Abstract
This study compared the performance of three rebound tonometers (iFalcon Tonovet, iCare Tonovet Plus, Reichert Tono-Vera Vet) for measuring intraocular pressure (IOP) in normal rabbit eyes, where a wide range of intraocular pressure (approx. 11–86 mmHg) was established and regulated via an intracameral needle connected to a saline manometer. Intraocular pressure measurements were performed on 10 eyes of New Zealand White rabbits using each of the three tonometers mentioned above. Measurements were taken at pressures ranging from 11 to 86 mmHg, yielding a total of 298 datasets. Statistical analysis of the intraocular pressure values obtained: iFalcon (32.5 ± 13.9 mmHg), iCare (33.6 ± 14.3 mmHg), Reichert (31.4 ± 12.0 mmHg) (p = 0.138). High overall agreement was observed (R2 0.95–0.98). However, trend differences existed: iFalcon slightly underestimated iCare, especially at higher IOP; Reichert read higher than iFalcon below 23 mmHg but lower above; Reichert also read higher than iCare below 18 mmHg but lower above. The novel iFalcon demonstrated stable performance. All three devices show good agreement for rabbit IOP monitoring, but clinicians should be aware of trend differences at low and high pressures, potentially applying corrections when switching devices during longitudinal studies.
Similar content being viewed by others
Introduction
Intraocular pressure (IOP) is a physiological parameter of critical importance in the assessment and management of ocular diseases1. Glaucoma, a chronic disease characterized by optic nerve damage and visual field defects due to elevated IOP, is a leading cause of irreversible blindness in humans and many veterinary species2. Indirect tonometry, such as applanation and rebound tonometers, is routinely used for non-invasive IOP measurement in clinical settings. Rebound tonometers are widely adopted in veterinary ophthalmology practice and research due to their ease of use, lack of requirement for corneal anesthesia, and suitability for small animals1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17. The principle involves an electromagnetically driven probe impacting the corneal surface, the IOP is determined based on the deceleration of the probe upon impact18. This rebound tonometry method was pioneered by Antti Kontiola in 1996. After extensive clinical validation, it is now widely used in ophthalmology.
Among commercially available rebound tonometers for animals, the iCare TonoVet Plus (hereafter referred to as iCare; Icare Finland Oy) (Fig. 1A) and the Reichert Tono-Vera Vet (hereafter referred to as Reichert; Reichert Technologies, USA) (Fig. 1C) are popular products. Their measurement accuracy has been extensively studied in dogs and other species, systematically validated against traditional manometry and applanation tonometers in vivo and ex vivo15,19,20. Studies indicate that several factors can influence IOP readings, such as the contact angle of the probe, the distance to the cornea, and central corneal thickness (CCT)9,21,22,23. Recently, a novel rebound tonometer, the iFalcon Tonovet (hereafter referred to as iFalcon; developed by iFalcon CO., Ltd), has entered the field of animal IOP measurement, providing a new option (Fig. 1B). Notably, this device represents a technical innovation: unlike conventional rebound tonometers relying on linear models, iFalcon employs a nonlinear physical model grounded in Hertz contact theory. By analyzing the dynamic indentation curve during probe-cornea impact and incorporating contact stiffness and impact energy into IOP calculations, this approach more physically represents the corneal impact process. Theoretically, it enables more accurate assessment of species-specific variations in corneal biomechanics.
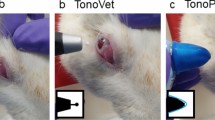
Measurement of IOP in a rabbit using rebound tonometers: (A) Novel iFalcon Tonovet. (B) iCare Tonovet Plus. (C) Reichert Tono-Vera Vet. When the tonometer is horizontally aligned with the cornea, the LED indicator light turns green for iFalcon and iCare, while Reichert displays a green horizontal line on the screen.
This study systematically evaluated the performance differences between the mainstream devices iCare and Reichert and the novel device iFalcon in normal rabbit eyes, utilizing a standardized post-mortem measurement protocol to eliminate in vivo confounding factors. However, the measurement consistency of these three devices in normal rabbits has not been systematically investigated, as previous studies have primarily focused on animals such as dogs and cats10,24. Knollinger et al. have demonstrated that rebound tonometers provide accurate estimates of intraocular pressure in clinically normal eyes of dogs and horses10. Given that rabbits are common experimental animals with distinct ocular structures (e.g., corneal thickness, globe size) differing from other species, directly applying results from other species may introduce bias.
This study aimed to compare the IOP measurements obtained with iFalcon, iCare, and Reichert in normal rabbits, analyzing the performance of these three rebound tonometers across a range of IOP values, to provide a basis for device selection in rabbit IOP measurement.
Results
Comparison of IOP measurements
A total of 298 valid IOP measurements were obtained (30 per eye; final valid data slightly less than 300 due to some measurements failing device validity criteria). The mean IOP ± SD was 32.5 ± 13.9 mmHg for iFalcon, 33.6 ± 14.3 mmHg for iCare, and 31.4 ± 12.0 mmHg for Reichert (Figs. 2, 3). No significant difference was found in the mean IOP measured by the three tonometers (p = 0.138). To ensure accuracy, Tukey’s simultaneous test was performed, confirming no significant differences in mean IOP between any pair of tonometers.
Box plots summarizing IOP measured with the three rebound tonometers. Overall means (± SD) were 32.5 ± 13.9 mmHg (iFalcon), 33.6 ± 14.3 mmHg (iCare), and 31.4 ± 12.0 mmHg (Reichert). Differences in mean IOP were non-significant, with iCare yielding the highest values, followed by iFalcon, and Reichert the lowest (p-value = 0.138, > 0.05). The box represents the interquartile range (IQR; middle 50% of data). The red center line is the median. The bottom and top of the box show the first and third quartiles, respectively. Outliers are shown as red circles.
Tukey’s simultaneous test for mean IOP differences. Black dots represent the mean difference between rebound tonometer pairs. The 95% confidence intervals (CI; horizontal lines) include zero (vertical dashed line). The differences iCare-Reichert (2.18 ± 1.06 mmHg; CI − 0.40, 4.76; p = 0.600), iFalcon-Reichert (1.12 ± 1.06 mmHg; CI − 1.46, 3.70; p = 0.567), and iFalcon-iCare (− 1.06 ± 1.06 mmHg; CI − 3.64, 1.52; p = 0.118) were all non-significant.
Bland-Altman analysis showed close agreement between the tonometers without systematic trends (Fig. 4). Linear regression analysis indicated good agreement between the devices, with R2 values ranging from 0.95 to 0.98 (Fig. 5). The highest R2 value (0.977) and closest agreement were observed between iFalcon and iCare. Despite good overall agreement, subtle but identifiable trend differences exist between the devices’ readings. Comparing iFalcon, iCare, and Reichert, the mean IOP measured by iFalcon lay between those measured by iCare and Reichert. iFalcon showed a slight underestimation compared to iCare, increasing with higher IOP. (Fig. 5A). When comparing iFalcon and Reichert, Reichert readings were slightly higher when IOP < 23 mmHg, while iFalcon readings were slightly higher when IOP > 23 mmHg (Fig. 5B). The difference between iCare and Reichert manifested as Reichert readings being higher when IOP < 18 mmHg and iCare readings higher when IOP > 18 mmHg. The determination of specific IOP values (18 mmHg, 23 mmHg) is the result after rounding the intersection point of the regression curve and the y=x reference line. Higher than the determination of specific IOP values is understood herein as high IOP as described, and lower than that as low IOP as described.
Bland–Altman plots comparing the three rebound tonometers. The estimated mean difference between two tonometers is represented by a black horizontal solid line. The 95% confidence interval (CI) is indicated by red dashed lines representing the upper (ULA) and lower (LLA) limits of agreement (mean ± 1.96 * SD). IOP measurements showed no systematic trend and were predominantly within the 95% CI. The estimated mean difference (± SD) was -1.06 ± 2.17 mmHg for iFalcon-iCare, 1.12 ± 3.26 mmHg for iFalcon-Reichert, and 2.18 ± 3.59 mmHg for iCare-Reichert.
Regression analysis of IOP between tonometers (including linear equations and R2 values). The solid black line indicates strong linear correlations exist between iFalcon-iCare and iFalcon-Reichert. The blue dashed lines mark the 95% prediction bands. Compared to the red dashed y = x line, iFalcon and iCare demonstrate good agreement. When comparing iFalcon and Reichert, iFalcon exhibits a tendency to underestimate lower IOP values and overestimate higher IOP values. The solid black line intersects the red dashed y = x line at an IOP value of approximately 23 mmHg.
Discussion
This study, through a systematic comparison of the novel iFalcon Tonovet, iCare Tonovet Plus, and Reichert Tono-Vera Vet rebound tonometers in normal rabbit eyes, is the first to clarify their characteristics of agreement and difference across a wide range of IOP values (11-86 mmHg). The core objective of this study is to evaluate the consistency and trend differences among different devices, especially in the performance within a wide range of intraocular pressures. We prefer to directly compare the three rebound tonometers cross-sect. It provides critical reference for device selection and clinical application in rabbit IOP measurement.
Rebound tonometers, particularly handheld designs, play a vital role in glaucoma diagnosis and management. For animal models (e.g., rabbit glaucoma research) and human patients, convenient, non-invasive IOP monitoring devices are key tools for early detection of IOP abnormalities and timely intervention. The portability of handheld devices is especially beneficial for achieving 24-hour dynamic IOP monitoring, which is irreplaceable for capturing IOP fluctuation peaks and comprehensively assessing glaucoma progression, overcoming the limitations of single clinic measurements7.
The repeated-measures ANOVA revealed no significant overall effect of the tonometer device on the IOP measurements (p = 0.138). Both Bland-Altman analysis and linear regression confirmed good overall agreement between them (R2 values 0.95–0.98). The highest agreement was between iFalcon and iCare (R2 = 0.977), likely attributable to their similar technical design and operation logic—both utilize LED lights for real-time probe angle feedback (iFalcon valid angle range approx. +5 to − 10 degrees), reducing measurement errors due to angular deviation. The agreement between iCare and Reichert (mean difference 2.18 ± 3.59 mmHg) may stem from Reichert’s advanced CMOS camera positioning system (precisely controlling distance: 6.0 ± 1.5 mm, angle: ±15 degrees), collectively ensuring measurement condition stability. This aligns with previous conclusions in other animals like dogs that “positioning aids enhance rebound tonometer agreement20.”
iFalcon Tonovet is mainly designed for large animals, including rabbits, cats, dogs, and horses. iFalcon Tonovet supports the vertical measurement mode. However, since the other products used for comparison in this study do not support vertical measurement, in this experiment, the horizontal measurement method was uniformly adopted and the vertical measurement function of iFalcon Tonovet was not demonstrated.
Notably, iFalcon’s innovation lies in its application of Hertz contact theory to nonlinearly model the probe-cornea interaction, departing from conventional linear approximations. Theoretically, this approach better accounts for interspecies variations in corneal stiffness, thereby enhancing measurement accuracy across diverse animal models. Practically, its capability for precise vertical alignment measurements of intraocular pressure constitutes a significant advantage, improving operational flexibility when examining uncooperative subjects or confined anatomical sites. However, further validation is required to establish the model’s applicability and precision across heterogeneous animal corneas.
The three tonometers demonstrated excellent agreement. The slight offsets and variations in intraocular pressure (IOP) values observed followed patterns consistent with those reported in previous studies on other animal species. Hodgson et al. compared the agreement among three rebound tonometers in both healthy dogs and dogs with glaucoma, which closely resembles our findings in healthy rabbits25. These trends may be related to differences in algorithm calibration within the device. Because different brands optimize rabbit eye biomechanical parameters (e.g., corneal elasticity, eyeball size) in different ways, resulting in response biases in the range of low and high intraocular pressure. The rebound tonometers of various brands are obtained in different species, and from a certain angle, it also reflects the good applicability of rebound tonometers in different species. And it has good repeatability under the operation of different measuring personnel. Icare Finland Oy, as the first brand to study rebound tonometers, iFalcon’s new rebound tonometer can achieve high consistency with iCare, which also reflects the advantages of iFalcon in using Hertz contact theory to model nonlinear probe-corneal interactions21,22,23.
This study utilized a standardized post-mortem measurement protocol. By performing immediate, alternate measurements on both eyes after euthanasia (30 measurements per eye completed within 15 min), confounding factors such as agitation and stress in live rabbits were effectively eliminated, which reduced intra-user variation and ensured data stability26. However, it should be noted that IOP may undergo a slight decline shortly after euthanasia (approx. 1–2 mmHg within 10 min). Given the extremely short interval between measurements with the three devices, their relative differences remain reliably informative.
The limitation of this study lies in the fact that the central corneal thickness was not measured. It is known that CCT is an important factor affecting the measurement values of tonometer. Future research should systematically evaluate the specific impact of corneal thickness and biomechanical properties on the measurement deviations of the three devices on live animal models. Furthermore, this study lacked a formal safety assessment, such as the evaluation of corneal epithelial lesions using fluorescein staining or histopathology.
Conclusion
This study confirms that iFalcon, iCare, and Reichert exhibit good overall agreement for IOP measurement in normal rabbit eyes, with the novel iFalcon device demonstrating stable performance. All three devices are suitable for long-term monitoring in clinical applications, holding positive significance, especially for achieving convenient and reliable 24-hour dynamic IOP monitoring. Future research should expand the rabbit sample, incorporate multi-operator comparisons, and utilize applanation tonometry as a reference standard to quantify device deviations. Particularly warranting exploration is the performance of the iFalcon nonlinear model in species with differing corneal biomechanics and in live dynamic monitoring scenarios.
Methods
Experimental animals
Five adult normal New Zealand white rabbits (3 females and 2 males) were selected, all with different genetic backgrounds. The median age is 6 months, with a range of 5–7 months. All rabbits were confirmed free of ocular disease (e.g., corneal lesions, lens opacities, glaucoma) by slit-lamp examination and indirect ophthalmoscopy. The rabbits were group-housed under identical environmental conditions at the Minsheng Fengda Life Science Park in Hangzhou, Zhejiang, China, on a 12-h light/12-h dark cycle and fed the same diet. The study adhered to the Association for Research in Vision and Ophthalmology (ARVO) Statement for the Use of Animals in Ophthalmic and Vision Research. The experimental protocol has been approved by IACUC and the Animal Management and Ethics Committee of Hangzhou Lifutai Biotechnology Co., Ltd., and complies with the “Guidelines for Ethical Review of Laboratory Animal Welfare” and “ARRIVE Guidelines”.
IOP measurements commenced immediately after euthanasia (performed according to AVMA Guidelines for the Euthanasia of Animals) to avoid interference from rabbit agitation and reduce stress during measurement. The experiment followed guidelines from the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC). Euthanasia was achieved via intravenous injection of sodium pentobarbital (100 mg/kg), and IOP measurements began within 5 min post-mortem.
Tonometry
Three rebound tonometers were used:
-
Novel iFalcon Tonovet (the rabbit setting)
-
iCare Tonovet Plus (Icare Finland Oy, the rabbit setting)
-
Reichert Tono-Vera Vet (Reichert Technologies, the rabbit setting)
All devices were calibrated by the manufacturer less than one year prior to the experiment. Following calibration, IOP was measured alternately on both eyes of freshly euthanized rabbits in the sequence iFalcon, iCare, Reichert. Shin et al. conducted relevant research data and statistics and found no statistically significant difference between the mean intraocular pressure in the right eye and the mean intraocular pressure in the left eye measured using a rebound tonometer. Therefore, this study will not distinguish between the left and right eyes27. Thirty IOP measurements were performed per eye per rabbit, resulting in 298 datasets (10 eyes × 30 rounds - 2 invalid datasets). A wide range of IOP values was generated using syringe manometry: the injection port was sealed with biological glue; IOP stabilized approximately 20 seconds after pressurization, and a set of measurements with all three devices was rapidly completed3,11,28,29.
For iFalcon and iCare, each measurement comprised 6 readings; the highest and lowest values were excluded, and the mean of the remaining 4 readings was recorded as the single measurement result. By default, Reichert averaged 3 readings after alignment; if the difference between readings exceeded 10%, additional readings were taken up to 6, and the mean of 3 qualifying readings was used25.
All measurements were performed by a single operator strictly according to the device manuals: iFalcon and iCare used LED lights to indicate probe angle validity (approximately +5 to -10 degrees). Reichert used its display to indicate distance (6±1.5 mm) and angle (±15 degrees). All data were collected between 3:00 PM and 10:00 PM over 6 days to minimize potential circadian rhythm effects on IOP21,26.
All three tonometers featured feedback mechanisms to ensure correct distance and probe alignment. iFalcon and iCare have displays indicating if the probe is too far or too close during each measurement; an LED on the probe tip indicates the tilt angle, turning green when within the valid range (approx. +5 to − 10 degrees) and red (preventing measurement) when outside. For both the iFalcon and iCare, the optimal probe-to-cornea distance is 4–8 mm. This range aligns with the manufacturers’ recommendations, and operators maintained a consistent distance during measurements9. Reichert’s ActiView positioning system uses a CMOS camera and two LEDs to actively guide the operator for correct alignment (Fig. 2)20. The display on the back of the instrument indicates the correct distance to the corneal apex (6.0 mm ± 1.5 mm) and centration. Additionally, a displayed probe angle indicator helps the operator maintain the probe angle within the horizontal tilt range (approx. − 15 to +15 degrees).
Statistics
Statistical analysis was performed using MATLAB software. A p-value < 0.05 was considered statistically significant. A repeated-measures ANOVA was performed to compare the overall differences in IOP readings among the three tonometers, with each rabbit eye treated as a subject and the measurements from the three devices as within-subject conditions. Tukey’s pairwise comparisons analyzed differences between specific device pairs. Bland-Altman plots assessed agreement between devices (calculating mean difference and 95% limits of agreement). Linear regression analysis evaluated the correlation between device readings (calculating R2 values).
Data availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
References
Komáromy, A. M. et al. The future of canine glaucoma therapy. Vet. Ophthalmol. 22(5), 726–740. https://doi.org/10.1111/vop.12678 (2019).
Weinreb, R. N., Aung, T. & Medeiros, F. A. The pathophysiology and treatment of glaucoma: A review. JAMA 311(18), 1901–1911. https://doi.org/10.1001/jama.2014.3192 (2014).
Barbosa, S. F. et al. TonoVet plus: Higher reliability and repeatability compared with Tono-Pen XL and TonoVet in rabbits. Vet. Ophthalmol. 25(4), 272–281. https://doi.org/10.1111/vop.12981 (2022).
Bertens, C. J. F. et al. Repeatability, reproducibility, and agreement of three tonometers for measuring intraocular pressure in rabbits. Sci. Rep. 11, 19217. https://doi.org/10.1038/s41598-021-98762-7 (2021).
Gloe, S., Rothering, A., Kiland, J. A. & McLellan, G. J. Validation of the Icare TONOVET plus rebound tonometer in normal rabbit eyes. Exp. Eye Res. 185, 107698. https://doi.org/10.1016/j.exer.2019.107698 (2019).
Pereira, F. Q., Bercht, B. S., Soares, M. G., da Mota, M. G. & Pigatto, J. A. Comparison of a rebound and an applanation tonometer for measuring intraocular pressure in normal rabbits. Vet. Ophthalmol. 14(5), 321–326. https://doi.org/10.1111/j.1463-5224.2011.00879.x (2011).
Ma, D. et al. Repeatability, reproducibility and agreement of intraocular pressure measurement in rabbits by the TonoVet and Tono-Pen. Sci. Rep. 6, 35187. https://doi.org/10.1038/srep35187 (2016).
Löbler, M. et al. Suitability and calibration of a rebound tonometer to measure IOP in rabbit and pig eyes. Vet. Ophthalmol. 14(1), 66–68. https://doi.org/10.1111/j.1463-5224.2010.00794.x (2011).
Rodrigues, B. D. et al. Intraocular pressure measurements using the TONOVET rebound tonometer: Influence of the probe-cornea distance. Vet. Ophthalmol. 24(Suppl 1), 175–185. https://doi.org/10.1111/vop.12832 (2021).
Knollinger, A. M., La Croix, N. C., Barrett, P. M. & Miller, P. E. Evaluation of a rebound tonometer for measuring intraocular pressure in dogs and horses. J. Am. Vet. Med. Assoc. 227(2), 244–248. https://doi.org/10.2460/javma.2005.227.244 (2005).
Wang, X., Dong, J. & Wu, Q. Twenty-four-hour measurement of IOP in rabbits using rebound tonometer. Vet. Ophthalmol. 16(6), 423–428. https://doi.org/10.1111/vop.12020 (2013).
Shim, J. et al. Comparative intraocular pressure measurements using three different rebound tonometers through in an ex vivo analysis and clinical trials in canine eyes. Vet. Ophthalmol. 24(Suppl 1), 186–193. https://doi.org/10.1111/vop.12771 (2021).
Iliev, M. E., Goldblum, D., Katsoulis, K., Amstutz, C. & Frueh, B. Comparison of rebound tonometry with Goldmann applanation tonometry and correlation with central corneal thickness. Br. J. Ophthalmol. 90(7), 833–835. https://doi.org/10.1136/bjo.2005.089870 (2006).
Oliveira, I. V. et al. Determination of intraocular pressure and Schirmer tear test and the comparison between the applanation tonometer (Tono-Pen AVIA) and the rebound tonometer (TonoVet Plus) in mini lionhead rabbits. Vet. Ophthal. 27(1), 53–60. https://doi.org/10.1111/vop.13150 (2024).
Zhang, H. et al. Validation of rebound tonometry for intraocular pressure measurement in the rabbit. Exp. Eye Res. 121, 86–93. https://doi.org/10.1016/j.exer.2014.02.004 (2014).
Takenaka, J., Mochizuki, H., Kunihara, E., Tanaka, J. & Kiuchi, Y. Evaluation of rebound tonometer for measuring intraocular pressure at deviated angle and position. Curr. Eye Res. 36(5), 422–428. https://doi.org/10.3109/02713683.2010.534574 (2011).
Lee, J. et al. Validation of recalibrated TONOVET plus and TONOVET pro in canine eyes compared to manometer and TONOVET. Vet. Ophthalmol. https://doi.org/10.1111/vop.70002.Advanceonlinepublication.doi:10.1111/vop.70002 (2025).
Kontiola, A. I. A new induction-based impact method for measuring intraocular pressure. Acta Ophthalmol. Scand. 78(2), 142–145. https://doi.org/10.1034/j.1600-0420.2000.078002142.x (2000).
Minella, A. L., Kiland, J. A., Gloe, S. & McLellan, G. J. Validation and comparison of four handheld tonometers in normal ex vivo canine eyes. Vet. Ophthalmol. 24(Suppl 1), 162–170. https://doi.org/10.1111/vop.12780 (2021).
Kapeller, L. E., Buckman, P. N., Wang, S. & Komáromy, A. M. Validation of the Reichert Tono-Vera Vet rebound tonometer in normal ex vivo canine eyes. Vet. Ophthalmol. 27(3), 290–293. https://doi.org/10.1111/vop.13213 (2024).
Garzón-Ariza, A., Guisado, A., Galán, A. & Martín-Suárez, E. Diurnal variations in intraocular pressure and central corneal thickness and the correlation between these factors in dogs. Vet. Ophthalmol. 21(5), 464–470. https://doi.org/10.1111/vop.12533 (2018).
de Oliveira, J. K., Montiani-Ferreira, F. & Williams, D. L. The influence of the tonometer position on canine intraocular pressure measurements using the Tonovet rebound tonometer. Open Vet. J. 8(1), 68–76. https://doi.org/10.4314/ovj.v8i1.12 (2018).
Guresh, A. M. et al. The effect of central corneal thickness on intraocular pressure values using various tonometers in the dog. Vet. Ophthalmol. 24(Suppl 1), 154–161. https://doi.org/10.1111/vop.12873 (2021).
Kiland, J. A. et al. Comparison of the TONOVET Plus, TonoVet, and Tono-Pen Vet tonometers in normal cats and cats with glaucoma. Vet. Ophthalmol. 26(5), 414–421. https://doi.org/10.1111/vop.13123 (2023).
Hodgson, K. J. et al. Comparison of three rebound tonometers in normal and glaucomatous dogs. Vet. Ophthalmol. 26(1), 31–38. https://doi.org/10.1111/vop.13043 (2023).
Lewin, A. C., Liu, C. C., Camacho-Luna, P., Alling, C. & Carter, R. T. Inter-user and intra-user variation of two tonometers in horses. Equine Vet. J. 52(4), 572–576. https://doi.org/10.1111/evj.13208 (2020).
Shin, Y. I. et al. Age-related changes of intraocular pressure in Dutch belted rabbits. Sci. Rep. 14, 12065. https://doi.org/10.1038/s41598-024-62097-w (2024).
Ben-Shlomo, G. & Muirhead, S. F. Estimation of intraocular pressure in normal canine eyes utilizing the newly introduced TonoVet Plus and TonoPen Avia, and their comparison to the established TonoVet. Vet. Ophthalmol. 24(Suppl 1), 171–174. https://doi.org/10.1111/vop.12747 (2021).
Mills, E. P. et al. Validation of a novel rebound tonometer (Tono-Vera Vet) in normal ex vivo rabbit eyes. Vet. Ophthalmol. 26(6), 555–559. https://doi.org/10.1111/vop.13142 (2023).
Acknowledgements
The authors gratefully acknowledge helpful discussions with Professor Z.C. Fan from Hunan University on the Hertzian contact method.
Funding
This work is supported by Science Foundation of Zhejiang Sei-Tech University (ZSTU) under Grant No. 24242151-Y.
Author information
Authors and Affiliations
Contributions
J F conceived the experiments, C W and Y S conducted the experiments, C W, J F and F S analyzed the results. All authors reviewed the manuscript. C W and Y S contributed to the work equally and should be regarded as co-first authors.
Corresponding author
Ethics declarations
Competing interests
Ji Fu is the Chief scientist of Hangzhou iFalcon Technology Co., Ltd. Chenghai Wang, Yunhao Su and Fushan Shi declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, C., Su, Y., Shi, F. et al. Comparison of three rebound tonometers in rabbits. Sci Rep 15, 44840 (2025). https://doi.org/10.1038/s41598-025-28990-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-28990-8