Abstract
We aimed to conduct a systematic analysis of the multi-target regulatory mechanisms of Ziyu glycoside I (Ziyu I) in cervical cancer progression using network pharmacology and molecular docking techniques. Additionally, we validated our findings using cellular experiments and animal models. The PhamMapper database was used to identify drug targets for Ziyu I, while cervical cancer-related disease targets were obtained from the DisGeNET, DrugBank, GeneCards, and OMIM databases. In vitro, the effects of Ziyu I on the proliferation, migration, apoptosis, and cell cycle were assessed using cultured HeLa and SiHa cervical cancer cells. Network pharmacology analysis identified 68 potential targets of Ziyu I in cervical cancer. Molecular docking findings revealed that Ziyu I had a stable binding ability to MAPK1, MAPK8, and MAPK14, which are key targets in the MAPK signaling pathway. Cellular experiments revealed that Ziyu I significantly inhibited the proliferation and migration of cervical cancer cells and promoted apoptosis and cell cycle arrest. Finally, a nude mice-loaded tumor assay revealed that Ziyu I had significant anti-cervical cancer activity in vivo. Ziyu I inhibits cervical cancer progression through a multitarget regulatory network involving MAPK signaling pathway-mediated inhibition of cell proliferation and migration, apoptosis activation, and cell cycle arrest.
Similar content being viewed by others
Introduction
Globally, cervical cancer has the fourth highest incidence and mortality rates among female malignancies. The disease burden is particularly high in low- and middle-income countries, where it is second to breast cancer in both incidence and mortality1. Human papillomavirus (HPV) infection, particularly high-risk HPV 16/18, is the main causative factor of cervical cancer2. Despite an increase in cervical cytological screening and HPV prophylactic vaccination coverage3,4, cervical cancer continues to pose a major public health threat. According to the World Health Organization’s Global Cancer Statistics Report 20231, approximately 623,000 new cases of cervical cancer and 341,000 mortality cases occur annually, with 89.2% of the disease burden concentrated in low- and middle-income countries. Currently, surgery, radiotherapy, chemotherapy, immunotherapy, and targeted therapy are primarily used to treat cervical cancer in clinical practice5,6, and these methods reduce mortality to some extent. However, despite the effectiveness of existing treatments, a significant proportion of patients still experience treatment-related adverse events, and some develop resistance7,8. Therefore, developing highly effective and low-toxicity drugs is urgently needed to overcome the limitations of existing treatments.
Chinese medicines have played an important role in disease treatment, and evidence has shown that their active ingredients exhibit significant antitumor effects9,10. Ziyu glycoside I (Ziyu I) is one of the main active components of Sanguisorba officinalis L. and possesses various pharmacological effects, including anti-inflammatory, antioxidant, antiviral, and antitumor effects11,12. Studies have shown that Ziyu I inhibits the proliferation of various tumors, including breast cancer, human retinoblastoma, and other malignant tumors13,14. A related study revealed that Ziyu I induced G2/M-phase blockade and endogenous/exogenous apoptosis in MDA-MB-231 breast cancer cells through the upregulation of p5313. However, to date, no studies have reported the role of Ziyu I in cervical cancer, and its specific anticancer mechanism has not yet been elucidated.
The concept of network pharmacology, first proposed by Hopkins in 2007, combines theories and methods from several disciplines, including bioinformatics, systems biology, pharmacology, and computer science, and has been widely used in drug discovery and disease mechanism research15. Our study aimed to investigate the potential molecular mechanisms of Ziyu I in cervical cancer using network pharmacology and molecular docking techniques to identify core targets and regulatory pathways. The antitumor activity of Ziyu I and its molecular mechanisms, including important biological processes, such as cell cycle regulation and apoptotic pathway activation, were further validated in a tumor-bearing mice model and cervical cancer cell lines through systematic in vivo and ex vivo pharmacodynamic experiments. The present study not only reveals the multi-targeting characteristics of Ziyu I but also provides an innovative drug candidate for combination therapy of cervical cancer.
Results
Ziyu glycoside I and the acquisition of cervical cancer targets
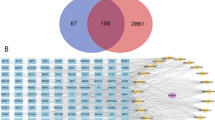
The molecular formula of Ziyu I was obtained from the TCMSP database (Fig. 1A). Eighty-eight potential targets of action were identified using PharmMapper. Subsequently, 215, 83, 5,267, and 168 targets related to cervical cancer were obtained from the DisGeNET, DrugBank, GeneCards, and OMIM databases, respectively. After integrating data from these four databases and removing duplicates, 5,431 cervical cancer-related targets were identified.
Network pharmacology analysis: (A) chemical structure of Ziyuglycoside I (Ziyu I), (B) Venn diagram of intersection of drug targets and disease targets, (C,D) Protein-protein interaction (PPI) network diagram, (E,F) screening of core targets, (G) Gene ontology (GO) enrichment analysis, and (H) Kyoto encyclopedia of genes and genomes (KEGG) pathway enrichment analysis.
Construction of protein-protein interaction network
The targets of action of Ziyu I were compared with those related to cervical cancer, identifying 68 cross-targets (Fig. 1B). The 68 cross-targets of plumbagin and cervical cancer were imported into the STRING database for analysis, and a protein-protein interaction (PPI) network of cross-targets between Ziyu I and cervical cancer was constructed (Fig. 1C). The network comprised 68 nodes and 321 edges. The nodes represent cross-targets and the edges indicate the interaction relationships between these targets. Nodes were ranked according to the degree value, with higher-degree nodes resulting in larger shapes and darker colors (Fig. 1D). Subsequently, initial screening of core targets was performed using three built-in plug-ins: cytoNCA, MCODE, and cytoHubba. The three groups of genes obtained intersected, and ESR1, EGFR, HSP90AA1, CASP3, SRC, ANXA5, MAPK1, MAPK8, and MAPK14 were identified as core targets (Fig. 1E,F).
Gene ontology and Kyoto encyclopedia of genes and genomes enrichment analysis
This study performed gene ontology (GO) and Kyoto encyclopedia of genes and genomes (KEGG) co-enrichment analyses of 68 drug-disease intersection targets using a bioinformatics platform to systematically analyze the antitumor mechanism of Ziyu I against cervical cancer. As illustrated in Fig. 1G, the GO functional clustering results indicated significant enrichment of biological processes (BP) in oxidation-related pathways, including the chemical stress response and reactive oxygen metabolism. Cellular components (CC) were primarily localized in critical signal transduction areas, such as the cytoplasmic vesicle lumen and membrane raft structure. Molecular functions (MF) analysis exhibited strong associations with steroid hormone receptor binding, serine/threonine kinase activity, and NADP oxidoreductase activity. KEGG pathway analysis indicated that the core targets were significantly enriched in the MAPK signaling pathway, regulation of reactive oxygen species, and apoptosis signaling pathway (Fig. 1H).
Molecular Docking and visualization
The binding ability of Ziyu I to core targets within the MAPK signaling pathway was assessed using the molecular docking technique. As shown in Fig. 2, the binding energies of Ziyu Saponin I with MAPK1 (ERK), MAPK8 (JNK), and MAPK14 (p38) were − 5.18 kcal/mol, -5.17 kcal/mol, and − 5.13 kcal/mol, respectively. The inhibition constants were 159.82 µM, 163.16 µM, and 172.76 µM respectively. These negative binding energies and µM-level inhibitory constants suggest that Ziyu I may regulate the activity of the MAPK pathway through its spontaneous binding effect.
Molecular docking: (A–C) Molecular docking maps of Ziyuglycoside I (Ziyu I) and MAPK1 (ERK), MAPK8 (JNK), and Ziyu I and MAPK14 (p38) proteins.
Ziyu glycoside I inhibits cervical cancer cell proliferation
To systematically evaluate the inhibitory effects of Ziyu I on the growth of cervical cancer cells, this study employed various experimental approaches. The results of the proliferation assay of CCK-8 cells revealed a concentration-dependent and significant decrease in cell viability after 24 h of treatment with Ziyu I (Fig. 3A). A concentration of 15 µM Ziyu I exhibited a significant inhibitory effect on cervical cancer cells and was therefore selected for the subsequent experiments. Inverted phase-contrast microscopy revealed that the cells in the drug-treated group exhibited typical morphological changes of apoptosis, including retraction of cell pseudopods, cytosolic crumpling, and disintegration of intercellular junctions (Fig. 3B). The plate clone formation assay further confirmed that the number of clones significantly reduced in a time-dependent manner after Ziyu I treatment (Fig. 3C). Western blot analysis revealed that drug treatment significantly downregulated the expression of the proliferation marker protein—proliferating cell nuclear antigen (Fig. 3D).
Ziyuglycoside I (Ziyu I) inhibits the proliferation of cervical cancer cells: (A) Cervical cancer HeLa and SiHa cells treated with different concentrations of Ziyu I for 24 h were examined for cell viability by CCK-8 assay. (B) After 24 h of Ziyu I treatment, morphological changes of HeLa and SiHa cells were observed and recorded, and typical images were acquired (200×). (C) Plate cloning assay was performed to evaluate the effect of Ziyu I treatment on the clonogenic ability of HeLa and SiHa cells. (D) Proliferating cell nuclear antigen (PCNA) protein expression levels were measured in HeLa and SiHa cells treated with Ziyu I for 24 h and 48 h.
Ziyu glycoside I inhibits cervical cancer cell migration
The regulatory effect of Ziyu I on the migration ability of cervical cancer cells was systematically evaluated by integrating a scratch healing assay, transwell migration analysis, and epithelial-mesenchymal transition (EMT) marker assay. Exposure to Ziyu I at 15 µM and 20 µM for 12–48 h resulted in concentration- and time-dependent suppression of scratch wound healing capacity in both HeLa and SiHa cells (Fig. 4A). Similarly, the number of cells penetrating through the 8 μm pore size polycarbonate membrane in the treated group was significantly lower than that of the control group as confirmed by Transwell migration analysis (Fig. 4B). Western blot analysis revealed that Ziyu I treatment significantly regulated the expression of EMT markers, upregulated the expression of the epithelial marker, E-cadherin, and downregulated that of the mesenchymal marker N-cadherin (Fig. 4C). These results indicate that Ziyu I effectively inhibited the migration of cervical cancer cells by reversing the EMT process.
Inhibition of cervical cancer cell migration by Ziyuglycoside I (Ziyu I): (A) Scratch assay to evaluate the effect of Ziyu I treatment on the migration ability of HeLa and SiHa cells, and photographs were taken at 0 h, 12 h, 24 h–0 h, 24 h, 48 h to record the area of cell migration (100×). (B) Transwell assay: Transwell chambers were used to determine the number of migrating cells in HeLa and SiHa cells after Ziyu I treatment (100×). (C) Western blot analysis of E-cadherin and N-cadherin protein expression levels in HeLa and SiHa cells treated with Ziyu I for 24 h and 48 h.
Ziyu glycoside I induces apoptosis in cervical cancer cells
To investigate the induction of apoptosis in cervical cancer cells by Ziyu I, the proportion of apoptotic cells was analyzed using Annexin V-fluorescein isothiocyanate (Annexin V-FITC) and propidium iodide (PI) double staining. Ziyu I treatment triggered time-dependent apoptotic responses in both cervical cancer cell lines, with HeLa cells exhibiting apoptosis rate escalation from 10.30% ± 0.85% to 17.89% ± 1.11% (24 h) and 23.86% ± 0.17% (48 h) and SiHa cells showing increases from 2.77% ± 0.41% to 8.28% ± 0.20% (24 h) and 16.65% ± 0.87% (48 h) (Fig. 5A). Western blotting analysis revealed that the expression levels of pro-apoptotic proteins p53, cleaved PARP1, and cleaved Caspase3 were significantly upregulated in the experimental group, while the expression of anti-apoptotic protein Bcl-2 revealed a significant downregulation trend (Fig. 5B).
Ziyuglycoside I (Ziyu I) induced apoptosis in cervical cancer cells: (A) Apoptotic cells were detected by flow cytometry combined with Annexin V-fluorescein isothiocyanate/propidium iodide (Annexin V-FITC/PI) staining in HeLa and SiHa cells treated with Ziyu I, and the data were analyzed using Flowjo software. (B) The expression levels of apoptosis-related proteins p53, Bcl-2, Cleaved Caspase3, and Cleaved PARP1 were detected by Western blot.
Ziyu glycoside I induces cell cycle arrest in cervical cancer
To investigate whether Ziyu I exerts antiproliferative effects by blocking the cell cycle, we examined the cell cycle distribution characteristics of HeLa and SiHa cells in the drug-treated and control groups by flow cytometry. The experimental results revealed that the proportion of HeLa and SiHa cells in the G2/M phase significantly increased after 24 h and 48 h of treatment with Ziyu I (Fig. 6A). Specifically, the proportion of HeLa cells in the G2/M phase increased from 10.33 ± 1.00% to 15.32 ± 0.34% and 19.88 ± 0.33%, respectively, whereas that of SiHa cells increased from 14.61 ± 1.28% to 24.43 ± 0.30% and 20.05 ± 0.06%, respectively. Accompanying the phenomenon of G2/M phase blockage, the corresponding proportion of cells in the G1 phase decreased significantly in both groups, from 73.69 ± 0.32% to 69.03 ± 0.33% and 64.64 ± 0.25% for HeLa cells and from 75.67 ± 1.56% to 65.81 ± 0.46% and 59.19 ± 0.38% for SiHa cells. Notably, while the proportion of HeLa cells in the S phase remained stable before and after drug treatment, the proportion of SiHa cells in the S phase exhibited a significant increase following a 48-h drug exposure. Western blot analysis revealed a significant downregulation in the expression levels of the key cell cycle regulatory proteins, Cyclin A2, and CDK1 (Fig. 6B).
Ziyuglycoside I (Ziyu I) induced cervical cancer cell cycle arrest: (A) HeLa and SiHa cells treated with 15 µM Ziyu I were subjected to cell cycle determination by flow cytometry and cell distribution in G0/G1, S, and G2/M phases was quantified using ModFit software. (B) The expression levels of cell cycle-related proteins Cyclin A2 and CDK1 were detected by Western blot.
Ziyu glycoside I exert anti-cervical cancer effects through the MAPK signaling pathway
Based on the results of the KEGG enrichment analysis, we initially hypothesized that Ziyu I exerts its anti-cervical cancer effects through the MAPK signaling pathway. To verify this hypothesis, we conducted western blotting experiments to detect changes in the expression levels and phosphorylation status of the key proteins—ERK, JNK, and p38—in the cervical cancer cell lines—HeLa and SiHa—after Ziyu I treatment. The results (Fig. 7) revealed that after Ziyu I treatment, the ratios of the phosphorylated forms of ERK and p38 to the total proteins were significantly upregulated, while the phosphorylation level of JNK was significantly decreased. These results indicate that Ziyu I may exert its inhibitory effect on cervical cancer by regulating the phosphorylation of key proteins in the MAPK pathway.
Effects of Ziyuglycoside I (Ziyu I) on cervical cancer cell function through the MAPK signaling pathway: The expression levels of ERK, JNK, and P38 proteins and their phosphorylation status in the MAPK signaling pathway were detected by Western blot in HeLa and SiHa cells treated with Ziyu I.
Ziyu glycoside I inhibits tumor growth in tumor-bearing mice
To systematically evaluate the in vivo antitumor effect of Ziyu I, a cervical cancer transplantation tumor model was constructed using HeLa cells. Compared to the blank control group, tumor growth was significantly inhibited in the Ziyu I administration group, suggesting that the compound has significant antiproliferative activity against cervical cancer (Fig. 8A–D). During the pharmacodynamic evaluation, the body weight fluctuations of the mice in the experimental groups were maintained within the normal physiological range, indicating that Ziyu I did not induce significant toxic side effects at the therapeutic dose (Fig. 8E, F).
Inhibition of HeLa tumor growth by Ziyuglycoside I (Ziyu I) in vivo: (A,B) 5 × 106 HeLa cells were subcutaneously injected into mice to form tumor xenografts and administered by intraperitoneal injection of 2 mg/kg Ziyu I every other day for 20 days. At the end of the experiment, mouse tumor images were acquired. (C) The tumor weights of the two groups of mice were compared at the end of the experiment. (D) Mouse body weight (g) was measured every 3 days for 20 days. (E) Tumor volume (mm2) was measured every 3 days for 20 days. (F) To evaluate the toxic and side effects of Ziyu I, the structural changes of mouse liver, kidney and tumor tissues were observed by hematoxylin-eosin (HE) staining (200×). (G) To evaluate the inhibitory effect of Ziyu I on HeLa tumors, the expression levels of Ki-67 in tumor tissues of the two groups of mice were detected by immunohistochemistry (IHC) (400×).
To further evaluate the potential toxicity of Ziyu I in mice at therapeutic doses, we performed HE staining analysis on the liver and kidney tissues of mice in the control and Ziyu I administration groups. The results showed no significant abnormalities in the liver and kidney histology of both groups of mice (Fig. 8G). Specifically, the liver lobules were structurally intact, connective tissue between lobules was evenly distributed, central and portal vein branches were arranged in an orderly manner, and hepatic cords showed a typical radial arrangement. The kidney glomeruli were structurally intact, and the epithelial cells of the renal tubules were arranged in an orderly manner, the interstitium had no inflammatory reaction, and the vascular morphology was normal. In addition, to further investigate the effect of Ziyu I on the tumors, we performed HE staining and Ki-67 immunohistochemical analysis on the tumor tissues of the two groups of mice. The HE staining results (Fig. 8G) showed that the tumor cells of the control group were densely packed, with varying sizes of cells, enlarged nucleoplasmic ratios, deep staining of the nuclei, and pathologic karyorrhexis. In contrast, the tumor tissues of the mice in the Ziyu I administration group were characterized by patchy necrotic areas, disorganized cell arrangement, contour disappearance, loss of cell outline, homogeneous red staining of the cytoplasm, and nuclear consolidation or lysis. Further, the expression level of Ki-67 in the tumor tissues of Ziyu I-dosed mice was significantly decreased (Fig. 8H).
Discussion
Cervical cancer, a common malignant tumor of the female reproductive system, is pathologically categorized into squamous cell carcinoma and adenocarcinoma of the cervix. Squamous cell carcinoma accounts for approximately 70%–80% of cervical cancer cases, with high morbidity and mortality16. Although radical surgery combined with radiotherapy can effectively control early-stage lesions, the 5-year survival rate for advanced or recurrent metastatic cervical cancer remains less than 17%17,18. In addition, despite systemic chemotherapy prolonging median survival, severe treatment-related toxicities, such as significant myelosuppression, often arise and ultimately lead to the development of a multidrug-resistant state19,20,21,22. Therefore, the development of novel therapeutic strategies has become a key scientific issue in gynecological oncology. Ziyu I is a type of triterpenoid saponin natural compound. The antitumor effects of Ziyu I are less studied than those of other similar compounds, such as Astragaloside IV23 and Saikosaponin D24. However, Astragaloside IV has been reported to have a relatively weak inhibitory activity against cervical cancer23, with its IC50 remaining at the mM level in the in vitro CCK-8 experiment. In animal experiments, Astragaloside IV was administered at doses of 12.5, 25, and 50 mg/kg for evaluation, with only the 25 mg/kg dose group showing a significant inhibitory effect on tumor volume. In contrast, Saikosaponin D exerts stronger anti-cervical cancer activity, but multiple studies25,26 have pointed out that it may cause significant neurotoxicity and hepatotoxicity, limiting its further clinical application.
In this study, 68 potential targets of Ziyu I in cervical cancer were identified through network pharmacology analysis using PharmMapper and other databases. These targets are involved in multiple biological processes and signaling pathways, suggesting that they possess the pharmacological characteristics of multi-target synergistic regulation. PPI network analysis identified 9 core targets, including ESR1, EGFR, HSP90AA1, and CASP3, as key mediators of Ziyu I treatment of cervical cancer. Mutations in the ligand-binding domain of ESR1 mediate endocrine therapy resistance in breast cancer by inducing constitutive activation of estrogen receptor alpha27. Clinical studies have confirmed that this mutation is significantly associated with accelerated tumor progression, as well as an increased risk of distant metastasis28,29. High EGFR expression is closely associated with tumorigenesis, development, and progression. Studies have shown that EGFR is significantly overexpressed in various solid tumors, including cervical cancer, esophageal cancer, and glioblastoma30. Currently, targeted therapies against EGFR have been widely studied and clinically applied31. Similarly, high expression of HSP90AA1 is significantly associated with poor prognosis in patients with hepatocellular carcinoma (HCC), whereas knockdown of HSP90AA1 not only significantly inhibits the proliferative viability of HCC cells but also enhances their sensitivity to chemotherapeutic agents32. CASP3, a core effector molecule in the execution phase of apoptosis, activates an irreversible caspase cascade by specifically cleaving PARP and other key substrates, ultimately leading to programmed cell death.
GO and KEGG33 enrichment analyses were further performed on the common targets of Ziyu I and cervical cancer. GO enrichment analysis revealed that the potential targets of Ziyu I were significantly enriched in the subcellular structural domains of the membrane lipid rafts, secretory granule lumen, and cytoplasmic vesicle compartments. Molecular functional annotation identified nuclear receptor coactivator, serine/threonine protein kinase, and oxidoreductase activities as core mechanisms of action. Biological process analysis further revealed that Ziyu I exerts its therapeutic effects by regulating the chemical stress response pathway and reactive oxygen species’ metabolic homeostasis. KEGG pathway enrichment results confirmed that Ziyu I exerts its anti-tumor effects mainly through the MAPK signaling pathway and apoptosis signaling network, which is a highly conserved signaling pathway in the cell and plays a major role in the regulation of biological processes, such as proliferation, differentiation, and apoptosis. The MAPK signaling pathway is a highly conserved signal transduction pathway in cells and plays a key role in regulating cell proliferation, differentiation, and apoptosis. ERK, JNK, and p38 are the three core members of the MAPK family, and their aberrant signaling activation has been shown to be closely related to the development of various malignant tumors34. In the apoptosis regulatory network, the regulation of ERK/JNK/p38 signaling can effectively stabilize the expression level of p53 protein, significantly enhance its transcriptional activation, upregulate the expression of the pro-apoptotic factor Bax, antagonize the anti-apoptotic function of Bcl-2, and ultimately mediate programmed cell death through mitochondrial pathway-dependent caspase cascade activation35,36,37. Several pharmacological studies have confirmed that small-molecule compounds derived from traditional Chinese medicines can effectively intervene in the malignant phenotype of cancer cells by modulating the ERK/JNK/p38 signaling network. Rotundifuran specifically activates the JNK/p38 MAPK signaling axis by inducing elevated levels of reactive oxygen species in HeLa and SiHa cervical cancer cells and significantly inhibiting ERK1/2 phosphorylation. This dual regulatory effect results in an elevated Bax/Bcl-2 protein ratio, triggers cytochrome c release from the mitochondria, and activates the caspase-9/3 cascade, ultimately inducing mitochondria-dependent apoptosis38. Similarly, curcumin significantly inhibited the phosphorylation and activation of the ERK and JNK signaling pathways by upregulating TFPI-2 gene expression, leading to the blockage of nuclear translocation of Snail1, a key regulator of EMT. This was accompanied by the upregulation of E-cadherin protein expression and downregulation of N-cadherin/vimentin expression, which ultimately resulted in a significant reduction in the migratory invasion ability of pancreatic cancer cells39. We selected key proteins in the MAPK pathway, ERK, JNK, and p38, for molecular docking studies. The findings revealed that the binding energy of Ziyu I to these proteins was low, which explains the mechanism of Ziyu I’s anti-cervical cancer action at the molecular level, which may be exerted by acting on the above-mentioned cores.
Based on the above predictive analyses, this study systematically verified the anti-cervical cancer effects of Ziyu I through ex vivo experiments using cervical cancer HeLa and SiHa cells, as well as HeLa cell-loaded mice as models. The effects of Ziyu I on the morphology of cervical cancer cells were observed using an inverted microscope, and the effects of Ziyu I on inhibiting cell proliferation and migration and inducing apoptosis and cycle arrest were comprehensively evaluated via the CCK-8 assay, clone formation assay, scratch assay, transwell migration assay, and flow cytometry. Protein expression changes in relation to the above-mentioned cellular phenotypes were further verified using western blotting. The findings revealed the changes in protein expression associated with these cellular phenotypes. The study findings further revealed that the phosphorylation levels of ERK and p38 proteins were significantly increased, and the phosphorylation level of JNK was reduced, suggesting that Ziyu I may regulate the MAPK signaling pathway to exert anti-cervical cancer effects. In addition, a nude mouse tumor assay revealed that Ziyu I also had significant anti-cervical cancer activity in vivo.
In conclusion, this study is the first to reveal the multi-target action of Ziyu I against cervical cancer by integrating network pharmacological prediction and molecular docking technology, suggesting that its mechanism may involve the synergistic regulation of the MAPK signaling pathway. In vitro experiments revealed that Ziyu I inhibits the proliferation and migration of cervical cancer cells in a concentration- and time-dependent manner, induces apoptosis, and exerts antitumor effects by blocking cell cycle progression. In vivo, Ziyu I significantly inhibited tumor growth in a nude mouse transplantation model, and no significant toxic side effects were observed. These results provide mechanistic insights into the role of Ziyu I in modulating cervical cancer progression via the MAPK signaling pathway, thereby establishing a robust pharmacological basis for its potential clinical translation.
Finally, this study has certain methodological limitations that warrant further investigation. To address these limitations and obtain more definitive evidence, we plan to implement a comprehensive experimental strategy, including cell-based overexpression and CRISPR/Cas9-mediated gene knockout approaches, in conjunction with cell thermal shift assays and small-molecule pull-down assays. These experiments will provide direct evidence for the interaction between Ziyu I and key MAPK signaling proteins.
Methods
Ziyu I and cervical cancer target screening
The molecular structure of Ziyu I was retrieved by searching for “Ziyuglycoside I” in the TCMSP database (https://old.tcmsp-e.com/tcmsp.php) and downloading the resulting Mol2 file. This file was imported into the PhamMapper database (https://lilab-ecust.cn/pharmmapper/submitfile.html) for target prediction. The identified targets were subsequently normalized. The corresponding gene names were obtained using the UniProt database (https://www.uniprot.org/). Meanwhile, “Cervical cancer” was used as the keyword in the DisGeNET database (https://disgenet.org/) and DrugBank database (https://go.drugbank.com/), as well as in the GeneCards database (https://www.genecards.org/) and OMIM database (https://www.omim.org/) to search for cervical cancer-related targets. In the GeneCards database, the median relevance score of 3.766 was used as the criterion to further screen out the highly relevant targets. The collected targets were then imported into the UniProt database for gene name normalization and duplicate removal to construct the final cervical cancer target dataset.
Construction of protein-protein interaction networks
Venn diagram analysis was performed using the Jvenn online analysis platform (https://www.bioinformatics.com.cn/static/others/jvenn/) to identify the overlapping genes of Ziyu I and cervical cancer disease targets. The obtained co-interacting targets were imported into the STRING database (https://string-db.org/), with the species parameter set to ‘Homo sapiens,’ a minimum PPI confidence threshold of 0.4, and the free nodes excluded. The PPI data were exported in TSV format and subsequently imported into Cytoscape 3.9.1 software for further analysis. Targets were ranked according to the node degree value, followed by a preliminary screening of core targets using three topological analysis algorithms, CytoHubba, cytoNCA, and MCODE, to obtain the candidate core gene sets. The three obtained gene sets were analyzed using a Venn diagram to identify common core target groups.
Gene ontology and Kyoto encyclopedia of genes and genomes enrichment analysis
Multidimensional functional annotation of the intersecting targets was performed using an online analysis platform (http://www.bioinformatics.com.cn/). A hypergeometric distribution test was used to conduct GO functional enrichment analysis, covering the three major categories of BP, MF, and CC. Simultaneously, the KEGG pathway enrichment analysis was conducted to identify relevant signaling pathways.
Molecular docking and visualization
Molecular docking simulations were performed using MAPK signaling pathway core proteins MAPK1 (ERK), MAPK8 (JNK), and MAPK14 (p38). The standard structure of Ziyu I (MOL006557) as a ligand molecule was obtained from the TCMSP database. The receptor protein crystal structure (PDB ID: 6SLG, 2XRW, 2FST) was downloaded from the RCSB PDB database (https://www.rcsb.org/) and the protein structure optimized using the PyMOL software, including the removal of crystalline water molecules and non-associated ligands. Receptor supplementation with hydrogen atoms was performed using AutoDockTools software and saved in pdbqt format. Final molecular docking simulations were performed using AutoDock Vina to calculate the binding energy between the ligand and the receptor, and a ligand-receptor 3D interaction map was generated using PyMOL.
Cell lines and drugs
Human cervical cancer cell lines, HeLa and SiHa, were purchased from Baidi Biotechnology (China) and cultured in vitro in Dulbecco’s Modified Eagle Medium (DMEM) (Gibco, USA) supplemented with 10% fetal bovine serum (FBS) (ExCell Bio, China) and 1% penicillin-streptomycin mixture (Solarbio, China). Cell culture conditions were set at 37 °C and 5% CO₂ saturated humid environment. Ziyu I (Purity: 99.42%) was purchased from MedChemExpress, USA, and a 50 mM stock solution was prepared using dimethyl sulfoxide, dispensed and stored at -80 °C for long-term use. Before experiments, the stock solution was diluted to the desired working concentration using the complete medium.
Cell viability assay
Cervical cancer cells in the logarithmic growth phase were harvested and seeded into 96-well culture plates containing 100 µL of complete media at a concentration of 5 × 104 cells/mL, followed by incubation at 37 °C overnight to ensure full adherence to the plate surface. Fresh complete medium containing increasing concentration of Ziyu I was then introduced individually, and incubation was extended for 24 h. After removal of the original medium, 100 µL of complete media supplemented with 10% CCK-8 reagent (APExBIO, USA) was introduced to each well, and incubation was maintained for 2 h. The absorbance value (OD value) of each well was measured at 450 nm using a Tecan Infinite 200 Pro Multifunctional Enzyme Labeller, and cell viability was calculated according to the following formula: viability = (OD experimental group - OD blank) / (OD control group - OD blank) × 100%.
Clone formation experiments
Cervical cancer cells in the logarithmic growth phase were seeded into 6-well plates at a density of 500 cells per well, with each well supplied with 2 mL of complete media containing 10% FBS, and grown at 37 °C with 5% CO₂ for 12 h to ensure full adherence to the surface. After treatment with 15 µM Ziyu I for 24–48 h, cells were replenished with fresh complete medium and cultured for 14 days to evaluate colony-forming ability. Upon completion of growth, the cells were progressively washed with pre-cooled phosphate buffered serum (PBS), fixed in 4% paraformaldehyde for 30 min at ambient temperature, and stained with 1% crystal violet for 15 min. After cleaning with distilled water and air-drying, the cells were enumerated using the ImageJ software.
Scratch experiment
Cell migration was assessed using a cell scratch assay system. A cervical cancer cell suspension at a concentration of 5 × 105 cells/mL was first inoculated at 80 µL/well in cell culture inserts (IBIDI, Germany) and incubated overnight in a constant temperature incubator to form a dense monolayer of cells. Images were recorded using an inverted microscope (Zeiss Axio Vert A1). Subsequently, the medium was replaced with a fresh medium containing 15 µM or 20 µM Ziyu I in 1% FBS, and the same field of view was imaged at 12 h, 24 h, and 48 h post-treatment. The change in scratch area was measured, and the cell migration rate was calculated using the formula: Cell migration rate = [1 - (end-point scratch area / initial scratch area)] × 100%.
Transwell migration experiments
Cervical cancer cells that were starved for 12 h, were resuspended in serum-free DMEM at a density of 2 × 105 cells/mL, and 200 µL of the suspension was added to the upper chamber of a Transwell (Corning, 3422). The lower chamber was injected with 700 µL of chemotaxis medium containing 20% FBS. After 24 h of incubation at a constant temperature, the cells were fixed with 4% paraformaldehyde for 15 min at room temperature, and the non-migrating cells in the upper chamber were mechanically removed using cotton swabs. Migrated cells in the lower chamber were stained with 0.1% crystal violet for 20 min, gently rinsed with distilled water, and air-dried at room temperature. Five randomly selected fields of view were photographed using an inverted microscope and counted using ImageJ software.
Annexin V-fluorescein isothiocyanate/propidium iodide staining assay
Apoptosis was quantified using Annexin V-FITC/PI double staining. Cervical cancer cells in the logarithmic growth phase were inoculated into 6-well plates at a density of 5 × 105 cells/well, cultured overnight to allow adherence to the wall, and then treated with 15 µM Ziyu I for either 24–48 h. Cells were collected and washed twice with precooled PBS before being resuspended in 500 µL of 10× Binding Buffer. Then, 5 µL of Annexin V-FITC and PI staining solution (BD Biosciences) were sequentially added, and the cells were incubated for 15 min away from light. Cell signals were acquired using a Thermo Fisher Attune NxT flow cytometer, and the proportion of apoptotic cells was analyzed using FlowJo v10.8.1 software.
Cell-cycle analysis
PI-stained cervical cancer cells in the logarithmic growth phase were seeded at a density of 5 × 105 cells/well in 6-well plates. After overnight incubation to allow adherence to the wall, Ziyu I was added and the cells were treated for either 24–48 h. Cells were collected and washed with pre-cooled PBS. Anhydrous ethanol was slowly added to a final concentration of 70% and fixed at 4 °C overnight. After centrifugation to remove the fixative, cells were resuspended in PBS and hydrated for 15 min at room temperature. Subsequently, 1 mL of PI staining working solution (LIANKE Bio) was added, and the cells were incubated for 30 min in the dark. Cell signals were collected using a flow cytometer and analyzed for cell cycle phase fitting using ModFit LT 5.0.
Western blot analysis
Western blotting technique was performed to detect the expression levels of target proteins. Treated cells were collected and lysed on ice for 30 min using pre-cooled radioimmunoprecipitation assay lysis buffer containing 2% protease and phosphatase inhibitors, and the supernatant was collected by centrifugation. Protein concentration was determined using the BCA method. Then, 30 µg of total protein was taken by SDS-PAGE electrophoresis and transferred to a 0.45 μm PVDF membrane. The membrane was blocked with rapid blocking solution for 30 min at room temperature and incubated with primary antibody overnight at 4 °C. The membrane was washed three times with tris-buffered saline with tween 20 (TBST), incubated with horseradish peroxidase (HRP)-labeled goat anti-dialytic antibody for 1 h at room temperature, washed three times with TBST, and developed with a photochemiluminescence kit. Signals were captured using a Tanon 5200 fully automated chemiluminescence imaging system, and grayscale analysis was performed using ImageJ software.
Mouse tumor-bearing model experiment
Animal experiments were approved by the Experimental Animal Ethics Committee of Youjiang Medical University for Nationalities (No. 2024081601), and the ARRIVE guidelines were followed. All animal experiments were conducted in strict compliance with the relevant guidelines and regulations. Five-week-old female SPF-grade BALB/c nude mice (Vital River, batch number 44829700037597), were selected for the study. The right axilla was subcutaneously inoculated with 100 µL of cervical cancer cell suspension at a concentration of 5 × 106 cells. Tumor growth was monitored daily, and after the volume reached 100–200 mm³ (V = 0.5×L×W²), the group was randomly categorized into two groups: the control group (n = 5) was injected intraperitoneally with the carrier solution, and the administration group (n = 5) was injected with an equal volume of the solution containing 2 mg/kg of Ziyu I, which was administered every 2 days. Body mass and tumor volume were recorded every 3 days. Tumor, liver, and kidney tissues were collected post-euthanasia via cervical dislocation performed under 4% isoflurane anesthesia for subsequent experimental analysis.
Hematoxylin and Eosin staining and immunohistochemical testing
Pathological analyses were performed using paraffin-embedded tissue sections and histochemical staining. Freshly isolated tissues were fixed with 4% paraformaldehyde at 4 °C for 24 h, followed by sequential gradient dehydration, xylene transparency, and paraffin embedding, and sliced into 4 μm-thick slices. Hematoxylin and eosin staining procedure was as follows: dewaxing with xylene, gradient hydration, Harris hematoxylin immersion staining for 8 min, 1% hydrochloric acid-ethanol differentiation for 5 s, returning to blue in running water for 1 h, 0.5% eosin staining for 3 min, gradient dehydration, and neutral tree resin sealing of slices. The immunohistochemical assay was as follows: after treatment with sodium citrate antigen repair solution, 3% H2O2 was incubated at room temperature for 10 min to eliminate endogenous enzyme activity, and 5% goat serum was sealed for 30 min. Drops of the Ki-67 monoclonal antibody were incubated overnight at 4 °C, rinsed in PBS, and incubated with HRP-labeled secondary antibody at 37 °C for 30 min. DAB chromatography. Hematoxylin was re-stained, dehydrated, made transparent, and then sealed.
Statistical analyses
Experimental data were obtained from three independent biological replicates (n = 3) and are expressed as mean ± standard deviation. Statistical analyses were performed using GraphPad Prism 9.5.1 (GraphPad Software, San Diego, CA, USA). Group comparisons were performed using the independent samples Student’s t-test. Statistical significance was defined as follows: *P < 0.05, **P < 0.01, ***P < 0.001, ns indicates no statistical difference (P ≥ 0.05).
Data availability
For the data used in this study, please contact the corresponding author.
References
Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 74, 229–263 (2024).
Ramakrishnan, S., Partricia, S. & Mathan, G. Overview of high-risk hpv’s 16 and 18 infected cervical cancer: pathogenesis to prevention. Biomed. Pharmacother. 70, 103–110 (2015).
Canfell, K., Smith, M., Saville, M. & Arbyn M. HPV screening for cervical cancer is reaching maturity. BMJ 377, o1303 (2022).
Mo, Y. et al. Prophylactic and therapeutic HPV vaccines: current scenario and perspectives. Front. Cell. Infect. Microbiol. 12, 909223 (2022).
Abu-Rustum, N. R. et al. NCCN guidelines(r) insights: cervical cancer, version 1.2024. J. Natl. Compr. Canc Netw. 21, 1224–1233 (2023).
Duska, L. R., Podwika, S. E. & Randall, L. M. Top advances of the year: cervical cancer. Cancer 130, 2571–2576 (2024).
Bhattacharjee, R. et al. Cellular landscaping of cisplatin resistance in cervical cancer. Biomed. Pharmacother. 153, 113345 (2022).
Grau-Bejar, J. F., Garcia-Duran, C., Garcia-Illescas, D. & Mirallas, O. Oaknin, A. Advances in immunotherapy for cervical cancer. Ther. Adv. Med. Oncol. 15, 17588359231163836 (2023).
Zhang, Y. et al. Antrodia cinnamomea exerts an anti-hepatoma effect by targeting PI3k/AKT-mediated cell cycle progression in vitro and in vivo. Acta Pharm. Sin B. 12, 890–906 (2022).
Nong, Y. et al. Oxymatrine inhibits PD-l1 by downregulating IFN-gamma to promote ferroptosis and enhance anti-PD-l1 efficacy in liver cancer. J. Hepatocell Carcinoma. 11, 2427–2440 (2024).
Jang, E., Inn, K. S., Jang, Y. P., Lee, K. T. & Lee, J. H. Phytotherapeutic activities of Sanguisorba officinalis and its chemical constituents: a review. Am. J. Chin. Med. 46, 299–318 (2018).
Sun, H. et al. Ziyuglycoside I attenuates collagen-induced arthritis through inhibiting plasma cell expansion. J. Ethnopharmacol. 294, 115348 (2022).
Zhu, X. et al. Ziyuglycoside I inhibits the proliferation of MDA-MB-231 breast carcinoma cells through inducing p53-mediated g2/m cell cycle arrest and intrinsic/extrinsic apoptosis. Int. J. Mol. Sci. 17, 1903 (2016).
Zhu, X. et al. Triggering p53 activation is essential in Ziyuglycoside i-induced human retinoblastoma WERI-rb-1 cell apoptosis. J. Biochem. Mol. Toxicol. 32 https://doi.org/10.1002/jbt.22001 (2018).
Hopkins, A. L. Network Pharmacology. Nat. Biotechnol. 25, 1110–1111 (2007).
Rositch, A. F. et al. Epidemiology of cervical adenocarcinoma and squamous cell carcinoma among women living with human immunodeficiency virus compared with the general population in the united States. Clin. Infect. Dis. 74, 814–820 (2022).
Gennigens, C. et al. Recurrent or primary metastatic cervical cancer: current and future treatments. ESMO Open. 7, 100579 (2022).
Kokka, F. et al. Hysterectomy with radiotherapy or chemotherapy or both for women with locally advanced cervical cancer. Cochrane Database Syst. Rev. 8, CD010260 (2022).
Liu, J. F. et al. Safety, clinical activity and biomarker assessments of Atezolizumab from a phase I study in advanced/recurrent ovarian and uterine cancers. Gynecol. Oncol. 154, 314–322 (2019).
Xu, Y. et al. Neoadjuvant chemotherapy increases the 5-year overall survival of patients with resectable cervical cancer: a systematic review and meta-analysis. Taiwan. J. Obstet. Gynecol. 60, 433–441 (2021).
Frumovitz, M. et al. Combination therapy with topotecan, paclitaxel, and bevacizumab improves progression-free survival in patients with recurrent high-grade neuroendocrine cervical cancer: a neuroendocrine cervical tumor registry (NeCTuR) study. Am. J. Obstet. Gynecol. 228, 445e1–445e8 (2023).
Li, F. et al. Autologous tumor-infiltrating lymphocyte mono-therapy can rapidly shrank tumor in Asian patient with stage III/IV cervical cancer: two cases report. Int. J. Womens Health. 16, 31–39 (2024).
Xia, C., He, Z. & Cai, Y. Quantitative proteomics analysis of differentially expressed proteins induced by Astragaloside IV in cervical cancer cell invasion. Cell. Mol. Biol. Lett. 25, 25 (2020).
Wang, Q. et al. Reactive oxygen species-mediated apoptosis contributes to chemosensitization effect of saikosaponins on cisplatin-induced cytotoxicity in cancer cells. J. Exp. Clin. Cancer Res. 29, 159 (2010).
Lixing, X. et al. Saikosaponin-d-mediated downregulation of neurogenesis results in cognitive dysfunction by inhibiting Akt/Foxg-1 pathway in mice. Toxicol. Lett. 284, 79–85 (2018).
Fan, G. et al. Saikosaponin D exacerbates acetaminophen-induced liver injury by sabotaging GABARAP-SNARE complex assembly in protective autophagy. Phytomedicine 138, 156409 (2025).
O’Leary, B. et al. The genetic landscape and clonal evolution of breast cancer resistance to Palbociclib plus fulvestrant in the PALOMA-3 trial. Cancer Discov. 8, 1390–1403 (2018).
Clatot, F. et al. Risk of early progression according to Circulating ESR1 mutation, CA-15.3 and CfDNA increases under first-line anti-aromatase treatment in metastatic breast cancer. Breast Cancer Res. 22, 56 (2020).
Freitag, C. E., Mei, P., Wei, L., Parwani, A. V. & Li, Z. ESR1 genetic alterations and their association with clinicopathologic characteristics in advanced breast cancer: a single academic institution experience. Hum. Pathol. 107, 80–86 (2021).
Liu, S. et al. Pan-cancer comprehensive analysis identified EGFR as a potential biomarker for multiple tumor types. Appl. Biochem. Biotechnol. 197, 1055–1072 (2025).
Singh, S., Sadhukhan, S. & Sonawane, A. 20 years since the approval of first EGFR-TKI, gefitinib: insight and foresight. Biochim. Biophys. Acta Rev. Cancer. 1878, 188967 (2023).
Wang, Z. et al. HSP90AA1 is an unfavorable prognostic factor for hepatocellular carcinoma and contributes to tumorigenesis and chemotherapy resistance. Transl Oncol. 50, 102148 (2024).
Kanehisa, M., Furumichi, M., Sato, Y., Matsuura, Y. & Ishiguro-Watanabe, M. KEGG: biological systems database as a model of the real world. Nucleic Acids Res. 53, 672–677 (2025).
Dhillon, A. S., Hagan, S., Rath, O. & Kolch, W. MAP kinase signalling pathways in cancer. Oncogene 26, 3279–3290 (2007).
Kang, Y. J. et al. MHY2251, a new SIRT1 inhibitor, induces apoptosis via JNK/p53 pathway in HCT116 human colorectal cancer cells. Biomol. Ther. (Seoul). 31, 73–81 (2023).
Lv, C., Lan, A., Fan, X., Huang, C. & Yang, G. Asperolide a induces apoptosis and cell cycle arrest of human hepatoma cells with p53-y220c mutant through p38 mediating phosphorylation of p53 (s33). Heliyon 9, e13843 (2023).
Yan, H. et al. Regorafenib inhibits EphA2 phosphorylation and leads to liver damage via the ERK/MDM2/p53 axis. Nat. Commun. 14, 2756 (2023).
Gong, G. et al. The cyr61 is a potential target for rotundifuran, a natural labdane-type diterpene from Vitex trifolia l., To trigger apoptosis of cervical cancer cells. Oxid. Med. Cell. Longev. 6677687 (2021). (2021).
Zhai, L. L. et al. Curcumin inhibits the invasion and migration of pancreatic cancer cells by upregulating TFPI-2 to regulate ERK- and JNK-mediated epithelial-mesenchymal transition. Eur. J. Nutr. 63, 639–651 (2024).
Acknowledgements
This study was supported by the Natural Science Foundation of Guangxi Province (Nos.2025GXNSFHA069073 and 2025GXNSFHA069187), the Scientific Research and Technology Development Plan of Baise (No.20241541), the Project to improve basic research ability of Young and middle-aged teachers of Guangxi Universities (2025KY0568), and the Innovation Project of Guangxi Graduate Education (NO.YCSW2024532).
Author information
Authors and Affiliations
Contributions
Shaofeng Huang wrote the manuscript and performed the analysis; Rong Wang and Yanlun Song wrote and revised the manuscript; Siyu Liao and Minmin Ou performed the analysis; Yuehua Huang, Zongyun Lin, and Fenglian Yang collected the data; Junli Wang revised the manuscript; Haimei Qin and Fenglian Yang designed the study. All the authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Huang, S., Wang, R., Song, Y. et al. Uncovering the anti-cervical cancer mechanism of Ziyuglycoside I via integrated network pharmacology molecular docking and experimental validation. Sci Rep 15, 36258 (2025). https://doi.org/10.1038/s41598-025-20129-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-20129-z