Abstract
Nanostructured lipid carrier represents a cutting edge drug delivery platform, offering enhanced encapsulation of lipophilic agents, high drug loading, controlled release and superior skin hydration effects due to occlusive properties. This study aimed to develop gel based NLC formulations by co-encapsulation of α-Tocopherol and α-Tocopheryl acetate (TTA) for enhanced anti-aging performance. Six formulations (TTA1–TTA6) were prepared using varying concentrations of solid and liquid lipids (lauric acid and oleic acid) and surfactant (Tween 80) with one placebo formulation (TTA0). NLC formulations were fabricated via hot melt encapsulation method and optimized based on particle size, polydispersity index and entrapment efficiency. The best optimised formulation underwent detailed characterization, including Dynamic Light Scattering, Fourier Transform Infrared Spectroscopy, X-ray Diffraction, Differential Scanning Calorimetry, and Thermo Gravimetric Analysis. All formulations showed high encapsulation efficiency (66–93%), particle sizes ranging 68–290 nm, spherical morphology, and thermal stability. A two phase release profile was seen including burst and then sustained release. Comparative in vitro and ex vivo evaluations of gels confirmed superior skin retention and minimal transdermal permeation. In vivo testing on human volunteers revealed significantly enhanced moisture retention (up to 68%) and improved skin elasticity for gel based NLC formulations (TTA2G) as compared to solution gel (TTAsolG). These results confirm the synergistic potential of co-encapsulation of TTA in nanostructured lipid carriers to improve topical delivery and anti-aging outcomes.
Similar content being viewed by others
Introduction
Lipid based nanocarriers are known to create an ultra thin layer over the skin, which promotes deeper penetration of actives into the stratum corneum, thereby enhancing both cosmetic performance and therapeutic outcomes1. Among these, Nanostructured Lipid Carriers (NLCs); a more advanced generation of Solid Lipid Nanoparticles; are distinguished by their capacity to hold and entrap drugs efficiently. This is attributed to their disordered lipid matrices, which support sustained drug release and heightened skin moisture retention. Their hybrid structure of solid and liquid lipids contributes to improved formulation stability, better transdermal drug flow, and a significant occlusive effect that limits transepidermal water loss2.
As the body’s largest organ, the skin plays a crucial intermediary role, managing the interaction between internal systems and environmental exposures3. The main barrier to effective drug permeation lies in the stratum corneum; a layered architecture that governs permeability and diffusion4. Persistent exposure to sunlight and airborne toxins subjects the skin to oxidative stress, accelerating premature aging signs such as wrinkles5. Skin hydration level; particularly within the stratum corneum; is vital for maintaining smoothness and overall dermatological health. Reactive oxygen species produced by UV, visible, and infrared radiations contribute not only to aging but also to skin malignancies. Antioxidants like vitamins A, C, and E, along with carotenoids, serve as the body’s defense line against these damaging species6.
With progress in nanotechnology, newer delivery systems such as liposomes, nanoemulsions, and lipid based nanoparticles have emerged as effective means for targeted drug application and controlled release in dermatological treatments7. α-Tocopherol, naturally present in the stratum corneum, is a powerful antioxidant recognized for mitigating UV induced oxidative damage and is gaining attention in both therapeutic and cosmetic fields8.
α-Tocopherol, with a molecular weight of 430.71 g/mol, is a lipid soluble antioxidant often found as a colorless to light yellow viscous oil. It is either odorless or has a faint, characteristic aroma. Due to its lipophilic composition, α-Tocopherol is water insoluble but readily dissolves in organic solvents like ethanol, chloroform, and acetone and its peak UV absorbance lies between 292 and 298 nm, making it suitable for spectrophotometric detection in formulation studies9. The esterified counterpart of α-Tocopherol is α-Tocopheryl acetate, which is slightly heavier with a molecular weight of 472.75 g/mol. It is typically a pale yellow to transparent oily liquid, nearly odorless, and shares a similar solubility profile such as insoluble in water but freely miscible with oils and organic solvents. It shows UV absorbance around 285 nm due to the chromanol ring, aiding in its analytical assessment10.
The skin endures constant oxidative challenges from UV radiation and environmental factors; accelerating the aging process. Both α-Tocopherol and its more stable ester form, α-Tocopheryl acetate, serve as potent antioxidants capable of defending skin tissues against photo induced damage. However, their practical use is hindered by concerns over chemical stability and skin permeation. To overcome these limitations, simultaneous incorporation of both forms into nanostructured gel carriers has been proposed as a modern solution to enhance their combined anti-aging potential and reshape approaches in cosmeceutical formulations11.
Recent scientific advances have focused on combining antioxidant agents within nano enabled delivery systems to combat oxidative skin aging. Embedding both α-tocopherol and α-tocopheryl acetate into lipid based matrices provides a strategic advantage, thus allowing dual antioxidant effects while addressing individual drawbacks like instability and poor water solubility. Although α-Tocopherol offers rapid free radical neutralization through its hydroxyl group but its instability is a known drawback. In contrast, α-Tocopheryl acetate, a more stable prodrug, undergoes enzymatic transformation in the skin, ensuring sustained antioxidant release and longer shelf life12.
Gelling agent and dispersion media interlink to form gel; a semisolid system. These are less greasy, non-toxic, non-invasive and inert to pharmaceuticals. Since they often have a local effect at the application site, so they show very few side effects. As they are less oily than other topical preparations such as cream, water in oil lotion and ointment so they are easier to wash off. Since they don’t pass through gastrointestinal tract, they eliminate food drug interactions13.
Using nanostructured gel system boosts the effectiveness of this dual action delivery by enabling better skin penetration through occlusion, maintaining release control, and safeguarding activities from environmental exposure. These formulations also show favorable rheological properties that improve ease of application and user compliance. Furthermore, such systems enhance skin hydration level that is a critical factor in preserving barrier integrity and elasticity, which ultimately contributes significantly to anti-aging results14.
From a formulation viewpoint, encapsulating both tocopherol derivatives within a lipid matrix make optimal use of their physicochemical compatibilities. This system can be further enhanced by integrating functional excipients like penetration enhancers, pH adjusters, and film-forming agents to optimize product performance. As concerns over aging skin rise in both medical and cosmetic industries, this nanotechnology based strategy promises the development of next generation topical treatment offering superior bioavailability, improved stability, and high consumer appeal.
Materials and methods
Materials
Combination of α-Tocopherol and α-Tocopheryl acetate (TTA) was used as an active agent. We purchased α-Tocopherol from Applichem Biochemic, Germany, α-Tocopheryl acetate was obtained as a kind gift from Merck laboratories, Pakistan. Lauric acid (solid lipid), ethanol, methanol, carbopol940 (gelling agent), triethanolamine were sourced from (Sigma Aldrich Germany), while Tween 80 (Daejung Korea) and oleic acid (Merck Germany)were used as surfactant and liquid lipid.
Selection of solid lipid, liquid lipid and surfactant
Lauric acid as a solid lipid and oleic acid as a liquid lipid was selected for preparing NLCs. For this purpose solubility of TTA was checked by using previously used method with slight modification15. Briefly 10 mg TTA was added in 5 g lauric acid and 5mL oleic acid in separate glass vials and heated up to 60 °C (to establish the temperature condition used for NLC preparation). Both vials were checked visually and solubility of TTA was confirmed by the absence of any crystal of lipid and attaining transparent or clear solution.
Tween 80 is a non-ionic surfactant that is most widely used for preparing dermal products16. It was selected as a surfactant out of three surfactants (other two were poloxomer407 and span60) in preliminary work on the basis of apparent clarity and stability of NLC formulation. Formulation was gel like and having milky appearance when used poloxomer407 and span60 respectively.
Fabrication of TTA loaded NLC formulations
The NLC formulations incorporating TTA were developed using the hot melt encapsulation technique Initially, the solid lipid, liquid lipid and active agent (TTA) were sequentially introduced into glass vials. The mixture was subjected to a controlled heating process, starting from 10 °C and gradually increasing up to the range of 55–60 °C, with continuous magnetic stirring to ensure homogeneity (at 500 rpm). In parallel, the aqueous phase; comprising of water and Tween 80; was heated to the same temperature range. This aqueous solution was then gradually added to the lipid phase under gentle stirring at 500 rpm to promote proper emulsification. Following this, the entire formulation was immediately transferred into an ice cold water bath, maintaining the temperature between 2 °C and 5 °C, to facilitate solidification and nanostructure formation17. For comparison purposes, solution containing free TTA (TTAsol) was also prepared in a similar manner as mentioned above for preparing NLC of TTA except using any lipid. Briefly; Tween 80 was added in a glass vial containing TTA and heated, water in another glass vial was also heated at the same temperature (heating conditions as mentioned above).
TTA2 nanoformulation gel (TTA2G) and TTA2 solution gel (TTAsolG) preparation
Carbopol940 was chosen for this research due to its exceptional gelling capabilities, making it highly suitable for the formulation of NLC gel at a 1% concentration. The powdered form of Carbopol940 was gradually incorporated into pre-formulated NLC formulation TTA2. This blend was subjected to continuous mixing using an overhead stirrer set at 950 rpm for approximately one hour to ensure uniform dispersion18. To adjust the pH of the resulting mixture to a physiologically acceptable range (5.5–6.5), triethanolamine was introduced drop by drop until neutralization was achieved and subsequently left undisturbed at ambient room temperature for eight hours to allow complete gel formation and stabilization19. In addition to NLC gel, solution gel (TTAsolG) was also prepared by directly dispersing gelling agent into the solution of TTA using the same protocol applied for NLC gel production20.
Drug loading capacity (DLC) and entrapment efficacy (EE)
To determine the percentage drug loading capacity (%DL) and percentage entrapment efficiency (%EE), approximately 2 mg of the prepared NLC formulation was centrifuged at 12,000 revolutions per minute for one hour. The obtained supernatant was carefully collected, rinsed with distilled water to eliminate any unbound drug residues, and centrifuged once more under the same conditions. Following this, the final supernatant was diluted appropriately, and its absorbance was measured using a UV Visible spectrophotometer. Prior wavelength scans had been conducted to identify the λmax value, revealing that TTA exhibited peak absorbance at 289 nm20.
%EE and %DL were calculated by using Eqs. (1) and (2)
WT = Total amount of TTA added in NLC, WF= Amount of free TTA in supernatant, WLip. = Amount of total lipid used.
Among the various formulations prepared, the one labeled TTA2 was identified as the most optimized due to its superior drug entrapment capacity, enhanced loading efficiency, and favorable particle size, along with its comparatively smaller particle size and lower polydispersity index (PDI). Based on these optimized parameters, this formulation was selected for comprehensive characterization studies. Subsequently, it was incorporated into a gel matrix to carry out more evaluations.
Particle size, Poly dispersity index, and zeta potential
To determine the size distribution of particles and zeta potential of the TTA NLC formulations, the DLS (dynamic light scattering) technique was employed, and a particle size of nano series (Malvern Instruments, ZEN3600) was used. From each formulation, a 1 mL sample was carefully transferred into a cuvette and appropriately diluted using distilled water to facilitate the particle size and PDI measurement. For measuring surface charge, an equal volume (1 mL) of each NLC formulation was introduced into an electrophoretic cell to assess the zeta potential of the nanoparticles20.
Drug release study
Using a membrane with a mol. wt. cut off value of 14 kda, the dialysis bag method was used to perform in vitro release behavior. After tying the tube on one end, the NLC formulation containing 2 mg of TTA was placed in a 60 mL solution of release media. Ethanol was added in a 1:1 into the acetate buffer (pH 5.8) as a release medium. Sink conditions were maintained with the addition of 0.5% Tween80. A 24 h release study was conducted at 37˚C and 100 rpm.
The amount of active agent present in each sample was derived through a standard curve. Release profile was constructed by plotting concentration vs. time curve. In this way the release profile of optimized formulation was obtained. Absorbance of different concentrations of TTA was taken at maximum absorbance wavelength in ethanol for constructing standard curve. Absorbance of samples was taken thrice then mean was used20.
Antioxidant activity by DPPH method
The antioxidant potential of TTA and TTA2 was evaluated using the 2,2-di phenyl-1-picryl hydrazyl (DPPH) free radical scavenging assay20. A standard DPPH stock solution was freshly prepared, and the test samples including; the pure compound (TTA) and NLC formulation (TTA2); were diluted with methanol to achieve a 1 mg/mL concentration based on the active constituents. For the assay, 3 mL of the DPPH solution was introduced into each prepared sample solution individually. Methanol alone was used as a control. The mixtures were then subjected to thorough shaking and incubated in complete darkness for 30 min to prevent light induced degradation. All experiments were conducted in triplicate to ensure accuracy and reproducibility. Each sample was checked for absorption at a wavelength of 517 nm using a UV visible spectrophotometer21. The efficiency of each formulation was expressed as the inhibition (%) of the DPPH radical, calculated using Eq. (3)
Transmission electron microscopy
NLC formulation TTA2 was initially diluted in a 1:10 ratio using distilled water and subsequently subjected to sonication for a duration of 5 min. Sonication with mild conditions of power and duration was done just to break down the agglomerates of particles. Following this, the dispersion was carefully applied onto a carbon coated copper grid. For contrast enhancement, the grid was negatively stained using a 2% solution of phospho tungstic acid. After staining, the sample containing grid was allowed to air dry under ambient conditions before being analyzed under a transmission electron microscope (JEM1010 JEOL) that was operated at 80 kV voltage20.
FTIR
Infrared spectral analysis was conducted to investigate potential chemical interactions among the constituents. Spectra were acquired for solid lipid, liquid lipid, pure TTA, and the optimized NLC formulation TTA2. Measurements were carried out using an FTIR spectrophotometer (Burker tensor series#27 made by Germany) across the wavelength range of 4000 to 400 cm−1, maintaining a transmittance range between 85% and 100%. A zinc selenide attenuated total reflectance (ATR) accessory was employed, and each sample underwent 16 scans to ensure spectral accuracy and reproducibility22.
XRD
Powdered X-ray diffraction was conducted for Lauric acid (solid lipid), physical mixture of Lauric acid + Oleic acid (solid lipid + liquid lipid) and TTA2 by using powder X-ray diffractometer (D8 Advance, Burker Axs, Germany). For this purpose TTA2 (NLC dispersion) was lyophilized by previously reported method23.
We were unable to conduct XRD analysis of TTA due to its highly viscous nature. This is the limitation of our study. XRD was performed by placing sample in sample holder and scanning was in 2θ angular range of 30° to 80°, at a consistent scanning rate of 0.3 s per step.This allowed the identification of any characteristic diffraction peak indicative of the formulation’s structural order24.
DSC and TGA
Thermal behavior of the developed NLC formulation was assessed using a Differential Scanning Calorimeter (Series diamond, PerkinElmer TG and DTA). The samples were weighed into standard alumina crucibles and analyzed under a continuous flow of nitrogen gas. The temperature program ranged from 30 to 500 °C, enabling the observation of melting, crystallization, and possible degradation events. Additionally, thermo gravimetric data was collected simultaneously to evaluate the percentage weight loss, facilitating an understanding of the formulation’s thermal stability and composition25.
Evaluation of gels
Organoleptic evaluation, pH and electrical conductance
To assess the stability profile, samples of both NLC gel and solution gel were stored under controlled environmental settings, including ambient room temperature (25 °C), refrigerated conditions (8 °C), and elevated temperature in an incubator (40 °C). The study spanned a total duration of 12 weeks, with samples being analyzed at specific intervals ranging from, after preparation to the 12th week. At each time point, critical physical attributes such as color, odor, overall aesthetic appearance, phase separation, pH, conductance and spreadability were carefully documented. pH and electrical conductance was measured by inserting the respective probe directly in to the gel sample26.
Rheological assessment
Rheological behavior of the gels was systematically analyzed by evaluating parameters such as viscosity, shear rate, and shear stress. All tests were conducted at ambient room temperature using spindle CP41(BrookField rheometer DVIII). Around 0.5 g of gel was placed in the rheometer’s sample holder, and measurements were carried out at rotational speeds ranging from 20 to 100 rpm. Each test was performed in triplicate to ensure statistical reliability27. The data obtained was further analyzed using the power law model to compute the consistency index (K) and flow behavior index (n) by using Eq. (4).
τ = Stress, K = Consistency index, D = Shear rate, n = flow index.
In vitro occlusion test
To evaluate the occlusive potential of the formulations, a simple in vitro occlusion test was performed. A glass beaker was filled with distilled water up to 50 mL, then covered with filter paper (measuring 18.8 cm²). A fixed amount (200 mg) of TTA2G and TTAsolG was evenly spread on filter paper. The beakers were subsequently sealed and weighed. Both beakers were then put inside an incubator maintained at 32 °C with a controlled relative humidity of 50% for a duration of 48 h. For comparison, a separate beaker containing only distilled water and covered with untreated filter paper served as the control. After the 48 h incubation period, both beakers were reweighed, and the loss in water weight due to evaporation was recorded. The percentage occlusion factor (F) for both gels was then computed using the Eq. (5)27.
A = Water loss from control beaker, B = Water loss from sample beaker.
Sun protection factor (SPF)
The sun protection factor (SPF) of each gel formulation was determined individually. For this purpose, one gram of the respective gel was accurately weighed and transferred into a volumetric flask. Ethanol was then added to the flask until the total volume reached 100 mL. This mixture was subjected to sonication for a duration of five minutes to ensure thorough dissolution and homogeneity. Subsequently, the resulted solution was filtered through a layer of cotton to remove any undissolved particles. The initial 20 mL of the filtrate was discarded to avoid contamination or impurities, and 10 mL of the remaining clear solution was carefully transferred into a second volumetric flask. Ethanol was again added to this flask to adjust the volume to 100 mL.
This step wise dilution ensures that the solution is uniform and free from any particulate matter that could interfere with the absorbance measurements. By preparing the solution in a controlled manner, the method guarantees accurate readings. The absorbance of the final diluted solution was recorded using a UV Visible spectrophotometer over the wavelength range of 290 nm to 320 nm. This wavelength range corresponds to the UVB region, which is primarily responsible for erythema or sunburn, making it the standard range for SPF evaluation. The SPF values were then computed using Eq. (6), which accounts for the intensity of solar radiation and the erythemal response of the skin28.
SPF = Sun protection factor (spectrophotometric), CF = Correction factor equals to 10, EE = Erythemal effect, I = Solar intensity, abs = Sample absorbance.
Ex vivo Franz diffusion cell study
For this study, the Franz type diffusion cell apparatus, with an area of 1.76 cm2 and receptor volume of 12 mL, was employed. Rat skin was chosen as the biological membrane due to its structural resemblance to human skin, as supported by prior studies20. The skin of healthy, male rat was used for ex vivo skin permeation study. The skin was obtained from the Pharmacology research lab of the Pharmacy department, The Islamia University of Bahawalpur. All experimental protocols were approved by the Institutional Research and Ethics Committee of The Islamia University of Bahawalpur under the reference # 94/S. We confirm that all the methods and experiments were reported according to the ARRIVE guidelines and all the methods were carried out in accordance with relevant guidelines and regulations.
The excised skin was divided into two sections and mounted between the donor and receptor compartments for the TTA2G and TTAsolG. Both gels were separately applied on the donor side to maintain consistent drug concentration. Acetate buffer adjusted to pH 5.5 (resembling skin pH) was filled in the receptor chamber and stirring was continued at 100 revolutions per minute to simulate physiological conditions. The entire system was maintained at a constant temperature of 32 °C by connecting it to a thermostatically controlled water bath. This study was conducted for 24 h at intervals of 1,2,4,6,8 and 24 h. After the predetermined time, 2 mL sample was withdrawn and replaced by fresh solution to preserve sink conditions throughout the experiment. Each collected sample was analyzed for drug concentration by UV spectrophotometer, measuring absorbance at 289 nm. Ultimately, the cumulative amount of drug that permeated per unit surface area of skin was plotted against time to evaluate the permeation profile29.
In vivo study
Study design
In the present study human participants were included for non-invasive in vivo study. This study was conducted according to the guidelines and regulations approved by the Institutional Research and Ethics Committee of the Islamia university of Bahawalpur under the reference # 94/S. All methods were carried out according to the declaration of Helsinki. The study was conducted over a period spanning from December 2018 through February 2019 and involved thirteen female volunteers aged between 28 and 38 years, all of whom voluntarily agreed to participate after reviewing and consenting to the study’s terms outlined in a formal consent document. Each participant underwent thorough screening to rule out any presence of skin infections or dermatological conditions prior to enrollment. The study protocol was clearly explained to all subjects in written form, and informed consent was obtained through their signatures on the protocol documentation20.All tests, including patch test, melanin and erythema measurement, skin moisture, elasticity, and sebum evaluation, were performed on the same group of thirteen female volunteers.
Burchard test (patch test)
Prior to initiating the main study, a patch test was carried out to assess any potential skin sensitivity or allergic reaction. This test involved applying patches to the volunteers’ forearms for a duration of 48 h. For this purpose, a 4*5 cm2 area was carefully outlined on both the right and left volar forearms. A patch containing one gram of TTA2G was applied to the marked area on the left forearm, while a similar patch with one gram of TTAsolG was placed on the right forearm. These patches were then securely covered with surgical dressing to ensure adherence. Participants were explicitly instructed not to remove the patches until the 48 h period has elapsed. After removal, the forearms were cleansed with normal saline. Erythema was then evaluated by the volunteers using a standardized scale marked with 0,1,2,3 indicating no redness, mild, moderate and severe erythema respectively. Volunteers reported these scores by themselves based on the presence of redness or itching at the patch sites30.
Application of gels
Volunteers were instructed on how to self apply the gels at night before sleeping on the right and left cheek as per the study directions. Each participant received two jars, one containing TTA2G and the other containing TTAsolG, clearly labeled for application on the respective cheek. Follow up visits were scheduled between the 2nd and 12th weeks for assessment and data collection30.
Skin melanin and erythema content measurement
Pigmentation (melanin content) and redness (erythema) of skin was quantified by Mexameter MPA580® and readings were recorded at 0 h (baseline) and then at intervals of 2 weeks up to 12 weeks31.
Skin moisture level measurement
Skin hydration level was assessed using a Corneometer MPA850®, which measures skin capacitance as an indicator of moisture content. Readings were obtained at baseline (0 h) and at the same subsequent intervals of 2, 4, 6, 8, 10, and 12 weeks after application of gels. For accuracy, the mean of three independent measurements was calculated and used in analysis32.
Skin elasticity measurement
Skin elasticity was monitored using the ElastometerEM25® instrument. Measurements were recorded at 0 h (baseline) and again at the follow-up time points of 2, 4, 6, 8, 10, and 12 weeks after application of the respective gels31.
Skin sebum level measurement
The SebumeterMS815® was utilized to quantify the sebum (skin oil) levels on the volunteers’ skin. Baseline measurements were taken at 0 h, followed by assessments at 2, 4, 6, 8, 10, and 12 weeks after gels application33.
Mathematical and statistical analysis
Changes observed in skin parameters; including melanin, erythema, elasticity, sebum, and moisture content; were calculated and analyzed mathematically using Eq. (7) to determine the significance of differences before and after treatment in all volunteers.
y = Value obtained at specific time, z = Value obtained at baseline.
Results for all the parameters were compiled after taking the mean ± SD of at least three individual values. To analyze variation of results among volunteers according to time intervals, the statistical test Two Way Analysis of variance was used. To check any significant difference in results attained after applying TTA2G and TTAsolG, Student’s t-test was used. Significance level was set at p < 0.0530. The statistical analysis was done by using Graphpad prism 7.00.
Results
TTA loaded NLC formulations and NLC gel
A total of six NLC formulations (designated TTA1 through TTA6) were synthesized, each containing the dual active agent TTA. For comparative analysis, a control formulation, labeled as TTA0, was also prepared under identical conditions but without incorporating any active agent. Based on the particle size and entrapment efficiency, TTA2 was selected for formulating NLC gel for further studies (Table 1).
Drug loading capacity (DL) and entrapment efficacy (EE)
The percentage of TTA entrapped (%EE) and loading efficiency (%DL) of all formulations (TTA1–TTA6) was found in the range of 84–93% and 4–8% respectively (Table 1).
Zeta potential and particle size determination
Zeta potential, polydispersity index and particle size of all six formulations were found in the range of 68 to 215 nm, 0.223 to 0.45 and − 25.8 to -14.71 respectively as compared to blank formulation TTA0 having 290 nm, 0.45, and − 20.6 respectively. All the results are shown in Table 1.
Antioxidant activity by DPPH method
Tocopherol and its derivatives are well-known antioxidants. This fact was confirmed by conducting antioxidant activity of α-Tocopherol and α-Tocopheryl acetate (1:1) and TTA2, the resultant antioxidant activity in terms of % inhibition (± SEM) was found 94.89% (± 0.15) and 93.53% (± 0.05) respectively.
Differential scanning calorimetric study
Thermal stability of ingredients was confirmed by DSC and TGA. It is used to confirm melting point of lipid and drug to lipid interactions. This study was conducted for pure TTA, solid lipid (lauric acid) and TTA2. The resulting thermograms of DSC, which are illustrated in Fig. 1a–c, provided insights into the heat flow behavior and phase transitions of the samples. Lauric acid, serving as the solid lipid base, exhibited two distinct endothermic transitions: the first occurred at approximately 43.07 °C with a heat flow of − 0.155 W/g, and the second was observed around 234.67 °C. To further understand their thermal degradation, the percentage of weight loss in response to increasing temperature was also recorded as shown in Fig. 1d–f.
DSC and TGA thermograms of (a,d) lauric acid, (b,e) α-Tocopherol + α-Tocopheryl acetate (TTA), and (c,f) NLC formulation TTA2. XRD spectra of (g) lauric acid, (h) oleic acid + lauric acid, and (i) TTA2. TEM micrograph of (j) TTA2, with arrows indicating nanoparticles in the size range of ~ 200 nm.
XRD
Crystallinity of ingredients was confirmed by powder X-ray diffraction. X-ray diffractograms for Lauric acid, physical mixture of Lauric acid + Oleic acid and TTA2 are shown in Fig. 1g–i respectively. Sharp diffraction peak of lauric acid was shown at 38.24° with high intensity at 82.69 counts. Certain peaks appeared in the physical mixture of Lauric acid + Oleic acid in the range of 38–45° where a prominent peak of lauric acid was shown with slight shift at level of 39.82° but with lower intensity 50.31. only one prominent peak was seen at level of 40.1° in case of TTA2 but intensity was very low at 9.9 count.
TEM
Transmission electron microscopic analysis of TTA2 was done to check surface morphology of NLC nanoparticles and also to confirm that the particles are in nanometer size range. TEM analysis of TTA2 showed nearly spherical and smooth surfaced nanoparticles. Particles shown in micrograph are in size range less than 200 nm (Fig. 1j).
FTIR
Compatibility of ingredients was confirmed by FTIR. It is an important parameter to detect any interaction of ingredients with lipids used. It was conducted for pure α-Tocopherol, α-Tocopheryl acetate, solid lipid and TTA2. FTIR spectra are shown in Fig. 2a–d respectively. Prominent peaks for α-Tocopherol were observed at wavenumbers 3566.93 cm−1, 2921.97 cm−1 and 2852.19 cm−1, 1376.07 cm−1,1277.48 cm−1, 1083.5 cm−1 (Fig. 2a). TTA2 showed peaks at 2910.01 cm−1 and 2840.20 cm−1 (Fig. 2d).
FTIR spectra for (a) α-Tocopherol, (b) α-Tocopheryl Acetate, (c) Lauric acid and (d) TTA2.
Drug release study
In vitro release study was performed by dialysis bag method for TTA2 as compared to TTAsol. 32% TTA was released within the first 4 h while a cumulative release of 85% was seen after 24 h from NLC formulation TTA2. TTAsol depicted a nearly 100% release profile within 4 h. Results are shown in Fig. 3a.
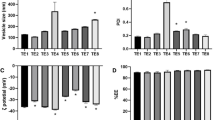
(a) In-vitro release profile of NLC formulation TTA2 and TTAsol; (b) Ex-vivo Franz diffusion cell study comparing TTA2G with TTAsolG; (c,e) pH and electrical conductance of TTA2G at different storage temperatures; (d,f) pH and electrical conductance of TTAsolG at different storage temperatures.
Evaluation of gels
Organoleptic evaluation of gels
Organoleptic evaluation of TTA2G and TTAsolG was conducted in terms of color, odor, look/feel/texture and liquefaction or phase separation. Both gels showed good stability at different storage temperatures for a study period of 12 weeks except at 40 °C; where gels turned hard and color change was also seen after 45 days. Detailed results are given in Table 2.
Ex vivo Franz diffusion cell study
Skin permeation study was conducted forTTA2G and TTAsolG. This type of study allows the measurement of release of active agent from nanoparticles through synthetic membrane or animal skin resembling human biological membranes. Cumulative.amount of TTA permeated by TTA2G was less than that of TTAsolG as shown in Fig. 3b.
pH and electrical conductance
pH and electrical conductance of TTA2G and TTAsolG was measured periodically under varying storage conditions (8 °C, 25 °C, and 40 °C) for a duration of 90 days. A gradual upward trend in pH (Fig. 3c, d) and electrical conductance (Fig. 3e, f) was observed across all samples over time.
Rheology
Results for rheological assessment regarding flow index and consistency index, by applying power law, of TTA2G and TTAsolG kept at different temperature conditions and different time points up to 12 weeks are shown in the form of rheograms between shear rate and shear stress (Fig. 4a, b at 8 °C, Fig. 4c, d at 25 °C, and Fig. 4e, f at 40 °C respectively). All samples of both gels showed non-Newtonian behavior of flow. The apparent viscosity of all formulations decreased upon shear application, with the most pronounced effects observed at 40 °C.
Shear rates vs. Shear stress rheograms and skin occlusion test. Rheogram of TTA2G and TTAsolG at (a,b) = 8 °C, (c,d) = 25 °C, (e,f) = 40 °C respectively and (g) Occlusion factor of TTA2G versus TTAsolG.
In vitro skin occlusion test
Occlusion factors of TTA2G and TTAsolG were checked after 6, 24 and 48 h. The result is shown in Fig. 4g. 58% occlusion factor was achieved for TTA2G as compared to TTAsolG that showed 8% only after 48 h.
Sun protection factor study
Sun protection factor of TTA2G and TTAsolG was conducted and more protection against UV rays was confirmed for TTA2G having SPF value of 3.70 than that of TTAsolG with SPF value 2.89. Detailed results are depicted in Table 3.
In vivo study
Burchard test (patch test)
Skin irritancy potential of TTA2G and TTAsolG was assessed by carrying out patch test on human volunteers. A decreasing trend in melanin and erythema index was seen for each volunteer individually by TTA2G as compared to increase in the values with TTAsolG. Mean value for both parameters was found to be -3.27% and − 7.65% with TTA2G and − 2.84% and − 0.63% with TTAsolG respectively.
Melanin and erythema index
Melanin and erythema level of each volunteer was found to be reduced gradually through out the study period by the use of TTA2G, while these parameters were either decreased irregularly or even increased in case of erythema level after using TTAsolG (Fig. 5a, b).
Mean% change in (a) Melanin (b) Erythema, (c) Moisture, (d) Elasticity and (e) Sebum levels by TTA2G versus TTAsolG at different time intervals.
Hydration level
The mean percentage change in moisture content of skin in volunteers was gradually increased in both cases of TTA2G and TTAsolG but results were found highly significant statistically for TTA2G which showed 68.5% moisture level after the 12th week while the level was found to be 27.6% by TTAsolG, as depicted in Fig. 5c.
Elasticity level
TTA2G showed consistent increase in elasticity level of skin with 13.36% change on 12th week of study, change in elasticity level by TTAsolG was found 3.47%. Change in elasticity level was found significantly high by TTA2G as compared to TTAsolG as Shown in Fig. 5d.
Sebum level
The characteristic features of the formulations developed for applying on the skin have a pronounced effect on the skin. These features are also associated with the release pattern of drugs. In order to maintain skin barrier properties, protecting it from drying and high level of sebum production, the pH of dermal preparations should be below 7 as the skin has a little bit of acidic pH ranging from 4 to 6. Sebum level of skin was gradually decreased after the use of TTA2G and TTAsolG (Fig. 5e). Suitable pH of TTA2 may be involved in it. Mean% reduction in sebum level was significant by both gels while insignificant in comparison of results of both gels to each other. Regarding sebum production, TTA2G achieved a notable 15.5% reduction by week 12, with TTAsolG also demonstrating a moderate decline of 11.6%. Both reductions were statistically significant (p < 0.05), although comparison between the two remained insignificant (p > 0.05).
Discussion
Selection of solid lipid and liquid lipid
Solid and liquid lipids act as the primary building blocks of nanostructured lipid carriers (NLCs). Fatty acids, which naturally serve as a lipid source, are widely used in the fabrication of lipid nanoparticles, including solid lipid nanoparticles (SLNs) and NLCs. Structurally, fatty acids are carboxylic acids composed of long hydrocarbon chains. They are typically divided into two categories: saturated fatty acids, which are solid at room temperature, and unsaturated fatty acids, which are liquid due to the presence of one or more double bond33.
Lauric acid is a medium chained, 12 carbon fatty acid with melting point 44°. In sebum some of the fatty acids are present naturally. Lauric acid has anti-inflammatory and antibacterial properties that help in combating skin bacteria, especially acne producing bacteria P.acnes. Oleic acid is a cis double bond at C-9. It is 18 carbon monounsaturated fatty acids. Lauric acid and oleic acid improved the penetration of many polar and nonpolar agents by rupturing the stratum corneum lipid bilayer in many investigations reported in literature34,35.Thats why in this study, lauric acid was employed as the solid lipid, whereas oleic acid was chosen as the liquid lipid for the preparation of NLC formulations.
Selection of method for preparing nanostructured lipid carriers
The hot melt encapsulation technique was utilized to prepare the NLC formulations. Lipophilic drugs can be easily distributed in the liquid blend of solid and liquid lipid after melting by this technique36.
Encapsulation efficiency
The elevated %EE values across all formulations (TTA1-TTA6) clearly indicate the suitability of the formulation method and the ratios of excipients used (Table 1). It is evident that by increasing the liquid lipid proportion in the lipid mixture; enhanced the entrapment efficiency and drug loading. Conversely, reducing the liquid lipid content resulted in lower %EE and %DL, likely due to an insufficient amount of solid lipid to retain TTA within the lipid core. Additionally, liquid lipids are more effective at dissolving active agents, thereby facilitating higher drug loading37. The %EE is also influenced by the concentrations of the drug and lipid mixture38, with higher entrapment efficiency observed at increased concentrations of TTA while maintaining a constant solid to liquid lipid ratio of nearly 7:3. Synthetic Tocopherol is a very viscous, light-sensitive liquid that irritates the skin. Because of this characteristic, Tocopherol is challenging for incorporating in topical or cosmetic products39. Since vitamin E has been linked to erythema multiform like responses, eczematous dermatitis, and contact urticaria, it may possibly do more harm than good when administered topically. In this research contact dermatitis was also reported40. In terms of shielding Tocopherol from UV deterioration, NLCs are thought to be more stable. Furthermore, research has shown that adding active ingredients to NLCs improve their chemical stability. Additionally, it is said that the crystalline lipid in the NLC reflects UV rays, protecting the skin from UV radiation41. High entrapment efficiency is advantageous as it is expected to minimize potential irritation caused by direct skin exposure to the active ingredient42.
Particle size
Particle size is in the form of average diameter. It is required to conduct in order to assure the development of particles in nanosized range. Particle size of all NLC formulations was in the range of 68.88 to 290 nm, suitable for dermal application. The particle size of lipid nanoparticles tended to increase with a higher proportion of solid lipid, attributed to greater surface tension of the particles. Particle size decreased linearly as the concentration of active constituent increased Table 1. Surfactant concentration significantly impacted particle size. In this study, by increasing Tween 80 concentration from 4.6% to 5% led to a decrease in particle size (TTA4 > TTA2), due to reduced interfacial tension between the lipid and aqueous phases. This reduction in interfacial tension results in the formation of smaller particles43. The influence of higher Tween 80 concentration in reducing particle size has been supported by other researches9,44. For TTA formulations, by increasing the solid lipid content while keeping the total lipid concentration constant caused an increase in particle size, likely due to increased viscosity of the system.
Poly dispersity index
Polydispersity Index (PDI) is a useful indicator of the breadth of size distribution in a unimodal system, where a value of 0 indicates a completely uniform particle dispersion and a value of 1 represents a highly heterogeneous, polydisperse distribution44,45. It is reported that a PDI range between 0.1 and 0.7 corresponds to uniform size distribution, while values above 0.7 indicate broad size distributions45. The PDI values for all NLC formulations (TTA1-TTA6) ranged from 0.223 to 0.543 as shown in Table 1, indicating monodispersed size distribution. PDI decreased with increasing surfactant concentration but increased linearly by increasing solid lipid content in the lipid blend.
Zeta potential
Zeta potential is the electric charge present on the surface of a particle that develops an electrical barrier. It plays a pivotal role in determining the physical stability of colloidal dispersions46. Furthermore, it reflects affinity for adjacent ions. It has been proposed that the nanoparticles having zeta potential value ± 30 are stable. The specific type of surfactants and the particle environment; including presence of various types of ions, their strengths and pH; may influence zeta potential value and charge on the particle47.
Zeta potential of all formulations TTA1 - TTA6 was found in the range of -14.71 to -25.8 Table 1. Although the formulations showed lower values of zeta potential, all of these were found stable at room temperature for up to 1 month. The reason for low zeta potential might be due to adsorption of Tween 80 on particles. Tween 80 also provides steric stabilization to the formulation48. The negative charge on the particles may be due to the presence of free fatty acids in liquid dispersion of NLCs because Tween 80 was used as surfactant that may also contain anionic contaminants, as described in literature47.
A noticeable increase in zeta potential was observed when the concentration of Tween 80 was raised from 4.6% to 5%. Notably, the control formulation (TTA0) exhibited a comparatively lower zeta potential than the most optimized NLC formulation TTA2.
Antioxidant activity by DPPH method
Tocopherol and its derivatives are reported for strong antioxidant activity. Tocopherol is one of the most efficient, membrane bound, lipophilic antioxidants. Tocopherol acts as an antioxidant either by slowing down the process of collagen breakdown or by donating its electrons to free radicals. Constant exposure of skin to UV rays results in reduction of skin’s natural antioxidant42. So antioxidants from outside sources are required to apply topically on the skin. Due to light resistant, viscous and irritative nature of synthetic tocopherol, it is not possible to apply directly on the skin. So encapsulation in nanoparticles is advantageous as reported in literature. So in the present study TTA was used to encapsulate in NLC. Antioxidant activity of pure TTA was conducted and strong action was shown which was 94%. After incorporating TTA in NLC; antioxidant activity of TTA2 was slightly reduced, showing a value 93% as described in Sect. 3.4. This reduction in antioxidant activity is insignificant. NLC proved to be the best carrier to protect the antioxidant activity of lipophilic agents.
DSC
According to the DSC result, the melting point of lauric acid was found 43.07 °C (Fig. 1a). In contrast, the combined TTA mixture did not reveal any sharp endothermic peaks (Fig. 1b), suggesting a significant reduction in crystallinity due to molecular entrapment. When these compounds were integrated into nano carriers, their melting transitions appeared at lower temperatures than that of pure lauric acid (Fig. 1c). This result shows the successful encapsulation of the active agent within the lipid matrix of the NLC as reported in literature49. This behavior is characteristic of lipid based nanosystem, where the incorporation of active ingredients disrupt the ordered structure of solid lipids.
TGA
Thermogravimetric analysis (TGA) offered additional insights into the thermal stability and degradation patterns. Lauric acid remained thermally stable up to 200 °C, after which weight loss was initiated. At approximately 300 °C, nearly 90% of the material had decomposed, confirming its resistance to thermal stress up to moderate temperatures (Fig. 1d). For TTA, a single step degradation profile was evident, with the compounds maintaining 99% of their mass up to 300 °C. By 380 °C, only about 8% of the original weight remained intact (Fig. 1e). TTA2 showed thermal stability up to 200 °C, beyond which decomposition began, culminating in a mass loss of approximately 95% by 300 °C (Fig. 1f).
XRD
Sharp diffraction peak of lauric acid was shown at 38.24° with high intensity 82.69 counts (Fig. 1g). Results confirmed the crystalline nature of lauric acid. Crystalline agents show sharp peaks with high intensity. Certain peaks appeared in the physical mixture of Lauricacid + Oleic acid in the range of 38- 44.5° where a prominent peak of lauric acid was shown with slight shift at level of 39.82° but with lower intensity 50.31 (Fig. 1h). This shift signifies a transformation of the lipid matrix from a crystalline to a more disordered structure, which is consistent with previous finding50. In comparison with the physical mixture, some peaks disappeared and only one prominent peak was seen at level of 40.1° in case of TTA2 but intensity was very low at 9.9 count (Fig. 1i). No other high intensity peak was seen, confirming no interaction of ingredients and successful encapsulation of TTA in the lipid matrix. Visible peaks but with low intensity show lesser crystallinity and near amorphous nature. These results are in accordance with previously reported result51.
TEM
Morphological observations confirmed that the NLC nanoparticles loaded with TTA were smooth and spherical, lacking any significant aggregation (Fig. 1j). Literature suggests that the shape of lipid nanoparticles is influenced by the purity of the lipids used. Particles synthesized using highly purified lipids often appear more cuboidal, whereas those prepared with polydispersed or mixed lipid systems tend to be spherical52,53. In the current study, the lipid matrix consisted of a blend of solid and liquid lipids, implying a polydispersed nature that favors spherical particle formation. According to previous findings, nanoparticles with diameters below 300 nm can partially penetrate deeper skin layers, whereas those under 70 nm can efficiently localize within both dermal and epidermal layers45. Based on particle size characterization, the nanocarriers developed in this study are expected to deposit primarily in the epidermis, making them well suited for delivering anti-aging agents with enhanced efficacy.
FTIR
Fourier Transform Infrared spectroscopy is a reliable analytical method used to assess potential chemical interactions between components within a formulation. It is suitable for analyzing a wide range of formulations54.
For α-tocopherol, an absorption peak was observed at 3566.93 cm−1, corresponding to hydroxyl stretching. Asymmetric and symmetric stretching vibrations of –CH3 and –CH₂ groups were found at 2921.97 cm−1 and 2852.19 cm−1 respectively. Additional signals included a methyl bending vibration at 1457.17 cm−1, phenyl group absorption at 1376.07 cm−1, –CH₂ scissoring at 1277.48 cm−1, and planar bending of the phenyl ring at 1083.5 cm−1. The trans = CH₂ stretching vibration was recorded at 917 cm−1, with C–O stretching observed between 1050 and 1250 cm−1 (Fig. 2a), in line with previously reported spectra.
The FTIR spectrum for α-Tocopheryl acetate (TA) is displayed in Fig. 2b. Key peaks include asymmetric and symmetric –CH3 and –CH₂ stretching vibrations at 2922.14 cm−1 and 2852.94 cm−1, phenyl group absorption and methyl bending at 1464.86 cm−1, methyl symmetric bending at 1376.53 cm−1, –CH₂ scissoring at 1264.76 cm−1, and planar phenyl bending at 1083.05 cm−1. A trans = CH₂ stretch appeared at 934.15 cm−155.
Lauric acid, known for its 12-carbon alkyl chain and carboxylic acid functionality, presented a strong carbonyl peak at 1695.56 cm−1. The –CH3 and –CH₂ stretching peaks were found at 2915.31 cm−1 and 2848.41 cm−1 respectively, with additional peaks at 933.97 cm−1 and 722.25 cm−1, indicative of long alkyl chains (> 3 carbon atoms) (Fig. 2c), found in accordance with previously found data56.
In the FTIR spectra of the TTA2, characteristic lauric acid peaks shifted slightly e.g., from 2915.31 cm−1 to 2910.01 cm−1 and 2848.25 cm−1 to 2840.20 cm−1 indicating successful encapsulation. The broader peak at 3355.66 cm−1 may be due to any free form of active agent (Fig. 2d). The spectra of TTA2 did not exhibit distinctive peaks of α-Tocopherol or α-Tocopheryl acetate, suggesting successful entrapment within the nanocarrier system and absence of interactions among active components. Literature verifies the post encapsulation disappearance of such peaks indicating effective drug loading57.
Drug release study
The drug release behavior of TTA from NLC formulation was investigated using the dialysis bag method. The aim of conducting this study was to know the release mechanism of TTA from nanoparticles. Within 4 h, TTA2 revealed 32% release, and after 24 h, 85% release was achieved. Within 4–6 h, TTAsol demonstrated rapid release and nearly 100% release as depicted in Fig. 3a. Three things can cause the release of active material from a NLC formulation: the receptor medium penetrating into the particle core, the active material diffusing from the particle core to external receptor media and the break down of particle shell or core. TTA on the surface of the corresponding nanocarrier or closer to the lipid mixture’s shell may be the cause of the initial burst release. The slow diffusion of active substances from the lipid mixture’s core can further explain the sluggish and continuous release. When applied to the skin, the nanocarrier is supposed to provide a regulated release after an initial rapid release58.
Such biphasic behavior is advantageous for topical delivery systems, offering an immediate therapeutic effect followed by controlled, prolonged release. When applied to skin, this mechanism suggests a quick onset of action accompanied by long term efficacy.
Evaluation of gels
Organoleptic evaluation of gels
Assurance of the physical integrity of cosmetic and pharmaceutical formulation is crucial for their effectiveness, consumer acceptance and shelf life. Various parameters including visual appearance, texture, pH, rheology and electrical conductivity serve as indicators of stability59. As per industry practices, these parameters were monitored under stress conditions to simulate long term storage. Both of the formulated gels (TTA2G and TTAsolG) were divided in to three equal parts and stored at three different temperature conditions: in refrigerator at 8 °C, ambient room temperature at 25 °C and accelerated temperature at 40 °C, over a 90 day period.
All the samples were systematically evaluated at scheduled intervals. Sensory evaluation, although subjective, remains a vital component of quality assessment due to its close alignment with consumer perception. No instrument can fully replicate the tactile, visual, and olfactory sensations experienced by users, hence the importance of this evaluation is evident60. Throughout the storage period, TTAsolG retained a transparent appearance, whereas TTA2G appeared off-white, as shown in Table 2. The texture remained smooth and consistent at 8 °C and 25 °C for all samples. However, at 40 °C, both TTA2G and TTAsolG developed a hardened texture. This transformation is likely due to moisture loss resulting from evaporation at elevated temperatures45. According to these results the best suitable storage temperature is room temperature or 25 °C.
Ex vivo skin permeation study
The ex vivo permeation analysis revealed a relatively low diffusion rate for TTA through the skin from both gels (TTA2G and TTAsolG). Interestingly, TTAsolG facilitated a higher drug permeation (0.17%) than TTA2G (0.0845%) after 24 h (Fig. 3b). These findings may be due to the structural integrity of the nanoparticles in gel matrix, which restricts active release but provides a more targeted and sustained effect.
pH and electrical conductance
Monitoring of change in pH is crucial, especially for dermal formulations, as deviation from skin-compatible pH can cause irritation or product degradation. The pH of freshly prepared TTA2G and TTAsolG was measured periodically under varying storage conditions (8 °C, 25 °C, and 40 °C) for a duration of 90 days. As depicted in Fig. 3c, d, a gradual upward trend in pH was observed across all samples over time. However, this increase remained within the physiologically acceptable range of 5.5–6.7 for healthy human skin31.
The incremental rise in pH may be attributed to the inclusion of triethanolamine; a cationic agent known for its interactions with the anionic polymer chains in carbopol based gel systems. These interactions can be intensified at higher temperatures, potentially altering the formulation’s ionic equilibrium and pH61.
An upward trend in conductivity was observed in all TTA2G and TTAsolG samples at each recorded time point (Fig. 3e, f). This consistent increase may be attributed to the coalescence of lipid globules62, which potentially influences the ionic mobility within the gel matrix.
Rheology
Graphs plotted between shear rate and shear stress revealed a non-Newtonian, shear thinning flow behavior. The apparent viscosity of all formulations decreased upon shear application, with the most pronounced effects observed at 40 °C (Fig. 4e, f) for TTA2G and TTAsolG respectively as compared to that of samples kept at 8 °C and 25 °C. These temperature dependent changes suggest enhanced molecular mobility and structural rearrangement within the gel matrix at elevated temperatures.
In vitro skin occlusion
The in vitro skin occlusion study revealed a superior occlusive effect of TTA2G over TTAsolG, as illustrated in Fig. 4g. At 48 h, TTA2G achieved a maximum occlusion factor of 58% as compared to just 8% for TTAsolG. These differences were statistically significant (P < 0.01), emphasizing the enhanced barrier forming ability of NLC gels. Occlusion, a key parameter in topical formulations, aids in moisture retention, contributing to anti-aging benefits through skin hydration enhancement63.
Using very small, highly crystalline particles, and low melting lipids result in the highest occlusion. It has been discovered that nanoparticles are fifteen times more occlusive than microparticles. An NLC containing cream was found to improve skin moisture in comparison to conventional cream. When the occlusive factor of an o/w emulsion and the NLC dispersion in the finished product were compared, the NLC dispersion showed five times greater effect than that of the o/w emulsion. There have been numerous reports of SLN and NLCs’ benefits for the dermal application of pharmaceutical and cosmetic products, including their occlusive features, enhanced skin hydration, encapsulation of poor water soluble active agents, modified release, better skin penetration linked to a targeting effect, and avoidance of systemic uptake64,65.
Sun protection factor study
Exposure of skin to ultraviolet rays of sun directs to irritation, redness, erythema and photoaging. Lipid based nanoparticles have protective effect against these factors. Whether the cosmetic or dermal product would be effective against harmful sun rays or not; can be judged by measuring sun protection factor66. In the present study SPF was measured and calculated for TTA2G and TTAsolG. A comparative view of SPF across all samples is shown in Table 3. More SPF value shown by TTA2G may be due to the encapsulation of TTA in the protective coating of lipids. Despite the numerical advantage of TTA2G, statistical analysis indicated no significant difference (p > 0.05).
In vivo study
Burchard test (patch test)
In addition to occlusion, a patch test and visual inspection for inflammation was conducted to assess skin irritation potential67. TTA2G reduced melanin levels by 3.27% and erythema level by 7.65%, suggesting a protective role against cutaneous irritation. The solution gel showed minimal changes, affirming the enhanced skin compatibility of nanocarrier formulation.
Melanin index
TTA2G exhibited a sustained reduction in melanin levels throughout the study, with a maximum decrease of 15% by day 90. In contrast, TTAsolG demonstrated inconsistent changes as shown in Fig. 5a. Two way ANOVA confirmed a statistically significant difference (p < 0.05) in melanin reduction between TTA2G and TTAsolG, while multiple comparison t-test revealed highly significant effects at weeks 4 and 6 (p < 0.01), and extremely significant changes at weeks 8, 10, and 12 (p < 0.0001). These outcomes are in alignment with the antioxidant and depigmenting actions of TTA.
In addition to combating the harmful effects; caused by free radicals; antioxidants have the ability to regulate pigmentation of skin68. α-Tocopherol is an efficient agent for skin whitening by regulating important enzymes such as protein kinase, tyrosine kinase. α-Tocopherol is also involved in protecting the skin against harmful sun rays69.
Erythema index
A steady decline in erythema levels was observed in TTA2G treated skin, with reduction of 15% and 17.6% recorded on days 60 and 90, respectively (Fig. 5b). Conversely, TTAsolG showed a persistent increase in erythema level, peaking at 2% by the end of the trial. The contrast was statistically significant (p < 0.05), and multiple t-test supported these findings (p < 0.00001) at all evaluated intervals. This may be due to the encapsulation of TTA in the lipid matrix.
Erythema is basically an expression of irritation or redness on the skin that can be triggered by elevated temperature of capillaries that are closer to the skin. Many factors such as use of specific drugs, having skin infection, more exposure to UV rays are involved in erythematic skin70. Anti erythematic effect shown by TTA2G is due to the antioxidant effect of encapsulated TTA in NLC while higher level of erythema shown by TTAsolG can be related to sensitivity of α-Tocopherol towards light and heat. These factors are involved in degradation of α-Tocopherol71.
Hydration level
TTA2G significantly boosted skin hydration, achieving a 68% increase in moisture level by the 12th week (Fig. 5c).
The agents used for keeping the skin moist work in two ways: (1) By developing an occlusive layer on skin to prevent trans epidermal water loss, (2) By making the skin suitable for entering the agents having ability to keep the skin hydrated72. In present study enhanced moisture level is attributed to the lipid based NLC forming an occlusive barrier, limiting trans epidermal water loss. This result also confirms the result obtained by in vitro occlusion test. There are numerous research articles proving the ability of lipid based nanoparticles to improve moisture content that is associated with reduction in appearance of wrinkles. This fact is ultimately the key factor involved in slowing down of aging process73.
Elasticity level
Skin elasticity was improved for both TTA2G and TTAsolG over the 12 week period, reaching maximum increase of 13.36% and 3.47%, respectively (Fig. 5d). Two way ANOVA confirmed the significance of results (p < 0.001). With the passage of time, skin starts to lose its elasticity. Aging of skin is associated with dryness and roughness that ultimately lead to appearance of wrinkles. Collagen present in skin plays an important role in the tightness of skin that can be disrupted by sun rays. Nanoparticles protect skin against harmful sun rays and antioxidants such as vitamin E and ascorbic acid are proven to protect skin’s elasticity69.
Sebum level
Acne is thought to be primarily caused by increased sebum production. After the application of TTA2G and TTAsolG, the sebum-suppressing action was seen, as illustrated in Fig. 5e. BY applying two way analysis of variance both gels showed decrease in sebum level at different time points. But TTA2G showed a more pronounced effect as compared to TTAsolG. According to reports, tocopherol and fatty acids play a role in suppressing 5α-reductase activity, which converts testosterone into dehydrotestosterone which is leading cause of sebum production and, ultimately, acne74. According to a study fatty acids such as oleic acid, linoleic acid etc. are involved in inhibiting the action of 5α-reductase so decreasing of excessive sebum production75. Reduction in sebum level may be due to the presence of fatty acids in TTA2G. However, an insignificant (p < 0.05) difference of result was shown after applying multiple t-test. To confirm the primary contributing factor, further investigation is needed.
Conclusion
In this study, we successfully developed a nanostructured lipid carrier based gel, TTA2G, which remained physically stable for three months at 25 °C. Both In vitro and In vivo tests confirm that the gel does not irritate human skin. The release profile, Ex vivo skin permeation, and SPF studies indicate its potential application as a sunscreen; however, further refinement in this area would benefit the cosmetics industry.
Importantly, the gel has the ability to retain skin moisture level, as demonstrated by In vitro occlusion test and In vivo moisture level study. Since more hydrated skin wrinkles more slowly, our gel formulation shows strong potential as an anti-aging product. One limitation of this study is the small sample size for the In vivo testing. We recommend future studies with a larger sample size from diverse demographic regions to better assess safety and efficacy. Additionally, comparing different novel drug delivery systems with encapsulated α-Tocopherol and α-Tocopheryl acetate in combination can open new avenues in drug development.
In conclusion, we found our NLC gel (TTA2G) to be a safe and suitable product for skin rejuvenation, depigmentation, sun protection, and anti-aging.
Data availability
The datasets used and/or analyzed during the current study would be available from the corresponding author on reasonable request.
References
Ahmad, A. S. & Sharma, R. Comparative analysis of herbal and allopathic treatment systems. Eur. J. Mol. Clin. Med. 7 (7), 2869–2876 (2020).
Viegas, C. et al. Solid lipid nanoparticles vs. nanostructured lipid carriers: a comparative review. Pharmaceutics 15 (6), 1593 (2023).
Papaccio, F., Caputo, S. & Bellei, B. Focus on the contribution of oxidative stress in skin aging. Antioxidants 11 (6), 1121 (2022).
Darvin, M. E. et al. Carotenoids in human skin in vivo: antioxidant and photo-protectant role against external and internal stressors. Antioxidants 11 (8), 1451 (2022).
Liu, T., Luo, G. & Xing, M. Biomedical applications of polymeric microneedles for transdermal therapeutic delivery and diagnosis: current status and future perspectives. Adv. Ther. 3 (9), 1900140 (2020).
Phatale, V. et al. Overcoming skin barriers through advanced transdermal drug delivery approaches. J. Controlled Release. 351, 361–380 (2022).
Stefanov, S. R. & Andonova, V. Y. Lipid nanoparticulate drug delivery systems: recent advances in the treatment of skin disorders. Pharmaceuticals 14 (11), 1083 (2021).
Budzianowska, A. et al. Antioxidants to defend healthy and youthful skin—current trends and future directions in cosmetology. Appl. Sci. 15 (5), 2571 (2025).
Camillo, L. et al. Alpha-tocopherol protects human dermal fibroblasts by modulating nitric oxide release, mitochondrial function, redox status, and inflammation. Skin Pharmacol. Physiol. 35 (1), 1–12 (2022).
Song, M. et al. Preparation and evaluation of liposomes co-loaded with doxorubicin, phospholipase D inhibitor 5-fluoro-2-indolyl Deschlorohalopemide (FIPI) and D-alpha Tocopheryl acid succinate (α-TOS) for anti-metastasis. Nanoscale Res. Lett. 14 (1), 138 (2019).
Ali, H. M. et al. Improving stabilization of α-tocopherol and α-tocopheryl acetate against oxidation, light and UV radiation by complexation with β-cyclodextrin and starch. J. Food Sci. Technol. 62 (1), 75–87 (2025).
Hong, R., Han, Y. & Chen, S. Advances in micro-and nano-delivery systems for increasing the stability, bioavailability and bioactivity of coenzyme Q10. Crit. Rev. Food Sci. Nutr. 1–18 (2025).
Un Nabi, S. A. A. et al. Pharmaceutical gels: a review. RADS J. Pharm. Pharm. Sci. 4, 40–48 (2016).
Kumar, A., Singh, L. V. & Singh, S. Phospholipid-α-tocopherol complexation based self-nanoemulsifying system: assessment of phase behaviour, lipolysis stability & apoptotic activity. BioNanoScience 15 (2), 1–16 (2025).
Mura, P. et al. Evaluation and comparison of solid lipid nanoparticles (SLNs) and nanostructured lipid carriers (NLCs) as vectors to develop Hydrochlorothiazide effective and safe pediatric oral liquid formulations. Pharmaceutics 13 (4), 437 (2021).
Elmowafy, M. et al. Enhancement of bioavailability and pharmacodynamic effects of thymoquinone via nanostructured lipid carrier (NLC) formulation. Aaps Pharmscitech. 17 (3), 663–672 (2016).
Sheybani, F. et al. Application of nanostructured lipid carriers containing α-tocopherol for oxidative stability enhancement of camelina oil. Ind. Crops Prod. 202, 117007 (2023).
Kesharwani, R. et al. Formulation and evaluation of solid lipid nanoparticle (SLN) based topical gel of Etoricoxib. J. Appl. Pharm. Sci. 6 (10), 124–131 (2016).
Sreeharsha, N. et al. Formulation optimization of Chitosan surface coated solid lipid nanoparticles of griseofulvin: A Box-Behnken design and in vivo Pharmacokinetic study. Eur. J. Pharm. Sci. 204, 106951 (2025).
Ijaz, M. & Akhtar, N. Fatty acids based α-Tocopherol loaded nanostructured lipid carrier gel: in vitro and in vivo evaluation for moisturizing and anti‐aging effects. J. Cosmet. Dermatol. 19 (11), 3067–3076 (2020).
Dasgupta, N. et al. Fabrication of food grade vitamin E nanoemulsion by low energy approach, characterization and its application. Int. J. Food Prop. 19 (3), 700–708 (2016).
Kumar, R., Siril, P. F. & Soni, P. Optimized synthesis of HMX nanoparticles using antisolvent precipitation method. J. Energ. Mater. 33 (4), 277–287 (2015).
Varshosaz, J., Eskandari, S. & Tabbakhian, M. Freeze-drying of nanostructure lipid carriers by different carbohydrate polymers used as cryoprotectants. Carbohydr. Polym. 88 (4), 1157–1163 (2012).
Soleimanifard, M. et al. Spanish Olive leaf extract-loaded nanostructured lipid carriers: production and physicochemical characterization by Zetasizer, FT‐IR, DTA/TGA, FE‐SEM and XRD. J. Food Process. Preserv. 43 (7), e13994 (2019).
Majeed, I. et al. Formulation and in-vitro characterization of capsaicin loaded ethosomes. Pak. J. Pharm. Sci., 32. (2019).
Sohail, M. et al. An approach to enhanced stability: formulation and characterization of solanum lycopersicum derived lycopene based topical emulgel. Saudi Pharm. J. 26 (8), 1170–1177 (2018).
Baptista, S., Baptista, F. & Freitas, F. Development of emulsions containing L-ascorbic acid and α-tocopherol based on the polysaccharide fucopol: stability evaluation and rheological and texture assessment. Cosmetics 10 (2), 56 (2023).
Mansuri, R. et al. Potential of natural compounds as sunscreen agents. Pharmacogn Rev. 15 (29), 47 (2021).
Bhaskar, K. et al. Lipid nanoparticles for transdermal delivery of flurbiprofen: formulation, in vitro, ex vivo and in vivo studies. Lipids Health Dis. 8 (1), 6 (2009).
Ali, A. et al. Anti-pollution cosmetic-based one-step formation of w/o/w multiple emulsion containing D-biotin for skin protection: fabrication and in vitro and in vivo evaluation. Drug Delivery Translational Res. 9 (6), 1117–1132 (2019).
Khiljee, T. & Akhtar, N. Investigation of antiaging and skin rejuvenation potential of phytoconstituents from pyrus communis loaded topical emulgel. Pak. J. Pharm. Sci., 32. (2019).
Farboud, E. S., Nasrollahi, S. A. & Tabbakhi, Z. Novel formulation and evaluation of a Q10-loaded solid lipid nanoparticle cream: in vitro and in vivo studies. Int. J. Nanomed. 611–617 (2011).
Czajkowska-Kośnik, A., Szymańska, E. & Winnicka, K. Nanostructured lipid carriers (NLC)-based gel formulations as Etodolac delivery: from gel Preparation to permeation study. Molecules 28 (1), 235 (2022).
Green, P. G., Guy, R. H. & Hadgraft, J. In vitro and in vivo enhancement of skin permeation with oleic and lauric acids. Int. J. Pharm. 48 (1–3), 103–111 (1988).
Rustan, A. C. & Drevon, C. A. Fatty acids: structures and properties. Encyclopedia Life Sci. 1, 1–7 (2005).
Kasongo, K. W., Müller, R. H. & Walker, R. B. The use of hot and cold high pressure homogenization to enhance the loading capacity and encapsulation efficiency of nanostructured lipid carriers for the hydrophilic antiretroviral drug, didanosine for potential administration to paediatric patients. Pharm. Dev. Technol. 17 (3), 353–362 (2012).
PAWBAKE, G. R. & SHIROLKAR, S. V. Formulation and evaluation of diacerein microemulgel topical drug delivery. Int. J. Pharm. Res. (09752366), 12(4). (2020).
Jannin, V., Musakhanian, J. & Marchaud, D. Approaches for the development of solid and semi-solid lipid-based formulations. Adv. Drug Deliv. Rev. 60 (6), 734–746 (2008).
Basiri, L., Rajabzadeh, G. & Bostan, A. α-Tocopherol-loaded niosome prepared by heating method and its release behavior. Food Chem. 221, 620–628 (2017).
Abla, M. & Banga, A. Formulation of Tocopherol nanocarriers and in vitro delivery into human skin. Int. J. Cosmet. Sci. 36 (3), 239–246 (2014).
Baumann, L. S. & Md, J. S. The effects of topical vitamin E on the cosmetic appearance of scars. Dermatol. Surg. 25 (4), 311–315 (1999).
Liu, J. et al. Solid lipid nanoparticles loaded with insulin by sodium cholate-phosphatidylcholine-based mixed micelles: Preparation and characterization. Int. J. Pharm. 340 (1–2), 153–162 (2007).
Thang, L., Hanh, N. & Duong, D. Study on cause-effect relations and optimization of exemestane-loaded nanostructured lipid carriers. Int. J. Pharm. Pharm. Sci. 9 (5), 68–74 (2017).
Gonzalez-Mira, E. et al. Design and ocular tolerance of flurbiprofen loaded ultrasound-engineered NLC. Colloids Surf., B. 81 (2), 412–421 (2010).
Danaei, M. et al. Impact of particle size and polydispersity index on the clinical applications of lipidic nanocarrier systems. Pharmaceutics 10 (2), 57 (2018).
Kim, J. Y., Song, M. G. & Kim, J. D. Zeta potential of nanobubbles generated by ultrasonication in aqueous alkyl polyglycoside solutions. J. Colloid Interface Sci. 223 (2), 285–291 (2000).
Madania, M. N. et al. The effect of surfactant on the characteristics of Curcumin-Loaded nanostructured lipid carriers: fluorescence and stability study. J. Pure Appl. Chem. Res. 14 (1), 1–12 (2025).
Shah, R. et al. Optimisation and stability assessment of solid lipid nanoparticles using particle size and zeta potential. J. Phys. Sci., 25(1). (2014).
Celebioglu, A. & Uyar, T. Electrospinning of polymer-free nanofibers from cyclodextrin inclusion complexes. Langmuir 27 (10), 6218–6226 (2011).
Pathak, P. & Nagarsenker, M. Formulation and evaluation of Lidocaine lipid nanosystems for dermal delivery. Aaps Pharmscitech. 10 (3), 985–992 (2009).
Das, S., Ng, W. K. & Tan, R. B. Are nanostructured lipid carriers (NLCs) better than solid lipid nanoparticles (SLNs): development, characterizations and comparative evaluations of clotrimazole-loaded SLNs and nlcs? Eur. J. Pharm. Sci. 47 (1), 139–151 (2012).
Nnamani, P. O. et al. Development of artemether-loaded nanostructured lipid carrier (NLC) formulation for topical application. Int. J. Pharm. 477 (1–2), 208–217 (2014).
Teeranachaideekul, V. et al. Cetyl palmitate-based NLC for topical delivery of coenzyme Q10–Development, physicochemical characterization and in vitro release studies. Eur. J. Pharm. Biopharm. 67 (1), 141–148 (2007).
Mohamed, M. A. et al. Fourier Transform Infrared (FTIR) spectroscopy, in Membrane Characterization p. 3–29 (Elsevier, 2017).
Nasiri, F., Faghfouri, L. & Hamidi, M. Preparation, optimization, and in-vitro characterization of α-tocopherol-loaded solid lipid nanoparticles (SLNs). Drug Dev. Ind. Pharm. 46 (1), 159–171 (2020).
Saric, S., Notay, M. & Sivamani, R. K. Green tea and other tea polyphenols: effects on Sebum production and acne vulgaris. Antioxidants 6 (1), 2 (2016).
Fang, Y. Z., Yang, S. & Wu, G. Free radicals, antioxidants, and nutrition. Nutrition 18 (10), 872–879 (2002).
Natarajan, J. V. et al. Sustained-release from nanocarriers: a review. J. Controlled Release. 193, 122–138 (2014).
Sarfraz, M. et al. Fabrication, organoleptic evaluation and in vitro characterization of cream loaded with carica Papaya seed extract. J. Cosmet. Dermatol. 23 (3), 1045–1054 (2024).
Akyar, I. Standard operating procedures (what are they good for?). Latest Res. Into Qual. Control. 12, 367–391 (2012).
Kulawik-Pióro, A., Kurpiewska, J. & Kułaszka, A. Rheological and sensory properties of hydrophilic skin protection gels based on polyacrylates. Int. J. Occup. Saf. Ergon. 24 (1), 129–134 (2018).
Pays, K. et al. Double emulsions: how does release occur? J. Controlled Release. 79 (1–3), 193–205 (2002).
Mandawgade, S. D. & Patravale, V. B. Development of SLNs from natural lipids: application to topical delivery of tretinoin. Int. J. Pharm. 363 (1–2), 132–138 (2008).
Jain, S., Mistry, M. A. & Swarnakar, N. K. Enhanced dermal delivery of acyclovir using solid lipid nanoparticles. Drug Deliv. Transl. Res. 1 (5), 395–406 (2011).
Sahu, K. L. et al. Advancements in Polycaprolactone nanoparticles for targeted drug delivery in breast cancer treatment: strategies and challenges. Next Nanatechnol. 8, P100206 (2025).
Nikolić, S. et al. Skin photoprotection improvement: synergistic interaction between lipid nanoparticles and organic UV filters. Int. J. Pharm. 414 (1–2), 276–284 (2011).
Farage, M. A. et al. Historical perspective on the use of visual grading scales in evaluating skin irritation and sensitization. Contact Dermat. 65 (2), 65–75 (2011).
Ma, W. et al. Chronological ageing and Photoageing of the fibroblasts and the dermal connective tissue. Clin. Exp. Dermatol. 26 (7), 592–599 (2001).
Augustyniak, A. et al. Natural and synthetic antioxidants: an updated overview. Free Radic. Res. 44 (10), 1216–1262 (2010).
KAUSAR, R. Development, characterization and non-invasive invivo evaluation of cosmetic emulgels loaded with strawberry, blackberry and raspberry fruit extracts. (PhD Thesis) (2017).
Hung, L. C. et al. An improved method for the preparations of nanostructured lipid carriers containing heat-sensitive bioactives. Colloids Surf., B. 87 (1), 180–186 (2011).
Leite et al. Hydrating effects of moisturizer active compounds incorporated into hydrogels: in vivo assessment and comparison between devices. J. Cosmet. Dermatol. 8 (1), 32–39 (2009).
Esposito, E. et al. Nanosystems for skin hydration: a comparative study. Int. J. Cosmet. Sci. 29 (1), 39–47 (2007).
Chen, W., Zouboulis, C. C. & Orfanos, C. The 5α-Recluctase system and its inhibitors: recent development and its perspective in treating Androgen-Dependent skin disorders. Dermatology 193 (3), 177–184 (1996).
Dobrev, H. Clinical and instrumental study of the efficacy of a new Sebum control cream. J. Cosmet. Dermatol. 6 (2), 113–118 (2007).
Acknowledgements
The authors extend their appreciation to Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2025R101), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia. We are also very thankful to all the reviewers of this manuscript. Their thoughtful feedback has not only improved this manuscript but has also provided us with valuable insights for our future research.
Funding
The research was funded by Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2025R101), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Musarrat Ijaz: Project management, Writing original draft; acquisition of data, figures preparation, data analysis, editing and rewriting, Asadullah Madni: Supervision and Conceptualization, Haji Muhammad Shoaib Khan: Revised the manuscript critically for important intellectual content, Tahreem Arshad: editing & rewriting, Proofreading and revising the manuscript critically for important intellectual content, Sidra Meer: Validation and software, Farhat Ijaz: Revising the manuscript and software, Naveed Akhtar: Supervision and Revising the manuscript critically for important intellectual content. Modhi O. Alotaibi : Data analysis and Funding, Areej Turkistani: Editing& rewriting, Gaber El-Saber Batiha: Funding acquisition, Validation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics declaration
All the experiments involving animal and human were carried out according to the guidelines and regulations approved by institutional research and ethics committee of The Islamia University of Bahawalpur under the reference # 94/S.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ijaz, M., Madni, A., Khan, H.M.S. et al. Nanostructured Lipid Carrier Based Gel Containing α-Tocopherol and α-Tocopheryl Acetate for Synergistic Cutaneous Antiaging Efficacy. Sci Rep 15, 42360 (2025). https://doi.org/10.1038/s41598-025-26277-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-26277-6