Abstract
This study investigates how adding graphite platelets (GP), with and without SiC, affects the oxidation resistance of ZrB₂ ceramics. The specimens—ZrB₂-10GP, ZrB₂-15GP, ZrB₂-15SiC, ZrB₂-30SiC, ZrB₂-30SiC-10GP, and ZrB₂-30SiC-15GP—were sintered at 1850 °C for 8 min via spark plasma sintering (SPS). For oxidation evaluation, the samples were exposed to air in the furnace at a temperature of 1450 °C for different times (30, 60, 90, 120, 180, 300 min). Also, the simultaneous thermogravimetric analysis (TGA) and differential thermal analysis (DTA) up to 1200 °C were applied to investigate the in situ oxidation. FESEM and EDS were utilized to perform microstructural and elemental analyses on the cross-sections of various oxide layers. It was disclosed that the GP and SiC both improve the oxidation resistance. The best oxidation resistances were obtained in the ZrB₂-30SiC and ZrB₂-30SiC-10GP with the lowest oxidation layer thicknesses of 38 ± 3 µm and 40 ± 4 µm, respectively. Also, the lowest oxidation resistance was obtained in pure ZrB2 with the highest oxide layer thickness of 129 ± 5 µm. The formation of an adherent oxide layer is the dominant factor for improving the oxidation resistance. The oxidation mechanism of all samples except ZrB2 was diffusion oxygen-controlled with a parabolic behavior. Also, TGA and DTA analysis showed a mass loss at the initial stage (\(\sim\) 200°C ), followed by mass gain at 709 °C and 1065°C.
Similar content being viewed by others
Introduction
Zirconium diboride (ZrB₂) is a binary compound of zirconium and boron, which belongs to Ultra High Temperature Ceramics (UHTCs); it is characterized by its high melting point (3224 °C), excellent hardness, and remarkable thermal and electrical conductivity. Due to these unique properties, ZrB₂ has garnered significant attention in various applications such as re-entry vehicles and aerospace1,2, furnace electrodes, microelectronics, cutting tools, metal crucibles, and solar collectors3.
Of course, one of the main challenges for the use of ZrB2 at high temperatures is its weak oxidation resistance; When ZrB2 is exposed to air oxygen, it is oxidized according to Eq. (1) and causes the formation of ZrO2 and B2O3 oxide layers.
At temperatures around 1273 K, the protection of the oxide layer decreases due to the preferential oxidation of B2O34.
In the temperature range of 1373–1673 K, the evaporation of B2O3 accelerated3,4 and led to form a porous ZrO2 layer and finally to the linear oxidation of ZrB2 substrate4.
To solve this problem, in recent years, various additives have been added to this ceramic in different amounts, such as SiC, MoSi2, VC5, Si3N4 3, HfB2, and graphite6,7.
The dense ultrahigh-temperature ZrB₂–15 vol.% SiC–5 vol.% WC ceramics were created through hot pressing at 2050 °C and 30 MPa. High-temperature oxidation resulted in to formation of a three-layer scale consisting of borosilicate glass, ZrO2 with other oxides (WO₃ and SiO₂), and boron and silicon-depleted materials. After oxidation at 1500 °C for 50 h, the scale was 85 μm thick, while at 1600°C for 2 h, it measured 84 μm. The dense scale enabled the material to retain 70% of its strength after oxidation at 1500°C and 50% after oxidation at 1600 °C, outperforming the base ceramics8.
Sintering ZrB₂-MoSi₂ ceramics at 1900–2150 °C results in beneficial microstructures with (Zr, Mo)B₂ solid solutions, improving performance during cyclic oxidation at 1650 °C. It revealed an interpenetrating microstructure of ZrO₂ micro-grains and nano-sized MoB particles, which effectively limits oxygen penetration to a few microns9.
Nils-Christian Petry10showed that polymer-derived ceramics enhance the high-temperature oxidation resistance of ZrB₂-based monoliths. They coated the ZrB₂ powder with SiCN, SiZrCN, or SiZrBCN and densified it at 1800 °C. Thermogravimetric analysis (TGA) at 1300°C assessed oxidation kinetics over 50 and 100 h. Results showed improved oxidation resistance compared to ZrB₂–SiC.
In our previous study11, the oxidation resistance of ZrB₂-SiC-based composites in the presence of various additives was investigated. Nine factors, including the contents of SiC, Cf, MoSi₂, HfB₂, and ZrC, as well as milling time and SPS parameters (temperature, time, pressure), were analyzed using the Taguchi design method. Oxidation tests were conducted at 1600 °C for 1 h. Results showed that ZrC negatively impacts oxidation resistance, while HfB₂ has a positive effect. Temperature was the most influential SPS parameter, while pressure and milling time had minimal effects. Other factors contributed varying percentages to oxidation resistance, with SiC being the most significant contributor at 12.8%.
Among the different additives, SiC is one of the best additives that not only improves oxidation resistance but also improves sintering ability, hardness, fracture toughness, bending strength, and shock resistance. Therefore, in this article, ZrB2-SiC composite was chosen as the base composite. Also, due to the positive influence of graphite on the oxidation resistance, it was chosen as reinforcement. The effect of graphite amount on the oxidation resistance of ZrB2-SiC composite at 1450 °C was investigated.
Experimental
Commercial SiC (25 μm particle size, 98.7% purity), and ZrB₂ (20 μm particle size, 99.5% purity) powders (were sourced from the Northwest Institute for Non-Ferrous Metal Research (China)) as well as GP (C > 99.9%, platelet diameter of 5–10 μm, US Research Nanomaterials, Inc.) were used as the initial materials. Eight composite samples were created based on the compositions and spark plasma sintering conditions outlined in Table 1. The powders were weighed and mixed using wet ball milling for 3 h at 250 rpm in ethanol, utilizing tungsten carbide cups and balls. After drying the powder mixtures on a magnetic heater, they were placed into graphite molds designed for sintering. The sintering process was conducted using spark plasma sintering technology (SPS: 20T-10, China) at 1850 ◦C for 8 min under 35 MPa pressure in a vacuum. The resulting disk-shaped composites were ground to eliminate any attached graphite foils and then machined using EDM technology for oxidation testing. The isothermal oxidation tests were performed in a box furnace (Azar Furnace, F64L, 1500 °C) at a constant temperature of 1450 °C for durations of 30, 60, 90, 120, 180, 300 min. Samples with the dimensions of 3*3*10 mm3 were applied for isothermal oxidation resistance. The relative mass change, used as an oxidation criterion, was calculated by dividing the mass change due to oxidation by the initial mass. For this purpose, the mass of samples before and after the oxidation was measured via a balance with an accuracy of four decimal places (RADWAG AS 520.R2 PLUS).
The non-isothermal oxidation test of the thermogravimetric analyzer (STA 504, Bahr, Germany) was primarily conducted to identify the critical temperatures for oxidation and to monitor the mass changes during heating, providing insights into the early stages of oxidation. The sample (within a weight of 10 mg) was heated at a rate of 10 °C/min from 50 to 1200 °C in a flowing air environment.
Microstructural and elemental analyses were conducted using Tescan Mira3 FESEM equipped with EDS capabilities. Phase identification of oxidized samples was carried out by X-ray diffraction (XRD: Philips, PW 1730, operating at 40 kV and 30 mA).
Results and discussion
GP effect
Phase identification and morphology of oxide scale
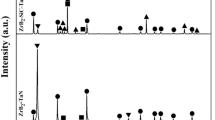
Figure 1 shows the typical XRD spectrum for Zr, Zr10GP, and Zr15GP samples after exposure to the air for 5 h at 1450 °C. It is worth noting that the XRD results of samples before oxidation were published in our previous works12. According to Fig. 1, it is revealed that the predominant phases in the microstructure of all samples are t-ZrO2 and m-ZrO2, which are formed by the oxidation of the ZrB2 phase (Eq. 1) and are consistent with the results of previous research4. It is obvious that besides the ZrO2, a few amount of B2O3 has been detected in the XRD results. This low B2O3 amount originates from its evaporation at the oxidation temperature of 1450 °C for 5 h. Also, some peaks belonging to the B2O3 were omitted in Zr10GP and Zr15GP in comparison with Zr, which can be associated with the positive effect of graphite in removing oxide impurities.
The XRD patterns of the Zr, Zr10GP, and Zr15GP samples, as well as the peak positions and their crystalline planes.
Figure 2 presents the FESEM images of the cross-section for the monolithic ZrB2 as well as ZrB2 containing 10 and 15 vol% GP after oxidation at 1450 °C for 5 h. The microstructure of all samples before oxidation is given in our previous works12,13. A three-layered structure can be distinguished through Fig. 2. According to the maps analysis for ZrB2 containing 10 and 15 vol% GP, which are given in Figs. 3 and 4, and their XRD results (Fig. 1), this layered structure consists of (1) a very thin layer of B2O3, (2) ZrO2, and (3) an unaffected substrate. In all samples, ZrO2 grains with a grey light color can be seen in the oxidized region.
FESEM images of cross-section of (a) Zr, (b) Zr10GP, and (c) Zr15GP after oxidation for 5h at 1450 °C.
Map analysis of the cross section of Zr10GP after oxidation for 5 h at 1450 °C as well as its EDS profile.
Map analysis of the cross section of Zr15GP after oxidation for 5h at 1450 °C as well as its EDS profile.
Many cavities exist in the microstructure of each sample according to Fig. 2 (indicated with white arrows), which can be seen more clearly in the map analysis for ZrB2 reinforced with 10 and 15 vol% GP (Figs. 3 and 4). These cavities are introduced in the microstructure as the result of incomplete densification, which is discussed elsewhere12,13 in detail, and evaporation of B2O3. ZrB2 transits to ZrO2 and B2O3 (l) through reaction (1) while it is exposed to air at a temperature of 1500 ◦C. According to the ZrB2 volatility diagram6 equilibrium pO2 of 1.9 × 10−11 Pa (log pO2 = − 10.73) is required for ZrB2 oxidation. For pO2 < 1.9 × 10−11 Pa, ZrB2 will be in equilibrium with the vapor species such as B2O3, B2O2, BO, and B6.
Since the vapor pressure of B2O3 (g) is much higher than any other species, it will preferentially vaporize as follows14.
Once the B2O3 (g) can escape through the oxide scale, the voids can be created and lead to form a porous skeleton of ZrO2. Of course, it may be that the B2O3 (g) with a high partial pressure of 2.7 *103 Pa, cannot exist quickly from the oxide scale; in this case, if its pressure overdue the ambient pressure, the air bubble may be created.
ZrB2 containing 10 and 15 vol% GP has a lower amount of voids with a smaller size (Fig. 1) compared with the free-GP sample (Zr). In addition, by increasing the GP amount from 10 to 15 vol%, the size and the amount of voids decrease, according to their maps analysis (Figs. 3 and 4). In other words, GP improves the oxidation resistance of ZrB2 by preventing the B2O3 evaporation (Eq. 2), through reaction (3) to some extent, and eventually reduces the creation of voids and bubbles. The thermodynamic calculation (conducted via HSC software) shows, reaction (3) occurs at 1600 °C due to its negative \(\Delta G\) = − 17.07 kcal.
Comparing the elemental map analysis shows, the amount of B2O3 created in the microstructure decreases with graphite addition, which is completely consistent with the XRD result.
Also, it is clear, the diffusion depth of oxygen decreases with GP addition. In other words, graphite prevents the evaporation of B2O3 through the reaction (4) and eventually leads to a decrease in the cavities or air bubbles formation in the microstructure, which act as paths for oxygen diffusion. So, by increasing the graphite addition, the amount of porosity is decreased, and the FESEM images and elemental analysis maps (Figs. 2 and 4) confirm this issue.
In Fig. 5, the amount of weight change of all samples after oxidation at 1450 °C for 5 h is presented. It can be seen that with the addition of graphite, the amount of weight change has been significantly reduced, and it has improved the oxidation resistance.
The weight changes of all samples after oxidation at 1450 °C for 5 h.
As previously discussed, the positive effect of graphite on oxidation resistance is due to its reaction with boron oxide, preventing its evaporation and ultimately the formation of porosity in the microstructure.
According to the FESEM images, the thickness of the oxide layer was measured by Image J software and listed in Table 2. As seen, by addition of graphite, up to 10 vol% and 15 vol%, the thickness of the oxide layer decreases to 112 µm and 80 µm from 129 µm in the Zr sample, respectively. This confirms the weight loss results obtained for these samples (Fig. 5).
SiC content
In Fig. 6, the FESEM images of the cross section of the ZrB2-15 vol% SiC and ZrB2-30 vol% SiC composites after oxidation at 1450 °C are presented. The XRD result of ZrB2-15 vol% SiC after oxidation at 1450 °C is given in Fig. 7. As seen, the key products formed during the oxidation of the composites include ZrO2-m, SiO2, and ZrSiO4, which were formed during reactions (1), (4), and (5), respectively. It is clear that the SiO2 + B2O3 layer plus the layer affected by oxidation in the sample containing 15 vol% % SiC (25 µm, 62 µm respectively) is more than the sample containing 30 vol% SiC (11 µm, 38 µm respectively) (Table 2). It discloses the enhancement in the oxidation resistance by increasing the amount of SiC. Also, according to Fig. 5, the weight changes after oxidation resistance at 1450 °C for 5 h, decrease noticeably in the presence of 15 vol% and 30 vol% SiC, which is completely following FESEM images.
FESEM images of the cross-section of ZrB2-SiC composites, (a) 15vol%SiC and (b) 30 vol% SiC after oxidation for 5h at 1450 °C.
The XRD pattern of the Zr15Si sample, as well as the peak positions and their crystalline planes.
The ability to resist oxidation is determined by a protective oxide layer that forms on the surface of the matrices. In some situations, this resistance is influenced by the gas pressure within the internal oxide layer. At the temperature of 1450 °C, the vapor pressures for B2O3 and SiO2 are roughly 100 Pa and 10–5 Pa, respectively, whereas ZrB2 has a vapor pressure of just 10–9 Pa15.
Due to its high vapor pressure, B2O3 is oxidized faster and, as mentioned before, either it leaves the matrix and causes porosity, or it remains in the matrix and causes bubbles. Due to the addition of SiC, the formed SiO2 reacts with B2O3 and forms a sticky borosilicate layer, thus preventing the evaporation and removal of B2O3 and improving oxidation resistance. It can be seen that compared to the sample containing graphite, ZrB2-Gr (Fig. 2), the microstructure of the sample is denser and lacks large pores. This issue indicates the more beneficial effect of SiC in comparison with the Gr addition.
Co-existence of SiC and GP
According to Fig. 5 (the weight changes against chemical composition), the addition of 10 vol% graphite to the Z30Si sample does not significantly change the oxidation resistance, while adding more amounts (15 vol%) results in to reduction of oxidation resistance.
According to the previous research6,16, it was reported that the graphite addition improves the oxidation resistance. The introduction of graphite into ZrB2-SiC could alter the development of a SiC-depleted layer beneath the outer SiO2-rich surface. Graphite boosts the carbon activity in comparison to nominally pure SiC, which is likely to impact the carbon monoxide pressure found beneath the SiO2 layers. Consequently, the addition of carbon may reduce the vaporization of SiC due to the increased pressure of carbon monoxide. Thus, the overall carbon monoxide pressure in equilibrium with SiC under its outer SiO2 layer is contingent on the carbon activity in the system. At around 1500 °C, studies have shown that the total pressure (pSiO + pO2) can rise dramatically (approximately 100 times) for carbon-saturated silicon carbide in contact with silica, as opposed to nominally pure silicon carbide6.
Jin et al.16 reported that a new ZrB phase exists in the oxide layer of ZrB2-SiC-G composite in addition to t-ZrO2 and m-ZrO2 during oxidation at 1500 °C and low oxygen partial pressure. It is presumably due to the incomplete oxidation.
Also, it was revealed through the oxidation time ascent, allotropic transformation occurred, and t-ZrO2 will be transformed to m-ZrO2 involuntarily, and ZrO2 with the monoclinic structure was detected as the dominant phase. The t-ZrO2 has a critical crystal size above which the allotropic transformation will occur. It was assumed, ZrB2 oxidized to t-ZrO2 through the reaction (1). Then, its crystal size grows by increasing the oxidation time, and it will be transformed to m-ZrO2 when it exceeds the critical size16,17.
The reason for the lack of positive effect of graphite addition on oxidation in the present study can be attributed to the high additive amounts of 40 vol% in Zr30Si10Gr (30 vol% SiC and 10 vol% Gr) and 45 vol% (30 vol% SiC and 15 vol% Gr). High amounts of additives make it difficult to distribute them uniformly in the matrix, thus causing microstructural inhomogeneity and reduced oxidation resistance.
In Fig. 8, the FESEM images of the cross-section of samples containing both graphite and SiC are given. It is clear; the microstructure is similar to ZrB2-SiC samples (Fig. 6), especially for the Zr30Si rather than Zr15Si sample, and it consists of a B2O3 layer with the distributed ZrO2 grains. According to these FESEM images, the thickness of the oxide layer was measured and listed in Table 2. As seen, firstly, with the addition of 10 vol% GP, the thickness of the oxide layer does not change significantly; however, by increasing the GP addition up to 15 vol%, it increases significantly to 64 ± 5 µm in Zr30Si15GP from 40 ± 4 µm in Zr30Si10GP. To investigate this issue, the elemental map distribution of Zr30Si15Gp is presented in Fig. 9. Oxygen has penetrated the microstructure to about 60 µm, which is consistent with the thickness of the oxidized layer reported in Table 2.
FESEM images of cross-section of ZrB2-SiC-GP composites, (a) 10vol%GP and (b) 15 vol% GP after oxidation for 5h at 1450 °C.
Map analysis of the cross section of Zr30Si15GP after oxidation for 5h at 1450 °C, as well as its EDS profile.
To investigate the in situ oxidation mechanism against temperature, the weight change and DTA curves for the Zr30Si10GP sample are presented in Fig. 10. It is observed that the weight of the sample decreases rapidly from about 200 °C to about 750 °C and then increases up to 1050°C. After that, it shows a decreasing behavior in the range of 1050–1065°C and finally continues to increase from 1065 to 1200 °C. The weight loss in the range of 200–750°C can be attributed to the GP oxidation reaction, which causes the formation of carbon monoxide/carbon dioxide gases that are released from the system and cause the weight of the sample to decrease. After that, the weight increase from 750 to 1050°C can be attributed to the oxidation of ZrB2 and the formation of B2O3 and ZrO2, which the peaks identified in the DTA curve in this temperature range indicate and confirming the completion of this reaction. Next, in the temperature range of 1050–1065 °C, the partial weight loss observed is due to the evaporation reaction of B2O3, which causes weight loss due to the exit of B2O3 from the system. Finally, the continuous weight increase of the sample from 1065 to 1200 °C can be attributed to the oxidation of SiC and the formation of SiO2. The formation of SiO2 causes weight increase both due to the formation of an oxide scale and due to the reaction with B2O3 and the formation of a borosilicate layer, and in the prevention of B2O3 evaporation.
Mass change and DTA curves of the Zr30Si10GP sample.
Malik et al.18 reported continuous mass gain (with two sharp increases at 740 °C and 1180 °C) in ZrB2-20 vol% SiC oxidized from 50 to 1300 °C during TGA analysis. The result of this research (mass gain with two sharp increases at 709 °C and 1065 °C) is completely consistent with them. The difference in weight changes in the initial stages (from 50 °C to around 740 °C) between the present study and the reference 18 is due to the presence of GP in the present study, as previously discussed.
Oxidation resistance
In Fig. 11, the weight change curves as a function of oxidation time at a temperature of 1450 °C for all samples are presented. It is clear that for all samples (except pure ZrB2, Zr), the weight change-temperature curves have two distinct regions: 1) a linear curve and 2) a nearly smooth curve.
The weight change curves of all samples against time for oxidation at a temperature of 1450 °C.
As observed in Fig. 11, the weight change rate over time decreases for all samples except pure ZrB₂ (Zr) as time increases. In linear and parabolic oxidation mechanisms, the weight change rate exhibits an ascending and descending trend with increasing time, respectively. Therefore, from the curves in Fig. 11, it can be concluded that the mechanism in the Zr10GP, Zr15GP, Zr30Si10GP, and Zr30Si15GP samples is parabolic, while in pure ZrB2, it is linear. For further investigation and confirmation of this matter, the following relationship was used.
In this equation: ∆W represents the weight change, n is the oxidation exponent, S is the surface area exposed to oxidation, k is the oxidation rate constant, It is the oxidation time.
According to Fig. 11, the calculated values of n were obtained as 1.42, 2.2, 2.07, 2.08, 2.001, 2.03, and 2.1 for Zr, Zr10GP, Zr15GP, Zr15Si, Zr30Si, Zr30Si10GP, and Zr30Si15GP samples, respectively. If the value of n is equal to 1, it indicates linear behavior, which means the oxidation mechanism is controlled by the reaction. In this case, a thick, unstable oxide layer forms on the surface of the sample due to the reaction of oxygen with the surface of the sample, leading to an increased oxidation rate over time. This mechanism occurs at higher temperatures.
If n is equal to 2, it indicates parabolic behavior, which means the oxidation mechanism is controlled by the diffusion of oxygen. In this case, a thick, stable oxide layer forms on the surface due to the reaction of oxygen with the sample, which resists further oxygen diffusion and results in resistance to oxidation and a decreased oxidation rate over subsequent times. This mechanism occurs at lower temperatures. So, it can be deduced that the oxidation mechanism of approximately all samples (except Zr) is parabolic.
The occurrence of the parabolic mechanism (which indicates better oxidation resistance) in samples containing Gr and SiC compared to pure ZrB2, which tends to follow a linear oxidation mechanism, can be attributed to the formation of an adherent oxide layer that hinders oxygen diffusion and further oxidation. In contrast, in the pure ZrB2 sample, the boron oxide layer does not form stably and tends to evaporate, leading to increased oxidation of the sample and resulting in a linear oxidation mechanism.
Conclusion
Samples containing graphite and silicon carbide, both individually and simultaneously, were fabricated at a temperature of 1850 °C using the SPS method. The investigation of the oxidation resistance of the samples at 1450 °C showed that the addition of graphite and silicon carbide individually significantly improves the oxidation resistance of pure zirconium diboride. Specifically, the thickness of the oxide layer decreased from 129 ± 5 µm in the pure zirconium diboride sample to 112 ± 5 µm, 80 ± 4 µm, 62 ± 5 µm, 38 ± 3 µm, 40 ± 4 µm, and 64 ± 5 µm in the Zr10GP, Zr15GP, Zr15Si, Zr30Si, Zr30Si10GP, and Zr30Si15GP samples, respectively. This improvement is attributed to the prevention of boron oxide evaporation and the formation of an adherent and oxidation-resistant oxide layer. Additionally, it was found that the simultaneous presence of silicon carbide and graphite in this study, due to the high addition amounts and lack of uniform distribution, resulted in a decrease in oxidation. It was determined that the weight changes are the result of weight loss due to the oxidation of carbon and the evaporation of B2O3, and weight gain due to the oxidation of ZrB2 and SiC.
Data availability
The data is available on reasonable request from corresponding author (zohre Balak : zbalak@iau.ac.ir).
References
Ghasilzadeh Jarvand, M. & Balak, Z. Oxidation response of ZrB2–SiC–ZrC composites prepared by spark plasma sintering. Synth. Sinter. 2, 191–197 (2022).
Shima, A. & Kazemi, M. Influence of TiN addition on densification behavior and mechanical properties of ZrB2 ceramics. Synth. Sinter. 3, 46–53 (2023).
Dodi, E., Balak, Z. & Kafashan, H. Oxidation-affected zone in the sintered ZrB2–SiC–HfB2 composites. Synth. Sinter. 2, 31–36 (2022).
Parthasarathy, T. A., Rapp, R. A., Opeka, M. & Kerans, R. J. A model for the oxidation of ZrB2, HfB2 and TiB2. Acta Mater. 55, 5999–6010 (2007).
Arab, S. M., Shahedi Asl, M., Ghassemi Kakroudi, M., Salahimehr, B. & Mahmoodipour, K. On the oxidation behavior of ZrB2–SiC–VC composites. Int. J. Appl. Ceram. Technol. 18, 2306–2313 (2021).
Rezaie, A., Fahrenholtz, W. G. & Hilmas, G. E. The effect of a graphite addition on oxidation of ZrB2–SiC in air at 1500 °C. J. Eur. Ceram. Soc. 33, 413–421 (2013).
Yang, B.-L. et al. Effect of nano-graphite on mechanical properties and oxidation resistance of ZrB2–SiC–graphite electrode ceramics. J. Iron. Steel Res. Int. 31, 1502–1513 (2024).
Vedel, D. V., Grigoriev, O. N., Mazur, P. V. & Osipov, A. E. Structure, strength, and oxidation resistance of ultrahigh-temperature ZrB2–SiC–WC ceramics. Powder Metall. Met. Ceram. 60, 60–68 (2021).
Silvestroni, L., Failla, S., Neshpor, I. & Grigoriev, O. Method to improve the oxidation resistance of ZrB2-based ceramics for reusable space systems. J. Eur. Ceram. Soc. 38, 2467–2476 (2018).
Petry, N.-C. et al. Oxidation resistance of ZrB2-based monoliths using polymer-derived Si(Zr,B)CN as sintering aid. J. Am. Ceram. Soc. 105, 5380–5394 (2022).
Balak, Z. & Azizieh, M. Oxidation of ZrB2-SiC composites at 1600 °C: Effect of carbides borides, silicides, and chopped carbon fiber. Adv. Ceram. Prog. 4, 18–23 (2018).
Asl, M. S. & Balak, Z. Fabrication and characterization of ZrB2 ceramic in presence of graphite platelet and SiC. SILICON 15, 6911–6919 (2023).
Savari, V., Balak, Z. & Shahedifar, V. Influence of graphite platelets with and without SiC on the densification and fracture toughness of ZrB2 ceramic sintered by SPS. Carbon Lett. 32, 1559–1566 (2022).
Chen, H. & Meng, S. High temperature oxidation behavior of ZrB2-SiC-graphite composite heated by high electric current. Adv. Mater. Res. 105–106, 162–164 (2010).
Zhang, X., Hu, P., Han, J. & Meng, S. Ablation behavior of ZrB2-SiC ultra high temperature ceramics under simulated atmospheric re-entry conditions. Compos. Sci. Technol. 68(7), 1718–1726 (2008).
Jin, H., Meng, S., Xinghong, Z., Qingxuan, Z. & Weihua X Oxidation of ZrB2–SiC-graphite composites under low oxygen partial pressures of 500 and 1500 Pa at 1800 °C. J. Am. Ceram. Soc. 1–7 (2016).
Basu, B. Toughening of yttria-stabilised tetragonal zirconia ceramics. Int. Mater. Rev. 50, 239–256 (2005).
Manab Mallik, K. K. R. & Mitra, R. Oxidation behavior of hot pressed ZrB2–SiC and HfB2–SiC composites. J. Eur. Ceram. Soc. 31, 199–215 (2011).
Author information
Authors and Affiliations
Contributions
Zohre Balak conducted the conception, measurements, manuscript composition, and writing, revised the manuscript and validated the data. Hamze Ghanbari Nezhad conducted the experimental design and measurement.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Nezhad, H.G., Balak, Z. Synergetic and alone influence of graphite platelet and SiC on the oxidation resistance of ZrB2 ceramics. Sci Rep 15, 41545 (2025). https://doi.org/10.1038/s41598-025-25484-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-25484-5