Abstract
Liver cirrhosis leads to portal hypertension and hepatic encephalopathy (HE). Bile duct ligation (BDL) is a valid model for developing HE in animal models. The present study investigated the protective effects of gallic acid (GA) against BDL-induced injuries in male rats. 64 male Wistar rats were randomly divided into eight equal groups: Sham, BDL, BDL+20 mg/kg GA, BDL+30 mg/kg GA, Sham+compound C (CC), BDL+CC, BDL+20 mg/kg GA+CC, and BDL+30 mg/kg GA+CC. The rats received GA by gavage and 6.2 µg of CC via bilateral intraventricular injection daily for four weeks. Serum ammonia, the expression of inflammatory cytokines and caspase-3 (Casp-3), and the number of degenerated neurons were significantly increased in BDL groups compared to the sham group. Antioxidant activity and adenosine monophosphate-activated protein kinase (AMPK) gene expression in the BDL groups significantly decreased compared to the sham group. GA significantly reduced serum ammonia, the expression of inflammatory cytokines, and Casp-3, as well as the number of degenerated neurons in the treated groups. GA significantly increased antioxidant activity and AMPK expression. In the groups that received CC, the improving effect of GA was reduced. GA could protect the hippocampus and cerebellum of male rats against the detrimental effects of BDL due to its antioxidant and anti-inflammatory properties and the activation of AMPK.
Similar content being viewed by others
Introduction
It is established that acute liver damage impairs detoxification function, leading to acute liver failure. This results in toxin accumulation, which affects multiple systems and causes reversible complications such as jaundice, electrolyte disorders, and hepatic encephalopathy (HE)1. Despite significant advances in therapeutic strategies, liver cirrhosis remains a formidable global health challenge, with 45–70% of patients progressing to advanced stages, and 10–20% requiring transjugular intrahepatic portosystemic shunt (TIPS) placement for complications2. The underlying mechanisms of HE are not yet fully elucidated. However, hyperammonemia, oxidative stress, inflammation, mitochondrial dysfunction, and astrocyte activation represent key contributors to its pathogenesis3,4. HE leads to personality disorders, depression, and astrocyte dysfunction5. Inflammation, oxidative stress, neurotoxins, and impaired blood–brain barrier (BBB) permeability play central roles in the pathogenesis of HE6. When ammonia accumulates due to impaired hepatic metabolism caused by enzymatic defects or acute liver injury. It crosses the BBB. This leads to astrocyte dysfunction, neuroinflammation, cerebral edema7. Elevated ammonia levels induce lipid peroxidation, generate reactive oxygen species (ROS) and malondialdehyde (MDA), and upregulate myeloperoxidase (MPO). Concurrently, they suppress key antioxidant defenses including glutathione (GSH) and catalase (CAT) activity amplifying oxidative damage in HE8,9. Injured hepatocytes generate ROS, stimulating Kupffer cells and hepatocytes to release pro-inflammatory cytokines. This amplifies hepatic inflammation, when compounded by hyperammonemia elevates systemic levels of key inflammatory mediators, including interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), and the immunoregulatory interleukin-10 (IL-10). These processes collectively drive neuroinflammation in HE10,11.
Hyperammonemia further promotes neuronal apoptosis via the intrinsic (mitochondrial) pathway. Elevated ammonia triggers mitochondrial permeability transition, leading to cytochrome c (Cyt c) release and subsequent caspase-3 (Casp-3) activation, a key executioner of apoptotic cell death in the brain3. In animal models, bile duct ligation (BDL) has been accepted by the International Society for HE and Nitrogen Metabolism (ISHEN) as a valid model for HE12. Due to dysfunction, a cirrhotic liver loses its detoxification capacity, leading to increased neurotoxin levels. Neurotoxins like ammonia can cross the BBB, causing hepatic encephalopathy. Also, inflammation and oxidative stress can significantly contribute to its pathogenesis13. Studies have shown that BDL rats with chronic liver failure develop decreased motor activity and cerebral edema, mimicking clinical manifestations and reflecting neurochemical features of hepatic encephalopathy in cirrhotic patients13. In rats with hepatic encephalopathy due to liver cirrhosis along with hyperammonemia, neuroinflammation is typically more pronounced in the cerebellum and hippocampus, that manifest memory impairment and motor coordination deficits14. Adenosine monophosphate (AMP) serves as the primary monitor of intracellular energy status due to its marked concentration fluctuations relative to adenosine triphosphate (ATP) and adenosine diphosphate (ADP). The AMP-activated protein kinase (AMPK), a serine/threonine kinase—acts as the central sensor for cellular AMP levels. AMPK is activated by energy-depleting stresses including oxidative stress, glucose deprivation, hypoxia, and ischemia, effectively functioning as a metabolic switch during ATP depletion15,16,17. Abnormal regulation of AMPK is increasingly recognized as a critical pathophysiological factor in multiple brain disorders including Alzheimer’s, amyotrophic lateral sclerosis, Parkinson’s, Huntington’s, and stroke18. The impairment of brain energy metabolism and dysregulation of cerebral AMPK signaling are central role to the pathophysiology of HE19. Dorsomorphin or compound C (CC) has been used for various medicinal purposes such as inhibiting the bone morphogenetic protein signaling (BMP) and stem cell differentiation, but it is mainly used as an inhibitor of the AMPK pathway20.
Gallic acid (3,4,5-trihydroxybenzoic acid, GA) is a naturally occurring polyphenolic compound abundantly present in fruits (grapes, berries), nuts (walnuts, hazelnuts, brazil nuts), and tea leaves21,22. It has antioxidant and anti-inflammatory properties and is employed in the treatment of hyperglycemia21,23,24. GA has demonstrated significant therapeutic potential in metabolic syndromes, particularly type 1 (T1DM) and type 2 diabetes (T2DM), primarily through activation of the AMPK pathway25. By activating the AMPK pathway in hepatocytes, the accumulation of fat in hepatocytes is prevented, thereby reducing apoptosis and the inflammatory response in hepatocytes and macrophages26. More recently, we indicated that GA improved memory and anxiety-like behaviors in rats with HE27. However, the mechanisms underlying the AMPK-mediated protective effects of GA on HE remain unclear. Therefore, the aim of this study was to demonstrate the effect of GA with antioxidant and anti-inflammatory properties on oxidative stress, apoptotic and inflammatory biomarkers, and AMPK expressions, and histopathological changes in the hippocampus and cerebellum following HE caused by BDL through activating the AMPK pathway.
Materials and methods
Drugs and chemicals
GA (CAS number: 149–91-7) and CC (CAS number: 866405–64-3) were purchased from the Sigma chemical company (Germany). The RNA extraction kit, cDNA synthesis kit, and real-time PCR master mix (Lot: RPM38) were acquired from the AnaCell company (Iran). Xylene (CAS number: CAS 1330–20-7) and entellan (CAS number: 107960) were purchased from Merck (Germany). Cresyl violet (CAS number: 10510–54-0) was bought from Merck (Germany).
Animals
All experimental procedures involving animals in this study were performed under the relevant guidelines and regulations of the ARRIVE guidelines28. All methods of working with laboratory animals in this study were performed in accordance with the relevant guidelines, and were approved by the Ethics Committee of Kerman University of Medical Sciences, Iran (approval number: IR.KMU.REC.1399.470). A total of 64 adult male albino Wistar-type rats weighing 220–250 g were kept in plastic cages in the animal house of the Neuroscience Research Center at Kerman University of Medical Sciences, under standard conditions including a temperature of 23°C, humidity of 40–50%, and 12 h of light and dark cycles. Throughout this period, the animals were allowed free access to water and food.
Experimental design
The sample size will be selected with a 95% confidence level using previous studies and the formula n = (DF / k) + 1. K represents the number of groups and n represents the volume or size of animals in the groups. Degrees of freedom (DF) which will usually be between 10 and 2029. The sample size was estimated using the equation approach of four rats per group. However, to ensure robust results and to estimate the possibility of animal mortality, the final sample size was increased to eight rats per group. Randomization was performed before the start of the study. After organizing the study groups, animals were randomly allocated to each study group, then the intervention began. In this experimental study, 64 male Wistar rats were randomly divided into eight equal groups30. The number of rats in this study was carried out according to the ARRIVE guidelines28, and was approved by the Ethics Committee of Kerman University of Medical Sciences.
Group 1: Sham, rat underwent laparotomy without BDL surgery.
Group 2: BDL, rats in this group underwent BDL surgery.
Group 3: BDL+ 20 mg/kg GA; rats received GA once daily by gavage for 28 days31.
Group 4: BDL+ 30 mg/kg GA; rats received GA once daily by gavage for 28 days32.
Group 5: Sham+CC.
Group 6: BDL+CC.
Group 7: BDL+ 20 mg/kg GA+CC.
Group 8: BDL+ 30 mg/kg GA+CC.
About 6.2 µg of CC was administered for each rat via bilateral intraventricular injection for four weeks on a daily basis (ventricles on the side of the brain) for all the receiving groups33. CC was dissolved in dimethyl sulfoxide (DMSO) + normal saline. Injection was started one week after stereotaxic surgery and intraventricular cannulation.
GA was dissolved in normal saline and its administration was started one day after BDL surgery.
If more than two animals in each group died, replacements were placed for the same group.
Surgical procedures
Stereotaxic cannula implants
Animals were anesthetized by an intraperitoneal (IP) injection of ketamine (87.5 mg/kg) and xylazine (12 mg/kg). Rats were fixed by the stereotaxic instrument. The scalp was shaved and cleaned with 70% ethanol. Then it was cut in the middle line and washed with normal saline. Stereotaxic coordinates were determined with rat brain atlas. Due to differences in the age and weight of rats, adjustment of the injection coordinates was required. Afterward, two small punctures were made by a micro dental drill in the parietal bone behind the bregma. The cannulas were fixed to the skull with two small screws and acrylic cement34.
BDL surgery
Animals were anesthetized by an IP injection of ketamine (87.5 mg/kg) and xylazine (12 mg/kg). BDL surgery was performed under sterile conditions. After shaving the abdomen and disinfecting this area, a midline incision was made. By removing the liver, the common bile duct appeared. Then ligation was performed in two places with 4-0 silk. One was ligated near the liver hilus and the other was ligated about 2 cm below the liver hilus. The common bile duct between the two ligature areas was cut. Finally, the abdominal muscles were sutured with 3-0 catgut chromic, and the skin was sutured with 3-0 nylon. Rats were euthanized four weeks (28 days) after BDL, and they were given free access to food and water during this time35. At the end of the study, the animals were anesthetized with ketamine/xylazine (87.5 mg/kg and 12 mg/kg, respectively) and euthanized after blood was collected from the right ventricle and the hippocampus was isolated. Then, the animals’ bodies were placed in a special well for animal euthanasia. All animal euthanasia procedures were performed according to the ethical checklist for working with laboratory animals of Kerman University of Medical Sciences, and were approved by the Ethics Committee of Kerman University of Medical Sciences.
Biochemical parameters
Evaluation of serum ammonia
Four weeks after BDL surgery, rats were anesthetized by ketamine/xylazine. They were placed in the supine position and their abdomens were incision from the midline, and then the diaphragm of the animals was opened from the abdominal cavity and the heart was accessed, and then blood was taken from the right ventricle of the animals with a syringe. The blood of the animals was stored in EDTA tubes placed on ice bags. After centrifugation, the plasma of the blood samples was separated. Lysed samples were discarded due to falsely elevated ammonia. Plasma was stored at the temperature of − 70 °C until the time of analysis. Plasma ammonia levels (NH3) were measured using an automatic analyzer (Biotecnica instrument BT1000) and ammonia kit (REF: BXC0376A) from the Biorexfars Company (Iran). Six serum ammonia measurements were considered for each group.
Preparation of tissue homogenates
Hippocampal and cerebellar tissues were washed with cold normal saline after isolation. Then they were homogenized ten times in phosphate-buffered solution (pH 7.0). The prepared homogeneous solution was centrifuged at 12,000 rpm and 4 °C for 15 min. The supernatant was separated and stored at − 70 °C for measuring oxidative stress biomarkers and antioxidant enzymes. These tests were performed in triplicate for four rats in each group. Total protein levels of homogenized tissues were measured by the Bradford method36.
Oxidative stress and antioxidant biomarkers in hippocampus and cerebellum tissues
Hippocampal and cerebellar MDA was measured using thiobarbituric acid (TBA). The mixture of homogenized tissue and the solution of TBA and trichloracetic acid (TCA) (35 mM TBA and 10% TCA in 125 mM HCl) were incubated in boiling water for 30 min, and then incubated at room temperature for 45 min. This mixture was centrifuged at 3000 rpm for 10 min. The absorbance of the supernatant was measured by a spectrophotometer at 532 nm37.
The hippocampus and cerebellum nitric oxide (NO) were measured using the method described by Gries et al. Briefly, the Gries solution (1% sulfonamide in 2.5% phosphoric acid solution) was mixed with the supernatant and then adsorbed with an ELISA reader at 560 nm38,39.
Hippocampal and cerebellar MPO were assessed by the O-dianisidine method. Briefly, a mixture containing the homogenized tissue supernatant, 0.1 mM phosphate buffer (pH 6.0), 0.01 mM hydrogen peroxide (H2O2), and 0.02 mM O-dianisidine was prepared and then adsorbed at 460 nm with an ELISA reader for 10 min40.
GSH was measured using the method reported by Rahman et al. Briefly, a mixture of hippocampal and cerebellar tissue supernatants along with the sulfhydryl reagent 5,5′-dithio-bis(2-nitrobenzoic acid) (DTNB) was placed in a 96-well plate, and then its absorbance was read with an ELISA reader at 412 nm41.
Hippocampal and cerebellar glutathione peroxidase (GPx) activity was measured according to the DeVore and Greene method. Briefly, the mixture supernatant, 50 mM KH2PO4 buffer, 0.5 mM EDTA, 1 mM GSH, 0.15 mM NADPH, 1.5 U glutathione reductase, 0.15 mM H2O2, and 1 mM NaN3 were incubated at 22 °C, and then the reduction in adsorption was determined by an ELISA reader at 340 nm42.
CAT activity in the hippocampus and cerebellum was measured using the Aebi method. Briefly, the reduced adsorption of the mixture of supernatant, 50 mmol/L potassium phosphate, 30 mmol/L hydrogen peroxide (H2O2) at 240 nm was read by spectrophotometry43.
RNA extraction and real-time quantitative polymerase chain reaction (PCR)
To evaluate the expression of TNF-α, IL-6, AMPK, and Casp-3 genes in different regions of the rat brain such as the hippocampus and cerebellum, total RNA in eight rats in each group was isolated by the TriZol reagent according to the manufacturer’s instructions. Then, RNA samples were treated with DNase to clear genomic DNA impurity. cDNA was generated from pure total RNA using Oligotex-30 and the cDNA synthesis kit (Roche, USA) in accordance with the manufacturer’s instructions. The amplification of cDNAs of each sample with triple repetition was performed using real-time PCR, the SYBR Green I master mix, and specific primers. Each cycle of PCR consists of denaturation, primer annealing, and primer extension. The initial step, i.e., denaturation (pre-incubation) was performed at 95 °C for 5 min in order to activate the DNA polymerase. Then 40 amplification cycles were performed at 95 °C for 10 s, a specific Tm for 20 s, and 72 °C for 10 s. The data were analyzed using Light Cycler 96 Software version 1.144. The primer sequences used for real-time PCR are listed in Table 1.
Histopathological studies
Tissue preparation
Four weeks after BDL, the animals were killed under anesthesia. Six hippocampus and cerebellum from each group were removed and fixed in 10% formalin. Tissue processing was performed with increasing degrees of alcohol for dehydration and clearing was performed with xylene. Then, paraffin was embedded for penetration. After that, paraffin blocks were created and sections with a thickness of 5 μm were prepared.
Nissl staining
Nissl staining was performed to evaluate the morphology of neurons in the CA1 region of the hippocampus and purkinje cells of the cerebellum. Tissue sections were deparaffinized with xylene and after rinsing with alcohol, they were stained with 0.1% cresyl violet solution for 5 min, 100% dehydrated in alcohol, clarified in xylene, and mounted using entellan. Degenerated and intact neurons in three visual fields in each rat were manually counted at 400 × magnifications. Then, the percentage of degenerated neurons in each group was calculated45.
Statistical analysis
The Shapiro–Wilk test showed that the data distribution is normal. If the data were homogeneous, Homogeneity of variance tests was verified using Levene’s test (p > 0.05).Data analysis was performed using one-way analysis of variance (ANOVA) with SPSS software version 16. Post hoc Tukey ̓s test for multiple comparisons was used to evaluate the statistical significance between different groups at P-values < 0.05. If the hypothesis of homogeneity of variances was rejected (Levene’s p < 0.05), the ANOVA Welch and Brown-Forsythe tests with Games-Howell post hoc test were used. Data are expressed as mean ± standard error of the mean (SEM). GraphPad Prism software version 6 was used to draw the diagrams.
Result
The effect of GA on serum ammonia in the BDL model in rats
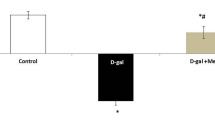
Serum ammonia was significantly higher in the BDL group than in the sham group (p < 0.0001). Serum ammonia levels were significantly lower in the BDL+ 20 mg/kg GA and BDL+ 30 mg/kg GA groups than in the BDL group (p < 0.01). In this study, serum ammonia was significantly higher in the BDL+CC group than in the sham+CC group (p < 0.0001). Serum levels of ammonia were significantly lower in the BDL+ 20 mg/kg GA+CC and BDL+ 30 mg/kg GA+CC groups than in the BDL+CC group (p < 0.05) (Fig. 1).
Effects of GA on serum ammonia in BDL-induced encephalopathy. Data are expressed as mean ± SEM (n = 8 per group). Values of this study were analyzed using one way ANOVA, followed by Tukey’s post hoc test. Significance level of the data was considered p value < 0.05. **** Shows a significant difference between the two groups with p < 0.0001, *** Shows a significant difference between the two groups with p < 0.001, ** Shows a significant difference between the two groups with p < 0.01,* Shows a significant difference between the two groups with p < 0.05.
The effect of GA on hippocampal and cerebellar oxidative stress parameters in the BDL model in rats
In our study, hippocampal and cerebellar MDA, NO, and MPO levels were significantly higher in the BDL group than in the sham group (p < 0.01). Hippocampal and cerebellar MDA was significantly lower in the BDL+ 20 mg/kg GA and BDL+ 30 mg/kg GA groups than in the BDL group (p < 0.0001). Hippocampal and cerebellar MPO levels were significantly reduced in the BDL+ 30 mg/kg GA group compared to the BDL group (p < 0.05). In this study, hippocampal MDA, MPO, and NO, as well as cerebellar MDA and MPO were significantly increased in the BDL+CC group compared to the sham+CC group (p < 0.0001). Hippocampal MDA and NO levels were significantly lower in the BDL+ 20 mg/kg GA+CC and BDL+ 30 mg/kg GA+CC groups than in the BDL+CC group (p < 0.05). Hippocampal MPO activities were significantly decreased in the BDL+ 30 mg/kg GA+CC group compared to the BDL+CC group (p < 0.05). Cerebellar MDA, NO, and MPO were significantly lower in the BDL+ 30 mg/kg GA+CC group than in the BDL+CC group (p < 0.05). Cerebellar MDA and hippocampal NO were significantly higher in the sham+CC group than in the sham group (p < 0.05). Hippocampal and cerebellar MDA were significantly increased in the BDL+CC group compared to the BDL group (p < 0.0001). Hippocampal and cerebellar MDA were significantly higher in the BDL+ 20 mg/kg GA+CC and the BDL+ 30 mg/kg GA+CC groups than in the BDL+ 20 mg/kg GA and the BDL+ 30 mg/kg GA groups, respectively (p < 0.0001) (Tables 2 and 3).
The effect of GA on hippocampal and cerebellar biochemical parameters in the BDL model in rats
Hippocampal and cerebellar GSH levels, GPx activity, and CAT activity were significantly lower in the BDL group than in the sham group (p < 0.0001). Hippocampal GSH levels and CAT activity were significantly higher in the BDL+ 20 mg/kg GA group than in the BDL group (p < 0.05). The administration of GA significantly increased cerebellar GPx and CAT in the BDL+ 20 mg/kg GA and BDL+ 30 mg/kg GA groups compared with the BDL group (p < 0.0001). Hippocampal and cerebellar GSH, GPx, and CAT were significantly lower in the BDL+CC group than in the sham+CC group (p < 0.05). The administration of GA significantly enhanced hippocampal and cerebellar GSH, GPx, and CAT in the BDL+ 20 mg/kg GA+CC and BDL+ 30 mg/kg GA+CC groups compared with the BDL+CC group (p < 0.05). In the present study, cerebellar and hippocampal GSH and GPx and cerebellar CAT were significantly reduced in the sham+CC group compared to the sham group (p < 0.05). Hippocampal GPx and CAT as well as cerebellar GSH and CAT were significantly lower in the BDL+CC group than in the BDL group (p < 0.05). Hippocampal GSH, GPx, and CAT, and cerebellar GPx and CAT were significantly decreased in the BDL+ 30 mg/kg GA+CC group compared to the BDL+ 30 mg/kg GA group (p < 0.05). Cerebellar GSH and CAT, as well as hippocampal GPx and CAT were significantly lower in the BDL+ 20 mg/kg GA+CC group than in the BDL+ 20 mg/kg GA group (p < 0.05) (Tables 2 and 3).
The effect of GA on the expressions of IL-6, TNF-α, AMPK, and Casp-3 genes in hippocampus and cerebellum in the BDL model in rats
The mRNA expressions of IL-6, TNF-α, and Casp-3 in the hippocampus and cerebellum were significantly higher in the BDL group than in the sham group (p < 0.05). On the other, the mRNA expressions of AMPK in the hippocampus and cerebellum were significantly lower in the BDL group than in the sham group (p < 0.05). The mRNA expressions of IL-6, TNF-α, and Casp-3 in the cerebellum and TNF-α and Casp-3 in the hippocampus were significantly reduced in the BDL+ 20 mg/kgGA and BDL+ 30 mg/kg GA groups compared to the BDL group (p < 0.05). In this study, the mRNA expression of AMPK in the hippocampus and cerebellum was significantly higher in the BDL+ 20 mg/kg GA and BDL+ 30 mg/kg GA groups than in the BDL group (p < 0.05). The mRNA expressions of IL-6, TNF-α, and Casp-3 in the hippocampus and cerebellum were significantly higher in the BDL+CC group than in the sham+CC group (p < 0.05). The mRNA expression of AMPK in the hippocampus and cerebellum was significantly lower in the BDL+CC group than in the sham+CC group (p < 0.05). The administration of GA significantly decreased the mRNA expression of cerebellar IL-6, TNF-α, and Casp-3 in the BDL+ 20 mg/kg GA+CC and BDL+ 30 mg/kg GA+CC groups compared with the BDL+CC group (p < 0.05). The administration of GA significantly reduced the mRNA expression of hippocampal TNF-α in the BDL+20 mg/kg GA+CC and BDL+ 30 mg/kg GA+CC groups compared with the BDL+CC group (p < 0.05). The mRNA expression of Casp-3 in the hippocampus was significantly lower in the BDL+ 20 mg/kg GA+CC group than in the BDL+CC group (p < 0.01). However, the administration of GA significantly increased the mRNA expression of cerebellar AMPK in the BDL+ 20 mg/kg GA+CC and BDL+ 30 mg/kg GA+CC groups compared with the BDL+CC group (p < 0.01). The mRNA expressions of cerebellar IL-6 and TNF-α and hippocampal TNF-α were significantly higher in the sham+CC group than in the sham group (p < 0.05). The mRNA expressions of cerebellar TNF-α and Casp-3 were significantly higher in the BDL+CC group than in the BDL group (p < 0.05). The mRNA expressions of cerebellar IL-6 and Casp-3 as well as hippocampal Casp-3 were significantly higher in the BDL+ 20 mg/kg GA+CC group than in the BDL+ 20 mg/kg GA group (p < 0.05). The mRNA expressions of cerebellar IL-6 and hippocampal Casp-3 were significantly higher in the BDL+ 30 mg/kg GA+CC group than in the BDL+ 30 mg/kg GA group (p < 0.01). The administration of CC significantly reduced the mRNA expression of cerebellar AMPK in the BDL+CC group compared with the BDL group (p < 0.05). The administration of CC significantly lowered the mRNA expression of hippocampal and cerebellar AMPK in the BDL+ 20 mg/kg GA+CC group compared with the BDL+ 20 mg/kg GA group (p < 0.001). Moreover, the administration of CC significantly decreased the mRNA expression of hippocampal and cerebellar AMPK in the BDL+ 30 mg/kg GA+CC group compared with the BDL+ 30 mg/kg GA group (p < 0.001) (Fig. 2).


Effects of Gallic acid the hippocampus and cerebellum mRNA expression levels of inflammatory markers such as IL-6 and TNF-α and apoptotic marker such as Caspase-3 and AMPK following BDL-induced encephalopathy. Data are expressed as mean ± SEM (n = 8 per group). Values of this study were analyzed using one way ANOVA, followed by Tukey’s post hoc test Significance level of the data was considered p value < 0.05. **** Shows a significant difference between the two groups with p < 0.0001, *** Shows a significant difference between the two groups with p < 0.001, ** Shows a significant difference between the two groups with p < 0.01,* Shows a significant difference between the two groups with p < 0.05.
The effect of GA on neuronal degeneration in the hippocampal CA1 region and cerebellum in the BDL model in rats
In the hippocampus and cerebellum, neuronal degeneration was significantly higher in the BDL group than in the sham group (p < 0.0001). The administration of GA significantly lowered neuronal degeneration in the hippocampus and cerebellum in the BDL+ 20 mg/kg GA and BDL+ 30 mg/kg GA groups compared with the BDL group (p < 0.05). In our study, neuronal degeneration in the hippocampus and cerebellum was significantly increased in the BDL+CC group than in the sham+CC group (p < 0.0001). The administration of GA significantly decreased neuronal degeneration in the hippocampus and cerebellum in the BDL+ 20 mg/kg GA+CC and BDL+ 30 mg/kg GA+CC groups compared with the BDL+CC group (p < 0.0001). The administration of CC significantly increased neuronal degeneration in the hippocampus and cerebellum in the BDL+ 30 mg/kg GA+CC group compared with the BDL+ 30 mg/kg GA group (p < 0.05) (Figs. 3 and 4).


Effects of Gallic acid on neuronal degeneration in the hippocampus CA1 in BDL-induced encephalopathy. Data are expressed as mean ± SEM (n = 7 per group). Values of this study were analyzed using one way ANOVA, followed by Tukey’s post hoc test Significance level of the data was considered p value < 0.05. **** Shows a significant difference between the two groups with p < 0.0001, *** Shows a significant difference between the two groups with p < 0.001, ** Shows a significant difference between the two groups with p < 0.01,* Shows a significant difference between the two groups with p < 0.05. Photographs show hippocampal pyramidal neurons in different groups. (A) Sham, (B) BDL, (C) BDL+ 20mg/kgGA, (D) BDL+ 30mg/kgGA, (E) sham+CC, (F) BDL+CC, (G) BDL+ 20mg/kgGA+CC, (H) BDL+ 30mg/kgGA+CC. The black arrow Shows degenerate neurons. (Nissl staining, magnification × 400).


Effects of Gallic acid on neuronal degeneration in the cerebellum in BDL-induced encephalopathy. Data are expressed as mean ± SEM (n = 7 per group). Values of this study were analyzed using one way ANOVA, followed by Tukey’s post hoc test Significance level of the data was considered p value < 0.05. **** Shows a significant difference between the two groups with p < 0.0001, *** Shows a significant difference between the two groups with p < 0.001, ** Shows a significant difference between the two groups with p < 0.01,* Shows a significant difference between the two groups with p < 0.05. Photographs show cerebellar purkinje in different groups. (A) Sham, (B) BDL, (C) BDL+ 20mg/kgGA, (D) BDL+ 30mg/kgGA, (E) sham+CC, (F) BDL+CC, (G) BDL+ 20mg/kgGA+CC, (H) BDL+ 30mg/kgGA+CC. The black arrow shows degenerate neurons. (Nissl staining, magnification × 400).
Discussion
In our study, BDL-induced HE increased oxidative stress, inflammatory cytokines, apoptosis, and neurological damage in the hippocampus and cerebellum of male rats. Receiving CC led to significantly higher oxidative stress and tissue damage in these regions. In this study, GA was found to reduce oxidative stress and inflammation in the hippocampus and cerebellum.
In our study, an increase in blood ammonia was observed four weeks after BDL. Hyperammonia in biliary cirrhosis increases oxidative stress and inflammatory cytokines while decreasing antioxidant activity in the brain35,46,47. Oxidative stress may cause neuronal apoptosis, which is a common pathological feature in neurological diseases. In addition, hippocampal synaptic changes cause memory and learning disorders48. Increased blood ammonia leads to inflammation, systemic oxidative stress changes in the morphology of neurons, and neuronal death. It also impairs memory and learning in HE47,49. An increase in factors such as ammonia, oxidative stress, bile acids, inflammatory cytokines, and lactate by disrupting Aquaporin 4 water channels cause an increase in BBB permeability. Which facilitates the entry of neurotoxic substances into the extracellular space of the brain. Neurotoxic substances have harmful effects on astrocytes and cause swelling of astrocytes by disrupting the metabolism of glutamate and glutamine. Astrocyte swelling affects astrocyte-neuron communication, thereby disrupting glutamatergic, and GABAergic neurotransmission systems, which ultimately causes cerebral edema and HE49. Oxidative stress is increased through the presence of intracellular and extracellular ROS in the central nervous system due to HE. Moreover, the antioxidant capacity of this part is reduced and inflammation occurs, which play an important role in disorders of the nervous system50. In the present study, BDL increased oxidative stress in the hippocampus and cerebellum. NO and MDA levels both increased in hippocampal and cerebellar tissues, while the activity of antioxidant enzymes such as GPx, CAT, and GSH was reduced. Elevated expression of inflammatory cytokines such as IL-6 and TNF-α, as well as increased expression of the Casp-3 gene, indicate an increase in inflammation and cellular damage in these tissues. They are also a sign of neuronal degeneration, swelling of astrocytes in the hippocampus and cerebellum, and inflammation of brain tissue. Systemic inflammation caused by the proinflammatory mediators and cytokines, including TNF-α, IL-1β, IL-6, IL-8, and IL-12, contributes to HE exacerbation and worsens neurological damage51.
The signaling system centered on AMPK is relatively complex, and the effects of activating or inhibiting this kinase are extensive52. From a physiological perspective, it controls cellular processes, which involve switching between anabolic and catabolic states and altering cell dynamics with a direct effect on genetic control and protein expression. When the cellular hemostatic apparatus is occupied by pathological forces, AMPK assumes a central position in inhibition, mostly indicating the direction of correction52. Ammonia disrupts energy metabolism by triggering the AMPK/ mammalian target of rapamycin (mTOR)/Unc-51 like autophagy activating kinase 1 (ULK1) pathway during autophagy53. Inflammation, increased inflammatory cytokines, increased serum ammonia, and CC decrease AMPK levels, resulting in neurological damage and cognitive, memory, and anxiety-like behaviors in rats54. It is well documented that CC can cause memory impairment and reduce learning by inhibiting AMPK in Alzheimer’s disease55. In our previous study, dorsomorphin by inhibiting AMPK in the hippocampus causes memory impairment in the BDL-induced encephalopathy model27. Hypoxic-ischemic injury in rat neonatal brain is activated by mTOR signaling, which may lead to cell death, whereas AMPK activation can suppress mTOR signaling. On the other hand, the induction of AMPK activation by sestrin2 can lead to the inhibition of mTOR signaling and reduced apoptosis after injury of hypoxic-ischemic encephalopathy in neonatal rats56. GA is a potential antioxidant that directly regulates the expression of antioxidant genes57. With its antioxidant properties, GA can play a role in nerve health and activation of AMPK58. In our study, the effect of GA on the increase of hippocampus antioxidant enzymes was not observed in the group of 30 mg/kg of GA, and this might have been due to the reduction in the amount of these enzymes in the tissue to reduce the high oxidative stress through free radical scavenging59,60. The anti-inflammatory mechanisms of GA are mainly mediated by the mitogen-activated protein kinase (MAPK) and nuclear factor kappa B (NF-кB) signaling pathways. Thus, by reducing the release of inflammatory cytokines, chemokines, adhesion molecules, and cellular permeation, GA attenuates the inflammatory response. Due to its remarkable drug activity, GA is considered to be a potential candidate for the treatment of various inflammatory diseases61. In the present study, due to GA’s antioxidant, anti-inflammatory, and anti-apoptotic properties, as well as its ability to activate AMPK25,61, it was able to increase the antioxidant capacity of hippocampal and cerebellar tissues and reduce the number of degenerated neurons, and inflammatory cytokines. On the other hand, GA could protect neurons against the deleterious effects of BDL in male rats. GA can prevent cisplatin-induced male reproductive toxicity by inhibiting oxidative stress, reducing DNA damage, reducing the expression of apoptotic genes such as Casp-3, Bax, and Bcl-2, and repairing structural and functional defects in the testes and epididymis62. In another study, GA reduced the expression of inflammatory mediators in liver tissue by activating the AMPK pathway and induced the expression of antioxidant enzymes, thereby reducing inflammation and the apoptosis of liver cells26. In the present study, GA, as an antioxidant with anti-apoptotic properties in hippocampal and cerebellar tissues, reduced the expression of Casp-3 in these tissues, and therefore, decreased tissue damage and dysfunction. The activation of AMPK by metformin significantly increases conduction velocity of the sciatic nerve and decreases the levels of inflammatory cytokines, such as IL-6 and TNF-α. However, concomitant use of CC with metformin reduces the beneficial effects of metformin63. GA by maintaining the integrity of the BBB, reduces brain oxidative stress, anxiety, and depression, helps the survival of neurons during cerebral ischemia/stroke, and increases nerve function over time64. In our study, AMPK expression was decreased in hippocampal and cerebellar tissues following BDL and dorsomorphin uptake. Dorsomorphin induces cell death, apoptosis and significantly induces ROS production. In the previous studies, it has been observed that AMPK inhibition by dorsomorphin accelerates cell death and apoptosis through increasing the level of ROS in p53 cells65,66. In the present study, the amount of oxidative stress, caspase-3 gene expression and nerve damage increased after receiving dorsomorphin, which may be due to the inhibition of AMPK by dorsomorphin. Following an increase in oxidative stress, AMPK increases, which itself acts to reduce stress67. Low concentration of dorsomorphin usually does not inhibit AMPK, while its high concentration inhibits AMPK even in the presence of AMPK activator, and also, dorsomorphin may exert its effects in inhibiting AMPK better when combined with cell damage and depletion of intracellular ATP27. In this study, GA significantly increased AMPK levels in hippocampal and cerebellar tissues in the BDL groups, but in the groups receiving dorsomorphin and GA, compared to the groups receiving GA, the potential effect of GA on AMPK activation decreased. Therefore, it can be concluded that dorsomorphin can partially reduce the healing effect of GA on hippocampal and cerebellar tissues. GA with increasing its antioxidant capacity, reduced oxidative stress caused by increased ammonia in brain tissue and reduced apoptosis and neuronal damage in hippocampal and cerebellar tissue. On the other hand, GA played an important role in reducing neuronal damage by increasing the expression of AMPK and reducing the expression of inflammatory cytokines.
Conclusion
In our study, due to its antioxidant and anti-apoptotic properties and the ability to activate AMPK, GA at the doses of 20 and 30 mg/kg was able to reduce oxidative stress, apoptosis, and inflammation in hippocampal and cerebellar tissues in the BDL-induced model of HE. It could also reduce neuronal damage. However, in the groups receiving CC, the anti-apoptotic and antioxidant properties of GA were reduced by inhibiting AMPK activity. It is recommended that future studies include ultrastructural histological studies, immunohistochemical studies such as the examination of astrocytes and microglia, and the examination of Bax and Bcl-2 gene expression.
Data availability
Upon a reasonable request, the data supporting the results of this article will be made available by the corresponding responsible author.
References
Jia, W. et al. Xiaochaihutang improves the cortical astrocyte edema in thioacetamide-induced rat acute hepatic encephalopathy by activating NRF2 pathway. Front. Pharmacol. 11, 382 (2020).
Ferenci, P. et al. Hepatic encephalopathy—definition, nomenclature, diagnosis, and quantification: Final report of the working party at the 11th World Congresses of Gastroenterology, Vienna, 1998. Hepatology 35(3), 716–721 (2002).
Bustamante, J. et al. Mitochondrial dysfunction as a mediator of hippocampal apoptosis in a model of hepatic encephalopathy. Mol. Cell. Biochem. 354(1), 231–240 (2011).
Jover, R. et al. Brain edema and inflammatory activation in bile duct ligated rats with diet-induced hyperammonemia: A model of hepatic encephalopathy in cirrhosis. Hepatology 43(6), 1257–1266 (2006).
Hajipour, S. et al. Thymoquinone improves behavioral and biochemical deficits in hepatic encephalopathy induced by thioacetamide in rats. Neurosci. Lett. 745, 135617 (2021).
Baraka, S. M. et al. Flavonoids from Barnebydendron riedelii leaf extract mitigate thioacetamide-induced hepatic encephalopathy in rats: The interplay of NF-κB/IL-6 and Nrf2/HO-1 signaling pathways. Bioorg. Chem. 105, 104444 (2020).
Parekh, P. J. & Balart, L. A. Ammonia and its role in the pathogenesis of hepatic encephalopathy. Clin. Liver Dis. 19(3), 529–537 (2015).
El-Baz, F. K., Elgohary, R. & Salama, A. Amelioration of hepatic encephalopathy using Dunaliella salina microalgae in rats: Modulation of Hyperammonemia/TLR4. BioMed Res. Int. 2021(1), 8843218 (2021).
Kwon, K. W. et al. Hepatoprotective effect of sodium hydrosulfide on hepatic encephalopathy in rats. Korean J. Physiol. Pharmacol. 23(4), 263–270 (2019).
Afifi, N. A. et al. Synergistic effect of aminoguanidine and l-carnosine against thioacetamide-induced hepatic encephalopathy in rats: Behavioral, biochemical, and ultrastructural evidence. Can. J. Physiol. Pharmacol. 99(3), 332–347 (2021).
Chen, B. et al. The critical role of hippocampal dopamine in the pathogenesis of hepatic encephalopathy. Physiol. Res. 70(1), 101 (2021).
Braissant, O. et al. Longitudinal neurometabolic changes in the hippocampus of a rat model of chronic hepatic encephalopathy. J. Hepatol. 71(3), 505–515 (2019).
Wu, K.-C. et al. Effect of sirolimus on liver cirrhosis and hepatic encephalopathy of common bile duct-ligated rats. Eur. J. Pharmacol. 824, 133–139 (2018).
Dadsetan, S. et al. Infliximab reduces peripheral inflammation, neuroinflammation, and extracellular GABA in the cerebellum and improves learning and motor coordination in rats with hepatic encephalopathy. J. Neuroinflammation 13, 1–14 (2016).
Jäger, S., Handschin, C., Pierre, J. S. & Spiegelman, B. M. AMP-activated protein kinase (AMPK) action in skeletal muscle via direct phosphorylation of PGC-1α. Proc. Natl. Acad. Sci. 104(29), 12017–12022 (2007).
Ramamurthy, S. & Ronnett, G. V. Developing a head for energy sensing: AMP-activated protein kinase as a multifunctional metabolic sensor in the brain. J. Physiol. 574(1), 85–93 (2006).
Salt, I. P., Johnson, G., Ashcroft, S. J. & Hardie, D. G. AMP-activated protein kinase is activated by low glucose in cell lines derived from pancreatic β cells, and may regulate insulin release. Biochem. J. 335(3), 533–539 (1998).
Liu, Y.-J. & Chern, Y. AMPK-mediated regulation of neuronal metabolism and function in brain diseases. J. Neurogenet. 29(2–3), 50–58 (2015).
Schousboe, A., Waagepetersen, H. S., Leke, R. & Bak, L. K. Effects of hyperammonemia on brain energy metabolism: Controversial findings in vivo and in vitro. Metab. Brain Dis. 29(4), 913–917 (2014).
Vucicevic, L. et al. Compound C induces protective autophagy in cancer cells through AMPK inhibition-independent blockade of Akt/mTOR pathway. Autophagy 7(1), 40–50 (2011).
Huang, D.-W., Chang, W.-C., Wu, J.S.-B., Shih, R.-W. & Shen, S.-C. Gallic acid ameliorates hyperglycemia and improves hepatic carbohydrate metabolism in rats fed a high-fructose diet. Nutr. Res. 36(2), 150–160 (2016).
John, J. A. & Shahidi, F. Phenolic compounds and antioxidant activity of Brazil nut (Bertholletia excelsa). J. Funct. Foods 2(3), 196–209 (2010).
Abdelwahed, A. et al. Study of antimutagenic and antioxidant activities of Gallic acid and 1, 2, 3, 4, 6-pentagalloylglucose from Pistacia lentiscus: Confirmation by microarray expression profiling. Chem. Biol. Interact. 165(1), 1–13 (2007).
BenSaad, L. A., Kim, K. H., Quah, C. C., Kim, W. R. & Shahimi, M. Anti-inflammatory potential of ellagic acid, gallic acid and punicalagin A&B isolated from Punica granatum. BMC Complement. Altern. Med. 17(1), 1–10 (2017).
Doan, K. V. et al. Gallic acid regulates body weight and glucose homeostasis through AMPK activation. Endocrinology 156(1), 157–168 (2015).
Tanaka, M. et al. Gallic acid inhibits lipid accumulation via AMPK pathway and suppresses apoptosis and macrophage-mediated inflammation in hepatocytes. Nutrients 12(5), 1479 (2020).
Jafaripour, L. et al. The effect of gallic acid on memory and anxiety-like behaviors in rats with bile duct ligation-induced hepatic encephalopathy: Role of AMPK pathway. Avicenna J. Phytomed. 12(4), 425 (2022).
Kilkenny, C., Browne, W. J., Cuthill, I. C., Emerson, M. & Altman, D. G. The ARRIVE guidelines animal research: Reporting in vivo experiments. PLoS Biol. 8(6), e1000412 (2010).
Alimohamadi, Y. & Sepandi, M. Sample size in animal studies (The number of laboratory animals in a Research study). Iran. J. Med. Microbiol. 16(2), 173–176 (2022).
Sen, A. et al. Effects of Myrtus communis extract treatment in bile duct ligated rats. J. Surg. Res. 205(2), 359–367 (2016).
Gandhi, G. R. et al. Gallic acid attenuates high-fat diet fed-streptozotocin-induced insulin resistance via partial agonism of PPARγ in experimental type 2 diabetic rats and enhances glucose uptake through translocation and activation of GLUT4 in PI3K/p-Akt signaling pathway. Eur. J. Pharmacol. 745, 201–216 (2014).
Kim, M. J. et al. Gallic acid, a histone acetyltransferase inhibitor, suppresses β-amyloid neurotoxicity by inhibiting microglial-mediated neuroinflammation. Mol. Nutr. Food Res. 55(12), 1798–1808 (2011).
Park, S., Kim, D. S., Kang, S. & Shin, B. K. Chronic activation of central AMPK attenuates glucose-stimulated insulin secretion and exacerbates hepatic insulin resistance in diabetic rats. Brain Res. Bull. 108, 18–26 (2014).
Zarei, M., Mohammadi, S., Jabbari, S. & Shahidi, S. Intracerebroventricular microinjection of kaempferol on memory retention of passive avoidance learning in rats: Involvement of cholinergic mechanism (s). Int. J. Neurosci. 129(12), 1203–1212 (2019).
Dhanda, S., Gupta, S., Halder, A., Sunkaria, A. & Sandhir, R. Systemic inflammation without gliosis mediates cognitive deficits through impaired BDNF expression in bile duct ligation model of hepatic encephalopathy. Brain Behav. Immun. 70, 214–232 (2018).
Kruger, N. The Bradford method for protein quantitation (Humana Press, Totowa, 1994).
Ahmadvand, H., Bagheri, S., Khosrobeigi, A. & Boshtam, M. Effects of olive leaves extract on LDL oxidation induced-CUSO 4 in vitro. Pak. J. Pharm. Sci. 25(3), 571–575 (2012).
Gries, A. et al. Inhaled nitric oxide inhibits human platelet aggregation, P-selectin expression, and fibrinogen binding in vitro and in vivo. Circulation 97(15), 1481–1487 (1998).
Dadpisheh, S., Ahmadvand, H., Jafaripour, L., Shati, H. & Bagheri, S. Effect of troxerutin on oxidative stress induced by sciatic nerve ischemia-reperfusion injury in rats. J. Kerman Univ. Med. Sci. 27, 338–347 (2020).
Khalatbary, A. R. & Ahmadvand, H. Effect of oleuropein on tissue myeloperoxidase activity in experimental spinal cord trauma. Iran. Biomed. J. 15(4), 164 (2011).
Rahman, I., Kode, A. & Biswas, S. K. Assay for quantitative determination of glutathione and glutathione disulfide levels using enzymatic recycling method. Nat. Protoc. 1(6), 3159–3165 (2006).
DeVore, V. & Greene, B. Glutathione peroxidase in post-rigor bovine Semitendinosus muscle. J. Food Sci. 47(5), 1406–1409 (1982).
Aebi, H. Catalase in vitro. Methods Enzymol 105, 121–126 (1984).
Pfaffl, M. W., Horgan, G. W. & Dempfle, L. Relative expression software tool (REST©) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Res. 30(9), e36 (2002).
Golshani, M., Basiri, M., Shabani, M., Aghaei, I. & Asadi-Shekaari, M. Effects of erythropoietin on bile duct ligation-induced neuro-inflammation in male rats. AIMS Neurosci. 6(2), 43 (2019).
Dhanda, S., Sunkaria, A., Halder, A. & Sandhir, R. Mitochondrial dysfunctions contribute to energy deficits in rodent model of hepatic encephalopathy. Metab. Brain Dis. 33(1), 209–223 (2018).
Cheon, S. Y., Jo, D., Kim, Y.-K. & Song, J. Long noncoding RNAs regulate hyperammonemia-induced neuronal damage in hepatic encephalopathy. Oxid. Med. Cell. Longev. 1, 7628522 (2022).
Hu, T. et al. Quercetin ameliorates diabetic encephalopathy through SIRT1/ER stress pathway in db/db mice. Aging (Albany NY) 12(8), 7015 (2020).
Ochoa-Sanchez, R. & Rose, C. F. Pathogenesis of hepatic encephalopathy in chronic liver disease. J. Clin. Exp. Hepatol. 8(3), 262–271 (2018).
Pierzchala, K. et al. Central nervous system and systemic oxidative stress interplay with inflammation in a bile duct ligation rat model of type C hepatic encephalopathy. Free Radical Biol. Med. 178, 295–307 (2022).
Lu, B. et al. Babao Dan improves neurocognitive function by inhibiting inflammation in clinical minimal hepatic encephalopathy. Biomed. Pharmacother. 135, 111084 (2021).
Shirwany, N. A. & Zou, M.-H. AMPK in cardiovascular health and disease. Acta Pharmacol. Sin. 31(9), 1075–1084 (2010).
Li Z, Miao Z, Ding L, Bao J, Teng X. Ammonia-induced energy metabolism disorder and autophagy via AMPK/mTOR/ULK1 pathway in chicken livers (2020).
Wang, D. et al. Stimulation of Wnt/β-catenin signaling to improve bone development by naringin via interacting with AMPK and Akt. Cell. Physiol. Biochem. 36(4), 1563–1576 (2015).
Rashtiani, S., Goudarzi, I., Jafari, A. & Rohampour, K. Adenosine monophosphate activated protein kinase (AMPK) is essential for the memory improving effect of adiponectin. Neurosci. Lett. 749, 135721 (2021).
Shi, X. et al. Sestrin2, as a negative feedback regulator of mTOR, provides neuroprotection by activation AMPK phosphorylation in neonatal hypoxic-ischemic encephalopathy in rat pups. J. Cereb. Blood Flow Metab. 37(4), 1447–1460 (2017).
Tanaka, Y., Obinata, H., Konishi, A., Yamagiwa, N. & Tsuneoka, M. Production of ROS by gallic acid activates KDM2A to reduce rRNA transcription. Cells 9(10), 2266 (2020).
Diaz, A. et al. Gallic acid improves recognition memory and decreases oxidative-inflammatory damage in the rat hippocampus with metabolic syndrome. Synapse 75(2), e22186 (2021).
Reshma, K., Ashalatha, V. R., Dinesh, M. & Vasudevan, D. Effect of ocimum flavonoids as a radioprotector on the erythrocyte antioxidants in oral cancer. Indian J. Clin. Biochem. 20, 160–164 (2005).
Rashidi, M. et al. Effects of vitamin E and zinc supplementation on antioxidants in beta thalassemia major patients. Iran. J. Pediatr. 21(1), 8 (2011).
Bai, J. et al. Gallic acid: Pharmacological activities and molecular mechanisms involved in inflammation-related diseases. Biomed. Pharmacother. 133, 110985 (2021).
Altındağ, F. & Meydan, İ. Evaluation of protective effects of gallic acid on cisplatin-induced testicular and epididymal damage. Andrologia 53(10), e14189 (2021).
Hasanvand, A. et al. Anti-inflammatory effect of AMPK signaling pathway in rat model of diabetic neuropathy. Inflammopharmacology 24(5), 207–219 (2016).
MirshekariJahangiri, H., Sarkaki, A., Farbood, Y., Dianat, M. & Goudarzi, G. Gallic acid affects blood-brain barrier permeability, behaviors, hippocampus local EEG, and brain oxidative stress in ischemic rats exposed to dusty particulate matter. Environ. Sci. Pollut. Res. 27(5), 5281–5292 (2020).
Ghanaatgar-Kasbi, S. et al. AMP-kinase inhibitor dorsomorphin reduces the proliferation and migration behavior of colorectal cancer cells by targeting the AKT/mTOR pathway. IUBMB Life 71(12), 1929–1936 (2019).
Le, D. D. T. et al. Inhibitory role of AMP-activated protein kinase in necroptosis of HCT116 colon cancer cells with p53 null mutation under nutrient starvation. Int. J. Oncol. 54(2), 702–712 (2019).
Kosuru, R. et al. AMPK contributes to cardioprotective effects of pterostilbene against myocardial ischemia-reperfusion injury in diabetic rats by suppressing cardiac oxidative stress and apoptosis. Cell. Physiol. Biochem. 46(4), 1381–1397 (2018).
Acknowledgements
The authors greatly appreciate the cooperation of the laboratory staff, Kerman Neuroscience Research Center and Lorestan University of Medical Sciences to conduct this research.
Funding
The Neuroscience Research Center of Kerman University of Medical Sciences, Iran, provided financial support for this project.
Author information
Authors and Affiliations
Contributions
Leila Jafaripour, Esmaeel Babaeenezhad, and Marzieh Maneshian participated in data collection and the draft of this manuscript. Khadijeh Esmaeilpour and Hamideh Bashiri performed data analysis and interpretation. Leila Jafaripour, Majid Asadi-Shekaari, Hassan Ahmadvand, and Esmaeel Babaeenezhad provided the conception and design of the study, wrote the article, and revised the article. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that there is no conflict of interest.
Ethical approval
All the stages of working with animals were approved by the Ethics Committee of Kerman University of Medical Sciences, Iran (approval number: IR.KMU.REC.1399.470).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jafaripour, L., Ahmadvand, H., Babaeenezhad, E. et al. Gallic acid alleviates hippocampus and cerebellum injuries in a rat model of hepatic encephalopathy. Sci Rep 15, 31080 (2025). https://doi.org/10.1038/s41598-025-15524-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-15524-5