Abstract
Prolonged exposure to stress alters brain pathophysiology by increasing inflammation leading to cellular and molecular dysfunction and initiation of neurobehavioral changes like anxiety, depression and cognitive impairment. Various signaling pathways and downstream cascades modulate the inflammatory responses within the Central Nervous System (CNS), notably cGAS-STING pathway and Nuclear factor kappa B (NF-κB) signaling are majorly affected. The present research aims to assess the consequence of modulation of cGAS with suramin in chronic unpredictable stress (CUS), and examining the impact on neurobehavioral alterations. For evaluating the impact of CUS and pharmacological interventions on neurobehavioral changes, Swiss albino mice of either sex were exposed to various stressors for 8 weeks. The behavioral traits for anxiety, depression and memory impairment were assessed by Elevated Plus Maze (EPM), Forced Swim Test (FST), Sucrose Preference Test (SPT), Tail Suspension Test (TST), Morris Water Maze (MWM) and Passive Avoidance Test (PAT). The level of corticosterone and neurotransmitters (dopamine and serotonin); various parameters of oxidative stress and neuroinflammation; acetylcholinesterase (AChE) activity and histological changes in hippocampus and cortex were also assessed. The results highlighted that administration of suramin (5, 10 and 20 mg/kg, i.p.) dose dependently improves neurobehavioral parameters, decreases corticosterone; elevates the level of neurotransmitters, and reduces the deleterious effects of CUS on oxidative stress and neuroinflammation; along with the histological improvement by inhibiting the cGAS-STING pathway and modulating NF-κB signaling as observed from decrease NF-κB levels in our study This study provides insights into suramin as a new treatment approach for mitigating the effects of chronic stress on restoring neurobehavioral changes.
Similar content being viewed by others
Introduction
Chronic stress has emerged as a significant public health concern, recognized for its effect on both physical and psychological well-being1,2. The body’s response to prolonged stress involves intricate neurobiological and immunological adaptations that lead to dysregulation of various cellular processes3,4. Chronic unpredictable stress (CUS) causes the persistent stimulation of the hypothalamic-pituitary-adrenal (HPA) axis and the subsequent overproduction of glucocorticoids, that promotes inflammation, by causing alteration in the pathophysiology of brain leading to neurodegeneration and neurobehavioral changes5,6. In addition, chronic stress-induced glucocorticoid elevation has been shown to impair hippocampal neurogenesis, disrupt synaptic plasticity, and increase vulnerability to neuroinflammatory insults, all of which are closely associated with the onset and progression of anxiety and depression-like behaviors7,8. Such maladaptive changes are further compounded by oxidative damage, mitochondrial dysfunction, and alterations in monoaminergic signaling9,10, highlighting the multifactorial nature of CUS-induced neuropathology. In Central Nervous System (CNS) various signaling pathways and downstream cascades are implicated in the modulation of the immunological responses11 notably cGAS-STING pathway and nuclear factor kappa B (NF-κB) signaling are majorly affected.
The cyclic GMP-AMP synthase (cGAS)-stimulator of interferon genes (STING) pathway, serves as an essential element of the innate immune system, has been recognized as a significant player in the progression of various neurodegenerative conditions characterized by chronic neuroinflammation12. This mechanistic pathway is initiated by dsDNA that infiltrates into the cytoplasm from injured neuronal cells, triggering a series of events that lead to the activation of STING and cGAS13,14. Once activated, STING triggers the downstream signaling cascades, including NF-κB & interferon regulatory factors (IRFs) along with the synthesis of type I interferons, which orchestrate the pathological response associated with inflammation15,16. NF-κB at the molecular level regulates the expression of genes vital for inflammation and cell survival17. Persistent activation of cGAS-STING/ NF-κB pathway due to continuous stress induces pathological condition by exacerbating inflammation18, increased oxidative stress19 and altered neurotransmitter levels which serve as hallmark of various neurobehavioral disorders20. Emerging studies suggest that modulation of this pathway can exert profound neuroprotective effects by attenuating microglial activation, reducing pro-inflammatory cytokine release, and restoring redox homeostasis21,22. These findings position the cGAS–STING/NF-κB axis as a promising therapeutic target in stress-related neuropathologies.
Suramin, is a complex chemical compound classified as polysulfonated naphthylurea, known for its potential role in neurodegeneration. It has the capacity to inhibit various pathways which are associated with neurological disorders23,24. Suramin is characterized as a potent inhibitor of various enzymes, especially cGAS. It directly binds to cGAS, therefore interfering with its enzymatic activity and prevents the synthesis of cyclic GMP-AMP (cGAMP), the second messengers that activates the STING pathway25. Suramin also regulates the neuroinflammatory responses by inhibiting purinergic signaling, which is often dysregulated in neurodegenerative disorders like Alzheimer’s and Parkinson’s diseases26,27. Previous studies have demonstrated its neuroprotective effects in conditions such as brain ischemia and neurodegenerative diseases by reducing oxidative stress, inhibiting glial cell activation, and promoting neuronal survival28,29. Notably, Suramin has also been reported to ameliorate behavioral deficits in rodent models of autism and neuroinflammation30, further supporting its capacity to modulate neuroimmune interactions. However, despite this growing body of evidence, its potential in stress-induced neuropathology has not been systematically explored and its effect on CUS induced neurobehavioral changes through cGAS-STING/ NF-κB pathway remains unknown.
Given the multifaceted nature of chronic stress pathology, targeting key inflammatory and oxidative pathways could provide a novel therapeutic approach. We hypothesized that suramin would attenuate CUS-induced neurobehavioral deficits by inhibiting the cGAS-STING/ NF-κB pathway, thereby reducing oxidative stress, neuroinflammation, and neurotransmitter dysregulation while restoring neuronal integrity. Thus, this study aimed to investigate the neuroprotective effects of suramin against CUS-induced anxiety, depression, and memory impairments by assessing behavioral, biochemical, and histological alterations.
Materials and methods
Animals
The present work included 25–30 g, 8–10 weeks old, Swiss albino mice of either sex in order to enhance the translational relevance of the finding. Animals were procured from Chitkara University, Rajpura, Punjab, India and were fed a normal pellet diet as required. Protocol for the current study was approved by Institutional Animals Ethics Committee (IAEC) of Chitkara University, Punjab, India (protocol number IAEC/CCP/24/01/PR-02). Animals were duly taken care by strictly following the guidelines issued by CCSEA (Committee for control and supervision of experimentation on animals), a committee governed by Ministry of Fisheries, Animal Husbandry and Dairying, Department of Animal Husbandry and Dairying, Government of India. The study is reported in accordance with the ARRIVE guidelines to ensure transparent and comprehensive reporting of animal research.
Drugs and chemicals
Serotonin (Cat No. 14927; Sigma Aldrich); Dopamine (Cat No. PHR1090; Sigma Aldrich); Corticosterone (Cat No. 27840; Sigma Aldrich); IL1-β (Cat No. KB3063; Krishgen Biosystem); IL-6 (Cat No. KB2068; Krishgen Biosystem); TNF-α (Cat No. E0117Mo; BT Lab); NF-κB (Cat No. K-02 2879; Kinesis Dx). Freshly prepared drug solutions were ensured for all experiments. Suramin was solubilized in water for injection (1 mg/ml) and administered in a dose range of 5 mg/kg, 10 mg/kg and 20 mg/kg; i.p.31,32. All other chemicals used in the study were of analytical grade and were purchased from standard laboratories.
Chronic unpredictable stress (CUS) protocol
The CUS protocol is a widely used preclinical model to induce neurobehavioral changes in mice by subjecting them to a sequence of unpredictable and variable stressors over a prolonged time. After 4 days of adaptation period, mice were given several stressors such as cold swim (animals were allowed to swim in water maintained at 12 °C for 5 min), tail pinch (soft artery clamp was applied 2 cm from base of tail for 1 min), food deprivation, water deprivation (12 h of deprivation), cage tilt (tilted at 45° angle for 1 h), physical restraint (restrained in ventilated restraining tubes for 10 min) and foot shock (0.6 mA for 1 s delivered for a period of 2 min) once a week for 56 days as mentioned in Table 133,34.
Experimental design
Animals were randomly assigned to seven groups (n = 10 per group) as follows: Group 1 – Control group; Group 2 – CUS group; Group 3 – Suramin per se (20 mg/kg, i.p.); Group 4 - Suramin-low + CUS (SUR-L, 5 mg/kg, i.p.); Group 5 – Suramin-medium + CUS (SUR-M, 10 mg/kg, i.p.); Group 6 – Suramin-high + CUS (SUR-H, 20 mg/kg, i.p.); Group 7 – Fluoxetine + CUS (20 mg/kg, p.o.). Suramin was freshly prepared in water for injection (1 mg/ml) and administered intraperitoneally once weekly. The chosen suramin doses (5–20 mg/kg) were based on previous preclinical protocols to cover a low-to-high range31,32. Fluoxetine (2 mg/ml) was freshly prepared in distilled water and administered orally once daily at a dose of 20 mg/kg. All the grouped animals were given stress according to the protocol described in Table 1, except the control group. The behavioral estimation was carried out on all the 10 animals of each group, furthermore 3 were used for histological estimations and 7 for biochemical studies. This design was intended to facilitate the direct correlation of behavioral outcomes with underlying neurochemical alterations. Behavioral and biochemical studies were performed as shown in Fig. 1.
Experimental protocol.
Estimation of behavioral parameters
Elevated plus maze (EPM)
For the estimation of anxiety like behavior the EPM test was performed on day 57. The apparatus consists of two open ‘and closed arms (30 × 5 cm) with central square compartment (5 × 5 cm). During the test, each mouse was positioned on the central square, facing one of the open arms and allowed to move across the maze for 5 minutes. The duration of stay in open arms and the number of entrances into the closed arms were both quantified35,36,37.
Tail suspension test (TST)
On day 57, TST was used to evaluate the depressive activity in mice. The animals were progressively hanged by lifting their tails from the hook on the apparatus about 50 cm above a level of the platform in order to prevent them from touching the ground or walls. The animals were observed for 6 min to record the immobility duration38.
Sucrose preference test (SPT)
The sucrose preference test, consist of two-bottle choice setup, which is an effective method for assessing anhedonia (major sign of serious depressive illness) in animals. This assessment entails measuring the animal’s ingestion of a sweetened solution, with a decrease in consumption indicates anhedonia-like behavior. The protocol comprises of two main sessions39.
Before the initiation of the CUS induction, mice were acclimatized to consume regular water from two bottles for a duration of 24-hour. subsequent to this acclimatization, one of the water bottles was switched with a 2.5% sucrose solution (w/v in water), and the animals were given unlimited access to both bottles for another 24 h. On the following day, individuals were given a 1-hour interval to choose between the two bottles—one containing plain water and other solution of sucrose. The placements of the bottles were changed for every two hours in order to avoid side preference. The intake of both sucrose & water was measured by weighing the bottles pre- and post-consumption. Sucrose preference was estimated as a percentage of the total liquid ingested using the equation:
Session 2: Testing Phase.
The mice were once again offered drinking water and sucrose solution together for one hour on the 57th day. The proportion of consumed sucrose during this test phase was calculated in the same way as in the baseline session, in order to compare stressed and control groups.
Forced swim test (FST)
FST assesses stress induced depression. During the training phase on day 57, the mice were placed in a glass container (60 cm height, 20 cm width) filled with water till 40 cm deep and maintained at (23 ± 2 °C) for 15 min. After 24-hour period, the test was performed for 6 min on each mouse, and they were monitored for immobility36,38.
Morris water maze (MWM)
This test was conducted as outlined by Morris and colleagues (1984) with few modifications40,41. The equipment consists of a large circular pool with 150 cm of diameter and a height of 35 cm with a platform placed 1 cm under the water surface. The circular pool was filled with water kept at 23–25֯ C, and harmless white dye was added to turn the water transparent so as to obstruct the platform visibility. The circular pool was partitioned into 4 equal virtual quadrants labelled Q1, Q2, Q3, and Q4. The acquisition trial test was conducted for 4 days, starting from day 53 to day 56, consisting of four trials a day for each mouse to locate the submerged platform. At the beginning of each trial, the mice were positioned in the pool and allowed to navigate for 120 s to locate the hidden platform; during this time, the escape latency (ELT) was recorded. On day 57, the platform was taken out from the pool, and the retrieval session was conducted, allowing the mice to move freely for 120 s, during which the duration of time spent in each quadrant was recorded. The mean average of time spent in target quadrants (TSTQ) was used to estimate memory retrieval42,43.
Passive avoidance test (PAT)
PAT was conducted to assess memory retention. On day 57, the acquisition trial of PAT was carried out. The animals were positioned on the platform and permitted to step on the floor, consisting of an electrical shock (0.5 mA) supplied for 5 s from the rods on the surface. After 24 h of the acquisition trial (day 58), the retention trial was carried out. The same procedure was followed as in the acquisition trial44. In both the acquisition and retention trials, the step-down latency time (sec) for each mouse was measured45.
Estimation of biochemical parameters
The blood sample was collected from mice (via cardiac puncture); and serum was used to estimate the level of corticosterone33. For biochemical assays, animals were euthanized using ketamine (i.p.) overdose in accordance with CCSEA-approved methods and ARRIVE guidelines. The brains were homogenized in a phosphate buffer (pH 7.4, 10% w/v) by using a homogenizer. After centrifugation at 3000 rpm for 15 min, clear supernatant was obtained to estimate AChE46, dopamine47, serotonin47, catalase48, TBARS49, GSH50,51, SOD50,52, MPO53. The levels of NF-κB, IL-1β IL-6 and TNF-α in homogenate of brain tissue were measured according to the protocol and instructions manual provided by Bioassay Technology Lab Elisa kit and Krishgen Biosystems Elisa kits.
Histopathology
For the histopathological estimation the brains of animals were removed and kept in 10% formalin solution for overnight. The blocks were prepared by embedding the tissue in paraffin followed by cutting the 5 μm thick section using microtome. The sections obtained were stained with hematoxylin and eosin and observed under light microscope (Olympus DP23 camera and were analyzed using Cell Sense software) at magnification of 10X, 20X and 40X for the histopathological changes in the hippocampus and cortex54,55.
Statistical analysis
In the current study data is presented as Mean ± S.E.M.; statistically analyzed by one-way ANOVA followed by Tukey’s multiple comparisons test in Graph pad prism 9. A value of p < 0.05 was considered statistically significant. Levels of significance are indicated as *p < 0.05, **p < 0.01, ***p < 0.001, and ****p < 0.0001.
Results
Effect of suramin on duration of stay and number of entries in open arm of EPM
Figure 2 demonstrates the impact of CUS and various treatments on the duration of time spent in open arms (Fig. 2A) along with the number of entrances into the closed arm (Fig. 2B) as assessed on day 57 of the study. As anticipated, CUS exposure led to a significant reduction in time spent in open arm of EPM as compared to control group indicating avoidance behavior reflecting an increased fear response and diminished exploratory activity. Conversely, suramin (5, 10 and 20 mg/kg, i.p.) administration, significantly increased the duration spent in open arms as compared to the CUS group in a dose-dependent manner, highlighting its efficacy in attenuating behavior akin to anxiety. Notably, suramin per se did not significantly alter the time spent relative to the control group. Additionally, fluoxetine (20 mg/kg; p.o.) administration significantly alleviated the symptoms of anxiety in CUS-exposed animals.
Effect of various treatments on (A) Time spent in open arm and (B) Number of closed arm entries of EPM in mice subjected to chronic unpredictable stress. Values are presented as mean ± S.E.M. (n = 10), analyzed by one-way ANOVA followed by Tukey’s multiple comparison test. p < 0.05 was considered statistically significant. ****p < 0.0001, **p < 0.01, *p < 0.05.
Effect of Suramin on sucrose preference
Figure 3 depicts the impact of CUS and several pharmacological treatments on sucrose preference, highlighting the hedonic responses. At baseline, sucrose preference was consistent across all groups, indicating no initial differences in reward-seeking behavior. CUS induction significantly decreased the preference for the sucrose intake relative to the control group, indicating a state of anhedonia and reduced motivation for pleasurable stimuli. Treatment with suramin (5, 10 and 20 mg/kg, i.p.) substantially and dose-dependently increases the consumption of sucrose solution compared to CUS group, underscoring its effectiveness in alleviating anhedonia-like symptoms. Suramin (20 mg/kg. i.p.), per se did not exhibit any substantial change in comparison to the control group. Furthermore, administration of fluoxetine (20 mg/kg, p.o.) also successfully reversed the diminished sucrose preference observed in animals exposed to CUS, indicating its role in restoring hedonic responsiveness.
Effect of various treatments on sucrose preference in mice subjected to chronic unpredictable stress. Values are presented as mean ± S.E.M. (n = 10), analyzed by two-way ANOVA followed by Tukey’s multiple comparison test. p < 0.05 was considered statistically significant. ****p < 0.0001, ***p < 0.001, **p < 0.01, *p < 0.05.
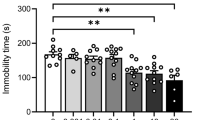
Effect of Suramin on immobility time in tail suspension test (TST)
TST exhibits increased immobility time in animals when they have no means of escape, which is considered an indicator of learned helplessness, a key feature often associated with depression. As depicted in Fig. 4, the CUS exposed rodents demonstrated a substantial increase in immobility time relative to control group, confirming depression-like behavior of animals. Administration of fluoxetine (20 mg/kg, p.o.) and suramin (5, 10 and 20 mg/kg, i.p.) showed significantly and dose-dependent decrease in the immobility time in comparison to CUS group indicating attenuation of despair and depressive symptoms. However, suramin (20 mg/kg. i.p.), per se substantially did not exhibit any effect on the immobility time relative to the control group.
Effect of various treatments on immobility time in TST in mice subjected to chronic unpredictable stress. Values are presented as mean ± S.E.M. (n = 10) and analyzed by one-way ANOVA followed by Tukey’s multiple comparison test. p < 0.05 was considered statistically significant. ****p < 0.0001, ***p < 0.001.
Effect of Suramin on immobility time in forced swim test (FST)
Figure 5 illustrate the effect of CUS and various pharmacological treatments on immobility time and behavioral despair in FST. Consistent with expectations, the CUS paradigm markedly increases immobility time as compared to the control group, validating its effectiveness in modeling depression-like behavior. Administration of suramin (5, 10 and 20 mg/kg, i.p.) led to a substantial and dose-dependent decrease in immobility time relative to the CUS group underscoring the considerable potential of suramin in targeting the core symptoms of depression. Suramin (20 mg/kg. i.p.), per se did not substantially alters the duration of immobility as compared to the control group. Additionally, treatment with fluoxetine (20 mg/kg,p.o.) effectively decreased the immobility time in animals exposed to CUS.
Effect of various treatments on immobility time in FST in mice subjected to chronic unpredictable stress. Values are presented as mean ± S.E.M. (n = 10) and analyzed by one-way ANOVA followed by Tukey’s multiple comparison test. p < 0.05 was considered statistically significant. ****p < 0.0001, **p < 0.01.
Effect of suramin on escape latency time (ELT) and time spent in target quadrant (TSTQ) in MWM
MWM evaluates the spatial learning and reference memory by estimating the ELT and TSTQ. As depicted in Table 2, the control group exhibited statistically lower day 56 ELT as compared to day 53 ELT depicting normal memory functions of animals. CUS group demonstrated impaired performance, as reflected by the increased ELT on day 56 relative to the day 56 ELT of the control group. However, suramin (20 mg/kg, i.p.), per se did not exhibit any substantial change in the latency time on day 56 in comparison to the control group. Treatment with suramin and fluoxetine reduced day 56 ELT relative to day 56 ELT of CUS group, indicating recovery of memory acquisition. However, during the retrieval test on day 57, there is substantial reduction in the time spent in target quadrant (TSTQ) by CUS group compared to the control group signifying a decrease in retention of spatial information and memory dysfunction (Fig. 6). The administration of suramin (5, 10 and 20 mg/kg, i.p.) substantially and dose-dependently enhanced the time spent in target quadrant as compared to CUS group. Suramin (20 mg/kg. i.p.), per se did not substantially alter the behavioral activity relative to the control group. Additionally, treatment with fluoxetine (20 mg/kg, p.o.) effectively improved TSTQ in animals subjected to CUS implying memory retrieval.
Effect of various treatments on TSTQ of MWM in mice subjected to chronic unpredictable stress. Values are presented as mean ± S.E.M. (n = 10) and analyzed by one-way ANOVA followed by Tukey’s multiple comparison test. p < 0.05 was considered statistically significant. ****p < 0.0001, **p < 0.01, *p < 0.05.
Effect of Suramin on stepdown latency time in passive shock avoidance task (PSA)
This test was used to assess memory and learning in mice by estimating stepdown latency time for associative learning, particularly in the context of fear conditioning. The latency time was reduced in CUS relative to the control group reflecting rodent’s inability to remember the association between stepping down and the negative consequence (shock) (Fig. 7). The groups treated with suramin (5, 10 and 20 mg/kg, i.p.) substantially and dose-dependently increased the latency time compared to the CUS group. Suramin (20 mg/kg. i.p.), per se did not show any significant change in comparison to the control group. The administration of fluoxetine (20 mg/kg, p.o.) substantially enhanced the latency time (Fig. 7) as compared to CUS induced mice indicating retention of fear conditioning and memory consolidation.
Effect of various treatments on stepdown latency time in passive avoidance task in mice subjected to chronic unpredictable stress. Values are presented as mean ± S.E.M. (n = 10) and analyzed by one-way ANOVA followed by Tukey’s multiple comparison test. p < 0.05 was considered statistically significant. ****p < 0.0001, **p < 0.01, *p < 0.05.
Effect of Suramin on brain ache activity
The Fig. 8 depicts that the CUS treated group demonstrated a substantial increase in levels of AChE activity relative to control group. Administration of suramin (20 mg/kg. i.p.), per se did not substantially affect the AChE activity as compared to the control group. Treatment with suramin (5, 10 and 20 mg/kg, i.p.) and fluoxetine (20 mg/kg, p.o.) exhibited significant and dose-dependently decrease in level of AChE activity as compared to CUS group.
Effect of various treatments on brain AChE activity in mice subjected to chronic unpredictable stress. Values are presented as mean ± S.E.M. (n = 6) and analyzed by one-way ANOVA followed by Tukey’s multiple comparison test. p < 0.05 was considered statistically significant. ****p < 0.0001, ***p < 0.001, **p < 0.01.
Effect of Suramin on brain corticosterone level
As depicted in Fig. 9 chronic exposure to stress significantly increases the corticosterone levels relative to the control group. Importantly, treatment with Suramin (5, 10 and 20 mg/kg, i.p.) and fluoxetine (20 mg/kg, p.o.) effectively lowered the elevated corticosterone levels triggered by CUS, underscoring its potential to modulate the dysregulation of the HPA axis. However, suramin per se did not show any significant change in corticosterone levels compared to control group.
Effect of various treatments on blood corticosterone levels in mice subjected to chronic unpredictable stress. Values are presented as mean ± S.E.M. (n = 6) and analyzed by one-way ANOVA followed by Tukey’s multiple comparison test. p < 0.05 was considered statistically significant. ****p < 0.0001, ***p < 0.001.
Effect of Suramin on oxidative stress markers (TBARS, SOD, GSH and catalase levels)
In current study, CUS significantly elevated TBARS levels while simultaneously decreasing GSH levels and the activity of catalase and SOD, relative to the control group (Fig. 10) indicating imbalance between oxidative stress parameters and protective antioxidant enzymes. Intriguingly, treatment with suramin (5, 10 and 20 mg/kg, i.p.) effectively counteracted the CUS-induced alterations lipid peroxidation and antioxidant enzyme activity thus confirming the protective effects of suramin against oxidative stress and lipid peroxidation. However, administration of suramin (20 mg/kg, i.p.), per se substantially did not affect the levels of oxidative markers relative to the control group. Treatment with fluoxetine (20 mg/kg, p.o.) resulted in positive modulation of oxidative markers when statistically compared to CUS-exposed animals.
Effect of various treatments on brain (A) GSH, (B) SOD, (C) catalase and (D) TBARS in mice subjected to chronic unpredictable stress. Values are presented as mean ± S.E.M. (n = 6) and analyzed by one-way ANOVA followed by Tukey’s multiple comparison test. p < 0.05 was considered statistically significant. ****p < 0.0001, ***p < 0.001, **p < 0.01, *p < 0.05.
Effect of Suramin on brain inflammatory markers (IL-1β, IL-6, TNF-α and NF-κB)
CUS exposure dramatically increased the levels of IL-1β, IL-6, TNF-α and NF-κB in the brain, relative to the control group (Fig. 11). This robust elevation underscores the significant inflammatory response triggered by CUS. Suramin (20 mg/kg, i.p.) per se yielded inflammatory marker levels that were statistically similar to those in the control group. Treatment with suramin (5, 10 and 20 mg/kg, i.p.) and fluoxetine (20 mg/kg, p.o.) significantly reduce the levels of IL-1β, IL-6, TNF-α, and NF-κB in a dose dependent manner, establishing the beneficial effect on inflammatory parameters.
Effect of various treatments on brain A) TNF-α, IL-6, IL-1β, NF-κB in mice subjected to chronic unpredictable stress. Values are presented as mean ± S.E.M. (n = 6) and analyzed by one-way ANOVA followed by Tukey’s multiple comparison test. p < 0.05 was considered statistically significant. ****p < 0.0001, ***p < 0.001, **p < 0.01, *p < 0.05.
Effect of Suramin on brain MPO levels
CUS induction significantly increased MPO levels in comparison to control group (Fig. 12). Treatment with suramin (5, 10 and 20 mg/kg, i.p.) and fluoxetine (20 mg/kg, p.o.) substantially and dose-dependently decreased the MPO levels compared to CUS group. Suramin (20 mg/kg. i.p.), per se did not substantially change the MPO levels relative to the control group.
Effect of various treatments on brain MPO levels in mice subjected to chronic unpredictable stress. Values are presented as mean ± S.E.M. (n = 6) and analyzed by one-way ANOVA followed by Tukey’s multiple comparison test. p < 0.05 was considered statistically significant. ****p < 0.0001, ***p < 0.001, **p < 0.01.
Effect of Suramin on brain serotonin and dopamine levels
CUS exposure exhibited a substantial decline in both serotonin and dopamine levels in comparison to the control group (Fig. 13), indicating that chronic stress disrupts neurotransmitter levels involved in mood regulation and emotional response. However, treatment with suramin (5, 10 and 20 mg/kg, i.p.) and fluoxetine (20 mg/kg, p.o.) substantially and dose-dependently restores these neurotransmitter reductions in CUS-exposed animals, suggesting Suramin’s capacity to re-establish neurotransmitter balance (Fig. 13). However, suramin (20 mg/kg. i.p.), per se substantially did not affect the serotonin and dopamine levels relative to the control group.
Effect of various treatments on brain (A) dopamine and (B) serotonin levels in mice subjected to chronic unpredictable stress. Values are presented as mean ± S.E.M. (n = 6) and analyzed by one-way ANOVA followed by Tukey’s multiple comparison test. p < 0.05 was considered statistically significant. ****p < 0.0001, ***p < 0.001, **p < 0.01.
Histopathological evaluation
CUS causes structural changes in the brains of mice. Histopathological evaluation reveals disruption in the molecular layer of the cortex and alterations in neuronal structures of the hippocampus (Figs. 14 and 15, Supplementary Fig. 1 and supplementary Fig. 2). The hippocampus shows changes in the neuronal layers and structural integrity of the CA regions, reflecting the potential neurodegeneration; while cortical sections exhibit neuronal shrinkage, vacuolation and reduced neuronal density in molecular layers (II-IV) of the cortex. Notably, treatment with suramin (5, 10 and 20 mg/kg, i.p.) and fluoxetine (20 mg/kg, p.o.) effectively ameliorated the CUS induced structural changes in cortical and hippocampal regions of brain. However, suramin (20 mg/kg, i.p.), per se did not show any disruption in molecular layer of cortex and hippocampus compared to the control group.
Effects of various treatments on hippocampus (10X and 40X) in mice subjected to chronic unpredictable stress.
Effects of various treatments on cortex region (10X and 20X) in mice subjected to chronic unpredictable stress.
Discussion
This study investigated the potential of suramin, a cGAS inhibitor, to mitigate CUS-induced neurobehavioral disorders and associated neurobiological changes in mice. Our findings demonstrate that suramin administration effectively attenuated CUS-induced neurobehavioral impairments, as evidenced by improvements in depression, anxiety, and memory. These behavioral improvements were accompanied by significant modulation of key neurobiological parameters, including reduced levels of corticosterone, restoration of neurotransmitter balance (dopamine and serotonin), reduced oxidative stress and neuroinflammatory markers, and modulation of acetylcholinesterase (AChE) activity in brain.
The CUS protocol serves as a fundamental preclinical animal model used to study behavioral neuroscience as an impact of intermittent unpredictable stressors affecting animals during several weeks33. The CUS elicit behavioral indicators which match those observed during anxiety, depression and cognitive impairment according to previous research findings56. The elevated plus maze is used to measure avoidance behavior which serve as an indication of anxiety through heightened fear response and decreased exploratory activity57. The standard sucrose preference test helps to evaluate anhedonia, a core depressive disorder symptom which reflects a diminished capacity to experience pleasure in rewarding experiences58. Furthermore, the evaluation of learned helplessness and despair behavior the essential depressive characteristics are estimated through forced swim tests (FST) and tail suspension tests (TST)58,59. The extent of cognitive impairment, characterized by impaired spatial memory along with learning deficits is estimated by Morris Water Maze (MWM) and the Passive Avoidance (PA) test60. In accordance with earlier research findings our study revealed that mice subjected to CUS exhibited decreased time spent in open arms along with diminished sucrose preference and increase in duration of immobility during FST and TST procedures. Moreover, the significant decrease in TSTQ of MWM accompanied by reduction in latency time of the PA test are significant representation of behavioral changes related to anxiety along with depression and cognitive impairment61.
These behavioral alterations observed in our study, in response to continuous exposure of stress, can be largely attributed to altered levels of corticosterone and neurotransmitters62. Chronic stress sensitizes the hypothalamic-pituitary-adrenal (HPA) axis, disrupting its normal feedback mechanisms and consequently elevating the corticosterone levels63,64. The increase in corticosterone subsequently triggers immune responses and inflammatory reaction cascade through cGAS-STING/ NF-κB pathway65, which in turn, results in degeneration of dopaminergic and serotonergic neurons which play a crucial role in mood and behavior66,67. The decrease in DA and 5HT in striatum, due to neurodegeneration, is the principal aspect underpinning the behavioral changes associated with anxiety and depression68,69. Moreover, the chronic activation of the cGAS-STING/ NF-κB pathway increased corticosterone is reported to cause the upregulation of AChE activity, therefore, influencing cholinergic signaling, associated with cognition and memory impairment, in the hippocampus1,70. The intricate sequence shows the cross talk between cGAS-STING/ NF-κB pathway and CUS-induced changes in neurobehavior. In alignment with previous research reports from other labs, our study demonstrated that the mice subjected to CUS showed a significant increase in corticosterone levels, accompanied by significant decrease in DA and 5HT68. The experimental results of this study71 showed enhanced activity of acetylcholinesterase (AChE). These biochemical changes are the indicators of behavioral abnormalities associated with anxiety, depression and cognitive dysfunction as enlisted by previously indicated behavioral parameters72.
Suramin administration not only enhanced the time spent in open arm of EPM and sucrose preference but also demonstrated significant improvement in despair-like behaviors measured through FST& TST. This enhancement of behavioral parameters demonstrates that suramin shows remarkable promise to treat the core symptomatic areas of anxiety and depression. Moreover, Suramin treatment leads to better spatial learning and memory function according to results from the MWM and Passive Avoidance test. Furthermore, Suramin significantly decreased the acetylcholinesterase (AChE) activity resulting in better cognitive performance. In addition, Suramin administration effectively lowered the elevated levels of corticosterone and simultaneously raising serotonin and dopamine concentrations in mice brain. The diverse therapeutic impact of Suramin demonstrates great potential against neurobehavioral changes induced by CUS due to the improvements in anxiety, depression, cognition, and neurochemical balance.
Neuroinflammation serve as the pivotal element in the etiology of neurobehavioral changes induced by CUS73,74. The inflammatory environment, triggered by the activation of cGAS-STING/ NF-κB pathway through the corticosterone release, substantially affects neuronal survival by various inflammatory mechanisms75,76. Moreover, the persistant inflammation induces the neurobehavioral changes, highlighting the contribution of neuroinflammatory processes in the pathogenesis of stress-related disorders77,78.The current study demonstrates that exposure to CUS causes a significant increase in inflammatory response in the brain, characterized by substantial increase in levels of pro-inflammatory cytokines IL-1β, IL-6, and TNF-α, as well as an elevation in the NF-κB79,80. Notably, treatment with suramin resulted in significant decrease in these inflammatory parameters. This reduction of neuroinflammation, may be principally attributed due to the ability of suramin to inhibit of cGAS-STING/ NF-κB pathway and, thereby, attenuating the subsequent inflammatory and immune cascade.
Oxidative stress, resulting from CUS, occurs due to the imbalance between antioxidant defense system and ROS, leading to mitochondrial dysfunction10,81. Impaired mitochondria, in turn, activates the cGAS-STING/ NF-κB pathway, increasing NF-κB release, which subsequently modulates antioxidant enzymatic activity and enhancing cellular resistance against oxidative damage22,82. In our study, CUS elevated oxidative stress as evident by diminished antioxidant activity and increased lipid peroxidation, as assessed by the measurement of key antioxidants including glutathione, superoxide dismutase, catalase, and TBARS. Remarkably, administration of Suramin effectively reversed the CUS-induced alterations by reducing TBARS and restoring GSH, catalase, and SOD levels, suggesting potent antioxidant mechanism. This effect appears to be mediated through inhibition of cGAS-STING/ NF-κB pathway which mitigates oxidative stress and reinforces the potent antioxidant mechanism underlying it neuroprotective effects in CUS induced neurobehavioral changes.
Chronic unpredictable stress (CUS) leading to release of inflammatory cytokines and decrease in antioxidant activity, contribute significantly to degeneration of neurons in hippocampus and structural alterations in the molecular layers of the cortex83,84.In a similar vein, our study reveals that cGAS-STING/ NF-κB pathway activation induces substantial neuronal loss, changes in the neuronal layers and structural integrity of the CA regions, reflecting the potential neurodegeneration, while cortical sections exhibit neuronal shrinkage, vacuolation and reduced neuronal density in molecular layers of the cortex. Notably, the administration of suramin, through inhibition of the cGAS-STING/ NF-κB pathway, effectively attenuated oxidative stress and neuroinflammation, resulting in marked amelioration of the histological alterations induced by CUS.
Suramin, a polycyclic compound, has recently gained attention for its ability to modulate the cGAS–STING/NF-κB signaling pathway, which plays a critical role in neurological disorders85,86. The cGAS–STING/NF-κB pathway regulates inflammatory and immune responses and has been implicated in several neurological conditions due to its complex and sometimes contradictory functions. Suramin has been shown to reduce cGAS activity by inhibiting its interaction with cytosolic DNA, thereby suppressing the STING-mediated inflammatory and immune response25. In addition, suramin exerts actions beyond inflammation. It modulates neurotrophic factors regulating neuronal survival and differentiation26, reduces oxidative stress, blocks the ATP excitatory system, and provides neuroprotection against brain ischemia28. Suramin also impedes amyloid-β aggregation, thereby attenuating neuroinflammation in Alzheimer’s disease27. Furthermore, it influences microglial activation, which is central to the pathology of multiple sclerosis and other neurodegenerative disorders87.
As highlighted in prior studies, the cGAS–STING/NF-κB pathway represents a crucial driver of chronic neuroinflammation in the CNS88. Upon activation in response to CUS, this pathway initiates a cascade culminating in the phosphorylation of Interferon Regulatory Factor 3 (IRF3), followed by the release of type I interferons and chemokines, and degradation of inhibitor of kappa B (IκB)89. The nuclear translocation of NF-κB subsequently amplifies the release of proinflammatory cytokines, exacerbating neuroinflammation16. Targeting this pathway is therefore a promising strategy to restore immune homeostasis, reduce inflammation, and regulate immunological responses in neurological conditions90. Based on this background, it was logical to further investigate the role of the cGAS–STING/NF-κB pathway and the modulatory effect of suramin to better understand the molecular mechanisms underlying neuroinflammatory responses in neurodegenerative disorders86.
The primary findings of our study indicate that suramin alleviated CUS-induced anxiety, depression, and cognitive impairment, while reducing oxidative stress, inflammation, neurotransmitter dysregulation, and corticosterone levels. These effects were supported by behavioral, histological, and biochemical evidence and were likely mediated through inhibition of the cGAS–STING/NF-κB pathway. The normalization of monoamine neurotransmitters (DA and 5-HT) observed in our study supports suramin’s ability to modulate neurotransmitter systems indirectly via immune regulation25. The associated AChE inhibition further suggests a procognitive mechanism, as cholinergic tone is closely tied to hippocampal function and memory1. This aligns with accumulating literature highlighting the beneficial effects of suramin in mitigating behavioral deficits74,75 linked to neuropsychiatric disorders, reinforcing its potential as a therapeutic candidate for CUS related changes. Collectively, these findings strengthen the pharmacological profile of suramin as a multi-target modulator of neuroinflammation, oxidative stress, and neurotransmission, supporting its therapeutic potential in neuropsychiatric and neurodegenerative disorders.
The pleiotropic nature of suramin, with its interactions across purinergic signaling, neurotrophic factors, and immune receptors, offers exciting possibilities for explaining our findings. While cGAS–STING/NF-κB inhibition emerges as the primary mechanism in our study, other pathways may also contribute in meaningful ways. Future investigations using pathway-selective inhibitors and gene-knockdown models will not only refine mechanistic clarity but also open new avenues for discovery. However, a notable limitation of this study is the absence of an extensive immunohistochemical analysis to ascertain the precise localization and quantification of pivotal markers associated with the cGAS-STING/NF-κB pathway, coupled with the dearth of in-depth gene expression profiling to further elucidate the molecular mechanisms supporting suramin’s neuroprotective effects.
Altogether, we have strong evidence indicating that suramin is effective in the prevention of behavioral, biochemical, and histological sequelae of chronic stress in mice by demonstrating that suramin improves the performance of stress related behavioural, biochemical and histological sequelae outcomes by inhibiting the cGAS-STING/NF-kB signaling cascade. Our results are scientifically significant as they highlight a new mechanism of action of suramin in neuropsychiatric disorders induced by stress. These findings provide the translational significance of CUS model to human mood and cognitive disorders. The clinical interest in cGAS-STING/NF-kB pathway inhibitors, is that our results provide a potentially important insight into the repurposing of suramin as a possible therapy of chronic stress-induced neuroinflammation that leads to behavioral dispositions.
Data availability
Data will be made available on request from the corresponding author (Amarjot Kaur Grewal).
References
Christoffel, D. J., Golden, S. A. & Russo, S. J. Structural and synaptic plasticity in stress-related disorders. Rev. Neurosci. 22, 535–549 (2011).
Liu, R. T. The Microbiome as a novel paradigm in studying stress and mental health. Am. Psychol. 72, 655–667 (2017).
Matosin, N., Cruceanu, C. & Binder, E. B. Preclinical and clinical evidence of DNA methylation changes in response to trauma and chronic stress. Chronic Stress 1, 247054701771076 (2017).
Salam, A. P., Borsini, A. & Zunszain, P. A. Trained innate immunity: A salient factor in the pathogenesis of neuroimmune psychiatric disorders. Mol. Psychiatry 23, 170–176 (2018).
Cenit, M. C., Sanz, Y. & Codoñer-Franch, P. Influence of gut microbiota on neuropsychiatric, disorders. World J. Gastroenterol. 23, 5486–5498 (2017).
O’Connor, T. G., Moynihan, J. A. & Caserta, M. T. Annual research review: The neuroinflammation hypothesis for stress and psychopathology in children – developmental psychoneuroimmunology. J. Child. Psychol. Psychiatry. 55, 615–631 (2014).
McEwen, B. S. Neurobiological and systemic effects of chronic stress. Chronic Stress 1, 2470547017692328 (2014).
Hill, A. S., Sahay, A. & Hen, R. Increasing adult hippocampal neurogenesis is sufficient to reduce anxiety and depression-like behaviors. Neuropsychopharmacology 40, 2368–2378 (2015).
Patki, G., Solanki, N., Atrooz, F., Allam, F. & Salim, S. Depression, anxiety-like behavior and memory impairment are associated with increased oxidative stress and inflammation in a rat model of social stress. Brain Res. 1539, 73–86 (2015).
Guo, C., Sun, L., Chen, X. & Zhang, D. Oxidative stress, mitochondrial damage and neurodegenerative diseases. Neural Regen Res. 8, 2003–2014 (2013).
Zhu, Y., Klomparens, E. A., Guo, S. & Geng, X. Neuroinflammation caused by mental stress: the effect of chronic restraint stress and acute repeated social defeat stress in mice. Neurol. Res. 41, 762–769 (2019).
Zhang, J. et al. Cytosolic DNA-sensing pathway: A novel link between inflammation and neurodegenerative diseases. Sci. China Life Sci. 56, 1058–1062 (2013).
Burdette, D. L. et al. STING is a direct innate immune sensor of Cyclic di-GMP. Nature 478, 515–518 (2011).
Konno, H., Konno, K. & Barber, G. N. Cyclic dinucleotides trigger ULK1 (ATG1) phosphorylation of STING to prevent sustained innate immune signaling. Cell 155, 688–698 (2013).
Sun, L., Wu, J., Du, F., Chen, X. & Chen, Z. J. Cyclic GMP–AMP synthase is a cytosolic DNA sensor that activates the type I interferon pathway. Science 339, 786–791 (2013).
Li, N. et al. STING–IRF3 contributes to lipopolysaccharide-induced cardiac dysfunction, inflammation, apoptosis and pyroptosis by activating NLRP3. Redox Biol. 24, 101215 (2019).
Zhao, Q., Wei, Y., Pandol, S. J., Li, L. & Habtezion, A. STING signaling promotes inflammation in experimental acute pancreatitis. Gastroenterology 154, 1822–1835 (2018).
Mead, A. L., Wong, T. T., Cordeiro, M. F. & Anderson, I. K. Assessment of retinal ganglion cell damage. Invest. Ophthalmol. Vis. Sci. 54, 2241–2251 (2013).
Mackenzie, K. J. et al. cGAS surveillance of micronuclei links genome instability to innate immunity. Nature 548, 461–465 (2016).
Sharma, O., Kaur Grewal, A., Khan, H. & Singh, T. G. Exploring the nexus of cGAS/STING pathway in neurodegenerative terrain: A therapeutic odyssey. Int. Immunopharmacol. 142(Pt B), 113205 (2024).
Newman, L. E. & Shadel, G. S. Pink1/Parkin link inflammation, mitochondrial stress, and neurodegeneration. J. Cell Biol. 217(10), 3327 (2018).
Yu, C. H. et al. TDP-43 triggers mitochondrial DNA release via mPTP to activate cGAS–STING in ALS. Cell 183, 636–649e18 (2020).
McGeary, R. P., Bennett, A. J., Tran, Q. B., Cosgrove, K. L. & Ross, B. P. Suramin: Clinical uses and structure–activity relationships. Mini Rev. Med. Chem. 8, 1384–1394 (2008).
von der Ahe, D. et al. Suramin-induced neurotoxicity: preclinical models and neuroprotective strategies. Molecules 23, 346 (2018).
Wang, M., Sooreshjani, M. A., Mikek, C., Opoku-Temeng, C. & Sintim, H. O. Suramin potently inhibits cGAMP synthase, cGAS, in THP1 cells to modulate IFN-β levels. Future Med. Chem. 10, 1301–1317 (2018).
Feng, C. W., Chen, N. F., Chan, T. F. & Chen, W. F. Therapeutic role of protein tyrosine phosphatase 1B in Parkinson’s disease via antineuroinflammation and neuroprotection in vitro and in vivo. Parkinsons Dis. 2020, 8814236 (2020).
Culibrk, R. A. et al. Impact of Suramin on key pathological features of sporadic alzheimer’s disease-derived forebrain neurons. J. Alzheimers Dis. 98, 301–318 (2024).
Kharlamov, A., Jones, S. C. & Kim, D. K. Suramin reduces infarct volume in a model of focal brain ischemia in rats. Exp. Brain Res. 147, 353–359 (2002).
Uhlmann, S. et al. Early glial cell reactivity in experimental retinal detachment: Effect of Suramin. Invest. Ophthalmol. Vis. Sci. 44, 4114–4122 (2003).
Naviaux, J. C. et al. Antipurinergic therapy corrects the autism-like features in the fragile X (Fmr1 knockout) mouse model. Mol. Autism 6, 1 (2015).
Naviaux, J. C. et al. Reversal of autism-like behaviors and metabolism in adult mice with single-dose antipurinergic therapy. Transl Psychiatry 4(6), e400 (2014).
Xiong, C., Liu, N., Fang, L., Zhuang, S. & Yan, H. Suramin inhibits the development and progression of peritoneal fibrosis. J. Pharmacol. Exp. Ther. 351(2), 373–382 (2014).
Monteiro, S. et al. An efficient chronic unpredictable stress protocol to induce stress-related responses in C57BL/6 mice. Front. Psychiatry. 6, 6 (2015).
Singh, V., Chauhan, G. & Shri, R. Anti-depressant like effects of quercetin 4’-O-glucoside from Allium cepa via regulation of brain oxidative stress and monoamine levels in mice subjected to unpredictable chronic mild stress. Nutr. Neurosci. (2021).
Verma, P., Hellemans, K. G. C., Choi, F. Y., Yu, W. & Weinberg, J. Circadian phase and sex effects on depressive/anxiety-like behaviors and HPA axis responses to acute stress. Physiol. Behav. 99, 276–285 (2010).
Singh, V., Krishan, P., Singh, N., Kumar, A. & Shri, R. Amelioration of ischemia-reperfusion induced functional and biochemical deficit in mice by Ocimum kilimandscharicum leaf extract. Biomed. Pharmacother. 85, 556–563 (2017).
Sohi, S., Shri, R. & Singh, V. Isolation and evaluation of rhoifolin: an anxiolytic flavonoid from Citrus paradisi Macfayden var. Redblush leaves. Nat. Prod. Res. 10, 1–9. (2025).
Vichaya, E. G. et al. Motivational changes that develop in a mouse model of inflammation-induced depression are independent of indoleamine 2,3 dioxygenase. Neuropsychopharmacology 44, 364–371 (2019).
Willner, P. The chronic mild stress (CMS) model of depression: history, evaluation and usage. Neurobiol. Stress. 6, 78–93 (2017).
Kumar, A., Kumar, A., Jaggi, A. S. & Singh, N. Efficacy of cilostazol, a selective phosphodiesterase-3 inhibitor, in a rat model of streptozotocin diabetes-induced vascular dementia. Pharmacol. Biochem. Behav. 135, 20–30 (2015).
Virk, D., Kumar, A., Jaggi, A. S. & Singh, N. Ameliorative role of rolipram, PDE-4 inhibitor, against sodium arsenite-induced vascular dementia in rats. Environ. Sci. Pollut Res. Int. 28, 63250–63262 (2021).
Singh, V., Shri, R., Krishan, P., Singh, I. P. & Shah, P. Isolation and characterization of components responsible for neuroprotective effects of Allium cepa outer scale extract against ischemia reperfusion induced cerebral injury in mice. J. Food Sci. 85, 4009–4017 (2020).
Singh, V. et al. 3-hydroxy-3′,4′,5′-trimethoxyflavone protects rats against lead-induced neurotoxicity via chelation and modulating oxidative stress, inflammatory and monoaminergic systems. J. Funct. Foods. 118, 106278 (2024a).
Kaur, R., Singh, V. & Shri, R. Anti-amnesic effects of ganoderma species: A possible cholinergic and antioxidant mechanism. Biomed. Pharmacother. 92, 1055–1061 (2017).
Pruthi, S., Kaur, K., Singh, V. & Shri, R. Improvement of cognitive function in mice by Citrus reticulata var. Kinnow via modulation of central cholinergic system and oxidative stress. Metab. Brain Dis. 36, 901–910 (2021).
Ellman, G. L., Courtney, K. D., Andres, V. Jr & Featherstone, R. M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 7, 88–95 (1961).
Parul, Mishra, A. et al. Chronic unpredictable stress negatively r]egulates hippocampal neurogenesis and promotes anxious depression-like behavior via upregulating apoptosis and inflammatory signals in adult rats. Brain Res. Bull. 172, 164–179 (2021).
Hadwan, M. H. & Najm, H. A. Data supporting the spectrophotometric method for the estimation of catalase activity. Data Brief. 6, 194 (2016).
Singh, V. et al. 5,7-dihydroxy-3′,4′,5′-trimethoxyflavone mitigates lead induced neurotoxicity in rats via its chelating, antioxidant, anti-inflammatory and monoaminergic properties. Food Chem. Toxicol. 189, 114747 (2024b).
Bhatia, N., Jaggi, A. S., Singh, N., Anand, P. & Dhawan, R. Adaptogenic potential of Curcumin in experimental chronic stress and chronic unpredictable stress-induced memory deficits and alterations in functional homeostasis. J. Nat. Med. 65, 532–543 (2011).
Sood, P., Singh, V. & Shri, R. Morus alba fruit extract and its fractions ameliorate streptozotocin-induced cognitive deficit in mice via modulating oxidative and cholinergic systems. Neurochem Res. 49, 52–65 (2024).
Singh, V., Sharma, S., Dhar, K. L. & Kalia, A. N. Activity guided isolation of anti-inflammatory compound/fraction from root of ricinus communis Linn. Int. J. Pharma Tech. Res. 5, 1142–1149 (2013).
Sood, P., Singh, V. & Shri, R. Morus Alba fruit diet ameliorates cognitive deficit in mouse model of streptozotocin-induced memory impairment. Metab. Brain Dis. 38, 1657–1669 (2023).
Fischer, A. H., Jacobson, K. A., Rose, J. & Zeller, R. Hematoxylin and eosin staining of tissue and cell sections. Cold Spring Harb. Protoc. pdb.prot4986 (2008). (2008).
Slaoui, M. & Fiette, L. Histopathology procedures: From tissue sampling to histopathological evaluation. Methods Mol. Biol. 691, 69–82 (2011).
Bijata, M., Bączyńska, E. & Wlodarczyk, J. A chronic unpredictable stress protocol to model anhedonic and resilient behaviors in C57BL/6J mice. STAR. Protoc. 3, 101659 (2022).
Bondi, C. O., Rodriguez, G., Gould, G. G., Frazer, A. & Morilak, D. A. Chronic unpredictable stress induces a cognitive deficit and anxiety-like behavior in rats that is prevented by chronic antidepressant drug treatment. Neuropsychopharmacology 33, 320–331 (2008).
Qiao, Y. et al. Effect of combined chronic predictable and unpredictable stress on depression-like symptoms in mice. Ann. Transl Med. 8, 942 (2020).
Fang, X. et al. Chronic unpredictable stress induces depression-related behaviors by suppressing AgRP neuron activity. Mol. Psychiatry 26, 2299–2315 (2021).
Gacar, N. et al. Beneficial effects of resveratrol on scopolamine but not mecamylamine induced memory impairment in the passive avoidance and Morris water maze tests in rats. Pharmacol. Biochem. Behav. 99, 316–323 (2011).
Zoladz, P. R. et al. Brief, pre-retrieval stress differentially influences long-term memory depending on sex and corticosteroid response. Brain Cogn. 85, 277–285 (2014).
Smith, S. M. & Vale, W. W. The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin. Neurosci. 8, 383–395 (2006).
Gądek-Michalska, A., Spyrka, J., Rachwalska, P., Tadeusz, J. & Bugajski, J. Influence of chronic stress on brain corticosteroid receptors and HPA axis activity. Pharmacol. Rep. 65, 1163–1175 (2013).
Wheelan, N. et al. Midlife stress alters memory and mood-related behaviors in old age: Role of locally activated glucocorticoids. Psychoneuroendocrinology 89, 13–22 (2018).
Decout, A., Katz, J. D., Venkatraman, S. & Ablasser, A. The cGAS–STING pathway as a therapeutic target in inflammatory diseases. Nat. Rev. Immunol. 21, 548–569 (2021).
Roy, A., Pahan, K. & He, S. The role of the cGAS-STING pathway in alzheimer’s disease pathogenesis. J. Neuroimmune Pharmacol. 15, 382–394 (2020).
Szego, E. M. et al. Constitutively active STING causes neuroinflammation and degeneration of dopaminergic neurons in mice. eLife 11, e81943 (2022).
Ahmad, A., Rasheed, N., Banu, N. & Palit, G. Alterations in monoamine levels and oxidative systems in frontal cortex, striatum, and hippocampus of the rat brain during chronic unpredictable stress. Stress 13, 355–364 (2010).
Nazmi, A. et al. Chronic neurodegeneration induces type I interferon synthesis via STING, shaping microglial phenotype and accelerating disease progression. Glia 67, 1254–1276 (2019).
Mohammadmirzaei, N., Rezayof, A. & Ghasemzadeh, Z. Activation of cannabinoid CB1 receptors in the ventral hippocampus improved stress-induced amnesia in rat. Brain Res. 1646, 219–226 (2016).
Maurer, S. V. & Williams, C. L. The cholinergic system modulates memory and hippocampal plasticity via its interactions with non-neuronal cells. Front. Immunol. 8, 1489 (2017).
Jiang, Y., Botchway, B. O. A., Hu, Z. & Fang, M. Overexpression of SIRT1 inhibits corticosterone-induced autophagy. Neuroscience 411, 11–22 (2019).
Hassamal, S. Chronic stress, neuroinflammation, and depression: An overview of pathophysiological mechanisms and emerging anti-inflammatories. Front. Psychiatry 14, 1130989 (2023).
Gao, D. Tetrahydroxy stilbene glycoside ameliorates neuroinflammation for alzheimer’s disease via cGAS–STING. Alzheimers Dement. 20(Suppl. 1), e084633 (2025).
Song, N., Dvoriantchikova, G., Rouger, M. & Falck, J. Central role of NLRP3 inflammasome activation in neuroinflammation and neurodegeneration. Nat. Rev. Immunol. 17, 138–144 (2017).
Gulen, M. F., Xue, W., Lison-Prado, S. & Rolot, M. Blocking the cGAS–STING pathway suppresses inflammation and protects against neuroinflammation and cognitive decline. Cell. Rep. 43, 109077 (2023).
Cohen, S. et al. Chronic stress, glucocorticoid receptor resistance, inflammation, and disease risk. Proc. Natl. Acad. Sci. USA 109, 5995–5999 (2012).
Ravi, M., Miller, A. H. & Michopoulos, V. The immunology of stress and the impact of inflammation on the brain and behavior. BJPsych Adv. 27, 158–165 (2021).
Vallabhapurapu, S. & Karin, M. Regulation and function of NF-κB transcription factors in the immune system. Annu. Rev. Immunol. 27, 693–733 (2009).
Hou, F. et al. MAVS forms functional prion-like aggregates to activate and propagate antiviral innate immune response. Cell 146, 448–461 (2011).
Forrester, S. J., Kikuchi, D. S., Hernandes, M. S., Xu, Q. & Griendling, K. K. Reactive oxygen species in metabolic and inflammatory signaling. Circ. Res. 122, 877–902 (2018).
Kim, H. et al. Targeting the cGAS–STING pathway: a promising approach to mitigate oxidative stress and neuroinflammation in neurodegenerative disorders. Antioxidants 12, 189 (2023).
Lamtai, M. et al. Chronic copper exposure leads to hippocampus oxidative stress and impaired learning and memory in male and female rats. Toxicol. Res. 36, 359–366 (2020).
Qiao, H. et al. Dendritic spines in depression: What we learned from animal models. Neural Plast. 2016, 8056370 (2016).
Brun, R., Blum, J., Chappuis, F. & Burri, C. Human African trypanosomiasis. Lancet 375, 148–159 (2010).
Wang, X. et al. Molecular mechanisms of cGAS–STING signaling in immunostimulation and neuroprotection. Front. Immunol. 12, 673109 (2021).
Illes, P., Rubini, P., Ulrich, H., Zhao, Y. & Tang, Y. Regulation of microglial functions by purinergic mechanisms in the healthy and diseased CNS. Cells 9, 1108 (2020).
Jauhari, A., Dey, D. & Sharma, H. Inflammatory responses in huntington’s disease pathogenesis: the role of cGAS–STING–IRF3 signaling. J. Neuroinflamm. 17, 273 (2020).
Hinkle, J. T., Seger, M. & Sharma, N. Modulation of the cGAS–STING signaling pathway in parkinson’s disease. Front. Immunol. 13, 622429 (2022).
Izquierdo, E., Quivy, V., Martínez, A. C. & Loginov, P. D. cGAS–STING regulation of neuroinflammation and neurodegeneration. Front. Cell. Infect. Microbiol. 13, 797742 (2023).
Acknowledgements
The authors acknowledge and appreciate the Ongoing Research Funding Program (ORF-2025-783), King Saud University, Riyadh, Saudi Arabia. The authors acknowledge and extend their appreciation to Chitkara University, Punjab, India for providing resources for completion of this study.
Funding
This research was supported by the Ongoing Research Funding Program (ORF-2025-783) at King Saud University in Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
O.S.: Investigation; A.K.G.: Conceptualization, Supervision and Writing—original draft; A.K.: Validation; H.K.: Formal analysis; V.S.: Methodology and Writing—review & editing; T.G.S.: Data curation and Resources; T.S.: Formal Analysis; S.F.A. Funding acquisition; H.A.A..-M.: Funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The experimental protocol for the current study was approved by Institutional Animals Ethics Committee (IAEC) of Chitkara University, Punjab, India (protocol number IAEC/CCP/24/01/PR-02). Animals were duly taken care by strictly following the guidelines issued by CCSEA (Committee for control and supervision of experimentation on animals), a committee governed by Ministry of Fisheries, Animal Husbandry and Dairying, Department of Animal Husbandry and Dairying, Government of India.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sharma, O., Grewal, A.K., Kumar, A. et al. Suramin protects against chronic stress-induced neurobehavioral deficits via cGAS–STING/NF-κB suppression. Sci Rep 15, 42810 (2025). https://doi.org/10.1038/s41598-025-27660-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-27660-z