Abstract
Pulmonary mucormycosis (PM) and invasive pulmonary aspergillosis (IPA) are both invasive pulmonary fungal infections with similar clinical manifestations. Few studies have focused on the expression of serum iron in patients with PM and IPA. A prospective, case‒control study was conducted between January 2023 and December 2024. Patients diagnosed with PM or IPA were assigned to the PM group or the IPA group, respectively. Clinical data were recorded for all enrolled patients. Serum iron, serum ferritin, unsaturated iron-binding capacity (UIBC), total iron-binding capacity (TIBC), and percentage transferrin saturation levels were determined via an automatic biochemical detector. A total of 58 patients with PM and 49 patients with IPA were enrolled. Nine patients in the PM group and one patient in the IPA group died during hospitalization. The serum iron, UIBC, and TIBC levels in the PM group were significantly lower than those in the IPA group (all p < 0.05). The serum ferritin level was significantly greater in the PM group than in the IPA group (p = 0.021). Binary logistic regression analysis revealed that only the TIBC level was an independent risk factor for PM [OR (95% CI) = 0.892 (0.839–0.947), p < 0.001]. Cox proportional hazards regression analysis revealed that the serum iron concentration [HR (95% CI) = 0.872 (0.784–0.969); p = 0.011] was an independent predictor of mortality risk in PM patients. Lower TIBC levels at admission were associated with a greater risk of PM, and reduced serum iron levels predict poor prognosis in patients with PM. Using IPA patients as controls, we preliminarily revealed aberrant iron metabolism in PM patients.
Trial registration: ClinicalTrials.gov ID NCT06495905, Date of registration July 11, 2024 (retrospectively registered).
Similar content being viewed by others
Introduction
Pulmonary mucormycosis (PM) and invasive pulmonary aspergillosis (IPA) are life-threatening opportunistic fungal infections that predominantly affect immunocompromised hosts, including individuals with hematologic malignancies, solid organ transplants, or uncontrolled diabetes1,2. Despite overlapping risk factors and clinical presentations, these infections exhibit distinct pathophysiological features and outcomes. Notably, compared with IPA, PM is associated with a greater incidence of massive hemoptysis, contributing to its elevated mortality rates3. The underlying mechanisms responsible for this disparity remain incompletely understood. However, the unique iron requirements of Mucorales (causative agents of mucormycosis) have garnered increasing attention4.
Mucorales exhibit a striking dependence on iron acquisition for growth and virulence. Experimental studies have demonstrated that Mucorales fungi possess efficient iron-scavenging mechanisms, including the production of high-affinity iron permeases and siderophores, enabling them to thrive in iron-rich environments5. In contrast, Aspergillus species, while also capable of utilizing host iron, appear to be less reliant on iron-dependent pathways for pathogenicity6. This iron dependency may explain the propensity of mucormycosis to invade blood vessels, leading to thrombosis, tissue necrosis, and subsequent massive hemoptysis. Clinical observations further support this hypothesis, as iron overload states (e.g., due to repeated transfusions or deferoxamine therapy) are well-established risk factors for mucormycosis but not aspergillosis7.
Despite these pathophysiological insights, comparative analyses of systemic iron metabolism in patients with PM versus IPA remain scarce. Current data are largely derived from in vitro or animal models, limiting their translational relevance to human disease8. Elucidating differences in serum iron parameters, including iron concentration, transferrin saturation, and ferritin levels, between these two groups could provide critical insights into disease mechanisms, prognostication, and therapeutic strategies. For example, abnormal serum iron levels in mucormycosis patients might reflect both fungal iron scavenging and host dysregulation, potentially serving as biomarkers for disease severity or targets for adjunctive therapies such as iron chelation.
This study aimed to compare the serum iron profiles of patients with proven PM and IPA, hypothesizing that PM patients exhibit distinct iron metabolic signatures correlated with clinical outcomes, including hemoptysis. By bridging this knowledge gap, our findings may inform novel diagnostic and therapeutic approaches to mitigate the high morbidity and mortality associated with these devastating fungal infections.
Methods
Study population
This prospective, case‒control study was conducted from January 2023 through December 2024 among patients hospitalized in Beijing Tsinghua Changgung Hospital. Patients diagnosed with PM were included in the PM group. PM was defined on the basis of the following criteria: the patient’s clinical manifestations and imaging findings were in line with the PM features; pathological examination of the lung tissue revealed mucormycosis hyphae; and (or) microscopic examination, culture and metagenomics next-generation sequencing (mNGS) of the tissue sample or bronchoalveolar lavage fluid (BALF) revealed mucormycosis.
Patients diagnosed with IPA were included in the IPA group. IPA was defined by the following criteria: the patient’s clinical manifestations and imaging findings were in line with IPA features; pathological examination of the lung tissue revealed Aspergillus sp.; and (or) microscopic examination, culture and metagenomics next-generation sequencing (mNGS) of the tissue sample or BALF revealed Aspergillus sp.
The exclusion criteria were age < 18 years or the presence of any of the following: pregnancy, end-stage renal or liver disease, or active tuberculosis.
Sample size calculation
In this study, we set the type I error/significance level (two-sided) at α = 0.05 and the type II error at β = 0.10 to provide 90% power. The test standard deviation was Zα = 1.96 and Zβ = 1.282. Assuming that in invasive pulmonary fungal infections, the incidence rates of PM and IPA were P0 = 0.10 and P1 = 0.70, respectively9. The sample size was calculated as follows:
R = \(\:\frac{{P}_{1}}{{P}_{0}};\:\)A = P1(1 − P0) + P0(1 − P1); B = (R − 1) P0(1 − P0); K = (A + B) (RA − B) − R(P1 − P0)2.
NPM = \(\:\frac{{\text{Z}}_{{\upbeta\:}}^{2}\text{K}\:+\:{\text{Z}}_{{\upalpha\:}}^{2}{(\text{A}\:+\:\text{B})}^{2}+2{\text{Z}}_{{\upalpha\:}}{\text{Z}}_{{\upbeta\:}}\:(\text{A}\:+\:\text{B})\sqrt{\text{K}}\:}{{\left({P}_{1}\:-\:{P}_{0}\right)}^{2}(\text{A}\:+\:\text{B})}\) = 44.
Assuming an approximate 1:1 sample size ratio between the PM group and the IPA group, a minimum of 44 patients should ultimately be enrolled in each group. All patients provided informed consent. This study was approved by the medical ethics committee of Beijing Tsinghua Changgung Hospital (No. 23617-0-01), all methods were carried out in accordance with the relevant guidelines and regulations.
Blood sample test
The peripheral blood sample tests were conducted by the laboratory department of our hospital. Clinical parameters, including the whole blood leukocyte count (WBC), hemoglobin (HGB), C-reactive protein (CRP), procalcitonin (PCT), and interleukin-6 (IL-6), were recorded for all recruited patients. The iron profile, including serum iron, unsaturated iron-binding capacity (UIBC), total iron-binding capacity (TIBC), percentage transferrin saturation, and serum ferritin, was tested by a biochemistry laboratory.
Electronic bronchoscopy examination
Electronic bronchoscopy examination was carried out by an experienced interventional pulmonologist. Bronchoalveolar lavage and/or biopsy were performed on the severely affected site. BALF samples were collected in sterile cups. Biopsy tissues were immediately placed in formalin solution.
Clinical diagnosis
A chest CT was jointly interpreted by at least one radiologist and one pulmonologist. Microscopic examination and fungal culture were carried out by the microbiology laboratory of our hospital. The mNGS results were interpreted by at least one experienced pulmonologist. For histopathology, the tissue sections were stained with HE, PAS, and GMS to identify fungal hyphae. The diagnosis of PM was made by two pathologists on the basis of histopathological examination.
Statistical analysis
Normally distributed continuous variables are expressed as the means ± standard errors of the means (SEMs), nonnormally distributed continuous variables are expressed as medians (interquartile ranges), and categorical variables are expressed as numbers (percentages). For equivalent variables with a normal distribution, independent Student’s t tests were used to compare two groups. Mann–Whitney U tests were used to compare categorical variables and abnormally distributed variables between two groups. Binary logistic regression was used to analyze the effect of the iron profile on Mucor infection. Cox proportional hazards regression analyses were used to analyze the effects of an array of variables on the 30-day survival of patients with PM during hospitalization.
A two-sided p value < 0.05 was considered statistically significant; confidence intervals (CIs) were set at 95%. Statistical analyses were performed by using GraphPad Prism version 7.00 software (GraphPad Software, La Jolla, CA, USA).
Results
1. Characteristics of the enrolled patients.
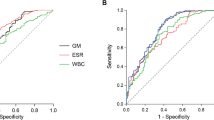
In total, 58 patients were diagnosed with PM in the PM group, and 49 patients were diagnosed with IPA in the IPA group. Clinical characteristics of all patients are presented in Table 1; Fig. 1.
The etiologies of the PM group were Rhizopus oryzae (forty-two patients), Rhizopus arrhizus (seven patients), and Rhizopus delemar (nine patients). The IPA group included Aspergillus fumigatus (twenty-seven patients), Aspergillus flavus (ten patients), Aspergillus terreus (seven patients), and Aspergillus niger (five patients).
Eight (13.8%) patients in the PM group died during hospitalization within 30 days, and one patient with PM died on the 43rd day of hospitalization. The main cause of death was confirmed to be rapid progression of PM (three patients), massive hemoptysis (five patients), and deterioration of underlying disease (one patient died of leukemia relapse). Only one patient died in the IPA group due to septic shock and deterioration of hepatocellular carcinoma.
In the PM group, only five (8.6%) patients were discharged with improved health conditions within 30 days (median: 21 days; range: 18–26 days), and forty-four (75.9%) patients were discharged after more than 30 days of hospitalization (median: 48 days; range: 41–71 days). In the IPA group, forty (81.6%) patients were discharged with improved health conditions within 30 days (median: 14 days; range: 9–28 days), and eight (16.3%) patients were discharged after more than 30 days of hospitalization (median: 47 days; range: 33–76 days).
Comparison of comorbidities between patients with PM and IPA.
2. Iron profile in each group.
Compared to the IPA group, the PM group had significantly lower serum iron [8.3 (5.5–12.7) vs. 11.8 (5.8–18.1) µmol/L, p = 0.039], UIBC [(21.8–8.9) vs. (26.5–10.5) µmol/L, p = 0.023], and TIBC [30.8 (25.5–38.9) vs. 41.5 (33.9–49.8) µmol/L, p < 0.001] levels. Conversely, serum ferritin was significantly higher in the PM group [901.3 (499.2–1869.0) vs. 688.1 (314.3–1015.0) µg/mL, p = 0.021]. Transferrin saturation did not differ significantly between the groups [25.3 (17.1–40.2) % vs. 28.7 (18.4–41.3) %, p = 0.584] (Fig. 2).
Histograms comparing the levels of serum iron-related markers in the PM group and IPA group. (A) Serum iron levels in the PM and IPA groups, p = 0.039. (B) Levels of unsaturated iron-binding capacity in the PM and IPA groups, p = 0.023. (C) Total iron-binding capacity in the PM and IPA groups, p < 0.001. (D) Percent transferrin saturation in the PM and IPA groups, p = 0.584. (E) Serum ferritin levels in the PM and IPA groups, p = 0.021.
3. Value of the iron profile in predicting PM.
Table 2 shows the results of binary logistic regression for patients suffering from PM. According to the univariate analysis, the ORs (95% CIs) of the serum iron, UIBC, TIBC, ferritin, and HGB levels were 0.942 (0.890–0.997), 0.950 (0.908–0.994)], 0.902 (0.855–0.952), 1.001 (1.000–1.001), and 0.976 (0.959–0.993), respectively, all p < 0.05. In the multivariate analysis, only the TIBC level was an independent risk factor for PM [OR (95% CI) = 0.892 (0.839–0.947), p < 0.001].
4. Prognostic value of the iron profile in patients with PM.
Table 3 shows the results of the Cox proportional hazards regression analysis of the effects on survival in PM patients. After adjusting for clinical data, only PCT [HR (95% CI) = 1.980 (1.283–3.057); p = 0.002)] and serum iron [HR (95% CI) = 0.872 (0.784–0.969); p = 0.011] could serve as independent predictors of mortality risk in PM patients.
Discussion
Invasive fungal infections (IFIs) pose a significant threat to immunocompromised individuals10, with Aspergillus and Mucorales being among the most prevalent causative agents. While IPA is relatively common, PM has a significantly higher mortality rate11. Both infections can manifest with similar respiratory symptoms, but a distinguishing clinical feature observed in PM is a propensity for hemoptysis. In this study, we presented two cohorts: a group of patients diagnosed with PM and a group of patients diagnosed with IPA. We also found that the PM group had a significantly greater incidence of hemoptysis than did the IPA group, with 8.6% of the PM patients dying from massive hemoptysis. The underlying reasons for this differential bleeding tendency remain poorly understood, hindering efforts to improve early diagnosis and targeted treatment strategies.
Traditionally, the angioinvasive nature of Mucorales is associated with a relatively high incidence of hemoptysis12. Mucorales exhibit a characteristic tropism for blood vessels, leading to thrombosis, infarction, and subsequent tissue necrosis, culminating in pulmonary hemorrhage13,14. However, the specific mechanisms that render Mucorales more angioinvasive and prone to causing hemoptysis than Aspergillus species are not fully understood. Emerging evidence suggests that iron metabolism may play a critical role in the pathogenesis of mucormycosis and could explain the increased risk of hemoptysis15. Iron is an essential nutrient for both the host and invading pathogens, and its availability can significantly influence the outcome of fungal infections16. Mucorales are known to be highly dependent on iron for their growth and virulence17. In contrast to Aspergillus, Mucorales possess efficient iron acquisition mechanisms, enabling them to thrive in iron-rich environments18. Therefore, we hypothesize that patients with PM may have abnormal iron metabolism levels compared with patients with IPA.
Studies on the expression of iron metabolism-related genes in PM or IPA are rare. During the COVID-19 pandemic, Mohan KH et al. compared serum iron indices (iron, ferritin, TIBC, UIBC and percentage transferrin saturation) between COVID-19 patients with mucormycosis (CAM) and without mucormycosis and reported lower TIBC levels in COVID-19 patients with mucormycosis, whereas the iron indices were not significantly different between CAM survivors and nonsurvivors19. Indu et al. examined serum ferritin levels in COVID-19 patients and reported that CAM patients had a significant increase in serum ferritin, which might be used as an early marker for screening for mucormycosis in COVID-19 patients20. In this study, we enrolled patients with IPA as a control group to highlight the distinct iron-seeking nature of mucormycosis and found that patients with mucormycosis had lower serum iron levels and lower UIBC and TIBC levels. However, some studies suggest that increased serum iron levels may be a risk factor for mucormycosis infection21,22. This discrepancy might be due to the different time points of the studies. In this study, almost all the patients with PM had received treatment at other hospitals for varying lengths of time before enrollment. It is postulated that serum iron metabolism profiles may vary across different phases of PM in affected individuals, with diminished serum iron levels potentially attributable to consumption.
Serum iron metabolism levels are intricately linked to overall health status and are subject to dynamic fluctuations influenced by a spectrum of physiological and pathological factors23. Notably, iron deficiency anemia and anemia caused by chronic disease have prominent effects24. Considering the protracted clinical courses of both PM and IPA, patients, serum iron levels are impacted by the dual burden of chronic disease-associated consumption and malnutrition, which frequently manifests as mild to moderate anemia. Consequently, hemoglobin levels and comorbidities were incorporated into the binary logistic regression analysis. The findings revealed that low TIBC levels remained an independent risk factor for PM even after adjusting for the confounding influence of hemoglobin and comorbidities. Of note, although ferritin levels were significantly elevated in the PM group compared to the IPA group, multivariate regression analysis revealed that ferritin did not independently contribute to the prediction of mucormycosis infection. While this finding contrasts with that of Indu et al.20, ferritin is generally recognized as a nonspecific inflammatory factor, and its levels are not strongly linked to the specific infectious agent.
Given that PM generally has a worse prognosis than IPA does, we conducted a further analysis to determine independent predictors of PM outcome. Using a Cox proportional hazards model, we identified elevated PCT and decreased serum iron as significant and independent risk factors for adverse PM outcomes. The negative association of elevated PCT with prognosis is well established in the literature, and this was also observed in our PM cohort, with higher PCT corresponding to greater disease severity. However, the prognostic literature on serum iron is quite heterogeneous. Ueda T et al.25. found that low serum iron was a marker of severity in acute decompensated heart failure patients, irrespective of hemoglobin or ferritin levels. Jie Shu et al.26. reported that high serum iron levels were associated with increased short- and long-term mortality in ICU patients with acute kidney injury. In cancer patients, Yang Y et al.27. reported that low serum iron levels were associated with a worse prognosis in gastric cancer patients treated with programmed death (PD-1) inhibitors and might be a quick and efficient biomarker to predict the efficacy of PD-1 inhibitors. Lan P, et al.28. reported that both low and high preoperative iron levels in stage II-III colorectal cancer patients were associated with unfavorable overall survival according to univariate and multivariate analyses. However, the prognostic role of serum iron in infectious disease remains relatively unexplored. Lan P et al.29. found that high serum iron levels were independently associated with increased 90-day mortality in patients with sepsis. In this study, we found that low serum iron levels are indicative of an unfavorable prognosis in patients with PM. Given that the PM patients included in this study presented with an existing disease course before admission, it would be optimal to assess serum iron levels at the onset of illness. Furthermore, serial measurements would better elucidate the potential role of iron metabolism in PM. Regrettably, our investigation only measured serum iron concentrations at the time of patient admission, which limits our ability to compare these levels to those preceding mucormycosis infection within the same individuals. Moreover, serial measurements are difficult, if not impossible, in many cases, particularly for PM patients who have received prior treatment at other hospitals before admission.
In conclusion, the serum iron, UIBC, and TIBC levels detected on admission are significantly lower in PM patients than in IPA patients, and a low TIBC level is a risk factor for PM. Decreased serum iron is predictive of a less favorable disease course in patients with PM. With IPA serving as a comparative control, this study revealed disrupted iron metabolism in patients with mucormycosis, providing a clinical basis for further investigation into the siderophilic nature of Mucorales. In immunocompromised patients with high clinical suspicion for pulmonary fungal infection, early detection of iron profile may aid in the timely identification of mucormycosis and the prompt recognition of high-risk individuals.
Data availability
The datasets generated and analyzed during the current study are not publicly available due to health privacy concerns but are available from the corresponding author upon reasonable request.
References
Muthu, V. et al. Has the mortality from pulmonary mucormycosis changed over time? A systematic review and meta-analysis. Clin. Microbiol. Infect. 27 (4), 538–549. https://doi.org/10.1016/j.cmi.2020.12.035 (2021).
Pardo, E. et al. Invasive pulmonary aspergillosis in critically ill patients with hematological malignancies. Intensive Care Med. 45 (12), 1732–1741. https://doi.org/10.1007/s00134-019-05789-6 (2019).
Alqarihi, A., Kontoyiannis, D. P. & Ibrahim, A. S. Mucormycosis in 2023: an update on pathogenesis and management. Front. Cell. Infect. Microbiol. 21, 13: 1254919. https://doi.org/10.3389/fcimb.2023.1254919 (2023).
Tanwar, M. et al. Comprehensive review on the virulence factors and therapeutic strategies with the aid of artificial intelligence against mucormycosis. ACS Infect. Dis. 10 (5), 1431–1457. https://doi.org/10.1021/acsinfecdis.4c00082 (2024).
Wang, J. & Pantopoulos, K. Regulation of cellular iron metabolism. Biochem. J. 434 (3), 365–381. https://doi.org/10.1042/BJ20101825 (2011).
Haas, H. Iron: a key nexus in the virulence of Aspergillus fumigatus. Front. Microbiol. 3, 28. https://doi.org/10.3389/fmicb.2012.00028 (2012).
Cassat, J. E. & Skaar, E. P. Iron in infection and immunity. Cell. Host Microbe. 13 (5), 509–519. https://doi.org/10.1016/j.chom.2013.04.010 (2013).
Alqarihi, A. et al. GRP78 and integrins play different roles in host cell invasion during mucormycosis. mBio 11 (3), e01087–e01020. https://doi.org/10.1128/mBio.01087-20 (2020).
Denning, D. W. Global incidence and mortality of severe fungal disease. Lancet Infect. Dis. 24 (7), e428–e438. https://doi.org/10.1016/S1473-3099(23)00692-8 (2024).
Pathakumari, B., Liang, G. & Liu, W. Immune defense to invasive fungal infections: A comprehensive review. Biomed. Pharmacother. 130, 110550. https://doi.org/10.1016/j.biopha.2020.110550 (2020).
Alexander, B. D. et al. Guidance on imaging for invasive pulmonary aspergillosis and mucormycosis: from the imaging working group for the revision and update of the consensus definitions of fungal disease from the EORTC/MSGERC. Clin. Infect. Dis. 72 (Suppl 2), S79–S88. https://doi.org/10.1093/cid/ciaa1855 (2021).
Skiada, A., Pavleas, I. & Drogari-Apiranthitou, M. Epidemiology and diagnosis of mucormycosis: an update. J. Fungi (Basel). 6 (4), 265. https://doi.org/10.3390/jof6040265 (2020).
Barker, K. R. et al. A practical workflow for the identification of aspergillus, fusarium, mucorales by MALDI-TOF MS: database, medium, and incubation optimization. J. Clin. Microbiol. 60 (12), e0103222. https://doi.org/10.1128/jcm.01032-22 (2022).
Zhang, M. et al. Pulmonary artery pseudoaneurysm caused by pulmonary mucormycosis. Am. J. Respir Crit. Care Med. 209 (7), 879–880 (2024).
Singh, A. et al. Lactoferrin, a potential iron-chelator as an adjunct treatment for mucormycosis - A comprehensive review. Int. J. Biol. Macromol. 187, 988–998. https://doi.org/10.1016/j.ijbiomac.2021.07.156 (2021).
Happacher, I. et al. Fungal siderophore metabolism with a focus on Aspergillus fumigatus: impact on biotic interactions and potential translational applications. Essays Biochem. 67 (5), 829–842. https://doi.org/10.1042/EBC20220252 (2023).
Nicolás, F. E. et al. Mucorales species and macrophages. J. Fungi (Basel). 6 (2), 94. https://doi.org/10.3390/jof6020094 (2020).
Stanford, F. A. et al. Expression patterns in reductive iron assimilation and functional consequences during phagocytosis of lichtheimia corymbifera, an emerging cause of mucormycosis. J. Fungi (Basel). 7 (4), 272. https://doi.org/10.3390/jof7040272 (2021).
Kumar, H. M. et al. Serum iron indices in COVID-19-associated mucormycosis: A case–control study. Mycoses 65 (1), 120–127. https://doi.org/10.1111/myc.13391 (2022).
Dp, I., Yadhav Ml, K. & Gs, C. Is serum ferritin an early marker for COVID-19-Associated mucormycosis?? Cureus. ; 15(3): e36734. (2023). https://doi.org/10.7759/cureus.36734
Pai, V. et al. Rhino-orbito-cerebral mucormycosis: pictorial review. Insights Imaging. 12 (1), 167. https://doi.org/10.1186/s13244-021-01109-z (2021).
Paidisetty, P. et al. Changing trend of risk factors of mucormycosis including diabetes, acidosis, and serum iron in the second wave of COVID-19. Indian J. Microbiol. 62 (4), 602–609. https://doi.org/10.1007/s12088-022-01038-5 (2022).
Billesbølle, C. B. et al. Structure of hepcidin-bound Ferroportin reveals iron homeostatic mechanisms. Nature 586 (7831), 807–811. https://doi.org/10.1038/s41586-020-2668-z (2020).
Pasricha, S. R. et al. Iron deficiency. Lancet 16 (10270), 233–248. https://doi.org/10.1016/S0140-6736(20)32594-0 (2021).
Ueda, T. et al. Serum iron: a new predictor of adverse outcomes independently from serum hemoglobin levels in patients with acute decompensated heart failure. Sci. Rep. 11 (1), 2395. https://doi.org/10.1038/s41598-021-82063-0 (2021).
Shu, J. et al. Elevated serum iron level is a predictor of prognosis in ICU patients with acute kidney injury. BMC Nephrol. 21 (1), 303. https://doi.org/10.1186/s12882-020-01965-9 (2020).
Yang, Y., Li, Y. & Chen, Z. Impact of low serum iron on treatment outcome of PD-1 inhibitors in advanced gastric cancer. BMC Cancer. 23 (1), 1095. https://doi.org/10.1186/s12885-023-11620-9 (2023).
Lan, P. et al. Preoperative iron status is a prognosis factor for stage II and III colorectal cancer. Int. J. Clin. Oncol. 26 (11), 2037–2045. https://doi.org/10.1007/s10147-021-01995-9 (2021).
Lan, P. et al. High serum iron level is associated with increased mortality in patients with sepsis. Sci. Rep. 8 (1), 11072. https://doi.org/10.1038/s41598-018-29353-2 (2018).
Acknowledgements
Not applicable.
Funding
This study was funded by the Beijing Hospitals Authority Clinical Medicine Development of special funding support, code: ZLRK202323.
Author information
Authors and Affiliations
Contributions
The roles of the authors in this study were as follows: XD-M obtained research funding. XW-H, J-X, L-L, LN-Z and QZ-L were in charge of sample preservation and reinforced the clinical data in the database. QZ-L conceived this study, analyzed the data, and wrote and edited the manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All the subjects provided informed consent. This study was approved by the medical ethics committee of Beijing Tsinghua Changgung Hospital (No. 23617-0-01).
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Luo, Q., He, X., Xu, J. et al. Reduced serum iron levels predict poor prognosis in pulmonary mucormycosis patients: a prospective, case‒control study. Sci Rep 15, 30395 (2025). https://doi.org/10.1038/s41598-025-15186-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-15186-3