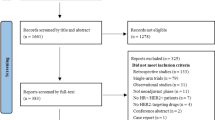
Abstract
In human epidermal growth factor receptor 2 (HER2) positive early breast cancer (EBC) patients, with residual invasive disease after neoadjuvant chemotherapy (NACT) plus HER2-targeted therapy, the KATHERINE trial demonstrated a 50% reduction in risk of recurrence or death with adjuvant trastuzumab emtansine (T-DM1) versus trastuzumab. A retrospective bicentric cohort study was set to assess the real-world efficacy and safety of T-DM1 as adjuvant therapy in HER2-positve EBC patients treated at Institut Curie Hospitals. N = 102 patients consecutively treated from September 2019 to January 2021 were included. All patients had previously received neoadjuvant trastuzumab (plus pertuzumab in one patient) and chemotherapy, N = 95 (93%) with anthracyclines. Post-neoadjuvant residual cancer burden was RCB I, II and III in N = 28 (27%), 62 (61%) and 11 (11%) patients, respectively, while N = 70 (69%) had an estrogen receptor-positive disease. After a median follow-up of 44 months (range 4.5–54), N = 7 (7%) patients experienced distant tumor recurrence, including central nervous system (CNS) metastases in N = 5 patients, leading to death in N = 2 patients, while no local recurrence was reported. The 4-years disease-free survival (DFS) rate was 92.5% (95%CI=[87;98]), lymph node involvement at diagnosis (cN+) being the only factor associated with a higher risk of relapse (p = 0.025, no event in the node negative group in our population). Dose reductions of T-DM1 were required in N = 34 patients (33%) after a median of 5 cycles, mainly because of peripheral neuropathy (N = 14), increased alanine and/or aspartate aminotransferase level (N = 9), and thrombopenia (N = 6). Adverse event leading to drug discontinuation occurred in N = 23 patients (23%), after a median of 9 cycles, mostly peripheral neuropathy (N = 10) and thrombopenia (N = 9). Grade 3–4 toxicity affected N = 9 patients (9%), with no related death to T-DM1. With a low relapse rate and although more dose reductions and treatment discontinuation were observed in our cohort, our results are consistent with those of the KATHERINE trial. CNS as the most frequent site of relapse points to a potential role for drugs with higher activity against CNS metastases.
Similar content being viewed by others
Introduction
Human epidermal growth factor receptor type 2 (HER2) positive breast cancer accounts for 15% of breast cancers. Although historically associated with higher rates of recurrence1, combinations of adjuvant chemotherapy and HER2-targeted therapy at early stage have remarkably improved the prognosis of HER2-positive early breast cancer (EBC)2,3. For stage I tumors, treatment is still based on initial surgery followed by adjuvant chemotherapy (12 weekly paclitaxel) and trastuzumab, given their excellent prognosis as demonstrated by the APT trial4. Treatment of stage II and III tumors is based on neoadjuvant chemotherapy (NACT), making it possible to differentiate prognosis groups according to chemosensitivity assessed by histological response, pathological complete response (pCR) being strongly associated with disease-free survival (DFS) and overall survival (OS)5. In patients with no pCR after neoadjuvant therapy with chemotherapy and trastuzumab +/- pertuzumab, trastuzumab emtansine (T-DM1) has been tested in the KATHERINE phase III trial as an adjuvant therapy6. T-DM1 is an antibody-drug conjugate (ADC) combining trastuzumab covalently linked to DM1, a microtubule inhibitor, and was until 2022 the standard of care second-line therapy for HER2-positive metastatic breast cancer7: its efficacy after resistance to trastuzumab +/- pertuzumab made it a good candidate for evaluation in the post-neoadjuvant phase. In the non-pCR population after NACT plus HER2-targeted therapy, KATHERINE demonstrated the benefit of T-DM1 (given every three weeks, for 14 cycles) as rescue adjuvant therapy, halving the relative risk of recurrence versus trastuzumab, leading to FDA approval in May 2019 and thus becoming the therapeutic standard in this post-neoadjuvant setting.
To frame T-DM1 efficacy and safety in a real-world context, we performed a retrospective analysis on patients who received T-DM1 for residual invasive disease after NACT plus HER2-targeted therapy at Institut Curie Hospitals from September 2019 to January 2021.
Patients and methods
The cohort and data collection was approved by Institut Curie IRB. Per French law, given the retrospective nature of the study, no informed consent was needed. All patients provided written consent for their clinical data to be used for research purpose.
Patients and treatment
This cohort included patients with HER2-positive (by local assessment) EBC (confirmed with 18-FDG PET-scan or chest, abdominal and pelvic CT-scan + Tc99m bone scintigraphy) treated with T-DM1 for residual invasive disease after NACT plus HER2-targeted therapy. T-DM1 was used per standard of care, i.e. initially started at 3.6 mg/kg, every three weeks, and dose reductions were possible at the following cycles per T-DM1 summary of product characteristics. T-DM1 was continued for 14 cycles, or until limiting toxicity, tumor recurrence, death, or patient decision. In case of T-DM1 discontinuation, trastuzumab could be given until cycle 14 in the absence of contraindications.
Data collection
Collected data included patients characteristics (birth date, sex, performance status, height, weight), tumor characteristics (date of diagnosis, tumor staging, histopathological features, tumor staging), medical history (neoadjuvant treatment, surgical modalities, adjuvant radiotherapy, adjuvant hormone therapy), information on T-DM1 prescription (start and end of treatment date, dosing adjustment, reason for dosing adjustment or discontinuation of treatment), safety (toxicities according to CTCAE v5.0, and those leading to dosing adjustment or discontinuation of treatment or death), and survival data. Data cutoff was February 22nd, 2024.
Endpoints and statistics
As for prior reports on other therapies8,9,10, the general purpose of this study was to report the efficacy and safety of new drugs in patients treated at Institut Curie Hospitals. For this study, we followed KATHERINE primary endpoint: DFS in the overall population, defined as the time from surgery to recurrence of breast cancer or death. Because of its retrospective nature, our study had no pre-specified power. Secondary endpoints were exploration of prognostic and predictive factors, and report safety data on a real-life population. The following prognostic factors for DFS were explored: age at baseline, performance status at baseline, body mass index at baseline, tumor grade, hormone receptor status, initial tumor size (cT), initial node involvement (cN), histological response according to residual cancer burden score (RCB), residual tumor size (ypT), and residual node involvement (ypN).
We reported binary and categorical variables as frequencies (percentages), and continuous variables as medians (interquartile). Patient characteristics were reported using descriptive analyses and compared using the χ2 test, Fisher’s exact test, or the Wilcoxon rank sum test. Survival curves for DFS were generated using the Kaplan-Meier method. Statistical significance was defined by a p < 0.05. All analyses were performed using R version 4.2.2.
Ethical approval.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required. This study was approved by the Institut Curie review board (DATA250007) and conducted in accordance with relevant guidelines and regulations. This article does not contain any studies with animals performed by any of the authors.
Consent to participate.
All patients provided written consent for their clinical data to be used for research purpose.
Results
Patient characteristics
This study included 102 patients consecutively treated in the Institut Curie Hospitals from September 2019 to January 2021. Baseline demographic and clinicopathological characteristics of patients are shown in Table 1. All patients were women, with a median age of 51 years. All patients had HER2-positive EBC, N = 70 patients (69%) had hormone positive disease (estrogen receptor and/or progesterone receptor ≥ 10%), and N = 59 pts (58%) had initial lymph node involvement. All patients had previously received neoadjuvant trastuzumab (plus pertuzumab in one patient) and chemotherapy, N = 95 (93%) with anthracyclines and taxanes, followed by surgery which was mostly mastectomy (N = 52; 51%; justified by tumour size, breast size, multifocality, retromammary location, extensive associated microcalcifications, extensive associated ductal carcinoma in situ, or genetic risk) and axillary lymph node clearance (N = 62; 61%). Post-neoadjuvant residual cancer burden was RCB I, II and III in N = 28 (27%), 62 (61%) and 11 (11%) patients, respectively. T-DM1 was given concomitantly with radiotherapy for N = 99 pts (97%) and endocrine therapy for N = 68 pts (67%). Patients received a median of 14 cycles. Trastuzumab was given to N = 19/23 patients who discontinued T-DM1.
Treatment efficacy and prognostic factors
After a median follow-up of 44 months (range 4.5–54), N = 7 patients (7%) experienced distant tumor recurrence, leading to death in N = 2 pts (2%), while no local recurrence was reported. The 4-years DFS rate was 92.5% (95%CI=[87;98]) (Fig. 1). The site of first metastatic relapse was central nervous system (CNS) in five patients (5%), liver in one patient, and mediastinal lymph nodes in the last case. Two of these relapses occurred during treatment with T-DM1. The only prognostic factor associated with relapse (Table 1) was lymph node involvement at diagnosis (p = 0.025, no event in the node negative group, Fig. 2), while other factors such as RCB (p = 0.7, Fig. 3) were not associated with DFS. It should be noted that one of the two patients with an invasive lobular carcinoma experienced a relapse (Table 1).
Disease-Free Survival.
Disease-Free Survival according to initial lymph node involvement (cN).
Disease-Free Survival according to RCB (residual cancer burden).
Safety
Summary of adverse events and impact on chemotherapy administration is shown in Table 2, and the complete list of adverse events in Table 3. Out of 102 patients treated with T-DM1, dose reductions were required in N = 34 patients (33%) after a median of 5 cycles, mainly because of peripheral neuropathy (N = 14), increased alanine and/or aspartate aminotransferase level (N = 9), and thrombopenia (N = 6). Adverse event leading to drug discontinuation occurred in N = 23 patients (23%), after a median of 9 cycles, mostly peripheral neuropathy (N = 10) and thrombopenia (N = 9). Grade 3–4 toxicity affected N = 9 patients (9%), with no related death to T-DM1.
Discussion
To our knowledge, this is the first real-world cohort to report survival and safety data in patients with HER2-positive EBC treated with T-DM1 for residual invasive disease after NACT plus HER2-targeted therapy. Median follow-up in our study is similar to that of the primary analysis of KATHERINE (44 versus 41 months). Another real-world study has been reported, but focused on the sociodemographic and clinicopathological characteristics of a Spanish cohort11.
The observed DFS in our cohort confirms the good prognosis of these patients, with a 4-years DFS rate of 92.5% (95%CI=[87;98]), in line with the 4-years DFS rate in KATHERINE (estimated 86.4%). Nevertheless, prolonged monitoring is needed, especially since our cohort was apparently enriched in ER+/HER2+ breast cancers, which could relapse later than ER-/HER2+. Such enrichment was also observed in KATHERINE, since ER-positivity is often associated with lower pCR rates among HER2-positive breast cancer12, and the latest update of KATHERINE showed patients could experience long terms relapses13.
In our cohort, lymph node involvement at diagnosis was unfavorably associated with an increased risk of relapse and most of these relapses (N = 5/7) occurred in the CNS. These data are aligned with outcomes reported in KATHERINE, in which nearly half of the relapses were in the CNS: 5.9% CNS relapses for 10.5% metastatic relapses at 3 years, 7% CNS relapses for 14.7% metastatic relapses on updated data at 8 years. These relapses could be explained both by mechanisms of resistance to T-DM114 and by poorer penetration of anti-tumor agents into the CNS through the blood-brain barrier15, and could be circumvented by new-generation therapies. It should also be noted that one of the two patients with an invasive lobular carcinoma in our cohort experienced a relapse. Although no conclusion can be drawn with such a small number of patients, this warrants further investigation given the lower chemosensitivity of lobular tumors16. There was no suggestion that patients with invasive lobular carcinoma experienced a different magnitude of benefit from adjuvant trastuzumab than those with invasive carcinoma of no special type17. Unfortunately, histological data are not available from studies with T-DM1 in either the early or advanced stages6,7,18.
Another aspect of our study was to report the toxicities of T-DM1 in a real-world unselected population. The observed toxicities were known and manageable toxicities but led to more dose reductions or treatment discontinuations than in the KATHERINE trial. In our cohort, dose reductions were required in 33% (versus 17.9% in KATHERINE) and adverse events leading to drug discontinuation occurred in 23% of patients (versus 14.3% in KATHERINE), with more frequent reductions at the minimum level of 2.4 mg/kg (11.4% in our cohort, 3.9% in KATHERINE). High rates of treatment discontinuations and dose reductions are often reported in real-life, due both to less selected populations and to the lifting of protocol constraints governing therapeutic adjustments. Main specific toxicities were known and routinely manageable, such as increased alanine and/or aspartate aminotransferase and thrombopenia. Interestingly, we report more peripheral neuropathy than in KATHERINE (41% versus 18.6% of any grade and 6% versus 1.4% of grade 3 respectively). Such higher toxicity signal may be related to more frequent neuropathy at baseline, bearing in mind that patients with grade ≥ 2 peripheral neuropathy – a frequent side effect of taxanes - were excluded from KATHERINE trial. However, we cannot rule out a bias due to the retrospective nature of the data collection, with peripheral neuropathy and its imputability poorly documented and wrongly attributed to T-DM1. Apart from this signal on neuropathy, no signal for a new toxicity has appeared in our cohort. Finally our cohort also illustrate the safety of concomitant administration of T-DM1 with adjuvant radiotherapy, confirming existing data19.
We believe our real-world data could contribute to the understanding of incoming clinical trial results with newer drugs. The first candidate drug in the post-neoadjuvant setting is trastuzumab deruxtecan (T-DXd), which previously demonstrated its superiority over T-DM1 in HER2-positive metastatic breast cancer patients20 and a good efficacy against CNS metastases21. The efficacy of T-DXd in the post-neoadjuvant setting in non-pCR patients is currently being assessed versus T-DM1 in the DESTINY-Breast05 trial (NCT04622319), a registrational phase III with results expected in 2025. The second candidate molecule is tucatinib, an anti-HER2 tyrosine kinase inhibitor that has demonstrated efficacy in combination with trastuzumab and capecitabine after resistance to T-DM1 in HER2-positive metastatic breast cancer patients22, and effectiveness on active CNS metastases23. The post-neoadjuvant combination of tucatinib and T-DM1 is being evaluated versus T-DM1 in the CompassHER2 phase III trial (NCT04457596).
Main limitations of our study include its retrospective nature and a small sample size. The retrospective and single-arm design, lacking a control group, limits the strength of the conclusions, and the comparison with the KATHERINE trial remains indirect. Selection bias cannot be excluded. With only 102 patients and seven relapse events, the study has limited power to detect prognostic factors or to perform robust multivariate analyses. Since our cohort was set up in two centers, both being referral tertiary centers for breast cancer in Paris greater area, results might also not be extrapolated to smaller primary care in France or elsewhere. Another limitation is the absence of exposure to neoadjuvant pertuzumab in our cohort: only one patient received neoadjuvant pertuzumab due to the lack of reimbursement for pertuzumab by the French healthcare system. As the majority of patients in the KATHERINE trial did not receive neoadjuvant pertuzumab prior to enrolment (81.5%), and neoadjuvant pertuzumab is still not reimbursed in France in 2025 as in many countries, this parameter does not compromise the value of reporting these real-world data. As discussed, T-DM1 was reported as an efficient rescue therapy six years ago (2019), therefore limiting our study median follow-up.
To conclude, the observed DFS in HER2-positive EBC patients treated with T-DM1 for residual invasive disease after NACT plus HER2-targeted therapy in a real-world cohort was consistent with the results of the KATHERINE trial, with the confirmation that CNS is the main site of relapse. Our data also confirm an acceptable real-life toxicity profile with known and manageable toxicities but which led to more dose reductions or treatment discontinuations than in the KATHERINE trial. Such data will help weighting the potential benefit of newer drugs now investigated in this specific clinical setting.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Esteva, F. J. & Hortobagyi, G. N. Prognostic molecular markers in early breast cancer. Breast Cancer Res. 6, 109 (2004).
Romond, E. H. et al. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl. J. Med. 353, 1673–1684 (2005).
von Minckwitz, G. et al. APHINITY - Adjuvant Pertuzumab and trastuzumab in early HER2-Positive breast cancer. N Engl. J. Med. 377, 122–131 (2017).
Tolaney, S. M. et al. Adjuvant Paclitaxel and trastuzumab for node-negative, HER2-positive breast cancer. N Engl. J. Med. 372, 134–141 (2015).
Spring, L. M. et al. Pathologic complete response after neoadjuvant chemotherapy and impact on breast cancer recurrence and survival: A comprehensive Meta-analysis. Clin. Cancer Res. 26, 2838–2848 (2020).
von Minckwitz, G. et al. KATHERINE - Trastuzumab emtansine for residual invasive HER2-Positive breast cancer. N. Engl. J. Med. 380, 617–628 (2019).
Verma, S. et al. EMILIA - Trastuzumab emtansine for HER2-positive advanced breast cancer. N Engl. J. Med. 367, 1783–1791 (2012).
Bello Roufai, D. et al. Alpelisib and fulvestrant in PIK3CA-mutated hormone receptor-positive HER2-negative advanced breast cancer included in the French early access program. Oncogene 42, 1951–1956 (2023).
De Moura, A. et al. Atezolizumab and Paclitaxel as first line therapy in advanced triple-negative breast cancer patients included in the French early access program. Sci. Rep. 13 (2023).
De Moura, A. et al. Sacituzumab Govitecan in metastatic triple-negative breast cancer patients treated at Institut curie hospitals: efficacy, safety, and impact of brain metastases. Breast Cancer. 31, 572–580 (2024).
Antolín Novoa, S. et al. Real world data on the demographic and clinicopathological profile and management of patients with early-stage HER2-positive breast cancer and residual disease treated with adjuvant trastuzumab emtansine (KARMA study). Cancer Treat. Res. Commun. 37, 100772 (2023).
Schettini, F. et al. HER2-enriched subtype and pathological complete response in HER2-positive breast cancer: A systematic review and meta-analysis. Cancer Treat. Rev. 84, 101965 (2020).
Loibl, S. et al. Phase III study of adjuvant ado-trastuzumab emtansine vs trastuzumab for residual invasive HER2-positive early breast cancer after neoadjuvant chemotherapy and HER2-targeted therapy: KATHERINE final IDFS and updated OS analysis. Presented at San Antonio Breast Cancer Symposium 2023, Abstract GS03-12 (2023).
Hunter, F. W. et al. Mechanisms of resistance to trastuzumab emtansine (T-DM1) in HER2-positive breast cancer. Br. J. Cancer. 122, 603–612 (2020).
Achrol, A. S. et al. Brain metastases. Nat. Rev. Dis. Primers. 5, 5 (2019).
Wilson, N., Ironside, A., Diana, A. & Oikonomidou, O. Lobular breast cancer: A review. Front. Oncol. 10, 591399 (2020).
Metzger-Filho, O. et al. Magnitude of trastuzumab benefit in patients with HER2-positive, invasive lobular breast carcinoma: results from the HERA trial. J. Clin. Oncol. 31, 1954–1960 (2013).
Krop, I. E. et al. Trastuzumab emtansine versus treatment of physician’s choice for pretreated HER2-positive advanced breast cancer (TH3RESA): a randomised, open-label, phase 3 trial. Lancet Oncol. 15, 689–699 (2014).
Salvestrini, V. et al. Safety profile of trastuzumab-emtansine (T-DM1) with concurrent radiation therapy: A systematic review and meta-analysis. Radiother Oncol. 186, 109805 (2023).
Cortés, J. et al. Trastuzumab Deruxtecan versus trastuzumab emtansine for breast cancer. N. Engl. J. Med. 386, 1143–1154 (2022).
Harbeck, N. et al. Trastuzumab Deruxtecan in HER2-positive advanced breast cancer with or without brain metastases: a phase 3b/4 trial. Nat Med 1–10 (2024).
Curigliano, G. et al. Tucatinib versus placebo added to trastuzumab and capecitabine for patients with pretreated HER2 + metastatic breast cancer with and without brain metastases (HER2CLIMB): final overall survival analysis. Ann. Oncol. 33, 321–329 (2022).
Lin, N. U. et al. Intracranial efficacy and survival with Tucatinib plus trastuzumab and capecitabine for previously treated HER2-Positive breast cancer with brain metastases in the HER2CLIMB trial. J. Clin. Oncol. 38, 2610–2619 (2020).
Acknowledgements
The authors acknowledge the Women’s Cancer Institute core grant to Institut Curie (ANR-23-IAHU-0006).
Author information
Authors and Affiliations
Contributions
AD wrote the main manuscript text. AD, OG, LC, FCB, TG have contributed to the study conception and design. AD, OG, LC, ABV, AB, DBR, PC, CS, DL, RG, JYP, LE, FCB, TG have made substantial contributions to data acquisition and data analyses. LC performed the statistical analyses. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
D Bello-Roufai reported consulting fees from AstraZeneca, MSD and EISAI; support for attending meetings from Daiichi-Sankyo. D Loirat reported consulting fees from AstraZeneca, Daiichi-Sankyo, Roche and Pfizer; support for attending meetings from AstraZeneca, Daiichi-Sankyo and Roche. JY Pierga reported consulting fees from AstraZeneca, Daiichi-Sankyo, Novartis, Lilly, Roche, MSD and Gilead; support for attending meetings from AstraZeneca, Novartis, Gilead and Daiichi-Sankyo. FC Bidard reported consulting fees from AstraZeneca, Daiichi-Sankyo and Roche; research grants from Pfizer and AstraZeneca; support for attending meetings from AstraZeneca and Daiichi-Sankyo. Other authors declared no competing interests with this research.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
De Moura, A., Gleizes, O., Cabel, L. et al. Efficacy and toxicity of post-neoadjuvant trastuzumab emtansine in HER2-positive breast cancer patients treated at Institut Curie Hospitals. Sci Rep 15, 39160 (2025). https://doi.org/10.1038/s41598-025-24849-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-24849-0