Abstract
Cholangiocarcinoma is a common malignant tumor that seriously endangers physical and mental health. Elevated serum ferritin levels are correlated with reduced survival and increased recurrence rates in various cancers. The present study aimed to evaluate the potential of serum ferritin as a prognostic biomarker in cholangiocarcinoma. Immunoassay was used to detect ferritin protein in the serum of 120 cholangiocarcinoma patients and to compare the diagnostic utility of the conventional tumour markers CEA, CA19-9 and CA125. The median DFS for all patients was 420 days (range, 1.0-51.8 months). Median survival was 991 days (range, 2.1–51.8 months) and 361 days (range, 1.0-51.8 months) in the low- and high-ferritin level groups, respectively (P < 0.001). Patients with cholangiocarcinoma with low ferritin expression (≤ 196.5 ng/ml) had significantly higher disease-free survival than those with high ferritin expression (> 196.5 ng/ml). The findings of multivariate analyses showed serum ferritin (P = 0.001), CA19-9 (P = 0.037), Tbil (P = 0.017), and Ki67 (P = 0.008), lymph node metastasis (P = 0.040) and no lymph node dissection (P<0.001) were considered as independent factors affecting the prognosis. Ferritin levels had statistically significant relationship between patient’s CA19-9 level, Vascular invasion, CD34/31 expression and Postoperative adjuvant therapy (P < 0.05). These results indicate that serum ferritin is prognostically associated with patients with cholangiocarcinoma and may serve as a valuable prognostic biomarker for patients with cholangiocarcinoma.
Similar content being viewed by others
Introduction
Originating from the bile duct epithelial cells, cholangiocarcinoma encompasses malignant growths within the liver and extends from the hepatic hilum to the distal end of the common bile duct. The disease occurs mainly among people between the ages of 50 and 70, with a ratio of 2.8 men to 1 women1. Though the incidence of bile tract cancer (BTC) is comparatively low, it carries a significant disease burden and is associated with unfavorable survival rates. Despite early detection and radical surgery for cholangiocarcinoma, studies have documented brief recurrence-free intervals (ranging from 11 to 20 months) and elevated recurrence frequencies (between 53–62%)2. Additionally, a majority of patients were first diagnosed with advanced cholangiocarcinoma cancer3. Therefore, understanding the factors related to cholangiocarcinoma recurrence or survival could have significant value in improving patient outcomes. Unlike other categories of cancer, progression of choalngiocarcinoma is closely connected to liver function. Tumour progression worsens hepatic function, but repeated treatments also impair hepatic function; hence, choosing appropriate treatments in clinical practice is challenging. For the past decade, standard treatment options for advanced hepatobiliary cancer have been limited4. Most patients undergo anticancer therapy, but the survival benefit is either limited or short-term. In the clinical practice, accurate survival forecasting is essential to provide optimal therapy to patients. Proteins in the blood of cancer patients can signal the presence and advancement of tumors. In medical practice, serum tumor markers like CEA, CA19-9, CA242, and CA50 have been validated as potential indicators for predicting survival outcomes in individuals with advanced cancers5,6. However, these biomarkers are inadequate to accurate prognosis prediction, with reports indicating inaccurate prediction in 45% of cases7,8,9. The complexity of these scoring systems makes it difficult to apply them widely in clinical practice, and thus, these biomarkers have not yet been widely adopted in clinical practice10.
Iron is an element important for promoting cell proliferation and growth, participating in cellular redox reactions and the generation of free radicals. Thus, iron is closely related to the onset and progression of tumours11. Elevated iron levels can increase oxidative stress in the tumor microenvironment, promoting tumor cell metastasis12. Ferritin is an iron storing protein found in large quantities in cells. A small fraction of ferritin, known as serum ferritin, is found in the bloodstream. This serum ferritin serves as an indicator of iron reserves, with 1 ng/ml correlating to approximately 8 mg of stored iron13. Tendencies show that serum ferritin levels increase with age and are generally more elevated in males compared to females14. The elevation of serum ferritin usually shows iron overload, but it is also elevated in cases of inflammation, liver disease and malignancy15,16. Previous studies have indicated that high serum ferritin levels are associated with a poorer prognosis in various cancers, including pancreatic17,18, colorectal19,20, lung21,22, peripheral T-cell lymphoma, and hepatocellular carcinoma8. However, the impact of serum ferritin on survival outcomes in patients with advanced cholangiocarcinoma remains unexplored.
This study aimed to assess the potential of serum ferritin levels to predict survival outcomes prospectively in patients with advanced cholangiocarcinoma.
Materials and methods
Study population
All cases were obtained from patients who had radical surgery and were pathologically diagnosed with cholangiocarcinoma postoperatively at the Department of Hepatobiliary Surgery and Oncology, The Second Affiliated Hospital of Kunming Medical University, between July 2013 and December 2023, with complete pathological data. All study subjects excluded those with pregnancy, infections, liver and renal insufficiency, diabetes, heart failure, cerebral hemorrhage, and other complications, as well as those with other types of tumors. The study was approved by the Ethics Committee of the second Affiliated hospital of Kunming medical university (No. PJ-2021–216), and was conducted in accordance with the Declaration of Helsinki promulgated by the Chinese government. All enrolled patients had complete clinicopathological data and follow-up information, and no missing key variables were identified; thus, a complete case analysis was employed.
Collection of clinical data and prognostic follow-up
All relevant medical data of the patients were captured from the hospital’s medical information record system, including age, gender, tumour pathological features (including lymph node metastasis, vascular invasion, Ki-67, CD34/31 expression, degree of tumour cell differentiation) and following treatments. Biochemical markers, including serum ferritin, CA19-9, CA125, CEA, pre-albumin(PA), total bilirubin (Tbil), albumin(ALB), neutrophil count(N), lymphocyte count(L), Monocyte count(M) and platelet count(P), were evaluated by peripheral blood test. Except for NLR (N/L), LMR (L/M), PLR (P/L) and serum ferritin, the cut-off values for all biochemical indicators were based on the reference ranges of the indicators.
Disease-free survival (DFS)
Disease-free survival (DFS) was as defined as the time from radical surgery to disease recurrence or death by any cause. When patient was out of follow-up or there is no record of death, the patient is censored. Patient survival time is defined as the period from the date of surgery to the final day of follow-up or to 31 December 2023 (the date on which survival was surveyed).
Statistical analysis
In this study, the X-tile software (Rimm Lab, Yale University, New Haven, CT, USA) was used to determine the optimal cut-off value for serum ferritin (196.5 ng/ml) through an exhaustive method. Patients’ DFS was measured using the Kaplan-Meier estimation method, and differences between survival values were compared using the log-rank test. Censoring on the right was implemented in survival analyses for incomplete time data. Chi-square tests were utilized for subgroup analyses regarding ferritin levels. Both univariate and multivariate analyses employed Cox proportional hazards modeling. Multivariate analyses used factors that were statistically significant in univariate analyses (P values ≤ 0.05) and controlled for confounders such as age and sex. Pearson’s correlation coefficient test was performed to assess the relationship between serum ferritin and other biochemical parameters. Data were analysed using SPSS (version 18.0; SPSS Inc.) P values ≤ 0.05 were considered statistically significant.
Results
Cholangiocarcinoma patients characteristics
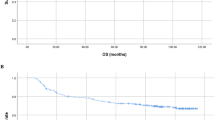
A series of 120 patients with cholangiocarcinoma were incorporated in this study, and their clinicopathological characteristics are shown in Table 1. The average age was 60.5 years, 35 cases were extrahepatic cholangiocarcinoma, and 85 were intrahepatic cholangiocarcinoma. The follow-up period ranged from 1 to 124 months (47.9 ± 32.1 months) (Fig. 1). As of December 31, 2023, a total of 14 patients (11.6%) had died.
Disease-free survival of enrolled patients.
Association between clinicopathological factors and prognosis in cholangiocarcinoma patients
In univariate Cox regression analysis (Table 2), clinicopathological factors such as serum ferritin, CA19-9 level, CA125 level, CEA level, PA level, PLR, TBIL, Ki67 expression level, lymph node metastasis, lymph node dissection or not and whether adjuvant therapy was applied were significantly associated with survival outcomes. The Kaplan-Meier survival curve is shown in Fig. 2.
Kaplan-Meier survival curve.
We further confirmed this correlation using the Cox regression model. The research indicated that patients with elevated serum ferritin levels had a 4.021-fold greater risk of tumor recurrence compared to those with lower levels (P = 0.001). The risk of tumour recurrence in patients with elevated serum CA19-9 levels was 1.944 times higher than in patients with low levels (P = 0.037). Patients with elevated total bilirubin levels faced a 0.423-fold increased risk of tumor recurrence compared to those with lower levels (P = 0.045). For cholangiocarcinoma, a Ki67 positivity above 30% correlated with a 2.228-fold increased risk of tumor recurrence versus those with Ki67 positivity at or below 30% (P = 0.008). The risk of tumour recurrence in cholangiocarcinoma patients with lymph node metastases was 1.849 times higher than in patients without lymph node metastases (P = 0.040); the risk of tumour recurrence in cholangiocarcinoma patients without lymph node clearance was 2.841 times higher than in patients without lymph node metastases (P<0.001). Serum ferritin (HR = 4.021, P = 0.001), CA19-9 (HR = 1.944, P = 0.037), Ki67 positive areas > 30% (HR = 2.402, P = 0.020), lymph node metastases (HR = 1.849, P = 0.040) and lymph node dissection (HR = 2.841, P<0.001) were significantly linked with DFS. We created a forest plot on the basis of the findings of the study (Fig. 3).
Forest flot according to the Cox regression model.
Relationship between iron concentration, clinicopathological factors, and prognosis in cholangiocarcinoma patients
We categorised ferritin levels into two groups, low and high, based on cut-off values to assess the relationship between cholangiocarcinoma prognosis and pathological factors. The outcome indicated that the median survival was 33.0 months (range: 2.1–51.8 months) in the low ferritin group, whereas the median survival was 12 months (range: 1.0-40.9 months) in the high ferritin group (P < 0.001). Disease-free survival was significantly shorter in patients with high ferritin than in those with low ferritin. As shown in Table 3, there were no meaningful differences in age, gender, CA125 level, CEA level, PA level, ALB level, Ki-67 expression, and lymphatic metastasis between the two groups (P > 0.05). However, higher serum ferritin levels were strongly linked to CA19-9 (P = 0.028), vascular invasion (P = 0.012), CD34/31 expression (P = 0.021), Tbil level(P = 0.031), and whether or not adjuvant therapy was administered after surgery (P = 0.008). These findings suggest that serum ferritin is an independent predictor of prognosis in patients with cholangiocarcinoma.
Discussion
In the study, we analysed the value of serum ferritin levels in predicting survival outcomes in patients with cholangiocarcinoma in this study. To sum up, our study identified ferritin, CA19-9, Ki-67, and lymph node involvement as significant factors associated with the prognosis of individuals suffering from cholangiocarcinoma. CA19-9 is a typical tumour marker. Many studies have shown that CA19-9 is essential for the diagnosis, efficacy monitoring and prognostic evaluation of cholangiocarcinoma23,24,25,26. Previous studies have suggested that tumour ferritin levels in cancer patients may be linked to prognosis. Regarding cholangiocarcinoma, Zhou et al. found that reduced iron levels were strongly associated with a favourable clinical prognosis in cholangiocarcinoma patients27. Song et al. reported that serum ferritin could be a predictive factor in patients with advanced hepatobiliary carcinoma treated with Korean medicine28. A meta-analysis by Lin et al. showed that both intrahepatic and extrahepatic cholangiocarcinoma patients with high serum ferritin levels were significantly associated with poor prognosis17. In line with these results, our research also discovered that increased pre-treatment serum ferritin (SF) levels correlated with inferior overall survival (OS), recurrence-free survival/progression-free survival/time to recurrence (RFS/PFS/TTR) in individuals with cholangiocarcinoma. Additional subgroup analyses revealed that elevated SF levels were significantly linked to a reduced disease-free survival (DFS) in cholangiocarcinoma patients, irrespective of tumor classification, ethnic background, SF value thresholds, tumor-node-metastasis (TNM) staging, or Newcastle-Ottawa Scale (NOS) scores. Consequently, pre-treatment SF could be regarded as a valuable prognostic indicator for individuals afflicted with cholangiocarcinoma.
We then examined the relationship between serum ferritin (SF) levels and various clinicopathological factors, categorizing patients into two groups—low and high—based on the World Health Organization’s guidelines for ferritin levels. The analysis showed that patients with high ferritin levels were significantly correlated with elevated CA19-9 (P = 0.028), high vascular invasion (P = 0.008), low CD34/31 expression (P = 0.038), and increased risk of postoperative adjuvant therapy and higher recurrence rate. CA19-9 is a notable tumor marker. CA19-9 is a carbohydrate antigen commonly association with cancers of the gastrointestinal tract and serves as an auxiliary diagnostic marker for malignant tumors such as pancreatic and gallbladder cancer, with significant utility in monitoring disease progression and recurrence29,30,31. Studies have shown that CA19-9 levels were significantly increased in the iron overload group compared to the ferritin-normal group. Correlation analysis showed that CA19-9 was strongly related to both iron overload and HbA1c32. After adjusting for confounding factors, iron overload and HbA1c were still found to be independently associated with CA19-9 levels. However, the mechanism by which ferritin influences CA19-9 still requires further research.
Serum ferritin maintains the iron homeostasis necessary for DNA synthesis and cell proliferation33. Due to differences in cell regeneration capacity, tumor cells require more iron than normal cells11. Serum ferritin binds to high molecular weight kininogen, thereby inhibiting its anti-angiogenic effects34. Coffman et al.32 discovered that ferritin promotes the colonisation of tumour endothelial cells, stimulates angiogenesis during tumour growth and enhancing tumour migration by antagonising the anti-angiogenic effects of HKa (cleaved high molecular weight kininogen)35. Our study showed that high ferritin levels are closely associated with high vascular invasion (P = 0.008) and low CD34/31 expression (P = 0.038), which are important markers of angiogenesis36.
Our research also revealed that ferritin is associated with the responsiveness to postoperative adjuvant treatments. It has been observed that in radiation-resistant cells from cancers of the breast, lung, bladder, glioma, and prostate, there is an accumulation of lipid droplets37. This accumulation leads to the release of specific lipids that inhibit iron-dependent cell death and subsequently increase ferritin expression38,39. Among the 58 patients in this study who underwent postoperative adjuvant therapy, those with elevated serum ferritin levels exhibited reduced progression-free survival (PFS) and a greater likelihood of treatment failure, suggesting a connection to a ferritin-mediated resistance mechanism. Regrettably, given the retrospective nature and single-center design of our study, the causal relationships cannot be definitively established at this stage. Therefore, additional external validation, along with further foundational and clinical research, is necessary to substantiate these observations.
The research encountered constraints, including its retrospective approach and small sample size. Additionally, it lacked iron-related biomarkers such as serum iron, transferrin, and total iron-binding capacity, which are not commonly measured in clinical practice. Therefore, there is a necessity for well-structured, prospective studies to validate the results of this investigation. The results indicated that elevated Tbil unexpectedly emerged as a protective factor. This finding contradicts conventional understanding and may be related to the limited sample size. Literature reports suggest that under specific circumstances, bilirubin may correlate with improved prognosis through its antioxidant properties that mitigate oxidative stress damage, or by inducing mitochondrial depolarization to promote tumor cell apoptosis40,41,42,43. A study limitation is the uncontrolled heterogeneity in adjuvant therapies, which we could not dissect due to small subgroup sizes. Furthermore, while serum ferritin is a significant prognostic biomarker, this observational study cannot establish causality. Thus, elevated ferritin is best interpreted as a composite indicator of tumor burden and concomitant pathologies, signifying poor prognosis rather than directly causing it. Its specific mechanistic role requires future study.
In summary, serum ferritin concentrations correlate with the outcome of cholangiocarcinoma. Our study revealed that among patients with cholangiocarcinoma, those with elevated serum ferritin levels (SF ≥ 196.5 ng/ml) had significantly poorer disease-free survival rates than those with lower levels. This suggests that serum ferritin might be a significant biomarker for predicting outcomes in cholangiocarcinoma. Moreover, additional research is needed to fully understand how serum ferritin affects the disease’s progression.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Brindley, P. J. et al. Cholangiocarcinoma Nat. Reviews Disease Primers 7(1): 65. (2021).
Chen, P-D., Chen, L-J., Chang, Y-J. & Chang, Y-J. Long-term survival of combined hepatocellular-cholangiocarcinoma: a nationwide study. Oncologist 26 (10), e1774–e1785 (2021).
Buckholz, A. P. & Brown, R. S. Cholangiocarcinoma: diagnosis and management. Clin. Liver Dis. 24 (3), 421–436 (2020).
Elvevi, A. et al. Clinical treatment of cholangiocarcinoma: an updated comprehensive review. Ann. Hepatol. 27 (5), 100737 (2022).
Cai, Q. Biomarker study of cholangiocarcinoma. (2022).
Shen, K. et al. Serum CA242: a biomarker for diagnosis, progression, and metastasis in multiple tumors. (2020).
Alkhateeb, A. A. & Connor, J. R. The significance of ferritin in cancer: anti-oxidation, inflammation and tumorigenesis. Biochim. Biophys. Acta (BBA) Rev. Cancer 1836 (2), 245–254. (2013).
Ramírez-Carmona, W. et al. Are serum ferritin levels a reliable cancer biomarker? A systematic review and meta-analysis. Nutr. Cancer. 74 (6), 1917–1926 (2022).
Moro, A. et al. The impact of preoperative CA19-9 and CEA on outcomes of patients with intrahepatic cholangiocarcinoma. Ann. Surg. Oncol. 27, 2888–2901 (2020).
Xiao-jie, L., Xu-ping, C. & Jun, Z. Predictive effect of serum CEA, CA125 combined with CA199 on prognosis of patients with advanced intrahepatic cholangiocarcinoma. J. Clin. Transfus. Lab. Med. 22 (4), 434. (2020).
Hsu, M. Y., Mina, E., Roetto, A. & Porporato, P. E. Iron: an essential element of cancer metabolism. Cells 9 (12), 2591 (2020).
Vela, D. Iron in the tumor microenvironment. Tumor Microenviron. Mol. Players Part A 39–51. (2020).
Moreira, A. C., Mesquita, G. & Gomes, M. S. Ferritin: an inflammatory player keeping iron at the core of pathogen-host interactions. Microorganisms 8 (4), 589 (2020).
Organization, W. H. Serum Ferritin Concentrations for the Assessment of Iron Status and Iron Deficiency in Populations (World Health Organization, 2011).
Hsu, C. C., Senussi, N. H., Fertrin, K. Y. & Kowdley, K. V. Iron overload disorders. Hepatol. Commun. 6 (8), 1842–1854 (2022).
DePalma, R. G., Hayes, V. W. & O’Leary, T. J. Optimal serum ferritin level range: iron status measure and inflammatory biomarker. Metallomics 13 (6), mfab030 (2021).
Lin, S. et al. Meta-analysis of the prognostic value of pretreatment serum ferritin in hepatobiliary and pancreas (HBP) cancers. BMJ open. 11 (5), e040801 (2021).
Wang, S-L. et al. Serum ferritin predicted prognosis in patients with locally advanced pancreatic cancer. Future Oncol. 11 (21), 2905–2910 (2015).
Urback, A. L. et al. Serum ferritin and the risk of early-onset colorectal cancer. World J. Gastrointest. Oncol. 16 (8), 3496 (2024).
Kim, H., Han, K., Ko, S-H. & An, H. J. Association between serum ferritin levels and colorectal cancer risk in Korea. Korean J. Intern. Med. 37 (6), 1205 (2022).
Guo, F. F., Cheng, S. J., Liu, Y. N. & Cui, J. W. The potential clinical significance of the serum iron and ferritin status in lung cancer patients. J. Nutritional Oncol. 5 (4), 189–195 (2020).
Gao, Y. & Ge, J-T. Prognostic role of pretreatment serum ferritin concentration in lung cancer patients: A meta-analysis. World J. Clin. Cases. 10 (33), 12230 (2022).
Wang, Z., Shi, Y., Xiong, G., Han, M. & Chen, X. The prognostic impact of preoperative CA19-9 on resectable cholangiocarcinoma: a comprehensive systematic review and meta-analysis. Discov Oncol. 15 (1), 773 (2024).
Lee, T., Teng, T. Z. J. & Shelat, V. G. Carbohydrate antigen 19 – 9 - tumor marker: Past, present, and future. World J. Gastrointest. Surg. 12 (12), 468–490 (2020).
Capuozzo, M. et al. Intrahepatic cholangiocarcinoma biomarkers: towards early detection and personalized Pharmacological treatments. Mol. Cell. Probes. 73, 101951 (2024).
Bekki, Y., Von Ahrens, D., Takahashi, H., Schwartz, M. & Gunasekaran, G. Recurrent intrahepatic Cholangiocarcinoma - Review. Front. Oncol. 11, 776863 (2021).
Zhou, S-N. et al. A new prognostic model covering all stages of intrahepatic cholangiocarcinoma. J. Clin. Translational Hepatol. 10 (2), 254 (2022).
Song, A., Eo, W., Kim, S., Shim, B. & Lee, S. Significance of serum ferritin as a prognostic factor in advanced hepatobiliary cancer patients treated with Korean medicine: a retrospective cohort study. BMC Complement. Altern. Med. 18, 1–10 (2018).
Luo, G. et al. Roles of CA19-9 in pancreatic cancer: Biomarker, predictor and promoter. Biochim. Et Biophys. Acta (BBA)-Reviews Cancer. 1875 (2), 188409 (2021).
Lakemeyer, L. et al. Diagnostic and prognostic value of CEA and CA19-9 in colorectal cancer. Diseases 9 (1), 21 (2021).
Sachan, A. et al. Raised CA19–9 and CEA have prognostic relevance in gallbladder carcinoma. Bmc Cancer. 20, 1–8 (2020).
Shang, X., Du, X., Fang, T., Zhang, R. & Tian, F. Effect of iron overload on the level of carbohydrate antigen 199 in type 2 diabetes mellitus. Zhonghua Yi Xue Za Zhi. 99 (22), 1722–1726 (2019).
Kotla, N. K., Dutta, P., Parimi, S. & Das, N. K. The role of ferritin in health and disease: recent advances and Understandings. Metabolites 12 (7), 609 (2022).
Tesfay, L., Huhn, A. J., Hatcher, H., Torti, F. M. & Torti, S. V. Ferritin blocks inhibitory effects of two-chain high molecular weight kininogen (HKa) on adhesion and survival signaling in endothelial cells. PLoS One. 7 (7), e40030 (2012).
Coffman, L. G., Parsonage, D., D’Agostino, R. Jr, Torti, F. M. & Torti, S. V. Regulatory effects of ferritin on angiogenesis. Proc. Natl. Acad. Sci. 106 (2), 570–575. (2009).
Kukreja, I., Kapoor, P., Deshmukh, R. & Kulkarni, V. VEGF and CD 34: A correlation between tumor angiogenesis and microvessel density-an immunohistochemical study. J. Oral Maxillofacial Pathol. 17 (3), 367–373 (2013).
Yang, T., Qiao, S. & Zhu, X. High-dose radiation-resistant lung cancer cells stored many functional lipid drops through JAK2/p-STAT3/FASN pathway. J. Cancer Res. Clin. Oncol. 149 (15), 14169–14183 (2023).
Tirinato, L. et al. Lipid droplets and ferritin heavy chain: a devilish liaison in human cancer cell radioresistance. Elife 10, e72943 (2021).
Rockfield, S. et al. Links between iron and lipids: implications in some major human diseases. Pharmaceuticals 11 (4), 113 (2018).
Li, N. et al. Elevated serum bilirubin levels are associated with improved survival in patients with curatively resected non-small-cell lung cancer. Cancer Epidemiol. 39 (5), 763–768 (2015).
Deng, C. C. et al. Unconjugated bilirubin is a novel prognostic biomarker for nasopharyngeal carcinoma and inhibits its metastasis via antioxidation activity. Cancer Prev. Res. (Phila). 9 (2), 180–188 (2016).
Keshavan, P. et al. Unconjugated bilirubin induces apoptosis in colon cancer cells by triggering mitochondrial depolarization. Int. J. Cancer. 112 (3), 433–445 (2004).
1 Chen, Z. et al. Bilirubin nanomedicines for the treatment of reactive oxygen species (ROS)-Mediated diseases. Mol. Pharm. 17 (7), 2260–2274 (2020).
Funding
This work was supported in part by the following grants and foundations: Yunnan Provincial Ten Thousand-Talent Program-Famous Doctor (YNWR-MY-2018-073); National Natural Science Foundation of China (81760430); Doctoral Research Project of the Second Affiliated Hospital of Kunming Medical University(2023BS03); Scientific Research Fund of Yunnan Provincial Department of Education (2024J0242). Applied Basic Research Joint special Fund project of Science and Technology Department of Yunnan Province and Kunming Medical University (202201AY070001-102); Yunnan Science and Technology Programme (202101AC070079);Kunming Medical University graduate Education Innovation Fund (2024S301).
Author information
Authors and Affiliations
Contributions
LC, S.Hu and XZ wrote the manuscript. S.Hu, LC, L.Liu and S.He conducted the experiments. LC, FS, HF and L.Li analyzed the data and collected the data. S.Hu, LC, L.Liu and XZ prepared the figures and conducted the statistical analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent for publication and consent to participate
Informed consent was obtained from all the participants involved in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, L., Hu, S., Liu, L. et al. Prognostic value of preoperative serum ferritin in cholangiocarcinoma patients. Sci Rep 15, 44288 (2025). https://doi.org/10.1038/s41598-025-27989-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-27989-5