Abstract
Metastatic breast cancer has historically had a poor prognosis, but oligometastatic breast cancer shows differing outcomes. We analyzed the factors affecting 5-year survival for patients undergoing upfront resection. We conducted a 10-year chart review from a tertiary referral hospital. Patients were followed for at least 5 years after surgery. Overall and progression free survival were calculated from date of surgery using cox-proportional hazards while logistic regression was used to evaluate the pre- and post-operative factors associated with surgery. 55 patients with a mean age of 50.4 years were included. Most patients had T4, N1/N2 stage disease on presentation. The most common site of metastasis was lungs. The median overall survival was 111 months with a 5-year survival rate of 64.6% (47.9%, 77.2%). There were 18 distant and 1 local recurrence and 20 deaths in our cohort leading to a median progression free survival of 36 months. N2 nodal disease, and liver metastasis were identified as poor prognostic markers while hormone receptors increased survival (p < 0.05), while locally advanced disease and triple positive tumor biology increased the risk of progression and death (p < 0.05). Patients with limited metastatic disease should be considered for upfront resection with the intent to cure, especially when triple positive tumor biology is present.
Similar content being viewed by others
Introduction
Breast cancer is the second most common cause of death in women globally. According to the Centre for Disease Control and Prevention, the incidence in the US was 7.9 per 100,000 in 20211. About 60% of breast cancer-related mortality is reported to be concentrated in lower- and middle-income countries2. As of 2019, Pakistan was had the highest rate of breast cancer-related mortality3. Generally, a large proportion of breast cancer-related mortality can be attributed to metastatic breast cancer1. Reyes et al. estimated that almost a third of breast cancer patients succumbed to widespread metastatic disease4.
Oligo-metastatic Breast Cancer (OMBC) is a sub-set which makes 20% of metastatic breast cancer. These are patients with limited number and sites of metastasis. Although no consensus exists on the definition of OMBS, most authors seem to agree with a version of the following definition that we used: metastatic lesions limited to one or two organs (excluding the primary resectable lesion), less than 5 lesions per organ, and diameter smaller than 5 cm per lesion5. Oligometastasis was first explained by Hellman in 1994 as restricted loco-regional tumor load with indolent behavior, for which he suggested aggressive management with chemotherapy and radiotherapy6. This concept was revisited in 2011, and it was understood that local cancer treatments are curative in a proportion of patients with metastasis, more commonly in oligometastatic cancer7. The OMBC phenotype has low metastatic potential and there is increasing evidence to advocate for aggressive local therapy for these patients in view of their increased survival and favorable prognosis.
Overall breast cancer survival has been improving with advances in the understanding of the biological behavior of the disease and the evolution in adjuvant treatment options8. Providing a treatment option aimed at an eventual disease-free state for OMBC patients is an exciting avenue as breast cancer treatment progresses9. Despite convincing retrospective data and ongoing prospective trials supporting radical surgery of local disease to improve survival outcomes, conflict in opinions and lack of treatment guidelines persist to treat this unique entity of patients10.
There is sparsity of local data on this subset of patients and there is a lack of recommended guidelines for these patients. In order to bridge this gap in literature, we aim to present our 10-year institutional experience in caring for patients with OMBC who underwent surgery of the primary lesion with the intention to cure. Our paper will elucidate the overall and progression free survival for these patients as well as the factors associated with it.
Materials and methods
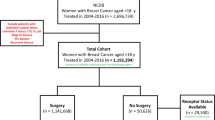
We conducted a retrospective chart review of patients who presented with oligometastatic disease at the time of diagnosis after staging work up, which included CT scan of the chest, abdomen and pelvis as well as a bone scan, magnetic resonance imaging, and positron emission tomography scan. Patients who underwent surgery at a single tertiary care hospital between 1st January 2009 and 31st December 2019 were included.
The included patients were more than 18 years old and labelled as stage IV disease de novo. Patients who received initial chemotherapy and/or adjuvant chemotherapy, radiation, hormonal therapy or immunotherapy were also included in the analysis. Patients who were pregnant on presentation or had incomplete medical records were excluded.
Ethical exemption was obtained for the use of records from the Aga Khan University ethical review committee (ERC# 2021-6152-19307).
Clinical and pathological data
Information on clinical and treatment characteristics of patients (Identification number, age, menopausal status, pre or post Neo adjuvant status, size of primary lesion-T, axillary nodal status-N, tumor biology—Estrogen receptor, Progesterone receptor, Her2Neu status, number and size of metastatic lesion, organ involved, type of primary surgery- mastectomy, wide local excision, oncoplastic breast conservation, adjuvant chemotherapy and radio therapy, number of years of follow up, any adverse event- local recurrence, increase in number or size of metastatic lesion, metastasis in any new organ was taken from patient file and electronic medical records. Patients with essential missing data were excluded from the analysis.
Follow-up and survival endpoints
Follow-up was quarterly for the initial two years and then biyearly from then on. All patients received a thorough history and examination at every visit to assess for signs and symptoms of local or distant recurrence. Radiological investigations including ultrasound, CT scan or a PET scan were also conducted where required.
Patients were followed until 22nd December 2024 for a complete five-year survival data set. Patients who had less than five years of survival data were labelled as lost to follow-up and censored. Disease-free-interval (DFI) was defined as the date of recurrence or death from the date of surgery.
Outcome was assessed as overall survival and progression free survival. Overall survival (OS) was defined as survival from the date of surgery until the date of death or the date of last follow-up. Progression free survival (PFS) was defined as survival from the date of surgery till the date of recurrence, death, or the date of last follow-up.
Data analysis
STATA (MP 17) was used for statistical analysis. Quantitative variables were reported as means (95% CI) or median (IQR) while categorical variables were reported as percentages (frequency). We compared patients who were censored vs the ones included to assess if censoring was informative.
Kaplan-Meir survival curves were generated to assess OS and PFS. Mean survival time was assessed and tested using log-rank test. Log-rank test was used to assess if censoring was informative. After testing the cox proportionality assumption, cox regression was performed to determine the association of clinical and treatment factors with OS and PFS, keeping confounding in consideration. They have been reported as hazard ratios with their respective 95% confidence intervals. P-value less than 0.05 is considered significant throughout the study.
Ethical considerations
The research was undertaken considering the principles detailed in the declaration of Helsinki. Ethical approval was obtained from the Ethical Review Committee at the Aga Khan University. Informed consent was waived by the Aga Khan University Ethical Review Committee.
Results
Our cohort consisted of 55 women who underwent surgery with curative intent between 1st January 2019 till 31st December 2019. We followed our participants for up to 15 years till 21st December 2024. The mean age at diagnosis was 50.4 (47.4, 53.4) years. About 73% of the women were peri or post-menopausal. The median size of the initial lesions was 6.0 cm, with a wide variability from 2 to 25 cm. In most patients (55.6%), the initial lesion was localized to the upper outer quadrant. Table 1 describes the characteristics of the included patients.
On the initial presentation, the majority (63.6%) of patients on presentation had a cT4 lesion. A similar proportion (54.6%) of the patients had clinically palpable mobile lymph nodes (N1). The most common kind of metastatic lesions at the initial presentation were lung metastasis (43.4%), followed by bone metastasis (35.9%); none of the patients in our study had brain metastasis. After surgery, most patients had a lower pathological T stage (p < 0.001). However, there was no significant change in the pathological N stage as compared to the clinical N stage (p = 0.07).
Initial chemotherapy was given to more than half of our cohort (56.4%; n = 31). The time lapse between diagnosis and surgery was 12.6 months for patients who did not undergo chemotherapy vs 14.2 months for those who underwent chemotherapy (p = 0.046). Most of the patients received Adriamycin and Cytoxan followed by Taxol (60.5%) while a fifth of the patients got Fluorouracil, Adriamycin, and Cyclophosphamide. About half of the patients (47.3%) received hormonal therapy with tamoxifen being the most common (29.1%).
Figure 1 shows the Kaplan–Meier curve for overall survival. The median follow-up time was 31 months (IQR = 8.2–93.4 months). The 5-year survival rate for our cohort is 64.6% (47.9%, 77.2%) with a median survival of 111 months. When adjusting for age, survival remained unaffected. However, it marginally increased to 68.0% when adjusting for the size of the mass. Patients who underwent mastectomy had a 61.6% chance of being alive at 5 years, although a similar comparison could not be made for patients who underwent breast conserving surgery as all but two of the patients were censored. The 5-year survival was similar at 60.2% for patients with grade 2 disease and 75.7% for those with grade 3 disease (p = 0.09).
Overall survival and 95% CI; numbers of patients censored are in parentheses.
Figure 2 shows the Kaplan Meier curve for progression free survival. The 5-year progression free survival was 49.2% (33.7%, 63.0%) with a median progression free survival of 36 months. There were 19 recurrences in our study. The cumulative hazard of survival stratified by menopausal status at diagnosis corresponds to 0.72 for premenopausal females as compared to post-menopausal females at the end of the study period (160 months).
Progression-free-survival and 95% CI; numbers of patients censored are in parentheses.
Table 2 describes the unadjusted and adjusted risk of death in our cohort after multivariate logistic regression. Considering preoperative clinical factors, the hazard of death was 4.7 and 5.8 respectively for patients with liver or bone metastasis (p < 0.05). Other factors like T and N stage at diagnosis were not significantly related to the risk of death. Moreover, there was no significant difference in outcome between patients who underwent initial chemotherapy vs upfront resection. Amongst the postoperative risk factors, pathological T staging was significant with a 29.3 times higher chance of death in patients with a pT3 stage (p < 0.05). Moreover ER/PR positive patients had a 0.025 hazard of death as compared to triple positive (ER/PR and HER2) patients (p < 0.05) after adjusting for pT and pN as well as tumor differentiation.
There were 19 recurrences during the 15-year follow-up period. The median disease-free-interval (DFI) was 13.2 months (IQR = 4.87–26.53). Liver metastasis increased the hazard of progression 300% (p < 0.05). The hazard of progression was 3.60 for pN3 stage as compared to pN0 stage (p < 0.05). After adjusted cox proportional hazards regression, triple positive tumor biology was associated with 6.8 times the hazard of progression as compared to hormone receptor positive disease while pT3 lesion had 21.8 times the hazard of progression as compared to pTis/pT1 disease (p < 0.05).
Discussion
To our knowledge, this study is the first looking at oligometastatic breast cancer patients in Pakistan. The 55 women included in our study form an integral cohort in the development of future recommendations for future treatment strategy. Despite the overall grim outlook of stage IV disease globally with a median survival of 2–3 years, our cohort showed a median survival of over 9 years11. These findings are encouraging for metastatic breast cancer patients. Although it is not possible to directly compare the survival difference between our cohort and other first line MBC patients, our findings support the hypothesis that patients with oligometastatic disease at presentation may benefit from aggressive local cure strategies.
Nguyeun et al. have shown that limited M1 disease is a good prognostic factor for patients with stage IV breast cancer with 5-year OS of 29.7%12. In a Dutch cohort of approximately 3500 women, the 10-year survival rate was 14.9% for patients with three or less metastatic lesions as opposed to 3.4% for those with more than three lesions13. Moreover, Lan et al. studied extracranial oligometastatic lesions and found the 5-year overall survival to be 58%, which is closer to our cohort14. Although the Chinese cohort showed better overall survival with upfront resection vs primary chemotherapy, no such difference was observed in our cohort, which may be due to the small number of patients who underwent upfront resection.
There is adequate retrospective data to suggest that patients with limited metastatic disease have a favorable prognosis when treated aggressively. However, evidence shows that this group of patients is diverse in their response to treatment. Indolent subtypes, longer disease-free interval, and low number of involved sites are seen to be predictors of longer OS15,16,17,18. Our data showed that the disease-free interval had no effect on survival. An RCT by Khan et al. failed to show any significant benefit in treating locoregionally as opposed to systemic therapy for oligometastatic disease19. However, the Turkish federation’s dataset favored survival at 40 months for the surgical resection arm (70% vs 55%, p < 0.01)20. The main difference between this paper and other negative trials is that patients underwent upfront locoregional surgery as opposed to surgery after systemic therapy for a limited period in the other studies. However, there is no landmark paper that exists to define guidelines for women with oligometastatic breast cancer, and there is sparsity of data from LMICs.
Cancer biology is also helpful since specific subtypes tend to metastasize to certain sites e.g., hormone-receptor positive disease and triple negative disease have a predilection for bones and the brain, respectively21. Oligometastasis is proposed to be an early, intermediate phase of metastatic progression, characterized by a limited number of tumors that have not yet acquired the genetic changes needed for extensive spread. Her2 positive disease as well as hormone receptor positive disease has been shown to have superior odds of survival, especially when targeted treatment is commenced immediately regardless of the date of surgery22. Our data was concurrent with these reports with an OS of 73% at 5 years for ER/PR positive disease. Moreover, bone metastasis, which is linked to hormone receptor positive disease, is seen to be favorable in larger datasets. Huang et al. showed the median OS to be 50 months vs 31 months for patients who underwent locoregional surgery and systemic therapy respectively (p < 0.001)23. Shein et al. also showed better OS with ER-positive disease, and pre-menopausal status24. Our patients were also about 95% more likely to survive with ER/PR positive disease when compared to other tumor biologies (p < 0.05). Triple negative tumor biology has been shown to have a higher hazard of progression and death, although our data failed to show such correlation25.
Ma and colleagues showed that systemic therapy combined with locoregional treatment was better than systemic therapy alone (median OS 70 vs 26 months; p < 0.001)26. They elucidated that a longer DFI and hormone receptor positive disease were predictors of good prognosis. ER positivity has been seen to allow longer survival in a Danish cohort as well as a systematic review27,28. Our participants also had better rates of survival with hormone receptor positive disease. This is largely due to the ability to modulate the effect of these hormones using a selective estrogen receptor modulator or an aromatase inhibitor in pre- and post-menopausal women respectively. Others have also reported a shorter DFI to be a predictor of poor prognosis25,28. Wu et al. described the effect of primary tumor resection in OMBC as seen in prospective and retrospective studies29. OMBC patients with limited metastasis had 40% higher chances of survival when treated with primary resection as opposed to systemic therapy alone. The results remained similar when stratified by number of sites (1,2, or ≤ 3 sites). There was no difference in survival in our study since most patients only had a single metastatic site. Nagasaki and colleagues reported 20-year overall survival at 34.1% for patients treated aggressively with OMBC30. The median overall survival was 184.5 months, longest reported for any cohort with OMBC. Compared to our cohort with an OS of 64.6% at 5 years, more than 83% of the patients survived amongst their participants. Moreover, patients who underwent any sort of chemotherapy also had better OS and PFS rates which underscores the importance of combining systemic therapy with local treatment. The treatment regimen for de novo OMBC is increasingly moving towards the inclusion of local and metastatic-directed radiotherapy with data showing OS of 87.3% and PFS of 60.5% at 3 years, which is better than our cohort31.
The site of metastatic lesions has been seen to affect survival in this cohort32. Liver metastasis generally has a poor prognosis with limited treatment modalities and strict eligibility criteria for patients33. However, with the advent of stereotactic body radiation and the option for hepatic resection as seen in metastasectomy for colorectal cancer, survival has improved for this group of patients although there is a need for randomized studies to study the effect of these interventions34. Our participants also had a higher hazard of death and progression with liver metastasis on presentation. Lung is one of the most common sites for metastasis and is one associated with the best outcomes, especially in resectable isolated pulmonary metastasis with 4-year OS reaching up to 82%35. The most common site, however, is the bone. It has the best prognosis of all metastatic breast cancer, given the increased predilection of hormone receptor positive disease to reside there which is responsive to readily available targeted therapy. This subtype of OMBC is also radiosensitive with most patients surviving without recurrence and 3-year OS more than 90%36,37. On the contrary, bone metastasis in our cohort increased the hazard of death (HR = 3.1) after adjusting for tumor size, age, initial chemotherapy, and menopausal status, although the result was not statistically significant (p > 0.05).
Our study had a few limitations. This was a retrospective chart review so no comment can be made on the effectiveness of the interventions due to the lack of a control group. There was a small cohort of patients who underwent initial chemotherapy and most of them became censored before the 5-year mark, so no meaningful analysis was possible. Data for management of metastatic sites was unavailable so no comparison could be made along those lines. However, one of the strengths of our study was that the lost to follow up group was not significantly different than the patients who were followed till death or the end of the study period (Table 3).
To conclude, this is the first reported group of participants from an LMIC who underwent aggressive treatment for oligometastatic breast cancer and were observed to have about 9 years of median survival. N2 disease as well as liver metastasis preoperatively and N2 disease postoperatively increased the risk of death while ER/PR positivity was protective for these patients. Although the cohort was small, these findings can be applied with caution to neighboring low-resource regions as well. Literature consistently shows that oligometastatic disease has better chances of survival with longer disease-free intervals and a low number of sites serving as good prognostic factors. Therefore, we propose that patients with 1–3 small metastatic lesions confined to a single organ will benefit from surgical resection with intent to cure given the high chances of survival in this cohort.
Data availability
Data availability: Data is available on reasonable request to the corresponding author.
References
CDC. Metastatic Female Breast Cancer Incidence. https://www.cdc.gov/united-states-cancer-statistics/publications/metastatic-breast-cancer.html#cdc_research_or_data_summary_res_data-trends-in-metastatic-female-breast-cancer-incidence-by-race-and-ethnicity.
Sung, H. et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 71, 209–249 (2021).
Xu, Y. et al. Global trends and forecasts of breast cancer incidence and deaths. Sci. Data 10, 334 (2023).
Reyes, D. K., Pienta, K. J., Reyes, D. K. & Pienta, K. J. The biology and treatment of oligometastatic cancer. Oncotarget 6, 8491–8524 (2015).
Gennari, A. et al. ESMO Clinical Practice Guideline for the diagnosis, staging and treatment of patients with metastatic breast cancer. Ann. Oncol. 32, 1475–1495 (2021).
Hellman, S. Natural history of small breast cancers. J. Clin. Oncol. 12, 2229–2234 (1994).
Weichselbaum, R. R. & Hellman, S. Oligometastases revisited. Nat. Rev. Clin. Oncol. 8, 378–382 (2011).
Heer, E. et al. Global burden and trends in premenopausal and postmenopausal breast cancer: a population-based study. Lancet Glob. Health 8, e1027–e1037 (2020).
Hafez, N. & Gettinger, S. Oligometastatic disease and local therapies: A medical oncology perspective. Cancer J. 26, 144–148 (2020).
Di Lascio, S. & Pagani, O. Oligometastatic breast cancer: A shift from palliative to potentially curative treatment?. Breast Care 9, 7–14 (2014).
Cardoso, F. et al. 5th ESO-ESMO international consensus guidelines for advanced breast cancer (ABC 5). Ann. Oncol. 31, 1623–1649 (2020).
Nguyen, D. H. A. et al. Limited M1 disease: A significant prognostic factor for stage IV breast cancer. Ann. Surg. Oncol. 19, 3028–3034 (2012).
Steenbruggen, T. G. et al. Characterization of oligometastatic disease in a real-world nationwide cohort of 3447 patients with de novo metastatic breast cancer. JNCI Cancer Spectr. 5, pkab010 (2021).
Lan, B. et al. Clinical features and prognostic factors for extracranial oligometastatic breast cancer in China. Int. J. Cancer 147, 3199–3205 (2020).
Fan, J., Chen, D., Du, H., Shen, C. & Che, G. Prognostic factors for resection of isolated pulmonary metastases in breast cancer patients: A systematic review and meta-analysis. J. Thorac. Dis. 7, 1441 (2015).
Planchard, D. et al. Uncertain benefit from surgery in patients with lung metastases from breast carcinoma. Cancer 100, 28–35 (2004).
Friedel, G. et al. Results of lung metastasectomy from breast cancer: prognostic criteria on the basis of 467 cases of the international registry of lung metastases. Eur. J. Cardiothorac. Surg. 22, 335–344 (2002).
Chen, F. et al. Clinical features of surgical resection for pulmonary metastasis from breast cancer. Eur. J. Surg. Oncol. 35, 393–397 (2009).
Khan, S. A. et al. Early local therapy for the primary site in de novo stage IV breast cancer: Results of a randomized clinical trial (EA2108). J. Clin. Oncol. 40, 978 (2022).
Soran, A. et al. Randomized trial comparing resection of primary tumor with no surgery in stage IV breast cancer at presentation: Protocol MF07-01. Ann Surg Oncol 25, 3141–3149 (2018).
Maki, D. D. & Grossman, R. I. Patterns of disease spread in metastatic breast carcinoma: Influence of estrogen and progesterone receptor status. AJNR Am. J. Neuroradiol. 21, 1064 (2000).
Soran, A. et al. Loco-regional treatment for intact primary tumor in patient with de novo metastatic breast cancer; Comments and concerns of ECOG-ACRIN 2108 trial. Eur. J. Breast Health 16, 158–159 (2020).
Huang, Z. et al. Surgery for primary tumor benefits survival for breast cancer patients with bone metastases: A large cohort retrospective study. BMC Cancer 21, 1–18 (2021).
Shien, T. et al. A randomized controlled trial comparing primary tumor resection plus systemic therapy with systemic therapy alone in metastatic breast cancer (PRIM-BC): Japan Clinical Oncology Group study JCOG1017. J. Clin. Oncol. 41, 523–523 (2023).
van Ommen-Nijhof, A. et al. Survival and prognostic factors in oligometastatic breast cancer. Breast 67, 14–20 (2023).
Ma, L. et al. Role of locoregional surgery in patients with de novo stage IV breast cancer: analysis of real-world data from China. Sci. Rep. 10, 1–8 (2020).
Steenbruggen, T. G. et al. Characterization of the tumor microenvironment of de novo oligometastatic breast cancer in a nationwide cohort. JCO Precis. Oncol. https://doi.org/10.1200/PO.22.00670 (2023).
van Ommen-Nijhof, A. et al. Prognostic factors in patients with oligometastatic breast cancer—A systematic review. Cancer Treat Rev 91, 102114 (2020).
Wu, C. et al. Primary tumor resection in de novo metastatic breast cancer from an oligometastatic perspective: A systematic review and meta-analysis. Science 27, 111224 (2024).
Nagasaki, E. et al. Long-term outcomes of oligometastatic breast cancer patients treated with curative intent: An updated report. Breast Cancer 28, 1051–1061 (2021).
Pujari, L. et al. Outcomes of de novo oligometastatic breast cancer treated with surgery of primary and metastasis directed radiotherapy. Am. J. Clin. Oncol. Cancer Clin. Trials https://doi.org/10.1097/COC.0000000000001129 (2024).
Zouki, D. N., Karatrasoglou, E. A., Pilichos, G. & Papadimitraki, E. Oligometastatic breast cancer: Seeking the cure by redefining stage IV disease?. Curr. Treat Options Oncol. 25, 1482–1494 (2024).
Zuo, Q., Park, N. H., Lee, J. K. & Erdogan, Z. M. Liver metastatic breast cancer: Epidemiology, dietary interventions, and related metabolism. Nutrients 14, 2376 (2022).
Hoffmann, K. et al. Liver resection for multimodal treatment of breast cancer metastases: Identification of prognostic factors. Ann. Surg. Oncol. 17, 1546–1554 (2010).
Yhim, H. Y. et al. Prognostic factors for recurrent breast cancer patients with an isolated, limited number of lung metastases and implications for pulmonary metastasectomy. Cancer 116, 2890–2901 (2010).
Miyata, M. et al. Salvage radiotherapy for solitary metachronous bone metastasis in patients with breast cancer. Anticancer Res. 37, 2575–2579 (2017).
Milano, M. T., Katz, A. W., Zhang, H. & Okunieff, P. Oligometastases treated with stereotactic body radiotherapy: Long-term follow-up of prospective study. Int. J. Radiat. Oncol. Biol. Phys. 83, 878–886 (2012).
Author information
Authors and Affiliations
Contributions
KS and LMV conceptualized the study. KS and MU wrote the first draft. SA and KS did data collection. MU did the formal data analysis. SA and LMV provided critical feedback for the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Shaikh, K., Uzair, M., Vohra, L.M. et al. Factors determining survival in oligometastatic breast cancer in a retrospective cohort study from a low and middle income country. Sci Rep 16, 3674 (2026). https://doi.org/10.1038/s41598-025-18342-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-18342-x