Abstract
This study evaluates plaque stability in atherosclerotic carotid plaques, which are key stroke contributors, by using radial wall strain (RWS) from digital subtraction angiography (DSA). It explores the Link between RWS, symptomatic stenosis, and endovascular treatment outcomes. In a single-center prospective study, 82 patients undergoing endovascular carotid atherosclerotic stenosis (CAS) treatment were assessed. Plaque vulnerability was analyzed using high-resolution magnetic resonance angiography and RWS from DSA images, examining the consistency and correlation of these methods, determining the optimal RWS threshold, and its relation to ischemic symptoms. A statistically significant correlation (p < 0.001) and concordance (Kappa = 0.447, p < 0.001) were observed between RWSmax and various aspects of plaque stability, as evaluated using high-resolution nuclear magnetic resonance. In severe CAS, RWSmax was higher in damaged plaques (17.7% vs. 12.7%, p < 0.001), with a non-significant trend in moderate CAS (15.7% vs. 10.8%, p = 0.068). Symptomatic CAS patients had higher RWSmax in vulnerable plaques (18% vs. 10%, p < 0.001), with a similar non-significant difference in asymptomatic patients (16.9% vs. 13.1%; p = 0.051). The optimal RWSmax cutoff for identifying vulnerability was 14.9% (AUC = 0.838; p < 0.001; 85.5% sensitivity, 74.1% specificity). RWSmax demonstrated excellent diagnostic accuracy across subgroups. More symptomatic patients had vulnerable plaques than asymptomatic ones [85.71% vs. 39.39%, p < 0.001], with higher median RWSmax in symptomatic patients [17.40% vs. 14.80%, p = 0.008]. Significantly more symptomatic than asymptomatic patients had RWSmax values ≥ 14.9% (77.55% vs. 48.48%, p = 0.006). The DSA-based RWS is a valuable index for the evaluation of CAS plaque vulnerability.
Trial Registration: The trial was registered to Chinese Clinical Trial Registry and the registration number is ChiCTR2400094665 (Registration date: 25/12/2024).
Similar content being viewed by others
Introduction
Ischemic stroke ranks among the foremost causes of mortality and morbidity on a global scale1. Extracranial carotid atherosclerotic stenosis (CAS) is a significant contributor to the incidence and recurrence of ischemic stroke worldwide2,3. Approximately 25% of ischemic strokes are associated with carotid plaque4. Current national and international guidelines, along with the expert consensus, advocate for endovascular intervention based on the severity of stenosis, with carotid balloon angioplasty and/or stenting being viable treatment modalities, depending on individual patient circumstances5,6,7. Nevertheless, an increasing body of clinical research indicates that the stroke recurrence risk is influenced not solely by the degree of stenosis, but also by the stability of atherosclerotic plaques, which plays a crucial role in the pathogenesis and recurrence of ischemic events8,9. Although the severity of stenosis is commonly used as a standard metric for the assessment of stroke risk, ischemic stroke can also arise from plaques causing only low-grade stenosis. Consequently, the perioperative assessment of the CAS extent and plaque stability is instrumental for the optimization of surgical strategies and selection of appropriate interventions to reduce complications10.
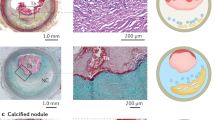
High-resolution magnetic resonance vessel-wall imaging (HR-VWI) enables the precise differentiation of plaque components, including the fibrous cap, intraplaque hemorrhage, Lipid-rich necrotic core, and calcification, for the achievement of accuracy rates of up to 90% in plaque composition analysis11. It is a powerful tool for the assessment of plaque vulnerability and the most suitable and cost-effective imaging technique for the characterization of vulnerable carotid plaques, facilitating early diagnosis and guiding clinical treatment12,13,14,15.
Digital subtraction angiography (DSA) is the current gold standard for the assessment of carotid stenosis16, as it not only offers insights into hemodynamics and collateral circulation but also provides the most direct imaging-based foundation for interventional therapy. Nonetheless, it has limitations in the evaluation of plaque stability.
In a recent report on a Coronary Artery Disease Study, researchers from multiple countries presented, for the first time on an international scale, an evaluation of plaque vulnerability utilizing artificial intelligence (AI) to calculate radial wall strain (RWS) values derived from individual digital subtraction angiography (DSA) images17. This method enables the comprehensive evaluation of coronary plaque stability, and results obtained with it show strong concordance with those obtained using optical coherence tomography (OCT)18,19. The pathophysiology of CAS plaques is analogous to that of coronary atherosclerosis, suggesting that RWS assessment could be feasible for CAS plaques, particularly given their larger diameters. To date, however, no report on this topic has been published. This study was conducted to investigate the correlation and consistency of DSA-derived RWS values and carotid HR-VWI–based plaque vulnerability classifications, and the predictive value of these measures for ischemic events.
Materials and methods
Design and participants
This single-center prospective study was conducted with consecutive patients with CAS undergoing Carotid balloon angioplasty and/or stenting at the Department of Neurology, Ordos City Center Hospital, China, between September 2022 and May 2024. The inclusion criteria were: (1) age ≥ 18 years; (2) DSA-based confirmation of CAS (≥ 50% symptomatic or ≥ 60% asymptomatic stenosis); and (3) several preoperative tests were conducted, such as carotid vascular ultrasound, magnetic resonance imaging (MRI), and HR-VWI. The exclusion criteria were: (1) the presence of tandem extracranial and/or intracranial stenosis (> 50%) in the ipsilateral carotid artery; (2) hepatic or renal insufficiency, or severe allergic reaction to the contrast agent; (3) cardiogenic embolism (e.g., atrial fibrillation, cardiac failure, or cardiac valvulopathy); (4) intracranial tumor, arteriovenous malformation, or aneurysm; and (5) unanalyzable DSA images of diseased vessels or incomplete clinical and/or imaging data. All patients received standardized medical therapy(Dual antiplatelet aggregation and statin lipid-lowering therapy) and carotid ultrasound during the perioperative period. The Ethics Committee of Ordos Central Hospital approved the study protocol (no. 2021-072), with all procedures conducted in accordance with the Declaration of Helsinki and relevant ethical standards.The patients or their family members provided written informed consent.
Data collection and group allocation
Data on patients’ general demographic and clinical characteristics and imaging data were collected. The patients were divided into symptomatic and asymptomatic groups according to ischemic event (ischemic stroke or transient ischemic attack) occurrence in the ipsilateral blood supply area of the responsible vessel within 6 months before hospitalization20.
HR-VWI–based plaque classification
HR-VWI examinations were performed using a Magnetom Verio 3.0-T MRI scanner (MAGNETOM Verio, Siemens Healthcare, Germany). Plaque types IV and V, according to the revised American Heart Association MRI classification standard for atherosclerotic plaques21, were defined as vulnerable, and other plaque types were defined as stable.
DSA examination, vascular stenosis classification, and RWS assessment
DSA examinations were performed using an ARTIS pheno device (Siemens). A contrast agent Omnipaque, GE Healthcare Pharmaceutical, Shanghai, China) with 350 mg iodine/ml was injected at a flow of 5 ml/s. Angiographic images were obtained at a rate of 7.5 frames/s. The Seldinger technique was used to perform selective angiography of the distal ends of the cerebral blood vessels and common carotid artery, including anteroposterior and, when necessary, oblique or three-dimensional imaging. A high-pressure injector (flow rate, 5 ml/s; total amount, 7 ml; pressure, 300 psi) was triggered manually to complete the acquisition.
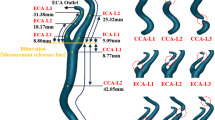
Using the DSA images, carotid stenosis was classified according to the standard of the North America Symptomatic Carotid Endarterectomy Trial22 as moderate (50–69%) or severe (70–99%).) The analysis of RWS based on angiographic image by one certified analyst who was blinded to patients’ clinical information and MRI data, using a commercially available software package (AngioPlus Core, Version 3, Pulse Medical). The standard operation procedure for RWSAngio analysis has been previously described17.Briefly, RWS was defined based on the maximal rate of change in a blood vessel’s diameter at a specific location throughout the entire arteriovenous cycle. Three image frames from the early cerebrovascular arterial, late arterial, and early venous phases were selected automatically. AI automatically delineated the vessel’s lumen contours on these images, and matched them to contours from other corresponding arteriovenous cycles to generate a longitudinal complete-cycle lumen diameter variation map for the blood vessel of interest. The RWS was taken as the relative diameter deformation at each position in the arteriovenous cycle, calculated as: RWS = (maximum diameter – minimum diameter)/maximum diameter. The lesion RWS was defined as the maximum radial wall strain (RWSmax) along the vessel segment of interest. All procedures were performed in accordance with the relevant guidelines and regulations. (Supplementary Appendix 1. Schematic diagram of RWSanalysis)
Statistical analysis
The R programming language was employed for data visualization, while statistical analyses were conducted using SPSS version 26.0. Data adhering to a normal distribution were summarized using the mean and standard deviation, and intergroup comparisons were performed using the t-test. For data not conforming to a normal distribution, the median and interquartile range [M (P25, P75)] were utilized, whereas categorical data were presented as frequencies and percentages [n (%)], with intergroup comparisons conducted via the chi-square test. Single-factor analysis was employed to examine the results, and parameters exhibiting statistically significant differences were further subjected to multivariate logistic regression analysis by sex. The relationship between RWS and plaque characteristics, as assessed by high-resolution vessel wall imaging (HR-VWI), was evaluated using box plots, and consistency was assessed using Cohen’s Kappa to further explore the association of RWS across different populations. To assess the diagnostic efficacy of RWSmax in various populations, patients were stratified into three categories: (1) moderate stenosis, (2) severe stenosis, and (3) asymptomatic. The study further aims to assess the sensitivity and specificity of RWS and HR-VWI in differentiating between vulnerable and non-vulnerable groups through receiver operating characteristic (ROC) analysis. The optimal threshold was established using the Youden index, and the predictive capability of RWS for ischemic events was examined via one-way binary logistic regression analysis, with plaque vulnerability as the independent variable and RWSmax as the dependent variable. Statistical significance was determined at a p-value of less than 0.05.
Results
Baseline clinical and pathological features
Of 147 potentially eligible patients screened, 82 patients with extracranial CAS were included in this study (Fig. 1). The median age of the patients was 70 years (IQR, 66.75–73.00 years), and 50 (60.97%) patients were male. The main vascular risk factors were hypertension [69 (84.15%) cases] and smoking [45 (54.88%) cases]. Severe and moderate stenosis were observed in 63 (76.83%) and 19 (23.17%) cases, respectively. HR-VWI revealed vulnerable plaques in 55 (67.07%) cases. Patient demographic characteristics, vascular risk factors, and imaging findings are summarized in Table 1.
Diagram of study flow. The entire procedure for the final screening of patients for enrollment is conducted in accordance with specified inclusion and exclusion criteria. HR-VWI, high-resolution magnetic resonance vascular-wall imaging; RWS, radial wall strain.
Correlation and consistency of RWSmax and HR-VWI measures of plaque vulnerability
Box plots showing the correlation between RWS and plaque properties assessed by HR-VWI. The distribution of vulnerable plaques, as determined by RWSmax values and HR-VWI–based assessment, differed significantly and correlated positively (p < 0.001; Fig. 2). The risk of plaque instability increased with the RWSmax value. The kappa consistency test showed good consistency between these values (κ = 0.447, p < 0.001).
Vulnerable plaque distribution, as evaluated by RWSmax calculation and HR-VWI.
To further evaluate the diagnostic efficacy of the RWSmax across different populations, patients were stratified into three groups: (1) moderate stenosis, (2) severe stenosis, and (3) asymptomatic. The identifications of vulnerable plaquesusing RWSmax values and HR-VWI demonstrated strong positive correlations for severe stenosis (r = 0.555, p < 0.001) and in the third group [odds ratio (OR), 4.249; 95% confidence interval (CI), 1.555–11.614; p = 0.005], but not for moderate stenosis (correlation coefficient r = 0.345, p = 0.184). In the subgroupanalysis, RWSmax values were higher for vulnerable than for stable plaques in the severe stenosis (17.7% vs. 12.7%, p < 0.001) and symptomatic (18.0% vs. 10.0%, p = 0.051) groups (Fig. 3B and D). Similar but non-significant trends were observed in the moderate stenosis (15.7% vs. 10.8%, p = 0.068) and asymptomatic (16.9% vs. 13.1%, p = 0.051) groups (Fig. 3A and C).
RWSmax distributions in the moderate vascular stenosis (A), severe vascular stenosis (B), asymptomatic (C), and symptomatic (D) groups.
Diagnostic efficiency
The area under the ROC curve (AUC) was 0.838 (95% CI, 0.740–0.936; p < 0.001; Fig. 4), means that the model has better classification performance. The optimal cutoff point, determined using the maximum Youden index, led to the identification of 14.9% of plaques as vulnerable with 85.5% sensitivity and 74.1% specificity.
ROC curve for predicting vulnerable plaques.
The optimal threshold for predicting lesion progression using RWSmax was14.9%. The analysis yielded an OR of 16.786 (95% CI, 5.361–52.558; p<0.001). An OR of 76.526 (95% CI, 10.871–538.702; p < 0.001) was obtained with an RWSmax of ≥ 14.9%, indicating a significantly elevated risk of plaque vulnerability. AUCs, accuracy, sensitivity, and specificity for subgroups are shown in Fig. 5.
Diagnostic efficacy in various subgroups. AUC, area under the curve; ACC, accuracy; SEN, sensitivity; SPE, specificity.
Relationship of RWSmax to symptomatic extracranial CAS
All participants were categorized into asymptomatic and symptomatic. Demographic characteristics and vascular risk factors did not differ significantly between the symptomatic and asymptomatic groups (p > 0.05; Table 2). Forty-two (85.71%) of 49 symptomatic stenoses were associated with vulnerable plaques, in contrast to 13 (39.39%) of 33 asymptomatic cases (p < 0.001). The mean RWSmax value was significantly higher in the symptomatic than in the asymptomatic group [17.40% (95% CI, 15.00–21.10%) vs. 14.80% (95% CI, 12.00–17.30%); p = 0.008]. Furthermore, a significantly larger proportion of patients in the symptomatic group than those in the asymptomatic group had RWSmax values ≥ 14.9% (77.55% vs. 48.48%, p = 0.006; Table 3). Binary logistic regression analysis showed strong positive associations between the two risk factors (RWSmax and symptomatic carotid stenosis) after correction in the different models (p < 0.01; Table 4).
Discussion
With the increasing global incidence of stroke, the precise prediction and identification of high-risk carotid plaques have become prominent areas of interest in clinical research. High-resolution magnetic resonance imaging (HR-MRI) is among the widely utilized noninvasive imaging techniques for evaluating carotid plaques, demonstrating high sensitivity and specificity in detecting vulnerable carotid plaques based on their composition15. This modality also facilitates the dynamic assessment of atherosclerotic lesion severity. Recently, a machine learning model utilizing computed tomography angiography (CTA) has been developed, which accurately identifies symptomatic carotid plaques. This model introduces the plaque hemorrhage-to-lipid volume ratio and the plaque hemorrhage volume percentage as novel key predictors23. Numerous studies have indicated that artificial intelligence enhances the efficiency and accuracy of identifying vulnerable plaques15,23,24. However, there remains a paucity of research onthe simultaneous AI-based assessment of carotid stenosis and plaque characteristics using digital subtraction angiography (DSA) images.
The inaugural multicenter international registry study in which AI was employed to quantify coronary artery disease demonstrated that lumen diameter stenosis and the non-calcified plaque volume, as assessed by coronary CTA, are significant predictors of major adverse cardiac events and mortality, and that the use of these metrics substantially enhances predictive accuracy for the traditional Diamond-Forrester score and other clinical risk factors25. Tu et al.17 examined correlations of the AI-generated DSA-based RWS with plaque composition and vulnerability characteristics and OCT-defined plaque vulnerability parameters [the lipid-to-cap ratio (LCR) and thin-cap fibroatheroma (TCFA)]. They found that an RWSmax of 12% (range 10–15.0%) correlated positively with the LCR (r = 0.584, p < 0.001) and lipid plaque loading (r = 0.411, p < 0.001) and negatively with fibrous cap thickness, and that RWSmax values > 12% were associated with a higher incidence of TCFA, thinner fibrous caps, and higher LCRs17. In another study, DSA- and OCT-derived RWS values correlated and agreed well (r = 0.91, p < 0 0.001), confirming the high reproducibility of RWS calculation18. The RWS has been shown to predict not only plaque vulnerability, but also the occurrence of composite vasculogenic endpoint events (vascular-related cardiac death and myocardial infarction, and the review endpoint of target-vessel revascularization) within 1 year of hemodialysis in patients with coronary artery disease26. In functional and plaque vulnerability assessment, the use of the RWS has incremental prognostic and predictive value for patients with delayed revascularization and reduces the risk of clinical event (target vessel failure, including cardiac death and target vessel-related myocardial infarction or ischemia-driven revascularization) occurrence20,27,28. Consistent with these findings, we observed significant correlations between RWSmax values and HR-VWI–based assessments of plaque vulnerability in patients with CAS, including those with severe stenosis and asymptomatic and symptomatic presentations, in this study. The risk of plaque vulnerability was 16.786 times greater for RWSmax values ≥ 14.9% than for those < 14.9%, and this increased risk remained significant in multifactorial analyses, consistent with findings from coronary artery studies17. Furthermore, a significantly larger proportion of symptomatic than asymptomatic stenoses was associated with vulnerable plaques, corroborating previous findings29. Larger RWSmax values were also associated with decreased plaque stability, and thus an increased risk of stroke.
We also found in this study that the RWSmax at the lesion site was significantly elevated in patients with symptomatic CAS relative to that in asymptomatic individuals. This finding implies that the prevalence of clinical symptoms is associated with an increased risk of plaque instability.This is consistent with other reported studies30. Consequently, the RWSmax may serve as a predictive marker for the occurrence of clinical ischemic events in patients with CAS. Regarding the threshold value, coronary-artery RWSmax values > 12% have been associated with a 3.49-fold increase in the relative risk of plaque instability17 and have been determined to predict the likelihood of future acute myocardial infarction events compared with lower RWSmax values20. Similarly, lesion-segment RWSmax values > 12.6% were found to effectively predict the progression of stenosis in critical lesions over a 2-year period27. A more recent study showed that RWSmax values > 14% directly impact the incidence of target vessel failure (target vessel-related cardiac death and myocardial infarction and clinically driven target vessel revascularization)28. In the context of carotid stenosis, the optimal RWSmax threshold is 14.9%. This discrepancy may be attributed to the larger diameter of the carotid lumen and the increased likelihood of vulnerable plaque deformation.
This study has several limitations that merit acknowledgment. Firstly, despite the prospective design of this investigation, the generalizability of the findings may be limited due to the relatively small sample size, which resulted from the exclusion of a significant number of subjects. To enhance the robustness and applicability of the results, future clinical trials should involve larger samples from multiple centers. Secondly, the study’s reliance on nuclear high-resolution magnetic resonance imaging (HR-MRI) instead of optical coherence tomography (OCT) or intravascular ultrasound for comparing carotid plaque properties may have introduced some uncertainty. Future research should incorporate additional diagnostic modalities to provide a more comprehensive evaluation of plaque characteristics. Thirdly, the exclusion of certain cases from the real-world scenario (RWS) analysis due to inadequate image quality may have unpredictably influenced the study’s findings. Fourthly, the performance of RWSmax is inevitably influenced by variations in the analyzed angle (resulting from vessel motion and twist), as well as by the angiographic projection and the quality of the angiographic images. Lastly, the absence of follow-up data precluded the assessment of long-term outcomes.
Conclusions
In conclusion, the AI-generated DSA-based RWS may be a valuable indicator of the biomechanical status of CAS plaque lesions. Our preliminary findings indicate that RWS may serve as a valuable tool for assessing and predicting plaque rupture, thereby endorsing its clinical application as a novel method for lesion evaluation.
Data availability
Data is provided within the manuscript.
References
Beylerli, O., Gareev, I., Shi, H. & Ilyasova, T. Effect of mesenchymal stem cell-derived exosomes on the inflammatory response after stroke. Brain Hemorrhages. 5 (5), 248–256 (2024).
Bonati, L. H., Jansen, O., de Borst, G. J. & Brown, M. M. Management of atherosclerotic extracranial carotid artery stenosis. Lancet Neurol. 21 (3), 273–283 (2022).
Wu, S. et al. Stroke in china: advances and challenges in epidemiology, prevention, and management. Lancet Neurol. 18 (4), 394–405 (2019).
Warlow, C., Sudlow, C., Dennis, M., Wardlaw, J. & Sandercock, P. Stroke. Lancet 362 (9391), 1211–1224 (2003).
Bonati, L. H. et al. Lal A, Ricco JB, Ringleb P, Taylor-Rowan M, Eckstein HH.: European Stroke Organisation guideline on endarterectomy and stenting for carotid artery stenosis. Eur. Stroke J. 6 (2), I–XLVII (2021).
Naylor, A. R. et al. (eds) : ‘s Choice - Management of Atherosclerotic Carotid and Vertebral Artery Disease: 2017 Clinical Practice Guidelines of the European Society for Vascular Surgery (ESVS). Eur J Vasc Endovasc Surg 55(1):3–81. (2018).
Eckstein, H. H. et al. Diagnosis, treatment and Follow-up in extracranial carotid stenosis. Dtsch. Arztebl Int. 117 (47), 801–807 (2020).
Thayabaranathan, T. et al. Global stroke statistics 2022. Int. J. Stroke. 17 (9), 946–956 (2022).
van Dam-Nolen, D. H. K. et al. Carotid plaque characteristics predict recurrent ischemic stroke and TIA: the PARISK (Plaque at RISK) study. JACC Cardiovasc. Imaging. 15 (10), 1715–1726 (2022).
Bir, S. C. & Kelley, R. E. Carotid atherosclerotic disease: A systematic review of pathogenesis and management. Brain Circ. 8 (3), 127–136 (2022).
Zhang, R. et al. Prediction of new cerebral ischemic lesion after carotid artery stenting: a high-resolution vessel wall MRI-based radiomics analysis. Eur. Radiol. 33 (6), 4115–4126 (2023).
Zhao, X. et al. Prevalence and characteristics of carotid artery high-risk atherosclerotic plaques in Chinese patients with cerebrovascular symptoms: A Chinese atherosclerosis risk evaluation II study. J. Am. Heart Assoc. 6(8), e005831 (2017).
Bos, D. et al. Atherosclerotic carotid plaque composition and incident stroke and coronary events. J. Am. Coll. Cardiol. 77 (11), 1426–1435 (2021).
Wu, G. et al. Co-existing intracranial and extracranial carotid atherosclerosis predicts large-artery atherosclerosis stroke recurrence: a single-center prospective study utilizing combined head-and-neck vessel wall imaging. Eur. Radiol. 33 (10), 6970–6980 (2023).
Han, N. et al. Imaging and hemodynamic characteristics of vulnerable carotid plaques and artificial intelligence applications in plaque classification and segmentation. Brain Sci. 13(1), 143 (2023).
Leng, X. & Leung, T. W. Collateral flow in intracranial atherosclerotic disease. Transl Stroke Res. 14 (1), 38–52 (2023).
Suo, S. et al. Angiography-based radial wall strain in carotid plaques and its association with plaque vulnerability. EuroIntervention 21 (4), e240–e243 (2025).
Huang, J. et al. Radial wall strain assessment from AI-Assisted angiography: feasibility and agreement with OCT as reference standard. J. Soc. Cardiovasc. Angiogr Interv. 2 (2), 100570 (2023).
Huang, J. et al. Optical coherence Tomography-Derived changes in plaque structural stress over the cardiac cycle: A new method for plaque Biomechanical assessment. Front. Cardiovasc. Med. 8, 715995 (2021).
Wang, Z. Q. et al. Angiography-derived radial wall strain predicts coronary lesion progression in non-culprit intermediate stenosis. J. Geriatr. Cardiol. 19 (12), 937–948 (2022).
Cai, J. M. et al. Classification of human carotid atherosclerotic lesions with in vivo multicontrast magnetic resonance imaging. Circulation 106 (11), 1368–1373 (2002).
Barnett, H. J. et al. Benefit of carotid endarterectomy in patients with symptomatic moderate or severe stenosis. North American symptomatic carotid endarterectomy trial collaborators. N Engl. J. Med. 339 (20), 1415–1425 (1998).
Pisu, F. et al. Machine learning detects symptomatic plaques in patients with carotid atherosclerosis on CT angiography. Circ. Cardiovasc. Imaging. 17 (6), e016274 (2024).
Scicolone, R. et al. Radiomics and artificial intelligence: general notions and applications in the carotid vulnerable plaque. Eur. J. Radiol. 176, 111497 (2024).
van Rosendael, A. R. et al. Rationale and design of the CONFIRM2 (Quantitative coronary CT angiography evaluation for evaluation of clinical outcomes: an international, multicenter Registry) study. J. Cardiovasc. Comput. Tomogr. 18 (1), 11–17 (2024).
Tu, S. et al. Short-Term risk stratification of Non-Flow-Limiting coronary stenosis by angiographically derived radial wall strain. J. Am. Coll. Cardiol. 81 (8), 756–767 (2023).
Li, C. et al. The association between angiographically derived radial wall strain and the risk of acute myocardial infarction. JACC Cardiovasc. Interv. 16 (9), 1039–1049 (2023).
Yang, S. et al. Relationship of coronary Angiography-Derived radial wall strain with functional significance, plaque morphology, and clinical outcomes. JACC Cardiovasc. Interv. 17 (1), 46–56 (2024).
Kopczak, A. et al. Complicated carotid artery plaques and risk of recurrent ischemic stroke or TIA. J. Am. Coll. Cardiol. 79 (22), 2189–2199 (2022).
Huang, Z. et al. Incremental prognostic value of Carotid Plaque-RADS over stenosis degree in relation to stroke risk. JACC Cardiovasc. Imaging 18(1), 77–89 (2025).
Acknowledgements
We thank Medjaden Inc. for the scientific editing of this manuscript.
Funding
This research was funded by the Inner Mongolia Autonomous Region Science and Technology Program under the Key Research and Development and Achievement Transformation Program (grant no. 2022YFSH0131) and the Key Technology Program (grant no. 2020SGG0480), Key Project of Science and Technology of Public Hospital Joint Research Fund(2024GLLH1329), Innovation Startup Fund for Returned Overseas Students in Inner Mongolia Autonomous Region, Ordos Science and Technology Innovation Leading Talents, Ordos City Industrial Innovation Talent Team and the 2024 Baotou Medical College Research and Innovation Program (grant no. BYKYCX202419).
Author information
Authors and Affiliations
Contributions
Yingchun Wu wrote the main manuscript text and prepared all figures. Ludi Fu, Wen Liu, Rihan Wu, Shu Tang, Zhixiang Wang, Jiajia Han, Yitai Liu and Xueyang Li collected data. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wu, Y., Fu, L., Liu, W. et al. Carotid atherosclerotic plaque vulnerability assessment from angiography-derived radial wall strain validated by MRI. Sci Rep 15, 34972 (2025). https://doi.org/10.1038/s41598-025-18916-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-18916-9