Abstract
Abiotic stresses are major constraints to sustainable agriculture. Among this salinity is a critical abiotic stress that severely affects crop productivity in arid and semi-arid regions. It impaired physiological and biochemical functions of plants. It also affects nutrient uptake and causes ionic disbalance. The main aim of this study was to evaluate the effectiveness of four selected exogenously applied amino acids (methionine, tryptophan, glycine betaine, and lysine) in mitigating salt stress and improving physiological performance of maize. Although amino acids are widely used as bio-stimulants but comparative evidence on their parameter specific effects under salinity is limited. The current study has been designed to investigate the effect of different concentrations (0, 25, 75, 100 ppm) of each amino acid in alleviating the effect of salt stress (120 mM NaCl) in Zea mays L. plants. Two factorial (amino acid treatments & salt stress) completely randomized design pot experiment with three replicates was conducted in control conditions. Salt stress reduced the morpho-physiological parameters along with antioxidant activities in Z. mays plants. Conversely, the applications of amino acids increased the growth parameters. Maximum shoot length and biomass were noted under the application of lysine. While maximum increase in root length, number of leaves and biomass of roots was observed under the application of glycine betaine, methionine, and tryptophan, respectively. Photosynthetic pigments were maximized under the influence of glycine betaine. Amino acids also triggered antioxidant activity and increased the production of metabolites. Like, total phenolics, total soluble proteins (TSP) and superoxide dismutase (SOD) were maximized by the application of glycine betaine. Similarly, catalase (CAT), peroxidase (POD) and flavonoids were maximized by the application of tryptophan, and total free amino acids (TFA) were maximized under the effect of methionine application. With increase in concentration of amino acids the studied parameters were also increased gradually. It is concluded that specific amino acids target distinct biochemical pathways to exhibit potential to induce tolerance against saline stress. These findings highlight the potential of amino acids, as bio-stimulants to enhance salt stress tolerance in maize. This indicates a role in boosting the plant’s antioxidant defense system under salt stress. However, validation under field conditions will be crucial to clarify molecular mechanisms to optimize the role of amino acids in sustainable crop production.
Similar content being viewed by others
Introduction
In recent times, the demand for cereals has been increased significantly with rapid increase in global population. Climate variations have disturbed the production of crops by increasing the severity and frequencies of biotic and abiotic stresses. Among abiotic factors, salinity is the main issue that possess severe risks on crop production1. Currently, 950 Mha of the total land area (10%) and 230 Mha of the total irrigated area (50%) are under salt stress around the globe2.
Plant bio-stimulants are labeled as metabolic enhancers, plant probiotics, plant strengtheners, biofertilizers and biostimulators3. In other words, bio-stimulants can be defined as products that enhance the nutritional processes of plants independently of the nutrient content of the product. These products such as humic substances, plant growth-promoting bacteria, amino acids, and seaweed extracts alter the plant’s physiological processes and support plant growth and development and stress responses upon application4. Amino acids are bio-stimulants which not only support protein synthesis but also drive key metabolic pathways that regulate cellular energy, redox balance, and immune cell function5. Among amino acids, methionine is a vital amino acid which involved in various physiological processes in plants. It acts as an effective regulator in the growth and development of plants under abiotic stress conditions6. Some reports have highlighted that foliar application of methionine showed positive interactions with plant pigments, osmolytes accumulation, increased uptake of potassium (K), calcium (Ca), phosphorous (P), and efficient removal of reactive oxygen species which ultimately results in improved growth of cowpea and wheat under water-deficient conditions7,8. Further, Lysine also contributes to several responses against biotic and abiotic stresses via the Sacharopine pathway (SACPATH)9. According to some recent reports, Iron and zinc-chelated fertilizers complexed with lysine amino acid result in improvement of growth and yield in crops like in Brassica napus and rice under salt and cadmium stress via an increase in plant biomass, gaseous exchange attributes, and antioxidant enzyme responses10,11,12. L-tryptophan, also known as ß-3-indolylalanine is very unique amino acid with an indole ring. Some studies have shown its promising results after spraying on soil and as seed priming agent. Plants taking up soil applied tryptophan directly via roots or soil microbes metabolized it into many products such as serotonin, niacin and auxins and these products are subsequently absorbed by plant roots13. The controlled use of tryptophan exerts positive effects on plant growth and yield due to continuous release of auxins14. Both foliar and soil-applied tryptophan showed a positive effect on auxin biosynthesis and its crosstalk with other phytohormones not only under normal conditions but also under stressful environments15. Tryptophan is absorbed by the leaf epidermis during foliar application and improves the growth and yield attributes in three bread wheat cultivars and lettuce plants as reported by16,17.
Over the years, researchers have explored various strategies to alleviate salt induced damages in crops. One promising approach is the application of bio-stimulants and osmo-protectants which guard the cellular apparatus of plants from stress-induced injuries without altering the normal metabolic functions at the cellular level. Previous studies have reported the beneficial effects of individual amino acids such as trehalose, phenolics, glycine betaine (GB), proline, sugars etc.18. GB is a quarterly ammonium compound which is synthesized endogenously in the chloroplast as a response to salt and drought environments19. Reports showed that foliar-applied GB results in increased biomass, leaf pigments, leaf area, proline, and primary and secondary metabolites, as well as a reduction in lipid peroxidation in Z. mays under water-deficient conditions20. In another study, foliar applied GB relieved the pepper plants from low temperature and low light stress via the reduction in photoinhibition and oxidative stress21. While Hamani et al.22 linked exogenous applied GB with stomatal characteristics such as photosynthetic rate (Pn), transpiration rate (Tr), intracellular CO2 concentration (Ci), and stomatal conductance under salt stress. Despite these advances a clear gap remains in understanding how different amino acids perform under identical salt stress conditions.so there is a need to access the practical applicability of these amino acids to support crop resilience in stress.
Maize (Zea mays L.) is the second largest widely cultivated crop and accounts for 1137 million tons of estimated global production with a significant contribution towards global food security23. It has significant economic value for GDP growth of a country. It is cultivated for forage, fodder, and cereals production. USA is the leading country to produce Z. mays. It has been proven in the Biphasic model of salinity-induced growth reduction which was presented by Munns et al.24 osmotic and ionic stresses are responsible for the reduction in cereals growth during the first and second phases, respectively. Salinity negatively effects on growth and yield of Z. mays attributes throughout plant life cycle which depends on the length and severity of the stress23,25. Similarly, in another study high salt concentration reduced the Z. mays kernel weight and yield significantly at the reproductive stage compared to non-saline conditions26. Changes in climate lead to alterations in soil fertility attributes such as nutrient levels, organic matter, temperature, moisture contents, and microbiota. To combat these challenges, sustainable agrotechnology can improve soil productivity27.
Salinity stress reduced cereals production on irrigated agricultural land throughout the world. Use of synthetic fertilizers is the major cause of saline stress in agricultural land. This stress led to declines plant growth and crop yield, for which various strategies have been employed to combat the salt stress in plants, including the use of plant growth regulators, Osmo protectants, and bio-stimulants. Among these amino acids have emerged as effective bio-stimulants due to their dual role as osmolyte and signaling molecule that regulate stress responsive pathways. Use of these bio-stimulants not only relieves salt stress but also enhanced growth and yield of crop plants. Replacing synthetic fertilizers with use bio-stimulants can overcome the effect and causes of salinity in agricultural land. In this study, four specific amino acids methionine, tryptophan, lysine, and glycine betaine were selected due to their known roles in enhancing antioxidant defense, promoting osmotic balance, and improving stress tolerance. while some studies have been reported showing individual effects of these amino acids on various crops, and thorough literature study indicates that focusing on all four amino acids in a single experiment under slat stress in maize is scarce, which supports the novelty of this work. The findings of comparative analysis of the effect of amino acids on growth, physio-biochemical attributes, and antioxidant defense mechanism of Z. mays plants under salt stress are expected to inform the development of efficient biostimulant based strategies for improving maize salt tolerance. The novelty of the present study lies in its direct applicability in the field and comparative analysis of multiple amino acids on a salt sensitive crop. To the best of our knowledge, this is the first study to systematically evaluate the comparative effects of these four amino acids on Z. mays under 120 mM NaCl stress, using both physiological and biochemical parameters. This comparative framework provides new insights into specific roles and relative effectiveness of amino acids in mitigating salt stress in maize plants.
Material and methods
Experimental design and layout
In this study, two factorial completely randomized design pot experiments with three replicates were conducted in control conditions in a green house. Conditions set for the experiment were 25/18 °C average day/night temperature with relative humidity of ~ 55% and 13/11 h light/dark period. Seeds of Z. mays cultivar (YH-5427) were obtained from Ayub Agriculture Research Institute (AARI), Faisalabad, Pakistan. The pot experiment was conducted in research area of the University of Lahore, Lahore, Pakistan. The garden soil used in this experiment was loamy in texture with ratio of 40:40:20 for sand, silt, clay, and pH ranging from ~ 7–7.8. Pots of ~ 25 cm height with ~ 15 cm diameter were use in this experiment. Pots were filled with ~ 6 kg of soil. Same sized healthy seeds (6–7) were sown in each pot at equal distance in early March and was harvested after 45–50 days. After germination, 4–5 seedlings were maintained at equal distance in each pot. Salt stress was imposed by irrigating with NaCl solution to reach a final salinity level of 120 mM started after 14 days of sowing. Irrigation was continued twice a week. Soil electrical conductivity (EC) was regularly monitored using a PC meter (ADWA AD8000 Professional multi parameter) and maintained within the range of 11.5–12.2 dS/m by adjusting the NaCl concentration as needed in subsequent irrigations. Meanwhile, control plants were irrigated with simple tap water. Amino acids (Methionine, Lysine, Tryptophan, Glycine betaine) of analytical grade (≥ 98% purity) and procured from Sigma-Aldrich (USA) were applied via foliar spray at different levels; 0 ppm for control, 25, 75 and 100 ppm prepared in 1% Tween-20. Tween-20 was used as surfactant and amino acids were applied in a way that plants get fully wet. Salt stress was applied to Z. mays plants at four leaf stages and maintained till the end of experiment by controlling EC of the pot soil. A week after salt stress treatment, foliar spray of amino acids was done twice with one week interval. Two weeks after last dosage of foliar application plants were uprooted for analysis of various growth, physiological and biochemical parameters as listed below. The plant samples were stored at 4 °C in refrigerator for later use in studying biochemical parameters.
Determination of Z. mays growth attributes
The total number of leaves were counted and noted for each plant. Shoot and root lengths of plants were measured using a scale. Fresh and dry biomass of shoot and root were weighed on an analytical weighing balance (Kern ABT).
Determination of photosynthetic pigments
Plant pigments such as chl a, chl b, total chlorophyll and carotenoids were determined according to the protocol of Arnon28 and Lichtenthaler29. For this purpose, Z. mays leave (0.5 g) were taken from each treatment in falcon tubes and extracted in 5 ml of 80% acetone. The extract was filtered and kept overnight at 4°C in the freezer. Absorbance was measured on UV/VIS spectrophotometer (Shanghai Metash Instruments Co., Ltd.) at wavelengths of 663, 645 and 480 nm for quantification of chl a, chl b, and carotenoids.
The concentrations were calculated using Arnon’s equation:
Determination of total phenolics and flavonoids
The total phenolic content was quantified in accordance with the methodology proposed by Zhang and Quantick30. Leaf samples (1 g) were subjected to extraction using hydrochloric acid–methanol (1%, 5 mL). The extract was filtered and subsequently diluted with hydrochloric acid–methanol to a final volume of 10 mL. The absorbance of the diluted extract was assessed at 280 nm to ascertain the total phenolic content, with quantification facilitated by a standard curve constructed using gallic acid.
The assessment of flavonoid content was conducted employing a modified approach as articulated by Beketov et al.31. The plant extract (0.2 mL) was combined with 90% ethanol (4.5 mL), 2% aluminum chloride (0.2 mL), and 33% acetic acid (0.1 mL). The resultant mixture was incubated in darkness for thirty minutes, after which the absorbance at 414 nm was measured spectrophotometrically.
Determination of total free amino acids and total soluble proteins
The quantification of free amino acid content was conducted utilizing a modified technique as delineated by Hamilton et al.32. Fresh foliage (0.1 g) was subjected to homogenization in a buffer solution with a pH of 7.0. The resulting homogenate was subsequently centrifuged at 8,000 rpm for ten minutes to segregate the supernatant. A reaction mixture was formulated by amalgamating the supernatant (1.0 mL) with 10% pyridine (1.0 mL) and 2% ninhydrin (1.0 mL). This composite was subjected to heating in a boiling water bath for thirty minutes, yielding a violet-hued solution. Following dilution with distilled water, absorbance at 570 nm was spectrophotometrically quantified.
The Bradford’s method33 was applied to ascertain the TSP content within the leaf samples. Bovine serum albumin was utilized as the standard, while Coomassie Brilliant Blue G-250 served as the dye. Frozen leaf specimens (0.5 g) were pulverized in liquid nitrogen and homogenized in 10 mL of 50 mM sodium phosphate buffer. The homogenate underwent centrifugation at 11,000 rpm for ten minutes at 4°C, and the supernatant (0.1 mL) was subsequently combined with Bradford reagent (3 mL). The absorbance at 595 nm was recorded to evaluate the total protein content, with quantification based on a standard curve derived from bovine serum albumin.
Determination of antioxidant enzymes activity
Leaf samples in phosphate buffer (pH 7.8) were utilized for the determination of antioxidant enzyme activity. The antioxidant potential of the extracts was assessed utilizing the 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging assay. A volume of 50 µL of the plant extract from each replicate was introduced into test tubes, succeeded by the incorporation of 14.50 mL of methanol and 5.0 mL of DPPH solution. The resultant mixtures were subjected to incubation in the absence of light at ambient temperature for a duration of 30 min. Butylated hydroxytoluene (BHT) at a concentration of 1 mM was employed as a positive control. After the incubation interval, the absorbance was quantitated at 517 nm relative to a blank control34. The percentage inhibition of the DPPH free radical attributed to each extract was computed utilizing the subsequent formula:
where, A0=absorbance of control, and A1=absorbance of sample.
Catalase(CAT) and Peroxidase(POD) activities were evaluated following the protocol established by Chance and Maehly35. For CAT, a reaction mixture was prepared comprising 25 mM potassium phosphate buffer (pH 6.8), 10 mM H₂O₂, and the enzyme extract to a total volume of 1 mL. The reduction in absorbance at 240 nm was monitored to measure the decomposition of hydrogen peroxide. In the case of POD, the reaction mixture included potassium phosphate buffer (25 mM, pH 6.8), 10 mM H₂O₂, guaiacol (0.05%), and diluted enzyme extract. POD activity was assessed by monitoring the oxidation of guaiacol at 470 nm.
In order to accurately assess the enzymatic activity of Superoxide Dismutase (SOD), a reaction solution was meticulously formulated, comprising 0.3 mL of methionine at a concentration of 130 mM, 0.3 mL of nitro blue tetrazolium at a concentration of 50 µM, 0.3 mL of EDTA-Na2 at a concentration of 100 µM, and lastly, 0.3 mL of riboflavin at a concentration of 20 µM, which collectively serve as the essential components for the enzymatic reaction to occur. Following the preparation of this reaction solution, a precise volume of 0.05 mL of the enzyme solution was carefully added to the reaction mixture, which was subsequently subjected to illumination under a light intensity of 4000 lx for a duration of 30 min, thereby facilitating the necessary photochemical reactions required for the assay. To assess the SOD activity, samples were monitored using a UV/VIS spectrophotometer, with absorbance readings taken at a wavelength of 560 nm36. Enzyme activities were expresses as Units per mg protein (U/mg protein).
Statistical analysis
Two factorial analyses of variance (ANOVA) with least significant difference (LSD) test at confidence level of p ≤ 0.05 was performed on R software. Bar graphs were also made on R. Data, presented in the bar graph is mean of three replicates ± standard errors. Bars sharing same letters are not significantly different at p ≤ 0.05. Pearson’s correlation and principal component analysis (PCA) were also performed using the “FactoMineR” and factoectra” packages in R version 4.2.1.
Results
Influence of foliar application of amino acids on growth indices of Z. mays under saline stress
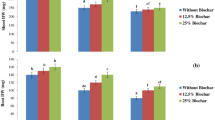
This study investigated the effects of foliar-applied amino acids on the growth parameters of Z. mays plants subjected to salt stress. Z. mays plants grown under salt stress exhibited a significant reduction in root and shoot length (23% and 32%, respectively), as well as fresh (51%) and dry biomass (40%), compared to control plants (Figs. 1 and 2). The number of leaves was also adversely affected (20%) by salt stress (Fig. 3A). Foliar application of various concentrations of amino acids, including methionine, tryptophan, lysine, and glycine betaine, resulted in a notable improvement in these growth indices. Among the amino acids tested, a concentration of 100 ppm consistently yielded the most pronounced positive effects on Z. mays growth under salt stress. Specifically, methionine, tryptophan, and lysine demonstrated significant increases in shoot and root length by 32% to 43%, compared to the salt-stressed control. Regarding biomass accumulation, foliar application of tryptophan and lysine at 100 ppm led to the greatest increase (69%) in fresh biomass of shoots. Glycine betaine was superior in terms of root fresh biomass (80%), followed by tryptophan (72%) and lysine (69%). For dry biomass, tryptophan (62%) and methionine (61%) exhibited the most favorable effects on root growth. The number of leaves per plant was also positively influenced (33–52%) by amino acids treatment, with methionine being the most effective (52%)(Table 1).
Effect of methionine, tryptophan, glycine betaine and lysine foliar spray on (A) root length, (B) root fresh weight and (C) root dry weight of Zea mays plants grown under salt stress (120 mM NaCl). Data presented in the graphs is the mean of three replicates ± standard errors. Bars sharing similar letters are not significantly different at p < 0.05.
Effect of methionine, tryptophan, glycine betaine and lysine foliar spray on (A) shoot length, (B) shoot fresh weight and (C) shoot dry weight of Zea mays plants grown under salt stress (120 mM NaCl). Data presented in the graphs is the mean of three replicates ± standard errors. Bars sharing similar letters are not significantly different at p < 0.05.
Effect of methionine, tryptophan, glycine betaine and lysine foliar spray on (A) number of leaves, (B) chlorophyll a and (C) chlorophyll b contents of Zea mays plants grown under salt stress (120 mM NaCl). Data presented in the graphs is the mean of three replicates ± standard errors. Bars sharing similar letters are not significantly different at p < 0.05.
Influence of foliar application of amino acids on photosynthetic pigments of Z. mays under saline stress
This study investigated the effects of foliar-applied amino acids on chlorophyll a (Fig. 3B), chlorophyll b (Fig. 3C), total chlorophyll (Fig. 4A), and carotenoid content (Fig. 4B) in Z. mays plants subjected to salt stress. Z. mays plants exposed to salt stress exhibited a significant reduction in all pigment parameters compared to control plants. Chlorophyll a, chlorophyll b, total chlorophyll, and carotenoids decreased by an average of 36.5%, 37.2%, 37.1%, and 23.8%, respectively, under stressed conditions. Foliar application of amino acids at various concentrations resulted in a notable increase in pigment content. Among the amino acids tested, tryptophan, methionine, lysine, and glycine betaine demonstrated varying degrees of effectiveness in mitigating salt stress-induced pigment degradation. At a concentration of 100 ppm, tryptophan was most effective (60%) in increasing chlorophyll content, followed by methionine (56%) and lysine (55%). Glycine betaine, however, exhibited the greatest enhancement (75%) in chlorophyll b content. For total chlorophyll and carotenoids, glycine betaine consistently outperformed the other amino acids, 66% and 71%, respectively.
Effect of methionine, tryptophan, glycine betaine and lysine foliar spray on (A) total chlorophyll and (B) carotenoids contents of Zea mays plants grown under salt stress (120 mM NaCl). Data presented in the graphs is the mean of three replicates ± standard errors. Bars sharing similar letters are not significantly different at p < 0.05.
Influence of foliar application of amino acids on total phenolics, flavonoids, total free amino acids and total soluble proteins in Z. mays under saline stress
Salt stress can induce physiological and biochemical changes in plants. This study investigated the effects of foliar-applied amino acids on the accumulation of TFA, TSP, phenolics, and flavonoids in Z. mays plants subjected to salt stress.
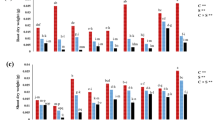
Z. mays plants grown under saline conditions exhibited a significant increase in TFA (37%), TSP (10%), phenolics (23%), and flavonoids (68%) compared to control plants. The magnitude of these increases varied depending on the specific amino acid treatment (Figs. 5 and 6A and B).
Effect of methionine, tryptophan, glycine betaine and lysine foliar spray on (A) total phenolics and (B) flavonoids contents of Zea mays plants grown under salt stress (120 mM NaCl). Data presented in the graphs is the mean of three replicates ± standard errors. Bars sharing similar letters are not significantly different at p < 0.05.
Effect of methionine, tryptophan, glycine betaine and lysine foliar spray on (A) total free amino acids, (B) total soluble proteins and (C) DPPH scavenging activity in Zea mays plants grown under salt stress (120 mM NaCl). Data presented in the graphs is the mean of three replicates ± standard errors. Bars sharing similar letters are not significantly different at p < 0.05.
Among the amino acids tested, methionine, tryptophan, lysine, and glycine betaine (GB) demonstrated varying levels of effectiveness in enhancing the accumulation of biochemical compounds under salt stress. Methionine was particularly effective in increasing TFA content by (41%), while tryptophan was superior in terms of TSP (14%) accumulation. For phenolics also, tryptophan showed the most pronounced increase (27%), followed by lysine (24%), glycine betaine (24%), and methionine (18%). In terms of flavonoids, glycine betaine (74%) and methionine (70%) were more effective than tryptophan (69%) and lysine (60%).
Influence of foliar application of amino acids on DPPH scavenging, catalase, peroxidase, and superoxide dismutase activities in Z. mays under saline stress
Z. mays plants subjected to salt stress exhibited a significant (p ≤ 0.05) increase in antioxidant activity when treated with varying concentrations (0, 25, 75, and 100 ppm) of amino acids. Compared to control plants, salt stress significantly (p ≤ 0.05) elevated enzymatic antioxidants, including CAT by 45%, POD by 39%, DPPH by 31%, and SOD by 42%.
Plants treated with 100 ppm amino acids demonstrated varying levels of CAT activity, with tryptophan exhibiting the highest increase (47%) followed by lysine (46%), methionine (44%), and glycine betaine (35%) relative to control plants under saline conditions. Similarly, POD activity was most elevated in plants treated with tryptophan (67%) and least (45%) with glycine betaine and lysine. DPPH levels increased under salt stress regardless of the amino acid applied. Lysine was the most effective in enhancing DPPH levels by 35%, followed by glycine betaine (32%), methionine (29%), and tryptophan (27%). SOD activity also followed a similar trend, with tryptophan (57%) and glycine betaine (55%) demonstrating the highest increases (Figs. 6C and 7).
Effect of methionine, tryptophan, glycine betaine and lysine foliar spray on (A) catalase, (B) peroxidase and (C) superoxide dismutase activity in Zea mays plants grown under salt stress (120 mM NaCl). Data presented in the graphs is the mean of three replicates ± standard errors. Bars sharing similar letters are not significantly different at p < 0.05.
Pearson’s correlation and principal component analysis
Data revealed that all the growth, biomass, enzymatic oxidants, metabolites, and photosynthetic pigment attributes of Z. mays plants were interrelated with significant correlations supporting the veracity of the conclusions. All the enzymatic attributes (SOD, CAT, DPPH and POD) and metabolites (primary and secondary) were negatively correlated with chlorophyll contents, biomass as well as growth parameters, in contrast, a significant positive correlation was noticed for chlorophyll and carotenoids contents with the seedling dry weight. while the phenolic contents were negatively correlated with the TSP (Fig. 8). These relationships highlight the tradeoff between stress induced metabolic activity and growth factors highlighting the role of amino acids in maintaining physiological balance under saline stress.
Pearson’s correlation among RL (Root length), RFW (Root fresh weight), RDW (Root dry weight), SL (Shoot length), SFW (Shoot fresh weight), SDW (Shoot dry weight), nL (number of leaves), Chl_a (Chlorophyll a), Chl_b (Chlorophyll b), TChl (Total chlorophyll), Carotenoids, Phenolics, Flavonoids, TFA (total free amino acids), TSP (total soluble proteins), DPPH (Scavenging activity), CAT (Catalase), POD (Peroxidase) and SOD (Superoxide dismutase) attributes studied in Zea mays plants grown in salt stress (120 mM) under the effect of methionine, tryptophan, glycine betaine and lysine foliar spray.
Principal component analysis (PCA) identified the two most dominant components, PC1 (56.69%) and PC2 (18.43%), expressing 75.12% of the total variability (Fig. 9A and B). The PCA biplot showed two distinct clusters of parameters. The first cluster, located in the positive PC1 and PC2 quadrant, comprised stress related parameters (e.g., SOD, phenolics, flavonoids) reflecting enhanced antioxidant and metabolite accumulation under stress condition. The second cluster, in the positive PC1 but negative PC2 quadrant, included growth parameters such as fresh root weight, chlorophyll content, root and shoot length expressing parameters linked with enhanced growth and photosynthetic parameters. This separation showed the mitigation of salt induced oxidative stress while positively enhanced plant biomass and photosynthetic pigments under the influence of amino acids supplementation. Numerical 1–32 represented various treatments employed in this study. Where numbers 1–16 represented treatments of Methionine, Tryptophane, Glycine, and Lysine at 0, 25, 75, and 100 (ppm) in control whereas 17–32 represented these treatments in stressed plants. For instance, methionine (100PPM) increased shoot length by 28%, root length by 24%, while photosynthetic pigments by 31% as compared to salt stressed plants. Likewise, tryptophan improved plant dry weight by 22%, while glycine and lysine increased carotenoid content by 19% and 21%, respectively. These quantitative improvements proved the efficacy of amino acids in alleviating salt stress in Zea mays L.
Principal component analysis. (A) percentage of explained variables in all principal components (PCs) and (B) PCA biplot (explaining contribution of variables in first two PCs). Length of arrows from origin point depicted contribution each parameter studied in this work. Alignment of arrows with PC1 and PC2 showed positive and negative correlations among parameters. Numerical 1–32 represented various treatments employed in this study. Where numbers 1–16 represented treatments of Methionine, Tryptophan, Glycine and Lysine at 0, 25, 75 and 100 (ppm) in control whereas 17–32 represented these treatments in stressed plants. (Various abbreviations used are same as given in Fig. 8).
Discussion
Plants have built up many mechanisms to withstand salt’s existence inside the cells or for the exclusion of salt from their cells. The response of crops towards salt stress and their strategies may vary from crop to crop22. In this regard, the exogenous application of amino acids is a well-known agriculture practice worldwide as it links with sustainable agriculture due to their amazing role in photosynthesis, nutrient absorption, antioxidant regulation and signaling pathways of plants which leads to good quality crops37. Therefore, we assessed the effectiveness of four exogenously applied amino acids on Z. mays plants in the upregulation of growth and biochemical processes exposed to salt stress. In the current study, it is recorded that the application of amino acids not only improves the growth indices in the control but also under salt stress which is detrimental to plant health same in the case of Z. mays plants. Rather than acting uniformly, each amino acid exhibits parameter-specific effects, showing their biochemical roles11,38. As, glycine betaine particularly enhanced chlorophyl b and stomatal conductance while tryptophan modulates catalase activity39. These differential effects suggest that specific amino acid target specific processes resulting in mechanistic explanation for their benefits40. This confers that foliar spray of amino acids improved growth attributes of Z. mays under salt stress.
In comparison to other physiological functions in plants, photosynthetic machinery is very sensitive to stress as pigments are reduced under stress. In the present study, chlorophyll content and carotenoids were positively influenced by the application of different concentrations of amino acids. In this regard, Hamani et al.22 also positively linked GB with stomatal characteristics such as an increase in transpiration rate (Tr), stomatal conductance (gs), intracellular CO2 concentration (Ci), and evolution of photosynthetic rate (Pn) in cotton seedlings. Therefore, it is suggested that GB positively influences photosynthetic machinery and related processes. Similarly, Yasmin et al.15 also reported that soil-applied tryptophan application increased the chlorophyll biosynthesis in Z. mays. It is found that amino acids are involved in the synthesis and stability of photosynthetic pigments. Furthermore, amino acids are also involved in the cofactors essential for activity of enzymes required for pigment biosynthesis41. Also, amino acids contributed to the synthesis of antioxidants42. Which in turn protect photosynthetic pigments from damage caused by oxidative stress. Amino acids application can also facilitate the bioavailability of micronutrients39 such as Fe and Mg which are essential of chlorophyll synthesis. Our study also showed the positive effect of amino acids on photosynthetic pigments of Z. mays plants in control and stress conditions.
Excessive synthesis of metabolites is always proved to be a shielding effort of the plant under a stressful environment, and the foliar spray of amino acids further increases their content which has been observed in the current study. Where phenolics and TSP were increased with the application of glycine betaine while flavonoids and TFA were increased in the effect of tryptophan and methionine, respectively, under salt stress. Our results are quite comparable with the reports of Rahman et al.43 who investigated the regulation of metabolites under foliar application of amino acids. It is believed that amino acids exogenous application can directly increase the pool of free amino acids within plant cells. Besides this, amino acids like methionine, tryptophan, glycine betaine, and lysine can also serve as the precursor of other amino acids and metabolites44, indirectly influencing the overall amino acids profile. Amino acids are building of proteins45, thus exogenous application of amino acids can stimulate protein synthesis. Furthermore, many amino acids can enhance enzyme activity involved in protein synthesis. These enzymes are also involved in other metabolic pathways responsible for the synthesis of secondary metabolites such as phenolics and flavonoids compounds5. This suggests that amino acids foliar spray enhance plant stress tolerance by increasing the production of primary as well as secondary metabolites, leading to improved growth under stress conditions.
Under stress conditions, the overproduction of reactive oxygen species (ROS) is always evident as a sign of oxidative stress which causes the improvement of enzymatic antioxidants. In the present study, CAT and superoxide were increased under the application of tryptophan while SOD and DPPH scavenging assay were enhanced under the effect of GB and lysine, respectively, under salt stress. In line with our findings, it has been reported previously, the involvement of GB in the efficient scavenging of excess free radicals by upregulating the CAT, POD and SOD enzyme activities via activating the ROS scavenging genes i.e. CaSOD, CaCAT, and CaPOD in Capsicum annum21. Other reports also highlighted the role of other amino acids in the activation of these enzymes7,46,47. Most of the antioxidant’s enzymes are proteins. Being building blocks of proteins, amino acids are directly involved in the synthesis of these enzymes45. Amino acids are also involved in protein turnover, i.e., replacement of damaged/degraded proteins. Thus, the degraded antioxidant enzymes are replenished by new one and their high level is maintained even in stress conditions with the application of amino acids which work as osmolytes to protect the cellular machinery of the plants18. Amino acids also act as signaling molecules, activating transcription factors that promote expression of antioxidant enzymes genes48. Amino acids also influence the synthesis or signaling hormones like abscisic acid (ABA), which are involved in plant stress tolerance. Foliar application of amino acids can regulate antioxidant defense mechanisms in plants also help in rapid accumulation of metabolites under stress by activating biosynthetic pathways and suppressing the catabolic processes48,49. These findings indicate that amino acids not only serve as precursors of proteins but also stimulate secondary metabolic pathways and antioxidant defense, thereby mitigating salt induced damage on Z. mays. This data could be useful for farmers and stockholders to formulate amino acids commercial preparations. Previous studies have typically focused on individual amino acids, while this side-by-side The novelty of this study lies in comparative evaluation of four biochemically distinct amino acids under saline conditions. Further, it is suggested to link or verify this data using the latest omics approaches and investigate the gene expression changes in Z. mays plants after amino acid application by high throughput techniques. There are some limitations related to this study that whole experiment was conducted under controlled conditions which may not fully reflect field experiments, and there was lack of molecular evidence to uncover gene regulation and protein level changes to support the physiological responses.
Conclusion
Amino acids, the fundamental building blocks of proteins, play a multifaceted role in plant growth, development, and stress tolerance particularly, glycine betaine and methionine. By directly supplying essential nutrients, regulating metabolic processes, and influencing the synthesis of secondary metabolites, amino acids can significantly enhance plant performance, suggesting the functional role of amino acids in enhancing physiological resilience under saline conditions. Foliar applications of amino acids have been shown to improve various aspects of plant physiology, including enhanced growth, improved stress tolerance, and increased secondary metabolite production. By supporting protein synthesis, enzyme activity, and nutrient uptake, amino acids can stimulate plant growth and development. Moreover, their ability to increase antioxidant enzyme activities, regulate hormone signaling, and provide Osmo protectants helps plants cope with adverse environmental conditions. In conclusion, amino acids are valuable tools for enhancing agricultural productivity and sustainability. This work supports the development of amino acid based foliar sprays as cost effective and ecofriendly strategy for mitigating the effects of salt stress in maize cultivations, particularly in regions affected by soil salinization also offer a potential alternative to synthetic growth regulators. Future field experiments and molecular validation will further help in product development to assess their long-term effects on soil health and crop productivity across different agroecological zones.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Shahid, S. A., Zaman, M. & Heng, L. Soil Salinity: Historical Perspectives and a World Overview of the Problem. In Guideline for Salinity Assessment, Mitigation and Adaptation Using Nuclear and Related Techniques 43–53 (2018) https://doi.org/10.1007/978-3-319-96190-3_2.
Giordano, M., Petropoulos, S. A. & Rouphael, Y. Response and defence mechanisms of vegetable crops against drought, heat and salinity stress. Agriculture 11, 463 (2021).
Du Jardin, P. Plant biostimulants: Definition, concept, main categories and regulation. Sci. Hortic. 196, 3–14 (2015).
Rouphael, Y. & Colla, G. Editorial: Biostimulants in agriculture. Front. Plant Sci. 11(124–133), 5 (2020).
Kelly, B. & Pearce, E. L. Amino assets: how amino acids support immunity. Cell Metab 32, 154–175 (2020).
Shahid, S. et al. Methionine-induced regulation of secondary metabolites and antioxidants in maize (Zea mays L.) subjected to salinity stress. Gesunde Pflanzen 75, 1143–1155 (2023).
Maqsood, F. M. et al. Methionine promotes the growth and yield of wheat under water deficit conditions by regulating the antioxidant enzymes, reactive oxygen species, and ions. Life 12, 969 (2022).
Merwad, A. R. M., Desoky, E. S. M. & Rady, M. M. Response of water deficit-stressed Vigna unguiculata performances to silicon, proline or methionine foliar application. Sci. Hortic. 228, 132–144 (2018).
Arruda, P. & Barreto, P. Lysine Catabolism Through the saccharopine pathway: Enzymes and intermediates involved in plant responses to abiotic and biotic stress. Front. Plant Sci. 11, (2020).
Okla, M. K. et al. Foliar application of iron-lysine to boost growth attributes, photosynthetic pigments and biochemical defense system in canola (Brassica napus L.) under cadmium stress. BMC Plant Biol 23, 648 (2023).
Ahmad, R. et al. Relief role of lysine chelated zinc (Zn) on 6-week-old maize plants under tannery wastewater irrigation stress. Int. J. Environ. Res. Public Health 17, 5161 (2020).
ul Aibdin, Z. et al. Combined effect of Zinc lysine and biochar on growth and physiology of wheat (Triticum aestivum L.) to alleviate salinity stress. Front Plant Sci 13, 1017282 (2023).
Mustafa, A., Imran, M., Ashraf, M. & Mahmood, K. Perspectives of using L-tryptophan for improving productivity of agricultural crops: A review. Pedosphere 28, 16–34 (2018).
Mohamed, M. F., Thalooth, A. T., Essa, R. E. Y. & Gobarah, M. E. The stimulatory effects of tryptophan and yeast on yield and nutrient status of wheat plants (Triticum aestivum) grown in newly reclaimed soil. Middle East J. Agric. Res. 7, 27–33 (2018).
Yasmin, H., Nosheen, A., Naz, R., Bano, A. & Keyani, R. L-tryptophan-assisted PGPR-mediated induction of drought tolerance in maize (Zea mays L.). J. Plant Interact. 12, 567–578 (2017).
Baqir, H. A. & AL-Naqeeb, M. A. S. Effect of some amino acids on tillering and yield of three bread what cultivars. Iraqi J. Agric. Sci. 50, 20–30 (2019).
Ibraheim, S. K. & Mohsen, A. A. M. Effect of mineral nitrogen levels and foliar spray with tryptophan concentrations on growth, yield and quality of lettuce plants. J. Plant Prod. 12, 517–525 (2021).
Sharma, A. et al. Phytohormones regulate accumulation of osmolytes under abiotic stress. Biomolecules 9, 285 (2019).
Nazar, Z. et al. Glycinebetaine-induced alteration in gaseous exchange capacity and osmoprotective phenomena in safflower (Carthamus tinctorius L.) under water deficit conditions. Sustainability 12, 10649 (2020).
Ashraf, M. H. P. J. C. & Harris, P. J. Photosynthesis under stressful environments: An overview. Photosynthetica 51, 163–190 (2013).
Li, N. et al. Foliar spraying of glycine betaine alleviated growth inhibition, photoinhibition, and oxidative stress in pepper (Capsicum annuum L.) seedlings under low temperatures combined with low light. Plants 12, 2563 (2023).
Hamani, A. K. M., Li, S., Chen, J. & others. Linking exogenous foliar application of glycine betaine and stomatal characteristics with salinity stress tolerance in cotton (Gossypium hirsutum L.) seedlings. BMC Plant Biol 21, 146 (2021).
Billah, M., Latif, M. A., Hossain, N. & Shalim Uddin, M. Evaluation and selection of salt tolerant hybrid maize under hydroponics culture. Research on Crops. 18, (2017).
Munns, R. et al. Energy costs of salt tolerance in crop plants. New Phytol. 225, 1072–1090 (2020).
Feng, G., Zhang, Z., Wan, C., Lu, P. & Bakour, A. Effects of saline water irrigation on soil salinity and yield of summer maize (Zea mays L.) in subsurface drainage system. Agric Water Manag 193, 205–213 (2017).
Kaya, C., Ashraf, M., Dikilitas, M. & Tuna, A. L. Alleviation of salt stressinduced adverse effects on maize plants by exogenous application of indoleacetic acid (IAA) and inorganic nutrients-A field trial. Aust. J. Crop. Sci. 7, 249–254 (2013).
Krajewska, J. B., Dlugosz, O., Salaga, M., Banach, M. & Fichna, J. Silver nanoparticles based on blackcurrant extract show potent antiinflammatory effect in vitro and in DSS-induced colitis in mice. Int. J. Pharm. 585, 119549 (2020).
Arnon, D. I. Copper enzymes in isolated chloroplasts Polyphenoloxidase in Beta Vulgaris. Plant Physiol. 24, 1–15 (1949).
Lichtenthaler, H. K. Chlorophylls and Carotenoids: Pigments of Photosynthetic Biomembranes. In Methods in Enzymology vol. 148 350–382 (Elsevier, 1987).
Zhang, D. & Quantick, P. C. Effects of chitosan coating on enzymatic browning and decay during postharvest storage of litchi (Litchi chinensis Sonn.) fruit. Postharvest Biol Technol 12, 195–202 (1997).
Beketov, E. V., Pakhomov, V. P. & Nesterova, O. V. Improved method of flavonoid extraction from bird cherry fruits. Pharm. Chem. J. 39, 316–318 (2005).
Hamilton, P. B., Van Slyke, D. D. & Lemish, S. The gasometric determination of free amino acids in blood filtrates by the ninhydrin-carbon dioxide method. (1943).
Bradford, M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254 (1976).
Koca, N. & Karaman, Ş. The effects of plant growth regulators and l-phenylalanine on phenolic compounds of sweet basil. Food Chem. 166, 515–521 (2015).
Chance, B. & Maehly, A. C. [136] Assay of Catalases and Peroxidases. {black Small Square}. Methods in Enzymology Vol. 2 (1955).
Beauchamp, C. & Fridovich, I. Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44, 276–287 (1971).
Teixeira, W. F. et al. Foliar and seed application of amino acids a_ects the antioxidant metabolism of the soybean crop. Front. Plant Sci. 8, 327 (2017).
Mosa, W. F., Ali, H. M. & Abdelsalam, N. R. The utilization of tryptophan and glycine amino acids as safe alternatives to chemical fertilizers in apple orchards. Environ. Sci. Pollut. Res. 28, 1983–1991 (2021).
Jacob, R. H., Afify, A. S., Shanab, S. M. & Shalaby, E. A. Chelated amino acids: biomass sources, preparation, properties, and biological activities. Biomass. Convers. Biorefin. 14, 2907–2921 (2024).
Dinkeloo, K., Boyd, S. & Pilot, G. Update on amino acid transporter functions and on possible amino acid sensing mechanisms in plants. In Seminars in cell & developmental biology Vol. 74 105–113 (Elsevier, 2018).
Senge, M. O., Ryan, A. A., Letchford, K. A., MacGowan, S. A. & Mielke, T. Chlorophylls, symmetry, chirality, and photosynthesis. Symmetry (Basel) 6, 781–843 (2014).
Monteiro, L. S. & Paiva-Martins, F. Amino Acids, Amino Acid Derivatives and Peptides as Antioxidants. In Lipid Oxidation in Food and Biological Systems: A Physical Chemistry Perspective 381–404 (Springer, 2022).
Rahman, S., Mehta, S. & Husen, A. Use of amino acids in plant growth, photosynthetic assimilation, and nutrient availability. In Biostimulants in Plant Protection and Performance 117–127 (Elsevier, 2024).
Kolukisaoglu, Ü. D-amino acids in plants: sources, metabolism, and functions. Int. J. Mol. Sci. 21, 5421 (2020).
Lopez, M. J. & Mohiuddin, S. S. Biochemistry, essential amino acids. In StatPearls [Internet] (StatPearls Publishing, 2024).
Oliveira, A. P. S. et al. Osmoregulatory and antioxidants modulation by salicylic acid and methionine in cowpea plants under the water restriction. Plants 12, 1341 (2023).
Usman, S. et al. Melatonin and arginine combined supplementation alleviate salt stress through physiochemical adjustments and improved antioxidant enzymes activity in Capsicum annuum L.. Sci. Hortic. 321, 112270 (2023).
Trovato, M., Funck, D., Forlani, G., Okumoto, S. & Amir, R. Amino acids in plants: regulation and functions in development and stress defense. Front. Plant Sci. vol. 12 772810 Preprint at (2021).
Teixeira, W. F. et al. Amino acids as stress reducers in soybean plant growth under different water-deficit conditions. J. Plant Growth Regul. 39, 905–919 (2020).
Acknowledgements
The authors express their gratitude for the financial support received from Ongoing Research Funding program, (ORF-2025-1245), King Saud University, Riyadh, Saudi Arabia.
Funding
This research was financed by Ongoing Research Funding program, (ORF-2025-1245), King Saud University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
MHA & ZZ; Experimentation, methodology, HB, NL, & BA; Formal analysis and data curation, NA & ZuN; Supervision and validation, SU; Formal analysis and statistical analysis, AAS; Resource acquisition and investigation, SS & EAM; Conceptualization, writing—original draft preparation and HOE & AA; Formal analysis and writing—revised draft preparation. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
We declare that the manuscript reporting studies do not involve any human participants, human data, or human tissues. So, it is not applicable. Our experiment follows with the relevant institutional, national, and international guidelines and legislation.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ahmad, M.H., Zafar, Z., Bibi, H. et al. Effect of exogenously applied amino acids on photosynthetic pigments, metabolites and enzymatic antioxidants in Zea mays L. subjected to salt stress. Sci Rep 15, 39025 (2025). https://doi.org/10.1038/s41598-025-25724-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-25724-8