Abstract
To compare the efficacy of stereotactic body radiotherapy (SBRT) and laparoscopic liver resection (LLR) for patients with single hepatocellular carcinoma (HCC) ≤ 5 cm. A retrospective analysis was conducted on a cohort of patients with HCC who underwent either LLR or SBRT at our centers. Recurrence-free survival (RFS) and overall survival (OS) were estimated using the Kaplan-Meier method and compared between groups using the log-rank test. To reduce selection bias, propensity score matching (PSM) was applied. Subgroup analyses were also performed to explore outcomes across different clinical and tumor-related characteristics. This retrospective cohort study included 383 patients from four hospitals between January 2017 and December 2019, with 268 patients in the LLR group and 115 in the SBRT group. After PSM adjustment, 98 patients were selected from each group for further analysis. The 1-, 3-, and 5-year OS rates were 94.9%, 88.8%, and 85.7% in the LLR group and 94.9%, 74.5%, and 67.3% in the SBRT group, respectively (p = 0.005). The 1-, 3-, and 5-year RFS rates were 86.7%, 71.4%, and 70.4% in the LLR group and 83.7%, 61.2%, and 53.1% in the SBRT group, respectively (p = 0.011). LLR provided better OS and RFS than SBRT. However, SBRT showed comparable OS and RFS outcomes with LLR for tumors ≤ 3 cm (p = 0.158; p = 0.418), and perivascular tumors (p = 0.082; p = 0.166). LLR is the first choice for treating single HCC ≤ 5 cm, while SBRT may be a favorable alternative for small and perivascular lesions.
Similar content being viewed by others
Introduction
Hepatocellular carcinoma (HCC) ranks as the sixth most common malignant tumor and the third leading cause of cancer-related mortality1. For patients with early-stage HCC, hepatectomy is currently the primary curative treatment2. In the past decade, laparoscopic liver resection (LLR) has matured as a safe and effective alternative to traditional open liver resection (OLR)3,4,5,6. As a result, LLR has increasingly become the preferred surgical approach in clinical practice. However, it is noteworthy that over 75% of HCC patients are ineligible for surgical intervention1,2.
Percutaneous ablation (PA) has firmly established itself as the standard therapeutic approach for achieving curative intent in small HCC (≤ 3 cm) among patients who are not suitable for surgical intervention. Nevertheless, the application scope of PA is significantly limited by various factors, including the location and diameter of the tumor7,8,9. Stereotactic body radiotherapy (SBRT) has emerged as a treatment option for HCC, yielding promising results. Usually, HCC patients are treated in line with the BCLC strategy, and per the most updated guideline, SBRT has a limited role for now for single HCC10. Numerous studies have demonstrated its effectiveness in reducing radiation-induced liver disease (RILD), enhancing local control (LC) rates, and improving survival rates11,12,13,14.
Several studies have reported on the effectiveness of SBRT versus liver resection (LR) for small HCC, but results are conflicting and limited. Small sample sizes and the lack of differentiation between open and laparoscopic procedures may explain these discrepancies15,16,17,18. However, after reviewing the available publications, we observed that few studies compared the long-term effects of LLR and SBRT. Given the significance of this comparison and the potential impact on clinical decision-making, this study aimed to conduct a large-scale multicenter investigation, comparing the clinical outcomes of LLR and SBRT for HCC no more than 5 cm.
Results
Patient characteristics
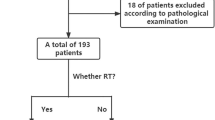
Between Jan 2017 and Dec 2019, a total of 383 eligible patients were identified. Of these, 268 were included in the LLR group, while 115 patients were assigned to the SBRT group. The study profile is illustrated in Fig. 1. Before PSM, the proportions of patients with hypertension, WBC < 4 × 109 /L, PLT counts ≤ 100 × 109 /L, TBIL > 17.1, PT > 13, and AFP > 400 in the LLR group were lower compared to the SBRT group. After PSM, each group comprised 98 patients. No statistically significant differences were observed in patient characteristics between the two groups. The general characteristics of the patient cohort are presented in Table 1.
The flow chart in the study. Abbreviations: HCC, hepatocellular carcinoma. LLR, laparoscopic liver resection. SBRT, stereotactic body radiotherapy. PSM, propensity score matching.
Survival analysis before and after PSM
Before PSM, the 1-, 3-, and 5-year OS rates were 96.6%, 89.4%, and 87.7% in the LLR group and 94.8%, 73.0%, and 66.1% in the SBRT group, respectively (Fig. 2A, p < 0.001). The 1-, 3-, and 5-year RFS rates were 89.9%, 76.1%, and 75.4% in the LLR group and 85.2%, 61.7%, and 54.8% in the SBRT group, respectively (Fig. 2B, p < 0.001).
Comparison of prognosis between LLR and SBRT Groups before PSM and after PSM. (A) OS before PSM; (B) RFS before PSM; (C) OS after PSM; (D) RFS after PSM. Abbreviations: OS, overall survival; RFS, recurrence-free survival; LLR, laparoscopic liver resection; SBRT, stereotactic body radiotherapy; PSM, propensity score matching.
After PSM, the 1-, 3-, and 5-year OS rates were 94.9%, 88.8%, and 85.7% in the LLR group and 94.9%, 74.5%, and 67.3% in the SBRT group, respectively (Fig. 2C, p = 0.005). The 1-, 3-, and 5-year RFS rates were 86.7%, 71.4%, and 70.4% in the LLR group and 83.7%, 61.2%, and 53.1% in the SBRT group, respectively (Fig. 2D, p = 0.011).
Subgroup analysis on the survival of HCC after PSM
In the subgroup of HCC ≤ 3 cm, there were no significant differences in OS and RFS between LLR and SBRT (p = 0.158; p = 0.418)(Fig. 3A&B). However, in the subgroup of HCC 3–5 cm, LLR provided significantly better OS and RFS than SBRT (p = 0.012, p = 0.01) (Fig. 3C&D).
Comparison of prognosis between LLR and SBRT Groups in tumor diameter subgroups after PSM. (A) OS in the ≤ 3 cm tumor diameter subgroup; (B) RFS in the ≤ 3 cm tumor diameter subgroup; (C) OS in the 3–5 cm tumor diameter subgroup; (D) RFS in the 3–5 cm tumor diameter subgroup. Abbreviations: OS, overall survival; RFS, recurrence-free survival; LLR, laparoscopic liver resection. SBRT, stereotactic body radiotherapy. PSM, propensity score matching.
In addition, in the subgroup of HCC with perivascular tumors, there were no significant differences in OS and RFS between LLR and SBRT (p = 0.082; p = 0.166) (Fig. 4A&B). But, in the subgroup of HCC with non-perivascular tumors, LLR provided significantly better OS than SBRT (p = 0.041). Although the RFS of LLR showed some advantage over SBRT, the difference was not statistically significant (p = 0.066) (Fig. 4C&D).
Comparison of prognosis between LLR and SBRT Groups in tumor location subgroups based on proximity to vascular structures after PSM. (A) OS in the perivascular tumor subgroup; (B) RFS in the perivascular tumor subgroup; (C) OS in the subgroup with tumor located in other liver segments; (D) RFS in the subgroup with tumor located in other liver segments. Abbreviations: OS, overall survival; RFS, recurrence-free survival; LLR, laparoscopic liver resection. SBRT, stereotactic body radiotherapy. PSM, propensity score matching.
Then, we categorized tumors into two anatomical subgroups: anteroinfero-lateral portion of the liver (segments 2, 3, 4b, 5, and 6) and postero-superior portion of the liver (segments 1, 7, 8, and 4a). For tumors located in the postero-superior portion, there was no statistically significant difference in OS or RFS between the LLR and SBRT groups (p = 0.185 and p = 0.428, respectively). For tumors located in the anteroinfero-lateral portion, the LLR group demonstrated a significantly better OS and RFS compared to SBRT (p = 0.015 and p = 0.013, respectively)(Fig. 5).
Comparison of prognosis between LLR or SBRT Groups in tumor location subgroups after PSM, according to technical difficulty of LLR. (A) OS in the anteroinfero-lateral portion tumor subgroup; (B) RFS in the anteroinfero-lateral portion tumor subgroup; (C) OS in the postero-superior portion tumor subgroup; (D) RFS in the postero-superior portion tumor subgroup. Abbreviations: OS, overall survival; RFS, recurrence-free survival; LLR, laparoscopic liver resection. SBRT, stereotactic body radiotherapy. PSM, propensity score matching.
Within the HBV (hepatitis B virus) subgroup, LLR was associated with significantly better OS compared to SBRT (p = 0.020), and showed a trend toward improved RFS, although the difference did not reach statistical significance (p = 0.084). In contrast, RFS and OS appeared generally comparable between LLR and SBRT within the HCV (hepatitis C virus), ALD (alcohol-related liver disease), and NASH (non-alcoholic steatohepatitis) subgroups (Figure S1).
Univariate and multivariable analyses of predictors of OS and RFS
After PSM, AFP < 400 and LLR were independent prognostic factors in improving OS, while non-perivascular tumor location, LLR, and PLT > 100 were independent prognostic factors in improving RFS in the Cox proportional hazard model (Table 2). We found that tumor diameter was not the influencing factor of OS and RFS.
Toxicity and complications
Among the 268 patients undergoing LLR, complications occurred in 22 patients, resulting in a complication rate of 8.2% (22/268). The specific complications were as follow: 7 patients experienced Grade 1 complications (including 6 cases of fever and 1 case of wound infection); 15 patients experienced Grade 2 complications (including 9 cases of ascites and 6 cases of minor bile leakage); 2 patients experienced Grade 3 complications (bile leakage requiring percutaneous drainage).
All 115 patients in the SBRT group completed the radiation therapy plan. Complications occurred in 7 patients, resulting in a complication rate of 6.1% (7/115). Grade 1 complications, including 5 cases of vomiting and 2 cases of anorexia. Grade 2 complications include 1 case of abdominal pain and 2 cases of ALT/AST elevation. No toxicities of Grade ≥ 3 were observed. All 7 patients recovered after drug treatment.
Therefore, no patients died from the toxicities or complications of LLR or SBRT (Table 3).
Discussion
To the best of our knowledge, this is the first study analyzing the long-term efficacy of SBRT compared to LLR for single HCC ≤ 5 cm. The present research indicates that LLR significantly outperformed SBRT in terms of OS and RFS, both before and after PSM. However, for small or perivascular tumors, LLR may lose its superiority, and the outcomes of both treatments were comparable. In terms of treatment-related side effects, LLR was associated with more adverse reactions compared to SBRT, which has been shown to be safe and well-tolerated in treating patients with HCC.
Previous studies have identified tumor diameter as an independent prognostic factor for HCC19,20,21. However, in our study focusing on HCC with a diameter ≤ 5 cm, we found that tumor diameter did not influence RFS and OS outcomes between LLR and SBRT. In the HCC ≤ 3 cm subgroup, we found that LLR and SBRT showed no significant differences in OS and RFS, which was consistence with previous research17. However, in the 3–5 cm subgroup, LLR demonstrated significantly higher OS and RFS compared to SBRT. A meta-analysis found that the OS of SBRT was superior to HCC lesions ≤ 3 cm compared to > 3 cm (p = 0.015)14. However, this study included patients with HCC lesions larger than 5 cm, which may have confounded the results and contributed to the observed difference in outcomes. Previous studies comparing LR and SBRT for single (≤ 5 cm) HCC have not analyzed tumor location in detail16,17,18. Perivascular HCC refers to neoplastic tissue that is either closely adhered to or located within 1 cm of major intrahepatic vessels and ducts22. This anatomical peculiarity complicates the achievement of adequate surgical margins, which are imperative for curative resection and must be maintained at a distance of ≥ 1 cm from the tumor edge. A prospective study has demonstrated that preoperative radiotherapy markedly enhances OS in perivascular HCC22. In the present study, no significant differences in OS and RFS were observed between LLR and SBRT in treating perivascular HCC. Conversely, for tumors located in non-perivascular regions, LLR demonstrated improved OS and comparable RFS when compared to SBRT. Previous findings suggest that the therapeutic efficacy of SBRT appears to be independent of tumor proximity to major vessels14. In contrast, for LLR, tumors situated away from vascular structures (non-perivascular) are more likely to allow for wider surgical margins, potentially contributing to better oncologic outcomes. This discrepancy in resection margin adequacy may partly explain the observed differences in treatment efficacy between LLR and SBRT. In perivascular regions, where achieving sufficient margins is technically challenging due to the proximity to vital vascular structures, LLR may be less advantageous. Meanwhile, SBRT, which relies on precise radiation targeting rather than physical excision, is less affected by tumor location relative to vascular anatomy. These differences highlight the importance of individualized treatment planning based on tumor characteristics and anatomical considerations.
Based on the IWATE criteria23, tumors located in the anteroinfero-lateral segments of the liver (segments 2, 3, 4b, 5, and 6) are generally considered more suitable for LLR, while those in the postero-superior segments (segments 1, 7, 8, and 4a) pose greater challenges2,4. This difficulty is mainly attributed to the limited accessibility of these regions through standard trocar placements and the restricted flexibility of laparoscopic instruments, which may compromise the ability to achieve adequate resection margins. In the present study, LLR was associated with significantly better OS and RFS than SBRT for tumors located in the anteroinfero-lateral segments. However, no significant differences were observed in tumors located in the postero-superior segments. Based on these findings, we suggest that while LLR may offer superior outcomes for HCCs ≤ 5 cm located in accessible regions of the liver, SBRT remains an effective alternative treatment option for tumors located in anatomically challenging areas such as the postero-superior segments or those in perivascular regions.
In terms of perioperative outcomes, the mean hospital stay was significantly shorter in the SBRT group (5.6 days) compared to the LLR group (10.5 days). The complication rate in the LLR group was 8.2%, while that of the SBRT group was 6.1%. These findings suggest that SBRT offers favorable short-term safety profiles and less invasiveness compared to LLR.
The present study has several limitations that warrant consideration. Firstly, the study design was retrospective, which introduces the potential for selection bias. Although PSM was utilized to mitigate the effects of baseline characteristic imbalances, the results should be further validated through well-designed prospective studies. Secondly, all study participants were from China, with approximately 80% of patients having concurrent HBV infection. We observed that LLR provides superior OS and comparable RFS to SBRT in the HBV-related subgroup. However, no significant differences were found between LLR and SBRT in patients with HCV-, ALD-, or NASH-related HCC. Given the small sample sizes in these subgroups, we believe these findings may be due to limited statistical power. Therefore, future multi-center studies including patients from diverse geographic and etiological backgrounds are planned to improve the external validity of our results. Lastly, our study exclusively included patients with early-stage solitary HCC and a diameter ≤ 5 cm. Therefore, the efficacy of LLR and SBRT for treating early-stage multifocal or larger diameter (≥ 5 cm) HCC remains unknown. Future research should aim to address these limitations to provide a more comprehensive understanding of the treatment outcomes across different patient populations and disease stages.
In conclusion, our study indicates that LLR provides better RFS and OS than SBRT in HCC patients with a single nodule ≤ 5 cm. However, for patients with solitary HCC ≤ 3 cm or located near major blood vessels, SBRT may be considered an effective and viable alternative treatment option.
Methods
Ethical considerations
The study was approved by our institution’s institutional ethics committee (No.:KXKS2025011). All methods were performed in accordance with the relevant guidelines and regulations, including the Declaration of Helsinki. Due to the study’s retrospective nature, the institutional ethics committee waived the need for written informed consent.
Patients
Eligibility criteria for inclusion in the study were as follows: (1) Diagnosis of HCC confirmed by a surgeon and/or radiologist via imaging studies, laboratory tests, or pathological examination; (2) presence of a single tumor with a maximum lesion diameter of 5 cm or less; (3) an Eastern Cooperative Oncology Group performance status (ECOG PS) score ranging from 0 to 2; (4) no history of prior therapy; (5) absence of portal vein tumor thrombosis and distant metastases; and (6) classification in Child–Pugh Class A or B (CP-A or CP-B).
The treatment plan was established by a multidisciplinary team (MDT), and patient preferences were also considered in determining the appropriate treatment modality. For patients with well-preserved liver function, laparoscopic liver resection was considered the primary option. If there were surgical contraindications such as thrombocytopenia, ascites, or inadequate coagulation, ablation was considered an alternative. When ablation posed significant risks (e.g., to the hepatic vasculature, main bile duct, or intestinal loop) or achieving an adequate resection or ablation margin was challenging, SBRT was contemplated.
The treatment procedure of SBRT group
Before performing SBRT with the G4 CyberKnife system (Accuracy, USA), each patient had four to six fiducial markers implanted. Following the determination of the treatment site through CT simulation, an oncologist delineated the gross tumor volume (GTV) and identified organs at risk (OARs), which included the stomach, liver (excluding tumorous areas), esophagus, duodenum, kidneys, spinal cord, and bowel. A planning target volume (PTV) was then created by expanding the GTV by 3–5 mm, ensuring that the OARs were avoided. Prescribed doses ranged from 48 to 54 Gy across 5–8 fractions, with the isodose curve covering 100% of the GTV. Treatment planning utilized the CyberKnife Multiplan system (version 4.0.2). The tolerance doses for the OARs were guided by the AAPM TG-101 report24.
The treatment procedure of LLR group
LLR was performed following established techniques as described in the literature25, with all procedures involving anatomical liver resection.
Toxicity reaction evaluation
Treatment-related toxicities were evaluated and graded based on the Common Terminology Criteria for Adverse Events (CTCAE) version 4.03 for SBRT. Additionally, post-surgical complications were assessed and classified according to the Clavien-Dindo classification system.
Follow-up study
Each patient was consistently monitored every 2–3 months during the initial years, and subsequently every 3 months until the end of this study or death, depending on which occurred first. The follow-up assessments comprised biochemical evaluations, such as liver function tests and serum alpha-fetoprotein levels, as well as imaging studies, including abdominal contrast-enhanced CT or MRI scans.
Definitions and evaluation of data
The primary endpoint was recurrence-free survival (RFS). The second endpoint was overall survival (OS). RFS was calculated beginning at the onset of LLR/SBRT until tumor recurrence. OS was assessed starting from the initiation of LLR/SBRT until the final follow-up appointment or mortality. Perivascular HCC refers to liver malignancies situated in distinct segments, including S1, S4, S5, and S8, where the neoplastic tissue is either intimately adhered to or situated within a proximity of 1 centimeter to major intrahepatic vessels and ducts22.
Statistical analysis
The utilization of propensity score matching (PSM) analysis aimed to decrease the impact of selection biases and potential confounding variables. The covariates included in the propensity score calculation were: age, white blood cell (WBC) count, platelet count (PLT), total bilirubin (TBIL), prothrombin time (PT), alpha-fetoprotein (AFP) level, tumor diameter, tumor location, and performance status. Following the PSM evaluation, instances were paired through 1:1 nearest neighbor matching with a caliper distance of 0.02 standard deviations of the logit of the PS. Cases that did not match were eliminated. OS and RFS were computed utilizing the Kaplan–Meier technique. The comparison between LLR and SBRT was conducted via the log-rank test. Univariate and multivariable hazard ratios were assessed employing the Cox proportional hazard model. The baseline variables of the two groups were compared using the χ2 test or Fisher’s exact test. A p < 0.05 was considered statistically significant. All statistical computations were carried out using R version 4.0.2 (Stanford University, CA, USA) and SPSS version 27.0 (IBM Corp., Armonk, NY, USA).
Data availability
Research data are stored in an institutional repository and will be shared upon request to the corresponding author.
References
Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249 (2021).
European Association for the Study of the Liver. EASL clinical practice guidelines: management of hepatocellular carcinoma. J. Hepatol. 69, 182–236 (2018).
Yoshida, H. et al. Current status of laparoscopic hepatectomy. J. Nippon Med. Sch. 86, 201–206 (2019).
Twaij, A. et al. Laparoscopic vs open approach to resection of hepatocellular carcinoma in patients with known cirrhosis: systematic review and meta-analysis. World J. Gastroenterol. 20, 8274–8281 (2014).
Zhou, Y-M., Shao, W-Y., Zhao, Y-F., Xu, D-H. & Li, B. Meta-analysis of laparoscopic versus open resection for hepatocellular carcinoma. Dig. Dis. Sci. 56, 1937–1943 (2011).
Yang, S-Y. et al. Perioperative and long-term survival outcomes of laparoscopic versus laparotomic hepatectomy for BCLC stages 0-A hepatocellular carcinoma patients associated with or without microvascular invasion: a multicenter, propensity score matching analysis. Hepatol. Int. 16, 892–905 (2022).
Chen, M-S. et al. A prospective randomized trial comparing percutaneous local ablative therapy and partial hepatectomy for small hepatocellular carcinoma. Ann. Surg. 243, 321–328 (2006).
N’Kontchou, G. et al. Radiofrequency ablation of hepatocellular carcinoma: long-term results and prognostic factors in 235 Western patients with cirrhosis. Hepatology 50, 1475–1483 (2009).
Lee, D. H. et al. Radiofrequency ablation of hepatocellular carcinoma as first-line treatment: long-term results and prognostic factors in 162 patients with cirrhosis. Radiology 270, 900–909 (2014).
Reig, M. et al. BCLC strategy for prognosis prediction and treatment recommendation: the 2022 update. J. Hepatol. 76, 681–693 (2022).
Kimura, T. et al. Stereotactic body radiotherapy for patients with small hepatocellular carcinoma ineligible for resection or ablation therapies. Hepatol. Res. 45, 378–386 (2015).
Sanuki, N. et al. Influence of liver toxicities on prognosis after stereotactic body radiation therapy for hepatocellular carcinoma. Hepatol. Res. 45, 540–547 (2015).
Su, T-S. et al. Stereotactic body radiation therapy for small primary or recurrent hepatocellular carcinoma in 132 Chinese patients. J. Surg. Oncol. 113, 181–187 (2016).
Bae, S. H. et al. Stereotactic body radiation therapy for hepatocellular carcinoma: Meta-Analysis and international stereotactic radiosurgery society practice guidelines. Int. J. Radiat. Oncol. Biol. Phys. 118, 337–351 (2024).
Su, T-S. et al. Long-Term survival analysis of stereotactic ablative radiotherapy versus liver resection for small hepatocellular carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 98, 639–646 (2017).
Nakano, R. et al. Hepatectomy versus stereotactic body radiotherapy for primary early hepatocellular carcinoma: A propensity-matched analysis in a single institution. Surgery 164, 219–226 (2018).
Sun, J. et al. Stereotactic body radiotherapy versus hepatic resection for hepatocellular carcinoma (≤ 5 cm): a propensity score analysis. Hepatol. Int. 14, 788–797 (2020).
Maher, A. M. et al. Comparison of outcomes following surgical resection, percutaneous ablation, or stereotactic body radiation therapy in early-stage, solitary, and small (≤ 3 cm) treatment-naïve hepatocellular carcinoma. Cancer Med. 13, e6978 (2024).
Wahl, D. R. et al. Outcomes after stereotactic body radiotherapy or radiofrequency ablation for hepatocellular carcinoma. J. Clin. Oncol. 34, 452–459 (2016).
Kim, N. et al. Stereotactic body radiation therapy vs. radiofrequency ablation in Asian patients with hepatocellular carcinoma. J. Hepatol. 73, 121–129 (2020).
Llovet, J. M. et al. Hepatocellular carcinoma. Nat. Rev. Dis. Primers. 2, 16018 (2016).
Wu, F. et al. Phase 2 evaluation of neoadjuvant Intensity-Modulated radiotherapy in centrally located hepatocellular carcinoma: A nonrandomized controlled trial. JAMA Surg. 157, 1089–1096 (2022).
Wakabayashi, G. What has changed after the Morioka consensus conference 2014 on laparoscopic liver resection? Hepatobiliary Surg. Nutr. 5, 281–289 (2016).
Benedict, S. H. et al. Stereotactic body radiation therapy: the report of AAPM task group 101. Med. Phys. 37, 4078–4101 (2010).
Abu Hilal, M. et al. The Southampton consensus guidelines for laparoscopic liver surgery: from indication to implementation. Ann. Surg. 268, 11–18 (2018).
Author information
Authors and Affiliations
Contributions
Conceptualization: Weixing Guo and Bin zhou.Funding acquisition: Shuqun Cheng.Resources: Bin zhou, Maolin Yan, Yunfei Duan, Jiazhou Ye and Shuqun Cheng.Investigation: Bin zhou, Zihui Ma, Yan Meng and Weixing Guo.Formal analysis: Bin zhou, Zihui Ma and Xi ChengWriting–original draft: All authors.Writing–review & editing: Weixing Guo.All authors contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhou, B., Ma, Z., Cheng, X. et al. Stereotactic body radiotherapy versus laparoscopic liver resection for single hepatocellular carcinoma ≤ 5 cm: a retrospective, multicenter, cohort study. Sci Rep 15, 40610 (2025). https://doi.org/10.1038/s41598-025-24386-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-24386-w