Abstract
The ICU environment is disruptive to a patient’s biological rhythms where sleep-wake cycles are often desynchronized from the environmental day-night changes. This puts patients at increased risk to develop delirium with consequent fiscal pressure for the health care system. An underappreciated dimension is how time-specific patient phenotypes in the critical care environment relate to clinical outcomes. We set out to analyze how rhythmic components (or the lack thereof) in physiological data streams sampled at high resolution in the ICU were associated with the future incidence of delirium and death. To offer cues for further interrogation into mechanism and risk prognosis, we examined differences in 24-hour fluctuations of clinical labs in ICU patient populations at risk. Rhythmic components using dipping ratios and JTK_CYCLE statistics were derived from 24-hour blood pressure and heart rate measurements available from ICU patient admissions recorded in the MIMIC IV database. Logistic adjusted regression models assessed the association between disrupted vital sign rhythms and the future incidence of delirium during the same hospital admission and death. Aggregation of numeric clinical lab measurements across the first 24 h from all patient admissions allowed modeling of rhythmic patterns and subsequent association studies to link potential biochemical mechanisms to perturbed vital sign rhythms and adverse ICU outcomes. Patients with reverse blood pressure dipping were at a 40% higher risk to have a diagnosis of delirium (Odds Ratio: 1.38, 95% CI 1.13–1.71) and a 13% increased risk of death (Odds Ratio: 1.13, 95% CI 1.02–1.26). Compared to the patient population with nocturnal blood pressure dip, reverse dippers showed 24-hour biochemistry profiles suggestive of altered circadian programs specifically in clinical parameters of renal, metabolic, and hemostatic function. Reverse blood pressure dipping can be an early sign for the future development of delirium in the ICU and is accompanied by disrupted biorhythms across multiple organ systems. Dampened and reversed heart rate and blood pressure rhythms are associated with a higher risk for death in ICU patients. Considering the inclusion of these risk factors in preventive care may improve patient outcomes and reduce burden on the health care system.
Similar content being viewed by others
Introduction
The environment in the intensive care unit (ICU) is disruptive to a patient’s biological rhythms. The most common concern in the ICU are sleep-wake cycles desynchronized from the environmental day-night changes. Large-scale quantification of disrupted sleep-wake cycles remains a challenge1. However, physiological oscillators, such as heart rate, blood pressure, and body temperature, are recorded in the ICU at high frequencies1,2,3 and may serve as proxy features to quantify the disruption of diurnal rhythms, allowing evaluation of the association of circadian disruption with clinical outcomes.
About a third of critically ill patients admitted to the ICU develop delirium, a condition described as a mental state of confusion with reduced awareness of surroundings4. Characteristic is a sudden onset over hours to days, and patients diagnosed with delirium face an increased incidence of dementia and mortality among other poor outcomes4,5,6. Delirium is multifactorial but notable risk factors for developing this condition include day to night disorientation and sleep deprivation which both indicate disrupted circadian programs4,7.
Parsing diurnal variability in time series data allows to identify time-specific differences and their association to disease outcomes. Indicators of diurnal variability include amplitude (half the distance from peak to trough), phase (the time-of-day at which the peak occurs), dipping ratio (the extent of the night-to-day change) and a p-value indicating whether the time-series is rhythmic or not. In 24-hour blood pressure monitoring, for example, the nocturnal dip is a commonly used metric to gauge a patient’s risk to develop cardiovascular and kidney disease8,9,10,11. However, more comprehensive methods to interrogate daily oscillations have been developed. Adopting the JTK_CYCLE algorithm that assesses the significance of rhythmic components, defined as repeat cycles in time series data where a steady increase towards a peak is followed by a steady decrease towards a trough12, we were able to improve risk stratification for patients with chronic renal insufficiency based on their 24-hour blood pressure profile13. For large cohort studies, we demonstrated the utility of rhythm measurement by associating dampened amplitudes of wrist temperature traces with the future onset of 73 diseases14. We extended this work to ask if we can identify rhythmic components in time series data collected in the ICU environment and how these relate to clinical outcomes.
We leveraged MIMIC IV as an electronic health record dataset to parse time-specific vital signs and patient outcomes. We found that in the ICU environment, patients with reverse blood pressure dipping - known to be a high-risk phenotype for CVD - were at higher odds to be diagnosed in the future with delirium and to have higher mortality compared to patients with blood pressure dipping at night. Furthermore, we discerned diurnal fluctuations in clinical labs and found perturbed rhythms in markers of kidney metabolism and hemostasis which offer cues for further interrogation into mechanism and risk prognosis.
Methods
The MIMIC-IV database
The Medical Information Mart for Intensive Care-IV (MIMIC-IV) database contains Electronic Health Record data of real ICU patient admissions from 73,181 ICU admissions at the Beth-Israel Deaconess Medical Center in Boston between 2008 and 201915,16. It contains vital signs recorded in patient charts at hourly intervals embedded in comprehensive in-hospital data such as diagnoses, procedures, diagnostic tests, medications, and intakes/outputs. Each patient is also followed up for out of hospital mortality for a maximum of 1 year post discharge from the last admission on record by tracking social security mortality data. The data were de-identified to maintain HIPAA standards with identifiers replaced by random ciphers. Dates were also shifted randomly such that a single date shift is applied to all data from the same individual. However, time of day and seasonality were preserved. We pulled the data from the study website (https://physionet.org/content/mimiciv/2.2/, version 2.2, downloaded January 5, 2023). The MIMIC IV study was approved by the institutional review board at the Beth Israel Deaconess Medical Center and was granted a waiver of informed consent. NE was granted access to the MIMIC-IV data after submitting a request to https://physionet.org/content/mimiciv/2.2/, along with proof of training in the ethical conduct of human subject research. All methods were performed in accordance with the relevant guidelines and regulations.
Rhythmic assessments
We extracted the hourly recordings of systolic blood pressure (both arterial and non-invasive), temperature (the body site of measurement is not further specified), and heart rate. We excluded any readings that were outside the range of 50 mmHg to 200 mmHg for systolic blood pressure, 30 °C to 43 °C for temperature, and 50 bpm to 150 bpm for heart rate. These criteria eliminated 0.25% of all blood pressure readings, 0.08% of temperature readings, and 1.17% of heart rate readings. For each individual vital sign, we only included ICU admissions that had at least 14 daytime and 6 nighttime measurements to ensure an representative coverage of both day and night9. We specified nighttime as the interval between midnight and 6 am and daytime as that between 6 am and midnight. Because only 20% of ICU admissions fulfilled our inclusion criteria for temperature measurements, we excluded temperature from further analysis to decrease the risk of selection bias (Figure S1 bottom panel). We calculated non-parametric Jonckheere-Terpstra-Kendall (JTK) p-values, amplitude, as well dipping ratio (nighttime mean divided by daytime mean) for included admissions. Values for each admission were binned by hour of the day and the mean of each bin was used as input to JTK, thereby ensuring all admissions had between 20 and 24 datapoints, allowing p-values comparisons across admissions13. We used the JTK p-value to determine rhythmicity for each time series of vital signs (p ≤ 0.05).
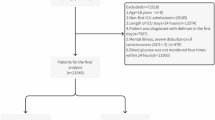
To account for non-linear associations with amplitude, we categorized amplitude based on the quartiles of the studied populations (Table S1). Dipping status for both blood pressure and heart rate was categorized as dippers (dipping ratio ≤ 0.9, a 10% or higher drop during the night), non-dippers (0.9 < dipping ratio ≤ 1, small change between the day and night) and reverse dippers (dipping ratio > 1, a rise during the night)9. To study whether the disruption of rhythms in the first 24 h is associated with future adverse outcomes, we limited our study to vital sign data collected only during the first 24 h of ICU admission. Admissions with less than 24 h between first and last measurements were excluded from analysis. For each analysis, patients fulfilling our rhythm quality control criteria for more than one admission had one of their admissions chosen at random for inclusion. Figure S2 graphically represents the temporal design of our study.
Clinical outcomes and statistical analyses
Incident delirium was the primary outcome of interest. MIMIC-IV reports results from each component of the confusion assessment method (CAM)17. CAM was developed as a clinical tool to detect delirium and showed high sensitivity (94–100%) and specificity (90–95%)18. While it should not be interpreted epidemiologically in the same way as an objective physiologic measurement, it is a widely used tool in clinical and research settings and is recommended for use by multiple practice guidelines19. In the MIMIC-IV database, CAM components assessed by healthcare providers are extracted from patient charts. We use these components as the bases for our delirium assessment. As per CAM guidelines, we defined a positive test as the presence of both a fluctuation in mental status from baseline and inattention and the presence of either a decreased level of consciousness or disorganized thinking17. For our delirium analysis, we included patients with a negative CAM in the first 24 h and with a repeat CAM after the first 24 h. We then ran a logistic regression model containing each tested rhythmic parameter individually with the components of the OASIS score (Oxford Acute Severity of Illness Score); variables: Pre-ICU length of stay, age, Glasgow coma scale, heart rate, mean arterial pressure (MAP), respiratory rate, temperature, urine output, mechanical ventilation, elective surgery20. To adjust better, vital sign components were computed as actual 24-hour averages and included in the model with normal cubic spline functions; other components were derived from the PostgreSQL code provided by the MIMIC code repository21. Because sedation and pressor use relate to both delirium and vital sign values, we included the Richmond Agitation-Sedation Score (RAS) and vasopressor use as covariates in our delirium model. We also studied mortality during the ICU visit or up to one month (30 days) afterwards from the day of admission using the same methodology adjusting for the same illness severity criteria in OASIS with the addition of vasopressor use as covariate. We specified 30 days as our time limit for this outcome to assess a longer-term outcome while staying in a time period that is still close to the time the included vital sign measurements were collected. Resulting p-values for both outcome analyses were adjusted for multiple testing by the Benjamini-Hochberg method22. To decrease the risk of missing true positives while we are exploring multiple rhythm metrics, a q-value cutoff of 0.1 was considered as the threshold for statistical significance. We report all p-values and q-values in the text and supplementary material. We also report results of Hosmer-Lemeshow tests (HL), variance inflation factor (VIF), and events per variable (EPV) as logistic regression diagnostics.
To explore rhythms in biochemical processes and their association with vital sign rhythms we aggregated all numeric clinical lab measurements across the first 24 h (the time period of vital sign assessments) from all patients with a vital sign rhythm pattern of interest after removing outlier values (beyond 1.5 times the second and third quartiles) and labs with less than 100 samples. If a patient had multiple visits, all were included, and dipping status was determined for each visit separately. We then ran a Cosinor analysis, with random intercept effects for subjects and admissions, using the lme4 R package23. To test for amplitude and rhythm differences among the patient groups, we used an F-test with the lmerTest package and Kenward-Rogers degrees of freedom to compare the nested model where both groups (dipping or reverse dipping) had separate cosinor fits to the model where all groups had the same cosinor fit but separate intercepts24. We considered a q-value cutoff of 0.1 as the threshold for significant rhythm differences across compared groups. The Cosinor method is widely used in circadian research to extract rhythm metrics from a time series such as, phase, amplitude, and a test statistic (p-value) assessing the whole time-series for rhythmicity. It can also be used to compare two time series for differences in rhythmicity. Of note, our method assesses rhythmicity in laboratory test values irrespective of where these values fall in comparison to normal clinical ranges.
Results
Patient data base characteristics
Data recordings from most ICU patient admissions met the day-/nighttime coverage criteria for blood pressure (45,287 out of 73,181, 61.9%) and for heart rate (47,130 out of 73,181, 64.4%), (Figure S3). Illness severity parameters showed that a less healthy population (significantly higher OASIS and SOFA scores) was included and analyzed after filtering for rhythm quality control criteria for both heart rate and blood pressure (Table 1). To assess any potential effect our selection criteria has on our outcomes, we ran univariate logistic regression models and found that patients fulfilling our selection criteria were not significantly more or less likely to test positive for delirium (β = − 0.11, p = 0.208) but were at higher risk to die within a month (β=-0.20, p < 0.001). Table 2 shows rhythmic parameter summary statistics for the included sample. After excluding repeat stays and stays with unmet CAM criteria, 7,346 out of 73,181 (10%) and 7,552 out of 73,181 (10.3%) patient admissions remained for blood pressure and heart rate, respectively, which also qualified for delirium analysis (Figure S3). Most patients (76%) had only one ICU admission (Figure S4). A total of 33,970 (46.4% of all admissions) and 35,101 (48% of all admissions) admissions were submitted to the mortality analysis for blood pressure (BP) and heart rate (HR), respectively (Figure S5).
Reverse blood pressure dipping phenotype predictive of future delirium
Incident delirium was detected in 15.9% (1,171 out of 7,346) and 15.7% (1,189 out of 7,552) of the included BP and HR ICU admissions, respectively (Figure S3). Patients with a reverse blood pressure dipping phenotype had a 38% greater risk for later testing positive for delirium during their ICU admission (Odds Ratio: 1.38, 95% CI 1.13–1.71, q = 0.0266, Fig. 1 left, Table S2) compared to patients with a dipping blood pressure profile. Patients with a non-dipping BP profile, in contrast, did not show this association when compared to BP dippers (Odds Ratio: 0.97, 95% CI 0.79–1.21, q = 0.845). Assessment of rhythmic components of BP suggested a link between non-oscillating blood pressure profile (JTK p-value > 0.05) and delirium detection (19% elevated risk), but this relationship has a higher false discovery rate (Odds Ratio: 1.19, 95%CI 1.01–1.42, q = 0.262). Though higher amplitudes of blood pressure oscillations showed a lower risk of delirium incidence compared to low amplitudes, this effect was small and non-significant (Odds Ratio: 0.69, 95%CI 0.78–1.15, q = 0.692 comparing JTK Amplitude Q1 versus Q4 in Fig. 1 left).
Associations between different rhythm parameters and the future development of delirium. The left panel displays results from the logistic regression models testing blood pressure rhythmic parameters, the right panel displays results from heart rate rhythmic parameters. Reference categories are Dippers, JTK p-value ≤ 0.05, and JTK Amplitude Q1 respectively. q-values are the results of multiple testing corrections of p-values using the Benjamini-Hochberg method. * q-value ≤ 0.1.
Examining heart rate, patients with a reverse dipping phenotype showed a 19% higher risk for later testing positive for delirium during their ICU admission (Odds Ratio: 1.19, 95% CI 0.97–1.46, q = 0.396, Fig. 1 right) but that effect was not statistically significant.
Dampened rhythms and reverse blood pressure dipping linked to increased risk of death
The mortality rate among patients with sufficient blood pressure recordings but excluding repeat ICU admissions was 15.9% (5,403 deaths within a month from ICU admission) compared to 16.3% (5,708 deaths) for the patient cohort meeting inclusion criteria for heart rate. Patients with a reverse dipping blood pressure profile had a 13% higher risk to die in the ICU setting within a month (Odds Ratio: 1.13, 95% CI 1.02–1.26, q = 0.07, Fig. 2, Table S3). Also, compared to patients with a blood pressure amplitude in the first quartile, those in the third quartile were at a significantly lower risk of death (Odds Ratio 0.88 95% CI 0.80–0.97, q = 0.07) and there was a non-significant trend in the same direction for those in the second and fourth quartile. Furthermore, patients with a more robust heart rate rhythm belonging to the third amplitude quartile were protected from dying (Odds Ratio: 0.83, 95% CI 0.75–0.92, q = 0.004) as compared to those in the lowest heart rate amplitude quartile (Fig. 2, Table S3), and patients in the second and fourth quartile had a non-significant trend in the same direction.
Associations between different rhythm parameters and 1 year mortality. The left panel displays results from the logistic regression models testing blood pressure rhythmic parameters, the right panel displays results from heart rate rhythmic parameters. Reference categories are Dippers, JTK p-value ≤ 0.05, and JTK Amplitude Q1 respectively. q- values are the results of multiple testing corrections of p-values using the Benjamini-Hochberg method. * q-Value ≤ 0.1.
Reverse blood pressure dipping is associated with multi-system rhythmic disruptions
We explored whether reverse blood pressure dipping correlates with altered diurnal variability in biochemical processes assessed by clinical laboratory measurements (Figure S6). The variety of clinical labs can provide insights into the function of multiple organ systems. By assessing lab rhythms in lab test values, we can estimate the rhythmic state of a specific organ system. Because a single patient would rarely have enough repeated laboratory measurements to cover a 24-hour time series, we analyzed measurements across all patients belonging to either blood pressure dippers (controls) or reverse dippers (cases) in a mixed effects cosinor model. Among 101 laboratory tests recorded in the first 24 h in patients with representative coverage of both day and night (at least 14 daytime and 6 nighttime measurements as used for blood pressure rhythm quality control criteria), a total of 45 laboratory tests demonstrated a statistically significant difference in rhythmicity in the cosinor fits comparing BP reverse dippers to dippers (q-value < 0.01, Fig. 3, Table S4, Figure S6). Candidates include hematocrit (q-value ≤ 0.001), hemoglobin (q-value < 0.001), arterial O2 pressure (q-value < 0.001), platelet count (q-value < 0.001), glucose (q-value < 0.001), creatinine (q-value < 0.001), prothrombin time (q-value < 0.001), INR (q-value < 0.001), lymphocytes (q-value = 0.002), WBC (q-value ≤ 0.001), PTT (q-value < 0.001), and monocytes (q-value < 0.001), all reported to be oscillatory though some with conflicting results particularly in PT, PTT, and INR25,26,27,28,29.
Significance levels of differences between blood pressure dipping statuses in cosinor fits of clinical laboratory measurements across the first 24 h. The red line indicates a q-value cutoff of 0.1 for statistical significance. The actual values behind the graph are in Table S4.
Next, we examined rhythmic characteristics of these 45 labs to ascertain differences and commonalities between blood pressure dippers and reverse dippers (Fig. 4A). Most of these clinical labs (n = 29, 69%) displayed significant diurnal variability consistently in both groups (cosinor q-value ≤ 0.1). About 14% (n = 6) were only rhythmic in dippers. Among these were albumin and cortisol. Figure 4B shows the significant dampening of albumin and cortisol rhythms seen by the marked effacements of both peaks and troughs. The diurnal fluctuations in serum albumin levels in a healthy population likely contribute to time-specific blood pressure regulation through associated changes in oncotic pressure30. This functional relationship might deteriorate in reverse dippers as suggested by the lack of oscillatory albumin serum concentrations (Fig. 4B). Cortisol’s diurnal secretion patterns in healthy volunteers likely drive the time-specific sensitization of the vasculature to the noradrenergic system31,32 a mechanism linked to blood pressure control33. Here, the lack of significant oscillations in reverse dippers compared to dippers may suggest that disrupted diurnal cortisol levels contribute to this pathology (Fig. 4B).
Amplitude and phase differences calculated from cosinor fits for laboratory measurements that showed statistically significant differences in cosinor fits between blood pressure dippers and reverse dippers. Values shown are relative to dippers and sorted by decreasing amplitude and phase differences. The actual values behind the graphs are in Table S4.
About 17% (n = 7) lab tests showed significant oscillations only in patients with the reverse blood pressure dipping phenotype (Fig. 4A). This is an unexpected finding and warrants further examination in future studies since time-of-day specific fluctuations have been reported in the healthy for the coagulation system34, immune system35, lactic acid and uric acid36,37. Conceivable, however, is that gain in diurnal fluctuations in the coagulation system are indicative of transient states of hypercoagulability, a known hemostatic abnormality in critical illness leading to poorer outcomes4,38.
A total of 29 clinical labs were detected as having significantly different rhythms between dippers and reverse dippers and yet were rhythmic in both. This indicates changes in rhythm parameters while maintaining rhythmicity.To examine the rhythmic components of these 29 clinical labs that were rhythmic in both dippers and reverse dippers we quantified cosinor amplitudes and acrophases (time of day of the peak) (Fig. 4C, Table S4). Kidney function as measured by serum creatinine showed a 60% decrease in amplitude in reverse dippers compared to dippers. This was accompanied by a phase advance of 11.8 h in reverse dippers compared to dippers. BUN similarly demonstrated a reduction in amplitude (60% in reverse dippers compared to dippers). Here, the phase differences were smaller (3.3 h in dippers respectively compared to dippers). Lab values indicative of acid base balance showed higher amplitude fluctuations in reverse dippers compared to dippers, for instance, 145% higher amplitude in arterial base excess, 76% in arterial pH amplitude and 36% in arterial CO2 pressure amplitude. Electrolyte levels diverged, for example, by a 11-hour phase advance in non-ionized calcium levels in reverse dippers compared to dippers accompanied by a 60% amplitude dampening. This contrasted with a 159% increase in ionized calcium amplitude in reverse dippers with minimal phase changes. Several hemostatic parameters showed distinct rhythmic components in reverse dippers compared to dippers. Prothrombin time (PT) fluctuated with a 333% higher amplitude in reverse dippers and amplitude was 40% higher in partial thromboplastin time (PTT). Thus, reverse blood pressure dipping was associated on a population level with perturbed rhythms in clinical parameters of renal, metabolic, and hemostatic function indicating disrupted rhythms in multiple organ systems.
Discussion
Our present study demonstrates that patients with a reverse blood pressure dipping phenotype had a 38% greater risk of later testing positive for delirium during their ICU admission compared to patients with the blood pressure dipping phenotype even with adjusting for illness severity, blood pressure levels, and for sedative and pressor use. We showed that patients with reverse blood pressure dipping and dampened amplitudes for blood pressure and heart rate within the first 24 h of ICU admission had a 13% higher risk of death within one month of their ICU admission. We postulate that the link between reverse BP dipping and delirium/death is accompanied by perturbed rhythms in clinical parameters of renal, metabolic, and hemostatic function, thus offering potential biomarkers for further interrogation of mechanism and risk prognosis.
Our finding that reverse blood pressure dipping precedes a positive test for delirium is supported by published data. We also take studies on long term cognitive decline and dementia into consideration since delirium is an established risk factor for developing chronic forms of cognitive impairment5. A longitudinal cohort study in older men in Sweden found a 64% higher risk for future dementia (and Alzheimer’s disease) among reverse dippers (Hazard Ratio: 1.64, 95% CI 1.14–2.34)39. The authors suggested reverse dipping as an independent risk factor for dementia (and Alzheimer’s)39. Several cross sectional studies support the link between reverse blood pressure dipping and chronic forms of cognitive decline40,41,42. A preliminary report of a functional study using the head-up tilt test detected a poorer autonomic function in participants with delirium compared to controls43.
The higher mortality we observed in patients with reverse blood pressure dipping and blunted diurnal rhythms of blood pressure and heart rate highlights how time series of vital sign measurements routinely assessed in the critical care environment harbor information to understand patients’ risk profile. Though our results strengthen comparable findings in the eICU Collaborative Research Database44, other studies in the same database surprisingly reported that a higher amplitude of heart rate oscillations was associated with elevated in-hospital mortality45. These discrepant findings might be in part driven by varying numbers of day- and nighttime measurements. Our study addresses this potential limitation by ensuring both nighttime and daytime periods were adequately represented in every included time series (at least 14 daytime and 6 nighttime measurements). Here, we advocate for a concerted effort to address research challenges in this space46.
Reverse blood pressure dipping is an alteration of the normal diurnal phenotype and is likely accompanied by time-specific changes in biochemical processes. Indeed, our results showed that reverse blood pressure dipping was associated with altered diurnal phenotypes in various clinical laboratory measurements. An important finding, for example, is the absence of rhythmic cortisol levels in reverse dippers (Fig. 4). While we cannot directly assess catecholamines in this database, activation of the hypothalamic pituitary axis usually accompanies increased sympathetic tone4. Based on that we speculate that a disturbance in cortisol rhythms is also mirrored by a disturbance in sympathetic tone that is oscillatory and plays a substantial role in regulating the diurnal variability in blood pressure47. This can be seen in the physiologic rise in both cortisol and catecholamines in the hours preceding the daily peak in blood pressure and can lead to the expectation that a disruption in cortisol and autonomic nervous system rhythms may be followed by disruptions in blood pressure rhythms47. Both elevated cortisol and sympathetic tone are associated with post-operative delirium4,48. Reverse blood pressure dipping can thus be a consequence of disturbed autonomic nervous system rhythms that may increase the risk for developing delirium and other adverse outcomes. Given the specific roles the autonomic nervous system exerts on different physiologic systems including metabolism, renal function, and lung function49, we speculate that the autonomic nervous system may thus represent a unifying candidate biomarker justifying further interrogation in future experimental studies9. Studies on the baroreflex support this direction of further exploration. The baroreflex has been proven to regulate diurnal rhythms in blood pressure50,51,52, Baroreflex sensitivity is reduced in critical illness53, potentially linking reverse blood pressure dipping to adverse outcomes in critical illness.
Our study has several strengths. We used a well curated, well documented, and commonly used database containing patient trajectories from a respected academic center. The size of the database permitted our application of strict inclusion criteria to ensure an adequate coverage of the first 24 h for our rhythmic assessments. The database also recorded time-stamped CAM assessments, a well validated commonly used instrument to detect delirium. This enabled us to assign delirium diagnoses with a higher level of certainty than relying on diagnostic codes, which might have missed true positives4. Further prospective cohort studies are needed to expand on our findings. Such studies should seek to confirm CAM positive results by a comprehensive specialist clinical evaluation, a step that is difficult in publicly available EHR datasets because CAM positivity is not equivalent to a delirium diagnosis. Our study design does not permit causal conclusions. Due to a low frequency of body temperature measurements and the consequent lack of confidence in their rhythmic assessments we had to abandon this biomarker. We also found that body temperature data were skewed; patients that had their temperature measured frequently were sicker. Another limitation inherent to this database is the observational design with a high degree of variability in patients, disease conditions, co-morbidities, and treatments. Overall, sicker patients might be over-represented since they likely had more interactions documented in the database due to higher care intensity. While our selection criteria did not appear to affect our delirium results, patients who fulfilled these criteria had a higher mortality rate indicating the higher risk for selection bias in our mortality analysis. Given the nature of this database, it is also difficult to assess a past history of neurologic disease. While pooling lab results from different patients is valuable to enable time-series statistics, options should be explored for repeat within-person biosampling. Lastly, the biomarker discovery is predetermined by the clinical lab panel.
Conclusion
Reverse blood pressure dipping can be an early sign for the future development of delirium in the ICU and is accompanied by disrupted biorhythms across multiple organ systems. Reverse dipping could be a measurable sign of an underlying state that later manifests in a positive test for delirium. Dampened and reversed heart rate and blood pressure rhythms are associated with a higher risk for death in ICU patients. Considering the inclusion of these risk factors in preventive care may improve patient outcomes and reduce burden on the health care system.
Data availability
Data used in this study is available for credentialed users at https://physionet.org/content/mimiciv/2.2/. All statistics derived from the dataset are available with the supplemental material attached to this manuscript. The code developed for this study is freely available at: https://zenodo.org/records/14750969.
Abbreviations
- BP:
-
Blood pressure
- CAM:
-
Confusion assessment method
- JTK:
-
Jonckheere–Terpstra–Kendall
- HR:
-
Heart rate
- OASIS:
-
Oxford acute severity of illness score
- PT:
-
Prothrombin time
- PTT:
-
Partial thromboplastin time
References
Reid, K. J. Assessment of circadian rhythms. Neurol. Clin. 37(3), 505–526. https://doi.org/10.1016/j.ncl.2019.05.001 (2019).
Hayter, E. A. et al. Distinct circadian mechanisms govern cardiac rhythms and susceptibility to arrhythmia. Nat. Commun. 12 (1), 2472. https://doi.org/10.1038/s41467-021-22788-8 (2021).
Smolensky, M. H., Hermida, R. C. & Portaluppi, F. Circadian mechanisms of 24-hour blood pressure regulation and patterning. Sleep. Med. Rev. 33, 4–16. https://doi.org/10.1016/j.smrv.2016.02.003 (2017).
Wilson, J. E. et al. Delirium. Nat. Rev. Dis. Primers 6(1), 90. https://doi.org/10.1038/s41572-020-00223-4 (2020).
Fong, T. G. & Inouye, S. K. The inter-relationship between delirium and dementia: The importance of delirium prevention. Nat. Rev. Neurol. 18(10), 579–596. https://doi.org/10.1038/s41582-022-00698-7 (2022).
Jackson, T. A., Wilson, D., Richardson, S. & Lord, J. M. Predicting outcome in older hospital patients with delirium: A systematic literature review. Int. J. Geriatr. Psychiatry. 31(4), 392–399. https://doi.org/10.1002/gps.4344 (2016).
Daou, M., Telias, I., Younes, M., Brochard, L. & Wilcox, M. E. Abnormal sleep, circadian rhythm disruption, and delirium in the ICU: Are they related? Front. Neurol. 11, 549908. https://doi.org/10.3389/fneur.2020.549908 (2020).
Komori, T., Eguchi, K., Saito, T., Hoshide, S. & Kario, K. Riser pattern is a novel predictor of adverse events in heart failure patients with preserved ejection fraction. Circ. J. 81(2), 220–226. https://doi.org/10.1253/circj.CJ-16-0740 (2017).
Rahman, M. et al. Prognostic significance of ambulatory BP monitoring in CKD: A report from the chronic renal insufficiency cohort (CRIC) study. J. Am. Soc. Nephrol. 31(11), 2609–2621. https://doi.org/10.1681/ASN.2020030236 (2020).
Sawamura, A. et al. Abnormal circadian blood pressure profile as a prognostic marker in patients with nonischemic dilated cardiomyopathy. Cardiology 136(1), 1–9. https://doi.org/10.1159/000446868 (2017).
Ueda, T. et al. Differences in blood pressure riser pattern in patients with acute heart failure with reduced mid-range and preserved ejection fraction. ESC Heart Fail. 6(5), 1057–1067. https://doi.org/10.1002/ehf2.12500 (2019).
Hughes, M. E., Hogenesch, J. B. & Kornacker, K. JTK_CYCLE: An efficient nonparametric algorithm for detecting rhythmic components in genome-scale data sets. J. Biol. Rhythms 25(5), 372–380. https://doi.org/10.1177/0748730410379711 (2010).
El Jamal, N. et al. Prognostic utility of rhythmic components in 24-h ambulatory blood pressure monitoring for the risk stratification of chronic kidney disease patients with cardiovascular co-morbidity. J. Hum. Hypertens. https://doi.org/10.1038/s41371-023-00884-0 (2024).
Brooks, T. G. et al. Diurnal rhythms of wrist temperature are associated with future disease risk in the UK biobank. Nat. Commun. 14(1), 5172. https://doi.org/10.1038/s41467-023-40977-5 (2023).
Johnson, A. et al. MIMIV-IV (version 2.2). Physionet. https://doi.org/10.13026/6mm1-ek67. (2023)
Johnson, A. E. W. et al. MIMIC-IV, a freely accessible electronic health record dataset. Sci. Data. 10(1), 1. https://doi.org/10.1038/s41597-022-01899-x (2023).
SK I. The Short Confusion Assessment Method (Short CAM) Training Manual and Coding Guide (Hospital Elder Life Program, 2014).
Inouye, S. K. et al. Clarifying confusion: the confusion assessment method. A new method for detection of delirium. Ann. Intern. Med. 113(12), 941–948. https://doi.org/10.7326/0003-4819-113-12-941 (1990).
Wei, L. A., Fearing, M. A., Sternberg, E. J. & Inouye, S. K. The confusion assessment method: A systematic review of current usage. J. Am. Geriatr. Soc. 56(5), 823–830. https://doi.org/10.1111/j.1532-5415.2008.01674.x (2008).
Johnson, A. E., Kramer, A. A. & Clifford, G. D. A new severity of illness scale using a subset of acute physiology and chronic health evaluation data elements shows comparable predictive accuracy. Crit. Care Med. 41 (7), 1711–1718. https://doi.org/10.1097/CCM.0b013e31828a24fe (2013).
Johnson, A. E., Stone, D. J., Celi, L. A. & Pollard, T. J. The MIMIC code repository: Enabling reproducibility in critical care research. J. Am. Med. Inf. Assoc. 25 (1), 32–39. https://doi.org/10.1093/jamia/ocx084 (2018).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. Royal Stat. Soc. Ser. B: Stat. Methodol. 57(1), 289–300. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x (1995).
Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting linear Mixed-Effects models Usinglme4. J. Stat. Softw. 67(1). https://doi.org/10.18637/jss.v067.i01 (2015).
Cornelissen, G. Cosinor-based rhythmometry. Theor. Biol. Med. Model. 11, 16. https://doi.org/10.1186/1742-4682-11-16 (2014).
Cristancho, E., Riveros, A., Sanchez, A., Penuela, O. & Boning, D. Diurnal changes of arterial oxygen saturation and erythropoietin concentration in male and female Highlanders. Physiol. Rep. 4(17). https://doi.org/10.14814/phy2.12901 (2016).
Van Cauter, E., Polonsky, K. S. & Scheen, A. J. Roles of circadian rhythmicity and sleep in human glucose regulation. Endocr. Rev. 18(5), 716–738. https://doi.org/10.1210/edrv.18.5.0317 (1997).
Eckerbom, P. et al. Circadian variation in renal blood flow and kidney function in healthy volunteers monitored with noninvasive magnetic resonance imaging. Am. J. Physiol. Ren. Physiol. 319(6), F966–F78. https://doi.org/10.1152/ajprenal.00311.2020 (2020).
Busza, A. et al. 0028 systematic review: time of day differences in complete blood count values. Sleep 46(Supplement_1), A12–A. https://doi.org/10.1093/sleep/zsad077.0028 (2023).
West, A. S., Schonsted, M. I. & Iversen, H. K. Impact of the circadian clock on fibrinolysis and coagulation in healthy individuals and cardiovascular patients—a systematic review. Thromb. Res. 207, 75–84. https://doi.org/10.1016/j.thromres.2021.09.011 (2021).
Jubiz, W., Canterbury, J. M., Reiss, E. & Tyler, F. H. Circadian rhythm in serum parathyroid hormone concentration in human subjects: Correlation with serum calcium, phosphate, albumin, and growth hormone levels. J. Clin. Invest. 51 (8), 2040–2046. https://doi.org/10.1172/JCI107010 (1972).
Yang, S. & Zhang, L. Glucocorticoids and vascular reactivity. Curr. Vasc Pharmacol. 2(1), 1–12. https://doi.org/10.2174/1570161043476483 (2004).
Weitzman, E. D. et al. Twenty-four hour pattern of the episodic secretion of cortisol in normal subjects. J. Clin. Endocrinol. Metab. 33(1), 14–22. https://doi.org/10.1210/jcem-33-1-14 (1971).
Holt-Lunstad, J. & Steffen, P. R. Diurnal cortisol variation is associated with nocturnal blood pressure dipping. Psychosom. Med. 69(4), 339–343. https://doi.org/10.1097/PSY.0b013e318050d6cc (2007).
Kapiotis, S. et al. Morning hypercoagulability and hypofibrinolysis. Diurnal variations in circulating activated factor VII, prothrombin fragment F1 + 2, and plasmin-plasmin inhibitor complex. Circulation 96(1), 19–21. https://doi.org/10.1161/01.cir.96.1.19 (1997).
Scheiermann, C., Kunisaki, Y. & Frenette, P. S. Circadian control of the immune system. Nat. Rev. Immunol. 13(3), 190–198. https://doi.org/10.1038/nri3386 (2013).
Devgun, M. S. & Dhillon, H. S. Importance of diurnal variations on clinical value and interpretation of serum urate measurements. J. Clin. Pathol. 45(2), 110–113. https://doi.org/10.1136/jcp.45.2.110 (1992).
Feneberg, R. et al. Synchronous fluctuations of blood insulin and lactate concentrations in humans. J. Clin. Endocrinol. Metab. 84(1), 220–227. https://doi.org/10.1210/jcem.84.1.5377 (1999).
Levi, M. & Opal, S. M. Coagulation abnormalities in critically ill patients. Crit. Care. 10(4), 222. https://doi.org/10.1186/cc4975 (2006).
Tan, X. et al. Reverse dipping of systolic blood pressure is associated with increased dementia risk in older men: A longitudinal study over 24 years. Hypertension 77(4), 1383–1390. https://doi.org/10.1161/HYPERTENSIONAHA.120.16711 (2021).
Gavriilaki, M. et al. Association between ambulatory blood pressure monitoring patterns with cognitive function and risk of dementia: A systematic review and meta-analysis. Aging Clin. Exp. Res. 35(4), 745–761. https://doi.org/10.1007/s40520-023-02361-7 (2023).
Shanahan, E. et al. 108abnormal diurnal blood pressure variability in patients with recent delirium. Age Ageing. 48(Supplement_1), i32–i5. https://doi.org/10.1093/ageing/afy204.01 (2019).
Paganini-Hill, A. et al. Blood pressure circadian variation, cognition and brain imaging in 90+ year-olds Front. Aging Neurosci. 11, 54. https://doi.org/10.3389/fnagi.2019.00054 (2019).
Shanahan, E. et al. 68 autonomic impairment and its manifestations in patients with delirium. Age Ageing. 48(Supplement_3), iii17–iii65. https://doi.org/10.1093/ageing/afz103.37 (2019).
Beyer, S. E., Salgado, C., Garcao, I., Celi, L. A. & Vieira, S. Circadian rhythm in critically ill patients: Insights from the eICU database. Cardiovasc. Digit. Health J. 2(2), 118–125. https://doi.org/10.1016/j.cvdhj.2021.01.004 (2021).
Yang, Z. et al. The impact of heart rate circadian rhythm on in-hospital mortality in patients with stroke and critically ill: Insights from the eICU collaborative research database. Heart Rhythm. 19(8), 1325–1333. https://doi.org/10.1016/j.hrthm.2022.03.1230 (2022).
Kervezee, L., Dashti, H. S., Pilz, L. K., Skarke, C. & Ruben, M. D. Using routinely collected clinical data for circadian medicine: A review of opportunities and challenges. PLOS Digit. Health 3(5), e0000511. https://doi.org/10.1371/journal.pdig.0000511 (2024).
El Jamal, N., Lordan, R., Teegarden, S. L., Grosser, T. & FitzGerald, G. The circadian biology of heart failure. Circ. Res. 132(2), 223–237. https://doi.org/10.1161/CIRCRESAHA.122.321369 (2023).
Mu, D. L. et al. High serum cortisol level is associated with increased risk of delirium after coronary artery bypass graft surgery: A prospective cohort study. Crit. Care. 14 (6), R238. https://doi.org/10.1186/cc9393 (2010).
Sullivan, R. P., Houser, S. R. & Koch, W. J. Neurotransmission: The autonomic and somatic motor nervous systems. in The Pharmacological Basis of Therapeutics 14th edn (eds Brunton, L. L., Knollmann, B. C.) Goodman & Gilman’s, McGraw-Hill Education, New York, NY, 2023).
Guasti, L. et al. Circadian blood pressure variability is associated with autonomic and baroreflex-mediated modulation of the sinoatrial node. Acta Cardiol. 60(3), 319–324. https://doi.org/10.2143/AC.60.3.2005011 (2005).
Hossmann, V., Fitzgerald, G. A. & Dollery, C. T. Circadian rhythm of baroreflex reactivity and adrenergic vascular response. Cardiovasc. Res. 14(3), 125–129. https://doi.org/10.1093/cvr/14.3.125 (1980).
Makino, M. et al. Circadian rhythms of cardiovascular functions are modulated by the baroreflex and the autonomic nervous system in the rat. Circulation 96(5), 1667–1674. https://doi.org/10.1161/01.cir.96.5.1667 (1997).
Carrara, M., Ferrario, M., Bollen Pinto, B. & Herpain, A. The autonomic nervous system in septic shock and its role as a future therapeutic target: A narrative review. Ann. Intens. Care 11(1), 80. https://doi.org/10.1186/s13613-021-00869-7 (2021).
Funding
Supported in part by the University of Pennsylvania Health System (UPHS). Research reported in this publication was supported by the National Center for Advancing Translational Sciences of the National Institutes of Health under Award Number UL1TR001878. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. GAF held a Merit Award from the American Heart Association during the course of this work.
Author information
Authors and Affiliations
Contributions
Conceptualization: N.E., C.S. Methodology: N.E., C.S., T.B., A.M., M.G. Supervision: G.F., C.S. Writing original draft: N.E., C.S. Writing—review and editing: C.S., G.F., T.B., A.M., M.G.. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
CS is the Robert L. McNeil Jr. Fellow in Translational Medicine and Therapeutics. GAF is the McNeil Professor of Translational Medicine and Therapeutics. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. The other authors declare no competing interests.
Ethical approval
The MIMIC IV study was approved by the institutional review board at the Beth Israel Deaconess Medical Center and was granted a waiver of informed consent.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
El Jamal, N., Brooks, T.G., Mrčela, A. et al. Critically ill patients with a reverse blood pressure dipping phenotype at increased risk for delirium and death. Sci Rep 15, 40433 (2025). https://doi.org/10.1038/s41598-025-23004-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-23004-z