Abstract
Primary biliary cholangitis (PBC) is an autoimmune liver disease in which macrophages play a critical role in disease progression. Vitamin D3 (VitD3) exerts immunomodulatory effects and can regulate the phenotype and function of macrophages. However, the role of VitD3 in modulating macrophage polarization during PBC progression remains unclear. In this study, the relationship between VitD3 levels and macrophage polarization in PBC patients was evaluated by bioinformatics as well as clinical validation, and the underlying mechanisms were investigated using a mouse model of PBC and an in vitro cellular model. Bioinformatics analysis of vitamin D-related genes involved in PBC disease. The number of M1-type macrophages is increased in the liver of PBC patients. After calcitriol treatment, the serum 25(OH)D3 level of PBC patients was increased, liver injury was alleviated, and the number of M1 macrophages in the liver was decreased. In the liver of PBC mice, VitD3 treatment significantly reduced the infiltration of inflammatory cells, especially M1 macrophages, and reduced the release of pro-inflammatory cytokines (TNF-α, IL-6, IL-1β), thereby alleviating liver injury. In vitro and in vivo experiments showed that VitD3 inhibited the expression of TLR4/NF-κB signaling pathway-related proteins TLR4 and MyD88 and the phosphorylation of p65 and IκB. In conclusion, VitD3 may attenuate M1 macrophage polarization through the TLR4/NF-κB signaling pathway, thereby ameliorating the liver inflammatory injury in PBC. This study provides novel insights into the immunomodulatory effects of VitD3 in PBC and highlights its potential therapeutic implications.
Similar content being viewed by others
Introduction
Primary biliary cholangitis (PBC) is a chronic, cholestatic autoimmune liver disease characterized by chronic, non-suppurative, destructive bile duct injury in the interlobular ducts1. Mitochondrial autoantigens are exposed after destruction of the intrahepatic bile duct epithelial cells2 and subsequently activate immune cells to produce a proinflammatory response, thereby accelerating liver inflammation and fibrosis in PBC3,4,5. Macrophages were reported to play a crucial role in liver homeostasis6. The liver macrophages are primarily increased through the proliferation of resident macrophages and the recruitment of circulating bone marrow-derived monocytes to the injured liver7,8. Macrophages possess a high degree of plasticity and can be polarized into two main phenotypes: classically activated (M1) macrophages and alternatively activated (M2) macrophages9. M1 macrophage polarization is characterized by the promotion of inflammatory responses10, while M2 macrophage polarization is involved in anti-inflammation, immune regulation, tissue remodeling, and fibrosis11,12. Several studies have indicated that M1/M2 macrophage polarization displays an abnormal imbalance in autoimmune diseases13,14. Dynamic changes in these two macrophage phenotypes are likely to be associated with liver inflammation and fibrosis. As an important immune cell component of the liver, macrophages are believed to play a critical role in disease progression and tissue damage. However, the function of macrophage polarization in PBC remains unclear.
Vitamin D3 (VitD3) is a fat-soluble vitamin15, is essential for the metabolism of phosphorus and calcium, the maintenance of normal calcium levels, and the promotion of healthy bone development16. Recently, VitD3 has been reported to exert immunomodulatory effects17. VitD3 deficiency is prevalent in many autoimmune diseases18,19,20,21. In our previous study, we found that VitD3 supplementation could effectively delay the progression of PBC22. VitD3 can promote the differentiation of monocytes into macrophages while also regulating macrophage activation and function. Arai et al.23. showed that the serum 25(OH)D3 level and VDR gene expression were significantly and independently associated with the severity of liver fibrosis. In non-alcoholic fatty liver disease, VitD3 can inhibit the polarization of M1 macrophages to reduce lipid accumulation in hepatocytes24. However, it remains unclear whether macrophage polarization in the liver of PBC patients is regulated by VitD3.
While VitD3 is currently employed as an adjuvant therapeutic agent in PBC to mitigate PBC-associated osteoporosis, its immunomodulatory effects on the disease pathogenesis remain to be fully elucidated. This study aimed to clarify the role of VitD3 in modulating macrophage polarization within the hepatic microenvironment of PBC and to elucidate the underlying mechanisms, thereby providing novel insights into its immunomodulatory properties and highlighting its potential implications for therapeutic strategies.
Materials and methods
In Silico prediction
The target genes of VitD and PBC were obtained from GeneCards database. The intersection of these genes, which are common targets for VitD and PBC, is shown in a Venn diagram drawn by the jvenn tool. Then, the PBC mRNA expression chip GSE79850 (including 16 PBC liver tissue samples and 8 normal liver tissue samples) was retrieved from Gene expression Omnibus (GEO) database. Based on this chip, the “limma” package in R language was used to identify the differentially expressed genes (DEGs) related to diabetes. To characterize their biological functions, we performed comprehensive enrichment analyses using Gene Ontology (GO), and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway databases. These analyses were conducted in R employing the “clusterProfiler” package for functional enrichment, supported by “org.Hs.eg.db” for annotation and “ggplot2” for visualization, with additional packages (“enrichplot”, “pathview”, “DOSE”) enhancing analytical capabilities. We used CIBERSORT to analyze the populations of macrophage cells from the GSE79850 dataset.
Patient samples
Blood samples were collected from 73 PBC patients at the Affiliated Hospital of Qingdao University. All patients were treated with ursodeoxycholic acid (UDCA), of which 44 patients were treated with vitamin D supplementation with calcitriol, and 29 patients were not treated with calcitriol. Blood samples were also collected from 40 volunteers as healthy controls, who had normal ranges of routine physical examination (fasting blood glucose, liver and kidney function tests, blood lipids, urine, and stools) and without any acute or chronic liver diseases during routine checkup at the same hospital. Research involving human subjects has been approved by the author’s Institutional Review Board (QYFYWZLL28980). Written informed consent was obtained from all participants. Human participants’ names and other HIPAA identifiers were removed from all sections of the manuscript, including supplementary information. The clinical data research was conducted in accordance with the Declaration of Helsinki. Serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), gamma-glutaryltransferase (GGT), total bilirubin (TBIL) and direct bilirubin (DBIL) in all subjects were detected by Hitachi 7600 series automatic biochemical analyzer. Peripheral blood macrophages were detected by flow cytometry. Serum 25(OH)D3 levels were quantified by chemiluminescence immunoassay (ARCHITECT 25-OH vitamin D kit, Abbott).
A mouse model of PBC
Female C57BL/6J wild-type mice aged 5–6 weeks and weighing 22–25 g were selected for PBC model construction. All animals received a standard laboratory diet. After a week of acclimatization, weight-matched mice were randomly assigned to three groups: (1) PBC group (n = 6); (2) Control group (n = 6); and (3) calcitriol (VitD3) treatment group (n = 6). All animals were housed under a 12/12-h light-dark cycle at room temperature (21 ± 2 °C). PBC was chemically induced in mice according to the protocol of Xu. et al.25. Briefly, PBC mice were intraperitoneally injected with a mixture of 2-OA-BSA and complete Freund’s adjuvant (CFA) at a concentration of 1 mg/mL on the first day of the first week, and then a mixture of 2OA-BSA and incomplete Freund’s adjuvant (IFA) at a concentration of 1 mg/mL were intraperitoneally injected every two weeks from the third week. From the third day of the first injection of 2-OA-BSA and CFA, the Polyinosinic: polycytidylic acid (PolyI: C, 5 mg/kg) was injected every three days until the end of modeling at week 8. The vitamin D treatment group was given 32.5 IU/kg calcitriol intragastric administration, divided into three times a week. Mice in the healthy control group were fed normally without any treatment. The weight of the mice during euthanasia was approximately 21–25 g. Histopathological examination, serum AMA antibody detection and liver function test were used to determine whether the mice were successfully established. AMA antibody in serum of mice was detected by indirect immunofluorescence (EUROIMMUN, China). Specifically, animal experiments were performed in compliance with the ARRIVE guidelines (https://arriveguidelines.org).
Euthanasia and tissue collection
Mice were euthanized by overdose of inhaled isoflurane. Briefly, animals were placed in an induction chamber and exposed to 4–5% isoflurane (in oxygen) until loss of consciousness. Deep anesthesia was confirmed by the absence of pedal reflex. Mice were then maintained under isoflurane anesthesia for an additional 2 min until cessation of respiration and cardiac arrest were verified.
Following euthanasia, a midline laparotomy was performed promptly. The entire liver was carefully excised by dissecting the connecting ligaments and vasculature. Liver lobes were separated and processed accordingly: portions for RNA/protein analysis were snap-frozen in liquid nitrogen and stored at -80 °C; samples for histology were fixed in 4% paraformaldehyde for 24 h at 4 °C before paraffin embedding.
Measurement of serum parameters in mice
To evaluate hepatic function, serum was collected from whole blood via retro-orbital bleeding after a 6-hour fast. The blood was allowed to clot at room temperature and subsequently centrifuged at 3,000 rpm for 15 min to separate the serum. The serum samples were then stored under refrigeration prior to analysis. Serum levels of ALT, AST, ALP, GGT, TBIL, and DBIL were measured using a fully automated biochemical analyzer (Hitachi 7100, Japan). All assays were performed in strict accordance with the manufacturers’ protocols. The serum levels of ALT, AST, and GGT were measured using the continuous monitoring method, while ALP was determined with the p-nitrophenyl phosphate substrate-AMP buffer method. TBIL and DBIL levels were quantified by the vanadate oxidation method.
Cell culture and stimulation
Human monocyte cell line (THP-1) cells were brought from Procell Life Science&Technology Co., Ltd. (Procell, Wuhan, China) and cultured with RPMI 1640 medium (Procell, Wuhan, China) containing 10% fetal bovine serum (FBS), 1% penicillin-streptomycin (P/S) and 0.05 nM β-mercaptoethanol. THP-1 cells were seeded in 6-well plates at a density of 1 × 106 cells per well. After inducing THP-1 cells with 100 ng/mL phorbol-12-myristate-13-acetate (PMA) (Yeasen, China) for 24 h, most of the cells adhered to the wall, which can be considered as polarized M0 macrophages. M0 macrophages were stimulated with LPS (100 ng/ml, Yeasen, China) and IFN-γ (20 ng/ml, Yeasen, China) to induce their polarization to M1 macrophages. Five concentrations (0 nM, 10nM, 50nM, 100nM and 300nM) of VitD3 were used to pretreated for 24 h before stimulated with LPS/IFN-γ.
Cell viability assay with CCK8
Cell Counting Kit-8 (CCK-8, Yeasen, China) was used to analyze the cell viability. THP-1 cells were seeded in 96-well plates with a density of 1.0 × 106 cells per well and stimulated with PMA to adhere for 24 h. Then different concentrations of VitD3 (0 M, 10nM, 50nM, 100nM and 300nM) were added to wells for 24 h. Cells were incubated with 10µL CCK-8 reagent per well for 2 h. The absorbance was measured at 450 nm with an automated microplate reader.
H&E staining
PBC, control and VitD3 treatment liver tissue from mice were removed and fixed in 10% formalin for 24 h at room temperature, dehydrated, transparentized and embedded in paraffin before sectioning into 4 μm-thick slices. Then, the sections were stained with H&E to evaluate biliary inflammation and cellular infiltration.
Immunohistochemistry
Liver tissue was obtained after mice were sacrificed. Formalin-fixed, paraffin-embedded tissue sections were deparaffinized and rehydrated. Blocking of endogenous avidin/biotin, and antigen retrieval was performed by boiling in 0.01 M sodium citrate for 24 min. The sections were then blocked with 5% BSA for 30 min, and then incubated with primary antibodies: anti-F4/80 (1:3000, Proteintech), anti-MCP-1 (1:500, Abcam) and anti-VDR (1:200, Affinity) at 4℃ overnight. Following washing, tissues sections were incubated with horseradish peroxidase conjugated anti-mouse or rabbit anti-bodies (CST) for 30 min at 37℃. Specific staining was detected by 3, 3- diaminobenzidine and examined by light microscopy. Tissue sections were evaluated using Image J.
Immunofluorescence
Liver sections were treated with 4% paraformaldehyde for 15 min and 0.2% triton for 10 min, and were then blocked with 5% goat serum for 60 min at room temperature. Tissue sections were then incubated with primary antibodies at 4℃ for overnight. The antibodies used were as follows: iNOS (1:200, Abcam), Arg-1 (1:200, Abcam), TLR4 (1:200, Proteintech). Subsequently, incubation with Alexa Fluor 488-labelled goat anti-mouse IgG and Alexa Fluor 647-labelled goat anti-rabbit IgG (Abcam, USA) was performed for 1 h at 37 °C in the dark. Nuclei were stained with 1 µg/mL DAPI. The images were captured using a confocal fluorescence microscope (Olympus, Tokyo, Japan). Image J software was applied for quantitative analysis.
Flow cytometry
THP-1 cells were seeded in 6-well plates at a density of 5 × 10⁵ cells/well and stimulated with 100 ng/mL PMA for 24 h to induce differentiation into M0 macrophages. Then they were treated with LPS (100 ng/mL) + IFN-γ (20 ng/mL) combined with different concentrations of vitamin D (0, 10, 50, 100 nM) for 24 h. After 0.25% trypsin digestion, cells were collected by cell scraping and centrifuged at 1000 rpm for 5 min to obtain cell precipitates. After washing with PBS, cells were first incubated with Fc receptor blocker at room temperature for 10 min to block nonspecific binding, followed by incubation with FITC-labeled anti-human CD11b antibody and PE-labeled anti-human CD80 antibody (Biolegend, San Diego, CA) in the dark for 30 min, respectively. After antibody staining, cells were washed twice with PBS, resuspended in 300 µL PBS, and assayed using an Agilent flow cytometer. Streaming data were analyzed with the use of NovoExpress software.
RNA extraction and quantitative PCR
TRIzol reagent (Yeasen, Shanghai, China) isolated total RNA from cells. Hifair® II 1st Strand cDNA Synthesis SuperMix (Yeasen, Shanghai, China) was used to synthesize complementary DNA, and Hieff® qPCR SYBR Green Master Mix (Low Rox Plus) (Yeasen, Shanghai, China) was used for qPCR. PCR amplification was conducted in triplicate for each sample, and the expression of target genes was normalized to GAPDH. Relative expression was determined using the 2−△△Ctmethod. Primer sequences were obtained from PrimerBank (https://pga.mgh.harvard.edu/primerbank/) and synthesized at SangonBiotechCo. Ltd. (Shanghai, China) (Table 1).
Western blot analysis
Total protein extracted from cells was used RIPA buffer (Yeasen, Shanghai, China). Proteins were separated by precast protein plus gel (4–20% Hepes-Tris) and then transferred to a PVDF membrane (Yeasen, China). At room temperature, the membrane was blocked with a blocking buffer (Beyotime Biotech, China) for 1 h. The PVDF membrane was subsequently cut and respectively incubated with the following antibodies at 4°Covernight: iNOS(1:2000; Abcam), VDR(1:1000; Abcam), TLR4(1:2000; Proteintech), MyD88(1:2000; Proteintech), NF-κBp65(1:1000; CST), p-p65(1:1000; CST), Iκb(1:1000; Affinity), p-Iκb(1:1000; Affinity),β-actin(1:10000; Abcam), GAPDH (1:30000; Proteintech). Subsequently, the bands were incubated with the following secondary antibodies for 1 h at 25 °C (1:20000; Abcam).
Statistical analysis
All data were presented as mean ± standard deviation. All analyses were performed with GraphPad Prism 8 (GraphPad, San Diego, CA). Each experiment was repeated independently for a minimum of three times. Shapiro-Wilk tests were used to assess the normality assumption of the data distribution. T-test was used for unpaired continuous variable data that conformed to a normal distribution. One-way ANOVA analysis determined the statistical difference between multiple groups with one variable. The statistical analysis data were considered to be statistically significant (p < 0.05).
Results
Bioinformatics analysis predicted VD through association with PBC disease
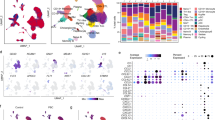
Through bioinformatics analysis, we obtained 9091 VitD-related genes and 2334 PBC-related genes from the GeneCards database. At the same time, 92 differentially expressed genes were found in PBC-related mRNA chip GSE79850. The three gene sets overlapped to obtain 60 candidate genes (Fig. 1A). We next performed functional annotation and enrichment analysis of overlapping genes. In addition, KEGG analysis showed that VitD and PBC-related genes were involved in signaling pathways. We found that these genes were significantly enriched in NF-κB pathway, Toll-like receptor signaling pathway, PI3K-Akt signaling pathway, TNF signaling pathway as well as JAK-STAT3 pathway (Fig. 1B). Gene Ontology (GO) analysis was performed on intersection genes, which revealed functional enrichment for biological process (BP), cellular components (CC), and molecular function (MF) (17) (Fig. 1C).
60 key genes and their functional analysis. The bubble shows significant items according to the P-value. (A) Venn diagram demonstrated the intersection set of VitD, PBC and DEGs. (B) KEGG pathway analysis. (C) The GO terms involved three components: biological process (BP), cellular component (CC), and molecular function (MF).
Analysis of macrophage infiltration in the liver of PBC patients
Immune infiltration in the liver of patients with PBC was characterized using the CIBERSORT algorithm on the GSE79850 dataset. The analysis revealed the presence of three distinct macrophage subtypes: M0, M1, and M2. Their infiltration levels were heterogeneous. Specifically, a significant increase was observed in M1 macrophage infiltration in the PBC liver (Fig. 2B). In contrast, the infiltration levels of M0 and M2 macrophages were not significantly elevated (Fig. 2A, C).
The infiltration of macrophages in the liver was assessed and compared between patients with PBC and healthy controls.
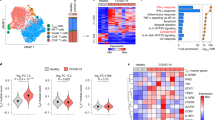
Calcitriol treatment can effectively reduce liver damage in patients with PBC
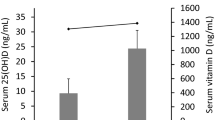
The demographic data, clinical and laboratory parameters of 40 Health controls, 44 Calcitriol - untreated PBC patients and 29 Calcitriol - untreated PBC patients were shown in Table 2. The serum levels of ALT (p < 0.05), AST (p < 0.01), ALP (p < 0.05), and GGT (p < 0.01) were significantly lower in the patients treated with calcitriol than in the patients not treated with calcitriol (Table 2; Fig. 3). The contents of TBIL and DBIL were not statistically different among the three groups. The serum 25(OH)D3 level of PBC patients was significantly lower than that of healthy controls (p < 0.0001) (Table 2; Fig. 3). The level of peripheral blood 25(OH)D3 in PBC patients treated with calcitriol was significantly higher than that in patients without calcitriol treatment (p < 0.0001) (Table 2; Fig. 3).
Serum levels of biochemical parameters and 25(OH)D3 were compared across the three groups: healthy controls (HC), calcitriol-treated PBC patients (PCT), and calcitriol-untreated PBC patients (PCN). *p<0.05; **p<0.01; ***p<0.001; ****p<0.0001.
Calcitriol treatment alleviates hepatic inflammation and cholangitis in patients with PBC
To test this hypothesis, we analyzed liver tissues from 4 PBC patients compared with 4 PBC patients treated with calcitriol (Fig. 4). HE staining revealed prominent inflammatory cell infiltration in portal areas, predominantly surrounding small bile ducts. Immunohistochemical analysis showed an increase in CD68+ macrophages and CK19+ cholangiocytes, along with bile duct proliferation, luminal narrowing, and ductular reaction. Following calcitriol treatment, we observed a significant reduction in periductal inflammatory cells and CD68+ macrophages, as well as decreased CK19+ cell density and attenuated bile ductular reaction.
Histological analysis via HE staining revealed periductal inflammatory cell infiltration in PBC patients. Immunohistochemistry demonstrated an elevated number of CD68+ (macrophages) and CK19+ (cholangiocytes) cells. Notably, calcitriol-treated PBC patients exhibited reduced inflammatory cell infiltration on HE staining, along with decreased CD68+ and CK19+ cell counts. Scale bar = 100 μm.
VitD3 reduces liver damage and liver inflammation in PBC mice livers
The flow chart of mouse model construction is shown in Fig. 5A. After 10 weeks, serum samples and liver tissues were collected. Liver injury and liver tissue inflammation were evaluated by serum liver function related indicators and HE histological staining. Compared with the healthy mice, the serum AMA of PBC group mice was positive, and ALT, AST, ALP, GGT, TBIL and DBIL levels were significantly increased (p<0.05) (Fig. 5B,C), indicating that the mouse PBC model was successfully established. Moreover, VitD3 treatment significantly reduced ALT, TBIL and DBIL levels (p<0.05). A typical picture of the appearance of the liver is shown in Fig. 5D. The liver surface of healthy mice was smooth, ruddy and soft in texture. In the PBC group, white inlay appeared on the liver surface, with swollen shape and dim color. The volume of white impaction on the liver surface was significantly reduced in VitD3 treatment group. HE results showed inflammatory cell infiltration in the portal area of the liver. Compared with healthy mice, PBC mice showed a large number of inflammatory cells infiltration in the portal areas of the liver (p<0.05) (Fig. 5E). Compared with mice without VitD3 treatment, VitD3 treatment can significantly reduce the infiltration of inflammatory cells in the liver portal area of PBC mice (p<0.05) (Fig. 5E).
VitD3 reduces liver damage and liver inflammation in PBC mice livers. (A) Workflow for inducing PBC mouse model; (B) The levels of serum ALT, AST, TBIL, DBIL, ALP, GGT and 25(OH)D3 in each group, n = 6; (C) Expression and quantification of AMA antibodies in peripheral blood of the three groups of mice, Scale bar = 10 μm, n = 3; (D) Representative photographs of mice liver in each group; (E) Representative micrographs of mice livers in three subgroups after HE staining, n = 6, Scale bar = 50 μm. Data are presented as mean ± SEM; *p<0.05; **p<0.01; ***p<0.001; ****p<0.0001.
VitD3 inhibits macrophage infiltration and reduce the release of macrophage-associated inflammatory factors in PBC mice livers
First, we investigated the macrophage infiltration in PBC livers. F4/80 is a marker of macrophage26. The F4/80 positive cells in PBC livers were significantly increased when compared with the control group (p<0.05) (Fig. 6A). VitD3 treatment significantly reduced F4/80 positive cells infiltration (p<0.05) (Fig. 6A). Next, there are enhanced levels of MCP-1 in PBC mice livers (p<0.05) (Fig. 6B), which are mainly released by inflammatory macrophages to recruit other inflammatory cells. VitD3 treatment reduced the expression of MCP-1 (p<0.05) (Fig. 6B). A large accumulation of VDR expression was observed around inflammatory cells near the bile ducts in PBC mice. After intervention with VitD3, compared with mice in the PBC group, the expression of VDR near inflammatory cells around the bile ducts in the liver decreased (p<0.05) (Fig. 6C). Furthermore, enhanced levels of secretory proinflammatory factors, including TNF-α, IL-1β, IL-6, IL-10 and TGF-β, in PBC livers (p<0.05) (Fig. 6D). Moreover, VitD3 treatment also decreased the expression of TNF-α, IL-1β and IL-6 (p<0.05) (Fig. 6D).
Macrophage infiltration and expression of related inflammatory cytokines in the liver of control, PBC and VitD3 treatment mice. (A) Immunohistochemistry staining of F4/80 in liver tissues, Scale bar = 50 μm, n = 6; (B) Immunohistochemistry staining of MCP-1 in liver tissues, Scale bar = 50 μm, n = 6; (C) Immunohistochemistry staining of VDR in liver tissues, Scale bar = 50 μm, n = 6; (D) The mRNA expression of TNF-α, IL-1β, IL-6, IL-10 and TGF-β were determined by RT-qPCR in PBC liver treated with or without VitD3, n = 6; Data are presented as mean ± SEM; *p<0.05; **p<0.01; ***p<0.001; ****p<0.0001.
VitD3 supplementation suppressed M1 macrophage infiltration in liver of PBC mice
Given VitD3 reduces inflammation and liver damage in PBC mice livers by inhibiting the infiltration of macrophages and the release of macrophage-associated inflammatory factors. This study investigated the effects of VitD3 on inflammatory macrophage polarization. The results showed that iNOS protein was highly expressed around the bile duct in PBC mice (p<0.05) (Fig. 7A). VitD3 markedly reduced the expression of iNOS (p<0.05) (Fig. 7A,B), a specific cell surface marker of inflammatory macrophages27. Moreover, the protein expression of Arg-1, a marker of M2 macrophages, was also examined. However, there was no significant difference among the three groups (p<0.05) (Fig. 7B). The untreated group exhibited substantial ductular reaction, characterized by solid cord-like CK19+ structures with narrow or absent lumens. In contrast, vitamin D intervention led to a marked improvement, restoring more regular CK19 expression and biliary architecture (Fig. 7C).
The liver macrophages of PBC mice are mainly polarized to M1 macrophage. (A) The expression of iNOS (red) and CK19 (green) in livers were examined by Immunofluorescence staining, Scale bar = 50 μm; (B) The expression of Arg-1 (red) and CK19 (green) in livers were examined by Immunofluorescence staining, Scale bar = 50 μm. (C) Biliary responses in the liver of PBC mice, Scale bar = 50 μm. *p<0.05; **p<0.01.
VitD3 inhibits the polarization of M1 macrophages by inhibiting the TLR4/NF-κB signaling in PBC mice liver
The TLR4/NF-κB signaling pathway mainly regulates the polarization of M1 macrophages, and its mediated macrophage polarization plays an important role in anti-inflammatory pathways. We performed fluorescence co-localization of macrophage M1 macrophage markers iNOS and TLR4 proteins in mouse liver. As shown in Fig. 8A, the expression of TLR4 in the liver of PBC mice was increased and co-localized with the expression of iNOS (p<0.05). The expression of TLR4 in the liver of mice with VitD3 treatment was lower than that in the without treatment group, and its co-expression with iNOS was reduced (p<0.05)( Fig. 8B). Similar results were observed for protein expression of TLR4 signaling pathway in the liver. The expression of TLR4, MyD88, pp65/p65 and pIκb/Iκb protein levels in the liver of PBC mice was significantly higher than that of the control group (p<0.05) (Fig. 8C, D). The expression of TLR4, MyD88, pp65/p65 and pIκb/Iκb protein decreased after VitD3 treatment (p<0.05) (Fig. 8C, D).
VitD3 suppressed the differentiation of macrophages to M1 through TLR4/NF-κB signaling pathway. (A,B) The expression of iNOS (red) and TLR4 (green) in livers were examined by Immunofluorescence staining, Scale bar = 50 μm, n = 6; Quantification assessment of iNOS and TLR4 fluorescent expression, n = 6; (C,D) The total proteins of the livers were extracted from liver tissue, and the expression levels of iNOS, TLR4, MyD88, p65, pp65, Iκb and p-Iκb were analyzed by Western blot, n = 3. *p<0.05; **p<0.01; ***p<0.001;****p<0.0001.
Cell viability assay with CCK-8
To further verify the effect of VitD3 on macrophage polarization, an in vitro macrophage polarization induction model was established in this study. Because THP-1 are now commonly used to study the function, mechanisms, and signaling pathways of macrophages28,29, herein, human THP-1 cells was chosen as research objects. For human THP-1 cells, PMA was applied to stimulate M0 differentiation, after which LPS and IFN-γ were used to induce M1-like macrophages (Fig. 9A). Prior to LPS/IFN-γ induction, M0 macrophages was protected with different concentrations of VitD3. THP-1 cells were round and transparent suspension cells. After 24 h of PMA stimulation, they adhered to the wall and were round or oval cells. When induced by LPS/IFN-γ for 6 h, the cells showed “fried egg” shape or irregular shape, pseudopodia appeared, and the surface was rough (Fig. 9B). The number of “fried egg” -like cells decreased under the microscope when 100nM VitD3 was used. The cells were fragmented and floating when pretreated with 300nM VitD3. As shown in Fig. 9C, when M0 macrophage cells were treated with different VitD3 concentrations from 10, 50, 100 to 300nM, the cell viability was almost 100% at VitD3 concentrations in the range of 0-100nM. The VitD3 concentration at 300nM had obvious effects on cell viability (p < 0.01). Based on the preliminary results, the experimental concentrations of VitD3 were finally determined to use 0-100nM.
An in vitro cell induction model was established. (A) Workflow for induction of M1 macrophages in vitro; (B) Morphological changes of macrophages treated with different conditions, Scale bar = 400 μm. (C) Effects of VitD3 on THP-1 cells viability. THP-1 cells were treated with different concentrations of VitD3 (0, 10, 50, 100, 300 nM), and cell viability was assessed by CCK-8 assay. Data are presented as mean ± SEM. *p<0.05.
VitD3 inhibits the polarization of M1 macrophages through the TLR4/NF-κB signaling pathway in vitro
For identification of macrophage differentiation in vitro, the CD11b+CD80+ cells were defined as M1 macrophages by flow cytometry. The results showed that the proportion of M1 macrophages was increased after induced by LPS/IFN-γ (Fig. 10A). Treatment of VitD3 could reduce the proportion of M1 macrophages, indicating that VitD3 may inhibit the polarization of M0 cells to M1 macrophages. As shown in Fig. 10B, after the stimulation of LPS/IFN-γ, the expression of inflammatory cytokines TNF-α, IL-6 and IL-1β were increased. Furthermore, VitD3 treatment significantly reversed the LPS/IFN-γ induced expression of these inflammatory cytokines in THP-1 cells (Fig. 10B). This phenomenon was particularly observed with 50 and 100nM concentration interventions. Correspondingly, the expressions of TLR4/NF-κB signaling pathway-related proteins were up-regulated in THP-1 cells treated with LPS/IFN-γ (Fig. 10C). VitD3 treatment significantly reduced LPS/IFN-γ induced TLR4, NF-κB, p-p65 and p-IκB protein expression. The level of these proteins showed similar changes as their inflammatory cytokines (Fig. 10D).
VitD3 inhibits the polarization of M1 macrophages through the TLR4/NF-κB signaling pathway in vitro. (A) Flow cytometry was used to detect the proportion of M1 macrophages under different conditions. CD11+CD80+ cells were defined as M1 macrophages. (B) RT-qPCR was used to detect the expression of pro-inflammatory cytokines in each group. (C) The typical Western blot for iNOS, TLR4, MyD88, p65, pp65, Iκb, p-Iκb and VDR from THP-1 cells treated with LPS and different concentrations of VitD3. (D) Quantitative analysis of iNOS, TLR4, MyD88, pp65/p65, p-Iκb/Iκb and VDR protein expression. *p<0.05; **p<0.01; ***p<0.001;****p<0.0001.
Discussion.
The association between vitamin D deficiency and autoimmune diseases has been well-established in multiple studies30,31,32. Our previous work further demonstrated a significant correlation between vitamin D deficiency and disease progression in patients with PBC22. However, the precise mechanistic role of vitamin D in PBC pathogenesis remains incompletely understood. Through an integrative approach combining genomics, clinical data, and experimental models, this study systematically investigated the potential role of vitamin D in PBC development and its underlying molecular mechanisms.
Our findings revealed that serum 25(OH)D3 levels were significantly lower in PBC patients compared to healthy controls, with the degree of deficiency being more pronounced than previously reported33,34. his observation may be attributed to the progressive destruction of small intrahepatic bile ducts, a hallmark pathological feature of PBC35, which impairs bile secretion and consequently reduces the absorption of fat-soluble vitamin D. Notably, serum vitamin D levels exhibited a significant negative correlation with liver function parameters (AST, ALT, and TBIL), with more severe deficiency observed in advanced-stage patients, suggesting that vitamin D status may serve as a biomarker for disease severity. Importantly, calcitriol treatment not only restored vitamin D levels but also significantly improved liver function indices and histopathological features, including portal inflammation and bile duct reaction, providing direct evidence for the hepatoprotective effects of vitamin D.
To our knowledge, this is the first study to identify a marked increase in M1-type macrophages in PBC liver tissue using hepatic sequencing data. The imbalance in macrophage polarization (M1/M2 ratio) has been implicated in various liver pathologies36,37. Our analysis demonstrated significant enrichment of M1 macrophages in PBC livers. Under conditions of disrupted hepatic homeostasis, macrophages undergo polarization38, adopting distinct functional phenotypes—classically activated (M1) and alternatively activated (M2) macrophages39 — that mediate diverse pathophysiological responses. The M1/M2 polarization imbalance is increasingly recognized as a key contributor to inflammatory diseases40,41. In PBC, cytotoxic M1 macrophages may drive the inflammatory cascade leading to autoimmune injury, particularly in the characteristic early-stage inflammatory damage of small intrahepatic bile ducts42. This finding aligns remarkably well with the urinary tract infection hypothesis of PBC pathogenesis proposed by Liang et al.43, as lipopolysaccharide (LPS) from Gram-negative bacteria is a potent inducer of M1 polarization, potentially explaining the observed M1 macrophage accumulation in PBC livers.
In our model, mice with early PBC exhibit biliary proliferation or liver injury similar to that of early PBC in humans. The infiltration of inflammatory cells around small intrahepatic bile ducts was reduced in PBC mice treated with vitamin D. These results suggest that vitamin D can significantly reduce liver inflammation in PBC. Macrophages play an important role in the occurrence and development of a variety of liver diseases36,37. When M1-type macrophages are activated, they release a large number of proinflammatory cytokines, leading to persistent aggravation of liver inflammation. M1-type macrophages over-accumulate in the liver of PBC mice, leading to increased levels of inflammatory factors TNF-α, IL-1β and IL-6. VitD3 treatment significantly inhibited the infiltration of M1 macrophages in the liver of PBC mice and reduced the release of inflammatory factors. Our in vitro experiments further showed that VitD3 had an inhibitory effect on LPS/IFN-γ induced M1 macrophage polarization, thereby reducing the expression of the M1 macrophage marker iNOS. Therefore, inhibition of M1-type macrophage activation may be involved in VitD3-mediated anti-inflammatory effects. In particular, we found that vitamin D inhibited the expression of MCP-1, a chemokine that plays a key role in the recruitment of macrophages to the site of bile duct injury. Monocyte chemoattractant protein-1 (MCP-1), a key factor for regulation of monocyte macrophage migration and infiltration, plays an important role in recruiting macrophages and stimulating their polarization44,45. MCP-1 is secreted by a variety of liver-resident cells including biliary epithelial cells, stellate cells, and hepatocytes46,47. MCP-1 induces macrophages to migrate to sites of biliary epithelial cell injury, polarizes the cells to an inflammatory phenotype, mediates the secretion of inflammatory factors, and aggravates the biliary epithelial cell inflammation. These observations suggest that bile duct inflammation may be related to M1 macrophages. Our results are the first to elucidate and localize the polarization typing and aggregation regions of macrophages in the liver of PBC patients. Therefore, identification of targets that regulate liver macrophage polarization may provide a potential novel therapeutic option for PBC.
Macrophage activation and differentiation are tightly controlled by multiple signaling molecules and mechanisms39. Of note, there is emerging evidence that the TLR4 pathway plays a role in macrophage-mediated inflammation48. Our study provides new insights into its complex regulation by vitamin D in the liver. TLR4, a pattern recognition receptor expressed on macrophages, activates NF-κB through the MyD88-dependent pathway, thereby promoting the release of multiple downstream inflammatory cytokines, including TNF-α, IL-1β, and IL-649. In the present study, genes associated with both vitamin D signaling and PBC were enriched in the TLR and NF-κB pathways. This led us to hypothesize that vitamin D might ameliorate hepatic inflammation in PBC by suppressing NF-κB signaling. To test this hypothesis, we assessed proteins related to the TLR4/NF-κB pathway in the livers of PBC model mice using immunofluorescence. We observed increased TLR4 expression in macrophages during liver injury. Vitamin D supplementation promoted VDR expression around bile ducts, concurrently suppressing the expression of TLR4, MyD88, and other pathway-related proteins, and inhibiting M1 macrophage polarization. These results indicate that vitamin D-mediated inhibition of the TLR4 pathway restrains M1 polarization, thereby attenuating liver inflammation in PBC. Mechanistically, it has been reported that vitamin D can influence direct interaction between VDR and IKKβ, disrupting IKK complex formation, inhibiting IKKβ phosphorylation and subsequent NF-κB pathway activation. It also blocks TNF-α-induced p65 nuclear translocation and IL-6 expression, revealing the molecular basis for VDR as a direct negative regulator of NF-κB signaling by targeting IKKβ50. Furthermore, VDR can competitively bind to TLR4 against the endogenous ligand HMGB1, indirectly suppressing immune and inflammatory responses51. All the above mechanisms may be involved in the regulation of TLR4/NF-κB signaling pathway in macrophages by vitamin D in the liver of PBC. In our PBC model, VDR expression was increased around bile ducts. Following VitD3 intervention, VDR expression was significantly reduced in these areas, accompanied by decreased expression of iNOS and MCP-1, consistent with the overall reduction in hepatic inflammatory cytokines. Concurrently, the expression of TLR4, MyD88, and NF-κB was suppressed, leading to reduced production of cytokines (TNF-α, IL-1β, IL-6). These data collectively suggest that VitD3 may modulates M1 macrophage polarization via the TLR4/NF-κB signaling pathway, substantially alleviating liver inflammation in PBC.
It is noteworthy that the immunomodulatory function of vitamin D3 is pleiotropic, extending well beyond its impact on macrophages. Published evidence indicates that it also modulates adaptive immune responses. For instance, vitamin D3 promotes the differentiation of CD4+ T cells, increasing the population of naïve T cells while reducing regulatory T cells (Tregs), thereby enhancing the cytotoxic activity of CD8+ effector T cells52. Furthermore, it suppresses the activation and differentiation of memory B cells, leading to decreased serum levels of IgM, IgG1, IgG3, and anti-dsDNA IgG53. Vitamin D3 also modulates inflammatory responses in other immune cells, such as NK cells54. Therefore, the mechanism elucidated in our study likely represents one component of the broader anti-inflammatory microenvironment orchestrated by vitamin D3 in vivo. The overall therapeutic effect may thus stem from the coordinated regulation of multiple targets—including macrophages, T cells, and B cells—collectively shifting the immune balance from a pro-inflammatory state towards a tolerogenic one. Future studies will focus on validating this integrated regulatory network within more complex in vivo settings.
This study has several limitations that should be acknowledged. First, the safety profile and immunomodulatory mechanisms of VitD3 within the hepatic microenvironment at both physiological and pharmacological concentrations remain to be fully elucidated. Second, the macrophage polarization is a transient process; thus, capturing its full dynamics necessitates future studies with denser temporal sampling to delineate the evolution of the M1/M2 equilibrium. One last point, the animal model was tested with only a single VitD3 dose. Subsequent investigations will employ systematic dose-response experiments to delineate the therapeutic window and determine the optimal dosage that provides the greatest benefit in modulating liver macrophage polarization with minimal risk (Fig. 11).
In PBC, hepatic macrophage activation through the TLR4/NF-κB signaling pathway drives M1 polarization, subsequently initiating a pro-inflammatory cascade characterized by elevated cytokine release that exacerbates hepatic inflammation. Conversely, VitD3 exerts therapeutic effects by suppressing TLR4/NF-κB pathway activation. This inhibition attenuates M1 macrophage differentiation, thereby ameliorating both inflammatory responses and liver injury in PBC pathogenesis.
Conclusions
In conclusion, this study has revealed for the first time that VitD3 can ameliorate the progression of PBC through modulation of hepatic macrophage polarization. Mechanistically, VitD3 exerts anti-inflammatory effects in the liver of PBC patients by suppressing M1 macrophage polarization through regulation of the TLR4/NF-κB signaling pathway. These findings provide novel insights into the immunomodulatory properties of VitD3 in PBC, and may offer a theoretical foundation for the development of therapeutic strategies to decelerate disease progression through VitD3-mediated macrophage polarization.
Data availability
The datasets presented in this study can be found in GEO repository (GSE79850, GPL19965 platform with gene PanCancer immune profiling panel, https://www.ncbi.nlm.nih.gov/geo/).
References
Carey, E. J., Ali, A. H. & Lindor, K. D. Primary biliary cirrhosis. Lancet (London England). 386(10003), 1565–1575. https://doi.org/10.1016/s0140-6736(15)00154-3 (2015).
Lleo, A., Marzorati, S., Anaya, J. M. & Gershwin, M. E. Primary biliary cholangitis: a comprehensive overview. Hep. Intl. 11(6), 485–499. https://doi.org/10.1007/s12072-017-9830-1 (2017).
Lleo, A. et al. Biliary apotopes and anti-mitochondrial antibodies activate innate immune responses in primary biliary cirrhosis. Hepatol. (Baltimore Md). 52(3), 987–998. https://doi.org/10.1002/hep.23783 (2010).
Hirschfield, G. M. & Gershwin, M. E. The immunobiology and pathophysiology of primary biliary cirrhosis. Annu. Rev. Pathol. 8, 303–330. https://doi.org/10.1146/annurev-pathol-020712-164014 (2013).
Tsuneyama, K., Baba, H., Morimoto, Y., Tsunematsu, T. & Ogawa, H. Primary biliary cholangitis: its pathological characteristics and immunopathological mechanisms. J. Med. Investig. 64(1.2), 7–13. https://doi.org/10.2152/jmi.64.7 (2017).
Krenkel, O. & Tacke, F. Liver macrophages in tissue homeostasis and disease. Nat. Rev. Immunol. 17(5), 306–321. https://doi.org/10.1038/nri.2017.11 (2017).
Li, X. et al. The flavonoid Quercetin ameliorates liver inflammation and fibrosis by regulating hepatic macrophages activation and polarization in mice. Front. Pharmacol. 9, 72. https://doi.org/10.3389/fphar.2018.00072 (2018).
Wynn, T. A. & Vannella, K. M. Macrophages in tissue Repair, Regeneration, and fibrosis. Immunity 44(3), 450–462. https://doi.org/10.1016/j.immuni.2016.02.015 (2016).
Msheik, Z., El Massry, M., Rovini, A., Billet, F. & Desmoulière, A. The macrophage: a key player in the pathophysiology of peripheral neuropathies. J. Neuroinflamm. 19(1), 97. https://doi.org/10.1186/s12974-022-02454-6 (2022).
Vasiliadou, I. & Holen, I. The role of macrophages in bone metastasis. J. Bone Oncol. 2(4), 158–166. https://doi.org/10.1016/j.jbo.2013.07.002 (2013).
Mantovani, A., Biswas, S. K., Galdiero, M. R., Sica, A. & Locati, M. Macrophage plasticity and polarization in tissue repair and remodelling. J. Pathol. 229(2), 176–185. https://doi.org/10.1002/path.4133 (2013).
Tang, Q. et al. Pirfenidone ameliorates pulmonary inflammation and fibrosis in a rat silicosis model by inhibiting macrophage polarization and JAK2/STAT3 signaling pathways. Ecotoxicol. Environ. Saf. 244, 114066. https://doi.org/10.1016/j.ecoenv.2022.114066 (2022).
Cutolo, M., Campitiello, R., Gotelli, E. & Soldano, S. The role of M1/M2 macrophage polarization in rheumatoid arthritis synovitis. Front. Immunol. 13, 867260. https://doi.org/10.3389/fimmu.2022.867260 (2022).
Chen, X., Zhu, L. & Wu, H. The role of M1/M2 macrophage polarization in primary sjogren’s syndrome. Arthritis Res. Therapy. 26(1), 101. https://doi.org/10.1186/s13075-024-03340-7 (2024).
DeLuca, H. F. Overview of general physiologic features and functions of vitamin D. Am. J. Clin. Nutr. 80(6 Suppl), 1689s–96s. https://doi.org/10.1093/ajcn/80.6.1689S (2004).
Pop, T. L., Sîrbe, C., Benţa, G., Mititelu, A. & Grama, A. The role of vitamin D and vitamin D binding protein in chronic liver diseases. Int. J. Mol. Sci. 23(18). https://doi.org/10.3390/ijms231810705 (2022).
Murdaca, G. et al. Emerging role of vitamin D in autoimmune diseases: an update on evidence and therapeutic implications. Autoimmun. rev. 18(9), 102350. https://doi.org/10.1016/j.autrev.2019.102350 (2019).
Dorji, S., Yangchen, S. & Chuki, P. Prevalence of vitamin D deficiency in patients with autoimmune rheumatic diseases visiting the rheumatology clinic at the National referral Hospital, Bhutan. SAGE open. Med. 12, 20503121231223313. https://doi.org/10.1177/20503121231223313 (2024).
Correa-Rodríguez, M. et al. Vitamin D levels are associated with disease activity and damage accrual in systemic lupus erythematosus patients. Biol. Res. Nurs. 23(3), 455–463. https://doi.org/10.1177/1099800420983596 (2021).
Song, F. et al. Vitamin D and CRP are associated in hospitalized inflammatory bowel disease (IBD) patients in Shanghai. Asia Pac. J. Clin. Nutr. 33(3), 370–380. https://doi.org/10.6133/apjcn.202409_33(3).0007 (2024).
Vahabi Anaraki, P. et al. Effect of vitamin D deficiency treatment on thyroid function and autoimmunity markers in hashimoto’s thyroiditis: A double-blind randomized placebo-controlled clinical trial. J. Res. Med. Sciences: Official J. Isfahan Univ. Med. Sci. 22, 103. https://doi.org/10.4103/jrms.JRMS_1048_16 (2017).
Wang, Z. et al. Serum vitamin D level is related to disease progression in primary biliary cholangitis. Scand. J. Gastroenterol. 55(11), 1333–1340. https://doi.org/10.1080/00365521.2020.1829030 (2020).
Arai, T. et al. Association of vitamin D levels and vitamin D-related gene polymorphisms with liver fibrosis in patients with biopsy-proven nonalcoholic fatty liver disease. Dig. Liver Disease: Official J. Italian Soc. Gastroenterol. Italian Association Study Liver. 51(7), 1036–1042. https://doi.org/10.1016/j.dld.2018.12.022 (2019).
Luo, W. J. et al. Vitamin D 1,25-Dihydroxyvitamin D(3) reduces lipid accumulation in hepatocytes by inhibiting M1 macrophage polarization. World J. Gastrointest. Oncol. 16(12), 4685–4699. https://doi.org/10.4251/wjgo.v16.i12.4685 (2024).
Xu, J. et al. Modulation of CXCR1 and CXCR3 expression on NK cells via Tim-3 in a murine model of primary biliary cholangitis. Mol. Immunol. 135, 342–350. https://doi.org/10.1016/j.molimm.2021.04.014 (2021).
Lefere, S., Devisscher, L. & Tacke, F. Targeting CCR2/5 in the treatment of nonalcoholic steatohepatitis (NASH) and fibrosis: opportunities and challenges. Expert Opin. Investig. Drugs. 29(2), 89–92. https://doi.org/10.1080/13543784.2020.1718106 (2020).
Italiani, P. & Boraschi, D. From monocytes to M1/M2 macrophages: phenotypical vs. Functional differentiation. Front. Immunol. 5, 514. https://doi.org/10.3389/fimmu.2014.00514 (2014).
Mohd Yasin, Z. N., Mohd Idrus, F. N., Hoe, C. H. & Yvonne-Tee, G. B. Macrophage polarization in THP-1 cell line and primary monocytes: A systematic review. Differ. Res. Biol. Divers. 128, 67–82. https://doi.org/10.1016/j.diff.2022.10.001 (2022).
Zubkova, E. S. et al. Features of the population of mouse peritoneal macrophages isolated after stimulation with Concanavalin A and thioglycolate. Bull. Exp. Biol. Med. 171(4), 532–540. https://doi.org/10.1007/s10517-021-05265-6 (2021).
Yang, Y. et al. Vitamin D protects silica particles induced lung injury by promoting macrophage polarization in a KLF4-STAT6 manner. J. Nutr. Biochem. 110, 109148. https://doi.org/10.1016/j.jnutbio.2022.109148 (2022).
Abu-Elnasr Awwad, A. et al. Impact of vitamin D in children with chronic tonsillitis (immunohistochemical study of CD68 polarisation and Proinflammatory cytokines estimation). Sci. Rep. 13(1), 8014. https://doi.org/10.1038/s41598-023-33970-x (2023).
Zhen, H. et al. VitA or VitD ameliorates bronchopulmonary dysplasia by regulating the balance between M1 and M2 macrophages. Biomed. pharmacotherapy = Biomedecine Pharmacotherapie. 141, 111836. https://doi.org/10.1016/j.biopha.2021.111836 (2021).
Agmon-Levin, N. et al. Vitamin D in primary biliary cirrhosis, a plausible marker of advanced disease. Immunol. Res. 61(1–2), 141–146. https://doi.org/10.1007/s12026-014-8594-0 (2015).
Guo, G. Y. et al. Serum vitamin D level is associated with disease severity and response to ursodeoxycholic acid in primary biliary cirrhosis. Aliment. Pharmacol. Ther. 42(2), 221–230. https://doi.org/10.1111/apt.13244 (2015).
EASL Clinical Practice Guidelines. The diagnosis and management of patients with primary biliary cholangitis. J. Hepatol. 67(1), 145–172. https://doi.org/10.1016/j.jhep.2017.03.022 (2017).
Ma, P. F. et al. Cytotherapy with M1-polarized macrophages ameliorates liver fibrosis by modulating immune microenvironment in mice. J. Hepatol. 67(4), 770–779. https://doi.org/10.1016/j.jhep.2017.05.022 (2017).
Yang, Y. et al. Crosstalk between hepatic tumor cells and macrophages via Wnt/β-catenin signaling promotes M2-like macrophage polarization and reinforces tumor malignant behaviors. Cell Death Dis. 9(8), 793. https://doi.org/10.1038/s41419-018-0818-0 (2018).
Wang, L., Lu, Q., Gao, W. & Yu, S. Recent advancement on development of drug-induced macrophage polarization in control of human diseases. Life Sci. 284, 119914. https://doi.org/10.1016/j.lfs.2021.119914 (2021).
Wang, C. et al. Macrophage polarization and its role in liver disease. Front. Immunol. 12, 803037. https://doi.org/10.3389/fimmu.2021.803037 (2021).
Zhou, F. et al. Kinsenoside attenuates osteoarthritis by repolarizing macrophages through inactivating NF-κB/MAPK signaling and protecting chondrocytes. Acta Pharm. Sinica B. 9(5), 973–985. https://doi.org/10.1016/j.apsb.2019.01.015 (2019).
Liu, J. et al. Macrophage polarization in periodontal ligament stem cells enhanced periodontal regeneration. Stem Cell Res. Ther. 10(1), 320. https://doi.org/10.1186/s13287-019-1409-4 (2019).
Younossi, Z. M. et al. Diagnosis and management of primary biliary cholangitis. Am. J. Gastroenterol. 114(1), 48–63. https://doi.org/10.1038/s41395-018-0390-3 (2019).
Liang, Y., Yang, Z. & Zhong, R. Smoking, family history and urinary tract infection are associated with primary biliary cirrhosis: A. Hepatol. Research: Official J. Japan Soc. Hepatol. 41(6), 572–578. https://doi.org/10.1111/j.1872-034X.2011.00806.x (2011).
Deshmane, S. L., Kremlev, S., Amini, S. & Sawaya, B. E. Monocyte chemoattractant protein-1 (MCP-1): an overview. J. Interferon Cytokine Research: Official J. Int. Soc. Interferon Cytokine Res. 29(6), 313–326. https://doi.org/10.1089/jir.2008.0027 (2009).
Xie, Z. et al. MCP1 triggers monocyte dysfunctions during abnormal osteogenic differentiation of mesenchymal stem cells in ankylosing spondylitis. J. Mol. Med. 95(2), 143–154. https://doi.org/10.1007/s00109-016-1489-x (2017).
Baeck, C. et al. Pharmacological Inhibition of the chemokine CCL2 (MCP-1) diminishes liver macrophage infiltration and steatohepatitis in chronic hepatic injury. Gut 61(3), 416–426. https://doi.org/10.1136/gutjnl-2011-300304 (2012).
Yan, M., Gu, S., Pan, C., Chen, Y. & Han, X. MC-LR-induced interaction between M2 macrophage and biliary epithelial cell promotes biliary epithelial cell proliferation and migration through regulating STAT3. Cell Biol. Toxicol. 37(6), 935–949. https://doi.org/10.1007/s10565-020-09575-9 (2021).
Duan, C. et al. Sirtuin1 inhibits calcium oxalate crystal-induced kidney injury by regulating TLR4 signaling and macrophage-mediated inflammatory activation. Cell. Signal. 112, 110887. https://doi.org/10.1016/j.cellsig.2023.110887 (2023).
Zhu, H. X. et al. Targeting pathogenic CD8(+) tissue-resident T cells with chimeric antigen receptor therapy in murine autoimmune cholangitis. Nat. Commun. 15(1), 2936. https://doi.org/10.1038/s41467-024-46654-5 (2024).
Chen, Y. et al. Vitamin D receptor inhibits nuclear factor κB activation by interacting with IκB kinase β protein. J. Biol. Chem. 288(27), 19450–19458. https://doi.org/10.1074/jbc.M113.467670 (2013).
Zhang, H., Yang, N., Wang, T., Dai, B. & Shang, Y. Vitamin D reduces inflammatory response in asthmatic mice through HMGB1/TLR4/NF–κB signaling pathway. Mol. Med. Rep. 17(2), 2915–2920. https://doi.org/10.3892/mmr.2017.8216 (2018).
Li, B. et al. Vitamin D3 reverses immune tolerance and enhances the cytotoxicity of effector T cells in coal pneumoconiosis. Ecotoxicol. Environ. Saf. 271, 115972. https://doi.org/10.1016/j.ecoenv.2024.115972 (2024).
Yamamoto, E. A. et al. Low levels of vitamin D promote memory B cells in lupus. Nutrients 12(2). https://doi.org/10.3390/nu12020291 (2020).
Amer, J., Salhab, A. & Abuawad, M. Restoring natural killer cell activity in lung injury with 1,25-hydroxy vitamin D(3): a promising therapeutic approach. Front. Immunol. 15, 1466802. https://doi.org/10.3389/fimmu.2024.1466802 (2024).
Funding
This work was supported by the National Natural Science Foundation of China (81971997).
Author information
Authors and Affiliations
Contributions
Zhan Wang: Conceptualization, Formal analysis, Investigation, Validation, Writing - original daft. Weize Gao: Methodology, Data curation, Software. Xuedi Cheng: Data curation. Nana Zhang: Data curation. Wenlong Lu: Data curation, Software. Yongxin Li: Formal analysis, Visualization. Mingjun Liu: Conceptualization, Investigation, Writing - review & editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Date availability
The datasets presented in this study can be found in GEO repository (GSE79850, GPL19965 platform with gene PanCancer immune profiling panel, https://www.ncbi.nlm.nih.gov/geo/).
Ethical approval
The acquisition of liver tissue information in this study was approved by the Ethics Committee of the Affiliated Hospital of Qingdao University (QYFYWZLL28713). All experiments are carried out in accordance with the health guidelines of the National Agency (Guidelines for the Care and Use of Laboratory Animals). The animal experimental protocols were approved by the Animal Experiment Ethics Committee of Viewsolid Biotech (approval no. VS2126A01149).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, Z., Gao, W., Cheng, X. et al. Vitamin D3 attenuates hepatic inflammation in primary biliary cholangitis by inhibiting TLR4/NF-κB mediated M1 macrophage polarization. Sci Rep 15, 44345 (2025). https://doi.org/10.1038/s41598-025-27911-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-27911-z