Abstract
This study aimed to clarify the role of tyrosine phosphatase SHP-1 on renal fibrosis and its underlying mechanisms. We used a unilateral ureteral obstruction (UUO) rat model to simulate renal fibrosis and achieved overexpression of SHP-1 through lentivirus injection. The effects of overexpression of SHP-1 on extracellular matrix (ECM) deposition, fibrosis degree and key indicators of glycolysis in renal tissue were evaluated. Results showed that overexpression of SHP-1 significantly alleviated renal fibrosis and ECM accumulation induced by UUO. SHP-1 overexpression reduced α-SMA, FN, collagen I, III, HK, PKM2, Bax expression, and lactate levels, while increasing glucose uptake and Bcl-2 expression. Bioinformatics analysis has shown that the transcription factors FOXO (FoxO3a and FoxO1) are downstream targets of the STAT3 signaling pathway, jointly regulating the glycolysis/gluconeogenesis pathway. Overexpression of SHP-1 effectively inhibited the phosphorylation of STAT3 (p-STAT3) and the expression of downstream FoxO3a/FoxO1. Mechanism studies have shown that overexpression of SHP-1 inhibits the STAT3 signaling pathway, generating effects similar to those of glycolytic inhibitors: inhibiting glycolysis, promoting cell proliferation, reducing cell apoptosis and collagen deposition, thereby alleviating renal fibrosis. In conclusion, overexpression of SHP-1 can effectively alleviate the progression of renal fibrosis by inhibiting the STAT3/FOXO signaling axis and its mediated glycolytic reprogramming. This provides new experimental evidence for the development of anti-fibrotic therapies targeting SHP-1.
Similar content being viewed by others
Introduction
Chronic Kidney Disease (CKD) is a major challenge to global health, its incidence is on the rise and it has become a common cause of death1. Renal fibrosis is the main pathological feature of CKD progression. Renal fibrosis is a group of chronic metabolic organ diseases with significant damage, characterized by an abnormal increase in myofibroblasts and excessive production and deposition of extracellular matrix (ECM) proteins, which can lead to cancer and death over time2. In the early stage of CKD, renal fibrosis is mainly manifested as glomerulosclerosis and tubulointerstitial fibrosis, and as the disease progresses, the degree of renal fibrosis gradually aggravates, ultimately leading to renal failure3. Thus, renal fibrosis plays a key role in the progression of CKD and provides an important indicator of disease prognosis and diagnosis. However, the mechanism of renal fibrosis progression has not been fully elucidated, and clinical treatment of renal fibrosis still lacks effective methods.
Myofibroblasts change their energy metabolism from oxidative phosphorylation to the glycolytic pathway during the pathological process of renal fibrosis, and this change in energy metabolism is conducive to promoting the phenotypic transformation of myofibroblasts and the continuous development of the pathological process of renal fibrosis4. Compared with oxidative phosphorylation, aerobic glycolysis can rapidly utilize ATP generated from glucose to ensure its rapid proliferation and promote the production of α-SMA and type I collagen, exacerbating the generation of ECM, and at the same time, a large amount of lactic acid produced during aerobic glycolysis can induce more myofibroblasts to aggregate and transdifferentiate, which further exacerbates the pathological process of renal fibrosis4,5. A UUO mouse model and transforming growth factor-β1 (TGF-β1)-induced fibroblasts have been found to reveal metabolic reprogramming of renal interstitial fibroblasts during renal fibrosis, which is characterized by a conversion from oxidative phosphorylation to aerobic glycolysis, and an elevated expression of HK and PKM2 in renal tissue and fibroblasts6. Wei et al. found that glycolysis inhibitors can inhibit renal interstitial fibrosis through different actions on fibroblasts and renal tubular cells4. Thus, metabolic reprogramming of mesenchymal fibroblasts promotes renal fibrosis and may be critical for the continued progression of CKD.
SH2 domain-containing protein tyrosine phosphatase 1 (SHP-1) is a non-receptor-type protein tyrosine phosphatase that is mainly responsible for negatively regulating the tyrosine phosphorylation signaling pathway in hematopoietic cells7. Some studies have found that SHP-1 can mediate signaling in cancer cells involved in tumor progression, and increasing SHP-1 function or expression needs to be further explored8. It has been shown that SHP-1 is negatively correlated with atrial fibrillation (AF)severity and that its overexpression inhibits AF through STAT3 dephosphorylation, thereby reducing atrial fibrosis, ROS generation, and ECM deposition9. Su et al. demonstrated in vitro that the SHP-1 agonist SC-43 significantly ameliorated liver fibrosis by upregulating SHP-1 and inhibiting phosphorylated STAT310. However, the expression of SHP-1 in renal fibrotic lesion tissues is currently unknown.
Therefore, in the present study, we first evaluated the effects of SHP-1 overexpression on renal tissue injury, glycolysis, ECM deposition, and the degree of renal fibrosis in a rat model of renal fibrosis constructed by the unilateral ureteral obstruction (UUO) method by injecting SHP-1 overexpressing lentivirus into the tail vein. We also screened and validated the key pathways of action by analyzing the effects of differential gene expression in renal tissues after overexpression of SHP-1 by raw letter analysis. Then a renal fibrosis cell model was constructed by TGF-β1 induction to further validate the mechanism of overexpression of SHP-1 on renal fibrosis.
Materials and methods
Cell culture
Rat proximal renal tubular epithelial cell line (NRK-52E cells) was purchased from Shanghai Cell Bank, Chinese Academy of Sciences. Cell culture conditions: 5 mL DMEM high glucose medium (SH30022.01B, Hyclone) containing 10% fetal bovine serum (10270-106, Gibco), 1% double antibody (containing 100 U/mL penicillin with 100 μg/mL streptomycin), and placed in an incubator at 37 °C with 5% CO2.
SHP-1 lentivirus construction and transfection
An overexpression lentiviral vector was constructed based on the sequence of rat SHP-1 (GI:4098326) and transfected into NRK-52E cells. The day before infection, cells were inoculated in 6-well plates, 3 × 105 cells/well, 2 mL per well, and incubated at 37 °C overnight. The virus was then removed from the refrigerator and melted on ice, the cells’ original medium was aspirated, 2 mL of fresh medium containing, the volume of virus required for optimal MOI was added to the cells, and the incubator was incubated. The infection medium was replaced with 2 mL of fresh medium after 24 h of incubation. The fluorescence expression was observed under a fluorescence microscope 72 h after cell infection. Transfection efficiency was verified by qRT-PCR assay.
Construction of animal model of renal fibrosis
Twenty-four SD rats (6-week-old, male, 220–240 g) were selected and raised under specific pathogen free (SPF) conditions. All animals were obtained from Hunan Slake Jingda. After adaptive culture, the renal fibrosis rat model was established by unilateral ureteral obstruction (UUO), and the specific methods are as follows: after anesthesia, the rats were placed in a supine position with their limbs and head fixed. The abdomen was trimmed and disinfected with 75% alcohol. A left abdominal incision was made, and the skin, muscles, and abdominal wall layers were cut open layer by layer to expose and separate the left ureter. Two ligatures were made with No. 4–0 silk thread, with the upper ligature point close to the lower pole of the left kidney. The ureter was cut at the two ligature points, and the skin was sutured layer by layer. In the sham group, only the left ureter was exposed but not ligated. Penicillin was given for anti-inflammatory treatment after surgery.
After the surgery, the animals were randomly divided into 4 groups: Sham (n = 6), UUO (n = 6), UUO+LV-NC (n = 6), and UUO+LV-SHP-1 (n = 6). The UUO+LV-NC and UUO+LV-SHP-1 groups were respectively injected with empty lentivirus or SHP-1-overexpressing lentivirus (108 TU/ml, 100 μl) via the tail vein immediately after UUO surgery for 14 days of intervention. The sham group and the UUO group were injected with the same amount of solvent simultaneously. After the experiment, 3% sodium pentobarbital 40 mg/kg was injected intraperitoneally for anesthesia, blood was collected, and kidney tissue was quickly collected.
qRT-PCR
Take each group of tissues or cells in an enzyme-free centrifuge tube of RNAlater reagent. Total RNA was extracted using a fully automated nucleic acid extractor and the accompanying kit (R1100, Wuhan Xavier Biotechnology Co., Ltd.) according to the kit instructions. Extracted RNA was tested for RNA concentration and purity by Nanodrop 2000. According to the instructions of the reverse transcription kit (G3330, Wuhan Xavier Biotechnology Co., Ltd.), RNA and the corresponding reagents were added sequentially, held at 37 °C for 60 min on the PCR instrument, and then held at 85 °C for 5 s after the end of the reaction, which was briefly centrifuged and then placed on ice. Fluorescence quantitative PCR: reaction conditions were pre-denaturation 95 °C, 30 s, cycling (40 times) 95 °C, 5 s, 60 °C, 30 s, melting curve 95 °C, 15 s 60 °C, 1 min 95 °C, 15 s 50 °C, 30 s. After the amplification reaction, conduct melting curve analysis to determine whether the product has non-specific amplification, analyze the amplification curve, and calculate the Ct value. Using GAPDH as the internal reference gene, the 2-ΔΔCT method was used to calculate the difference in mRNA expression levels between each group. The primers are shown in Table 1
Periodic acid-schiff stain (PAS)
Rat kidney tissue was stained using PAS kit (G1280, Solarbio, Beijing, China). The kidney tissue sections were dewaxed with xylene, graded ethanol and distilled water, dewaxed with iodic acid oxidation solution for 5 min, dewaxed with Schiff’s reagent for 15 min, and stained with hematoxylin (DH0010, Beijing leagene biotech.co., ltd) for 1 min, differentiate in 1% hydrochloric acid ethanol for 3 s, dehydrate, seal with transparent seal, seal with neutral gum, observe under microscope and collect pictures. PAS glycogen positivity is purple-red and is used to evaluate changes in the basement membrane and mesangial matrix. Referring to the research of Liu et al.11, scores were given based on the degree of lesion (renal tubular atrophy, injury, and renal tubulointerstitial fibrosis), with a score range of 0 to 3:0 points (no injury). 1 point (degree of injury <20%); 2 points (Injury degree ranging from 20 to 50%); 3 points (injury degree >50%).
Masson
Kidney tissue sections were stained with 0.5% hematoxylin for 5 min, washed, alcohol fractionated with 0.5% hydrochloric acid for 20 s, washed, and stained with Masson’s complex staining solution for 5 min. The samples were washed with 0.2% acetic acid solution, reacted with 5% phosphomolybdic acid solution for 5 min, washed with 0.2% acetic acid solution, immersed in 2% aniline blue solution for 20 s, washed with anhydrous ethanol and dried, and sealed with neutral dendrimer, and observed under the microscope and pictures were collected. Collagen staining in the interstitial region of the kidney appeared blue and was used to assess renal fibrosis. Masson staining kit (G1340) was purchased from Solarbio (Beijing, China). The image J software was used to analyze the fibrobic area.
TUNEL
The pre-treated tissue sections were deparaffinized, hydrated and endogenous catalase blocked, rinsed with distilled water and PBS, and then spiked with Proteinase K working solution (G1205, Solarbio). After rinsing with PBS and adding TUNEL reaction mixture, the reaction was completed by adding converter-POD to the specimen after rinsing with PBS, rinsing with PBS after the reaction, and then adding freshly prepared DAB (DA1010, Solarbio) to develop the color, rinsing with tap water, re-staining with hematoxylin, rinsing with distilled water, and then slicing, dehydrating and sealing the specimen for observation and photographing under the microscope. The image J software was used to analyze the rate of positive cells.
Immunohistochemistry
Rat kidney tissues were dehydrated and wax-embedded by the usual procedure. Paraffin sections were deparaffinized and dehydrated with ethanol; 3% H2O2 was added and incubated at 37 °C for 10 min to block the endogenous peroxidase activity. Phosphate buffer solution (PBS) was rinsed 3 times × 5 min; antigen repair was performed, and goat serum was incubated with α-SMA (1:50, Proteintech) and FN (1:500, Proteintech) antibodies overnight at 4 °C. PBS rinse 3 times × 5 min; add biotin labeling, incubate at 37 °C for 20 min; PBS rinse 3 times × 5 min; add horseradish peroxidase labeling, incubate at 37 °C for 30 min. PBS washed 3 times × 5 min; newly prepared DAB was added for color development, hematoxylin re-staining for 10 s, PBS counterblue, dehydration, transparency, and sealing. Photographs are taken through a microscope and the relevant parts of the sample are collected and analyzed.
Biochemical testing
Glucose (A154-1-1) and lactate (A154-1-1) levels in each group were detected by using the Nanjing Jianjian Bioengineering Institute kit. The experimental steps were carried out strictly according to the instructions of the kit.
Immunofluorescence
Kidney tissue samples were paraffin-embedded, cut into thin slices, deparaffinized, hydrated, heated in citrate buffer, incubated with drops of 3% H2O2 for 15 min, and serum was closed at 37 °C for 30 min. SHP-1 (PAB32009, bioswamp) antibody was added dropwise and incubated at 4 °C overnight. After dropwise addition of fluorescent secondary antibody and incubation at 37°C for 1 h, DAPI re-staining for 10 min, dropwise addition of anti-fluorescence quencher to seal the film, and then photographed using fluorescence microscope.
Western-blot
The BCA Protein Assay Kit is used to detect the concentration of proteins in the supernatant collected after lysis of cells with RIPA Lysis Buffer. In SDS/PAGE, protein samples were separated on 12% sodium dodecyl sulfate polyacrylamide gels and transferred to PVDF membranes with 5% skimmed milk closed for 2 h at room temperature. The membrane was incubated with primary antibodies α-SMA (PAB30319, 1:1000), FN (PAB42262, 1:1000), Collagen I (PAB34241, 1:1000), Collagen III (PAB46099, 1:1000), HK2 (PAB30271, 1:1000), PKM2 (PAB31790, 1:1000), Bax (PAB46088, 1:1000), Bcl-2 (PAB30041, 1:1000), SHP-1 (PAB32009, 1:1000), and GAPDH (PAB36269, 1:1000) were incubated overnight at 4 °C. After washing the membrane 3 times with TBST, the membrane was incubated with secondary antibody for 1 h at room temperature. After color development by ECL, the image was exposed in a chemiluminescence imaging detector. All antibodies were purchased from Bioswamp (Wuhan, China). The densitometric values of the bands were calculated using Image J. GAPDH was used as a reference and each assay was repeated 3 times.
Bioinformation analysis
We searched the GEO database for rat transcriptome sequencing data using the keyword “renal fibrosis”. Find data set GSE216376 containing kidney tissue transcriptome sequencing data from 3 UUO-induced model rats and 3 sham-operated control rats. Expression matrices were constructed from the transcriptome sequencing data of three UUO-induced rat models (UUO) and three sham-operated control rats (Sham), and then normalized to obtain the normalized expression matrices. Inter-sample correlations and PCA analyses were calculated based on the normalized expression matrix. The UUO and Sham groups were analyzed for differences using limma software and volcano plots were drawn. ClusterProfiler software was used to perform GO functional enrichment analysis of the screened differentially expressed genes. ClusterProfiler software was used to perform GO functional enrichment analysis and KEGG enrichment analysis12,13,14 of the screened differentially expressed genes.
Construction of renal fibrosis cell model
Based on the well-cultured NRK-52E cells, 2 ng/ml TGF-β1 was used to induce 0, 24, 48, and 72 h respectively. CCK8 was used to detect cell activity, and qPCR was used to detect the mRNA expression of SHP-1 and α-SMA to determine the optimal induction time of TGF-β1 and simulate and construct a renal fibrosis cell model.
Cell grouping and processing
To study the effect of SHP-1 overexpression pathway-mediated glycolysis on renal fibrotic cells, we divided the cells into the following six groups: control group (normal cultured cells), model group (induced with 2 ng/mL of TGF-β1 for 48 h), LV-NC group (transfected with LV-NC after inducing with 2 ng/mL of TGF-β1 for 48 h), LV-SHP-1 group (transfected with LV-SHP-1 after inducing with 2 ng/ml of TGF-β1 for 48 h), 2-DG group (transfected with 2 ng/mL of TGF-β1 for 48 h after induction with 10 mM 2-DG) and 2-DG + LV-SHP-1 group (after induction with 2 ng/ml of TGF-β1 for 48 h, 10 mM 2-DG was added for 1 h before transfection of lv-SHP-1). TGF-β1 (100–21) was purchased from PeproTech.
To investigate the effect of SHP-1 overexpression on TGF-β1-induced renal fibrosis cells by targeting this pathway to mediate glycolysis, we divided the cells into the following six groups: control group (normal cultured cells), model group (induced with 2 ng/mL of TGF-β1 for 48 h), LV-NC group (transfected with LV-NC after inducing with 2 ng/ml of TGF-β1 for 48 h), LV-SHP-1 group (transfected with 2 ng/ml of TGF-β1 after inducing with 2 ng/ml of TGF-β1 for 48 h), Inh-STAT3 group (treated with 2 ng/ml of TGF-β1 for 48 h after induction, added 10 μM S3I-201 treatment), Inh-STAT3 + LV-SHP-1 (induced with 2 ng/ml of TGF-β1 for 48 h, pre-treated with 10 μM S3I-201 for 1 h before transfection of lv-SHP-1).
Cell counting Kit-8 (CCK8)
Cells were collected from each group, the concentration of cell suspension was adjusted, and the cells were inoculated into 96-well plates, 3 × 103 cells/well, 100 μL per well, and incubated at 37 °C in a 5% CO2 incubator overnight to make the cells adherent to the wall. The cells in each group were treated according to the different groupings and the culture was continued to the corresponding time. Then the cell culture plate was removed, and 10 μL of CCK8 solution was added to each well, and the incubation was continued for 4 h. The cell culture plate was then incubated for 4 h. The cell culture plate was then incubated for 4 h. Absorbance values of the wells were measured at 450 nm on the ELISA.
Flow cytometry
NRK-52E cells in logarithmic growth phase were taken as 1 × 106 cells resuspended in culture medium, 300 g, and centrifuged at 4 °C for 5 min. Add 1mL of pre-cooled PBS, gently blow to mix the cells, and centrifuge at 300g for 5min at 4 °C. Cells were resuspended in 500 μL of diluted 1 × Annexin V Binding Buffer. Add 5 μL of Annexin V-APC staining solution and 5 μL of 7-AAD staining solution (100 μg/mL)), mix gently, and incubate for 20 min at 4 °C away from light. flow-through assay was performed immediately. Analysis was performed using NovoExpress analysis software.
Statistical analysis
Statistical analysis was performed using Graph Pad Prism 8.0 software, with K-S test data conforming to normal distribution and ANOVA data conforming to chi-square. Data were expressed as mean ± standard deviation (mean ± SD), t-test was used for two-by-two comparisons, and ANOVA was used to compare means between groups. Differences were considered statistically significant at P < 0.05.
Results
Effects of overexpression of SHP-1 on renal injury and apoptosis in UUO rats
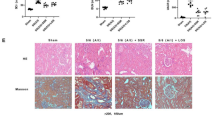
We first constructed a lentiviral vector for overexpression of SHP-1. Compared with the LV-NC group, the expression of SHP-1 in the cells of the LV-SHP-1 group was significantly increased (Fig. 1A). This result shows that the overexpression SHP-1 lentiviral vector was successfully constructed. Then we constructed a UUO rat model. Through PAS staining, it was found that compared with the Sham group, the mesangial matrix of rats in the UUO group and UUO+LV-NC group significantly accumulated. Compared with the UUO+LV-NC group, the accumulation of mesangial matrix in the UUO+LV-SHP-1 group was significantly inhibited (Fig. 1B & E). At the same time, Masson staining found that compared with the Sham group, the interstitial fibrosis in the kidney tissue of rats in the UUO group and UUO+LV-NC group was severe. Compared with the UUO+LV-NC group, the degree of renal tissue fibrosis in the UUO+LV-SHP-1 group was reduced (Fig. 1C & F). In addition, Tunel staining showed that compared with the Sham group, the apoptotic area of rat kidney tissue in the UUO group and UUO+LV-NC group increased significantly. Compared with the UUO+LV-NC group, the apoptotic area of renal tissue in the UUO+LV-SHP-1 group was significantly reduced (Fig. 1D & G). This result shows that overexpression of SHP-1 can significantly inhibit the apoptosis of renal tissue in UUO rats and improve the degree of renal fibrosis.
Effect of overexpression of SHP-1 on renal injury and fibrosis in UUO rats. (A) Transfection efficiency of overexpression of SHP-1 was verified by qRT-PCR. (B & E) Changes in renal injury in rats were evaluated by PAS staining. (C & F) Degree of fibrosis was evaluated by Masson staining. (D & G) Apoptosis of renal tissues in rats was evaluated by TUNEL.
Effect of overexpression of SHP-1 on renal fibrosis and glycolysis in UUO rats
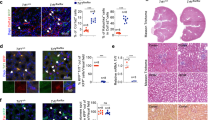
Next, we examined renal fibrosis marker protein expression. The expression of α-SMA and fibronectin (FN) was significantly increased in the kidney tissues of UUO rats compared with the Sham group. The expression of α-SMA and FN was significantly lower in the kidney tissues of rats in the UUO+LV-SHP-1 group compared with the UUO+LV-NC group. The results of immunohistochemistry and western-blot were consistent (Fig. 2A and B). Meanwhile, the protein expression of both Collagen I and Collagen III was significantly upregulated in the UUO group compared to the Sham group. The protein expression of both Collagen I and Collagen III was significantly down-regulated in the UUO+LV- SHP-1 group compared with the UUO+LV-NC group. In addition, glucose levels were significantly lower and lactate levels were significantly higher in the UUO group compared to the Sham group. Glucose levels were significantly increased and lactate levels were significantly decreased in the UUO+LV- SHP-1 group compared with the UUO+LV-NC group (Fig. 2C). And the mRNA and protein expression of HK and PKM2 were significantly increased in the kidney tissues of rats in the UUO group. Compared with the UUO+LV-NC group, mRNA and protein expression of HK and PKM2 were significantly reduced in the kidney tissues of rats in the UUO+LV- SHP-1 group. Examination of apoptotic proteins revealed decreased expression of Bcl-2 and increased expression of Bax in kidney tissues of UUO rats. Compared with the UUO+LV-NC group, the expression of Bcl-2 was increased and the expression of Bax was decreased in the kidney tissues of rats in the UUO+LV- SHP-1 group. In addition, the protein expression of SHP-1 was significantly reduced in kidney tissues of UUO rats. The mRNA expression of SHP-1 was significantly increased in rat kidney tissues after transfection with LV- SHP-1 (Figs. 2B and 3B). Further detection by immunofluorescence revealed that the fluorescence intensity of SHP-1 was significantly weakened in the UUO group. The fluorescence intensity of SHP-1 was significantly enhanced in rats in the UUO+LV- SHP-1 group compared with the UUO+LV-NC group (Fig. 3A). This result suggests that overexpression of SHP-1 may affect renal fibrosis by regulating glycolysis in renal tissues of UUO rats.
Effect of overexpression of SHP-1 on renal fibrosis and glycolysis in UUO rats. (A) The effect of α-SMA and FN protein expression in renal tissues was detected by immunohistochemistry. (B) The protein expression of α-SMA, FN, Collagen I, Collagen III, HK, PKM2, Bcl-2, and Bax was detected by western-blot in renal tissues. (C) Serum glucose level and lactate production in rats were detected by biochemical kits. (D) Expression of HK and PKM2 was detected by qRT-PCR.
Effect of overexpression of SHP-1 on the changes of SHP-1 in kidney tissues of UUO rats. (A) Localization of SHP-1 in kidney tissues detected by immunofluorescence. (B) Detection of mRNA expression of SHP-1 in kidney tissues of rats by qRT-PCR.
SHP-1 target and pathway screening and validation
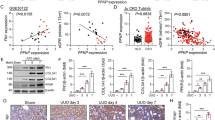
Next, we performed a biosignature analysis of SHP-1 action targets and pathways. As shown in Fig. 4A, the correlation results show that the similarity between the Sham and UUO groups is less than the similarity within the groups, and the samples are more reproducible. The UUO and Sham groups were analyzed for differences using limma software, and differential genes were screened according to |log2FoldChange|≥1 and pvalue≤ 0.05 as the threshold. The differential gene volcano map is shown in Fig. 4B. Differential genes were analyzed for GO function enrichment, which includes molecular function (MF), cellular component (CC) and biological process (BP). Among the MFs were RNA polymerase II-specific DNA-binding transcription factor binding, ubiquitin-like protein ligase binding, transcriptional coregulator activity, and regulation of apoptotic processes in epithelial cells (Fig. 4C). We then performed KEGG enrichment analysis on the screened differential genes. In the enriched pathway STAT3 was found to regulate FOXO in the FOXO signaling pathway, and FOXO is involved in gene regulation in downstream glycolysis and glucose de novo related pathways (Fig. 5A). Therefore, we validated the STAT3/FOXO pathway. It was found that the expression of p-STAT3, FoxO3a and FoxO1 was significantly up-regulated in the kidney tissues of UUO rats compared with the Sham group. The expression of p-STAT3, FoxO3a and FoxO1 was significantly down-regulated in the kidney tissues of rats in the UUO+LV- SHP-1 group compared with the UUO+LV-NC group (Fig. 5B).
Bio-confidence screening of SHP-1 targets and pathways. (A) Inter-sample correlation and PCA analysis were calculated based on the normalized expression matrix. (B) Differential analysis of UUO and Sham groups was performed by limma software, and volcano plots were drawn. Differential genes were screened according to the threshold values of |log2FoldChange|≥ 1 and p value ≤ 0.05. (C) ClusterProfiler software was used to perform GO functional enrichment analysis of the screened differentially expressed genes.
Screening of the SHP-1 pathway of action was validated. (A) ClusterProfiler software was used to analyze the KEGG enrichment of the screened differentially expressed genes. (B) Protein expression of the pathway-associated factors STAT3, p-STAT3, FoxO3a, and FoxO1 was detected by Western-blot.
Effect of overexpression of SHP-1 on cell viability and apoptosis of renal fibrosis cells
We then constructed a renal fibrosis cell model. The viability of the cells was significantly reduced after 24, 48 and 72 h of TGF-β1 induction, and the expression of both SHP-1 and α-SMA was significantly increased, which was most effective at 48 h of induction (Fig. 6A and B). Therefore, we chose TGF-β1 induction for 48 h for subsequent experiments. Next, we found that the viability of the cells in the model group was significantly reduced and the apoptotic rate was significantly increased compared to the control group. Compared with the LV-NC group, cells in the LV- SHP-1 and DG groups showed a significant increase in cell viability and a significant decrease in apoptosis rate. Compared with the LV- SHP-1 and DG groups, cells in the DG+LV- SHP-1 group showed a significant increase in cell viability and a significant decrease in apoptosis rate (Fig. 6C–E). This result suggests that overexpression of SHP-1 promotes the cell proliferation of renal fibrotic cells and inhibits apoptosis.
Effect of overexpression of SHP-1 on value-added and apoptosis of renal fibroblasts. (A) Cell viability was detected by CCK8. (B) mRNA expression of SHP-1, α-SMA was detected by qRT-PCR. (C) The viability of cells in each group was detected by CCK8. (D–E) Apoptosis rate of cells in each group was detected by flow cytometry.
Effect of overexpression of SHP-1 on cellular renal fibrosis and glycolysis
Glucose uptake was decreased and lactate levels were increased in the cells of the model group compared to the control group. Compared with the LV-NC group, glucose levels were increased and lactate levels were decreased in the cells of the LV- SHP-1 and DG groups. Glucose levels were increased and lactate levels were decreased in the cells of the DG+LV- SHP-1 group compared with the LV- SHP-1 and DG groups (Fig. 7A). Meanwhile, mRNA expression of HK and PKM2 was significantly increased in the cells of the model group. The mRNA expression of HK and PKM2 was significantly lower in the cells of the LV- SHP-1 and DG groups compared with the LV-NC group. The mRNA expression of HK and PKM2 was significantly lower in the cells of the DG+LV- SHP-1 group compared with the LV- SHP-1 and DG groups (Fig. 7B). In addition, protein expression of α-SMA, FN, Collagen I, Collagen III, HK, PKM2 and Bax was significantly up-regulated and that of Bcl-2 was significantly down-regulated in the cells of the model group compared with the control group. The protein expression of α-SMA, FN, Collagen I, Collagen III, HK, PKM2 and Bax was significantly down-regulated and that of Bcl-2 was significantly up-regulated in the cells of the LV- SHP-1 and DG groups compared with that of the LV-NC group. The protein expression of α-SMA, FN, Collagen I, Collagen III, HK, PKM2, and Bax was significantly down-regulated and that of Bcl-2 was significantly up-regulated in the cells of the DG+LV- SHP-1 group compared to the LV- SHP-1 group and the DG group (Fig. 7C). These results suggest that overexpression of SHP-1 can regulate the glycolysis and apoptosis processes in renal fibroblasts, thereby alleviating renal fibroblast injury.
Effect of overexpression of SHP-1 on cellular renal fiber and glycolysis. (A) Glucose level and lactate production were detected by biochemical kits. (B) mRNA expression of HK and PKM2 in cells was detected by qRT-PCR. (C) The protein expression of α-SMA, FN, Collagen I, Collagen III, HK, PKM2, Bcl-2 and Bax in cells was detected by Western-blot.
Overexpression of SHP-1 affects renal fibrotic cells through STAT3/FOXO pathway
To further verify the occurrence of its mechanism, we introduced STAT3 inhibitors. Compared with the LV-NC group, the proliferation ability of cells in the LV-SHP-1 group and the Inh-STAT3 group was significantly increased, and the apoptosis rate was significantly reduced. Compared with the LV-SHP-1 group and the Inh-STAT3 group, the proliferation ability of cells in the Inh-STAT3+LV-SHP-1 group was significantly increased, and the apoptosis rate was significantly reduced (Fig. 8A–C). Compared with the LV-NC group, the glucose levels in the cells of the LV-SHP-1 group and the Inh-STAT3 group increased, and the lactate levels decreased. Compared with the LV-SHP-1 group and the Inh-STAT3 group, the glucose level in the cells of the Inh-STAT3+LV-SHP-1 group increased and the lactate level decreased (Fig. 8D). Compared with the LV-NC group, the expression of HK and PKM2 in cells of the LV-SHP-1 group and Inh-STAT3 group was significantly reduced. Compared with the LV-SHP-1 group and the Inh-STAT3 group, the expression of HK and PKM2 in the cells of the Inh-STAT3+LV-SHP-1 group was significantly reduced (Fig. 8E). In addition, compared with the LV-NC group, the protein expressions of α-SMA, FN, Collagen I, Collagen III, HK, PKM2 and Bax in the cells of the LV-SHP-1 group and Inh-STAT3 group were significantly down-regulated, and the expression of Bcl-2 Protein expression was significantly increased. Compared with the LV-SHP-1 group and Inh-STAT3 group, the protein expression of α-SMA, FN, Collagen I, Collagen III, HK, PKM2 and Bax in the cells of the Inh-STAT3+LV-SHP-1 group was significantly down-regulated. The protein expression of Bcl-2 was significantly up-regulated (Fig. 8F). Overexpression of SHP-1 can affect the glycolysis, apoptosis and fibrosis degree of renal fibrotic cells.
Overexpression of SHP-1 affects renal fibrotic cells through STAT3/FOXO pathway. (A) Cell proliferation was detected by CCK8 assay. (B and C) Detection of cell apoptosis by flow cytometry. (D) Biochemical kit detects glucose levels and lactate production. (E) Detection of HK and PKM2 expression in cells by qRT-PCR. (F) The protein expression of α-SMA, FN, Collagen I, Collagen III, HK, PKM2, Bcl-2 and Bax in cells was detected by Western-blot.
Overexpression of SHP-1 affects cellular renal fibrosis through the STAT3/FOXO pathway
Compared with the LV-NC group, the expressions of p-STAT3, FoxO3a and FoxO1 were significantly down-regulated in the LV-SHP-1 group and Inh-STAT3 group. Compared with the LV-SHP-1 group and Inh-STAT3 group, the expression of p-STAT3, FoxO3a and FoxO1 in the cells of the Inh-STAT3+LV-SHP-1 group was significantly down-regulated (Fig. 9). This result indicates that overexpression of SHP-1 may alleviate renal fibrosis through glycolysis mediated by the STAT3/FOXO pathway.
Detection of protein expression of pathway-related factors STAT3, p-STAT3, FOXO and p-FOXO through Western-blot.
Discussion
CKD is characterized by extracellular matrix deposition and is a major global health problem. Its incidence is on the rise and it has become a common cause of death15. Renal interstitial fibrosis (RIF) is considered as a common phenomenon in almost all types of chronic kidney disease CKD. In this study, we demonstrated that SHP-1 is low-expressed in both UUO rats and renal fibrotic cells. Overexpression of SHP-1 can promote cell proliferation, inhibit cell apoptosis and glycolysis, reduce collagen deposition, and reduce the degree of renal fibrosis. In terms of mechanism, it may be related to the expression inhibition of p-STAT3, FoxO3a and FoxO1.
The left UUO method for constructing animal models of renal fibrosis has the advantages of simplicity, ease of use, and reliable results, and is the classical method for preparing renal fibrosis models16,17. In this study, we found that overexpression of SHP-1 inhibited the accumulation of thylakoid matrix, reduced collagen deposition, inhibited apoptosis, and ameliorated renal fibrosis in renal tissues of UUO rats. During renal fibrosis, myofibroblasts accumulate in large numbers in the renal mesenchyme and their phenotype changes to α-SMA-positive, and this type of cell is able to synthesize α-SMA and Collagen I in large quantities and inhibit the degradation of ECM, which leads to the over-accumulation of ECM and exacerbates the pathological process of renal fibrosis18. FN, as a major extracellular matrix glycoprotein, not only maintains the morphology and stability of the ECM, but also conveys signals regulating cell proliferation and differentiation and apoptosis by binding to integrin receptors on the cell membrane, which in turn is involved in the production and deposition of the ECM19. It has been demonstrated that localized FN and collagen accumulation can promote the formation of fibroblasts, and renal fibrosis20. This study demonstrated that overexpression of SHP-1 significantly reduced the expression of α-SMA, FN, Collagen I and Collagen III in the renal tissue of UUO rats. This is similar to the results of previous studies. Yiqi Juanshen decoction can reduce the expression of SMA, FN, and Collagen I in the renal tissue of CKD rats, improve the renal function of rats, and reduce renal pathological damage and fibrosis21. Hou et al.22 found that Neuropilin 1 (NRP1) expression alleviates UUO-induced interstitial fibrosis by reducing the expression of α-SMA, FN, and Collagen I.
Studies have proven that glycolysis is an important pathogenic process in renal fibrosis, and abnormal glucose metabolism will promote the progression of renal fibrosis4,23. In this project, we found that glucose uptake was reduced, lactate levels were increased, and the expression of glycolysis-related enzymes HK and PKM2 was upregulated in the kidneys of UUO rats, confirming the metabolic shift to aerobic glucose metabolism in renal fibrosis. Overexpression of SHP-1 can reverse this phenomenon. Studies have shown that treatment of mice with the glycolysis inhibitor 2-deoxyglucose (2-DG) can improve proximal tubular epithelial cell proliferation, cystogenesis, and renal fibrosis24. In this study, overexpression of SHP-1 and 2-DG can promote the proliferation of renal fibrosis cells, inhibit cell apoptosis and glycolysis, and improve the degree of renal fibrosis. And the effect of combined use is better than the effect of using alone. It is suggested that overexpression of SHP-1 may mediate the progression of renal fibrosis by inhibiting glycolysis. Similar to previous findings, Yang et al.25 demonstrated that phosphofructokinase 1 platelet isoform (PFKP) mediates the progression of renal interstitial nephrofibrosis by regulating glycolysis in proximal tubular epithelial cells. Yang et al.26 demonstrated that targeting bone marrow glycolysis is a novel approach to inhibit renal fibrosis. Ding et al.23 also demonstrated that the expression of glycolytic enzymes was up-regulated in the kidney of UUO mice, the amounts of proteins indicated by glucose uptake and lactate production was increased and positively correlated with the fibrotic process, and that the inhibition of aerobic glycolysis by 2-DG suppressed the activation of renal mesangial fibroblasts and renal fibrosis in TGF-β1-treated kidneys.
Bioinformatics analysis was performed to further explore the underlying mechanisms. In the enriched pathway, STAT3 was found to regulate FOXO in the FOXO signaling pathway, which is involved in the gene regulation of downstream glycolysis and glucose de novo related pathways. Therefore, we validated it by a combination of in vivo and in vitro experiments. The results revealed that overexpression of SHP-1 inhibited the expression of p-STAT3 and p-FOXO in kidney tissues and renal fibroblasts of UUO rats. STAT3 is a transcription factor that regulates many downstream target genes27. It has been demonstrated that activated STAT3 is an important transcription factor in the pathogenesis of pulmonary and renal fibrosis28,29. Chen et al.30 found that interleukin 6 (IL-6) signaling prevents renal fibrosis by inhibiting STAT3 activation. Rao et al.31 found that the interaction of β-catenin with Foxo could be a new target for the treatment of renal fibrosis. Guo et al.32 demonstrated that STAT3 knockdown reduces FOXO promoter activity and FOXO expression, thereby affecting apoptosis and ameliorating encephalitis. Thus, our study demonstrated that overexpression of SHP-1 may alleviate renal fibrosis by inhibiting STAT3/FOXO pathway-mediated glycolysis.
Conclusion
In this study, we demonstrated that overexpression of SHP-1 may alleviate renal fibrosis by inhibiting STAT3/FOXO pathway-mediated glycolysis by constructing a UUO rat model and a cellular fibrosis model, thus further elucidating the mechanism of action of SHP-1 in alleviating the progression of renal fibrosis. This discovery highlights the potential of SHP-1 as a novel anti-fibrotic therapeutic target. Compared with existing strategies (such as the indirect benefits of SGLT2 inhibitors by improving metabolic disorders, or the broad-spectrum anti-fibrotic effect of pirfenidone), targeted activation of SHP-1 may offer a more direct therapeutic approach to the core fibrotic signaling pathways (such as STAT3). However, the development of effective SHP-1 agonists faces significant challenges. The primary difficulty is to achieve their tissue-specific delivery (especially renal targeting) to avoid potential systemic off-target effects. It should be noted that this study has not yet fully verified the core position of this pathway through the rescue experiment of STAT3 overexpression. This will be a key direction for future research to more precisely clarify the specific mechanism of action of SHP-1 in renal fibrosis and its feasibility as a therapeutic target.
Data availability
Data is provided within the manuscript.
References
Ruiz-Ortega, M., Rayego-Mateos, S., Lamas, S., Ortiz, A. & Rodrigues-Diez, R. R. Targeting the progression of chronic kidney disease. Nat Rev Nephrol 16(5), 269–288. https://doi.org/10.1038/s41581-019-0248-y (2020).
Sisto, M., Ribatti, D. & Lisi, S. Organ fibrosis and autoimmunity: The role of inflammation in TGFβ-dependent EMT. Biomolecules https://doi.org/10.3390/biom11020310 (2021).
Djudjaj, S. & Boor, P. Cellular and molecular mechanisms of kidney fibrosis. Mol. Aspects Med. 65, 16–36. https://doi.org/10.1016/j.mam.2018.06.002 (2019).
Wei, Q. et al. Glycolysis inhibitors suppress renal interstitial fibrosis via divergent effects on fibroblasts and tubular cells. Am. J. Physiol. Renal Physiol. 316(6), F1162–F1172. https://doi.org/10.1152/ajprenal.00422.2018 (2019).
Wang, M. et al. Ceria nanoparticles ameliorate renal fibrosis by modulating the balance between oxidative phosphorylation and aerobic glycolysis. J. Nanobiotechnol. 20(1), 3. https://doi.org/10.1186/s12951-021-01122-w (2022).
Yin, X. N., Wang, J., Cui, L. F. & Fan, W. X. Enhanced glycolysis in the process of renal fibrosis aggravated the development of chronic kidney disease. Eur. Rev. Med. Pharmacol. Sci. 22(13), 4243–4251. https://doi.org/10.26355/eurrev_201807_15419 (2018).
Hao, F., Wang, C., Sholy, C., Cao, M. & Kang, X. Strategy for leukemia treatment targeting SHP-1,2 and SHIP. Front. Cell Dev. Biol. 9, 730400. https://doi.org/10.3389/fcell.2021.730400 (2021).
Lim, S., Lee, K. W., Kim, J. Y. & Kim, K. D. Consideration of SHP-1 as a molecular target for tumor therapy. Int. J. Mol. Sci. https://doi.org/10.3390/ijms25010331 (2023).
Zang, X. et al. SHP-1 alleviates atrial fibrosis in atrial fibrillation by modulating STAT3 activation. Exp. Biol. Med. (Maywood) 248(11), 979–990. https://doi.org/10.1177/15353702231165717 (2023).
Su, T. H. et al. Src-homology protein tyrosine phosphatase-1 agonist, SC-43, reduces liver fibrosis. Sci. Rep. 7(1), 1728. https://doi.org/10.1038/s41598-017-01572-z (2017).
Liu, M. et al. Analysis of the potential biological mechanisms of geniposide on renal fibrosis by network pharmacology and experimental verification. BMC Pharmacol. Toxicol. 26(1), 17. https://doi.org/10.1186/s40360-025-00855-w (2025).
Kanehisa, M. & Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucl. Acids Res. 28(1), 27–30. https://doi.org/10.1093/nar/28.1.27 (2000).
Kanehisa, M. Toward understanding the origin and evolution of cellular organisms. Protein Sci.: A Publ. Prot. Soci. 28(11), 1947–1951. https://doi.org/10.1002/pro.3715 (2019).
Kanehisa, M., Furumichi, M., Sato, Y., Kawashima, M. & Ishiguro-Watanabe, M. KEGG for taxonomy-based analysis of pathways and genomes. Nucl. Acids Res. 51(D1), D587–D592. https://doi.org/10.1093/nar/gkac963 (2023).
Humphreys, B. D. Mechanisms of renal fibrosis. Annu. Rev. Physiol. 80, 309–326. https://doi.org/10.1146/annurev-physiol-022516-034227 (2018).
Jung, G. S., Hwang, Y. J., Choi, J. H. & Lee, K. M. Lin28a attenuates TGF-β-induced renal fibrosis. BMB Rep. 53(11), 594–599. https://doi.org/10.5483/BMBRep.2020.53.11.153 (2020).
Chevalier, R. L., Forbes, M. S. & Thornhill, B. A. Ureteral obstruction as a model of renal interstitial fibrosis and obstructive nephropathy. Kidney Int. 75(11), 1145–1152. https://doi.org/10.1038/ki.2009.86 (2009).
Tang, P. M. et al. Neural transcription factor Pou4f1 promotes renal fibrosis via macrophage-myofibroblast transition. Proc. Natl. Acad. Sci. U S A 117(34), 20741–20752. https://doi.org/10.1073/pnas.1917663117 (2020).
Wu, M. et al. Renal asymmetric dimethylarginine inhibits fibrosis. FEBS Open Bio 10(10), 2003–2009. https://doi.org/10.1002/2211-5463.12949 (2020).
Moridaira, K. et al. ACE inhibition increases expression of the ETB receptor in kidneys of mice with unilateral obstruction. Am. J. Physiol. Renal Physiol. 284(1), F209-217. https://doi.org/10.1152/ajprenal.00352.2001 (2003).
Tan, K. et al. Yiqi Juanshen decoction alleviates renal interstitial fibrosis by targeting the LOXL2/PI3K/AKT pathway to suppress EMT and inflammation. Sci. Rep. 15(1), 4248. https://doi.org/10.1038/s41598-025-86622-7 (2025).
Hou, L. & Du, Y. Neuropilin 1 promotes unilateral ureteral obstruction-induced renal fibrosis via RACK1 in renal tubular epithelial cells. Am. J. Physiol. Renal Physiol. 325(6), F870–F884. https://doi.org/10.1152/ajprenal.00069.2023 (2023).
Ding, H. et al. Inhibiting aerobic glycolysis suppresses renal interstitial fibroblast activation and renal fibrosis. Am. J. Physiol. Renal. Physiol. 313(3), F561-f575. https://doi.org/10.1152/ajprenal.00036.2017 (2017).
Cao, H. et al. Tuberous sclerosis 1 (Tsc1) mediated mTORC1 activation promotes glycolysis in tubular epithelial cells in kidney fibrosis. Kidney Int. 98(3), 686–698. https://doi.org/10.1016/j.kint.2020.03.035 (2020).
Yang, S. et al. Inhibition of PFKP in renal tubular epithelial cell restrains TGF-β induced glycolysis and renal fibrosis. Cell Death Dis. 14(12), 816. https://doi.org/10.1038/s41419-023-06347-1 (2023).
Yang, Q. et al. Myeloid PFKFB3-mediated glycolysis promotes kidney fibrosis. Front Immunol. 14, 1259434. https://doi.org/10.3389/fimmu.2023.1259434 (2023).
Heinrich, P. C., Behrmann, I., Müller-Newen, G. & Schaper, F. Interleukin-6-type cytokine signalling through the gp130/Jak/STAT pathway. Biochem. J. 334(Pt 2), 297–314. https://doi.org/10.1042/bj3340297 (1998).
O’Donoghue, R. J. et al. Genetic partitioning of interleukin-6 signalling in mice dissociates Stat3 from Smad3-mediated lung fibrosis. EMBO Mol. Med. 4(9), 939–951. https://doi.org/10.1002/emmm.201100604 (2012).
Pechkovsky, D. V. et al. STAT3-mediated signaling dysregulates lung fibroblast-myofibroblast activation and differentiation in UIP/IPF. Am. J. Pathol. 180(4), 1398–1412. https://doi.org/10.1016/j.ajpath.2011.12.022 (2012).
Chen, W. et al. Blocking interleukin-6 trans-signaling protects against renal fibrosis by suppressing STAT3 activation. Theranostics 9(14), 3980–3991. https://doi.org/10.7150/thno.32352 (2019).
Rao, P. et al. Promotion of β-Catenin/Forkhead Box protein O signaling mediates epithelial repair in kidney injury. Am. J. Pathol. 191(6), 993–1009. https://doi.org/10.1016/j.ajpath.2021.03.005 (2021).
Guo, F. et al. Japanese encephalitis virus induces apoptosis by inhibiting Foxo signaling pathway. Vet. Microbiol. 220, 73–82. https://doi.org/10.1016/j.vetmic.2018.05.008 (2018).
Funding
This project was supported by Natural Science Foundation of Hubei Province (No.2020CFB215) and Postdoctoral Program of General Hospital of Central Theater Command (No. BSH021).
Author information
Authors and Affiliations
Contributions
Hongzhe Tian: Article writing and revision; Bo Liu: Experimental analysis; Wei Liu: Data collation; JiaRong Yang: Literature review; Tiejun Pan: Article design.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statement
This study was approved by the Ethics Committee of Wuhan Myhalic Biotechnology Co., Ltd (Approval Number: HLK-20221011-001). The study is reported in accordance with ARRIVE guidelines and all methods were performed in accordance with the relevant guidelines and regulations.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tian, H., Liu, B., Liu, W. et al. SHP1 overexpression ameliorates renal fibrosis via inhibition of STAT3 signaling and glycolysis reprogramming. Sci Rep 15, 40429 (2025). https://doi.org/10.1038/s41598-025-21520-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-21520-6