Abstract
The efficacy and safety of transdermal dissolvable microneedle array patch (MNAP) in enhancing topical anesthesia delivery was studied in transfusion-dependent thalassemia children who had intravenous cannulation (IVC) for blood transfusion. We also compared the use of skin conductance algesimeter index (SCAI) as a pain assessment tool against the visual analog score (VAS) in these children with ages between 6 and 17 years, who had the topical anesthetic cream (EMLA) applied on the hand dorsum before IVC. This was a double-blind, randomized crossover trial involving 19 children with each child randomized to four intervention arms sequentially: MNAP and 1 finger-tip-unit (FTU) EMLA applied for 30 min (A), MNAP and 0.5 FTU EMLA for 30 min (B); MNAP and 1 FTU EMLA for 15 min (C); and no MNAP (sham) patch and 1FTU EMLA for 30 min (D), before IVC. The primary and secondary endpoints were, respectively, VAS and SCAI scores measured immediately after IVC. The MNAP with 1 FTU EMLA for 30 min resulted in significantly lower VAS scores than sham patch [MeanMNAP (SE),1.839 (0.560) vs. [Meansham (SE): 3.063 (0.534); p = 0.048], but not SCAI [MeanMNAP (SE): 0.200 (0.024) versus Meansham (SE): 0.203 (0.026); p = 0.926]. The SCAI was not significantly correlated with VAS for all groups (r = -0.040 to 0.378; p (all) > 0.05). Only 1 participant reported pruritus as a mild adverse event. The MNAP is a safe adjunct method to enhance effectiveness of pain relief from EMLA skin anesthetic before IVC.
Similar content being viewed by others
Introduction
Thalassemia is one of the most commonly inherited hematological conditions afflicting Malaysian children. It is estimated that thalassemia affects 2.1 per 1000 live births, and the majority of them are blood-transfusion dependent and on chelation therapy1,2. Due to the maturational process in the physiology of the neurophysiological system, children may be more vulnerable to acute and long term procedural-related pain, which may lead to avoidance behavior and later poor therapeutic compliance3,4. Topical anesthesia in the form of pre-procedural skin application of local anesthetic agents such as the eutectic mixture of lidocaine and prilocaine (EMLA) is the standard pain relief method before skin puncture, but its effectiveness is time-dependent and likely compromised in a busy clinic setting. Thus, a shorter time from application to the desired anesthetic effect will not only ensure patient satisfaction but also optimize operational efficiency. An innovative transdermal delivery system is warranted to better render relief of pain from frequent intravenous cannulation (IVC) to transfuse blood for thalassemia children. Advancements in microfabrication technology have revolutionized the transdermal drug delivery system through the creation of microneedles. A MNAP consists of hundreds of micron-sized, out-of-plane projections that resemble hypodermic needles. The microneedles perturb the epidermal stratum corneum (SC) layer slightly by forming numerous transcutaneous conduits, thus improving the skin permeability for better drug absorption and enhanced therapeutic benefits. Biocompatible MNAP has recently been advocated for pediatric patients6. Moreover, MNAP is also cost-effective with minimal adverse effects, making it a suitable alternative for rapid transdermal drug delivery in children. Besides, maltose, a natural carbohydrate, has excellent biodegradability and well-recognized safety profiles, making it extensively utilized in pharmaceutical formulations7,8.
Besides, to evaluate pain relief effectiveness, a reliable pain scoring system is required. An alternative, more objective pain assessment modality that does not rely on the variable, age-dependent cognitive development and nociceptive experience of children is thus preferred. This could potentially reduce the subjectivity and challenges associated with the commonly used and established self-reported pain assessment tools such as the Visual Analog Scale (VAS) and Revised Faces Pain Scale5,6,7,8,9,10,11. Recently, an innovative objective pain assessment apparatus, the Pain Monitor device has been developed. It detects cutaneous electrical conductance alterations that are caused by nociception-induced hidrosis12. By continuously capturing the serial activation of the skin sympathetic nervous system, pain intensity is measured by the nociceptive nerve firing rate (skin conductance algesimeter index, SCAI), quantified in microSiemens per second (µS/s) as the response to a painful stimulus. However, the correlation between the objective SCAI measurement and the subjective VAS scale for pain assessment has not been sufficiently investigated in pediatric patients.
In this trial, we hypothesized that the biocompatible, biodegradable, and dissolvable maltose-based microneedle array patch (MNAP), when used as an adjunct to topical EMLA cream application, is safe and provides more effective pain relief during intravenous cannulation for blood transfusion in children with thalassemia. In addition, we included an exploratory secondary objective to examine whether the skin conductance algesimeter index (SCAI), an objective physiological pain indicator, could complement or correlate with the traditional Visual Analogue Scale (VAS) in pediatric pain assessment.
Methods
Study design
This was a phase II randomized, active-controlled, crossover, exploratory clinical trial involving children with transfusion-dependent thalassemia requiring regular IVC to transfuse blood at the Hospital Canselor Tuanku Muhriz (HCTM), Malaysia, between September 2021 and August 2022. The trial was approved by the Universiti Kebangsaan Malaysia (UKM) Research Ethics Committee (Approval reference and date: UKM PPI/111/8/JEP-2021-578–19/08/2021) and registered in ClinicalTrials.gov (NCT05078463; Registration date: 20/09/2021) as well as the Malaysian National Medical Research Registry (NMRR) (NMRR-ID-21-01989-47G; Registration date: 22/11/2021). All study procedures were conducted in accordance with the ethical principles of medical research as laid out by the World Medical Association (WMA)’s Declaration of Helsinki. The trial participants were not involved in the trial design, conduct and reporting of the trial findings.
Eligibility criteria
The inclusion criteria were children with thalassemia between the ages of 6 and 17 years requiring regular IVC to be transfused blood, whilst those with history of allergy and sensitization to EMLA or other study materials [e.g., maltose, polyvinyl alcohol (PVA), polyethylene terephthalate (PET)], exposed to any form of anesthesia within 24 h before the trial, or with generalized skin disorders and not cooperative or assenting were excluded.
Investigated medical products and methods of administration
Solid maltose microneedle fabrication
The MNAP (Hayashibara, Okayama, Japan) size approximates 10 mm × 10 mm, contains 36 microneedles with 1 mm needle gap between each of them. The MN dimension was designed to be approximately 400 μm in height, with a base width of 100 μm and a 3 μm tip radius. The standard deviation of needle heights within the patch was controlled to be less than 3%. The total patch thickness is, therefore, 0.8 mm.
The microneedles were grown on a soft cushion made of PVA material (Kanto Chemical, Tokyo, Japan), with a PET patch (Acrysunday, Tokyo, Japan) supporting the soft PVA patch. The base patch spans 125% more than the MNAP, with an estimated patch size of 15 mm × 15 mm. The entire patch size is 1.7 cm (width) × 4 cm (length) × 0.5 cm (height) (Fig. 1A), with the microneedle array and individual microneedle morphology shown in the magnified optical (Fig. 1B) and scanning electron microscopy (SEM) images (Fig. 1C), respectively. The details and configuration of the MNAP have been previously published in our trial protocol13.
The maltose-based microneedle array patch (MNAP). (A) The transparent patch contains multiple microscopic maltose microneedles arranged in a uniform grid on a polyvinyl alcohol-polyethylene terephthalate (PVA-PET) base. (B) Magnified view (approximately 10X) of a selected region from the same patch shown in A, highlighting the uniform microneedle geometry and consistent alignment across the array. Each microneedle exhibits a sharp, tapered tip and smooth sidewalls, designed to minimize insertion force and achieve reliable penetration through the stratum corneum. (C) Scanning electron microscopy (SEM) images of individual maltose microneedles showing well-defined conical tips and uniform height. Each microneedle measures approximately 400 μm in height and is designed to penetrate stratum corneum and viable epidermis without reaching dermal nociceptors.
Methods of skin analgesic intervention
This trial uses EMLA cream (AstraZeneca, Cheshire, UK) as the topical anesthesia. EMLA is a eutectic emulsion mixture of lidocaine and prilocaine in a 1:1 ratio (each gram of EMLA contains 25 mg lidocaine and prilocaine each). The EMLA anesthetic efficacy was evaluated based on the pain scores recorded immediately after IVC.
EMLA cream was applied for approximately 30 min before IVC, which reflects common practice in our clinical setting due to our high patient turnover with a large number of children requiring timely IVC on the same day, despite the manufacturer’s recommendation of a 60-minute application time for full dermal anesthesia14. Besides, the shorter application time is supported by previous research, which has demonstrated that meaningful dermal analgesia can be achieved with 30 min of EMLA application15,16,17 The trial participants received their blood transfusion based on the national guidelines18. Each participant was randomized to one of the 19 intervention sequences mentioned in our published protocol13. There was a four-week washout period before administering the subsequent interventions (Fig. 2).
The trial workflow showing four representative randomization sequences to illustrate how the crossover trial was conducted. A total of 19 unique randomization sequences were generated for the trial, but only four are displayed here for simplicity of visual presentation. Intvn: Intervention. Invtn A: MN + 1 FTU EMLA 30 min application; Intvn B: MN + 0.5 FTU EMLA 30 min application; Intvn C: MN + 1 FTU EMLA 15 min application; Intvn D: Sham patch + 1 FTU EMLA 30 min (control).
The four-week washout period corresponded to the participants’ routine monthly blood transfusion schedule, ensuring that each intervention was performed at a separate transfusion visit. This interval was deemed sufficient to eliminate any carry-over effects from the preceding cannulation, as procedural pain sensitization in children typically resolves within a few days.
The interventionist first marked a grid area of 1 cm × 1 cm at the dorsum of the hand, which had been identified as the IVC site. The MNAP was pressed firmly by thumb force within the marked grid area for 5 s before EMLA application. The sensor probe of the Pain Monitor (Med-Storm Innovation AS, Oslo, Norway) was applied to the participant’s palm of the opposite hand to record the baseline SCAI before the cannulation procedure began (Fig. 3A). The interventionist, who was not a trial investigator, subsequently applied the EMLA cream according to the assigned quantity, 1 FTU (approximately 0.68 g/cm2) or 0.5 FTU (approximately 0.369 g/cm2) shortly after the MNAP application. For the no-MNAP or sham group, an empty PVA-containing PET sham patch was also firmly pressed by thumb force for 5 s to maintain the same tactile similarity and masking integrity. The skin area of intervention was subsequently covered with an adhesive dressing (Tegaderm; 3 M, Maplewood, MA, USA) after EMLA cream application for either 15–30 min based on the allocated group. The attending healthcare personnel (healthcare provider) then cleaned and inserted the IVC (22Fr-gauge cannula) as per the usual practice, without knowledge of the assigned intervention.
Methods and tools used for pain assessment. (A) Application of the sensor probe to assess pain based on the skin conductance algesimeter index (SCAI) using the Pain Monitor. (B) The Visual Analog Scale (VAS) Ruler and the Pain Scores between the range of 0 and100 mm. No pain: 0–4 mm, Mild pain: 5–44 mm, Moderate pain: 45–74 mm, Severe pain: 75–100 mm.
Consent, randomization and blinding
Parental written informed consent and the child’s assent were obtained after the trial eligibility assessment at the pediatric daycare ward of HCTM. Simple randomization was performed by the trial statistician using the randomizer package version 20.0 implemented on the R platform. A total of nineteen possible randomization sequences were generated by the trial statistician and each sequence represented a distinct order of the four intervention arms in the crossover design13. The randomization list was concealed from all trial investigators, including those who screened for eligible subjects to be enrolled in the study. After consent and assent were obtained, the recruiting staff contacted the statistician by telephone, and the participant was then assigned to one of the pre-generated sequences according to the randomization list. This procedure preserved allocation concealment and prevented any foreknowledge of group assignment by the investigators or clinical staff. The participants, care providers, and outcome assessors were masked, whilst the statistician and interventionist were unmasked to the interventions assigned to the participants. The interventionist and the care provider were different individuals. The interventionist applied the MNAP or sham patch according to the randomization list, whereas the care provider performed the intravenous cannulation without knowledge of the assigned intervention. The MNAP and PVA-containing PET sham patch are identical in shape, size, color, thickness and adhesiveness and were applied with the same thumb pressure for five seconds. Besides, the microneedles were only 400 μm in height and did not produce any noticeable discomfort, thereby preserving tactile similarity and ensuring masking integrity among participants and assessors.
Baseline and background information retrieval
Upon enrolment and before the administration of trial interventions, baseline clinical and demographic profiles (age, gender, ethnicity, thalassemia inheritance, comorbidities, anthropometric measurements) of the participants were collected and recorded in a case report form (CRF).
Data collection and pain assessment tools
To lessen inter-rater variability, a single investigator (rater) performed all endpoint measurements and data collection. The rater was not tasked with managing the participants clinically. The rater briefed each study participant about the VAS, and how to report the degree of pain using the VAS ruler (Fig. 3B). Each study subject reported his/her perceived score on the VAS to the rater immediately after IVC. With regard to the SCAI, the rater underwent several training sessions with the Pain Monitor device manufacturer (Med-Storm Innovation AS, Oslo, Norway) to operate the device, record and evaluate the results in several pilot dummy runs before commencing the full trial protocol. Full details on the measurements with SCAI and VAS scores are described in our previously published protocol13. Both VAS and SCAI measurements were obtained immediately after the first intravenous cannulation attempt, regardless of its success. Pain assessment, therefore, reflected the initial puncture stimulus, eliminating variation related to repeated attempts as a potential confounder.
The skin conductance algesimeter index (SCAI) quantifies the number of skin conductance peaks per second (µS/s) that reflect sympathetic nervous system activity triggered by nociceptive stimuli. According to the manufacturer’s calibration reference (Pain Monitor, Med-Storm Innovation AS, Oslo, Norway), SCAI values between 0.00 and 0.07 µS/s correspond to no pain, 0.13–0.21 µS/s to mild pain, 0.27–0.33 µS/s to moderate pain, and 0.40–0.70 µS/s to severe pain. These thresholds were derived from validation studies correlating SCAI readings with established subjective pain scores in postoperative and intensive care populations.
Endpoint and safety assessments
The primary endpoint is the VAS mean score differences based on pairwise comparisons among different intervention arms (A vs. B, A vs. C, A vs. D, B vs. C, B vs. D, C vs. D) immediately after IVC insertion. The secondary endpoint is the mean SCAI differences measured using the Pain Monitor device (Med-Storm Innovation AS, Oslo, Norway) based on similar pairwise comparisons after IVC insertion.
For safety assessment, the trial participants were subject to trial withdrawal at any time during the trial period if the participants experienced a sudden (expected or unexpected) severe complication or adverse event. Any such withdrawal was documented on the adverse event section of the case report form (CRF), and the participant was followed up for outcome assessment and included in the final analysis according to their originally assigned group, following the modified intention-to-treat (ITT) principle.
All adverse events (AEs) and serious adverse events (SAEs) were recorded and graded in accordance with the Common Terminology Criteria for Adverse Events (CTCAE) version 5, as well as the US FDA’s Toxicity Grading Scale for Healthy Adult and Adolescent Volunteers Participating in Preventive Vaccine Clinical Trials. The participants were withdrawn if they experienced CTCAE grade 3–4 adverse events such as severe or life-threatening erythema, itchiness or tenderness). The participants were followed up for up to 48 h after the intervention was administered via phone calls.
Sample size calculation
The sample size was calculated using G*Power version 3.1.9.6 (Universitat Kiel, Germany, February 2020). We utilized the F-test within-group comparison sub-option and fixed the number of groups and measurements to 1 and 4, respectively, since all subjects would receive all intervention arms in a crossover fashion. Based on prior guidelines, moderate-to-high correlations between interventions (r = 0.7) were assumed19. In our original protocol, we assumed the non-sphericity correction (ε) of 0.7511. However, based on the pilot run data and prior guidelines, an ε value of 1 is also considered statistically appropriate and justifiable and was eventually opted for20,21. We then fixed the type I error rate and study power at 0.05 and 0.80, respectively. Therefore, a sample size of 19 subjects was required for this study. Assuming a 10% drop-out rate, the total sample size would be 21 (ntotal = 21).
Statistical analysis
The data were descriptively summarized in mean and standard deviation (SD), or median and interquartile range (IQR) for numerical variables, or frequency and percentage for categorical variables. No multiple imputation was carried out if the missing value rate was under 5%. The Shapiro-Wilks test was utilized to assess data normality. Little’s test was performed to examine whether the missingness patterns follow the missing completely at random (MCAR) assumption. No interim analysis was conducted and no stopping guidelines for early trial termination due to futility or efficacy had been pre-specified. Besides, no subgroup or sensitivity analysis was performed.
A generalized linear mixed effect (GLMM) model with an identity link function was used to estimate the differences in mean VAS score (primary outcome) and SCAI (secondary outcome) between the intervention groups after controlling for the participant’s age, sex, treatment sequence, period effect and baseline VAS or SCAI scores, respectively (all possible confounders). Pearson’s correlation coefficient was then employed to assess the correlations between the SCAI and VAS scores for each intervention group. Bonferroni correction was employed to address multiple testing issues.
For each analysis, the significance level is fixed at 0.05 (two-tailed) and a 95% CI is reported for each parameter estimate. All analyses were conducted using SPSS (IBM Statistics for Windows, Version 26.0, Armonk, NY: IBM Corp).
Results
Participant recruitment
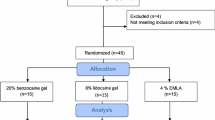
From the patient registry, we identified 96 thalassemia children receiving transfusions at the Thalassemia Daycare Unit, HCTM, who were potentially eligible for this study. Forty-nine thalassemia children were excluded for not fulfilling the age range (9 thalassemia patients aged less than 6 years old and 40 patients older than 18 years old). Sixteen children were further excluded for not being transfusion-dependent thalassemia. Another 11 thalassemia children refused trial participation. Eventually, only 20 participants consented to trial participation and were randomized to the different intervention sequences.
Overall, 16 out of 20 recruited trial participants (80%) completed the entire intervention sequences and had complete recorded observations for all trial endpoints and interventions. One participant was transferred to another hospital after randomization but before receiving the first intervention. The participant was thus considered to have withdrawn from the trial and excluded from analysis under the mITT principle. One patient did not complete the whole set of interventions because this participant was transferred to another hospital to continue care after the first set of interventions. Two children did not complete the entire intervention sequence. One child did not have SCAI readings during two sessions due to a temporary malfunction of the Pain Monitor device. Another child demonstrated significant procedural distress during two assessment sessions, characterized by crying and agitation, which precluded reliable completion of the VAS assessment. Despite repeated clarification by the rater, the child changed responses several times and appeared unable to focus on the task. The reported VAS scores were therefore excluded to maintain data veracity. However, all the other data from these three affected participants were still included in the analyses for the mITT purposes (Fig. 4). All MNAP and PVA-containing PET sham patches were administered by medically trained procedurists following the procedures described in the “Methods” section.
The modified CONSORT diagram for crossover design depicting trial participant recruitment flow.
Baseline characteristics of study participants
The mean age of the children is 11.95 years, with more than three-quarters being male and of Malay ethnicity. Most of the participants were beta-thalassemia major. All participants received blood transfusions on a four-weekly basis, and the chelation therapy prescribed was predominantly deferasirox (Table 1). Besides, all participants did not receive any concomitant analgesic therapy that could have influenced the trial endpoints (VAS and SCAI).
Primary outcome: pain assessment by visual analog scale (VAS) for all intervention groups versus control
For VAS, each MNAP intervention group showed lower marginal mean scores compared with the sham patch, indicating a consistent reduction in perceived pain intensity. Among these, the combination of MNAP and 1 FTU EMLA applied for 30 min recorded the lowest mean VAS and was the only intervention demonstrating a statistically significant reduction compared with the sham patch (mean difference: -1.224 (95% CI -2.441, -0.007), p = 0.048). The magnitude of this reduction corresponds to the minimal clinically important difference of more than 1 point for procedural pain reported in the literature, suggesting that the analgesic effect was not only statistically but also clinically relevant19. All model assumptions (residuals’ linearity, normality, independence and homoscedasticity) are met (Table 2).
Secondary outcome: pain assessment by skin conductance algesimeter index (SCAI) for all intervention groups versus control
For SCAI, the MNAP groups also show lower readings compared to the control sham patch. The combined MNAP and 1 FTU EMLA application for the 30-minute arm had the lowest mean SCAI among all interventions. However, all mean SCAI differences between the MNAP groups were very small, resulting in statistically non-significant comparisons. All model assumptions (residuals’ linearity, normality, independence and homoscedasticity) are met (Table 3).
Secondary outcome: correlation between VAS and SCAI
There are statistically non-significant and poor negative-to-positive correlations between VAS and SCAI (range: -0.040 to 0.378, p > 0.05). Hence, SCAI does not correlate well with VAS (Table 4).
Safety profile
Only 1 participant (5.3%) reported grade 1 (mild) itchiness immediately after MNAP application, before EMLA was applied. The itchiness completely and spontaneously resolved without treatment within one-hour post-MNAP application without any treatment and did not recur in subsequent interventions involving either MNAP or EMLA, suggestive of minor mechanical irritation from the cutaneous penetration rather than a true hypersensitivity reaction. No serious adverse event (AE) or suspected unexpected serious adverse reaction (SUSAR) was reported by the trial participants. The acceptability of the MNAP appeared satisfactory since no participant dropped out of the trial due to intervention-related AE.
Discussion
This study demonstrates that the MNAP mediates a deeper skin anesthetic effect by EMLA, as shown by the significantly lower VAS scores in patients who received the MNAP compared to the sham patch, when both were administered 1 FTU EMLA for 30 min. The observed reduction of about 1.2 points in VAS between the MNAP + 1 FTU EMLA group and the sham patch aligns with the recognized minimal clinically important difference for procedural pain22. This supports that the improvement, while modest in absolute terms, was likely perceptible and meaningful to the participants. This result is consistent with the underlying mechanism of MNAP creating minute conduits in the skin SC layer that could facilitate the more extensive dispersion of the drug applied topically for enhanced nociceptor blockage.
No significant differences in VAS pain scores were observed when the MNAP was combined with the EMLA dose reduced by half (0.5 FTU) or the EMLA applied only for 15 min, as compared to the sham patch control with similar EMLA conditions. These findings suggest that the MNAP application is still dependent on a certain threshold of adequacy in EMLA® dosage and application time for effective skin anesthesia. Although the manufacturer recommends a 60-minute application of EMLA to achieve skin anesthesia to a depth of 3mm14, such duration may not be feasible in high-volume pediatric transfusion services. Hence, prolonged pre-procedural waiting time may compromise the timely delivery of care. Therefore, EMLA was applied for approximately 30 min, which was considered a practical and effective compromise in this clinic outpatient setting. Furthermore, previous studies have also shown that meaningful dermal analgesia can be achieved with a 30-minute application time15,16,17. Our trial thus designed in the interest of patient comfort while maintaining service delivery to ensure a pragmatic adaptation with the existing management practice for transfusion-dependent thalassemic children18.
A recent animal study by Liu and colleagues utilized a similar biodegradable polylactic acid (PAL)-based MNAP design to enhance lidocaine delivery23. They showed that PAL-MNAP hastened the lidocaine’s onset of action from 40 min to 10 min in their murine model based on the Von Frey hair filaments test results. However, this application is still in the early pre-clinical development phase and many practical challenges, for instance, the extent of required skin penetration depth in pediatric patients for successful anesthetic delivery, satisfactory biocompatibility profile and patient acceptance and compliance in children are still unclear24.
Another study, an open-label non-randomized trial investigated novel pain-reducing modalities in transfusion-dependent thalassemic children. Chan et al. demonstrated that the thumbtack infusion system (Thalaset needle) was significantly superior to the standard butterfly vein scalp system in reducing the pain associated with desferrioxamine infusion in these patients. However, this study was not assessing pain associated with venipuncture and IVC25. However, the pain assessment was based on a non-validated scoring system and only non-confounder-adjusted (univariable) analyses were presented. Instead, our trial addressed inter-patient variability in pain threshold by using the same patient in cross-over interventions, which demonstrated for the first time that a novel biocompatible maltose-based MNAP could potentially enhance pain-reducing effects of EMLA cream in this unique pediatric population that requires frequent IVC and skin puncture procedures. Hence, our biocompatible and dissolvable maltose-based MNAP design has clear potential for delivering topical analgesics and may address some of the above-mentioned challenges.
We speculate that similar findings in improved pain relief with MNAP and 1 FTU EMLA for 30 min when pain severity was measured using the SCAI was that the SCAI method, although previously shown to detect nociceptive responses during heel lancing and vaccinations in neonates and infants26,27, was very sensitive, and the readings in microSiemens/s over a short duration may not be able to distinguish differences in the measurement of pain sustained during brief procedures such as IVC. Hellerud and Storm26 and Harrison et al.27 indeed showed that skin conductance reliably rises with pain in neonates, for example, during heel-prick capillary blood sampling or vaccinations. However, these procedures involve a relatively long-lasting, whole-foot stimulus, whereas IV cannulation in our thalassemia children aged 6 to 17 years is a brief, localized venipuncture stimulus. The sympathetic surge from a quick IV puncture is therefore very transient and, hence, SCAI may not capture distinct conductance differences under these conditions, supporting our finding of limited SCAI sensitivity in this context.
The SCAI method has been used more extensively for the detection and monitoring of pain responses during anesthesia for surgical operative procedures or in the intensive care setting12. The SCAI findings in this study were within the expected physiological range reported by the device manufacturer. The mean SCAI values across all intervention arms (0.179–0.203 µS/s) fell within the mild pain range defined by the Pain Monitor reference scale (0.13–0.21 µS/s). Despite this, the correlations between SCAI and VAS were weak and inconsistent, indicating that the two measures capture different aspects of the pain experience. While the VAS reflects subjective perception and higher cortical processing of pain, the SCAI records peripheral sympathetic activation, which may fluctuate rapidly and be influenced by multiple physiological or environmental factors. Besides, the VAS-SCAI poor correlations could also be attributed to the fact that our thalassemic children underwent multiple blood transfusions that potentially had caused significant cutaneous iron deposits. As a result, the excessive iron deposits in the skin may interfere with the cutaneous electrical skin conductance. Previous reports demonstrated that regular blood transfusions in beta-thalassemia major children caused xerosis and scarring28,29. Consequently, we speculate that these conditions may increase the cutaneous electrical impedance, which could result in the observed VAS-SCAI discordance. Further trials on different pediatric populations and longer pain-sustaining procedures are required to further evaluate the SCAI method as a valid or reliable pain assessment tool in children.
Our trial involving children with thalassemia has several limitations. First, the overall sample size was small, which may limit the statistical power. Besides, the missing observations for inevitable reasons e.g. hospital transfers for further care and a dwindling number with this condition, plus the refusal to assent and lack of cooperativeness during some of the intervention days affected our study sample size. However, the GLMM approach used for data analysis still produced unbiased estimates with satisfactory preservation of study power, especially since our data follows an MCAR pattern30. Finally, patient acceptance of our MNAP could not be formally evaluated due to the absence of a validated survey instrument.
Nonetheless, our study establishes the novelty of a biodegradable and dissolvable MNAP that appears to be safe for application in the pediatric population and potentially enhances the delivery of topical skin analgesics or other medications. This pilot study will pave the way for further optimization of the MNAP for other applications such as hormone or even vaccine delivery. Currently, we are fabricating a lignocaine-incorporated MNAP so that this anesthetic agent can be delivered into the skin to block the nociceptive receptors. Hopefully, MNAP could improve the transdermal delivery of this drug with minimal pain compared to that induced by hypodermic needle used to infiltrate the skin.
Conclusions
The dissolvable maltose-based MNAP appears safe and was able to enhance the pain relief from skin analgesic when applied with the usually recommended regimen of the EMLA cream before IVC for blood transfusion in children with thalassemia. Future trials are needed to confirm these findings in a healthy pediatric population or children without a chronic debilitating disease. The SCAI as a tool for pain assessment during short procedures such as IVC, does not appear to be interchangeable with the VAS method for children between 6 and 17 years old and may not be particularly reliable for conditions that could alter the skin autonomic or conductance system.
Data availability
The datasets used and/or analysed that support the findings of this study are available from the corresponding author upon reasonable request.
References
George, E. Beta-thalassemia major in Malaysia, an ongoing public health problem. Med. J. Malaysia. 56, 397–400 (2001).
Mohd Ibrahim, H. et al. Observational study on the current status of thalassaemia in malaysia: a report from the Malaysian thalassaemia registry. BMJ Open. 10, e037974 (2020).
Orenius, T., LicPsych, Säilä, H., Mikola, K. & Ristolainen, L. Fear of injections and needle phobia among children and adolescents: an overview of psychological, behavioral, and contextual factors. SAGE Open. Nurs. 4, 2377960818759442. https://doi.org/10.1177/2377960818759442 (2018).
Franck, L. S., Greenberg, C. S. & Stevens, B. Pain assessment in infants and children. Pediatr. Clin. North. Am. 47, 487–512. https://doi.org/10.1016/s0031-3955(05)70222-4 (2000).
Pancekauskaitė, G. & Jankauskaitė, L. Paediatric pain medicine: pain Differences, recognition and coping acute procedural pain in paediatric emergency room. Med. (Kaunas). 54, 94. https://doi.org/10.3390/medicina54060094 (2018).
Duarah, S., Sharma, M. & Wen, J. Recent advances in microneedle-based drug delivery: special emphasis on its use in paediatric population. Eur. J. Pharm. Biopharm. 136, 48–69. https://doi.org/10.1016/j.ejpb.2019.01.005 (2019).
Kolli, C. S. & Banga, A. K. Characterization of solid maltose microneedles and their use for transdermal delivery. Pharm. Res. https://doi.org/10.1007/s11095-007-9350-0 (2008). 25, 104 – 13.
Waghule, T. et al. Microneedles: a smart approach and increasing potential for transdermal drug delivery system. Biomed. Pharmacother. 109, 1249–1258. https://doi.org/10.1016/j.biopha.2018.10.078 (2019).
Bardol, M., Pan, S., Walker, S. M., Standing, J. F. & Dawes, J. M. Pharmacokinetic pharmacodynamic modeling of analgesics and sedatives in children. Paediatr. Anaesth. 33, 781–792 (2023).
Shields, B. J., Palermo, T. M., Powers, J. D., Grewe, S. D. & Smith, G. A. Predictors of a child’s ability to use a visual analogue scale. Child. Care Health Dev. 29, 281–290. https://doi.org/10.1046/j.1365-2214.2003.00343.x (2003).
Hicks, C. L., von Baeyer, C. L., Spafford, P. A., van Korlaar, I. & Goodenough, B. The faces pain Scale-Revised: toward a common metric in pediatric pain measurement. Pain 93, 173–183. https://doi.org/10.1016/s0304-3959(01)00314-1 (2001).
Storm, H. Changes in skin conductance as a tool to monitor nociceptive stimulation and pain. Curr. Opin. Anaesthesiol. 21, 796–804. https://doi.org/10.1097/aco.0b013e3283183fe4 (2008).
Abdul Jalal, M. I. et al. Transdermal Maltose-Based microneedle patch as adjunct to enhance topical anesthetic before intravenous cannulation of pediatric thalassemic patients receiving blood transfusion: a randomized controlled trial protocol. J. Clin. Med. 11, 5291. https://doi.org/10.3390/jcm11185291 (2022).
Aspen Pharmacare Ireland Ltd. EMLA Cream 5% (5 g pack) – summary of product characteristics. (2024, accessed 11 Nov 2025). https://www.medicines.org.uk/emc/product/1762/smpc.
Bjerring, P. & Arendt-Nielsen, L. Depth and duration of skin analgesia to needle insertion after topical application of EMLA cream. Br. J. Anaesth. 64, 173–177. https://doi.org/10.1093/bja/64.2.173 (1990).
Hopkins, C. S., Buckley, C. J. & Bush, G. H. Pain-free injection in infants. Use of a lignocaine-prilocaine cream to prevent pain at intravenous induction of general anaesthesia in 1-5-year-old children. Anaesthesia. 43, 198–201 (1988).
Yu, Z. et al. Pharmacological and non-pharmacological interventions in management of peripheral venipuncture-related pain: a randomized clinical trial. BMC Pediatr. 23, 58. https://doi.org/10.1186/s12887-023-03855-z (2023).
Clinical Practice Guidelines. Management of transfusion-dependent Thalassaemia. Kuala Lumpur: Ministry of Health, Malaysia (2009, accessed 31 Dec 2023). https://mpaeds.my/wp-content/uploads/2018/03/CPG-Management-of-Transfusion-Dependent-Thalassaemia.pdf.
Mukaka, M. M. Statistics corner: a guide to appropriate use of correlation coefficient in medical research. Malawi Med. J. 24, 69–71 (2012).
Prajapati, B., Dunne, M. & Armstrong, R. Sample size Estimation and statistical power analyses. Optometry Today. 16, 10–18 (2010).
Beck, T. W. The importance of a priori sample size Estimation in strength and conditioning research. J. Strength. Conditioning Res. 27, 2323–2337. https://doi.org/10.1519/jsc.0b013e318278eea0 (2013).
Olsen, M. F. et al. Pain relief that matters to patients: systematic review of empirical studies assessing the minimum clinically important difference in acute pain. BMC Med. 15, 35. https://doi.org/10.1186/s12916-016-0775-3 (2017).
Liu, Y. et al. Toward a solid microneedle patch for rapid and enhanced local analgesic action. Drug Deliv Transl Res. 14, 1810–1819. https://doi.org/10.1007/s13346-023-01486-6 (2024).
Chakraborty, C., Bhattacharya, M. & Lee, S. S. Current status of microneedle array technology for therapeutic delivery: from bench to clinic. Mol. Biotechnol. 66, 3415–3437. https://doi.org/10.1007/s12033-023-00961-2 (2024).
Chan, G. C., Ng, D. M., Fong, D. Y., Ha, S. Y. & Lau, Y. L. Comparison of subcutaneous infusion needles for transfusion-dependent thalassemia patients by the intrapersonal cross-over assessment model. Am. J. Hematol. 76, 398–404. https://doi.org/10.1002/ajh.20103 (2004).
Hellerud, B. C. & Storm, H. Skin conductance and behaviour during sensory stimulation of preterm and term infants. Early Hum. Dev. 70, 35–46. https://doi.org/10.1016/s0378-3782(02)00070-1 (2002).
Harrison, D., Boyce, S., Loughnan, P., Dargaville, P. & Storm, H. Johnston L. Skin conductance as a measure of pain and stress in hospitalised infants. Early Hum. Dev. 82, 603–608. https://doi.org/10.1016/j.earlhumdev.2005.12.008 (2006).
Zulfiqar, Z. et al. Cutaneous manifestations in patients with Beta-Thalassemia major. J. Mother. Child. 27, 142–146. https://doi.org/10.34763/jmotherandchild.20232701.d-23-00047 (2023).
Fahmey, S. S., Taha, G., El-Refaey, A. & Adly, S. Skin disorders in Egyptian children with β-Thalassemia major. J. Trop. Pediatr. 64, 104–109. https://doi.org/10.1093/tropej/fmx035 (2018).
Miller, M. L., Roe, D. J., Hu, C. & Bell, M. L. Power difference in a χ2 test vs generalized linear mixed model in the presence of missing data - a simulation study. BMC Med. Res. Methodol. 20, 50. https://doi.org/10.1186/s12874-020-00936-w (2020).
Acknowledgements
We thank Dr Goh Chee Seong from Alnair Labs Corporation, Tokyo, Japan for his technical expertise in designing and fabricating the microneedle array patch. We also thank Drs Natalie Ling Sze Jane and Phan Yong Hong for their technical assistance.
Funding
This research was funded by the Malaysian Ministry of Science, Technology and Innovation Prototype Research Grant Scheme (MOSTI-PRGS), grant number PRGS/2/2020/TK05/UKM/01/1.
Author information
Authors and Affiliations
Contributions
MIAJ & CFC: Study conceptualization, methodology development, resources (pilot testing), data analysis and interpretation, manuscript review; KSO & KCF: methodology development, resources (pilot testing), manuscript review and editing; AAH: resources (microneedle fabrication), data collection, project funding; CFD: resources (microneedle fabrication), data collection and project funding; MRB, PCO, ASAR: resources (microneedle fabrication) and data collection; TYL: resources (microneedle fabrication) and data collection; XYC: resources (pilot testing) and data collection; DSCL: resources (pilot testing), data collection and provision of clinical care; FCC: resources (pilot testing) and data collection. All authors have read and approved the published version of this manuscript. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jalal, M.I.A., Ooi, K.S., Foo, K.C. et al. Randomized double-blind crossover trial of a transdermal maltose dissolvable microneedle patch for topical anesthesia before intravenous cannulation for thalassemia children. Sci Rep 16, 671 (2026). https://doi.org/10.1038/s41598-025-30279-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-30279-9