Abstract
Early identification of high-risk recurrence patients is crucial as it can provide information for treatment decisions, achieve personalized treatment, and maximize the benefits for locally advanced rectal cancer (LARC). The goal of this study was to explore the impact of ROI selection on the repeatability of spectral detector CT (SDCT) imaging biomarkers and to investigate and visualize an interpretable multidimensional radiological-angiogenesis-clinicopathological integrated model (RACIM) for predicting disease-free survival (DFS) in LARC. 204 LARC patients who underwent SDCT scanning prior to any anticancer treatment were retrospectively included. Two observers independently measured the iodine concentrations and normalized iodine concentration (NIC) at venous/delayed phases (VP/DP) using two different ROI protocols (2D vs. 3D). Cox regression methods were applied to determine the independent risk predictors associated with DFS, which were used to develop a prediction model. Kaplan–Meier survival curves were used to explore the performance of RACIM for personalized risk stratification. The models’ performance was assessed using the area under the receiver operating characteristic (ROC) curve (AUC). The SHapley Additive exPlanation (SHAP) algorithm was used to attribute interpretability to the optimal prediction model. The 3D-ROI method achieved satisfactory interobserver accuracy. Multivariate Cox regression analyses indicated that perineural invasion, CD105, histological grade and NICVP3D values were independent predictors of DFS. On this basis, a multidimensional RACIM was constructed and achieved AUCs of 0.95 and 0.93 in the training and validation cohorts, respectively. The RACIM-based classifier demonstrated good performance for discriminating patients at high and low risk of recurrence, and patients in the high-risk group were found to have a favorable response to postoperative adjuvant chemotherapy (P = 0.036). Moreover, SDCT-derived 3D quantitative biomarker displayed as the most important feature in SHAP, indicating the importance of quantitative radiological features in personalized risk stratification. This study laid the foundation for the large-scale standardized clinical application of SDCT in the future, and provided an optional novel approach for postoperative risk stratification and even chemotherapy beneficiaries selection in LARCs.
Similar content being viewed by others
Introduction
In the past decade, the development of total mesorectal excision and the widespread use of neoadjuvant chemoradiotherapy have significantly decreased the locoregional recurrence rate of locally advanced rectal cancer (LARC)1,2,3 but 25–40% of LARC patients still experience systemic recurrence after surgery4. To reduce the incidence of systemic recurrence, the guidelines recommend postoperative adjuvant chemotherapy for LARCs with high-risk clinicopathological factors such as T4 tumor stage, poorly differentiated tumors, obstruction or perforation at the time of diagnosis, lymphovascular invasion (LVI), perineural invasion (PNI), positive margins, and fewer than 12 lymph nodes examined after surgery5. Valentini’s nomogram (VN), which includes pathological stage, surgical procedure, and adjuvant chemotherapy status, was developed for prognostic prediction of LARC6 and endorsed by the American Joint Committee on Cancer. However, its performance is still limited (local recurrence, c-index 0.68; distant metastasis, c-index 0.73)7. In addition, whether high-risk patients classified by these clinicopathological factors can benefit from adjuvant chemotherapy after surgery remains controversial8,9. At present, radiological imaging is widely used for early diagnosis and prognostic factor detection due to its advantages of being noninvasive, visible, dynamic, and comprehensive for evaluating tumor lesions. Tumor angiogenesis is also an important indicator for predicting patient prognosis10,11,12. Hence, integrating multidimensional information from clinical-imaging-angiogenesis data to construct a fusion prediction model might provide better performance for postoperative risk stratification and selection of chemotherapy benefit populations.
In fact, imaging evaluation relies on visual assessment by radiologists, and there may be low consistency in diagnostic results among radiologists, especially junior radiologists with limited experience. Spectral detector CT (SDCT) has become a popular research topic due to its ability to not only obtain anatomical information from conventional CT but also provide various quantitative parameter information based on material composition analysis13. However, the majority of studies based on SDCT have used 2D-ROI delineation at the maximum level of tumors14,15,16,17,18. Due to the heterogeneity of LARC and the uneven distribution of neovascularization in tumors, qualified imaging markers must be able to reveal information on all lesions and be stable. Therefore, early evaluation using reproducible 3D-SDCT imaging biomarkers may become an important parameter for improving prognostic prediction.
Angiogenesis is one of the core markers of cancer19. Multiple studies have shown that tumor angiogenesis, such as that of CD34 and CD105, and the level of vascular endothelial growth factor (VEGF) were important indicators for predicting patient prognosis10,11,12but the prognostic value of different angiogenesis parameters in RC remains controversial20,21,22. The identification of optimal angiogenesis biomarkers associated with the prognosis of LARC may provide additional prognostic value for the construction of prognostic models.
In this regard, we analyzed the impact of different ROI selection methods (2D vs. 3D) on the measurement of SDCT imaging biomarkers, explored the complementary prognostic value of different angiogenesis biomarkers, and established an interpretable model based on the optimal ROI protocol for individualized prediction of postoperative risk stratification in LARC patients. We further assessed and compared the prognostic value of our model to that of the VN model and tested its ability to determine the benefits of postoperative adjuvant chemotherapy in LARC patients.
Materials and methods
Patients
All studies complied with the Declaration of Helsinki. This retrospective study was approved by the institutional review boards of Tianjin Medical University Cancer Institute & Hospital (EK20240180), and patients informed consent was waived. All experiments were performed in accordance with relevant guidelines and regulations.
We retrospectively reviewed the data of rectal cancer patients who underwent pretreatment SDCT examination. Retrospective data between March 2019 and March 2021 were used to identify eligible patients according to the following inclusion criteria: (1) biopsy-proven untreated rectal cancer; (2) available pre-treatment pelvic SDCT images from the PACS; (3) standard neoadjuvant therapy followed by surgical resection with histopathological confirmation; and (4) no concurrent malignant tumors or prior cancer treantment. The exclusion criteria were as follows: (1) lost to follow-up within 3 years; or (2) had incomplete records of imaging, clinical and pathological data. The detailed information on preoperative treatment, surgery, and postoperative adjuvant therapy are shown in Supplementary Material 1.
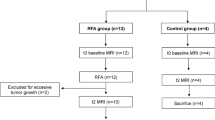
Two rectal cancer cohorts were analyzed: 204 patients were randomly classified into training (n = 143) and validation (n = 61) cohorts at a 7:3 ratio, using the Python package scikit-learn, in Python 3.10.9. Figure 1 presents the flowchart for patient selection and the workflow of this study.
After surgical resection, all patients were followed up every 3–6 months for at least 3 years. The follow-up measurements included the CEA level and a contrast CT/MRI scan. The outcome of this study was 3-year disease-free survival (DFS), which was defined as the period between the date of surgery and the first evidence of clinical recurrence and/or distant metastases, as confirmed by imaging or histopathology. Patients were divided into recurrent and non-recurrent groups based on recurrence status within 3 years.
Flowchart of patient’s enrollment and the workflow of the study.
SDCT protocols and imaging feature acquisition
Rectal unenhanced and two-phase contrast-enhanced scans were performed by a dual-layer detector spectral CT (IQon Spectral CT, Philips Healthcare) in the supine position with hands above the head. First, under a tube voltage of 120 kVp, nonenhanced scanning was obtained following scout imaging. The nonionic contrast medium iohexol was applied as a contrast agent at a concentration of 1.2 mL/kg and an injection rate of 2.5 mL/s through the median cubital vein. The acquisition times in the venous phase (VP) and the delayed phase (DP) were 60–70 s and 90 s after contrast agent administration, respectively. The acquisition and reconstruction parameters are shown in Supplementary Material 2.
Spectral images were obtained on an IntelliSpace Portal Workstation (ISP version 10, Philips Healthcare) for further investigation. Two radiologists with abundant abdominal expertise (10 years and 12 years) and who were blinded to the clinical data, measured the quantitative imaging biomarkers independently. Two different ROI protocols were used. (1) For 2D-ROI, a round ROI was placed at the greatest axial diameter and adjacent to the upper and lower slices of the rectal cancer lesions, and then the average iodine concentration (IC) values on the iodine-based images were calculated (Fig. S1a,b,e,f). (2) For 3D-ROI, the volume of interest along the entire enhanced tumor margin at all visible tumor levels was outlined (Fig. S1c,d,g,h), and the average IC values were calculated. Additionally, the normalized iodine concentration (NIC) refers to the quotient of the lesion IC divided by the the same layer of external iliac artery IC to minimize patient variability (NIC = IClesion/ICartery).
Clinicopathological parameters
Clinicopathological parameters, including age, sex, hypertension history, family history, histologic grade, pathologic T, N stage, microsatellite instability status, HIF-1α expression, Ki-67 level, LVI, PNI, surgical procedure and postoperative adjunctive history, were collected by reviewing the medical records of the patients (Supplementary Material 3).
Tumor angiogenesis parameters
Tumor angiogenesis was assessed via the CD34-based microvessel density (MVD), CD105-based MVD, and VEGF level.
Immunohistochemistry was performed to stain selected specimens using monoclonal mouse anti-human CD34 and CD105 antibodies and monoclonal rabbit anti-human VEGF antibody (Supplementary Material 4,5).
Model construction
The “xgboost” package in R was used to build the model, with the learning rate set to 0.1 and the objective function specified as binary logistic regression.
Statistical analyses
All the statistical calculations were conducted using SPSS 19.0, MedCalc 20.014 or R 6.1 (http://www.R-project.org). Quantitative variables are expressed as the mean ± SD. The data of the two observers were tested with the interclass correlation coefficient (ICC) and Bland-Ahman plot. Continuous variables were compared using the t-test or Mann-Whitney U tests, while categorical variables were compared using the χ2 or Fisher exact tests, as appropriate. For continuous variables with missing values, use mean, median, or mode for imputation; For categorical variables, use mode interpolation. Cox proportional hazards model was used for univariate and multivariate analyses, effect sizes were reported as hazard ratios (HRs) with 95% confidence intervals (CIs). The DeLong test was adopted to compare the differences between parameters. The optimal cutoff value was determined using X-tile (3.6.1) for dividing patients into high-risk and low-risk groups. Patient stratification in different risk groups was evaluated using Kaplan‒Meier curve analysis and the log-rank test. In order to control bias and achieve pseudo-randomization, propensity score matching (PSM) using R was used to rematch the study samples. To establish an interpretable prediction model, SHapley Additive exPlanation (SHAP) was used to provide an explanation for our predictive model, which calculated the importance of ranking features from the final model23. P < 0.05 was considered to indicate statistical significance.
Results
Patient characteristics
The baseline data of all 204 patients and their tumors are summarized in Table 1. The median follow-up times for DFS were 899.5 days in the training cohort (interquartile range [IQR]: 385.0–1257.0 days) and 950.4 days in the validation cohort (IQR: 499.0–1304.5 days).
Sixty-five of the 204 patients experienced disease recurrence, 33 (50.8%) of whom experienced systemic disease recurrence (8 in the lung, 20 in the liver, 2 in the bone and 3 in both the liver and lung), 21 (32.3%) of whom experienced locoregional disease recurrence, and 11 (16.9%) of whom experienced mixed disease. Among them, 20 patients were confirmed by surgery, while the other 45 patients were diagnosed based on radiological characteristics. 137 (67.2%) patients were treated with postoperative adjuvant fluorouracil-based chemotherapy.
Overall, the average CD34-based MVD of all the lesions was 40.19 ± 6.89; for CD105-based MVD, it was 28.25 ± 5.50.
2D- vs. 3D-ROI interobserver agreement
Among the two ROI methods, the 3D-ROI method had the best interobserver agreement (ICC of 0.826–0.960) (Table 2). The Bland-Altman analysis showed that all the imaging features measured by the 3D-ROI method were more concentrated than those measured by the 2D-ROI method, indicating that the 3D-ROI analysis had a smaller consistency interval and better accuracy in repeated measurements by different readers (Fig. 2). Therefore, the average values of 3D-quantitative imaging features calculated by the two radiologists were used for further analysis.
Predictive factors for DFS
In the univariate analyses of DFS, clinicopathological parameters (histologic grade, pT stage, pN stage, CEA, HIF-1α, LVI, and PNI), SDCT features (NICVP3D and NICDP3D values) and angiogenesis parameters (CD34, CD105, and VEGF) were associated with DFS. According to the multivariate analysis, clinicopathological parameters (PNI, histologic grade), SDCT features (NICVP3D values) and angiogenesis parameters (CD105) were found to be independent predictors in the training cohort (P < 0.05, Table 3).
Bland-Altman analysis between the two observers using two different ROI methods. (a) ICVP3D. (b) ICDP3D. (c) NICVP3D. (d) NICDP3D. (e) ICVP2D. (f) ICVP2D. (g) NICVP2D. (h) NICDP2D.
Model construction and comparison
A multidimensional radiological-angiogenesis-clinicopathological integrated model (RACIM) was established based on the above prediction variables (PNI, CD105, histologic grade and NICVP3D values), which predicted the probability of disease recurrence for each individual patient. Multivariate analysis was used to construct a clinical model that included histologic grade, HIF-1α, LVI and PNI; an angiogenesis model that included CD105; and a radiological model that included NICVP3D values. The receiver-operating characteristic (ROC) curves of the different models for the entire cohort are shown in Fig. 3. The ROC curves revealed that the radiological model (NICVP3D) had an AUC of 0.85 (95% CI, 0.78–0.91), a sensitivity of 78.4%, and a specificity of 79.3%. According to the X-tile, the optimal cut-off value of the NICVP3D was identified as 0.345. The combined model achieved excellent predictive performance, with AUCs of 0.95 (95% CI, 0.92–0.98) and 0.93 (95% CI, 0.85-1.00) in the training and validation cohorts, respectively (Table 4). The AUC of the combined model was obviously greater than that of the radiological (P = 0.0004, P = 0.0393), angiogenesis (P < 0.0001, P = 0.0091) and clinical models (P = 0.0471, P = 0.0088) in all cohorts.
Receiver operating characteristic curve (ROC) analysis for the prediction models in the training (a) and validation cohorts (b).
Additionally, a VN model with pathological stage, surgical procedure, and adjuvant chemotherapy status was also built for comparison. Compared with the VN model (AUC: 0.77, 95% CI: 0.70–0.85; AUC: 0.76, 95% CI: 0.59–0.92; AUC: 0.72, 95% CI: 0.59–0.85), our radiological and RACIM models exhibited superior performance in the training (AUC: 0.85, 95% CI: 0.78–0.91, P = 0.0160; AUC: 0.95, 95% CI: 0.92–0.98, P < 0.0001) and validation (AUC: 0.83, 95% CI: 0.73–0.93, P = 0.4217; AUC: 0.93, 95% CI: 0.85-1.00, P = 0.0428) (Table 4). Moreover, the calibration plots of RACIM model showed that the estimations had good agreement with the actual observations (Fig. 4a,b). The decision curve analysis curves revealed that the RACIM model achieved moderately better net benefit than other models over the relevant threshold range in all cohorts (Fig. 4c, d).
Calibration curves and decision curves of different models. (a) Calibration curves in training cohort. (b) Calibration curves in validation cohort. (c) Decision curves in training cohort. (d) Decision curves in validation cohort.
Patient risk stratification
We divided patients into high- and low-risk groups according to the X-tile-generated optimum cutoff value (0.389) of the RACIM, which significantly differed in terms of DFS in the training cohort (log-rank test, P < 0.001) (Fig. 5a). Then, we performed the same analyses to stratify patients in the validation cohort to determine the prognostic value of the RACIM. Consistent with the training cohort, significant differences in DFS were observed between the two groups in validation cohort (log-rank test, P = 0.001) (Fig. 5b). Table 5 showed the selected prediction parameters in RACIM-classified high and low-risk groups.
To test the ability of the RACIM to identify patients who may benefit from postoperative adjuvant chemotherapy, subgroup analyses of patients receiving adjuvant chemotherapy were further performed. Notably, in the RACIM-classified high-risk group, postoperative adjuvant chemotherapy was significantly associated with a treatment benefit (P = 0.036), while adjuvant chemotherapy did not improve survival in any of the 204 patients (P = 0.400) or in patients with any high-risk clinicopathological features (P = 0.400, Fig. 6).
The Kaplan Meier survival analysis curve stratified the prognosis of patients according to the RACIM-based classifier. (a) Training cohort. (b) Validation cohort.
Effect of postoperative adjuvant chemotherapy in different subgroups, which were stratified by the receipt of chemotherapy. (a) All cases group. (b) RACIM-classified high risk group. (c) Any high-risk clinicopathological features group.
Model interpretability with SHAP
In this study, we employed the SHAP algorithm to endow our RACIM with global and local interpretability. As observed in the plot, the SDCT imaging indicator NICVP3D was the most important risk factor, followed by CD105, PNI, and histologic grade (Fig. 7a,b).
Figure 7c,d shows the SHAP model force plot of two male participants who had TNM stage IIIB disease, depicting how NICVP3D, CD105 and clinicopathological characteristics affect the ability of the model to predict recurrence risk at the individual level.
Model interpretability of the RACIM for the prediction of disease-free survival (DFS) with SHAP in the training cohort. (a) Feature importance plot listing the most significant variables in descending order. (b) Summary plot of the impact of features on model decision-making and the interactions between features in the model. SHAP force plots of two participants with high (c) and low (d) risk of DFS. Yellow dots represent higher eigenvalues and purple dots represent lower eigenvalues.
Discussion
In this study, we first demonstrated that SDCT-derived 3D quantitative biomarkers achieved more satisfactory interobserver accuracy than 2D parameters and could reliably predict DFS in LARC patients. Compared to other angiogenesis parameters (CD34 and VEGF), CD105 was the only independent angiogenesis factor for DFS. We then developed and validated a multidimensional RACIM combining clinicopathological, SDCT and angiogenesis parameters (PNI, histologic grade, NICVP3D, and CD105) for the individualized prediction of DFS and achieved better performance than traditional clinicopathological parameters. More importantly, the proposed RACIM successfully identified patients who were most likely to benefit from postoperative adjuvant therapy.
Over the past few years, many scholars have attempted to identify new biomarkers that can predict patient prognosis and help identify patients who are suitable for postoperative adjuvant chemotherapy in LARCs. Mounting evidence has revealed the predictive value of SDCT features in rectal cancer14,15,16,17,18. Previous studies selected only the maximum level of the tumor for ROI delineation, ignoring tumor heterogeneity and phenotypic variations. In contrast, our study showed that the 3D-ROI method has the best consistency and repeatability, thereby ensuring the reliability and stability of the results. In addition, our NICVP3D model was superior to any other parameter or VN model for predicting recurrence. Through the variance importance plot of the SHAP algorithm, we found that NICVP3D is also the most important feature, confirming the necessity of incorporating imaging features into conventional clinical features for risk stratification and laying the foundation for the large-scale standardized clinical application of SDCT in the future.
Notably, in our study, the NIC was an independent factor for DFS compared to the IC, and the venous-phase NIC outperformed the delay-phase NIC in terms of prognosis, which is consistent with the results of other studies24,25,26,27. The reason for these results may be that the NIC removed the potential influence of individual circulation on the IC, as well as the differences in tumor blood supply during different phases of contrast-enhanced CT. The iodine quantitative imaging biomarkers in the VP could well reflect the microcirculation of tumors and may reflect the distribution of contrast agents in the interstitial space25,27. However, other studies have also reported the predictive value of IC and NICDP in rectal cancer26. We speculated that this difference in the results may be attributable to the use of different ROI selection methods, although further investigations are needed before widespread implementation of these methods in clinical practice.
Angiogenesis is an important component of tumor progression; thus, our study incorporated angiogenesis parameters into the constructed predictive model. Notably, compared to VEGF and CD34, CD105-based MVD was the only independent factor for DFS, confirming that CD105 can be regarded as a better prognostic marker for rectal cancer, which is similar to the findings of a previous study28. CD105, a 180 kDa transmembrane glycoprotein, is a coreceptor of TGF-β and is the key factor in the TGF-β pathway and angiogenesis29,30. In contrast to CD31, CD34 and factor-related antigens, CD105 is highly expressed only in neovascular endothelial cells, while it is almost undetectable in the blood vessels of normal tissues31. Therefore, it is reasonable to explain why the average value of CD105-based MVD was lower than that of CD34-based MVD in the present study.
Another focus of this study was the selection of patients who benefited from adjuvant chemotherapy after surgery. According to our subgroup analyses, we found that patients in the RACIM-based high-risk group achieved a better survival benefit from adjuvant chemotherapy, whereas patients in the low-risk group did not. Furthermore, this study revealed that adjuvant chemotherapy failed to significantly improve survival outcomes in either the overall patient cohort or subgroups stratified by conventional high-risk clinicopathological parameters. These findings reaffirm the inherent limitations of traditional clinicopathological risk assessment in identifying optimal candidates for postoperative chemotherapy. Importantly, our novel RACIM-based classifier demonstrated superior predictive capacity for therapeutic stratification, enabling more personalized adjuvant treatment decisions through precision patient selection.
As a preliminary effort, the study has a few limitations. First, as a retrospective study, it was characterized by unavoidable selection bias. Second, the data were based on small numbers of patients from one center and lacked external validation. For this reason, future studies with more samples are needed to verify the model, and integrated research with antiangiogenic agents and neoadjuvant therapies are promising future directions. Finally, manual segmentation was time-consuming. To our knowledge, the IntelliSpace Portal workstation 12 has achieved semiautomatic volumetric ROI delineation, which will provide convenient technical support for further validation of our results.
Future impact
To translate the promising performance of the RACIM into routine clinical practice, prospective multicenter validation in diverse patient cohorts is essential. This will rigorously confirm its generalizability, stability across different SDCT platforms and operators, and real-world prognostic value. Integration into the clinical workflow could involve incorporating the RACIM score calculation into radiology reporting systems or hospital information systems (HIS/RIS/PACS) upon SDCT examination completion. The visualized risk stratification output could then be readily available within multidisciplinary team meetings, directly informing discussions on adjuvant therapy intensity (e.g., escalating or de-escalating chemotherapy) for individual LARC patients. Furthermore, exploring the development of automated or semi-automated tools for NICVP3D measurement within SDCT would enhance standardization, reduce observer variability, and facilitate the seamless adoption of this quantitative biomarker into future prospective studies and clinical practice, ultimately paving the way for its role in optimizing LARC treatment strategies.
Conclusion
The 3D-SDCT analysis protocol improved interobserver consistency, and the NICVP obtained by this method has the potential for predicting DFS. Moreover, our study highlighted the prognostic value of CD105-based MVD in rectal cancer. Furthermore, the multidimensional RACIM combined with SDCT, angiogenesis and clinicopathological parameters could offer an optional novel approach to improve the postoperative risk stratification in LARC patients. Our prediction model provided individual-level interpretability through the SHAP algorithm, providing a valuable decision-making basis for personalized postoperative therapeutic strategies.
Data availability
The data can be accessed upon request with a research proposal submitted to the investigator via the corresponding author.
Abbreviations
- DFS:
-
Disease-free survival
- DP:
-
Delayed phase
- IC:
-
Iodine concentration
- LARC:
-
Locally advanced rectal cancer
- LVI:
-
Lymphovascular invasion
- MVD:
-
Microvessel density
- NIC:
-
Normalized iodine concentration
- PNI:
-
Perineural invasion
- RACIM:
-
Radiological-angiogenesis-clinicopathological integrated model
- ROC:
-
Receiver-operating characteristic
- SDCT:
-
Spectral detector CT
- SHAP:
-
SHapley Additive exPlanation
- VEGF:
-
Vascular endothelial growth factor
- VN:
-
Valentini’s nomogram
- VP:
-
Venous phase
References
Sebag-Montefiore, D. et al. Preoperative radiotherapy versus selective postoperative chemoradiotherapy in patients with rectal cancer (MRC CR07 and NCIC-CTG C016): a multicentre, randomised trial. Lancet 373, 811–820 (2009).
Rödel, C. et al. Preoperative chemoradiotherapy and postoperative chemotherapy with fluorouracil and oxaliplatin versus fluorouracil alone in locally advanced rectal cancer: initial results of the German CAO/ARO/AIO-04 randomised phase 3 trial. Lancet Oncol. 13, 679–687 (2012).
Breugom, A. J. et al. Adjuvant chemotherapy for rectal cancer patients treated with preoperative (chemo) radiotherapy and total mesorectal excision: a Dutch colorectal cancer group (DCCG) randomized phase III trial. Ann. Oncol. 26, 696–701 (2015).
Fokas, E. et al. Tumor regression grading after preoperative chemoradiotherapy for locally advanced rectal carcinoma revisited: updated results of the CAO/ARO/AIO-94 trial. J. Clin. Oncol. 32, 1554–1562 (2014).
National Comprehensive Cancer Network. NCCN Guidelines Rectal Cancer. National Comprehensive Cancer Network. (Version 6. 2023). (Accessed 23 December 2023). https://www.nccn.org/professionals/physician_gls/pdf/rectal.pdf.
Valentini, V. et al. Nomograms for predicting local recurrence, distant metastases, and overall survival for patients with locally advanced rectal cancer on the basis of European randomized clinical trials. J. Clin. Oncol. 29, 3163–3172 (2011).
Amin, M. B. et al. The eighth edition AJCC cancer staging manual: continuing to build a Bridge from a population-based to a more personalizedapproach to cancer staging. CA Cancer J. Clin. 67, 93–99 (2017).
Sargent, D. J., Andre, T. & Grothey, A. Futher evaluating the benefit of adjuvant chemotherapy for colon cancer. J. Clin. Oncol. 34, 3711–3712 (2016).
Pahlman, L. A. et al. Should the benefit of adjuvant chemotherapy in colon cancer be re- evaluated? J. Clin. Oncol. 34, 1297–1299 (2016).
Hwang, I. et al. Tumor-associated macrophage, angiogenesis and lymphangiogenesis markers predict prognosis of non-small cell lung cancer patients. J. Transl Med. 18, 443 (2020).
Fox, S. B. Tumour angiogenesis and prognosis. Histopathology 30, 294–301 (1997).
Zheng, S. et al. Identification of the angiogenesis related genes for predicting prognosis of patients with gastric cancer. BMC Gastroenterol. 21, 146 (2021).
Parakh, A. et al. Dual-energy CT images: pearls and pitfalls. Radiographics 41, 98–119 (2021).
Cao, Y. et al. Development of a dual-energy spectral computed tomography-based nomogram for the preoperative discrimination of histological grade in colorectal adenocarcinoma patients. J. Gastrointest. Oncol. 12, 544–555 (2021).
Al-Najami, I., Lahaye, M. J., Beets-Tan, R. G. H. & Baatrup, G. Dual-energy CT can detect malignant lymph nodes in rectal cancer. Eur. J. Radiol. 90, 81–88 (2017).
Al-Najami, I., Drue, H. C., Steele, R. & Baatrup, G. Dual energy CT-a possible new method to assess regression of rectal cancers after neoadjuvant treatment. J. Surg. Oncol. 116, 984–988 (2017).
Wu, J. et al. The value of single-source dual-energy CT imaging for discriminating microsatellite instability from microsatellite stability human colorectal cancer. Eur. Radiol. 29, 3782–3790 (2019).
Fan, S. X. et al. Correlations between the iodine concentrations from dual energy computed tomography and molecular markers Ki-67 and HIF-1α in rectal cancer: A preliminary study. Eur. J. Radiol. 96, 109–114 (2017).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Abdalla, S. A. et al. Prognostic relevance of microvessel density in colorectal tumours. Oncol. Rep. 6, 839–842 (1999).
Akagi, K. et al. Estimation of angiogenesis with anti-CD105 immunostaining in the process of colorectal cancer development. Surgery 131 (Suppl 1), S109–S113 (2002).
Nogués, A. et al. Endoglin (CD105) and VEGF as potential angiogenic and dissemination markers for colorectal cancer. World J. Surg. Oncol. 18, 99 (2020).
Lundberg, S. M. & Lee, S. I. A unified approach to interpreting model predictions. In Proceedings of the 31st international conference on neural information processing systems. 4768-77. (Neural Information Processing Systems Foundation, Inc. (NIPS), 2017).
Li, J. et al. Spectral CT-based nomogram for preoperative prediction of perineural invasion in locally advanced gastric cancer: a prospective study. Eur. Radiol. 33, 5172–5183 (2023).
Liang, P., Ren, X., Gao, J., Chen, K. & Xu, X. Iodine concentration in spectral CT: assessment of prognostic determinants in patients with gastric adenocarcinoma. Am. J. Roentgenol. 209, 1033–1038 (2017).
Gao, W. et al. Association between extramural vascular invasion and iodine quantification using dual-energy computed tomography of rectal cancer: a preliminary study. Eur. J. Radiol. 158, 110618 (2023).
Chen, X. H. et al. Spectral computed tomography in advanced gastric cancer: can iodine concentration non-invasively assess angiogenesis? World J. Gastroenterol. 23, 1666–1675 (2017).
Pammar, C., Nayak, R. S., Kotrashetti, V. S. & Hosmani, J. Comparison of microvessel density using CD34 and CD105 in oral submucous fibrosis and its correlation with clinicopathological features: an immunohistochemical study. J. Cancer Res. Ther. 14, 983–988 (2018).
Langer, E. M. et al. ZEB1-repressed MicroRNAs inhibit autocrine signaling that promotes vascular mimicry of breast cancer cells. Oncogene 37, 1005–1019 (2018).
Fonsatti, E., Nicolay, H. J. M., Altomonte, M., Covre, A. & Maio, M. Targeting cancer vasculature via endoglin/CD105: a novel antibody-based diagnostic and therapeutic strategy in solid tumours. Cardiovasc. Res. 86, 12–19 (2010).
Dallas, N. A. et al. Endoglin (CD105): A marker of tumor vasculature and potential target for therapy. Clin. Cancer Res. 14, 1931–1937 (2008).
Funding
This study was supported by National Natural Science Foundation of China (Grant No. 82302180); Chinese National Key Research and Development Project (2021YFC2500400 and 2021YFC2500402); Tianjin Key Medical Discipline (Specialty) Construction Project (TJYXZDXK-009 A).
Author information
Authors and Affiliations
Contributions
S.F., L.Q. and X.C. conceived and designed the study. S.F., J.L., X.Y., L.Q. acquired clinical data. S.F., L.X., L.Q., X.C. acquired and analyzed imaging data.S.F. performed the statistical analysis. S.F. and X.C. drafted the manuscript. All authors revised and amended critical parts of the manuscript. All authors contributed to the interpretation of the data and approved the final version of this manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Fan, S., Liu, J., Yang, X. et al. 3D spectral CT-based fusion model predicts prognosis and postoperative adjuvant chemotherapy benefit in locally advanced rectal cancer. Sci Rep 15, 28836 (2025). https://doi.org/10.1038/s41598-025-14851-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-14851-x