Abstract
Unio pictorum (L. 1758) and Unio tumidus (Philipsson, 1788) are common bivalve molluscs from the Unionidae family, with significant ecological importance in aquatic ecosystems. Their shells are essential for species identification and can also be used to assess changes in population structure, individual growth, and body form under varying environmental conditions. The aim of this study was to: (i) compare the individual size and shape of shells, (ii) assess length growth, and (iii) analyse population structures (based on length and age) of the bivalve molluscs Unio pictorum and Unio tumidus between specimens from the early medieval period (EMS) and those currently found (MS) in the Oder River near Szczecin, Poland. EMS shells were collected from archaeological excavations in cultural layers dated to the 10th and 11th centuries. They were located at three sites in Szczecin, approximately 500–700 m from the Oder River. MS specimens were collected using a bottom dredge between 12 and 21 May 2024 from sites within 1,000 m of the excavation area. For both groups, measurements included size, age, growth (using the von Bertalanffy model), and morphometric characteristics (length [L], height [H], width [W]), as well as the Elongation Index and Convexity Index. The shells of Unio pictorum and U. tumidus from the modern sample (MS) exhibited statistically significantly greater length, width, and height compared to those from the early medieval sample (EMS). At the same time, lower values of the Convexity Index and, in U. pictorum, the Elongation Index were recorded in the MS group. The length and age structure of both species in the MS group was dominated by larger and older individuals relative to the EMS group. The growth of contemporary Unio pictorum and U. tumidus can be considered moderate compared to other present-day populations, with maximum predicted ages of eight and nine years, respectively, and asymptotic lengths (L∞) of 93.85 mm and 87.03 mm. In contrast, in the EMS group, the maximum predicted age and asymptotic length were 10 years and 70.12 mm for U. pictorum, and 6 years and 96.25 mm for U. tumidus, respectively. Higher values of the φ′ and P growth indices in the MS group indicate that both species are currently growing at a faster rate than their early medieval counterparts (EMS). The observed differences in individual and population characteristics of Unio pictorum and U. tumidus between present-day specimens (MS) and those from the early Middle Ages (EMS) are attributed to long-term changes in temperature, nutrient availability, and water regime over the past 1,000 years.
Similar content being viewed by others
Introduction
Fifty-five species of bivalve molluscs have been recorded in the inland waters of Europe1of which 16 belong to the Unionidae family2. Unionidae represent a major group of freshwater bivalves, accounting for approximately two-thirds of all freshwater bivalve species3. Among their representatives are Unio pictorum (L., 1758) and U. tumidus (Philipsson, 1788), both commonly found in European waters4. Unio pictorum is distributed from western Europe (including Spain and Portugal) to the Urals and the Caucasus, with its southern range extending into Romania and Bulgaria4. In contrast, U. tumidus has a more limited range, occurring in European waters from France to Russia4. Both species are common in standing and flowing waters4,5typically found near shores where they form large shoals alongside Anodonta anatina6. In such aggregations, the wet biomass of bivalves can exceed 11 kg per m², with densities reaching up to 80 individuals per m²7. These species prefer sandy or muddy substrates, avoiding gravel and soft silt, and are frequently found among macrophytes8.
Unio tumidus and U. pictorum are typically filter feeders9. This feeding strategy contributes to water purification by removing suspended solids from the water column and reducing the abundance of microorganisms10which is crucial for the functioning of aquatic ecosystems11. According to 6, the filtration rate of Unio pictorum ranges from 3.2 to 4.6 L·h⁻¹ per individual, while for U. tumidus it ranges from 2.1 to 2.4 L·h⁻¹. Furthermore,12 note that selective filtration of planktonic organisms by bivalves can lead to restructuring of planktonic communities. In ecosystems, bivalves also serve as hosts for parasites13,14. Conversely, their larval stage (glochidia) is parasitic on various fish species15. Additionally, these bivalves are used by ostracophilic fish, such as Rhodeus sericeus roseus, as sites for egg deposition4. According to4U. tumidus is highly resistant to water pollution, including heavy metals, and can even survive in the vicinity of sewage discharge sites.
Shells serve as a protective element for the bivalve body16 and play a significant habitat-forming role in aquatic environments16,17,18. They also provide attachment surfaces for numerous epizootic organisms19. Shells are a key feature in the identification of individual bivalve species. For instance, Unio tumidus is distinguished by its broadly rounded anterior and tapering posterior shell, while U. pictorum displays a much more elongated shell shape4. Additionally, Unio tumidus typically has a greater shell height at the same length compared to U. pictorum20. However, shell morphology in bivalves may be influenced by various factors, including habitat conditions, sex, parasitic infections, and genetic variation20,21.
Bivalves are estimated to have first appeared during the Cambrian period, approximately 541 million years ago22with historical records of their presence dating back to antiquity17. For over 6,000 years, these organisms have served as a source of food and material for ornamental objects among communities living near water bodies17. Their shells have been found in refuse pits of early medieval settlements situated near water, where inhabitants discarded food waste23,24. However, their remains were never predominant at these sites, as fish consistently formed the core of the human diet17,25. Despite their limited dietary role, bivalve shells from archaeological contexts provide valuable material for study, offering insights into species biology and local environmental conditions26. These remains allow for the analysis of long-term changes in bivalve morphology23,27as well as more abrupt changes over shorter time spans28,29. Numerous archaeological sites across Europe have yielded such findings, particularly in Central Europe23,30. Among the best-known sites are excavations in Wolin on the Szczecin Lagoon23as well as those in Szczecin, where remains of aquatic animals fished from the Oder River during the early medieval period have also been uncovered31. Compared to the Szczecin Lagoon, the Oder River has a higher water flow rate (approximately 570–580 m³·s⁻¹)32,33 and contains elevated levels of nutrients and biogens that support high mussel density and influence population structure32,33.
The bivalves Unio pictorum and U. tumidus recovered from early medieval excavations along the Oder River have not previously been the subject of zoological research. Their discovery enabled an analysis of changes in selected biological and population traits over the past millennium. The aim was to: (i) compare individual size and shell shape, (ii) assess length growth, and (iii) examine population structures (in terms of length and age) of Unio pictorum and U. tumidus between specimens from the early medieval period (EMS) and those found today (MS) in the Oder River near Szczecin.
Results
Shell morphology
Table 1 presents the main characteristics describing the linear dimensions and weight of both bivalve species from each period (EMS and MS groups). In both Unio pictorum and U. tumidus, modern individuals were characterized by statistically significantly greater shell length, width, and height, as well as higher individual shell weight (Mann–Whitney U test, p < 0.0001).
The Elongation Index and Convexity Index values for both bivalve species are presented in Table 2. The results indicate that, for both species, individuals from the EMS group had more convex shells, as reflected by higher Convexity Index values. Additionally, in U. pictorum, a higher Elongation Index was recorded for individuals from the early medieval period, suggesting that their shells were more elongated compared to those from the MS group.
The regression equations for the linear parameters of Unio pictorum and U. tumidus shells in the EMS and MS subgroups are presented in Table 3. The high regression coefficient values (r₁), ranging from 0.877 to 0.992, along with p-values < 0.00001, indicate that all models were statistically significant. The shell growth of both Unio pictorum and U. tumidus from both periods can be classified as allometric.
Length structure of the mussel population
Among U. pictorum from the EMS group consisting of 153 specimens, the 40.0–50.0 mm length class was the most common, representing 58.5% of individuals. In contrast, in the MS group containing 162 specimens, the 50.0–60.0 mm and 60.0–70.0 mm classes dominated, together accounting for 97.5% of the specimens. For U. tumidus, the EMS group of 182 specimens was primarily composed of individuals in the 30.0–40.0 mm and 40.0–50.0 mm length classes, which together made up 76.0% of the sample. In the MS group comprising 175 specimens, individuals in the 50.0–60.0 mm and 60.0–70.0 mm classes predominated, comprising 73.8% of the total (Fig. 1). Overall, larger specimens of both Unio pictorum and U. tumidus were recorded in the modern populations compared to those from the early medieval period.
Distribution of length (L) of Unio pictorum (A) and U. tumidus (B) shells from the Early Middle Ages (EMS) and present day (MS).
Age and growth rate
In terms of the age structure of U. pictorum, the highest abundance of mussels in the EMS group was found at age 5+, which accounted for 49.0%, while in the MS group they were at age 6+ (53.7%). For U. tumidus, the EMS group was dominated by individuals aged 3+ (55.5%), while the MS group was dominated by individuals aged 4+ (36.0%) (Fig. 2).
Distribution of age of Unio pictorum (A) and U. tumidus (B) shells from the Early Middle Ages (EMS) and present day (MS).
The growth of bivalve molluscs from the EMS and MS groups for Unio pictorum (A) and U. tumidus (B), based on the von Bertalanffy equation, is illustrated in Fig. 3, with the equation parameters provided in Table 4. The positioning of the growth curves indicates that individuals from the MS group of both species reached greater lengths at corresponding ages compared to those from the EMS group. Additionally, the higher values of the φ′ and P indices in the MS group, as shown in Table 5, suggest that the modern populations (MS) of both species exhibit faster growth rates than their early medieval counterparts (EMS).
Comparison of growth rates of the bivalves Unio pictorum (a) and U. tumidus (b) from the early Middle Ages (EMS) and present day (MS).
Discussion
Unio pictorum and U. tumidus, found in European waters, are characterized by a maximum shell length of up to 140 mm for both species1. In Polish waters, the shell length of Unio tumidus typically ranges from 60 to 100 mm (with a maximum of 130 mm), while for U. pictorum it ranges from 70 to 100 mm (with a maximum of 138 mm)4. The measurements obtained for the populations of these species in the Oder River fall within these reported ranges. However, in early medieval specimens, both the mean shell dimensions (length, width, and height) and their ranges were smaller compared to modern individuals. Our findings, showing that contemporary specimens are larger than those from the past, are consistent with the results of24who compared modern bivalves with specimens dated to around 850 BC. The authors suggest that archaeological samples containing many small shells indicate successful recruitment, whereas no evidence of recruitment was found among the large individuals in modern samples35. In the case of the EMS and MS specimens analysed in this study, reproduction and recruitment of Unio pictorum and U. tumidus currently inhabiting the Oder River appear to be sufficient, as indicated by the high densities of these bivalves5.
Miller et al.36and Peacock37 also reported larger sizes in modern bivalves compared to the same species found in Holocene archaeological sites. These differences are likely the result of long-term environmental changes, including increasing eutrophication—now accelerated by human activities35,38 — as well as temperature fluctuations39. Currently, elevated water temperatures and high levels of nutrients and food availability in the Oder River40 may be contributing to the faster growth (see Fig. 3) and larger body sizes (Table 1; Fig. 1) observed in present-day bivalves. Conversely, the smaller sizes of Unio pictorum and U. tumidus from the early medieval period may be influenced by differential preservation, differential archaeological recovery methods, or both41. Generally, smaller, denser, and thicker mussel shells tend to preserve better than thinner ones, which are more prone to damage42. Furthermore, since modern shells were collected by hand rather than by sieving sediments, larger specimens may have been more easily detected and collected43.
The bivalves shell morphology is an important taxonomic feature. In the nineteenth century, approximately 4,000 species were described based on shell morphology alone44though in recent years the number of recognized species has been revised45. However, the high morphological plasticity of shells remains a significant challenge, as it often leads to taxonomic misidentification46,47. Numerous studies have highlighted the influence of long-term climate change on bivalve shell size and form39,48. According to49water temperature and productivity are the most important environmental factors shaping shell morphology. In addition50, emphasize the role of river discharge, turbulence, and water depth, as also noted by51 and52. It is likely that changes in environmental conditions in the Oder River over the past millennium53 have influenced the shell morphology of Unio pictorum and U. tumidus. This is particularly plausible given that, as54 observed, shell morphology is phenotypically plastic and primarily shaped by environmental conditions. Consequently, individuals from modern populations in the Oder River—where trophic and food resources are currently abundant40—had longer, wider, and higher shells than those from the early medieval period. They also exhibited lower Convexity Index values, and, in the case of U. pictorum, a higher Elongation Index compared to early medieval specimens, which likely inhabited less fertile environments53. Nonetheless, it should be acknowledged that phenotypic plasticity in response to environmental variation also has a genetic component55 and should not be viewed merely as environmental noise obscuring the relationship between heritability and phenotype56.
It is generally accepted that individual bivalve growth follows the von Bertalanffy growth curve23,57,58. Bivalves continue to grow throughout their lives, and in temperate climates, a period of winter growth arrest—when water temperatures fall below approximately 12 °C59—leads to the formation of an annual growth ring, visible as a dark band on the shell. However, as noted by60ring formation may also be triggered by environmental disturbances. Despite this, bivalve growth patterns offer valuable insights into their living conditions and historical population dynamics61. The lengths achieved by individuals in different age groups reflect environmental factors such as water quality and food availability62,63which explains the considerable variation in growth observed for Unio pictorum and U. tumidus (Table 6).
The lengths attained in individual years of life by Unio pictorum and U. tumidus from the Oder River are consistent with data reported for other populations of these species (Table 6), and the von Bertalanffy growth parameters calculated in this study align with those from other research sites (Table 7). Generally, both species exhibit their highest growth rates in the first year of life, ranging from 15 to 23 mm for Unio pictorum and from 13 to 22 mm for U. tumidus. In the subsequent two years, growth rates average around 10 mm for both species and gradually decline thereafter. Based on annual growth rates, individuals of both species currently inhabiting the Oder River can be classified as moderate-growing bivalves, similar to populations in Wicken Lode, England65and the Szczecin Lagoon23. The calculated growth coefficients for Unio pictorum (K = 0.156–0.170) and U. tumidus (K = 0.129–0.172) are also within the average range for these species and fall within the broader ranges reported for European populations: K = 0.121–0.245 for Unio pictorum and K = 0.075–0.286 for U. tumidus28,65.
However, clear differences in shell length growth were observed between the early medieval (EMS) and modern (MS) specimens of bivalves from the Oder River, particularly in U. pictorum. This is supported by the higher values of the growth indices φ′ and P in the MS group (Table 5). These differences are likely attributable to changes in environmental conditions in the river, especially water temperature and fertility, both of which significantly influence bivalve growth62. According to67the period between 950 and 1250 AD, known as the Medieval Climate Anomaly (MCI), was characterized by relatively warm conditions in Europe. Nevertheless, water temperatures in the Oder River during that time were likely slightly lower than present-day levels40. Additionally, a study by53 indicates that water fertility (i.e., productivity) in the Oder River was considerably lower in the early medieval period. These conditions likely contributed to the differences in growth observed between the two time periods. As noted by62freshwater mussels generally grow more slowly and live longer at higher latitudes, where lower air temperatures prevail. Reduced water temperatures limit bivalve feeding rates68; in winter, although filtration continues, food particles are often rejected as inedible seston69. This can lead not only to growth stagnation but also to increased mortality during colder periods70. In contrast, higher water temperatures promote phytoplankton development—a primary food source for bivalves—thus enhancing their growth rates71,72,73.
An important factor influencing the growth of filter-feeding organisms, in addition to food availability, is the nutrient content of the water63. According to data presented by53the process of eutrophication in the Oder River began in the 8th century AD, coinciding with the destruction of oak and beech forests in the area of present-day Szczecin. Initially slow, eutrophication accelerated significantly during the post-industrial era, and today the Oder is considered a highly eutrophicated river with elevated nutrient levels40. This increase in nutrient content may enhance food availability for filter-feeding bivalves, enabling them to attain larger body sizes60.
In addition, differences in bivalve growth may also result from long-term changes in the hydrological regime24. According to74the period during which the EMS mussel groups originated was marked by frequent floods and river surges in Poland, leading to increased water flow. Haag and Rypel62 suggest that such increases in flow may be negatively correlated with mussel growth. In contrast, under present-day conditions of reduced flow and lower water levels, a positive correlation between flow and growth has been observed, as slower currents can enhance food availability for filter feeders63,68. However, higher energy demands for survival under high-flow conditions may counteract growth75. Additionally, the instability of flow regimes during the large floods of the early medieval period74,76 may have further limited bivalve growth compared to the more stable, low-flow conditions of the 21st century77. A positive relationship between bivalve growth and flow stability was also demonstrated by78who found that greater stability in water flow was associated with increased bivalve size.
The production of these mussels is affected by various factors, including water pollution, habitat degradation, a decline in the number of host fish and the presence of invasive species79,80. Water pollution significantly reduces the production of filter-feeding mussels and limits their presence81. Unfortunately, the section of the Oder River under study is located in a heavily industrialised area (cities of Szczecin and Police). Industrial activity significantly impacts water quality, causing major increases in pollution and periodic decreases in oxygen content40. Decreases in oxygen content82 and increases in pollution83 lead to reduced mussel production and high mortality rates, particularly among juveniles. The regulatory changes made to the river since the 19th century, including the concrete reinforcement of riverbanks, the removal of aquatic vegetation, and dredging, are important factors limiting the production of these organisms in the Oder River and leading to the degradation of their habitats84. These works have been most intensive in the past 50 years, resulting in a decrease in macrozoobenthos species diversity, as well as changes to the resources and structure of ichthyofauna85,86. Ichthyofauna, especially benthic species such as bream, roach and tench, can reduce mussel production by feeding on them87. However, fish also promote the movement of mussel larvae (in the glochidium stage) by carrying them, thus facilitating the colonisation of new water bodies88.
Competition with other bivalves, particularly species capable of colonising a variety of underwater structures – such as the zebra mussel Dreissena polymorpha – plays a significant role in shaping Unionidae biomass89. Large concentrations of this invasive species are currently present in the lower Oder River, including the Szczecin Lagoon. Despite the pollution of the Oder by a chemical plant in the town of Police, D. polymorpha populations have been recorded at levels reaching approximately 91,000 tonnes90.
Thus, increased temperatures, higher water fertility, and other consequences of climate change – including declining water levels and reduced flow – may be key factors influencing bivalve growth over the long term39,48. These factors also appear to have played a major role in shaping the evolution of individual size, shell morphology, growth patterns, and population structure of Unio pictorum and U. tumidus in the Oder River over the past millennium.
Methods
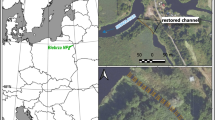
The study material consisted of shells of the bivalves Unio pictorum and U. tumidus, obtained from archaeological excavations (early medieval EMS group) and modern specimens harvested during fishing with a bottom dredge in the Oder River in 2024 (MS group). A total of 315 specimens of Unio pictorum (153 medieval and 162 modern) and 355 specimens of U. tumidus (181 medieval and 174 modern) were analysed. The EMS group comprised shells recovered from archaeological excavations at three sites: (1) Szczecin Mścięcino (located at an unspecified distance from the Oder River), (2) Szczecin Rynek Warzywny, and (3) Szczecin Zamek Książąt Pomorskich (both situated approximately 500 m from the Oder River) (Fig. 4). The shells were obtained from cultural layers dated to the 10th and 11th centuries31. Shells from the MS group were collected from bivalves caught between 12 and 21 May 2024 using a bottom dredge at sites in the Oder River within 1,000 m of the archaeological site.
Location of early medieval excavation sites (1 - Szczecin Mścięcino, 2 - Szczecin Zamek, 3 - Szczecin Rynek Warzywny) and 4 - fishing sites of the currently occurring mussels Unio pictorum and U. tumidus (Szczecin River Odra).
Methods
The collected bivalves were identified to species level based on the criteria provided by4. Morphometric measurements were taken following the scheme proposed by65as modified by23using a NOWA 2000 (China) electronic calliper (± 0.01 mm) connected to a computer. The measurements included maximum shell length (L), maximum shell height (H), and maximum shell width (W) (Fig. 5;23 ).
An analysis of the relationships H ~ L, W ~ L and W ~ H was conducted following65 and the type of growth for H and W was assessed23. Based onShingleton (2010), the relationship is called isometric when b = 1, positively allometric when b > 1, and negatively allometric when b < 1.
Shells were weighed using an Axis electronic scale (Radwag, Poland) with a precision of 0.01 g. The age of the bivalves was estimated by counting the growth rings visible on the shell surface after cutting the shells into thin sections.93. Growth rates were determined using the von Bertalanffy growth function57,58 as shown in Eq. (1):
where Lt is length (mm) at time t (age in years), L∞ is length (mm) at time infinity (the predicted mean maximum length for the population), K is a growth constant which describes the rate at which L∞ is attained (mm, year[- [1) t is age (years) and to is the time at which length = 0.
The parameters of the von Bertalanffy equation were calculated in the R programming environment in the FSA, nlstools, magrittr, dplyr packages94.
Following34the maximum age that the studied bivalve species can reach in this water body was calculated according to Eq. (2):
where Amax is the maximum age and Lm is the maximum length.
The phi-prime index (φ′ -Eq. (3)) and overall growth performance (index P – Eq. (4)) were also calculated95:
These indices are now widely used to assess the health of bivalves96,97.
In addition, Elongation index (Eq. (5)) and Convexity index (Eq. (6) were calculated according to98,99:
where: L - individual shell length, H- individual shell height.
where: W - individual shell width, H - individual shell height.
Statistical analyses
The Shapiro–Wilk and Levene tests were used to assess the normality of distribution and homogeneity of variance, respectively, while the Breusch–Pagan test was applied to check for heteroscedasticity100. Comparisons between clams from the early medieval (EMS) and modern (MS) samples were conducted using the Mann–Whitney U test (also known as the Wilcoxon rank-sum test)100. A p-value ≤ 0.05 was considered statistically significant. If this threshold was met, the null hypothesis of no differences between EMS and MS samples was rejected in favour of the alternative hypothesis indicating statistically significant differences.
In addition, following the methodology of101regression analysis of the linear parameters was conducted using the SMATR package in the R programming language. This package was also used to determine the type of growth by comparing the calculated b value to the reference value of 1, as proposed by92. A p-value < 0.05 was considered statistically significant, allowing the rejection of the null hypothesis that there was no difference between the calculated b value and 1. All statistical analyses were performed using R software (version 4.4.1;102 ) along with the required packages.
Data availability
Dataset available upon request from corresponding author.
References
Collas, F. P. L., Buijse, A. D., Hendriks, J. A., van der Velde, G. & Leuven, R. S. E. W. Sensitivity of native and alien freshwater bivalve species in Europe to climate-related environmental factors. Ecosphere 9, e02184 (2018).
Araujo, R. & de Jong, Y. Fauna europaea: Mollusca - Bivalvia. Biodivers Data J 3, (2015).
Lopes-Lima, M. et al. Phylogeny of the most species-rich freshwater bivalve family (Bivalvia: unionida: Unionidae): Defining modern subfamilies and tribes. Mol. Phylogenet Evol. 106, 174–191 (2017).
Piechocki, A. & Wawrzyniak-Wydrowska, B. Guide To Freshwater and Marine Mollusca of Poland (Bogucki Wydawnictwo Naukowe, 2016).
Szlauer-Łukaszewska, A., Andrzejewski, W., Gierszal, H. & Urbańska, M. Co-occurrence of sinanodonta woodiana with native Unionidae in the lower Oder. Oceanol. Hydrobiol. Stud. 46, 244–248 (2017).
Piechocki, A. Reproductive biology of Unio pictorum (Linnaeus) and U. tumidus Philipsson in the Pilica river (Central Poland). Heldia 4, 53–60 (1999).
Abraszewska-Kowalczyk, A. Unionid bivalves of the Pilica river catchment area. Folia Malacol. 3, 99–173 (2002).
Killeen, J. J., Aldridge, D. & Oliver, G. Freshwater Bivalves of Britain and Irelanedvol. 82 (National Museum of Wales, 2004).
Kryger, J. & Riisgård, H. U. Filtration rate capacities in 6 species of European freshwater bivalves. Oecologia 1, 34–38 (1988).
Chowdhury, G. W., Zieritz, A. & Aldridge, D. C. Ecosystem engineering by mussels supports biodiversity and water clarity in a heavily polluted lake in dhaka, Bangladesh. Freshw. Sci. 35, 188–199 (2016).
Zieritz, A., Hoffman, J. I., Amos, W. & Aldridge, D. C. Phenotypic plasticity and genetic isolation-by-distance in the freshwater mussel Unio pictorum (Mollusca: Unionoida). Evol. Ecol. 24, 923–938 (2010).
Thorp, J. H. & Casper, A. F. Potential effects on zooplankton from species shifts in planktivorous mussels: a field experiment in the St Lawrence river. Freshw. Biol. 47, 107–119 (2002).
Cichy, A., Urbańska, M., Marszewska, A., Andrzejewski, W. & Żbikowska, E. The invasive Chinese pond mussel Sinanodonta woodiana (Lea, 1834) as a host for native symbionts in European waters. J. Limnol. 75, 288–296 (2016).
Pavluchenko, O. V. & Yermoshyna, T. V. Parasites of unionid molluscs (Bivalvia, Unionidae) and their effect on the body of molluscs. Regul. Mech. Biosyst. 8, 482–488 (2017).
Ćmiel, A. M. et al. The size and shape of parasitic larvae of naiads (Unionidae) are not dependent on female size. Sci. Rep. 11, 23755 (2021).
Chakraborty, A., Parveen, S., Chanda, D. K. & Aditya, G. An insight into the structure, composition and hardness of a biological material: the shell of freshwater mussels. RSC Adv. 10, 29543–29554 (2020).
Atkinson, C. L. et al. Gains and gaps in knowledge surrounding freshwater mollusk ecosystem services. Freshw. Mollusk Biology Conserv. 26, 20–31 (2023).
Gutiérrez, J. L., Jones, C. G., Strayer, D. L. & Iribarne, O. O. Mollusks as ecosystem engineers: the role of shell production in aquatic habitats. Oikos 101, 79–90 (2003).
Bołtruszko, J. S. Epizoic communities of rotifera on freshwater bivalves. Oceanol. Hydrobiol. Stud. 39, 75–82 (2010).
Njimerie, M. E. Shell Morphology of the Unionid Mussels (Anodonta Anatina, Unio Pictorum and Unio tumidus) in Relation To Gender and Trematode Parasitism (University of Jyväskylä, 2015).
Zając, K., Zając, T. & Ćmiel, A. What can we infer from the shell dimensions of the thick-shelled river mussel Unio crassus? Hydrobiologia 810, 415–431 (2018).
Li, L., Zhang, X., Yun, H. & Li, G. Complex hierarchical microstructures of cambrian mollusk pelagiella: insight into early biomineralization and evolution. Sci. Rep. 7, 1935 (2017).
Czerniejewski, P., Dąbrowski, J., Wawrzyniak, W., Brysiewicz, A. & Surma, O. Shell morphology, growth and longevity of Unio tumidus (Bivalvia: Unionidae) from an archaeological site and contemporary population inhabiting the Oder estuary. Hydrobiologia 848, 3555–3569 (2021).
Ollard, I., Ballantyne, R. & Aldridge, D. C. Freshwater mussel (Unio pictorum) shells reveal hydrological and environmental change from 1300 BC to the present day. Glob Chang. Biol. 30, e17532 (2024).
Quaempts, E. J., Jones, K. L., O’Daniel, S. J., Beechie, T. J. & Poole, G. C. Aligning environmental management with ecosystem resilience: a first foods example from the confederated tribes of the umatilla Indian reservation, oregon, USA. Ecol. Soc. 23, art29 (2018).
Nędzarek, A., Czerniejewski, P. & Tórz, A. A comparison of the concentrations of heavy metals in modern and medieval shells of swollen river mussels (Unio tumidus) from the Szczecin lagoon, SW Baltic basin. Mar. Pollut Bull. 163, 111959 (2021).
Ożgo, M., Bogucki, Z. & Nowakowska, M. Shells of Unio tumidus (Bivalvia: Unionidae) from an archaeological site and contemporary population inhabiting the same lake. Pol. J. Ecol. 4, 839–844 (2012).
Ollard, I. & Aldridge, D. C. Declines in freshwater mussel density, size and productivity in the river Thames over the past half century. J. Anim. Ecol. 92, 112–123 (2023).
Lewandowski, K. & Kołodziejczyk, A. Long-term changes in the occurrence of unionid bivalves in a eutrophic lake. Folia Malacol. 22, 301–309 (2014).
Ślusarska, K. Wild resources in the economy of bronze and early iron ages between Oder and bug Rivers – Source overview. Open. Archaeol. 7, 177–210 (2021).
Chełkowski, Z. Pozostałości ichtiologiczne z Badań Wykopaliskowych Na Grodzisku Wczesnośredniowiecznym w Szczecinie [Ichthyological remains from excavations conducted on early medieval city of Szczecin] (in Polish). Materiały Zachodniopomorskie. 11, 551–561 (1965).
Friedland, R. et al. Managing eutrophication in the Szczecin (Oder) Lagoon-Development, present state and future perspectives. Front. Mar. Sci. 5, 521 (2019).
Wolnomiejski, N. & Witek, Z. The Szczecin Lagoon Ecosystem: the Biotic Community of the Great Lagoon and its Food Web Model (Versita, 2013).
Ziuganov, V., Zotin, A. A., Nezlin, L. & Tretiakov, V. The Freshwater Pearl Mussels and their Relationship with Salmonid Fish (VNIRO Publishing House, 1994).
Mitchell, J. & Peacock, E. A. Prehistoric freshwater mussel assemblage from the big sunflower river, sunflower county, Mississippi. Southeast. Nat. 13, 626–638 (2014).
Miller, E. J., Tomasic, J. J. & Barnhart, M. C. A comparison of freshwater mussels (Unionidae) from a late-archaic archeological excavation with recently sampled verdigris river, kansas, populations. Am. Midl. Nat. 171, 16–26 (2014).
Peacock, E. Assessing bias in archaeological shell assemblages. J. Field Archaeol. 27, 183–196 (2000).
Fritts, A. K., Fritts, M. W., Haag, W. R., DeBoer, J. A. & Casper, A. F. Freshwater mussel shells (Unionidae) chronicle changes in a North American river over the past 1000 years. Sci. Total Environ. 575, 199–206 (2017).
Lundquist, S. P., Worthington, T. A. & Aldridge, D. C. Freshwater mussels as a tool for reconstructing climate history. Ecol. Indic. 101, 11–21 (2019).
Sługocki, Ł. & Czerniawski, R. Water quality of the Odra (Oder) river before and during the ecological disaster in 2022: A warning to water management. Sustainability 15, 8594 (2023).
Peacock, E. & Chapman, S. Taphonomic and biogeographic data from a plaquemine shell midden on the Ouachita river, North Louisiana. Southeast. Archaeol. 20, 44–55 (2001).
Wolverton, S., Randklev, C. R. & Kennedy, J. H. A conceptual model for freshwater mussel (family: Unionidae) remain preservation in Zooarchaeological assemblages. J. Archaeol. Sci. 37, 164–173 (2010).
Vaught, C. C. et al. 1997 A comparison of the effectiveness of timed searches vs quadrat sampling in mussel surveys. in Proceedings of a UMRCC Symposium Conservation & Management of Freshwater Mussels II. Initiatives for the Future (eds. Cummings, K. S., Buchanan, A. C., Mayer, C. A. & Naimo, T. J.) (1997).
Graf, D. L. & Cummings, K. S. Review of the systematics and global diversity of freshwater mussel species (Bivalvia: Unionoida). J. Molluscan Stud. 73, 291–314 (2007).
Lopes-Lima, M. et al. Conservation status of freshwater mussels in europe: state of the Art and future challenges. Biol. Rev. 92, 572–607 (2016).
Shea, C. P., Peterson, J. T., Wisniewski, J. M. & Johnson, N. A. Misidentification of freshwater mussel species (Bivalvia:Unionidae): contributing factors, management implications, and potential solutions. J. North. Am. Benthol Soc. 30, 446–458 (2011).
Morais, P., Rufino, M. M., Reis, J., Dias, E. & Sousa, R. Assessing the morphological variability of Unio Delphinus spengler, 1783 (Bivalvia: Unionidae) using geometric morphometry. J. Molluscan Stud. 80, 17–23 (2013).
Holland, H. A., Schöne, B. R., Lipowsky, C. & Esper, J. Decadal climate variability of the North sea during the last millennium reconstructed from bivalve shells (Arctica islandica). Holocene 24, 771–786 (2014).
Hastie, L. C., Young, M. R. & Boon, P. J. Growth characteristics of freshwater Pearl mussels, Margaritifera margaritifera (L). Freshw. Biol. 43, 243–256 (2000).
Hornbach, D. J., Kurth, V. J. & Hove, M. C. University of Notre Dame variation in freshwater mussel shell sculpture and shape along a river gradient. Am. Midl. Nat. 164, 22–36 (2010).
Amyot, J. P. & Downing, J. A. Endo- and epibenthic distribution of the unionid mollusc Elliptio complanata. J. North. Am. Benthol Soc. 10, 280–285 (1991).
Green, R. H., Bailey, R. C., Hinch, S. G., Metcalfe, J. L. & Young, V. H. Use of freshwater mussels (Bivalvia: Unionidae) to monitor the nearshore environment of lakes. J. Great Lakes Res. 15, 635–644 (1989).
Okupny, D. et al. How well multi-indicator palaeo-environmental studies Meet the needs of research on settlements, on the example of the early medieval settlement complex in szczecin: methodological problems and evaluating interpretation value. Acta Geogr. Lodziensia. 112, 97–121 (2022).
Zieritz, A., Mahadzir, F. N., Chan, W. N. & McGowan, S. Effects of mussels on nutrient cycling and bioseston in two contrasting tropical freshwater habitats. Hydrobiologia 835, 179–191 (2019).
Via, S. et al. Adaptive phenotypic plasticity: consensus and controversy. Trends Ecol. Evol. 10, 212–217 (1995).
Pigliucci, M. Evolution of phenotypic plasticity: where are we going now? Trends Ecol. Evol. 20, 481–486 (2005).
Vincenzi, S., Jesensek, D. & Crivelli, A. J. Biological and statistical interpretation of size-at-age, mixed-effects models of growth. R Soc. Open. Sci. 7, 192146 (2020).
Sansom, B. J., Atkinson, C. L. & Vaughn, C. C. Growth and longevity estimates for mussel populations in three Ouachita mountain rivers. Freshw. Mollusk Biology Conserv. 19, 19–26 (2016).
Dettman, D. L., Reische, A. K. & Lohmann, K. C. Controls on the stable isotope composition of seasonal growth bands in Aragonitic fresh-water bivalves (unionidae). Geochim. Cosmochim. Acta. 63, 1049–1057 (1999).
Versteegh, E. A. A., Troelstra, S. R., Vonhof, H. B. & Kroon, D. Oxygen isotope composition of bivalve seasonal growth increments and ambient water in the rivers Rhine and Meuse. Palaios 24, 497–504 (2009).
Pickard, C., Boroneant, A. & Bonsal, C. Molluscan remains from early to middle holocene sites in the iron gates reach of the danube, Southeast Europe. in In Molluscs in Archaeology: Methods, Approaches and Applications (ed Allen, M.) 179–194 (Oxbow Books, Oxford, (2017).
Haag, W. R. & Rypel, A. L. Growth and longevity in freshwater mussels: evolutionary and conservation implications. Biol. Rev. 86, 225–247 (2011).
Vaughn, C. C. & Hakenkamp, C. C. The functional role of burrowing bivalves in freshwater ecosystems. Freshw. Biol. 46, 1431–1446 (2001).
Ercan, E., Gaygusuz, Ö., Tarkan, A. S., Reichard, M. & Smith, C. The ecology of freshwater bivalves in the lake Sapanca basin, Turkey. Turkish J. Zool. 37, 730–738 (2013).
Aldridge, D. C. The morphology, growth and reproduction of Unionidae (Bivalvia) in a Fenland waterway. J. Molluscan Stud. 65, 47–60 (1999).
Abdelsaleheen, O., Taskinen, J. & Kortet, R. Reproductive cycle, fecundity and growth of the freshwater mussel Unio tumidus (Bivalvia: Unionidae) from lake viinijärvi, Finland. J. Molluscan Stud. 90, eyae024 (2024).
Mann, M. E. et al. Global signatures and dynamical origins of the little ice age and medieval climate anomaly. Sci. (1979). 326, 1256–1260 (2009).
Schöne, B. R., Dunca, E., Mutvei, H. & Norlund, U. A 217-year record of summer air temperature reconstructed from freshwater Pearl mussels (M. margaritifera, Sweden). Quat Sci. Rev. 23, 1803–1816 (2004).
Hawkins, A. J. S., Salkeld, P. N., Bayne, B. L., Gnaiger, E. & Lowe, D. M. Feeding and resource allocation in the mussel mytilus edulis: evidence for time-averaged optimization. Mar. Ecol. Prog Ser. 20, 273–287 (1985).
Kornijów, R., Pawlikowski, K., Drgas, A., Rolbiecki, L. & Rychter, A. Mortality of post-settlement clams Rangia cuneata (Mactridae, Bivalvia) at an early stage of invasion in the vistula lagoon (South Baltic) due to biotic and abiotic factors. Hydrobiologia 811, 207–219 (2018).
Wetzel, R. G. Limnology. Lake and Rivers Ecosystems (Academic, 2001).
Carey, C. S., Jones, J. W., Hallerman, E. M. & Butler, R. S. Determining optimum temperature for growth and survival of laboratory-propagated juvenile freshwater mussels. N Am. J. Aquac. 75, 532–542 (2013).
Dąbrowski, J., Czerniejewski, P., Brysiewicz, A. & Więcaszek, B. Morphometrics, growth and condition of the invasive bivalve Rangia cuneata during colonisation of the Oder estuary (North-Western Poland). Water (Basel). 15, 3331 (2023).
Macklin, M. G. et al. Past hydrological events reflected in the holocene fluvial record of Europe. Catena (Amst). 66, 145–154 (2006).
Strayer, D. L. Freshwater Mussel Ecology (University of California, 2008).
Perșoiu, I. & Perșoiu, A. Flood events in Transylvania during the medieval warm period and the little ice age. Holocene 29, 85–97 (2019).
Cushway, K. C., Harris, A. E., Piercy, C. D., Mitchell, Z. A. & Schwalb, A. N. Go with the flow: impacts of high and low flow conditions on freshwater mussel assemblages and distribution. PLoS One 19, (2024).
DuBose, T. P., Patten, M. A., Holt, A. S. & Vaughn, C. C. Latitudinal variation in freshwater mussel potential maximum length in Eastern North America. Freshw. Biol. 67, 1020–1034 (2022).
Verma, A. K., Rahman, A., Hussain, S. & Singh, N. S. Freshwater mussels as multifaceted ecosystem engineers: insights into their ecological importance, bioindication, and economic contributions. Water (Basel). 17, 1629 (2025).
Dudgeon, D. et al. Freshwater biodiversity: importance, threats, status and conservation challenges. Biol. Rev. 81, 163–182 (2006).
Haag, W. R. North American Freshwater Mussels (Cambridge University Press, 2012). https://doi.org/10.1017/CBO9781139048217
Hyvärinen, H. S. H., Sjönberg, T., Marjomäki, T. J. & Taskinen, J. Effect of low dissolved oxygen on the viability of juvenile margaritifera margaritifera: hypoxia tolerance ex situ. Aquat. Conserv. 32, 1393–1400 (2022).
Cope, W. G. et al. Understanding the influence of multiple pollutant stressors on the decline of freshwater mussels in a biodiversity hotspot. Sci. Total Environ. 773, 144757 (2021).
Warcholak, P. Krótki Rys historyczne regulacji Rzeki Odry. Zeszyty Naukowe Poliechniki Zielonogórskiej. 118, 185–188 (1998).
Brysiewicz, A., Czerniejewski, P. & Sieczko, L. An assessment of maintenance works and their impact on macroinvertebrate communities and long-term recolonization to small lowland watercourses. J. Environ. Manage. 367, 121849 (2024).
Brysiewicz, A. & Czerniejewski, P. The effect of maintenance works on ichthyofauna in the context of hydrochemical conditions of small watercourses of central and North-Western Poland. J. Ecol. Eng. 20, 82–89 (2019).
Lammens, E. H. R. R. & Hoogenboezem, W. Diets and feeding behaviour. in Cyprinid Fishes (eds Winfield, I. J. & Nelson, J. S.) 353–376 (Springer Netherlands, Dordrecht, doi:https://doi.org/10.1007/978-94-011-3092-9_12. (1991).
Modesto, V. et al. Fish and mussels: importance of fish for freshwater mussel conservation. Fish Fish. 19, 244–259 (2018).
Pilotto, F., Sousa, R. & Aldridge, D. C. Is the body condition of the invasive zebra mussel (Dreissena polymorpha) enhanced through attachment to native freshwater mussels (Bivalvia, Unionidae)? Sci. Total Environ. 553, 243–249 (2016).
Stańczykowska, A., Lewandowski, K. & Czarnoleski, M. Backhuys Publishers, Leiden, Distribution and densities of Dreissena polymorpha in Poland – past and present. in The Zebra Mussel in Europe (eds. van der Verde, G., Rajagopal, S. & bij de Vaate, A.) 119–127 (2010).
Rizhinashvili, A. L. On the relationships between absolute and allometric shell growth in unionid mussels (Bivalvia, Unionidae) from European Russia. Inland. Water Biol. 1, 241–247 (2008).
Shingleton, A. W. & Allometry The study of biological scaling. Nat. Educ. Knowl. 3, 2 (2010).
Jonsson, A., Bertilsson, A. & Rydgård, M. Spatial distribution and age structure of the freshwater unionid mussels anodonta anatina and Unio tumidus: implications for environmental monitoring. Hydrobiologia 711, 61–70 (2013).
Ogle, D. H. Introductory Fisheries Analyses with R (Chapman & Hall, 2016).
Mour, P. et al. Age and growth of a highly successful invasive species: the Manila clam Ruditapes philippinarum (Adams & reeve, 1850) in the Tagus estuary (Portugal). Aquat. Invasions. 12, 133–146 (2017).
Peharda, M., Župan, I., Bavčević, L., Frankić, A. & Klanjs̆ček, T. Growth and condition index of mussel Mytilus galloprovincialis in experimental integrated aquaculture. Aquac Res. 38, 1714– (2007).
Yildiz, H., Vural, P., Acarli, S. & Condition Index Meat yield and biochemical composition of mediterranean mussel (Mytilus galloprovincialis lamarck, 1819) from Canakkale strait, Turkey. Alinteri J. Agric. Sci. 36, 308–314 (2021).
Caill-Milly, N., Bru, N., Mahé, K., Borie, C. & D’Amico, F. Shell Shape Analysis and Spatial Allometry Patterns of Manila Clam (Ruditapes philippinarum) in a Mesotidal Coastal Lagoon. J Mar Biol 281206 (2012). (2012).
Klishko, O. K., Lopes-Lima, M., Bogan, A. E., Matafonov, D. V. & Froufe, E. Morphological and molecular analyses of anodontinae species (Bivalvia, Unionidae) of lake Baikal and transbaikalia. PLoS One. 13, e0194944 (2018).
Lewis, N. D. 100 Statistical Tests in R. Easy R Series (Heather Hills, 2013).
Warton, D. I., Duursma, R. A., Falster, D. S. & Taskinen, S. Smatr 3– an R package for Estimation and inference about allometric lines. Methods Ecol. Evol. 3, 257–259 (2012).
R Core Team. A language and environment for statistical computing: R Foundation for Statistical Computing.Vienna. (2023). https://www.R-project.org/
Acknowledgements
The authors are thankful to Derek H. Oggle for his help in the interpretation of growth rate assessment using the von Bertalanffy method.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Contributions
P.C. conduced fieldwork; J.D., P.C., A.B. conceptualized the research; J.D. formal data analysis; J.D., P.C., A.B prepared draft manuscript; A.B. and P.C. reviewed and commented on the manuscript; A.B and P.C. edited throughout. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Statement confirming
The reporting in the manuscript follows the recommendations in the ARRIVE guidelines.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dąbrowski, J., Czerniejewski, P. & Brysiewicz, A. Millennial-scale changes in size and growth of Unio pictorum (L. 1758) and U. tumidus (Philipsson, 1788), in the Oder river. Sci Rep 15, 30576 (2025). https://doi.org/10.1038/s41598-025-16251-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-16251-7