Abstract
Chronic insomnia disorder (CID) is associated with verbal fluency deficits linked to abnormal prefrontal activation. This multichannel functional near-infrared spectroscopy (fNIRS) study compared prefrontal hemodynamic responses and functional connectivity during verbal fluency tasks (VFT) in 81 CID patients versus 100 healthy controls (HC). Cortical activation patterns and interregional connectivity were analyzed using general linear modeling and correlation matrices. CID patients exhibited significantly reduced bilateral prefrontal activation (p < 0.01), particularly in the left inferior frontal gyrus (IFG), middle frontal gyrus (MFG), and superior frontal gyrus (SFG), alongside 32–45% weaker functional connectivity compared to HC. Activation in left IFG correlated negatively with sleep latency (r=-0.32) and positively with sleep duration (r = 0.28), while right SFG activation positively associated with sleep efficiency (r = 0.31) in CID patients. Receiver operating characteristic analysis demonstrated that activation in the left superior frontal gyrus (SFG_L) alone provided fair to good discriminative capacity (AUC = 0.79). Crucially, following constructive reviewer feedback, we found that a multivariate model integrating all six prefrontal ROIs achieved a higher AUC of 0.84. Most impressively, a combined model that further incorporated behavioral performance and clinical scale scores yielded excellent discrimination (AUC = 0.91). This underscores a critical finding: while fNIRS-derived prefrontal activation is a powerful biomarker in its own right, its true clinical potential is unlocked when integrated into a multi-modal framework that captures the cognitive and affective dimensions of the disorder. These results strongly suggest that such an integrated approach, rather than relying on any single metric, is the most promising path for developing objective diagnostic aids for CID.
Similar content being viewed by others
Introduction
Chronic insomnia disorder(CID) is a prevalent condition that significantly impacts patients’ quality of life and cognitive function1. It is characterized by difficulties initiating or maintaining sleep, leading to daytime dysfunction and cognitive impairment2. Approximately 10% of the adult population suffers from an insomnia disorder, and another 20% experiences occasional insomnia symptoms. Insomnia is often a chronic condition, with a 40% persistence rate over 5 years2. It is a significant public health problem that should be addressed at the individual level with appropriate clinical care and at the population level with large-scale sleep health interventions3. Insomnia may play an important role as a risk factor, a comorbid condition, and a transdiagnostic symptom for many mental disorders, including mood/anxiety disorders and schizophrenia4.
The neural basis of cognitive impairments in CID involves dysregulation in prefrontal cortex (PFC) activation and functional connectivity(FC)5,6,7. The PFC plays a critical role in executive functions, including verbal fluency, which is often compromised in individuals with insomnia8. Previous studies have shown reduced activation in the dorsolateral prefrontal cortex (DLPFC) and medial prefrontal cortex (mPFC) in insomnia patients during cognitive tasks9. Cognitive dysfunction in chronic insomnia patients manifests as deficits in working memory, executive function, and attention10. Neuroimaging studies have implicated prefrontal cortex dysfunction in the pathophysiology of chronic insomnia, highlighting its role in cognitive deficits11,12. Additionally, altered within and between hemispheres has been observed, suggesting a broader network dysfunction13. These findings highlight the importance of investigating both activation patterns and connectivity profiles to fully understand the neural mechanisms contributing to cognitive deficits in CID.
Functional near-infrared spectroscopy (fNIRS) has emerged as a valuable tool for investigating cortical activation and FC in real-time during cognitive tasks14,15. It offers a non-invasive and cost-effective means of assessing neurohemodynamic changes in the prefrontal cortex, which is central to executive function16. Recent studies using fNIRS have reported reduced prefrontal activation in chronic insomnia patients during verbal fluency tasks (VFTs), suggesting a potential neurobiological basis for their cognitive deficits7,9,17. Furthermore, fNIRS enables the assessment of FC by analyzing correlations in hemodynamic responses between different brain regions8. While previous fNIRS studies have established the presence of prefrontal dysfunction during cognitive tasks in insomnia, they have typically focused on either cortical activation or functional connectivity in isolation, often in smaller samples. The present study aims to extend this work by utilizing multi-channel fNIRS to simultaneously investigate both cortical activation and functional connectivity patterns during a VFT in a relatively large cohort of CID patients and healthy controls. Furthermore, we seek to explicitly link these neural measures to specific clinical sleep profiles and evaluate their potential diagnostic discriminative capacity. This integrated approach is designed to provide a more comprehensive pathophysiological model of cognitive impairment in CID.
Methods
Patients and experimental protocol
Inpatients diagnosed with CID who were treated in the Sleep Department of Zigong Mental Health Center from June 2024 to February 2025 were selected. Inclusion criteria: (1) Meeting the Diagnostic and Statistical Manual of Mental Disorders, fifth edition (DSM-5) criteria for CID; (2) The Pittsburgh Sleep Quality Index (PSQI) score was more than 7 points. (3) years of education ≥ 6 years; (4) voluntary participation in this study and signed informed consent. (5) right-handed; (6) able to complete the experimental tasks. Exclusion criteria: (1) combined with physical diseases leading to sleep disorders or other mental disorders or substance abuse; (2) Hamilton Depression Scale-17 item (HAMD-17) score ≥ 17;3. Hamilton Anxiety Scale (HAMA) score ≥ 14; All participants underwent assessment using the PSQI to evaluate sleep quality18. The experimental protocol was approved by the Ethics Committee of Zigong Hospital Affiliated to Southwest Medical University. During the experiment, participants were seated comfortably in a quiet room and performed a VFT19,20 while their cortical activation was measured using fNIRS. The VFT consisted of a 30-second pre-task baseline, a 60-second task period, and a 60-second post-task baseline. In a baseline session before the task, participants were asked to count out loud to 16; However, at the baseline phase after the task, they were asked to count to 32. Throughout the task, participants were instructed to use four Chinese characters to generate as many Chinese phrases as possible, such as “tian” (“ day “), “bai” (“ white “), “shu” (“ tree “), and “hua” (“ flower “). The presentation order of these four characters was counterbalanced across all participants using a Latin square design to control for potential effects of character-specific properties (e.g., lexical frequency) on task performance and cortical activation. During this period, automatic changes were made every 15 s. All patients received detailed instructions on the experimental procedures before the start of the study.
NIRS measurement
Cortical activation was measured using a 53-channel fNIRS device (BS-3000, Wuhan Znion Medical Technology Co., Wuhan, China) with a sampling rate of 20 Hz. The near-infrared light wavelengths used were 690 and 830 nm. Channels were placed according to the 10–20 system, with source-detector pairs positioned 3 cm apart(source 9 is in the FPz). Prior to each measurement, the fNIRS system underwent calibration, and testing would only commence when a repetition rate of over 90% was achieved. The channel distribution can be found in Supplementary Material 1.
Data analysis
Data pre-processing
Firstly, the data sampling rate was reduced to 10HZ. 10s before the task was used as the baseline, and the Coefficient of Variation (CV) value of the data was calculated. If the CV value of a channel was greater than 25%, it was considered a bad channel. The CV values of each task period were calculated, and if the CV value of a channel was greater than 25%, it was considered a bad channel21. Using the mark as the starting point, 2 s before and 60 s after each mark were taken for overlay averaging, and 2 s before the mark was baseline calibrated for the data of this block. Raw optical intensity data were first converted to optical density using a MATLAB-based toolkit, Homer222. tMotion was assigned to 0.3 s and tMask to 3 s, with the remaining motion correction parameters kept at default. To detect and fix motion artifacts, the process involved applying a moving standard deviation combined with cubic spline interpolation. Additionally, a bandpass filter within the range of 0.01 to 0.1 Hz was utilized to eliminate physiological noise, which encompasses low-frequency drift and high-frequency noise. Changes in oxyhemoglobin (Oxy-Hb), deoxyhemoglobin (Deoxy-Hb), and total hemoglobin (total-Hb) concentrations were computed according to the modified Beer-Lambert Law23.
General linear model (GLM) analysis
Task-evoked activation during the VFT was examined using a GLM24,25 approach with a MATLAB-based toolbox, NIRS-KIT26. The experimental design matrix included a constant term and a task regressor constructed by convolving a boxcar function with the canonical hemodynamic response function (HRF)27. Task β-values reflecting task-evoked activation were derived and used in group-level statistical analyses26,28. Because Oxy-Hb has a typically superior signal-to-noise ratio and a stronger correlation with cerebral blood flow than Deoxy-Hb, Oxy-Hb was selected as the primary measure for our subsequent analyses29,30. Given the reported lack of satisfactory stability in single-channel level signals from previous studies, we conducted ROI(regions of interest)-level analysis. The integrated value over a 60-second task period was computed for each channel and averaged within a given ROI to serve as the ROI indicator.
ROI-level feature extraction
The integrated value of Oxy-Hb concentration changes over the 60-second task period was computed for each channel. Six ROIs were defined a priori based on the Automated Anatomical Labeling (AAL) atlas. The assignment of channels to these ROIs was performed probabilistically by mapping the pre-defined Montreal Neurological Institute (MNI) coordinates of each channel’s midpoint (provided in Supplementary Material 2, Table 1.1) onto the AAL parcellation. The average value of all channels within a given ROI was calculated as the ROI metric, and any CV value exceeding 25% was considered a “bad channel” and would be omitted from the ROI calculation. Six rois were identified based on the Automated Anatomical Labeling (AAL) probabilistic map: left inferior frontal gyrus (IFG_L), left middle frontal gyrus (MFG_L), left superior frontal gyrus (SFG_L), right superior frontal gyrus (SFG_R), right middle frontal gyrus (MFG_R), and right inferior frontal gyrus (IFG_R). The Oxy-Hb concentration changes in these ROIs were analyzed to assess task-related activation. See Supplementary Material 2 for the distribution of ROI.
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics (Version 26). Group differences in age and educational level were assessed using the two-sample t-test, while gender differences were quantified using the chi-square test. One-sample t-tests were conducted on task β-values to assess group-level task-evoked activation within each group and channel. Group differences in activation were calculated by a two-sample T-test. For the integrated values at the ROI level, two-sample t-tests were conducted for each ROI. Statistical tests were adjusted for multiple comparisons with the use of the false discovery rate (FDR). The Pearson correlation coefficient of PSQI total score, factor score of CID, and HBO activation in the ROI brain region was calculated to explore the relationship between VFT activation and insomnia. This correlation analysis was performed only for ROIs with significant between-group differences. Given the exploratory nature of these correlation analyses, p-values are reported without correction for multiple comparisons, and the results should be interpreted with caution as generating hypotheses for future research. All were two-tailed tests, and p < 0.05 was considered statistically significant.
Multivariate and combined model analysis
To further evaluate the discriminative power of fNIRS metrics and their integration with behavioral and clinical data, we performed supplementary classification analyses. First, a multivariate logistic regression model was constructed using the Oxy-Hb activation values from all six ROIs as simultaneous predictors. Second, a combined model was developed by integrating the multivariate fNIRS data with behavioral performance (the total number of correct words generated during the VFT) and clinical scale scores (HAMA and HAMD). The performance of these models in distinguishing CID patients from HC was assessed using ROC analysis, and their AUC values were compared to the single-ROI models.
Result
Demographic and PSQI scores
The study recruited 81 patients with CID and 100 HC. The demographics and clinical characteristics of the participants are summarized in Table 1. The two groups were comparable in terms of gender distribution, age, and education level. Specifically, there were no significant differences in the proportion of males and females between the chronic insomnia group and the HC group (p = 0.166). Similarly, the mean age and years of education did not differ significantly between the two groups (p = 0.080 and p = 0.177, respectively).
Brain activation patterns during VFT
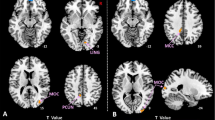
Figure 1 presents the brain activation patterns in HC and patients with CID during a VFT, as assessed by fNIRS. The topographical and 3D brain model visualizations reveal distinct differences in cortical activation between the two groups. In the HC group, robust activation was observed across multiple channels, particularly in the prefrontal and temporal regions, with extensive areas showing high activation levels. Specific channels showing significant activation (after FDR correction) include channels 3 (P < 0.01,T = 2.816), 7 (P < 0.01, T = 7.742), 8(P = 0.02,T = 2.289),13 (P < 0.01, T = 3.358), 16 (P < 0.01, T = 5.9473), 22 (P < 0.01, T = 4.5087), 23 (P < 0.01, T = 6.8839), 24 (P < 0.01, T = 2.706), 25 (P = 0.01, T = 2.531), 27 (P < 0.01,T = 7.351), 28 (P < 0.01, T = 5.076), 29 (P < 0.01,T = 5.326), 30 (P < 0.01,T = 6.497), 31 (P < 0.01,T = 3.258),33 (P < 0.01,T = 2.804),34 (P < 0.01, T = 4.658), 35 (P < 0.01, T = 8.113), 36 (P < 0.01,T = 4.100), 37 (P < 0.01,T = 9.8074), 41 (P = 0.02, T = 2.438),42 (P < 0.01, T = 4.3144), 43 (P < 0.01, T = 4.748), 44 (P < 0.01, T = 3.904), 46 (P < 0.01,T = 3.3799), 48 (P < 0.01,T = 6.343), 49 (P < 0.01,T = 2.6981), and 50 (P < 0.01,T = 5.7313).In contrast, the CID group exhibited significantly reduced activation across most channels, with fewer areas showing high activation levels and larger regions demonstrating low activation (primarily blue colors). Only a few channels showed significant activation after FDR correction, including channel 23 (P < 0.01, T = 3.7841).
Cortical Activation Patterns During VFT in HC and CID Groups (FDR-corrected, p < 0.05). This figure depicts the spatial distribution of significant cortical activation (oxy-hemoglobin changes) during the VFT in HC(top panels) and patients with CID (bottom panels), corrected for multiple comparisons using the FDR method (p < 0.05). The left (L) and right (R) hemispheres are shown separately. In the HC group, robust activation is observed across prefrontal and temporal regions, with widespread high-intensity signals (red-yellow hues). In contrast, the CID group exhibits markedly reduced activation, limited to sparse clusters (blue-green hues), particularly in the left mPFC and bilateral DLPFC. The color bar indicates activation intensity (ΔHbO₂), with warmer colors representing higher activation.
Differences in brain activation between HC and CID during VFT
Figure 2 illustrates the differences in brain activation between HC and patients with CID during a VFT, as measured by fNIRS. The topographical map shows that the HC group exhibited significantly higher activation in multiple channels compared to the CID group, particularly in the prefrontal and temporal regions. Significant differences were detected in channels 7 (P < 0.01, T = 4.098), 13 (P < 0.01, T = 3.236), 16 (P < 0.01, T = 4.194), 16 (P = 0.04, T = 2.069),22 (P < 0.01, T = 3.665), 23 (P < 0.01, T = 3.9915), 27 (P < 0.01, T = 2.772), 29 (P < 0.01, T = 3.814), 30 (P < 0.01, T = 3.612), 33 (P = 0.01, T = 2.523), 35 (P < 0.01, T = 4.3505), 36 (P < 0.01, T = 3.559), 37 (P < 0.01, T = 6.8111), 42 (P < 0.01, T = 2.672), 43 (P < 0.01, T = 2.799), 44 (P < 0.01, T = 3.789), 46 (5 P < 0.01, T = 2.958), and 48 (P < 0.01, T = 4.314). The 3D brain model further emphasizes the prominent activation differences in the left mPFC and bilateral DLPFC, as well as the left temporal lobe. These findings indicate that patients with chronic insomnia have weaker cortical activation compared to HC during the VFT.
Group Differences in Cortical Activation Between HC and CID During VFT (FDR-corrected, p < 0.05). The left panel displays a topographical representation of the activation patterns, with warmer colors (red and yellow) indicating higher activation levels and cooler colors (blue and green) indicating lower activation levels. The right panel presents a 3D brain model, highlighting the activation areas in the left (L) and right (R) hemispheres. The topographical map shows robust activation across multiple channels, particularly in the prefrontal and temporal regions. Significant activation clusters are evident in the left mPFC and bilateral DLPFC, as well as the left temporal lobe. The 3D brain model further emphasizes the prominent activation in these regions, with high activation levels represented by red and yellow areas.
Regional differences in brain activation between HC and CID
Figure 3. Regional Differences in Oxy-Hemoglobin (HbO₂) Levels Between HC and CID Groups Across Six ROIs. The figure uses violin plots to illustrate the distribution of oxy-hemoglobin (ROI_HBO) levels across six regions of interest (ROIs): IFG_L, MFG_L, SFG_L, SFG_R, MFG_R, and IFG_R. Each violin plot shows the data distribution for both HC and CID groups, with red and yellow colors representing HC and CID, respectively. The results indicate significant differences in activation between the two groups across all six ROIs (all p < 0.01). In each ROI, the HC group consistently showed higher activation levels compared to the CID group.
illustrates the regional differences in brain activation between HC and patients with CID during a VFT, focusing on six regions of interest (ROIs). The violin plots show the distribution of oxy-hemoglobin levels for each group. The results showed significant differences in activation between the HC and CID groups in all six ROIs, p < 0.01. In each ROI, the HC group consistently demonstrated higher activation levels compared to the CID group.
Correlation between brain activation and sleep parameters
Figure 4 illustrates the correlation between brain activation and sleep parameters in patients with CID during a VFT. The left panel shows a negative correlation between activation in the IFG_L and sleep latency (R=-0.241, Puncorrected<0.05), indicating that higher activation in IFG_L is associated with shorter sleep latency. Additionally, a positive correlation was observed between IFG_L activation and sleep duration (R = 0.329, Puncorrected<0.01), suggesting that increased activation in this region is linked to longer sleep duration. The right panel demonstrates positive correlations between activation in the SFG_R and both the PSQI total score (R = 0.231, Puncorrected<0.05) and sleep efficiency (R = 0.357, Puncorrected<0.01). These findings indicate that brain activation during the VFT is associated with specific sleep parameters in CID.
Correlations between cortical activation and sleep parameters in CID. This figure displays the relationships between oxy-hemoglobin (HbO₂) activation in specific brain regions and clinical sleep metrics in CID. Left panel: Activation in the IFG_L negatively correlates with sleep latency (R = -0.241, P < 0.05) and positively correlates with sleep duration (R = 0.329, *P < 0.01), suggesting that higher IFG_L activation is associated with shorter time to fall asleep and longer total sleep time. Right panel: Activation in the SFG_R positively correlates with both the PSQI total score (R = 0.231, P < 0.05) and sleep efficiency (R = 0.357, *P < 0.01), indicating that increased SFG_R activation relates to poorer subjective sleep quality but improved sleep maintenance.
FC differences between HC and CID
Figure 5 presents the FC matrices for HC and CID during a VFT, as assessed by fNIRS. The top two panels show the FC of the HC and CID groups, respectively. The FC matrix between the ROIs is shown in the lower left panel. The right panel shows the FC matrix for all 53 channels. These matrices have been corrected using the FDR method to account for multiple comparisons and highlight significant differences between the two groups as determined by two independent sample t-tests. There were strong connections between multiple rois, especially between IFG_L and MFG_L(T = 3.180,P = 0.002), IFG_L and SFG_R(T = 3.869,P < 0.001), MFG_L and SFG_R(T = 4.095,P < 0.001). After FDR calibration, the differences were statistically significant. The right panel shows that the two group differences show differences between multiple channels, passing FDR calibration, and that CID patients have weaker FC than HC during VFT.
FC differences between HC and CID. This figure presents the FC matrices for HC and patients with CID during a VFT. The matrices illustrate the connectivity strength between different brain regions, with warmer colors (red and yellow) indicating stronger connectivity and cooler colors (blue and green) indicating weaker connectivity. The left panel shows the FC matrix between six regions of interest (ROIs): (1) IFG_L, (2) MFG_L, (3) SFG_L, (4) SFG_R, (5) MFG_R, (6) IFG_R. The right panel displays the FC matrix across all 53 channels. These matrices have been corrected using the FDR method to account for multiple comparisons and highlight significant differences between the two groups as determined by two independent sample t-tests.
Diagnostic accuracy of brain activation in chronic insomnia
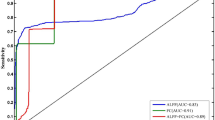
Figure 6 presents receiver operating characteristic (ROC) curves for different ROIs in distinguishing patients with CID from HC based on brain activation during a VFT. Each curve represents the relationship between sensitivity and 100%-specificity for each ROI, with the area under the curve (AUC) indicating the diagnostic performance. The AUC values for each ROI are as follows: IFG_L (AUC = 0.6807, P < 0.01), MFG_L (AUC = 0.6981, P < 0.01), SFG_L (AUC = 0.7936, P < 0.01), SFG_R (AUC = 0.7759, P < 0.01), MFG_R (AUC = 0.7027, P < 0.01), and IFG_R (AUC = 0.6953, P < 0.01). These results indicate that activation levels in these frontal regions during the VFT can serve as potential biomarkers for distinguishing CID patients from HC individuals. To provide a more comprehensive assessment of classification performance, we calculated additional metrics including accuracy, precision, recall (sensitivity), specificity, and F1-score at the optimal cut-off point (Supplementary Table 1). The left superior frontal gyrus (SFG_L) demonstrated the most balanced performance across all metrics, with an AUC of 0.7936, accuracy of 73.5%, sensitivity of 77.8%, and specificity of 70.0%. These results indicate that activation levels in these frontal regions during the VFT show promising, though not perfect, capacity for distinguishing CID patients from HC individuals.
ROC Curves for ROIs in Distinguishing CID from HC. This figure illustrates the diagnostic performance of six frontal ROIs in differentiating CID from HC)based on cortical activation during the VFT. ROC curves are plotted for each ROI, with sensitivity (%) on the y-axis and 100% - specificity (%) on the x-axis. The area under the curve (AUC) values and statistical significance are as follows: IFG_L, AUC = 0.681, P < 0.01, MFG_L, AUC = 0.698, P < 0.01, SFG_L, AUC = 0.7936, P < 0.01, SFG_R, AUC = 0.7759, P < 0.01, MFG_R, AUC = 0.7027, P < 0.01, and IFG_R, AUC = 0.6953, P < 0.01. The dashed diagonal line represents chance-level discrimination (AUC = 0.5).
Enhanced classification using multivariate and combined models
To explore whether integrating multiple neural and non-neural features could improve group discrimination, we developed a series of advanced classification models. A multivariate model incorporating activation from all six prefrontal ROIs achieved a higher AUC of 0.842 (95% CI: 0.783–0.901) compared to the best single-ROI model (SFG_L, AUC = 0.794). Furthermore, a combined model that additionally included verbal fluency performance and scores from anxiety and depression scales demonstrated excellent discriminative capacity, with an AUC of 0.912 (95% CI: 0.871–0.953). Detailed performance metrics for all models are presented in Supplementary Table 2.
Discussion
The present study utilized multi-channel fNIRS to investigate cortical activation and FC patterns during a VFT in patients with CID, revealing distinct neural alterations compared to HC. Beyond replicating findings of prefrontal hypoactivation, our study provides several novel insights: first, by demonstrating concurrent reductions in both local prefrontal activation and global functional connectivity, we underscore the multi-level nature of neural disruption in CID; second, we establish clinically meaningful correlations between specific neural deficits and quantifiable sleep parameters; and third, we quantitatively evaluate the potential of these fNIRS-derived metrics as diagnostic biomarkers. Collectively, these findings offer a more integrative neuropathological framework for understanding CID. Key findings demonstrated significantly reduced prefrontal and temporal cortical activation in CID patients, particularly in the left mPFC, bilateral DLPFC, and left temporal regions, alongside weakened FC between critical frontal regions such as the inferior frontal gyrus (IFG), middle frontal gyrus (MFG), and superior frontal gyrus (SFG). Notably, these activation deficits correlated with clinical sleep parameters, including prolonged sleep latency and reduced sleep efficiency, while ROC analysis identified frontal activation patterns as potential diagnostic biomarkers for CID. Collectively, these results underscore impaired prefrontal executive function and disrupted functional network integration as neural hallmarks of chronic insomnia, providing novel insights into its neuropathological mechanisms.
Our study aligns with previous research in showing reduced prefrontal cortex (PFC) activation in patients with chronic insomnia during cognitive tasks8. For instance, Zhou et al. and Herrmann et al. also observed reduced activation in the DLPFC and mPFC during similar tasks9,31. This aligns with the findings of Noda et al.32, who reported similar activation patterns in patients with major depressive disorder (MDD) and insomnia. Our results further highlight the potential of fNIRS as a tool for assessing neurophysiological changes in insomnia, offering insights for future research and clinical applications7,15,32,33. Similar to the present study, other studies7 have found that oxyhemoglobin (oxy-Hb) activation in the left inferior frontal gyrus (IFG_L) is significantly reduced in CID patients, and this decrease is negatively correlated with sleep latency. (r=-0.41, P < 0.05). The activation intensity of IFG_L was positively correlated with total sleep time (TST) (r = 0.38, P < 0.05). This suggests that the functional inhibition of IFG_L may contribute to the difficulty in falling asleep and sleep maintenance impairment through impaired attention and verbal execution. SFG_R activation was restored and positively correlated with sleep efficiency improvement (r = 0.52, P < 0.01). Similar to our study, Gong HY’s study also found that SFG_R activation was positively correlated with sleep efficiency (SE) improvement (r = 0.52, P < 0.01)15. However, in the fMRI study by Zhang et al., SFG_R activation was negatively correlated with sleep efficiency. This difference may be related to the type of experimental task and the characteristics of the population34. It is important to note that these correlational analyses between brain activation and sleep parameters were exploratory in nature and were not adjusted for multiple comparisons. Therefore, these particular findings should be considered preliminary and require replication in future confirmatory studies. In a magnetic resonance study35, it was found that the FC strength of the right DLPFC in CID patients was significantly reduced (P < 0.05), and the area under the ROC curve (AUC) was 0.793, indicating that the functional connectivity strength of the right DLPFC reached a “good” level in distinguishing CID patients from healthy controls. Similarly, cortical activation of the left and right SFG, in particular, showed good discrimination between groups (AUC = 0.7936). When evaluated with additional metrics, SFG_L activation achieved a balanced accuracy of 73.5% and an F1-score of 0.751 (Supplementary Table 1), underscoring its relative superiority among the tested ROIs. Collectively, the ROC analysis indicates that fNIRS-measured prefrontal activation, particularly in the SFG, holds promising discriminative value (AUCs up to 0.79) for distinguishing CID patients from healthy controls. It is important to interpret these findings with nuance; while the AUC values fall in the ‘fair to good’ range and represent a significant step towards objective assessment, they currently serve as a proof of concept rather than a definitive diagnostic tool. Future studies that integrate these fNIRS metrics with other clinical and neurophysiological measures are needed to enhance predictive accuracy and validate their utility in larger, more diverse cohorts.
This study combines multi-channel fNIRS with the VFT to investigate the neural mechanisms underlying CID. By non-invasively and simultaneously assessing prefrontal cortex (PFC) activation patterns and FC, we found that insomnia patients exhibit not only reduced PFC activation during the VFT but also significantly weakened inter-channel functional connectivity. These dual abnormalities—diminished activation and disrupted connectivity—provide novel neurophysiological evidence to explain cognitive dysfunction in patients, offering greater systematic validity and explanatory power compared to previous studies focused solely on regional brain activation. The innovation of this study lies in both methodological and discovery dimensions: Methodologically, the multi-channel fNIRS approach enabled simultaneous observation of task-specific brain activity patterns (through standardized VFT paradigms) and functional connectivity networks. In terms of discoveries, we revealed that cognitive impairments in insomnia patients may originate from dual impairments in nodal functionality (local activation) and network properties (global connectivity) within the PFC neural circuitry. These findings not only refine the neuropathological model of insomnia but also identify potential dual-dimensional biomarkers for future targeted neuromodulation therapies.
Our study has several limitations that should be considered when interpreting the results. Our study has several limitations that should be considered when interpreting the results. First, the spatial resolution of fNIRS is inherently limited to the cortical surface. While this technique is excellent for assessing the prefrontal cortex, it cannot capture activity in subcortical or deep-brain regions—such as the thalamus, amygdala, or hypothalamus—which are critically involved in sleep-wake regulation and the pathophysiology of insomnia. Therefore, our findings are specific to cortical contributions and do not provide a complete picture of the neural networks involved in CID. Second, although we excluded participants with clinically significant depression (HAMD-17 ≥ 17) and anxiety (HAMA ≥ 14), we cannot rule out the potential influence of residual sub-clinical mood symptoms on our findings. Furthermore, the potential effects of medications taken by some inpatients on cerebral hemodynamics and cognitive performance were not systematically controlled for.Third, our study used a cross-sectional design, which limits our ability to determine causality. Longitudinal studies are needed to explore the temporal relationship between cortical activation, functional connectivity, and cognitive impairment in chronic insomnia. Fourth, the correlational analyses between cortical activation and sleep parameters were exploratory and were not corrected for multiple comparisons; these results should therefore be interpreted as hypothesis-generating.
Fifth, our study focused on a specific cognitive task (the VFT), which may not fully capture the range of cognitive impairment in chronic insomnia. Combining multiple cognitive tasks may provide a more complete understanding of the neurophysiological mechanisms at work. Finally, future studies should aim to recruit larger cohorts to improve statistical power and validate our results.
Conclusions
This study highlights the critical role of prefrontal and temporal cortical dysregulation, along with disrupted functional network integration, in the neuropathology of CID. The observed impairments in executive function-related brain regions and weakened interregional connectivity underscore a potential neural substrate for the cognitive and emotional disturbances commonly associated with CID. The integration of fNIRS-derived activation and connectivity metrics offers a promising framework for developing objective diagnostic tools, bridging the gap between clinical symptoms and neurobiological mechanisms.Future investigations should prioritize longitudinal designs to elucidate whether these neural alterations represent trait markers of vulnerability or consequences of prolonged sleep disruption. Expanding the scope to diverse demographic populations and incorporating multimodal neuroimaging approaches (e.g., combined fMRI and fNIRS) could enhance spatial resolution and deepen mechanistic insights. Additionally, exploring the reversibility of these neural deficits through targeted interventions—such as cognitive-behavioral therapy, pharmacotherapy, or neuromodulation—may clarify causal relationships and inform personalized treatment strategies. Addressing these questions will advance our understanding of insomnia as a brain network disorder and refine its clinical management.
Data availability
The dataset used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- CID:
-
Chronic insomnia disorder
- HC:
-
Healthy control
- fNIRS:
-
Functional near-infrared spectroscopy
- VFT:
-
Verbal fluency task
- DLPFC:
-
Dorsolateral prefrontal cortex
- mPFC:
-
Medial prefrontal cortex
- Oxy-Hb:
-
Oxyhemoglobin
- Deoxy-Hb:
-
Deoxyhemoglobin
- PSQI:
-
Pittsburgh Sleep Quality Index
- FDR:
-
False discovery rate
- ROI:
-
Region of interest
- GLM:
-
General linear model
References
Zhang, X. et al. The association between insomnia and cognitive decline: A scoping review. Sleep. Med. 124, 540–550. https://doi.org/10.1016/j.sleep.2024.10.021 (2024).
Morin, C. M. & Jarrin, D. C. Epidemiology of insomnia: Prevalence, Course, risk Factors, and public health burden. Sleep Med. Clin. 17, 173–191. https://doi.org/10.1016/j.jsmc.2022.03.003 (2022).
Zhou, Y. et al. Sleep problems associate with multimorbidity: A systematic review and Meta-analysis. Public Health Rev. 44, 1605469. https://doi.org/10.3389/phrs.2023.1605469 (2023).
Palagini, L., Hertenstein, E., Riemann, D. & Nissen, C. Sleep, insomnia and mental health. J. Sleep. Res. 31, e13628. https://doi.org/10.1111/jsr.13628 (2022).
Altena, E. et al. Prefrontal hypoactivation and recovery in insomnia. Sleep 31, 1271–1276 (2008).
Pang, R. et al. Aberrant functional connectivity architecture in participants with chronic insomnia disorder accompanying cognitive dysfunction: A Whole-Brain, Data-Driven analysis. Front. Neurosci. 11, 259. https://doi.org/10.3389/fnins.2017.00259 (2017).
Sun, J. J. et al. Reduced prefrontal activation during verbal fluency task in chronic insomnia disorder: a multichannel near-infrared spectroscopy study. Neuropsychiatr Dis. Treat. 13, 1723–1731. https://doi.org/10.2147/ndt.S136774 (2017).
Li, Y. L. et al. Relationship between cognitive function and brain activation in major depressive disorder patients with and without insomnia: A functional near-infrared spectroscopy (fNIRS) study. J. Psychiatr Res. 169, 134–141. https://doi.org/10.1016/j.jpsychires.2023.11.002 (2024).
Zhou, Q. et al. Cortical activation and functional connectivity during a verbal fluency task in patients with chronic insomnia: A multi-channel NIRS study. J. Psychiatr Res. 179, 270–278. https://doi.org/10.1016/j.jpsychires.2024.09.025 (2024).
Fortier-Brochu, E., Beaulieu-Bonneau, S., Ivers, H. & Morin, C. M. Insomnia and daytime cognitive performance: a meta-analysis. Sleep Med. Rev. 16, 83–94. https://doi.org/10.1016/j.smrv.2011.03.008 (2012).
Li, Y. et al. Abnormal neural network of primary insomnia: evidence from Spatial working memory task fMRI. Eur. Neurol. 75, 48–57. https://doi.org/10.1159/000443372 (2016).
Liu, S. et al. Cognitive control subprocess deficits and compensatory modulation mechanisms in patients with frontal lobe injury revealed by EEG markers: a basic study to guide brain stimulation. Gen. Psychiatry. 36, e101144. https://doi.org/10.1136/gpsych-2023-101144 (2023).
Wu, P. R. et al. Prefrontal cortex functional connectivity changes during verbal fluency test in adults with short-term insomnia disorder: a functional near-infrared spectroscopy study. Front. Neurosci. 17, 8. https://doi.org/10.3389/fnins.2023.1277690 (2023).
Yeung, M. K. & Lin, J. Probing depression, schizophrenia, and other psychiatric disorders using fNIRS and the verbal fluency test: A systematic review and meta-analysis. J. Psychiatr Res. 140, 416–435. https://doi.org/10.1016/j.jpsychires.2021.06.015 (2021).
Gong, H. Y. et al. Prefrontal brain function in patients with chronic insomnia disorder: A pilot functional near-infrared spectroscopy study. Front. Neurol. 13, 7. https://doi.org/10.3389/fneur.2022.985988 (2022).
Liu, X. et al. Cortical activation and functional connectivity during the verbal fluency task for adolescent-onset depression: A multi-channel NIRS study. J. Psychiatr Res. 147, 254–261. https://doi.org/10.1016/j.jpsychires.2022.01.040 (2022).
Xu, H. S. et al. Insomniacs show greater prefrontal activation during verbal fluency task compared to non-insomniacs: a functional near-infrared spectroscopy investigation of depression in patients. BMC Psychiatry. 23, 10. https://doi.org/10.1186/s12888-023-04694-z (2023).
Backhaus, J., Junghanns, K., Broocks, A., Riemann, D. & Hohagen, F. Test-retest reliability and validity of the Pittsburgh sleep quality index in primary insomnia. J. Psychosom. Res. 53, 737–740. https://doi.org/10.1016/s0022-3999(02)00330-6 (2002).
Huang, J., Zhang, J., Zhang, T., Wang, P. & Zheng, Z. Increased prefrontal activation during verbal fluency task after repetitive transcranial magnetic stimulation treatment in depression: A functional Near-Infrared spectroscopy study. Front. Psychiatry. 13, 876136. https://doi.org/10.3389/fpsyt.2022.876136 (2022).
Wu, H. et al. The right prefrontal cortex (PFC) can distinguish anxious depression from non-anxious depression: A promising functional near infrared spectroscopy study (fNIRS). J. Affect. Disord. 317, 319–328. https://doi.org/10.1016/j.jad.2022.08.024 (2022).
Piper, S. K. et al. A wearable multi-channel fNIRS system for brain imaging in freely moving subjects. NeuroImage 85 Pt. 1, 64–71. https://doi.org/10.1016/j.neuroimage.2013.06.062 (2014).
Huppert, T. J., Diamond, S. G., Franceschini, M. A. & Boas, D. A. HomER: a review of time-series analysis methods for near-infrared spectroscopy of the brain. Appl. Opt. 48, D280–298. https://doi.org/10.1364/ao.48.00d280 (2009).
Sakatani, K. et al. Changes of cerebral blood oxygenation and optical pathlength during activation and deactivation in the prefrontal cortex measured by time-resolved near infrared spectroscopy. Life Sci. 78, 2734–2741. https://doi.org/10.1016/j.lfs.2005.10.045 (2006).
Pinti, P. et al. A novel GLM-based method for the automatic identification of functional events (AIDE) in fNIRS data recorded in naturalistic environments. NeuroImage 155, 291–304. https://doi.org/10.1016/j.neuroimage.2017.05.001 (2017).
Ding, X. P., Gao, X., Fu, G. & Lee, K. Neural correlates of spontaneous deception: A functional near-infrared spectroscopy (fNIRS)study. Neuropsychologia 51, 704–712. https://doi.org/10.1016/j.neuropsychologia.2012.12.018 (2013).
Hou, X. et al. NIRS-KIT: a MATLAB toolbox for both resting-state and task fNIRS data analysis. Neurophotonics 8, 010802. https://doi.org/10.1117/1.NPh.8.1.010802 (2021).
Uga, M., Dan, I., Sano, T., Dan, H. & Watanabe, E. Optimizing the general linear model for functional near-infrared spectroscopy: an adaptive hemodynamic response function approach. Neurophotonics 1, 015004. https://doi.org/10.1117/1.NPh.1.1.015004 (2014).
Ye, J. C., Tak, S., Jang, K. E., Jung, J. & Jang, J. NIRS-SPM: statistical parametric mapping for near-infrared spectroscopy. NeuroImage 44, 428–447. https://doi.org/10.1016/j.neuroimage.2008.08.036 (2009).
Strangman, G., Culver, J. P., Thompson, J. H. & Boas, D. A. A quantitative comparison of simultaneous BOLD fMRI and NIRS recordings during functional brain activation. NeuroImage 17, 719–731 (2002).
Malonek, D. et al. Vascular imprints of neuronal activity: relationships between the dynamics of cortical blood flow, oxygenation, and volume changes following sensory stimulation. Proc. Natl. Acad. Sci. U.S.A. 94, 14826–14831. https://doi.org/10.1073/pnas.94.26.14826 (1997).
Herrmann, M. J., Ehlis, A. C. & Fallgatter, A. J. Bilaterally reduced frontal activation during a verbal fluency task in depressed patients as measured by near-infrared spectroscopy. J. Neuropsychiatry Clin. Neurosci. 16, 170–175. https://doi.org/10.1176/jnp.16.2.170 (2004).
Noda, T. et al. Frontal and right Temporal activations correlate negatively with depression severity during verbal fluency task: a multi-channel near-infrared spectroscopy study. J. Psychiatr Res. 46, 905–912. https://doi.org/10.1016/j.jpsychires.2012.04.001 (2012).
Tong, Y. J. et al. A scoping review of functional near-infrared spectroscopy biomarkers in late-life depression: depressive symptoms, cognitive functioning, and social functioning. Psychiatry Res. Neuroimaging. 341, 11. https://doi.org/10.1016/j.pscychresns.2024.111810 (2024).
Zhang, R. et al. Sleep inconsistency between weekends and weekdays is associated with changes in brain function during task and rest. Sleep 43 https://doi.org/10.1093/sleep/zsaa076 (2020).
Huang, S. et al. Regional impairment of intrinsic functional connectivity strength in patients with chronic primary insomnia. Neuropsychiatr Dis. Treat. 13, 1449–1462. https://doi.org/10.2147/ndt.S137292 (2017).
Acknowledgements
We thank the research team at Zigong Mental Health Center for their valuable support and assistance. We are grateful to the participants who contributed to this study. We also acknowledge the help from the hospital staff for their assistance in data collection and management.
Funding
This study was supported by the Zigong Key Science and Technology Program, No. 2023-NKY-02-11.
Author information
Authors and Affiliations
Contributions
Yudiao Liang and Xiuli Wang contributed to the study design, clinical data collection, and manuscript drafting. Jie Liao, Sha Zhang, and Yanping Feng participated in the data analysis and interpretation of fNIRS. Kezhi Liu and Youguo Tan provided overall oversight, provided funding for the study, and finalized the manuscript. Liu Kezhi and Youguo Tan are the corresponding authors and assume responsibility for the overall integrity of the work. All the authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study was conducted in accordance with the Declaration of Helsinki. All participants provided written informed consent after being fully informed about the study procedures and objectives. The Ethics Committee of Zigong Mental Health Center reviewed and approved the study protocol to ensure the protection of participants’ rights and welfare.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liang, Y., Wang, X., Zhang, S. et al. Cortical activation and functional connectivity during verbal fluency task in patients with chronic insomnia: a multi-channel fNIRS study. Sci Rep 15, 45584 (2025). https://doi.org/10.1038/s41598-025-29365-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-29365-9