016/j.jembe.2017.01.017 (2017).
Tuya, F., Wernberg, T. & Thomsen, M. Habitat structure affect abundances of labrid fishes across temperate reefs in south-western Australia. Environ. Biol. Fish. 86, 311–319 (2009).
Wernberg, T. et al. Impacts of climate change on marine foundation species. Annual Rev. Mar. Sci. 16, 247–282. https://doi.org/10.1146/annurev-marine-042023-093037 (2024).
UNEP. United Nations Environment Programme and Norwegian Blue Forest Network Global Report on Kelp Forests. United Nations Environment Programme in (2023).
Serisawa, Y. et al. Morphometric study of Ecklonia Cava (Laminariales, Phaeophyta) sporophytes in two localities with different temperature conditions. Phycological Res. 50, 193–199 (2002).
Mabin, C., Gribben, P., Fischer, A. & Wright, J. Variation in the morphology, reproduction and development of the habitat-forming Kelp Ecklonia radiata with changing temperature and nutrients. Mar. Ecol. Prog. Ser. 483, 117–131. https://doi.org/10.3354/meps10261 (2013).
Gerard, V. A. Ecotypic differentiation in light-related traits of the Kelp Laminaria Saccharina. Mar. Biol. 97, 25–36 (1988).
Gerard, V. A. & DuBois, K. R. Temperature ecotypes near the Southern boundary of the Kelp laminaria Saccharina. Mar. Biol. 97, 575–580. https://doi.org/10.1007/bf00391054 (1988).
Pessarrodona, A., Moore, P. J., Sayer, M. D. J. & Smale, D. A. Carbon assimilation and transfer through kelp forests in the NE Atlantic is diminished under a warmer ocean climate. Global Change Biology https://doi.org/10.1111/gcb.14303 (2018).
Wernberg, T. et al. Decreasing resilience of Kelp beds along a latitudinal temperature gradient: potential implications for a warmer future. Ecol. Lett. 13, 685–694 (2010).
van Son, T. C. et al. Achieving Reliable Estimates of the Spatial Distribution of Kelp Biomass. Front. Mar. Sci. https://doi.org/10.3389/fmars.2020.00107 (2020).
Pessarrodona, A. et al. Homogenization and miniaturization of habitat structure in temperate marine forests. Glob. Change Biol. 27, 5262–5275. https://doi.org/10.1111/gcb.15759 (2021). https://doi.org:.
Smale, D. A., Burrows, M. T., Moore, P., O’Connor, N. & Hawkins, S. J. Threats and knowledge gaps for ecosystem services provided by Kelp forests: a Northeast Atlantic perspective. Ecol. Evol. 3, 4016–4038. https://doi.org/10.1002/ece3.774 (2013).
Bolton, J. The biogeography of kelps (Laminariales, Phaeophyceae): a global analysis with new insights from recent advances in molecular phylogenetics. Helgol. Mar. Res. 64, 263–279. https://doi.org/10.1007/s10152-010-0211-6 (2010).
Sheridan, J. A. & Bickford, D. Shrinking body size as an ecological response to climate change. Nat. Clim. Change. 1, 401–406 (2011). http://www.nature.com/nclimate/journal/v1/n8/abs/nclimate1259.html#supplementary-information
Clauss, M., Dittmann, M. T., Müller, D. W. H., Meloro, C. & Codron, D. Bergmann′s rule in mammals: a cross-species interspecific pattern. Oikos 122, 1465–1472. https://doi.org/10.1111/j.1600-0706.2013.00463.x (2013).
Gardner, J. L., Heinsohn, R. & Joseph, L. Shifting latitudinal clines in avian body size correlate with global warming in Australian passerines. Proc. Royal Soc. B: Biol. Sci. 276, 3845–3852. https://doi.org/10.1098/rspb.2009.1011 (2009).
Caruso, N. M., Sears, M. W., Adams, D. C. & Lips, K. R. Widespread rapid reductions in body size of adult salamanders in response to climate change. Glob. Change Biol. 20, 1751–1759. https://doi.org/10.1111/gcb.12550 (2014).
Shackell, N. L., Frank, K. T., Fisher, J. A., Petrie, B. & Leggett, W. C. Decline in top predator body size and changing climate alter trophic structure in an oceanic ecosystem. Proc. Royal Soc. B: Biol. Sci. 277, 1353–1360 (2010).
Timofeev, S. F. Bergmann’s principle and Deep-Water gigantism in marine crustaceans. Biology Bull. Russian Acad. Sci. 28, 646–650. https://doi.org/10.1023/A:1012336823275 (2001).
Barber, V. A., Juday, G. P. & Finney, B. P. Reduced growth of Alaskan white Spruce in the twentieth century from temperature-induced drought stress. Nature 405, 668–673. https://doi.org/10.1038/35015049 (2000).
Franks, S. J. & Weis, A. E. A change in climate causes rapid evolution of multiple life-history traits and their interactions in an annual plant. J. Evol. Biol. 21, 1321–1334. https://doi.org/10.1111/j.1420-9101.2008.01566.x (2008).
Moles, A. T. et al. Global patterns in plant height. J. Ecol. 97, 923–932. https://doi.org/10.1111/j.1365-2745.2009.01526.x (2009).
Bolton, J. J. & Lüning, K. Optimal growth and maximal survival temperatures of Atlantic Laminaria species (Phaeophyta) in culture. Mar. Biol. 66, 89–94. https://doi.org/10.1007/bf00397259 (1982).
Assis, J., Lucas, A. V., Bárbara, I. & Serrão, E. Future climate change is predicted to shift long-term persistence zones in the cold-temperate kelp Laminaria hyperborea. Marine Environmental Research https://doi.org/10.1016/j.marenvres.2015.11.005 (2016).
tom Dieck, I. & de Oliveira, E. C. The section digitatae of the genus laminaria (Phaeophyta) in the Northern and Southern atlantic: crossing experiments and temperature responses. Mar. Biol. 115, 151–160. https://doi.org/10.1007/BF00349397 (1993).
Krause-Jensen, D. & Duarte, C. M. Expansion of vegetated coastal ecosystems in the future Arctic. Front. Mar. Sci. https://doi.org/10.3389/fmars.2014.00077 (2014).
Filbee-Dexter, K., Wernberg, T., Fredriksen, S., Norderhaug, K. M. & Pedersen, M. F. Arctic Kelp forests: diversity, resilience and future. Glob. Planet Change. 172, 1–14. https://doi.org/10.1016/j.gloplacha.2018.09.005 (2019). https://doi.org:
Wernberg, T., Smale, D. A. & Thomsen, M. S. A decade of climate change experiments on marine organisms: procedures, patterns and problems. Glob. Change Biol. 18, 1491–1498. https://doi.org/10.1111/j.1365-2486.2012.02656.x (2012).
Sjøtun, K., Fredriksen, S., Lein, T. E., Rueness, J. & Sivertsen, K. Population studies of Laminaria hyperborea from its Northern range of distribution in Norway. Hydrobiologia 260–261, 215–221. https://doi.org/10.1007/bf00049022 (1993).
Diehl, N. et al. Photoperiod and temperature interactions drive the latitudinal distribution of laminaria hyperborea (Laminariales, Phaeophyceae) under climate change. J. Phycol. 60, 1237–1255. https://doi.org/10.1111/jpy.13497 (2024). https://doi.org:
Smale, D. A. et al. Linking environmental variables with regional-scale variability in ecological structure and standing stock of carbon within UK Kelp forests. Mar. Ecol. Prog. Ser. 542, 79–95 (2016).
Graham, M. H., Vasquez, J. A. & Buschmann, A. H. Global ecology of the giant Kelp macrocystis: from ecotypes to ecosystems. Oceanogr. Mar. Biol. Annu. Rev. 45, 39–88 (2007).
Gerard, V. A. Growth and utilization of internal nitrogen reserves by the giant Kelp macrocystis pyrifera in a low-nitrogen environment. Mar. Biol. 66, 27–35. https://doi.org/10.1007/BF00397251 (1982).
Bartsch, I. et al. The genus Laminaria sensu lato: recent insights and developments. Eur. J. Phycol. 43, 1–86 (2008).
Layton, C. et al. Resilience and stability of Kelp forests: the importance of patch dynamics and environment-engineer feedbacks. PLOS ONE. 14, e0210220. https://doi.org/10.1371/journal.pone.0210220 (2019).
Wernberg, T., Kendrick, G. A. & Toohey, B. D. Modification of the physical environment by an Ecklonia radiata (Laminariales) canopy and implications for associated foliose algae. Aquat. Ecol. 39, 419–430 (2005).
Clark, R. P., Edwards, M. S. & Foster, M. S. Effects of shade from multiple Kelp canopies on an understory algal assemblage. Mar. Ecol. Prog. Ser. 267, 107–119 (2004).
Eckman, J. E., Duggins, D. O. & Sewell, A. T. Ecology of understory Kelp environments I. effects of Kelps on flow and particle transport near the bottom. J. Experimental Mar. Biology Ecol. 129, 173–188 (1989).
Graham, M. H. Effects of local deforestation on the diversity and structure of Southern California giant Kelp forest food webs. Ecosystems 7, 341–357 (2004).
Reed, D. C. & Foster, M. S. The effects of canopy shading on algal recruitment and growth in a giant Kelp forest. Ecology 65, 937–948 (1984).
Velimirov, B. & Griffiths, C. L. Wave-induced Kelp movement and its importance for community structure. Bot. Mar. 22, 169–172 (1979).
Kennelly, S. J. Effects of Kelp canopies on understorey species due to shade and scour. Mar. Ecol. Prog. Ser. 50, 215–224 (1989).
Norderhaug, K. M. et al. Ecosystem-level effects of large-scale disturbance in Kelp forests. Marine Ecol. Progress Series ITRS, ITRSav4 (2020).
Lovas, S. M. & Torum, A. Effect of the Kelp laminaria hyperborea upon sand Dune erosion and water particle velocities. Coast. Eng. 44, 37–63 (2001).
Zarco-Perello, S., Wernberg, T., Langlois, T. J. & Vanderklift, M. A. Tropicalization strengthens consumer pressure on habitat-forming seaweeds. Sci. Rep. 7, 820. https://doi.org/10.1038/s41598-017-00991-2 (2017).
O’Brien, J. M. & Scheibling, R. E. Nipped in the bud: mesograzer feeding preference contributes to Kelp decline. Ecology 97, 1873–1886. https://doi.org/10.1890/15-1728.1 (2016).
Filbee-Dexter, K. et al. Carbon export is facilitated by sea urchins transforming Kelp detritus. Oecologia 192, 213–225. https://doi.org/10.1007/s00442-019-04571-1 (2020).
Duggins, D., Simenstad, C. & Estes, J. Magnification of secondary production by Kelp detritus in coastal marine ecosystems. Science 245, 170–173. https://doi.org/10.1126/science.245.4914.170 (1989).
Vilas, D. et al. Kelp-carbon uptake by Arctic deep-sea food webs plays a noticeable role in maintaining ecosystem structural and functional traits. J. Mar. Syst. 203, 103268. https://doi.org/10.1016/j.jmarsys.2019.103268 (2020).
Wernberg, T., Vanderklift, M. A., How, J. & Lavery, P. S. Export of detached macroalgae from reefs to adjacent seagrass beds. Oecologia 147, 692–701 (2006).
Pedersen, M. F. et al. Detrital carbon production and export in high latitude Kelp forests. Oecologia https://doi.org/10.1007/s00442-019-04573-z (2019).
Pessarrodona, A. et al. Global seaweed productivity. Sci. Adv. 8, eabn2465. https://doi.org/10.1126/sciadv.abn2465 (2022).
Filbee-Dexter, K. et al. Carbon export from seaweed forests to deep ocean sinks. Nat. Geosci. https://doi.org/10.1038/s41561-024-01449-7 (2024).
Thomsen, M. S. et al. Secondary foundation species enhance biodiversity. Nat. Ecol. Evol. 2, 634–639. https://doi.org/10.1038/s41559-018-0487-5 (2018).
Norderhaug, K. M., Christie, H. & Fredriksen, S. Is habitat size an important factor for faunal abundances on Kelp (Laminaria hyperborea)? J. Sea Res. 58, 120–124. https://doi.org/10.1016/j.seares.2007.03.001 (2007). https://doi.org:.
Edgar, G. J. & Aoki, M. Resource limitation and fish predation: their importance to mobile epifauna associated with JapaneseSargassum. Oecologia 95, 122–133. https://doi.org/10.1007/BF00649515 (1993).
Norderhaug, K., Christie, H., Fosså, J. & Fredriksen, S. fish–macrofauna interactions in a Kelp (laminaria hyperborea) forest. J. Mar. Biol. Association United Kingd. 85, 1279–1286. https://doi.org:undefined (2005).
Christie, H., Fredriksen, S. & Rinde, E. Regrowth of Kelp and colonization of epiphyte and fauna community after Kelp trawling at the Coast of Norway. Hydrobiologia 376, 49–58 (1998).
Steen, H., Moy, F. E., Bodvin, T. & Husa, V. Regrowth after Kelp harvesting in Nord-Trøndelag, Norway. ICES J. Mar. Science: J. Du Conseil. 73, 2708–2720. https://doi.org/10.1093/icesjms/fsw130 (2016).
Raybaud, V. et al. Decline in Kelp in West Europe and climate. PLoS ONE. 8, e66044. https://doi.org/10.1371/journal.pone.0066044 (2013).
Takao, S., Kumagai, N. H., Yamano, H., Fujii, M. & Yamanaka, Y. Projecting the impacts of rising seawater temperatures on the distribution of seaweeds around Japan under multiple climate change scenarios. Ecol. Evol. 5, 213–223. https://doi.org/10.1002/ece3.1358 (2015).
Arafeh-Dalmau, N. et al. Extreme Marine Heatwaves Alter Kelp Forest Community Near Its Equatorward Distribution Limit. Front. Mar. Sci. https://doi.org/10.3389/fmars.2019.00499 (2019).
Voerman, S. E., Llera, E. & Rico, J. M. Climate driven changes in subtidal Kelp forest communities in NW Spain. Mar. Environ. Res. 90, 119–127. https://doi.org/10.1016/j.marenvres.2013.06.006 (2013)
Rogers-Bennett, L. & Catton, C. A. Marine heat wave and multiple stressors tip bull Kelp forest to sea urchin barrens. Sci. Rep. 9, 15050. https://doi.org/10.1038/s41598-019-51114-y (2019).
Filbee-Dexter, K. et al. Marine heatwaves and the collapse of marginal North Atlantic Kelp forests. Sci. Rep. 10, 13388. https://doi.org/10.1038/s41598-020-70273-x (2020).
Tanaka, K., Taino, S., Haraguchi, H., Prendergast, G. & Hiraoka, M. Warming off Southwestern Japan linked to distributional shifts of subtidal canopy-forming seaweeds. Ecol. Evol. 2, 2854–2865. https://doi.org/10.1002/ece3.391 (2012).
Bennett, S., Wernberg, T., Arackal Joy, B., de Bettignies, T. & Campbell, A. H. Central and rear-edge populations can be equally vulnerable to warming. Nat. Commun. 6, 10280. https://doi.org/10.1038/ncomms10280 (2015).
Wernberg, T. et al. Genetic diversity and Kelp forest vulnerability to Climatic stress. Sci. Rep. 8, 1851. https://doi.org/10.1038/s41598-018-20009-9 (2018).
Vranken, S. et al. Genotype-environment mismatch of Kelp forests under climate change. Mol. Ecol. 15, 3730–3746 (2021).
Duarte, B. et al. Climate Change Impacts on Seagrass Meadows and Macroalgal Forests: An Integrative Perspective on Acclimation and Adaptation Potential. Front. Mar. Sci. https://doi.org/10.3389/fmars.2018.00190 (2018).
Anderson, M. J., Gorley, R. N. & Clarke, K. R. PERMANOVA + for PRIMER: Guide to software and statistical methods. 2nd edition edn, 214 (PRIMER-E Ltd, (2008).
Tyberghein, L. et al. Bio-ORACLE: a global environmental dataset for marine species distribution modelling. Glob. Ecol. Biogeogr. 21, 272–281. https://doi.org/10.1111/j.1466-8238.2011.00656.x (2012).
Assis, J. et al. Bio-ORACLE v2.0: extending marine data layers for bioclimatic modelling. Glob. Ecol. Biogeogr. 27, 277–284. https://doi.org/10.1111/geb.12693 (2018).
Leclerc, J. C. et al. Local flexibility in feeding behaviour and contrasting microhabitat use of an omnivore across latitudes. Oecologia 196, 441–453. https://doi.org/10.1007/s00442-021-04936-5 (2021).
de Bettignies, T. et al. T., La Rivière M. Case report for Kelp forests habitat. 39 (2021). (OSPAR 787/2021.
Wernberg, T. et al. In into the Blue: Securing a Sustainable Future for Kelp Forests9–27 (United Nations Environment Programme, 2023).
Acknowledgements
This study was supported by grants from the Norwegian Research Council (KFD, KMN, TW, AJS: ‘BlueConnect’, ‘GecoKelp’, ‘BlueArc’), the National Research Foundation of South Africa (AJS, JJB, RA, TW), The Australian Research Council (TW, KFD: DP190100058, DE1901006192, DP220100650), and The University of Western Australia (TW, TdB). We are grateful for diving, boating and sample processing assistance from the Seaweed Research Unit in South Africa and the core dive team at the marine biological station of Roscoff, France.
Funding
Open access funding provided by Institute Of Marine Research
Author information
Authors and Affiliations
Contributions
Conceived the study: TW, TdB, KFD, AJS. Field work and data collection: TW, TdB, JCL, LL, HCC, DCD, MDR, KFD, AJS. Analysed data and wrote the first draft: TW, KFD, AJS. Discussed results and edited the manuscript: all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
41598_2025_13950_MOESM1_ESM.docx (download DOCX )
Supplementary Material 1
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wernberg, T., Filbee-Dexter, K., de Bettignies, T. et al. Smaller plants in warmer water could have implications for future Kelp forests. Sci Rep 15, 28616 (2025). https://doi.org/10.1038/s41598-025-13950-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-13950-z