Abstract
Red blood cell distribution width (RDW), recognized as a biomarker for systemic inflammation and oxidative stress, has been associated with various health outcomes, including depression. However, the precise relationship between RDW and depression, particularly in individuals with differing cancer statuses, remains ambiguous. In this cross-sectional study, data from 32,467 adults aged 18 and older from the NHANES 2005–2018 cohort were analyzed. Depressive symptoms were evaluated using the Patient Health Questionnaire-9 (PHQ-9), where a score of 10 or above indicated moderate-to-severe depression. Multivariate logistic regression models assessed the association between RDW and depression, accounting for demographic, lifestyle, clinical, and laboratory covariates. Subgroup analyses considered cancer status, revealing that a higher RDW was associated with depression in the general population [adjusted odds ratio (OR), 1.04; 95% confidence interval (CI), 1.01–1.07]. This link persisted for individuals without cancer (adjusted OR, 1.05; 95% CI, 1.02–1.08) but not for those with self-reported cancer history (adjusted OR, 0.97; 95% CI, 0.89–1.06), indicating significant interaction differences (P for interaction = 0.012). These results suggest RDW may serve as a cost-effective marker associated with depression, particularly in individuals without cancer, and highlight the need for further exploration of its implications and mechanisms across various clinical contexts.
Similar content being viewed by others
Introduction
Red cell distribution width (RDW), a measure of the variability in red blood cell size, has traditionally been used in diagnosing hematological disorders. However, recent studies have highlighted its broader role as a marker of systemic inflammation and oxidative stress1,2. Elevated RDW levels have been linked to poor outcomes in chronic diseases such as cardiovascular conditions, diabetes, and cancer3,4,5,6. Despite growing interest, the relationship between RDW and mental health, particularly depressive symptoms, remains underexplored7.
Depression, a prevalent mental health disorder, is a leading cause of global disability. Its pathogenesis involves complex interactions between genetic, environmental, and biological factors, with systemic inflammation emerging as a key contributor8,9. Elevated RDW, as a surrogate marker for inflammatory states, has been hypothesized to be associated with depression. Mechanistically, systemic inflammation and oxidative stress may disrupt erythropoiesis and impair red blood cell homeostasis, thereby increasing RDW levels. These processes have also been implicated in the etiology of depressive disorders, suggesting a possible link between elevated RDW and depression10,11.
Moreover, RDW has demonstrated significant associations with other inflammatory biomarkers, such as C-reactive protein (CRP) and interleukin-6 (IL-6), which are known to play roles in depressive pathophysiology12,13. These findings underscore the potential utility of RDW as a cost-effective and accessible biomarker associated with depression. However, studies investigating this association have primarily focused on general populations, with limited attention to specific subgroups, such as individuals with cancer.
Cancer patients represent a unique population characterized by chronic inflammation, anemia of chronic disease, and high psychological distress. These factors might collectively elevate RDW levels and contribute to an increased prevalence of depressive symptoms3,14,15. Additionally, cancer-related systemic inflammation and comorbidities may amplify the complex interplay between RDW and depression, necessitating further investigation16,17.
The National Health and Nutrition Examination Survey (NHANES), a comprehensive and nationally representative dataset, offers an ideal framework for exploring these associations. By integrating demographic, clinical, and laboratory data, NHANES enables robust evaluations of health determinants across diverse populations18. Utilizing NHANES data from 2005 to 2018, this study aims to clarify the relationship between RDW and depressive symptoms, with a specific focus on differences between cancer and non-cancer populations.
Specifically, this study has two objectives: (1) to assess the independent association between RDW and depression in the general population after adjusting for confounders, and (2) to evaluate the effect modification by cancer status on the RDW-depression relationship. By addressing these objectives, this research seeks to enhance the understanding of RDW as a potential biomarker for depression and provide insights into tailored intervention strategies for vulnerable populations.
Materials and methods
Study design and population
This was a cross-sectional study based on continuous public data from NHANES, with all data details taken from the official NHANES website (https://www.cdc.gov/nchs/nhanes/about_nhanes.htm). It conducted extensive household interviews and collected population baseline information and health questionnaire data. Physical examination and collection of blood samples was done through a mobile examination center (MEC). Serum specimens were stored in appropriate frozen (−30 °C) conditions until transported to the Laboratory Sciences Division of the National Center for Environmental Health at the Centers for Disease Control and Prevention for testing. Detailed specimen collection and handling procedures are documented in the NHANES Laboratory/Medical Technician Procedures Manual (LPM). Our study population was adults (≥ 18 years old) from the 2005–2018 NHANES, all of whom completed interviews and assessments at the MEC. Data on depressive status, RDW, and cancer status were excluded if missing. At the same time, we exclude women who are pregnant. The final study population of 32,467 (16,300 female and 16,167 male) participated in the retrospective study. The NHANES survey protocol was approved by the National Center for Health Statistics Research Ethics Review Committee. All participants provided written informed consent, and all methods were carried out in accordance with relevant guidelines and regulations.
RDW
RDW was measured from EDTA-anticoagulated whole blood using automated hematology analyzers (Coulter DxH series) in NHANES-certified laboratories following standardized protocols. Blood samples were processed within 24–48 h of collection under refrigerated conditions (4 °C) to ensure stability, with rigorous quality control measures including daily calibration. RDW was analyzed both as a continuous variable (per 1% increment) and categorically (T1 < 12.6%, T2:12.7–13.4%, T3 > 13.5%) to assess its association with depression.
Depressive symptoms and cancer
In NHANES, the Patient Health Questionnaire (PHQ-9), a 9-item screening instrument that measures the frequency of various depressive symptoms in the past 2 weeks, was used to assess participants’ depressive status19. Each of the 9 items consists of a 4-point scale, with 0 meaning “not at all,” 1 meaning “a few days,” 2 meaning “more than half the time,” and 3 meaning “almost every day,” with a total score of 0 to 27. Based on the PHQ-9 score, a score of 10 was used as the cut-off point for inclusion in the depression group, indicating moderate to severe depressive symptoms. 88% sensitivity and 88% specificity of the PHQ-9 for suggesting moderate to severe depressive symptoms were obtained20.
Cancer status was defined based on self-reported responses to the NHANES questionnaire item: ‘Have you ever been told by a doctor or other health professional that you had cancer or a malignancy of any kind?’ Participants answering ‘Yes’ were classified as having cancer. This definition included both primary and recurrent cases but did not distinguish between active treatment, remission, or time since diagnosis due to data limitations.
Covariates
The NHANES database contains lifestyle information and personal medical history based on standardized questionnaires. Age, gender, race, education level, marital status, household income to poverty ratio (PIR), body mass index (BMI), smoking status, drinking status, and laboratory results including white blood cells (WBC), red blood cell count (RBC), albumin, alanine aminotransferase (ALT), aspartate aminotransferase (AST), blood urea nitrogen (BUN), creatinine, total cholesterol (TC) and comorbid diabetes, hypertension and coronary heart disease. Race was categorized as Mexican-American, non-Hispanic white, non-Hispanic black, other Hispanic, or other. Educational level was categorized as less than high school, high school or equivalent, and above high school. According to standardized protocols, BMI was calculated as weight in kilograms divided by height in meters squared and was categorized as < 25.0, 25.0–30.0, and ≥ 30.0 kg/m2. Smoking status was categorized as never smokers, former smokers, and current smokers. Participants who had never smoked a cigarette in their lives were labeled as “never smokers.” Those who had previously smoked ≥ 100 cigarettes but were no longer smoking were considered as “former smokers.” Participants who had previously smoked ≥ 100 cigarettes and were currently smoking at the time of the interview were classified as “current smokers.” Never drinkers, former drinkers, and current drinkers were all separated into three categories participants who had had < 12 drinks in their lives were labeled as “never drinkers.” Participants who had had ≥ 12 drinks in 1 year and did not drink last year were considered to be “former drinkers”. Individuals who have consumed more than 12 drinks and continued drinking in the past year are referred to as “current drinkers.” Medication use was categorized as “no” for individuals not taking any medication, “antidepressants drugs” for those taking antidepressants, and “other drugs” for those taking medications other than antidepressants. Cases of diabetes were defined based on self-reported questionnaires and the American Diabetes Association’s criteria21. The criteria were as follows: FPG ≥ 126 mg/dL, glycosylated hemoglobin (HbA1c) ≥ 6.5%, 2-hour plasma glucose from an oral glucose tolerance test (OGTT) ≥ 200 mg/dL, current use of insulin or diabetes pills to lower blood glucose, and self-reported questionnaire data indicating a physician diagnosis of diabetes. In addition, self-reported current use of antihypertensive medication or physician diagnosis was used to define hypertension. Coronary heart disease is based on the “Ever told you had coronary heart disease” question, and the answer is yes.
Statistical analysis
Multivariate logistic regression analyses were adopted to assess the independent association between RDW and the prevalence of depression between all participants, with and without cancer participants. An extended logistic model approach was used for different covariates adjusted models. In multivariate logistic regression, RDW was analyzed as a continuous and categorical variable, with model 1 adjusting for age and gender and model 2 adjusting for variables from model 1 as well as clinical indications including BMI, race, education level, smoking status, drinking status and PIR, model 3 was adjusted for model 2 plus WBC, RBC, Albumin, ALT, AST, BUN, creatinine, TC and diabetes, hypertension, coronary heart disease and medication use. Multiple imputation was applied for covariates with missing values (< 10%). All the variables have no multicollinearity (Supplementary Table 1). More importantly, the relationship between participants’ RDW and depression was compared between the non-cancer and cancer group. Interaction among subgroups was inspected by the likelihood ratio test. Descriptive analyses were used for all participants, based on cancer status. Categorical variables are expressed as frequencies (%). For continuous data, normal distributions were expressed as mean ± standard deviation (SD) or skewed data were expressed using median and interquartile range (IQR), as applicable. Variables were compared using independent samples t-test (normal distribution) and Mann-Whitney U test (skewed distribution) and chi-square test (categorical variables), respectively. All analyses were performed using the free statistical software version 2.122 and the statistical software packages R4.2.2 (The R Foundation, http://www.R-project.org). P < 0.05 was considered statistically significant (two-tailed test).
Results
Study population
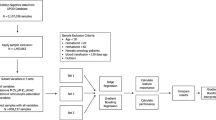
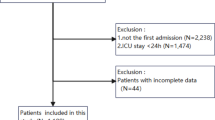
This study extracted data from NHANES 2005–2018 in which 70,190 potential participants completed interviews and underwent MEC screening were included in our study. Participants younger than 18 years of age and missing PHQ-9 scores (n = 33,731) were excluded. After excluding participants with missing data for RDW (n = 1,464), cancer status (n = 2,001) and pregnant women (n = 527), the remaining 32,467 participants were included in our analysis and were classified into two categories based on cancer status. The flowchart of the study population selection is presented in Fig. 1.
The flowchart of the study.
Baseline characteristics
The age of all participants was 50.1 ± 17.7 years and 50.2% were female. Mean baseline RDW was 13.3 ± 1.4%. Detailed characteristics of the population as determined by cancer status are shown in Table 1. Individuals who are older, have a relatively high PIR, have a higher BMI, and have a high school education or less appear to have a higher cancer prevalence. The prevalence of depression in cancer and non-cancer patients was 10.5% and 8.7%, respectively. Notably, the cancer group demonstrated a significantly higher RDW (13.6 ± 1.5%) compared to the non-cancer group (13.3 ± 1.4%) (P < 0.001). The prevalence of diabetes, hypertension and coronary heart disease among the participants were all higher in the cancer patients. Interestingly, current rates of smoking and drinking appear to be lower among cancer patients, which may be the reason for abstinence due to cancer.
Univariate and multivariate analyses of RDW and depression
Gender, race, education level, marital, PIR, BMI, smoking status, drinking status, RBC, RDW, albumin, WBC, ALT, AST, BUN, TC, medication use, comorbid diabetes, hypertension and coronary heart disease were significantly associated with prevalence of depression, as shown in Supplementary Table 2. Table 2 showed that after adjusting for different confounders, such as age, gender, race, education level, PIR, marital, BMI, smoking status, drinking status, WBC, RBC, Albumin, ALT, AST, BUN, creatinine, TC, medication use, diabetes, hypertension and coronary heart disease, RDW was positively associated with depression in all four models. The odds ratio (OR) for RDW was consistently significant in all different models, regardless of whether RDW was analyzed as a continuous variable or as three tertiles.
When RDW was assessed as a continuous variable, after adjusting for all covariates in Table 1, it was observed that the prevalence of depressive group (OR, 1.04, 95% CI: 1.01–1.07) increased by 4% with higher RDW (per 1-unit increment) (Model 3, Table 2). When RDW was analyzed as three tertiles, the adjusted OR for depressive group was 1.01 (95% CI:0.91–1.13) and 1.16 (95% CI: 1.04–1.29) for T2 and T3 in model 3 with T1 as reference. In the same models and analyses, the adjusted OR for participants without cancer in T2 and T3 was 1.04 (95% CI: 0.93–1.16) and 1.20 (95% CI: 1.07–1.34) respectively, with T1 as reference. Accordingly, RDW was positively associated with depression in all models for the overall population and for participants without self-reported cancer (P for trend < 0.05). However, this association was not significant among individuals with self-reported cancer.
Subgroup analyses by different cancer status
After adjusting for age, gender, race, education level, PIR, marital, BMI, smoking status, drinking status, WBC, RBC, Albumin, ALT, AST, BUN, creatinine, TC, medication use, diabetes, hypertension and coronary heart disease, higher RDW levels were significantly associated with a higher prevalence of depression in the group without cancer (OR, 1.05; 95% CI, 1.02–1.08), but not in the cancer group (OR, 0.97; 95% CI, 0.89–1.06). There was an interaction between RDW and depression in patients with cancer and those without cancer when RDW was transformed into a categorical variable (P-value for the likelihood ratio test for the interaction was P = 0.026) (Table 3). Furthermore, to account for the potential influence of inflammatory markers, we further included CRP as an additional covariate. The results remained consistent with the main findings (Supplementary Table 3).
Discussion
In this study, we investigated the association between RDW and depressive symptoms using data from the NHANES 2005–2018 cohort. Our findings demonstrated a significant positive relationship between RDW and depression in the general population, consistent across multiple adjustment models. Notably, this association was pronounced in participants without cancer but not in those with cancer, suggesting that cancer status may modify the relationship between RDW and depression.
Elevated RDW has been previously identified as a marker of systemic inflammation and oxidative stress2, both of which play crucial roles in the pathogenesis of depression10,23. Mechanistically, systemic inflammation disrupts erythropoiesis by impairing iron metabolism and erythrocyte maturation, leading to increased RDW. This inflammatory state, characterized by elevated pro-inflammatory cytokines (e.g., IL-6, TNF-α), is implicated in depression through direct effects on the brain, including neuroinflammation, hypothalamic-pituitary-adrenal (HPA) axis dysregulation, and reduced neurogenesis8,12,24. Inflammation-induced disruptions in erythropoiesis may lead to increased RDW, reflecting the inflammatory burden underlying depressive symptoms12. Additionally, RDW has been associated with other biomarkers of inflammation, such as CRP and IL-6, which promote blood-brain barrier permeability and microglial activation, exacerbating depressive phenotypes12,13,24. Our findings align with this biological framework, further supporting the potential role of RDW as a surrogate marker for depression and highlighting its utility in reflecting shared pathways between hematologic dysregulation and mood disorders7,10,23.
The subgroup analysis revealed a nuanced relationship between RDW and depression based on cancer status. While RDW was significantly associated with depression in the non-cancer group, this relationship was not observed in participants with cancer. Cancer patients typically exhibit elevated RDW due to anemia of chronic disease, chemotherapy-induced hematological changes, and chronic systemic inflammation25,26. These factors may obscure the specific contribution of RDW to depressive symptoms in this population. Furthermore, psychological stress and other unique aspects of cancer-related depression may involve distinct mechanisms, such as neuroendocrine dysregulation and cytokine release16,24. The lack of association between RDW and depression in cancer patients may reflect heterogeneity in cancer types, stages, or treatment status, which NHANES data do not capture. For example, active treatment or advanced disease could disproportionately elevate RDW due to anemia of chronic disease, obscuring its relationship with depression. Future studies should stratify cancer patients by treatment and remission status to clarify these mechanisms.
Interestingly, the higher prevalence of depression in cancer patients (10.5%) compared to non-cancer participants (8.7%) underscores the importance of addressing mental health in this vulnerable group. While RDW was not independently associated with depression in cancer patients, it remains a valuable biomarker for assessing overall disease burden and inflammation in this population. Further studies are warranted to explore alternative biomarkers or mechanisms driving depression in cancer patients.
Our study has several strengths. The use of a large, nationally representative cohort enhances the generalizability of our findings. The availability of comprehensive covariate data from NHANES allowed for robust adjustments to minimize confounding bias. Additionally, this study is one of the few to examine the interaction between cancer status and the RDW-depression relationship, contributing to the growing understanding of RDW as a biomarker in different clinical contexts.
However, several limitations should be noted. First, the cross-sectional design precludes causal inference. Longitudinal studies are needed to establish whether elevated RDW precedes depressive symptoms or vice versa. Second, despite adjusting for a comprehensive set of covariates, residual confounding from unmeasured factors (e.g., dietary habits, inflammatory disease or nutritional factors) may persist. For instance, certain medications (e.g., iron supplements or chemotherapy agents) could influence RDW independently of depression, potentially biasing our results. Third, while NHANES employs standardized protocols, measurement errors in RDW or PHQ-9 scores are possible. RDW variability may arise from pre-analytical factors (e.g., sample handling delays), though NHANES mitigates this through strict laboratory procedures. Similarly, depression was assessed via self-reported PHQ-9, which, despite its validated sensitivity (88%), may misclassify cases due to subjective reporting or comorbid conditions (e.g., anxiety). Fourth, the classification of medication use was necessarily simplified. The ‘other’ category encompassed a heterogeneous group of individuals taking various non-antidepressant medications. These simplifications highlight the need for future research to further investigate the effects of medications. Finally, cancer status was self-reported and lacked details on stage, treatment, or remission, which could mask heterogeneity in the RDW-depression relationship among cancer subtypes. Future studies should incorporate clinical records to refine cancer classifications and explore mediation by treatment-related anemia or inflammation.
Conclusions
This study provides evidence for a positive association between RDW and depressive symptoms, particularly in individuals without cancer. The findings highlight the potential of RDW as a cost-effective and accessible marker associated with depression. Future research should explore the longitudinal relationship between RDW and depression, investigate alternative markers in cancer populations, and elucidate the biological mechanisms underlying these associations.
Data availability
The datasets generated and analyzed during the current study are available on the NHANES website: https://wwwn.cdc.gov/nchs/nhanes/Default.aspx.
Abbreviations
- OR:
-
odds ratio
- CI:
-
confidence interval
- PIR:
-
the ratio of family income to poverty
- BMI:
-
body mass index
- WBC:
-
white blood cells
- RBC:
-
red blood cell count
- RDW:
-
red cell distribution width
- ALT:
-
alanine aminotransferase
- AST:
-
aspartate aminotransferase
- BUN:
-
blood urea nitrogen
- TC:
-
total cholesterol
- CRP:
-
C-reactive protein
References
Salvagno, G. L., Sanchis-Gomar, F., Picanza, A. & Lippi, G. Red blood cell distribution width: A simple parameter with multiple clinical applications. Crit. Rev. Clin. Lab. Sci. 52, 86–105 (2015).
Patel, K. V., Ferrucci, L., Ershler, W. B., Longo, D. L. & Guralnik, J. M. Red blood cell distribution width and the risk of death in middle-aged and older adults. Arch. Intern. Med. 169, 515–523 (2009).
Lu, X. et al. Prognostic significance of increased preoperative red cell distribution width (RDW) and changes in RDW for colorectal cancer. Cancer Med. 12, 13361–13373 (2023).
Aslam, H. et al. The role of red cell distribution width as a prognostic marker in chronic liver disease: A literature review. Int. J. Mol. Sci. 24, 3487 (2023).
Hong, J. et al. Impact of red cell distribution width and red cell distribution width/albumin ratio on all-cause mortality in patients with type 2 diabetes and foot ulcers: a retrospective cohort study. Cardiovasc. Diabetol. 21, 91 (2022).
Felker, G. M. et al. Red cell distribution width as a novel prognostic marker in heart failure: data from the CHARM program and the Duke databank. J. Am. Coll. Cardiol. 50, 40–47 (2007).
Xi, L. et al. Association of hemoglobin-to-red blood cell distribution width ratio and depression in older adults: A cross sectional study. J. Affect. Disord. 344, 191–197 (2024).
Dowlati, Y. et al. A meta-analysis of cytokines in major depression. Biol. Psychiatry. 67, 446–457 (2010).
Leonard, B. E. Inflammation and depression: a causal or coincidental link to the pathophysiology? Acta Neuropsychiatr. 30, 1–16 (2018).
Shafiee, M. et al. Depression and anxiety symptoms are associated with white blood cell count and red cell distribution width: A sex-stratified analysis in a population-based study. Psychoneuroendocrinology 84, 101–108 (2017).
Dai, M. et al. Predictive value of red blood cell distribution width in poststroke depression. Comput Math Methods Med 2021, 8361504 (2021).
Miller, A. H., Maletic, V. & Raison, C. L. Inflammation and its discontents: the role of cytokines in the pathophysiology of major depression. Biol. Psychiatry. 65, 732–741 (2009).
Harsanyi, S., Kupcova, I., Danisovic, L. & Klein, M. Selected biomarkers of depression: what are the effects of cytokines and inflammation? Int. J. Mol. Sci. 24, 578 (2022).
Lu, F., Pan, S., Qi, Y., Li, X. & Wang, J. The clinical application value of RDW, CA153, and MPV in breast cancer. Clin Lab. 67, 277-283 (2021).
Chen, W., Xin, S. & Xu, B. Value research of NLR, PLR, and RDW in prognostic assessment of patients with colorectal cancer. J Healthc Eng. 2022, 7971415 (2022).
Spiegel, D. & Giese-Davis, J. Depression and cancer: mechanisms and disease progression. Biol. Psychiatry. 54, 269–282 (2003).
Polityńska, B. et al. Is depression the missing link between inflammatory mediators and cancer? Pharmacol. Ther. 240, 108293 (2022).
Mao, Y. et al. Association of serum 25-hydroxyvitamin d concentrations with all-cause and cause-specific mortality among individuals with depression: A cohort study. J. Affect. Disord. 352, 10–18 (2024).
Manea, L., Gilbody, S. & McMillan, D. A diagnostic meta-analysis of the patient health Questionnaire-9 (PHQ-9) algorithm scoring method as a screen for depression. Gen. Hosp. Psychiatry. 37, 67–75 (2015).
Kroenke, K., Spitzer, R. L. & Williams, J. B. The PHQ-9: validity of a brief depression severity measure. J. Gen. Intern. Med. 16, 606–613 (2001).
American Diabetes Association. Standards of medical care in diabetes–2010. Diabetes Care. 33 (Suppl 1), S11–61 (2010).
Mao, Y., Li, X., Zhu, S. & Geng, Y. Association between dietary fiber intake and risk of depression in patients with or without type 2 diabetes. Front. Neurosci. 16, 920845 (2022).
Kriegmair, V., Sigrist, C., Vöckel, J., Kaess, M. & Koenig, J. Red cell distribution width and depressive symptoms: A failed replication in female adolescents. World J. Biol. Psychiatry. 24, 754–759 (2023).
Bortolato, B. et al. Depression in cancer: the many biobehavioral pathways driving tumor progression. Cancer Treat. Rev. 52, 58–70 (2017).
Zahorec, R. Ratio of neutrophil to lymphocyte counts–rapid and simple parameter of systemic inflammation and stress in critically ill. Bratisl Lek Listy. 102, 5–14 (2001).
Tchekmedyian, N. S. Anemia in cancer patients: significance, epidemiology, and current therapy. Oncol. (Williston Park). 16, 17–24 (2002).
Acknowledgements
We thank Dr. Yafei Mao (Fengfeng General Hospital of North China Medical & Health Group) for helping in this revision.
Author information
Authors and Affiliations
Contributions
Yulan Geng. and Ziming Liu. conceived and designed the study. Yanming Li., Hongliang Hu., and Shumin Zhu. drafted the manuscript. Yanming Li., Hongliang Hu., Lisha Wang., Zining Liu., and Xintong Pang. collected and organized data. Ziming Liu. and Yanming Li. performed the statistical analysis and interpreted the data. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The survey protocol was approved by the Research Ethics Review Board of the National Center for Health Statistics (https://www.cdc.gov/nchs/nhanes/ irba98.htm). The NCHS Research Ethics Review Board reviewed and approved NHANES, and all survey participants provided written informed consent. Therefore, no further ethical approval and informed consent were required.
NHANES 2017–2018 | Protocol #2018-01 Effective beginning October 26, 201 |
Continuation of Protocol #2011-17 Effective through October 26, 2017 | |
NHANES 2015–2016 | Continuation of Protocol #2011-17 |
NHANES 2013–2014 | Continuation of Protocol #2011-17 |
NHANES 2011–2012 | Protocol #2011-17 |
NHANES 2009–2010 | Continuation of Protocol #2005-06 |
NHANES 2007–2008 | Continuation of Protocol #2005-06 |
NHANES 2005–2006 | Protocol #2005-06 |
Consent for publication
Since all NHANES data were de-identified, the study did not require the approval of the institutional board review committee or the informed consent of participants.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, Y., Hu, H., Liu, Z. et al. Correlation of red cell distribution width with depression among adults with and without self-reported history of cancer. Sci Rep 15, 41520 (2025). https://doi.org/10.1038/s41598-025-25383-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-25383-9