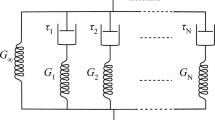
Abstract The female perineum plays a critical role in several physiological processes. During childbirth, this region undergoes significant stretching, resulting in perineal trauma in over 90% of vaginal deliveries. Despite advances in obstetric care, predicting and preventing such injuries remains challenging. Computational models offer a non-invasive approach to investigate female pelvic biomechanics. However, their reliability depends on a deeper understanding of perineal mechanics, an area that remains understudied. This study addresses this gap by characterizing the mechanical behavior of the human female perineal body. Perineal samples were obtained post-mortem from six women and subjected to stress-relaxation tests and ultimate tensile tests up to failure. A genetic algorithm and finite element simulations calibrated the material parameters of a visco-hyperelastic model to fit the experimental data. Stress-relaxation tests demonstrated a 40% stress reduction after 900 seconds, highlighting the tissue’s time-dependent behavior. The ultimate Cauchy stress was 224.70 ± 131.69 kPa at a stretch of 1.97. Histology revealed preferential fiber alignment with the direction of physiological stretching. Three ranges of material parameters were calibrated, enabling the data to be used in future computational simulations. The findings provide valuable insights into the mechanics of the female perineum, facilitating more accurate simulations. This study contributes to ongoing efforts to enhance the understanding, prevention, and management of pelvic floor dysfunctions. Graphic Abstract
Similar content being viewed by others
Introduction
The perineal region forms a diamond-shaped area bound by the pubic symphysis, the tip of the coccyx, and both ischial tuberosities. Within this region lies the perineum, located centrally between the commissure of the labia and the anus. The main anatomical structure in the perineum forms the perineal body, a structure composed of dense connective tissue and interspersed with muscle fibers. It serves as an attachment point for several key muscles, including the pelvic floor muscles (PFM), the external anal sphincter, the deep and superficial transverse perineal muscles, and the bulbospongiosus muscles1. As a region that undergoes significant stretching during childbirth, studies indicate that the incidence of perineal trauma may exceed 90% in females who deliver vaginally2. Third- or fourth-degree perineal tears, classified as obstetric anal sphincter injuries (OASIS), affect a substantial proportion of postpartum women, with an incidence ranging from 1% to 11%3. Perineal trauma is associated with considerable postnatal morbidity, including pelvic organ prolapse and urinary incontinence, and patients are at increased risk of recurrent severe perineal lacerations in subsequent deliveries4.
Several factors contribute to the risk of perineal trauma during childbirth. While certain aspects, such as ethnicity, nulliparity, and maternal age, are unchangeable, other factors must be considered: these include fetal macrosomia, the use of epidural anesthesia, prolonged second stage of labor, instrumental delivery, and the practice of episiotomy5. Even a modest reduction in perineal trauma would positively impact a significant number of women, leading to lower childbirth costs and reduced demand for extended medical care6. Despite extensive discussions in the medical field, childbirth-related trauma remains difficult to predict to date. Identifying specific injuries often requires costly post-delivery examinations that are not typically part of the standard care process. As such, healthcare providers could benefit from non-invasive quantitative methods to help identify potential injury sites or predict the types of injuries that may occur. Early detection and monitoring could facilitate timely interventions, thereby reducing the risk or severity of pelvic floor disorders, which often manifest years or even decades after delivery and significantly impact women’s quality of life.
Computational models provide a quantitative approach to analyze the physiology of labor, overcoming the limitations of physical models and invasive procedures7. Finite element simulations are often used to replicate the biomechanical behavior of the distinct structures involved in childbirth, providing a deeper understanding of the mechanisms of vaginal delivery and the associated risks of injury8. A critical aspect of these models is the mechanical characterization of the soft tissues involved, which often exhibit complex, non-linear behavior. Nevertheless, owing the complexity of the mathematical models required to accurately capture this behavior, or the lack of experimental data, researchers often rely on simplified assumptions, such as focusing only on linear elastic or isotropic hyperelastic properties. While these simplifications facilitate modeling, they severely limit the accuracy of computational models. This limitation can be mitigated by performing mechanical experiments to obtain precise, tissue-specific properties that allow for more realistic simulations.
However, even when experimental tests can be conducted to characterize tissues, ethical and logistical challenges arise, particularly when dealing with soft biological tissues in sensitive contexts as childbirth. Direct experimentation on the human perineum during pregnancy is neither feasible nor ethical due to the significant risks it poses to both mother and fetus. This leaves post-mortem examination as one of the few viable options, although it has its own limitations, such as potential tissue degradation, changes in mechanical properties, or limited sample availability. Contemporary research also involves the use of animal models to mimic the mechanical behavior of human pelvic floor components9,10. While these models offer valuable insights, they cannot fully replicate the complexity of human anatomy and physiology, potentially leading to discrepancies in tissue characterization.
Several studies have attempted to non-invasively estimate the mechanical properties of the perineal body, primarily using elastography techniques. For instance, Chen et al. (2015)11 measured the mechanical properties of the perineal body in 20 nulliparous women, estimating its elastic modulus. Similarly, Li et al. (2024)12 quantified the perineal body elasticity using transperineal shear wave elastography. However, these methods only capture the elastic properties of the tissue, which does not fully encompass its complex mechanical behavior. As previously mentioned, animal models are commonly used in research due to their anatomical similarities to the human female pelvic region, with ewe and porcine tissues often selected. Kochova et al. (2019)13 investigated the microstructure and elasticity of the perineal body by conducting uniaxial tension tests on pregnant and postmenopausal ewes. More recently, Lallemant et al. (2024)14 performed uniaxial tensile tests on sows’ perineal body tissue to gain further insights into the mechanical properties contributing to perineal tears during childbirth. Furthermore, Kochova et al. (2019)15 conducted the only study, to our best knowledge, which performed tensile tests to determine the load-deformation properties of human female post-mortem perineal tissues, alongside histological assessment. Despite these contributions, all of these studies lack a detailed analysis of the hyperelastic and viscoelastic properties of the perineum, critical factors that significantly influence its mechanical behavior.
Therefore, the primary goal of this study was to thoroughly characterize the hyperelastic and viscoelastic mechanical behavior of the human female perineum, specifically focusing on the perineal body. This was achieved through uniaxial tensile tests and stress-relaxation tests on post-mortem tissue. The experimental data were then used to calibrate the material parameters of a visco-hyperelastic constitutive model, providing a detailed representation of the tissue’s mechanical properties. The data obtained contribute to a deeper understanding of the biomechanical behavior of the perineal region and enable more accurate mechanical characterization of these tissues in computational models. Ultimately, this study provides a valuable reference for the mechanical properties of pelvic tissues, supporting simulations of aging-related dysfunctions and pelvic floor disorders, and offering insight into factors that contribute to tissue vulnerability in elderly women.
In summary, the main contributions of this paper are:
-
Development of a experimental protocol tailored to perineal tissue, addressing the challenges of its softness and small dimensions, while providing standardized procedures for collection, storage, and preparation that may also be adapted to other delicate soft tissues.
-
A deeper understanding of the mechanical behavior of perineal tissue. This includes an analysis of its deformation characteristics, stress-strain relationship, relaxation behavior over time, and its maximum load-bearing capacity before failure.
-
Mechanical characterization of the perineum using a visco-hyperelastic anisotropic constitutive model. The determined material parameters can be used for finite element simulations to accurately model the mechanical behavior of the perineal body.
-
Establish a reference dataset of the mechanical properties of the human female perineum. This dataset can be used for future studies and computational modeling, particularly for applications such as understanding postpartum dysfunction, as pelvic organ prolapse, and developing interventions for pelvic tissue disorders.
Results
The data acquired during the relaxation and ultimate tensile tests is available at Zenodo repository16. The dataset includes the samples that were considered in the results presented in this study.
Relaxation tests
Of the seventeen samples tested, thirteen successfully completed the protocol. Specimens were excluded if they ruptured during the first 75% loading cycle or exhibited insufficient relaxation during the relaxation period. The latter were excluded primarily because they consisted mostly of fat tissue. Since the majority of specimens failed during the first loading cycle at 150% strain, relaxation data was collected at the 75% strain level.
The stress response during these loading and unloading cycles is illustrated in Fig. 1. The stress decreased from the first peak to the final one by 78.3%, highlighting the material’s response to cyclic loading.
Representative cyclic loading and unloading response of the tissue prior to the relaxation tests, illustrating the evolution of Cauchy stress across repeated cycles. The pink line represents a spline interpolation fitted to the peak stress values of each cycle.
Following the seventh loading cycle, the displacement was held constant at 75% strain for 900 seconds. The Cauchy stress-time curve for the experiments, along with its averaged response, is presented in Fig. 2a. Additionally, the results are presented in terms of normalized stress (Fig. 2b), calculated as the ratio of the current stress to the peak stress. By the end of the 900-second period, the material demonstrated an average stress-relaxation of 39.76%, highlighting its time-dependent behavior.
Average response of the relaxation tests: (a) Cauchy Stress (kPa) and (b) Normalized Stress (current stress/peak stress) of all the experiments (dashed lines).
Ultimate tensile tests
For ultimate the tensile tests, the displacement of the actuators was used to calculate stretch and stress, as the markers measured with the video extensometer deformed strongly resulting in insufficient quality for reliable analysis. Of the seven samples tested, five successfully completed the protocol, while the other two slipped during testing, which made the data collected nonviable. From the force-displacement data, the ultimate tensile displacement was determined to be 11.43 mm, while the ultimate tensile force reached \(0.291 \pm 0.182~\)N. The results are represented in terms of Cauchy stress (calculated in kPa) as a function of stretch, as shown in Fig. 3. The maximum Cauchy stress for the average curve was determined to be \(224.70 \pm 131.69\) kPa, corresponding to a stretch of 1.966.
Average Cauchy stress-stretch curve from the ultimate tensile tests. Averaged response: mean ± SD (standard deviation).
Histologic assessment
Histological analysis was performed on all specimens included in the stress-relaxation and ultimate tensile test results. Figure 4a presents the original histological section of the sirius red staining, while 4b presents the color-coded visualization after orientation analysis based on sirius red staining of the perineal specimens. Fiber orientation was analyzed employing Fiji17 with the ImageJ plugins Colour Deconvolution 218,19 and OrientationJ20. Blue and green tones indicate fibers oriented in the horizontal direction, and red tones represent fibers oriented in the vertical direction.
(a) Representative histological section stained with Sirius Red. (b) Post-processed histological section of perineal specimens at 20x magnification. Blue and green tones represent fibers oriented in the horizontal direction, while red tones represent fibers oriented in the vertical direction. (c) Polar histogram of the collagen fibers. Radial distance represents fiber density, while the angle, in degrees, indicates fiber orientation. Shaded bars show the distribution of fiber densities across directions.
The histological assessment enabled the analysis of fiber density and orientation in the examined tissues. The preferential fiber direction is illustrated in Fig. 4c, with the majority of fibers predominantly aligned horizontally, corresponding to the direction of tissue stretching. The mean orientation of the fibers is approximately 19 degrees. In the qualitative evaluation of muscle content assumed in vivo, most specimens exhibited medium fiber content, while two showed high fiber content, and one displayed low fiber content.
Calibration of constitutive parameters
The micro-genetic algorithm developed by Vila Pouca et al. (2020)21 was used to calibrate the constitutive parameters of the Holzapfel-Gasser-Ogden (HGO) constiutive model, which was coupled with the generalized Maxwell model to account for viscous effects and capture the visco-hyperelastic behavior. Figure 5 presents the calibrated constitutive model with the experimental data. Due to the variability observed in both the hyperelastic and viscoelastic responses, parameter calibration was performed for three cases: minimum, mean and maximum stress relaxation, as well as the corresponding stresses from the tensile tests. This approach enabled the derivation of three sets of parameters, each representing distinct types of tissue behavior. The calibrated parameters are presented in Table 1.
Calibration of the constitutive model parameters using experimental data. (a) Fitting to the minimum, mean, and maximum stress-relaxation test curves. (b) Fitting of the minimum, mean and ultimate tensile test curves.
The mean squared error for the viscoelastic response is \(1.31 \times 10^{-5}\), \(1.13 \times 10^{-4}\) and \(5.94 \times 10^{-5}\) for cases 1, 2 and 3, respectively. For the hyperelastic calibration, the errors obtained were 1.66 \(\times 10^{-5}\) \(\hbox {MPa}^2\), 8.02 \(\times 10^{-6}\) \(\hbox {MPa}^2\) and 1.08 \(\times 10^{-6}\) \(\hbox {MPa}^2\) for the same cases.
Discussion
This study presents an experimental approach to investigate the hyperelastic and viscoelastic properties of the female perineum, a critical structure within the pelvic cavity that plays a fundamental role in various physiological processes. Accurate mechanical characterization of the perineum can be essential for developing reliable biomechanical models that simulate the mechanical behavior of the pelvic cavity, providing insights into tissue vulnerability and contributing to the prediction, prevention, and management of pelvic organ prolapse and other age-related pelvic floor dysfunctions. The present study conducted uniaxial tensile and stress-relaxation tests on post-mortem perineal tissue samples to explore the tissue’s mechanical behavior. The findings presented here aim to enhance the understanding of perineal biomechanics, providing valuable insights that could help unveil the mechanisms underlying pelvic floor disorders, supporting the development of preventive strategies and therapeutic interventions to promote long-term maternal health. The raw mechanical characterization data is available to interested researchers at Zenodo. Given the specificity of the data, assistance can be provided for its interpretation and usage, along with the necessary documentation. This will ensure that others can accurately utilize the dataset for further analysis and development in the field.
The relaxation test results indicate that the perineal body exhibits viscoelastic behavior, as evidenced by the high stress relaxation when a constant displacement is maintained for 900 seconds. Specifically, the stress decreased by approximately 40% during this period, as illustrated in Fig. 2. Viscoelasticity is a critical property of biological tissues, particularly in the female pelvis, as it allows tissues to accommodate larger deformations with reduced risk of damage22. This characteristic is especially relevant during childbirth, where the relaxation of pelvic tissues facilitates progressive dilation as labor advances23. However, to the best of our knowledge, no stress-relaxation curves for the perineal body are currently available in the literature for direct comparison of its viscoelastic behavior.
The ultimate tensile tests revealed that the specimens exhibited non-linear hyperelastic behavior, as demonstrated by the stress-stretch curve (Fig. 3). The specimens demonstrated high deformability, with a non-linear relationship between force and displacement. Limited data exist in the literature regarding the mechanical failure properties of the perineum. Kochova et al. (2019)13 reported ultimate strain and engineering stress values of 1.4 and 171.5 kPa, respectively, for the postmenopausal human female perineal body. In contrast, the specimens tested in the current study exhibited a lower ultimate strain of 0.95 before failure, while the ultimate tensile engineering stress, approximately 120 kPa, was within a comparable range. These discrepancies may arise from differences in sample dimensions, as the present study utilizes specimens with smaller width and thickness. Additionally, the experimental setup differs, as our study uses a phosphate-buffered saline (PBS) bath during testing to prevent the sample from drying throughout the process. Another possible explanation is that, despite both studies involving elderly women, the mean age in our study is higher.
The mechanical properties obtained from the tensile tests in this study can also be compared to those reported in animal models, which are often used as surrogates for human tissues in biomechanical research. For instance, Lallemant et al. (2024)14 performed uniaxial tensile tests on porcine perineal tissues, identifying the vagina and anal mucosa as the stiffest tissues, while the internal and external anal sphincters were more extensible and less stiff. The stress values reported for latter structures fall within a similar range to those observed in our human tissue specimens. This suggests that certain porcine tissues may serve as reasonable analogs for studying the mechanical behavior of human perineal structures, although differences in anatomy and tissue composition must be considered. Additionally, Kochova et al. (2019)15 conducted tensile tests on the perineal body of postmenopausal ewes, reporting stress values that are comparable to those obtained in our study. However, the ewe tissue exhibited slightly higher rigidity and sustained greater strain before failure. These comparisons highlight the potential utility of animal models for preliminary investigations into perineal biomechanics, while also underscoring the importance of human tissue data for accurate characterization. Despite the similarities, the observed differences in mechanical behavior between human and animal tissues emphasize the need for caution when extrapolating findings from animal studies to human clinical applications.
The histological analysis of the perineal specimens provided valuable insights into the structural composition and fiber orientation of the tissue, which are critical factors influencing its mechanical behavior. The Sirius Red collagen staining revealed a dense network of collagen fibers, predominantly aligned along the direction of tissue stretching, as illustrated in the polar histogram (Fig. 4c). This preferential fiber orientation aligns with the anatomical function of the perineal body, which undergoes significant stretching during childbirth. The alignment of fibers in the direction of loading likely contributes to the tissue’s ability to withstand high stress, as observed in the ultimate tensile tests. However, the high variability in fiber density, observed directly from the measurements, and in fiber orientation across specimens may also explain the differences in mechanical responses, such as the stress relaxation behavior observed in the relaxation tests. The presence of well-organized collagen fibers supports the tissue’s viscoelastic properties, allowing it to reduce stress over time, which is crucial for accommodating the large deformations experienced during childbirth. These histological findings complement the mechanical data, providing a more comprehensive understanding of the perineal body’s biomechanical behavior. In future work, the constitutive model could also be extended to account for collagen fiber dispersion using the HGO formulation. Incorporating fiber orientation heterogeneity could allow a more physiologically realistic representation of the tissue’s anisotropic mechanical behavior and improve the predictive accuracy of the simulations.
The calibration of material properties demonstrated the feasibility of fitting numerical simulations to experimental data to achieve accurate mechanical characterization. In this study, the minimum, mean, and maximum tissue responses were calibrated based on the variability in the mechanical behavior of the same tissue across different women, with these responses representing the lowest, average, and highest experimental curves, respectively. This variability can be primarily attributed to inter-individual differences among donors, including age-related physiological changes and variations in tissue composition. All specimens were obtained from elderly women, a population in which pelvic tissue quality is affected by factors such as reduced collagen cross-linking, altered elastin content, and progressive remodeling of the extracellular matrix. In addition, the absence of detailed obstetric or clinical histories further contributes to the heterogeneity observed across samples. These donor-related factors influence the microstructure and viscoelastic behavior of pelvic tissues, introducing variability that reflects the natural physiological diversity of the studied population rather than methodological inconsistencies. This underscores the critical need for patient-specific models to account for anatomical and mechanical differences unique to each individual. In the calibration process of the relaxation tests, a small deviation from the experimental curves is observed in the numerical results, with a faster initial decay followed by an approach to an equilibrium value slightly lower than the experimental data. The inclusion of an additional time constant within the Maxwell model framework was considered to address this discrepancy. However, the overall trend remained unchanged. Despite this, the numerical curves show very good agreement with the experiments, and the associated errors are minimal. Additionally, the relaxation curves exhibited slight variations across stress levels, suggesting a transition from a predominantly single-mode to a multi-mode relaxation behavior at higher stresses. This observation indicates that the tissue displays stress-dependent viscoelastic effects, which could be more accurately represented by models incorporating variable or non-linear relaxation spectra24. The values reported in Table 1 correspond to the optimal parameters obtained from the calibration of the constitutive model through a genetic algorithm for each specimen. The genetic algorithm provides point estimates rather than statistical confidence intervals. While parameter variability could in principle be assessed via repeated calibration runs or sensitivity analysis, this was beyond the scope of the present study. Therefore, the reported values represent single optimal estimates. Nevertheless, significant advancements in experimental mechanics are still required to effectively capture the hyperelastic and viscoelastic behavior of tissues in vivo. Addressing this challenge is essential for improving the fidelity of models used and potentially implementing them in clinical applications.
The primary limitation of this study is that all specimens were obtained from elderly female donors, whose perineal tissue may exhibit mechanical properties substantially different from those of younger, nulliparous, or pregnant women. Age-related changes, including reduced collagen content, altered elastin structure, and hormonal variations, are known to influence the mechanical behavior of soft tissues. Consequently, caution is warranted when extrapolating these results to younger populations. Previous research has explored the impact of aging on the biomechanical properties of soft tissues, particularly pelvic tissues. For instance, Chantereau et al. (2014)25 demonstrated that aging and potential trauma contribute to changes in the mechanical behavior of pelvic floor tissues, resulting in increased stiffness in most of the tissues tested. These findings are crucial as they could facilitate the development of mathematical models that relate the mechanical properties of younger and older tissues26. Such models would allow researchers to extrapolate the characteristics of younger tissues based on the more readily available samples from elderly individuals.
Additional limitations must be acknowledged in this study. First, the small sample size (eighteen samples from six individuals) limits its generalizability and the ability to account for inter-individual variability in tissue properties, which may have led to biased or incomplete conclusions. Second, the perineal tissue samples were frozen prior to analysis, a common preservation method that may alter the mechanical and structural properties of biological tissues. Nonetheless, literature suggests that this influence is minimal27,28. Regarding the freezing process, the tissues were stored frozen for a relatively long period, which may have slightly influenced their mechanical properties and contributed to some variability between samples. In addition, the occurrence of one or more freeze–thaw cycles could also have had an effect on tissue integrity and mechanical behavior29. Third, although the perineal body contains muscle fibers, only the passive properties of the tissue were analyzed. However, this scenario can be compared to cases where women receive epidurals during labor, which lead to muscle relaxation. Furthermore, the mechanical properties were tested in only one direction. While this aligns with the primary anatomical stretching direction of the tissue, it may limit the acquisition of critical information regarding tissue heterogeneity. Future work should focus on testing the tissue in the transverse direction and also under biaxial loading, allowing for a more comprehensive validation of the anisotropic response of the tissue. Also, the use of a standardized geometry in the finite element simulations for parameter calibration may be considered a limitation, as small geometric variations exist between specimens, and the model does not fully capture the unique geometry of each individual specimen. The absence of histological assessment throughout the experimental protocol also represents a limitation. Since microstructural analysis was not performed at different stages, particularly after preconditioning, it is not possible to determine with certainty whether any microstructural damage occurred in the tissue. Moreover, all experiments in this study were performed at a single deformation rate. Therefore, rate-dependent viscoelastic effects, which may influence tissue stiffness at different loading rates, were not assessed. Nonetheless, previous studies have demonstrated that tissue stiffness can vary with loading rate due to rate-dependent viscoelastic behavior30. Lastly, the marker data could not be incorporated into the ultimate tensile test analysis, which may have led to less precise measurements. As the samples demonstrated stress concentration near the grips, causing damage to the areas near the clamps, it would have been more accurate to measure the stress data at the midsection of the samples.
Despite these limitations, this study presents a pipeline for acquiring experimental data from post-mortem tissues and integrating it into a computational model for biomechanical simulations. Our findings demonstrate that the perineal body exhibits a highly non-linear, time-dependent viscoelastic response when subjected to mechanical stress. By fitting experimental data to a constitutive model using Abaqus® finite element software, we successfully characterized the tissue’s mechanical behavior. Incorporating realistic tissue characterization into computational models can help identify regions of the pelvic floor that are most susceptible to stress or deformation, particularly in elderly women who are at increased risk of pelvic organ prolapse and other pelvic floor dysfunctions. This predictive capability could inform the development of targeted interventions, such as individualized pelvic floor rehabilitation programs, supportive devices, or preventive strategies to reduce the progression of prolapse and associated complications. Furthermore, integrating these models into clinical training and decision-making could improve healthcare providers’ understanding of the biomechanical factors contributing to pelvic floor dysfunction, ultimately supporting better prevention, management, and care for aging women.
Methods
The protocol begins with sample collection and storage, followed by specimen preparation for experimental testing, including uniaxial tensile and relaxation tests. The final step involves calibrating the material parameters.
Sample collection and storage
Perineal samples were obtained from six females who while alive had provided written informed consent to participate in a post-mortem body donation program. The study was reviewed and approved by the Ethics Committee of the Medical University of Graz (36–197 ex 23/24) through an expedited review process. All experiments were performed in accordance with relevant guidelines and regulations. Tissues were obtained from females aged 72 to 101 years at the time of death (87 ± 9.8, mean ± standard deviation). The following clinical data is known from the donors: height (162.9 ± 2.7 cm), weight (70.7 ± 15.6 kg), body mass index (26.6 ± 5.4 kg/\(\hbox {m}^2\)).
The samples were collected post-mortem, pre-cooled at \(4^{\circ }\)C for 1 hour and stored frozen at \(-80^{\circ }\)C for 7–120 days. During sample collection, first skin and subcutaneous tissue were removed, then surrounding structures were identified for guidance, and the perineal body was dissected up to a depth of 20 mm. The region of interest in anterior-posterior orientation was defined as between the inner vaginal wall and the inner rectal wall. Laterally the region of interest was extended to approximately 30 mm per side to avoid damage of the perineal body during dissection. The orientation of the perineal body was marked with surgical suture.
To ensure uniform sample preparation for mechanical testing, the perineal samples were thawed at room temperature for only as long as necessary to enable cutting. The region of interest, the perineal body, was then carefully dissected. The areas closest to the vaginal wall and the anal sphincter were identified, and all samples were refrozen at \(-80^{\circ }\)C in such a way that the sphincter region was flat and facing downward. This ensured that all specimens would be cut in the same direction. During the process, the samples were kept constantly hydrated with PBS.
Specimen preparation
To maximize the number of specimens retrievable from each sample and ensure uniform sample thickness, a Leica CM1950 Cryostat was used to cut each frozen sample into 0.6 mm sections, the maximum value within the trimming thickness range of this instrument. The specimens were embedded in optimal cutting temperature (OCT) compound to provide structural support and stability and to prevent damage. The sections were kept hydrated with PBS before and immediately after cutting. Each section was placed on a glass microscope slide, numbered sequentially from 1 to the total number of sections, starting from the sphincter side, and then returned to the freezer at \(-80^{\circ }\)C until it was time for specimen preparation for testing. This process is demonstrated in Fig. 6a.
On the day of testing, each specimen was thawed, a process that only took a few minutes due to their small thickness. The specimens were then cut into a dog-bone shape using a customized dog-bone punch created for this purpose. As each sample measured approximately 25 to 30 mm per side, the final dimensions of each specimen were 22 mm in total length, 12 mm in gauge length, 3 mm in width, and 0.6 mm in thickness, as shown in Fig. 6b. The samples were hydrated with PBS throughout the preparation phase.
Following the shaping process, each specimen was placed on a preparation table. A vinyl polysiloxane dental impression material was then applied to both the top and bottom surfaces of the specimen. Once the material solidified, it was carefully cut to register the cross-sectional area, which was subsequently scanned for further analysis. This methodology was implemented in accordance with the same principles of standardized soft tissue testing described by Scholze et al. (2020)31.
To prevent slippage during testing, custom-designed 3D-printed clamps were employed, following the approach outlined by Scholze31. The testing setup included a preparation table, clamps featuring flat pyramid structures optimized for securing thin soft tissue samples, and holder arms designed to attach the clamps and specimen securely to the testing machine, as demonstrated in Fig. 6b. The clamps and preparation table were fabricated using polylactic acid filament, while the holder arms were made from thermoplastic polyurethane for increased flexibility.
Before placing the specimen in the testing machine, a set of black markers was drawn in the middle of each specimen using tissue marker dye. These markers were essential for using a video extensometer to obtain precise strain measurements. By measuring strain directly on the specimen’s surface with the integrated digital extensometer of the Zwick/Roell biaxial testing machine, this technique eliminates potential errors caused by machine deformation or material slippage, ensuring more accurate data collection.
Experimental protocol
The study was conducted in two phases. The first phase involved performing relaxation tests, in which each specimen was stretched up to 75% and 150% of its gauge length to evaluate the viscoelastic mechanical properties of the perineal body. The selection of these stretch values was based on preliminary tests with a limited number of samples while considering realistic stretch values during birth32. In the second phase, ultimate tensile tests were carried out to determine the failure mechanical properties of the tissue.
After completing all sample preparation steps, seventeen specimens were allocated for stress-relaxation tests and seven for ultimate tensile tests. For the former, two to three specimens were tested per donor, while for the latter, at least one specimen was tested per donor. All tests were conducted using a Zwick/Roell biaxial tester machine equipped with four 100 N load cells, each with a resolution of 0.002 N. The machine was operated in uniaxial mode, calibrated, and controlled using the testXpert III software. The complete set up of the testing machine is presented in Fig. 6c.
Demonstration of the experimental protocol, from initial sample preparation to final testing. (a) Sample preparation steps: the initial perineal sample (1) was cut and inserted into the cryostat machine (2) to produce sections that were placed on microscope slides (3). (b) Representation of the perineal specimen dimensions and its integration into the testing system, including the 3D-printed clamps, holders arms, and the specimen with black markers. (c) Specimen positioned in the 3D-printed clamps and machine grips, immersed in PBS bath, prepared for testing.
For stress calculations, molds of the cross-sections were scanned at 600 DPI using a commercial scanner (CanoScan LiDE 400). The resulting images were then processed using ImageJ software, where a threshold was applied to differentiate the dental impression material from the remaining area formed by the specimen’s shape. This remaining area corresponds to the specimen’s cross-section, and its area was automatically calculated by the software. The engineering stress (or first Piola-Kirchhoff stress, P) was calculated, which is given by the force in the current configuration divided by the area in the non-deformed reference configuration. To calculate the Cauchy stress tensor, \(\sigma\), the following equation was considered:
where J is the Jacobian, assumed to be 1 since we are assuming an incompressible behavior of soft tissues which is a valid approximation due to the water amount33, and F is the deformation gradient for the purely isochoric uniaxial deformation.
In the uniaxial case, which is the formulation considered in the present work, the Cauchy stress is given by:
in which f corresponds to the applied force, \(A_0\) is the cross-sectional area in the undeformed (reference) configuration, and \(\lambda\) is the stretch ratio in the direction of the applied load.
Relaxation tests
The relaxation tests were displacement-controlled and followed a defined protocol involving seven loading and six unloading cycles at 75% strain, applied at a rate of 3 mm/min. The preconditioning served the purpose to create a more realistic baseline and account for preconditioning as soft tissues in the living body are also being loaded periodically. After the cycles, the displacement was held constant for 900 seconds34. The sample was then stretched to 150%, and another set of seven loading and six unloading cycles to 75% was performed. At 150%, the displacement was again held constant for 900 seconds. After the relaxation test, the sample was stretched until failure, with force-displacement data recorded at 0.1-second intervals. A representation of the protocol implemented is presented in Fig. 7.
Displacement-controlled profile for the cyclic loading and relaxation test protocol.
Ultimate tensile tests
Similarly, the ultimate tensile tests were also displacement-controlled, with specimens stretched until failure at a rate of 1 mm/min, and force-displacement data were acquired at 0.1 second intervals. For the ultimate tensile test, preconditioning of the samples was not performed before testing. Throughout the mechanical tests, the specimens were kept hydrated in a \(37^{\circ }\) C bath containing PBS.
Histological assessment
Samples for histology were taken adjacent to the center of the dog-bone sample. Samples were fixed with 4% formaldehyde for 24 h and then stored in PBS at \(4^{\circ }\)C until further processing. Tissue samples were processed with a KOS Rapid Tissue Processor (Milestone SRL, Valbermbo, Italy) using the pre-programmed setup for Histoprocessing (Fixation: 4% formaldehyde, Dehydration: Ethanol-Isopropanol- Wax). After paraffin embedding, 5-\(\mu\)m sections were obtained using a rotary microtome (Leica RM2255, Nussloch, Germany).
Sirius red staining for collagen was performed according to routine procedure (Weigert’s hematoxylin 15 min; Picro-sirius red 60 min)35. Brightfield imaging was performed under controlled conditions, maintaining consistent brightness and contrast settings across all samples to ensure reliable comparability. Similarly, Masson-Goldner-Trichrome staining was performed according to standard protocol (Weigert’s hematoxylin 2 min; Goldner staining solutions 5 min each). After dehydration in graded solutions of alcohol, slides were cover slipped in DPEX mounting media (Electron Microscopy Science, Hatfield, PA, USA). Throughout the process, xylol was replaced with Histolab Clear (Sanova, Vienna, Austria). Images were taken using a digital slide scanner (Slideview VS200, Olympus, Tokio, Japan) or a camera-equipped microscope (BX-43, Olympus, Tokio, Japan) using a 20x dry lens.
Evaluation of histological stains
Collagen fiber orientation was evaluated employing Fiji17 with the ImageJ plugins Colour Deconvolution 218,19 and OrientationJ20 on Sirius red-stained sections. All imaged sections were evaluated using the same color parameters and consistent fiber thickness using a custom-written macro. The derived histograms of fiber orientation were used to calculate the fiber orientation mean and standard deviation using Python, which were used as an input for the computational model.
Muscle content was evaluated qualitatively due to variance in staining intensity in Masson-Goldner stained sections. Sections were categorized as containing low, medium, or high muscle fiber content.
Calibration of the constitutive parameters
The experimental data were used to calibrate the material parameters of a visco-hyperelastic constitutive model. Finite element simulations were conducted in Abaqus®, modeling a specimen that replicated the geometry and boundary conditions of the experimental setup. The material parameters were iteratively optimized to reproduce the mechanical response of the specimen, and the resulting stress–relaxation and stress–stretch curves were extracted from the simulations. The optimization procedure was implemented in MATLAB, while the finite element analyses were executed in Abaqus®. Simulation results were automatically transferred to MATLAB for analysis, establishing an Abaqus®–MATLAB interface for automated parameter calibration. The simulated curves were then compared with the experimental results to evaluate and refine the calibration. The optimization was performed using a previously developed genetic algorithm21. The different components of this process, including the constitutive model, finite element simulations, and optimization algorithm, are described in detail in the following sections.
Finite element simulation
To obtain numerical data, simulations were conducted to replicate the experimental protocol of the mechanical tests. The geometrical model used for parameter calibration, shown in Fig. 8, represents the specimen from the experimental tests, with dimensions detailed in Fig. 6b. To simplify the analysis, only one-eighth of the specimen was modeled, leveraging symmetry boundary conditions. The final mesh comprises 592 elements of type C3D8H. The fibers were assumed to have a principal orientation aligned with the direction of the applied stretch. The engineering stress in the direction of the applied load was used as the comparison variable with experimental data, and the average response of all finite elements within the gauge length was considered.
Finite element model used for simulating experimental tests, integrated with a genetic algorithm for calibrating the constitutive model parameters. The finite element model consists of a eighth-symmetry of the specimen. (a) Full specimen mesh. (b) Eighth-model with symmetry planes. (c) Applied boundary conditions and direction of the applied stretch.
Material model
Based on the experimental data obtained, a transversely isotropic hyperelastic response with viscoelastic behavior was assumed. The HGO model was considered for the hyperelastic behavior and the generalized Maxwell model for the viscous effects36,37.The constitutive behavior was implemented in Abaqus® using a previously defined user-defined material subroutine (UMAT). The free-energy function is given by Equation 3.
where \(\overline{\Psi }_{m}\) is the isochoric matrix contribution (Equation 4), \(\overline{\Psi }_{f}\) the collagen fibers contribution (Equation 5), and \(\Psi _{vol}\) the volumetric contribution (Equation 6).
where \(c_{10}> 0\) and \(k_1> 0\) are stress-like material parameters, and \(k_2> 0\) is a dimensionless parameter. \(\overline{I}_1 = trace(\overline{{\textbf {C}}})\) is the first invariant of the isochoric right Cauchy-Green strain tensor, given by \({\textbf {C}}={\textbf {F}}^T{\textbf {F}}\), where F is the deformation gradient, and \(\overline{I}_4 = \overline{\lambda ^2}\) is an invariant arising directly from the anisotropy that is equal to the square of the stretch in the fiber direction. Regarding the volumetric contribution, J = det(F) corresponds to a measure of the volume variation, and D is a constitutive parameter that allows to describe the tissue as near-incompressible, with a value of \(1 \times 10^{-5}\) \(\hbox {MPa}^{-1}\) used.
The viscoelastic contribution is represented by the generalized Maxwell model. The implementation includes the addition of a dissipative potential to the strain energy function (Equation 7), describing the non-equilibrium state.
The configurational free energy \(\Upsilon _\alpha\) is a function of the isochoric right Cauchy-Green strain tensor, \(\overline{{\textbf {C}}}\), and a set of strain-like internal variables denoted by \(\varvec{\Gamma }_\alpha\). The latter characterizes the relaxation and/or creep behavior of the material. The viscoelastic behavior is modeled by \(\alpha\)=1,...,m viscoelastic processes.
The second Piola-Kirchhoff stress tensor, given by \({\textbf {S}} = J {\textbf {F}}^{-1} \varvec{\sigma } {\textbf {F}}^{-T}\), can be decomposed into an equilibrium part and a non-equilibrium part characterized by the elastic response of the system and the viscoelastic response, respectively, as demonstrated in Equation 8.
where \({\textbf {S}}^\infty _{vol}\), \({\textbf {S}}^\infty _{m}\) and \({\textbf {S}}^\infty _{f}\) denote the volumetric, matrix and fiber contribution of the second Piola-Kirchhoff stress tensor, and \({\textbf {Q}}_{\alpha }\) correspond to the isochoric non-equilibrium stresses arising from the viscous effects:
where \(\tau _\alpha\) and \(\beta _\alpha\) consist of the relaxation time and free-energy parameters, representing the viscoelastic properties derived from the Maxwell model with \(\alpha\) parallel elements.
Genetic algorithm
The algorithm begins by generating a random population of five elements, where each element represents a unique set of material parameters. The fitness of each element is evaluated using a specific evaluation function, and the fittest element is retained for the next generation, which is produced through a crossover operation. The population is then checked for diversity. If there is insufficient diversity, the population is reinitialized randomly, except for the fittest individual, who is retained for the next generation. This iterative process continues until the algorithm converges.
In the present work, the elements of the population were the material parameters of the constitutive model described in the previous section, and the fitness of each element was evaluated using a least squares evaluation function, \(F_e\), between the experimental data and the solution from the numerical simulation.
where n is the number of considered experimental points and \(P_i^e\) and \(P_i^\Psi\) are respectively the Cauchy stresses in the direction of the applied stretch from the experimental data and from the numerical simulation in Abaqus®.
Calibration
To calibrate the visco-hyperelastic material, the viscous properties of the Generalized Maxwell model were initially determined using the mean normalized relaxation curve obtained from the relaxation part of the experiments, as presented in Fig. 2b. In the calibration process, only the relaxation of the tissue was considered, not including the cyclic loading procedure performed prior to the stress-relaxation test. Normalization ensures that stress-relaxation is expressed as a percentage, independent of the absolute values and the hyperelastic parameters, which are unknown at this stage. The normalization was performed by dividing each data point by the maximum value of the relaxation curve.
Once the viscous parameters were successfully determined, the next step involved calibrating the hyperelastic properties. This was done by comparing the model’s predictions with the absolute values obtained from the Cauchy stress – stretch experimental data (Fig. 3). By fitting the model to the mean of the experimental curves, the hyperelastic parameters were determined, completing the material calibration process.
Data availability
The experimental data from this study have been deposited in the Zenodo repository (https://doi.org/10.5281/zenodo.15407814).
References
Drake, R., Wayne Vogl, A. & Mitchell, A. W. M. Gray’s Anatomy for Students 5th edn (Elsevier, Philadelphia, MO, 2023).
Kimmich, N., Yeo Te-ying, A., Zimmermann, R. & Furrer, E. How do sustained birth tears after vaginal birth affect birth tear patterns in a subsequent birth?. J. Perinat. Medicine 48, 335–344. https://doi.org/10.1515/jpm-2020-0007 (2020).
Dudding, T., Vaizey, C. & Kamm, M. Obstetric Anal Sphincter Injury: Incidence, Risk Factors, and Management. Annals Surg. 247, 224–237. https://doi.org/10.1097/SLA.0b013e318142cdf4 (2008).
Riva, D. & Minini, G. (eds.) Childbirth-Related Pelvic Floor Dysfunction 1st edn (Springer International Publishing, Cham, Switzerland, 2016).
Doumouchtsis, S. Childbirth Trauma 1st edn (Springer, London, UK, 2016).
Santoro, G. A., Wieczorek, A. P. & Sultan, A. H. (eds.) Pelvic Floor Disorders: A Multidisciplinary Textbook 2nd edn (Springer International Publishing, Cham, Switzerland, 2021).
Hoyte, L. & Damaser, M. Biomechanics of the female pelvic floor (Academic Press, London (UK; San Diego, 2016).
Chen, S. & Grimm, M. J. Childbirth Computational Models: Characteristics and Applications. J. Biomech. Eng. 143, 050801. https://doi.org/10.1115/1.4049226 (2021).
Couri, B. M., Lenis, A. T., Borazjani, A., Paraiso, M. F. R. & Damaser, M. S. Animal models of female pelvic organ prolapse: lessons learned. Expert. Rev. Obstet. & Gynecol. 7, 249–260. https://doi.org/10.1586/eog.12.24 (2012).
Rynkevic, R. et al. The effect of consecutive pregnancies on the ovine pelvic soft tissues: Link between biomechanical and histological components. Annals Anat. - Anat. Anzeiger 222, 166–172. https://doi.org/10.1016/j.aanat.2018.12.002 (2019).
Chen, L., Low, L. K., DeLancey, J. O. & Ashton-Miller, J. A. In vivo estimation of perineal body properties using ultrasound quasistatic elastography in nulliparous women. J. Biomech. 48, 1575–1579. https://doi.org/10.1016/j.jbiomech.2015.02.056 (2015).
Li, X. et al. Assessment of perineal body properties in women with stress urinary incontinence using Transperineal shear wave elastography. Sci. Reports 14, 21647. https://doi.org/10.1038/s41598-024-72429-5 (2024).
Kochová, P. et al. The histological microstructure and in vitro mechanical properties of pregnant and postmenopausal ewe perineal body. Menopause 26, 1289–1301. https://doi.org/10.1097/GME.0000000000001395 (2019).
Lallemant, M. et al. In vitro biomechanical properties of porcine perineal tissues to better understand human perineal tears during delivery. Acta Obstet. et Gynecol. Scand. 103, 1386–1395. https://doi.org/10.1111/aogs.14791 (2024).
Kochová, P. et al. The histological microstructure and in vitro mechanical properties of the human female postmenopausal perineal body. Menopause 26, 66–77. https://doi.org/10.1097/GME.0000000000001166 (2019).
Author. Experimental Data on the Hyperelastic and Viscoelastic Properties of Human Female Perineal Tissue [Data set], https://doi.org/10.5281/zenodo.15407814 (2025).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682. https://doi.org/10.1038/nmeth.2019 (2012).
Landini, G., Martinelli, G. & Piccinini, F. Colour deconvolution: stain unmixing in histological imaging. Bioinformatics 37, 1485–1487. https://doi.org/10.1093/bioinformatics/btaa847 (2021).
Ruifrok, A. C. & Johnston, D. A. Quantification of histochemical staining by color deconvolution. Anal. Quant. Cytol. Histol. 23, 291–299 (2001).
Rezakhaniha, R. et al. Experimental investigation of collagen waviness and orientation in the arterial adventitia using confocal laser scanning microscopy. Biomech. Model. Mechanobiol. 11, 461–473. https://doi.org/10.1007/s10237-011-0325-z (2012).
Vila Pouca, M., Parente, M., Natal Jorge, R. & Ashton-Miller, J. Investigating the birth-related caudal maternal pelvic floor muscle injury: The consequences of low cycle fatigue damage. J. Mech. Behav. Biomed. Mater. 110, 103956. https://doi.org/10.1016/j.jmbbm.2020.103956 (2020).
Abramowitch, S. & Easley, D. Introduction to Classical Mechanics. In Biomechanics of the Female Pelvic Floor, 89–107, https://doi.org/10.1016/B978-0-12-803228-2.00004-0 (Elsevier, 2016).
Jing, D., Ashton-Miller, J. & DeLancey, J. A subject-specific anisotropic visco-hyperelastic finite element model of female pelvic floor stress and strain during the second stage of labor. J. Biomech. 45, 455–460. https://doi.org/10.1016/j.jbiomech.2011.12.002 (2012).
Anssari-Benam, A., Bader, D. L. & Screen, H. R. Anisotropic time-dependant behaviour of the aortic valve. J. Mech. Behav. Biomed. Mater. 4, 1603–1610. https://doi.org/10.1016/j.jmbbm.2011.02.010 (2011).
Chantereau, P. et al. Mechanical properties of pelvic soft tissue of young women and impact of aging. Int. Urogynecology J. 25, 1547–1553. https://doi.org/10.1007/s00192-014-2439-1 (2014).
Natali, A., Pavan, P., Carniel, E., Dario, P. & Izzo, I. Characterization of soft tissue mechanics with aging. IEEE Eng. Medicine Biol. Mag. 27, 15–22. https://doi.org/10.1109/MEMB.2008.919492 (2008).
Delgadillo, J. O. V., Delorme, S., El-Ayoubi, R., DiRaddo, R. & Hatzikiriakos, S. G. Effect of freezing on the passive mechanical properties of arterial samples. J. Biomed. Sci. Eng. 03, 645–652. https://doi.org/10.4236/jbise.2010.37088 (2010).
Fischer, B., Kurz, S., Höch, A. & Schleifenbaum, S. The influence of different sample preparation on mechanical properties of human iliotibial tract. Sci. Reports 10, 14836. https://doi.org/10.1038/s41598-020-71790-5 (2020).
Ekiert, M., Karbowniczek, J., Stachewicz, U. & Mlyniec, A. The effect of multiple freeze-thaw cycles on the viscoelastic properties and microstructure of bovine superficial digital flexor tendon. J. Mech. Behav. Biomed. Mater. 120, 104582. https://doi.org/10.1016/j.jmbbm.2021.104582 (2021).
Anssari-Benam, A., Bader, D. L. & Screen, H. R. C. A combined experimental and modelling approach to aortic valve viscoelasticity in tensile deformation. J. Mater. Sci. Mater. Medicine 22, 253–262. https://doi.org/10.1007/s10856-010-4210-6 (2011).
Scholze, M. et al. Standardized tensile testing of soft tissue using a 3D printed clamping system. HardwareX 8, e00159. https://doi.org/10.1016/j.ohx.2020.e00159 (2020).
Moura, R., Oliveira, D. A., Parente, M. P., Kimmich, N. & Natal Jorge, R. M. A biomechanical perspective on perineal injuries during childbirth. Comput. Methods Programs Biomed. 243, 107874. https://doi.org/10.1016/j.cmpb.2023.107874 (2024).
Vito, R. P. & Dixon, S. A. Blood Vessel Constitutive Models–1995–2002. Annu. Rev. Biomed. Eng. 5, 413–439. https://doi.org/10.1146/annurev.bioeng.5.011303.120719 (2003).
Peña, E. et al. Experimental study and constitutive modeling of the viscoelastic mechanical properties of the human prolapsed vaginal tissue. Biomech. Model. Mechanobiol. 9, 35–44. https://doi.org/10.1007/s10237-009-0157-2 (2010).
Rittié, L. Method for Picrosirius Red-Polarization Detection of Collagen Fibers in Tissue Sections. In Rittié, L. (ed.) Fibrosis, vol. 1627, 395–407, https://doi.org/10.1007/978-1-4939-7113-8_26 (Springer New York, New York, NY, 2017).
Holzapfel, G. A., Gasser, T. C. & Ogden, R. W. A new Constitutive Framework for Arterial Wall Mechanics and a Comparative Study of Material Models. In Cowin, S. C. & Humphrey, J. D. (eds.) Cardiovascular Soft Tissue Mechanics, 1–48, https://doi.org/10.1007/0-306-48389-0_1 (Kluwer Academic Publishers, Dordrecht, 2004).
Vila Pouca, M., Ferreira, J., Oliveira, D., Parente, M. & Natal Jorge, R. Viscous effects in pelvic floor muscles during childbirth: A numerical study. Int. J. for Numer. Methods Biomed. Eng. 34, e2927. https://doi.org/10.1002/cnm.2927 (2017).
Acknowledgements
The authors gratefully acknowledge the support from Portuguese Foundation of Science under the Grants 10.54499/2021.05876.BD and 10.54499/2022.09480.BD, the Junior Researcher Contract 10.54499/2020.01522.CEECIND/CP1612/CT0001, the funding of Project UIDB/50022/2020, and the Erasmus+ Short PhD Mobility Program. The authors also thank Noah Frank, Julia M. A. Puchleitner and Michael Pretterklieber for support during sample obtainment, and Melika Muzaferovic-Majdanac for support during histological processing.
Funding
This work was funded by the Portuguese Foundation of Science under the Grants 10.54499/2021.05876.BD and 10.54499/2022.09480.BD, the Junior Researcher Contract 10.54499/2020.01522.CEECIND/CP1612/CT0001, and the funding of Project UIDB/50022/2020.
Author information
Authors and Affiliations
Contributions
R.M. and A.S. conceived the original idea. R.M. conducted the experiments. A.S. and T.P. supervised the experiments. R.M. wrote the manuscript with support from D.O., M.C. and A.S. R.M., M.C., D.O., and A.S. analyzed the results. A.S., M.P. and N.H. supervised the project. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Moura, R., A. Oliveira, D., R. Carvalho, M. et al. Experimental characterization of the non-linear viscoelastic properties of human female perineal tissue. Sci Rep 15, 42483 (2025). https://doi.org/10.1038/s41598-025-26576-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-26576-y