Abstract
Yellow mushrooms (Floccularia luteovirens), which grow in high-altitude regions, are a component of traditional Tibetan medicine and have high nutritional value. In this study, the basic quality indices and volatile organic compounds (VOCs) of yellow mushrooms with different cap opening stages were determined. The volatile compounds (VOCs) were determined by headspace solid-phase microextraction (HS-SPME) combined with gas chromatography-mass spectrometry (GC-MS) technology. The results demonstrated that as the degree of cap opening increased, both the weight of individual mushrooms and their respiratory intensity exhibited an upward trend. However, there was no significant difference in firmness, and the color change was not obvious. A total of 99 VOCs were detected in yellow mushrooms. Principal component analysis (PCA) and hierarchical cluster analysis (HCA) successfully differentiated the three groups of yellow mushrooms based on VOCs, indicating the presence of differentially volatile organic compounds (DVOCs) among the groups. Further, through orthogonal partial least squares discriminant analysis (OPLS-DA), hierarchical cluster analysis (HCA), venn diagram and K-means cluster analysis, the intergroup DVOCs and the specific high-abundance DVOCs were obtained. It revealed the differences in VOCs of yellow mushrooms with different cap opening stages, providing theoretical data for further exploring the quality changes of yellow mushrooms during storage and the traceability of their origin.
Similar content being viewed by others
Introduction
As the world’s third pole, the Qinghai-Tibet Plateau is home to unique vegetation types and ecosystems. Floccularia luteoviren also known as the yellow mushroom. It is extensively found in the alpine meadows of Qinghai and Tibet and stands as one of the most representative fungi in the Qinghai-Tibet Plateau’s alpine meadow ecosystem, possessing significant economic value and both medicinal and edible applications1,2,3,4. Usually, the yellow mushroom produces fruiting bodies in July and August in the alpine meadows of the Qinghai-Tibet Plateau. The primary growth habitat of yellow mushroom is characterized by low temperatures and high altitudes. Researchers have found that the distribution range of the yellow mushroom is from 28°93′N to 37°69′N and from 90°4′E to 102°1′E, and it grows at an altitude of 3,000 to 4,300 m in alpine meadows or grasslands. It has strict requirements for its growth environment5,6,7,8. So far, there has been no successful precedent of artificial cultivation.
Aroma serves as a relatively crucial quality indicator in mushrooms and constitutes a significant factor affecting consumer acceptance. Therefore, studies on volatile organic compounds (VOCs) has always been a hot topic in the field of aroma9. Edible mushrooms commonly encompass sulfur-containing compounds, octane compounds, aldehydes, ketones, esters and heterocyclic compounds as major volatile classes10,11,12,13. The attributes of flavor of mushrooms like aromatic, delicious, meaty, sweet is a crucial reason for their popularity14. The VOCs of different edible mushroom varieties, growth conditions (light, altitude, etc.), various parts of the fruiting bodies, and samples at different growth and development stages show considerable variation. In the initial stage, it had a slightly fruity flavor and the aroma becomes increasingly rich. Before the maturation stage, the concentration of C8 VOCs and ketones gradually decreased, and then the mushroom flavor became more intense15. The aroma is generally the result of the combined interaction of multiple components, rather than a single component acting alone16,17. Zhu and Jung et al. found that some C8 (such as 1-octen-3-ol and 1-octen-3-one, etc.) VOCs were the unique flavor components of mushrooms14,18. Some sulfur-containing compounds, terpenoids and benzaldehyde derivatives all played important roles in the VOCs of mushrooms19,20. These VOCs were usually qualitatively and quantitatively determined by gas chromatography-mass spectrometry (GC-MS), and then the key VOCs or characteristic VOCs were identified by multivariate statistical analysis methods17,21.
At present, most of the research on yellow mushrooms focuses on their medicinal effects, antioxidant capacity, genetic analysis, and growth environment22,23,24,25. However, research on the VOCs and differentially volatile organic compounds (DVOCs) of yellow mushrooms is limited, and there is also a lack of analysis on the quality and VOCs of yellow mushrooms at different cap opening stages. This research aims to compare the basic qualities and identify the VOCs of yellow mushrooms at different cap opening stages, establish the connection between yellow mushrooms with different cap opening stages and VOCs, and provided theoretical data for establishing fingerprint profiles of yellow mushrooms using VOCs in the future, as well as predicting the optimal harvest period and post-harvest treatment of yellow mushrooms.
Results and discussion
The basic quality of yellow mushrooms
As shown in Table 1, there were differences in the weight of individual yellow mushrooms different cap opening stages. The weight of individual mushrooms increased as the cap opened further. This phenomenon could be attributed to the continuous growth and development of the yellow mushrooms, during which they transitioned from a closed state to full opening, resulting in an enlargement of the mushroom body. These findings are consistent with observational data. However, firmness decreased while respiratory intensity increased with the degree of cap opening, indicating that as the yellow mushroom grows, its respiration becomes more activeand maturation progresses, which is consistent with its natural developmental pattern.Yellow mushrooms are prone to browning, especially after picking, making their color and overall appearance extremely crucial for quality assessment and commercial value2. The instrumental chroma values are useful for uncovering the true color reflection of these produce. The color value L* represents brightness and is inversely proportional to the depth of the color, a* represents redness and b* represents yellowness. As shown in Table 1, with the increase of cap opening intensity of the mushrooms, the L*, a*, b*, and C* values showed a downward trend, suggesting a tendency towards the achromatic area in the color space. Combined with the h* value analysis, the color points all remained in the yellow area, and only the b*, C*, and h* values of the MNO and MAO showed significant differences. This indicated that as the cap opening of yellow mushrooms increased, their color remained yellow, and certain indicators showed no significant differences, with no signs of post-harvest browning. This suggests that browning typically occurs after harvesting and may be associated with water loss and oxidation26.
Identification of VOCs in yellow mushrooms
From the analysis of the total ion flow diagram (Fig. S1) of the VOCs of yellow mushrooms and the qualitative classification of each peak using the spectrum database, a total of 99 VOCs were identified (Table S1). These aroma-active compounds were mainly described as mushroom-like, earthy, green, woody, fatty, fruity, sulfurous, sweet, cabbage-like and herbal odor, which were greatly related to alcohols, ketones, aldehydes, esters, alkenes, alkanes, aromatic hydrocarbons, furans, acids and ethers. Thus, the final aroma profile of yellow mushrooms results from the combined contribution of diverse odor compounds9. These included 19 esters, 16 ketones, 22 alcohols, 13 aldehydes, 9 alkenes, 9 alkanes, 4 aromatic hydrocarbons, 3 furans, 3 ethers, and 1 acid. The most diverse VOCs were observed in the MAO (77 species), followed by the MSO (75 species) and the MNO (66 species). As shown in Fig. 1A, alcohols were the most abundant in the MNO, with a total of 16 species, accounting for 24.24% of the total quantity. Esters and aldehydes followed, each with 11 species, accounting for 16.67% of the total quantity. In the MSO, esters were the most abundant (15 species), accounting for 20.00% of the total number, followed by alcohols (14 species, 18.67%) and ketones (12 species, 16.00%). In the MAO, 16 kinds of esters were predominant, accounting for 20.78% of the total quantity, followed by alcohols (15 species, 19.48%), then aldehydes and ketones (11 and 10 species, respectively). Acids accounted for the lowest proportion among the three groups of yellow mushrooms.
The analysis of the content of VOCs in yellow mushrooms (Fig. 1B) revealed that the total content of alcohols were the highest, at 2009.59 µg/kg FW, accounting for 43.17% of the total content. Esters followed, with a content of 1056.91 µg/kg FW, representing 20.88% of the total content. Aldehydes were next, with a content of 838.78 µg/kg FW, accounting for 18.02% of the total content. The proportions of other categories were below 10%. Furans, ethers and acids contents were the lowest, at 51.01 µg/kg FW, 42.96 µg/kg FW and 9.24 µg/kg FW, respectively. This indicated that alcohols, esters and aldehydes were the major VOCs with high content in yellow mushrooms. Similar findings have been reported in other fungi species27,28,29,30,31.
The results revealed that as the degree of cap opening increased, the content of alcohols decreased, while esters increased, accompanied by a rise in both the diversity and total concentration of VOCs. Once the caps were fully opened, the mushroom’s odor became more intense. In nature, edible fungi emit different VOCs at various developmental stages to mediate interactions and establish ecological relationships with surrounding organisms. As the fruiting body matures, the overall VOC content tends to increase32.
Types and contents of VOCs in yellow mushroom with different cap opening stages. (A) The species proportion of VOCs in different groups. (B) The content proportion of VOCs in different groups.
Analysis of common and unique VOCs
According to the Upset analysis (Fig. 2A), a total of 47 common VOCs were identified across the three groups of yellow mushrooms. These VOCs included 9 esters, 5 ketones, 10 alcohols, 7 aldehydes, 2 alkenes, 5 alkanes, 4 aromatic hydrocarbons, 2 furans and 3 ethers as illustrated in (Fig. 2B). There were 26 unique VOCs, both MNO and MSO included 6 species, and MAO included 14 species. These findings indicated that the VOCs of yellow mushrooms were influenced by different cap opening stages. While some VOCs remained stable, an increase in the degree of cap opening was associated with a greater diversity of VOCs. Furthermore, the energy metabolism of mushrooms at different developmental stages was closely linked to their VOCs. The total VOCs content was also correlated with enzymatic activity related to energy metabolism, adenosine triphosphate (ATP) levels, and freshness, all of which varied with maturity33.
The common and unique VOCs in yellow mushroom with different cap opening stages. (A) The upset diagram among groups. (B) Common and unique VOCs among groups.
PCA and HCA of VOCs
PCA represented the features of multi-dimensional metabolomics data through several principal components. Therefore, differences between various groups could be observed through PCA plots34,35. In this study, PCA was performed on 99 VOCs from three groups of yellow mushrooms with different cap opening stages (Fig. 3A). The groups were completely separated and distributed across different quadrants. The first principal component accounted for 46.35% of the original dataset, revealing that MAO was significantly distinct from the other groups. The second principal component explained 30.55% of the dataset, with MSO being separated from the remaining groups. These results indicate that MAO exhibited the greatest differences, followed by MSO, suggesting that VOCs varied among yellow mushrooms at different cap-opening stages.
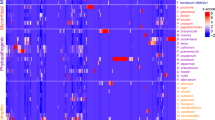
HCA could group samples without prior knowledge of sample information, eliminating human interference35. The accumulation pattern of VOCs in yellow mushrooms with different cap opening stages was analyzed through a cluster heat map (Fig. 3B). HCA results revealed significant differences in VOCs among the groups, which were categorized into three distinct clusters. In cluster 1, VOCs levels were highest in MNO, with minimal differences between MSO and MAO. Cluster 2 showed the highest VOCs levels in MAO, followed by moderate levels in MNO and the lowest in MSO. In cluster 3, VOCs levels were again highest in MAO, moderate in MSO, and lowest in MNO. Different biological replicates also clustered together, indicating good homogeneity among biological replicates and high reliability of the data. MAO was the last to cluster together, which were consistent with the PCA analysis results.
PCA (A) and HCA (B) of VOCs in yellow mushroom with different cap opening stages.
Screening of DVOCs
OPLS-DA analysis was a multivariate statistical analysis method with supervised pattern recognition, which could effectively eliminate the influence of factors irrelevant to the study and screen differential metabolites36. OPLS-DA was used to conduct pairwise comparisons among the MNO, MSO and MAO, and then draw score plots. They were obtained through 200 substitutions, the model validation of MNO vs. MSO showed R2Y = 1 and Q2 = 0.986 (Fig. 4A), the model validation of MSO vs. MAO showed R2Y = 1 and Q2 = 0.981 (Fig. 4B) and the model validation of MNO vs. MAO showed R2Y = 1 and Q2 = 0.986 (Fig. 4C). The closer the Q2 value was to 1, the more stable and reliable the model was.
OPLS-DA moder validation of VOCs in yellow mushroom with different cap opening stages. (A) MNO vs. MSO (B) MSO vs. MAO (C) MNO vs. MAO.
According to the OPLS-DA results, with VIP ≥ 1 and P ≤ 0.05 as the criteria37DVOCs among the three groups and between the two groups were screened out (Table S2). A total of 75 DVOCs were detected, with 47 identified between the MNO and MSO groups. The VIP score plot (Fig. 5A) showed VOCs with VIP ≥ 1, indicating both increase and decrease.trends Among them, 14 DVOCs were significantly increase, including esters (3 species), ketones (3 species), alcohols (3 species), aldehydes (1 species), alkanes (2 species), aromatic hydrocarbons (1 species) and ethers (1 species), and 33 DVOCs were significantly down-regulated, including esters (6 species), ketones (5 species), alcohols (6 species), aldehydes (5 species), alkenes (4 species), alkanes (3 species), furans (1 species), ethers (2 species) and acids (1 species). From the DVOCs clustering heat map (Fig. 6A), the DVOCs with high-abundance between MNO and MSO could be clearly distinguished. Combined with Fig. 2B, the high-abundance DVOCs specific to MNO were identified as 18,18’-bi-1,4,7,10,13,16-hexaoxacyclononadecane, 3,6,9,12-tetraoxatetradecan-1-ol, (2 S,13 S)-12,13-dihydroxy-1,4,7,10-tetraoxacyclotetradecane and hexaethylene glycol. The high-abundance DVOCs specific to MSO were identified as 4-hydroxy-2-butanone, quinoxalin-2-one, decahydro-3-(3,3-dimethyl-2-oxobutenylideno)-, 1-nonanol and butanal, 3-methyl-. These specific high-abundance DVOCs could be used as characteristic VOCs for the identification of MNO and MSO.
It was also found that there were 48 DVOCs between MSO and MAO. According to the VIP score plot (Fig. 5B), compounds with a VIP ≥ 1 and their increase and decrease trends were clearly presented. Among these, 8 VOCs were significantly up-regulated, including esters (2 species), ketones (1 species), aldehydes (3 species) and ethers (2 species), and 40 VOCs were significantly down-regulated, including esters (5 species), ketones (11 species), alcohols (12 species), aldehydes (6 species), alkenes (4 species) and alkanes (2 species). The DVOCs with high-abundance between MSO and MAO was clearly distinguished from the clustering heat map (Fig. 6B). Combined with Fig. 2B, the high-abundance DVOCs specific to MSO included 4-hydroxy-2-butanone, quinoxalin-2-one, decahydro-3-(3,3-dimethyl-2-oxobutenylideno)- and 3-hydroxy-3-phenylbutan-2-one. The high-abundance DVOCs specific to MAO included 4-hydroxybutyl acrylate, thiocyanic acid, methyl ester, 2-heptanone, 2-acetonylcyclopentanone, (+)-2-bornanone, (-)-car-3-en-2-one, 4-nonanol, dl-2-phenyl-1,2-propanediol, (18 S,19 S)-18,19-dihydroxy-1,4,7,10,13,16-hexaoxocycloencosane, 2,4-dihydroxybenzaldehyde, 2TMS derivative, 3-nonene, (E)- and 1,3-hexadiene, 3-ethyl-2-methyl-. These specific high-abundance DVOCs could be used as characteristic DVOCs for identifying MSO and MAO.
A total of 59 DVOCs were identified between MNO and MAO. According to the VIP score plot (Fig. 5C), compounds with a VIP ≥ 1, along with their increase and decrease trends were highlighted. Among these, 17 DVOCs were significantly up-regulated, including esters (5 species), ketones (2 species), alcohols (2 species), aldehydes (5 species), aromatic hydrocarbons (2 species), and ethers (1 species). In contrast, 42 DVOCs were significantly down-regulated, comprising esters (8 species), ketones (8 species), alcohols (11 species), aldehydes (2 species), alkenes (7 species), alkanes (4 species), furans (1 species), and acids (1 species). The DVOCs with high-abundance between MNO and MAO could be clearly distinguished from the clustering heat map (Fig. 6C). Combined with Fig. 2B, the high-abundance DVOCs specific to MNO included 3,6,9,12-tetraoxatetradecan-1-ol, hexaethylene glycol, 2-[2-[2-[2-[2-[2-[2-(2-hydroxyethoxy)ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethanol, (2s,13s)-12,13-dihydroxy-1,4,7,10,13,16-hexaoxocyclooctadecane and 18,18’-bi-1,4,7,10,13,16-hexaoxacyclononadecane. The high-abundance DVOCs specific to MAO included 4-hydroxybutyl acrylate, thiocyanic acid, methyl ester, 2-heptanone, 2-acetonylcyclopentanone, (+)-2-bornanone, (-)-car-3-en-2-one, 4-nonanol, dl-2-phenyl-1,2-propanediol, 2-[2-[2-[2-[2-[2-[2-(2-hydroxyethoxy)ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethanol, (18s,19s)-18,19-dihydroxy-1,4,7,10,13,16-hexaoxocycloencosane, 2,4-dihydroxybenzaldehyde, 2TMS derivative, 3-nonene, (E)- and 1,3-hexadiene, 3-ethyl-2-methyl-. These specific high-abundance DVOCs could be used as characteristic DVOCs for identifying MNO and MAO.
Through the aforementioned analysis, it could be concluded that the specific high-abundance VOCs distinguishing MNO from MSO and MAO were 3,6,9,12-tetraoxatridecan-1-ol (2s,13s)-12,13-dihydroxy-1,4,7,10-tetraoxacyclotetradecane and hexaethylene glycol. MSO was distinguished from MNO and MAO by the specific high-abundance of DVOCs such as 4-hydroxy-2-butanone and quinoxalin-2-one, decahydro-3-(3,3-dimethyl-2-oxobutenylidene)-. Additionally, MAO was distinguished from MNO and MSO, by the specific high-abundance of DVOCs like 4-hydroxybutyl acrylate, thiocyanic acid methyl ester, 2-heptanone, 2-acetonylcyclopentanone, (+)-2-bornanone, (-)-car-3-en-2-one, 4-nonanol, dl-2-phenyl-1,2-propanediol, (18s,19s)-18,19-dihydroxy-1,4,7,10,13,16-hexaoxocycloencosane, 2,4-dihydroxybenzaldehyde, 2TMS derivative, 3-nonene, (E)- and 1,3-hexadiene, 3-ethyl-2-methyl-. These findings suggest that yellow mushrooms cap not open (MNO) or slightly open (MSO) exhibited fewer high-abundance DVOCs, whereas fully opened cap yellow mushrooms displayed a marked increase in the number of the high-abundance of DVOCs. This increase is likely due to the activation of complex metabolic pathways as the mushroom matures. These pathways involve fatty acid metabolism, amino acid metabolism, lentinic acid metabolism, terpenoid biosynthesis, and various other biochemical reactions, collectively leading to the production of a substantial array of VOCs. For instance, linoleic acid and alpha-linolenic acid within the cells were enzymatically broken down by lipoxygenase, forming intermediate compounds 9-hydroperoxyoctadecadienoic acid (9-HPOD), 10-hydroperoxyoctadecadienoic acid (10-HPOD) and 13-hydroperoxyoctadecadienoic acid (13-HPOD). Following this, 10-HPOD was reduced to C8 compounds through hydrogen peroxide lyase, whereas 9-HPOD and 13-HPOD were transformed into C9 aldehydes and C6 aldehydes, respectively. These aldehyde compounds were further metabolized into C9 alcohols and C6 alcohols via alcohol dehydrogenase. Moreover, these alcohol compounds were subsequently converted into corresponding ester compounds by alcohol acyltransferase. Notably, the pathway in which 10-HPOD generates C8 compounds was exclusive to fungi, possibly accounting for why the aroma of fresh mushrooms differs from that of plants38,39. Additionally, factors like postharvest conditions, maturity stages and even distinct parts of the mushroom considerably impacted its VOC profiles21,40,41,42,43,44.
VIP score plot of DVOCs in yellow mushroom with different cap opening stages. (A) MNO vs. MSO. (B) MSO vs. MAO. (C) MSO vs. MAO.
HCA of DVOCs in yellow mushroom with different cap opening stages. (A) MNO vs. MSO. (B) MSO vs. MAO. (C) MSO vs. MAO.
Venn diagrams effectively simplify complex data relationships, making them more intuitive and facilitating a clearer understanding of the potential logical connections between datasets. Therefore, this approach was performed on DVOCs for the comparisons between MNO vs. MSO, MSO vs. MAO, and MNO vs. MAO (Fig. 7). Among these comparisons, there were 13 overlapping DVOCs (Table S3), including 2 esters, Methyl formate and Propanoic acid, 2-methyl-, nonyl ester, which possessed medium-lasting wine-like and sweet rose-like aromas45. Two alcohols, 1-pentanol and 1-hexanol, exhibited clear and tender fusel oil-like scents46. Three ketones, namely 3-octanone, 3-ethyl-2-nonanone and pyrrolidine-2,4-dione, had less sweet and herbaceous characteristics47. Three aldehydes, pentanal, 2-methyl-, pentanal and hexanal, contributed to cabbage-like, fried onion, and green grass scents48. Two alkenes, hexane, 1,2,3-trimethoxy- and 2-nonene, (Z)-, and one ethers, 15-crown-5, further enriched the aroma profile.
Venn diagram of DVOCs between two groups.
K-means cluster analysis of DVOCs
K-means cluster analysis, an unsupervised method commonly used for grouping samples or metabolites based on their characteristics49was applied to analyze the accumulation patterns of 75 DVOCs in yellow mushrooms with different cap opening stages (Fig. 8 and Table S4). Based on the clustering results, DVOCs were categorized into five classes. Class 1 included 13 DVOCs whose concentration increased with the degree of cap opening, reached their highest levels in MAO, suggesting that these DVOCs were key contributors to the MAO group’s profile. Representative compounds in this class included 4-hydroxybutyl acrylate, ethyl 4-(ethyloxy)-2-oxobut-3-enoate, decanoic acid, methyl ester, propanoic acid, 2-methyl-, nonyl ester, 9,12-octadecadienoic acid (Z, Z)-, methyl ester, 4,6-dimethylheptan-2-one, 1-hexanol, amylene hydrate, pentanal, 2-methyl-, 4-nonene, (Z)-, styrene, hexane, 1-methoxy- and hexanoic acid. Class 5 comprised 12 DVOCs whose concentration decreased with increasing cap opening degree. Notably, some DVOCs were particularly abundant in the MSO (Class 2 and 3) and MAO (Class 4). Importantly, Class 3 DVOCs perfectly represent the down-regulated compounds in the MSO vs. MAO and MNO vs. MAO comparisons.
K-means diagrams of the DVOCs.
Materials and methods
Experimental materials
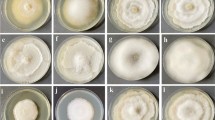
The yellow mushrooms were harvested in the early morning of July 22, 2024 (summer) in Naqu City, Tibet Autonomous Region, China. The region’s average altitude is 4700 m, with summer air oxygen content at 58% of sea level. The average daytime temperature is 14 °C, while the average nighttime temperature is 1 °C. it has annual rainfall of approximately 420 mm, and an annual illumination time of 2800 h. Summer accounts for most of the rainfall and light exposure. Immediately after harvesting, the yellow mushrooms were placed in portable refrigerators, transported to the laboratory, and divided into three groups based on their degree of cap opening: mushroom cap not open(MNO), there was no gap between the mushroom umbrella and the mushroom stem; mushroom cap slightly open (MSO), there was a gap of 0–5 mm between the mushroom umbrella and the mushroom stem; mushroom cap all open (MAO), there was a gap of more than 5 mm between the mushroom umbrella and the mushroom stem. As shown in Fig. 9, each experiment was repeated three times.
The yellow mushroom with different cap opening stages. (A) MNO. (B) MSO. (C) MAO.
Determination of single mushroom weight
Twenty yellow mushrooms were randomly selected from each experimental group and weighed with a precision of 0.01 g. The process was repeated three times, there were a total of 180 mushrooms in three groups.
Determination of firmness
The firmness of the yellow mushrooms was measured using a TMS-PRO texture analyzer (FTC Co., USA) with a measuring range of 250 N. A 2 mm probe was used, with a puncture distance of 6 mm and a puncture speed of 30 mm/s. Twenty mushrooms were randomly selected from each experimental group, and the puncture location was at the center of the yellow mushroom cap. Firmness measurements were repeated three times, there were a total of 180 mushrooms in three groups.
Determination of respiratory intensity
Respiratory intensity was determined using a Shimadzu GC-2010Plus (Tokyo, Japan). The chromatographic conditions were as follows: injector temperature 80 °C, column oven temperature 100 °C, detector temperature 160 °C, and reformer temperature 360 °C. In each experimental group, 300 g of yellow mushrooms were sealed in a 2.2 L container for 1 h, and 1 mL of gas was extracted for measurement. CO2 concentration was recorded, and the procedure was repeated three times, there were a total of 2700 g mushrooms in three groups.
Determination of color difference
The Konica Minolta CR-400 colorimeter (Tokyo, Japan) was used to measure the color of 20 yellow mushrooms in each group, aligning with the center of the mushroom cap. The instrument automatically records the color difference values (L*, a*, b*, C* and h*). Measurements were repeated three times, there were a total of 180 mushrooms in three groups.
Sample Preparation for VOCs determination
For each group of yellow mushrooms, 20 were taken respectively, weighed and equal volume of deionized water was added. The mixture was homogenized, and 100 g of the sample was weighed. Then, 36 g of NaCl was added and thoroughly mixed. Next, 5 g of the mixed sample was combined with 50 µL of a 0.04 g/L standard solution of 3-Nonanon and transferred into an SPME vial. Each sample was collected in triplicate, there were a total of 180 mushrooms in three groups.
HS-SPME conditions
A divinylbenzene/carboxen/polydimethylsiloxane (DVB/CAR/PDMS) SPME fiber with a thickness of 50/30 µm, obtained from Sigma-Aldrich (Supelco, Bellefonte, PA, USA), was used for the extraction of VOCs. The AOC 6000 auto-sampler (Shimadzu, Tokyo, Japan) was employed to extract VOCs from the samples. Prior to each analysis, the fiber was subjected to conditioning at 250℃ for a duration of 10 min. To promote the dissociation of VOCs, the samples were maintained at 45℃ for 15 min. Following this, the SPME fiber was placed in the headspace of the vial for 15 min at 80℃, with an agitator speed set at 300 rpm, to facilitate the extraction process50.
GC-MS analysis
A Shimadzu 2010plus gas chromatograph coupled with a QP2020 mass spectrometry system (Shimadzu, Tokyo, Japan) was utilized for sample analysis. The VOCs adsorbed on the fiber were thermally desorbed at 200℃for 1 min in the injector port using splitless mode. The chromatographic separation was carried out on an HP-INNOWAX capillary column (60 m×0.25 mm×0.25 μm, Agilent Technologies, Santa Clara, CA, USA). The temperature program began at 40℃and was maintained for 1 min, followed by a ramp of 2℃/min up to 200℃(with a 1-minute hold), and subsequently increased at 10℃/min to reach 230℃, where it was held constant for 10 min. Helium gas (purity 99.999%) served as the carrier gas, flowing at a constant rate of 1.0 mL/min50.
MS detection was conducted under electron ionization (EI) conditions with an ionization energy of 70 eV. The ion source temperature was set to 230℃, and the mass spectra were acquired over the range of m/z 50–500. Compound identification was achieved by comparing spectral similarity against the NIST 11s library database, requiring a matching score exceeding 90% and combined with the retention index (RI), the quality of peak matching for each VOC was carefully verified.
Statistical analysis
Quantification for VOCs was performed by the internal standard method, in which the content of each compound was normalized to that of 3-Nonanon. Peak area was measured by integration and was used for quantitative analysis. The calculation method adopts the semi-quantitative method with internal standards.
For data analysis and graphic presentation, Excel 2010 (Microsoft, Redmond, WA, USA) software was employed. An upset map and venn were created using online analysis software (Visual website, China). To explore clustering patterns and establish connections between groups and VOCs, PCA and HCA were carried out with Origin 2021 (MathWorks, Natick, MA, USA). Additionally, OPLS-DA was executed utilizing SIMCA version 14.0 (Umetrics, Umea, Sweden) and R (version 3.5.1).
Conclusion
In this study, the basic quality indices and VOCs of Tibetan yellow mushrooms with different cap opening stages were systematically evaluated. The results showed that as the degree of cap opening increased, both the weight of individual mushrooms and their respiratory intensity followed an upward trend. However, the variance in individual mushroom weight was substantial, reflecting significant individual differences. Additionally, no significant changes were observed in firmness and color, indicating that these basic quality parameters were not closely correlated with the degree of cap opening or associated quality characteristics. Using HS-SPME coupled with GC-MS, a total of 99 VOCs were identified across all groups, including 66 in the MNO, 75 in the MSO, and 77 in the MAO. The content of VOCs was highest in the MAO, followed by the MSO and MNO. Further analysis revealed 47 common VOCs and 26 unique VOCs among the three groups. PCA revealed distinct clustering patterns for the three groups, highlighting differences in VOCs that could be used to differentiate yellow mushrooms based on their cap opening stages. HCA further confirmed the separation of the three groups, and the MAO was finally clustered together. OPLS-DA identified a total of 75 DVOCs, with 47, 48, and 59 high-abundance of DVOCs detected between pairwise comparisons (MNO vs. MSO, MSO vs. MAO and MNO vs. MAO, respectively). Thirteen common DVOCs were also identified. K-means clustering classified these DVOCs into five categories, with Category 1 being the key DVOCs influencing the MAO. Notably, certain DVOCs were particularly enriched in the MSO (Categories 2 and 3) and MAO (Category 4). Category 5, which contained 12 DVOCs, exhibited decreasing abundance with increasing cap opening degree. These findings suggest a systematic pattern in the VOCs and their abundances across the three groups, encompassed both differences in common VOCs and unique VOCs. This study established a standardized identification framework for yellow mushrooms, provided a theoretical and scientific basis for post-harvest evaluation and artificial cultivation.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Xing, R. et al. Genetic variation and phylogenetic relationships of the ectomycorrhizal Floccularia luteovirens on the Qinghai-Tibet plateau. J. Microbiol. 55, 600–606 (2017).
Liu, Z., Jiao, Y., Lu, H., Shu, X. & Chen, Q. Chemical characterization, antioxidant properties and anticancer activity of exopolysaccharides from Floccularia luteovirens. Carbohydr. Polym. 229, (2020).
Ni, Y. et al. The research status and prospects of Floccularia luteovirens: A mycorrhizal fungus with edible fruiting bodies. J. Fungi. 9 (11), 1071 (2023).
Gan, X. et al. Draft genome assembly of Floccularia luteovirens, an edible and symbiotic mushroom on Qinghai-Tibet plateau. G3-Genes Genomes Genet. 10 (4), 1167–1173 (2020).
Xing, R. et al. Microbial communities inhabiting the Fairy ring of Floccularia luteovirens and isolation of potential mycorrhiza helper bacteria. J. Basic. Microbiol. 58 (6), 554–563 (2018).
Liu, Z., Lu, H., Zhang, X. & Chen, Q. The genomic and transcriptomic analyses of Floccularia luteovirens, a rare edible fungus in the Qinghai-Tibet plateau, provide insights into the taxonomy placement and fruiting body formation. J. Fungi. 7 (11), 887 (2021).
Guo, J. et al. The molecular mechanism of yellow mushroom (Floccularia luteovirens) response to strong ultraviolet radiation on the Qinghai-Tibet plateau. Front. Microbiol. 13, (2022).
Xing, R. et al. Fine-scale genetic diversity and genet dynamics of the Fairy ring fungus Floccularia luteovirens on the Qinghai-Tibet plateau. Fungal Ecol. 60, 101194 (2022).
Xing, R. et al. Key components, formation pathways, affecting factors, and emerging analytical strategies for edible mushrooms aroma: A review. Food Chem. 438, 137993 (2024).
Kalac, P. Chemical composition and nutritional value of European species of wild growing mushrooms: A review. Food Chem. 113 (1), 9–16 (2009).
Sun, L. B. et al. Advances in Umami taste and aroma of edible mushrooms. Trends Food Sci. Technol. 96, 176–187 (2020).
Tagkouli, D. G. et al. Volatile profiling of pleurotus eryngii and pleurotus ostreatus mushrooms cultivated on agricultural and agro-industrial by-products. Foods 10 (6), 1287 (2021).
Zhou, T. H. et al. The flavor quality of dried lentinus Edodes with different species and drying methods (charcoal roasting and naturally drying). J. Food Meas. Charact. 14 (1), 613–622 (2020).
Zhu, R. et al. The flavors of edible, mushrooms: a comprehensive review of volatile organic compounds and their analytical methods. Crit. Rev. Food Sci. Nutr. 64 (16), 5568–5582 (2022).
Song, W. et al. Characterization of aroma dynamic changes during six developing stages of yellow cultivar flammulina filiformis based on E-Nose, GC x GC-TOF MS, GC-IMS, and GC-O-MS. Food Biosci. 61, (2024).
Tian, R., Liang, Z., Wang, Y. & Zeng, N. Analysis of aromatic components of two edible mushrooms, Phlebopus portentosus and Cantharellus yunnanensis using HS-SPME /GC-MS. Results Chem. 4, (2022).
Selli, S., Guclu, G., Sevindik, O. & Kelebek, H. Variations in the key aroma and phenolic compounds of Champignon (Agaricus Bisporus) and oyster (Pleurotus Ostreatus) mushrooms after two cooking treatments as elucidated by GC-MS-O and LC-DAD-ESI-MS/MS. Food Chem. 354, (2021).
Jung, M. Y. et al. Characterization of volatile profiles of six popular edible mushrooms using headspace-solid-phase Microextraction coupled with gas chromatography combined with chemometric analysis. J. Food Sci. 84 (3), 421–429 (2019).
Kleofas, V. et al. Aroma profile of the anise-like odour mushroom Cortinarius odorifer. Flavour. Frag J. 30 (5), 381–386 (2015).
Li, W. et al. The anabolism of sulphur aroma volatiles responds to enzymatic and non-enzymatic reactions during the drying process of Shiitake mushrooms. Food Chem. 371, (2022).
Guo, Y. et al. Characteristic volatiles fingerprints and changes of volatile compounds in fresh and dried Tricholoma Matsutake singer by HS-GC-IMS and HS-SPME-GC-MS. J. Chromatogr. B 1099, 46–55 (2018).
Ni, Y. et al. Selection and validation of reference genes for normalization of gene expression in Floccularia luteovirens. Fungal Biol. 128 (1), 1596–1606 (2024).
Fu, Y. et al. Metabolomics reveals Dopa melanin involved in the enzymatic Browning of the yellow cultivars of East Asian golden needle mushroom (Flammulina filiformis). Food Chem. 370, (2022).
Ng, T. B. & Wang, H. X. A homodimeric laccase with unique characteristics from the yellow mushroom Cantharellus cibariusi. Biochem. Biophys. Res. Commun. 313 (1), 37–41 (2004).
Vinogradov, E., Petersen, B. O., Duus, J. O. & Wasser, S. The structure of the glucuronoxylomannan produced by culinary-medicinal yellow brain mushroom (Tremella mesenterica ritz.:fr., Heterobasidiomycetes) grown as one cell biomass in submerged culture. Carbohydr. Res. 339 (8), 1483–1489 (2004).
Xiao, M. et al. Influence of drying methods on the morphological features, microstructural properties, and antioxidant performance of Floccularia luteovirens: a metabolomic analysis. J. Fungi. 11 (1), 78 (2025).
Lu, B., Perez-Moreno, J., Zhang, F., Rinaldi, A. C. & Yu, F. Aroma profile of two commercial truffle species from Yunnan and sichuan, china: inter- and intraspecific variability and shared key compounds. Food Sci. Hum. Wellness. 10 (2), 163–173 (2021).
Xu, X. et al. Identification of dihydro-β-Ionone as a key aroma compound in addition to C8 ketones and alcohols in Volvariella Volvacea mushroom. Food Chem. 293, 333–339 (2019).
Zhuang, J. et al. Comparative flavor profile analysis of four different varieties of Boletus mushrooms by instrumental and sensory techniques. Food Res. Int. 136, (2020).
Hou, Z. et al. Effects of drying temperature on Umami taste and aroma profiles of mushrooms (Suillus Granulatus). J. Food Sci. 87 (5), 1983–1998 (2022).
Splivallo, R., Ottonello, S., Mello, A. & Karlovsky, P. Truffle volatiles: from chemical ecology to aroma biosynthesis. New. Phytol. 189 (3), 688–699 (2011).
Liu, Q. et al. Coating Shiitake mushrooms (Lentinus edodes) with a polysaccharide from Oudemansiella radicata improves product quality and flavor during postharvest storage. Food Chem. 352, (2021).
Liu, Q. et al. Relationship between flavor and energy status in Shiitake mushroom (Lentinula edodes) harvested at different developmental stages. J. Food Sci. 86 (10), 4288–4302 (2021).
Wang, Y. et al. Difference in volatile aroma components of Stropharia rugosoannulata under two cultivated environments investigated by SPME-GC-MS. Foods 12 (14), 2656 (2023).
Feng, Y. et al. Effects of different monochromatic lights on Umami and aroma of dried suillus granulatus. Food Chem. 404, (2023).
Gao, F., Xie, W., Zhang, H., Li, S. & Li, T. Variations of quality and volatile components of corels (Morchella sextelata) during storage. J. Plant. Physiol. 290, (2023).
Wei, J. et al. Changes in the metabolome and nutritional quality of pulp from three types of Korla fragrant Pears with differentappearances as revealed by widely targeted metabolomics. Plants 12, 3981 (2023).
Guo, Q. et al. Comparative analysis of volatile profiles in four pine-mushrooms using HS-SPME/GC-MS and E-nose. Food Control 134, (2022).
Kalogiouri, N. P. et al. Headspace solid-phase Microextraction followed by gas chromatography-mass spectrometry as a powerful analytical tool for the discrimination of truffle species according to their volatiles. Fron Nutr. 9, (2022).
Xia, N. et al. Genome-wide association analysis of 1-octen-3-ol content related to soymilk off-flavor in soybean seed. Crop Pasture Sci. 70 (2), 133–139 (2019).
Li, Q. et al. Chemical compositions and volatile compounds of Tricholoma Matsutake from different geographical areas at different stages of maturity. Food Sci. Biotechnol. 25 (1), 71–77 (2016).
Cho, I. H., Namgung, H. J., Choi, H. K. & Kim, Y. S. Volatiles and key odorants in the pileus and stipe of pine-mushroom (Tricholoma Matsutake Sing). Food Chem. 106 (1), 71–76 (2008).
Orban, A., Weber, A., Herzog, R., Hennicke, F. & Ruehl, M. Transcriptome of different fruiting stages in the cultivated mushroom Cyclocybe Aegerita suggests a complex regulation of fruiting and reveals enzymes putatively involved in fungal Oxylipin biosynthesis. Bmc Genom. 22 (1), (2021).
Qi, D. et al. Study on the effects of rapid aging technology on the aroma quality of white tea using GC-MS combined with chemometrics: in comparison with natural aged and fresh white tea. Food Chem. 265, 189–199 (2018).
Martinez, R., Valderrama, N., Moreno, J. & de Bruijn, J. Aroma characterization of grape juice enriched with grapevine by-products using thermomaceration. Chil. J. Agric. Res. 77 (3), 234–242 (2017).
Umano, K., Hagi, Y. & Shibamoto, T. Volatile chemicals identified in extracts from newly hybrid citrus, Dekopon (Shiranuhi Mandarin suppl. J). J. Agric. Food Chem. 50 (19), 5355–5359 (2002).
Song, W. et al. Variations in key aroma compounds and aroma profiles in yellow and white cultivars of Flammulina filiformis based on gas chromatography-mass spectrometry-olfactometry, aroma recombination, and omission experiments coupled with odor threshold concentrations. Foods 13 (5), 684 (2024).
Nielsen, G. S. & Poll, L. Determination of odor active aroma compounds in freshly cut Leek (Allium ampeloprasum var. Bulga) and in long-term stored frozen unblanched and blanched Leek slices by gas chromatography olfactometry analysis. J. Agric. Food Chem. 52 (6), 1642–1646 (2004).
Li, S. et al. Machine learning-enhanced flavoromics: identifying key aroma compounds and predicting sensory quality in sauce-flavor Baijiu. Food Chem. 475, (2025).
Wang, Y. et al. Identification of volatile organic compounds and analysis of aroma characteristics in ten Pear syrups. Foods 13 (20), 3223 (2024).
Funding
This work was Supported by Science and Technology Projects of Xizang Autonomous Region, China (XZ202401ZY0039).
Author information
Authors and Affiliations
Contributions
Y.Y. conceived the idea and designed the experiments, and provided technical assistance. Y.W. Writing Original draft. W. T. provided technical assistance, and corrected the manuscript. W. Z. technical assistance. X. S. analyzed the data. X. W. performed most of the experiments.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, Y., Tong, W., Zhang, WH. et al. Quality characteristics and volatile organic compound profiling of yellow mushrooms at different cap opening stages. Sci Rep 15, 29961 (2025). https://doi.org/10.1038/s41598-025-15137-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-15137-y