Abstract
The hepatitis C virus (HCV) is recognized as a significant global public health concern due to its complex transmission dynamics and long-term health consequences. In this study, a stochastic delay differential model was examined to enhance the understanding of HCV transmission. Time delays were incorporated into the mathematical model to represent incubation periods, while stochastic perturbations were introduced to reflect random environmental and demographic variations. The model was formulated to account for key compartments, including susceptible individuals, acute and chronic infections, and recovered individuals, along with disease-induced mortality and progression rates. By representing delays associated with incubation periods and asymptomatic stages, the model was used to explore HCV transmission mechanisms under stochastic influences. Additionally, strategies for disease control through vaccination and treatment were investigated. Stochastic fluctuations were included to capture uncertainties arising from environmental and demographic factors. Using stochastic Lyapunov functional techniques, the existence of a unique global solution was established, and conditions for disease extinction, persistence, and the existence of stationary distributions were derived. Optimal control strategies were developed with the goal of minimizing infection prevalence and associated intervention costs, focusing on measures such as treatment, public health education, and vaccination. The optimal control trajectories under the influence of delays and stochastic effects were determined by applying Pontryagin’s Maximum Principle (PMP), thereby ensuring practical relevance. Numerical simulations were conducted to demonstrate the effects of time delays and stochastic variables on HCV dynamics and to highlight the effectiveness of the proposed control strategies. Overall, this work provides important insights into the interplay between stochastic processes, time delays, and optimal interventions, offering a comprehensive framework for the effective management and potential eradication of HCV epidemics.
Similar content being viewed by others
Introduction
The HCV poses a major global health challenge, impacting millions of individuals and contributing to severe liver conditions such as cirrhosis and hepatocellular carcinoma. Approximately 50 million individuals worldwide live with chronic HCV infection, with an estimated every year, 1 million new cases are recorded. According to the World Health Organization (WHO), about 242, 000 people died from hepatitis C in 2022, primarily due to complications like cirrhosis and primary liver cancer1. The insidious nature of HCV, marked by an extended incubation period and a frequently asymptomatic early stage, makes early detection and management particularly difficult. Compounding these challenges are uncertainties in transmission dynamics driven by demographic and environmental factors, which hinder efforts to control the virus effectively. Developing comprehensive mathematical models that address these complexities is essential for understanding HCV dynamics and designing targeted intervention strategies. Delay differential equations (DDEs) have gained prominence in recent years as a valuable tool for modeling diseases with incubation periods or delayed response mechanisms, see2 and the references therein. These equations allow for a realistic depiction of biological and epidemiological processes, such as the time lag between infection and symptom onset or delays in immune system responses3. Additionally, stochastic differential equations (SDEs) capture the randomness inherent in disease transmission, influenced by factors such as environmental variability and demographic fluctuations4. By integrating these approaches, stochastic delay differential equations (SDDEs) provide a powerful framework for studying the dynamics of HCV and similar diseases in real-world conditions.
This study focuses on developing and analyzing a stochastic delay differential epidemic model to better understand the HCV control techniques and propagation dynamics. The model incorporates time delays representing the incubation period and asymptomatic stages, along with stochastic perturbations to account for environmental uncertainties. The primary objectives include examining the existence and uniqueness of solutions, establishing threshold conditions for disease extinction and persistence, and analyzing the effects of delays and stochasticity on disease progression. To address practical challenges in HCV control, the study also introduces optimal control strategies emphasizing vaccination and treatment interventions. Optimal control theory offers a structured approach to identifying strategies that minimize disease prevalence and intervention costs while maximizing public health outcomes. By examining the interactions between stochastic influences, time delays, and control measures, the study aims to identify effective strategies for mitigating HCV epidemics.
The dynamics of HCV transmission, characterized by varying incubation periods and treatment responses, require advanced mathematical tools for a comprehensive understanding and effective management of the epidemic. This paper introduces a novel approach to modeling HCV dynamics using stochastic delay differential equations (SDDEs), which account for both random variations in disease transmission and critical delays inherent in infection processes. Unlike traditional deterministic models, the stochastic framework incorporates randomness in disease spread while addressing significant biological and clinical delays, such as the incubation period and response to treatment. By integrating optimal control theory with SDDEs, the model provides a robust tool for designing and assessing intervention strategies. This combined approach allows for realistic predictions and the development of more effective control measures. Given the availability of highly effective direct-acting antiviral therapies and the WHO’s goal to eradicate HCV by 2030, this framework is particularly relevant for guiding public health initiatives.
To further enhance the understanding and management of HCV dynamics, researchers have explored models that incorporate additional complexities and uncertainties. Zhang et al.5 introduced a nonlinear epidemiological model for HCV that incorporates parametric uncertainties, extending beyond earlier optimal strategies. The primary objective of their proposed control scheme was to reduce the populations of those who are chronically afflicted and those who are ignorant of their susceptibility, even in the presence of parameter uncertainties. Martcheva and Chavez6 presented a simpler mathematical model consisting of three compartments: infected intensely, persistently, and vulnerable. Their model was based on various epidemiological observations. Building on foundational HCV modeling efforts, Yuan and Yang7 extended the traditional framework by introducing an exposed class, proposing that susceptible individuals transition to the exposed compartment upon contact with infected individuals. This addition provided a more nuanced representation of early-stage infections. Zhang and Zhou8 further enhanced the biological realism of the model by incorporating disease-induced mortality, thus aligning the mathematical structure more closely with real-world HCV outcomes. Continuing this trajectory, Hu and Sun9 developed a four-compartment model that, for the first time, included the recovered population, acknowledging the importance of immunity in shaping epidemic dynamics. Complementing these continuous models, Okosun and Makinde10 proposed a nonlinear HCV epidemic model that divided the population into Susceptible (S), Exposed (E), Acutely Infected (I), Treated (T), and Chronically Infected (V) compartments. Their approach integrated screening and drug efficacy as key control mechanisms and utilized Pontryagin’s Maximum Principle (PMP) to derive optimal intervention strategies. In a discrete-time setting, Abdul et al.11 analyzed the dynamics of a difference equation model for HCV, uncovering transitions between stable and unstable states that highlighted the system’s sensitivity to parameter variations and the importance of targeted interventions. In parallel with these deterministic modeling advancements, recent years have seen increasing attention to the role of stochasticity and uncertainty in disease dynamics. Anwarud Din12 introduced a stochastic delay differential equation (SDDE) model to analyze the bifurcation behavior of the Hepatitis B virus, revealing that environmental noise can significantly suppress disease spread. Building upon this, Anwarud Din and Yongi Jin13 examined the impact of stochastic controls in an HIV/AIDS model and demonstrated the effectiveness of intervention strategies under uncertainty. Broadening the scope beyond viral diseases, Wajahat et al.14 and Ullah et al.15 proposed fractional-order models to study antacid-induced allergies, emphasizing how fractional dynamics provide critical insights for managing immunological responses. Further, Qura et al.16 investigated stationary distributions in stochastic epidemic systems with nonlinear noise, offering insights into long-term equilibrium behavior. Shah et al.17 applied similar stochastic frameworks in the context of worm transmission within wireless sensor networks, showcasing the cross-disciplinary relevance of such models. These studies collectively emphasize the necessity of integrating time delays, environmental noise, and fractional-order dynamics into epidemic models to enhance predictive accuracy and policy relevance. In particular, Khan et al.18 argued that modeling incubation periods and delays as variables, rather than categorizing disease stages rigidly, provides greater flexibility and realism. Additionally, external environmental factors such as temperature, rainfall, and humidity play a critical role in modulating viral transmission. Unlike deterministic models that offer only expected values, stochastic differential equations (SDEs) enable the exploration of both average behavior and the variability around it, capturing the probabilistic nature of real-world epidemics. As Ji and Jiang19 noted, this probabilistic perspective yields a richer and more accurate understanding of infectious disease dynamics, especially under fluctuating environmental conditions. Khan et al.18 suggested that it may be more suitable to model the incubation period and time delay as variables for forecasting how infections would behave dynamically, rather than dividing the disease into two distinct stages. Furthermore, the pace at which illnesses, particularly viral infections in humans, spread is greatly influenced by external environmental elements including temperature, rainfall, and air humidity. Unlike deterministic models, SDEs provide more insightful outcomes by capturing both the expected results and the distribution of long-term dynamic behavior’s likelihood. This approach offers deeper insights into disease transmission compared to deterministic models, which only produce predicted values (Ji and Jiang,19). Zhao and Jiang,20 proposed simple stochastic epidemic models with Brownian motion perturbations that have proven effective in deriving threshold conditions for controlling disease transmission, helping determine whether a disease will eventually die out. Effective disease control requires more than just ensuring system stability; mathematical modeling of epidemic spread plays a critical role in preventing and managing outbreaks (Din et al., Li, Zaman21,22,23; Yusuf et al., 202124). When deterministic systems are converted into stochastic systems, stochastic analysis provides better tools for evaluating system stability and transient dynamics (Liu et al.,25). Qi and Meng26 examines whether HCV will be eradicated under the combined effects of random fluctuations and time delays, and how time delays influence disease extinction. They focus on understanding the effect of time delay rather than simply analyzing virus decay. Specifically, our study extends previous works by incorporating both time delays and stochastic perturbations into the HCV transmission dynamics, which are often overlooked or treated separately in earlier models. While many deterministic models have provided foundational insights into disease spread, they typically do not capture the inherent randomness in disease transmission due to environmental or demographic fluctuations. Our SDDEs model not only addresses this gap but also incorporates optimal control strategies under stochastic influences, thereby offering a more realistic and applicable framework for policy and intervention design. Recent studies, such as those by Liu and Guo27 and Wafer28, have investigated aspects of stochasticity but have not fully integrated both stochasticity and time delays with optimal control. In contrast, our work develops a robust framework that merges these critical aspects and analyzes their joint impact on the dynamics and control of HCV. This comprehensive approach allows for a deeper understanding of how delays in disease progression and random perturbations affect the outcomes of public health interventions.
In response to disease outbreaks, optimal control methods have garnered significant interest, whether applied to deterministic or stochastic models. These methods aim to reduce the number of infected individuals through external interventions, primarily immunization of vulnerable populations and treatment of infected individuals. By using the Hamilton-Jacobi-Bellman approach, optimal control can be framed as an optimization problem with constraints, minimizing the cost of reducing infections across society.
In this paper we suggest a stochastic delayed HCV model that incorporates a nonlinear transmission rate represented by \(\beta S(t) (I(t - \tau ) + C(t - \tau ))\). The model also integrates white noise to account for parameter perturbations and the effects of external environmental variability. Our primary focus is to analyze the stability of the system using stochastic Lyapunov functional theory, emphasizing the conditions for disease extinction and the characterization of its stationary distribution. Additionally, we examine the role of time delay in regulating control strategies. To mitigate disease transmission in a stochastic environment, The main control methods that we take into consideration are the effectiveness of vaccinations and treatments.
This paper structured as in Section 2 present the delayed stochastic epidemic model for HCV, detailing its formulation and underlying assumptions. In Section 3, we provide a theoretical analysis, including proofs of the existence and uniqueness of solutions, along with the derivation of threshold conditions. Sections 5 and 5.1 focus on the development of optimal control strategies in both deterministic and stochastic contexts. There are numerical simulations displayed in Section 6 to validate the theoretical findings and demonstrate the effects of stochastic elements and temporal delays on the dynamics of illness. Finally, Section 7 highlights the key insights and discusses their relevance to HCV control and management strategies.
The HCV model that incorporates a delay
According to Jing et al.29 suggest the HCV epidemic model,
In this study, the model given in equation (1) describes the transmission dynamics of the HCV using a deterministic compartmental framework. The total population N(t) is divided into four epidemiological classes: susceptible individuals S(t), those acutely infected with HCV I(t), those chronically infected C(t), and recovered individuals R(t). The susceptible population increases through recruitment (birth or immigration) at a constant rate \(\Lambda\) and decreases due to infection and natural death. Infection occurs through effective contact with both acutely and chronically infected individuals, captured by the bilinear incidence term \(\beta S(t)(I(t) + C(t))\), where \(\beta\) denotes the transmission rate. Once infected, individuals enter the acute class I(t), where they may either recover naturally or progress to chronic infection. The progression occurs at rate \(\gamma\), with a fraction p advancing to the chronic class C(t), while the remaining fraction \((1-p)\) recover and move to the recovered class R(t). Natural death occurs in all compartments at a constant rate \(\alpha\), while chronically infected individuals also experience disease-induced mortality at an additional rate \(\mu\). The recovered individuals are assumed to have immunity and do not return to the susceptible class. This compartmental structure effectively captures the biological progression of HCV from infection to chronic disease or recovery and incorporates both natural and disease-induced mortality. The biological interpretations of the parameters in model (1) are summarized in Table 1.
Theorem 1
The deterministic system (1) with non-negative initial conditions \(S(0) \ge 0\), \(I(0) \ge 0\), \(C(0) \ge 0\), \(R(0) \ge 0\). Then, the system has a unique, positive, and bounded solution for all \(t \ge 0\).
Proof
Let \(\textbf{X}(t) = (S(t), I(t), C(t), R(t))^\top\) and define the vector field \(\textbf{F}(\textbf{X})\) from the right-hand side of (1). Each component of \(\textbf{F}\) is continuously differentiable on \(\mathbb {R}_+^4\), and thus \(\textbf{F}\) is locally Lipschitz continuous in \(\textbf{X}\). By the Picard-Lindelöf theorem, a unique local solution exists. We show that the solution remains non-negative for all \(t \ge 0\).
-
When \(S(t) = 0\): \(\frac{dS}{dt} = \Lambda> 0 \Rightarrow S(t) > 0\) for \(t > 0\).
-
When \(I(t) = 0\): \(\frac{dI}{dt} = \beta S(t)C(t) \ge 0\) implies \(I(t) \ge 0\).
-
When \(C(t) = 0\): \(\frac{dC}{dt} = p\gamma I(t) \ge 0\) implies \(C(t) \ge 0\).
-
When \(R(t) = 0\): \(\frac{dR}{dt} = (1-p)\gamma I(t) \ge 0\) implies \(R(t) \ge 0\).
Thus, if the initial conditions are non-negative, the solution remains non-negative for all \(t \ge 0\). Now the total population: \(N(t) = S(t) + I(t) + C(t) + R(t)\). Then,
Adding the right-hand sides of (1):
Solving the differential inequality:
we get the upper bound:
Hence, all compartments S(t), I(t), C(t), R(t) are bounded above by \(\frac{\Lambda }{\alpha }\), and the solution exists globally. \(\square\)
The deterministic system given by Equation (1) has been previously studied by Jing et al.29, the disease-free equilibrium (DFE) and endemic equilibrium were explicitly derived, and the local stability of the DFE was analyzed using the basic reproduction number \(R_0\). They demonstrated that the disease-free equilibrium \(P_0\) is locally and globally asymptotically stable when \(R_0 < 1\) and unstable when \(R_0 > 1\), and the endemic equilibrium point \(P^*\) is locally and globally asymptotically stable when \(R_0 > 1\). These results form the classical basis for understanding the threshold dynamics of the model, upon which the current stochastic analysis is built.
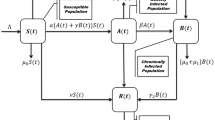
Environmental fluctuations play a crucial role in shaping the transmission dynamics of HCV, introducing random disturbances that influence the spread of the disease. To capture these effects, we incorporate time delays and environmental variability into the model (2). Specifically, the environmental noise is represented as multiplicative white noise, with the intensity parameters \(\eta _i\) corresponding to standard Brownian motion processes \(W_i(t)\), where \(i = 1,2,3,4\). These stochastic terms are state-dependent, meaning their magnitude is proportional to the size of the corresponding population compartments (e.g., \(\eta _1 S(t) dW_1(t)\)), reflecting realistic environmental impacts on disease transmission and progression. By formulating a system of coupled SDEs, as illustrated in Fig. 1, we capture both the inherent time delays and random environmental influences. This form of multiplicative noise ensures biological realism by preserving positivity and scaling fluctuations appropriately with population size. These noise terms capture variations such as:
-
1.
Changes in transmission due to environmental factors (e.g., public awareness, hygiene).
-
2.
Sudden outbreaks or reductions due to unpredictable events.
-
3.
Variability in recovery or mortality rates due to differing healthcare access or natural resistance.
By varying the values of \(\eta _i\), we can simulate different levels of randomness in the population dynamics and investigate how such fluctuations affect disease persistence, extinction, and control strategy effectiveness. This modeling approach provides a more accurate and robust framework for understanding HCV dynamics and for designing effective control strategies in uncertain environments.
Flowchart of the HCV transmission stochastic model (2).
The infection rate terms \(\beta S(t)\big (I(t-\tau ) + C(t-\tau )\big )\) in model (2) incorporate a delay to account for the time it takes for acutely infected and chronically infected individuals to influence the susceptible population.
The next equations (3) provide the model (2) starting values,
here the set C is represented by the Lebesgue operator mapping from \([-\tau , 0]\) to \(\mathbb R_+^4\).
Analyzing stationary data for a positive solution for model (2)
Let us define a complete probability space \((\Omega , \mathscr {F}, \mathbb {P})\), endowed with a filtration \(\{\mathscr {F}_t\}_{t \ge 0}\) that adheres to the standard assumptions (see30):
-
(i)
The filtration \(\{\mathscr {F}_t\}\) is right-continuous;
-
(ii)
It is non-decreasing in t;
-
(iii)
All \(\mathbb {P}\)-null events are included in \(\mathscr {F}_0\).
Additionally, the interior of the nonnegative orthant in \(\mathbb {R}^l\) is defined as:
Now consider the following SDE in l dimensions (see4 for further reference):
with the initial state \(\textbf{Z}(t_0) = \textbf{Z}_0 \in \mathbb {R}^l\). Here, \(\textbf{B}(t)\) represents an n-dimensional Brownian motion defined on the same probability space.
Let \(V(\textbf{Z}, t)\) denote a function belonging to the class \(\mathscr {C}^{2,1}(\mathbb {R}^l \times [t_0, \infty ))\), i.e., V is twice continuously differentiable with respect to the spatial variable \(\textbf{Z}\), and once with respect to the temporal variable t.
The infinitesimal generator \(\mathscr {L}\) associated with the SDE is defined as:
Accordingly, when the operator \(\mathscr {L}\) acts on V, we obtain:
where:
Applying Itô’s Lemma to the function \(V(\textbf{x}(t), t)\), we arrive at the SDE:
In the next theorem, we prove that the solution to the SDE is global. Additionally, we conduct a similar analysis as done in Zhang et al.31 and Din and Li22 to focus on the stationary analysis of the positive solutions of model (2), and their existence and uniqueness.
Theorem 2
Given any initially value that satisfies constraints in (3), there is a unique solution for the stochastic model (2) for any \(t \ge - \tau\). Additionally, there is a one percent chance that this solution will stay inside \(\mathbb R_+^4\). In other words, for every \(t \ge - \tau\), \((S(t), I(t), C(t), R(t)) \in \mathbb R_+^4\) is almost surely (a.s.) true.
Proof
The coefficients satisfying condition (3) are continuous and locally Lipschitz, which guarantees the existence of a distinctive local solution (S(t), I(t), C(t), R(t)) for the system (2) over \(t \in [-\tau , t_e)\), where \(t_e\) represents the explosion time. Let \(a_0\) be a sufficiently large nonnegative number such that the initial values in (3) remain within the interval\(\left[ \frac{1}{a_0}, a_0\right]\). For any nonnegative integer \(a \ge a_0\), the stopping time can then be defined as follows:
We must confirm that \(t_e = \infty\) a.s. in order to prove the solution’s universal property. We define \(\inf \phi = \infty\) for this, where \(\phi\) is a null set. Based on its definition, it is clear that \(t_a\) is rising monotonically as \(a \rightarrow \infty\). Let \(t_\infty \le t_e\) and \(t_\infty = \lim _{a\rightarrow \infty } t_a\). The next step is to verify that \(t_\infty = \infty\) is nearly certainly true. In contrast, two positive constants T and \(\varepsilon \in (0,1)\) would exist such that the following circumstances are satisfied:
Therefore, for integers \(a_1\), we have \(P\{t_a \le T\} > \varepsilon\) for all \(a \ge a_1 \ge a_0\). Then, we define
Define V as a function mapping from \(\mathbb {R}_+^4\) to \(\mathbb {R}_+\), where the constant \(c > 0\) will be specified later. It is clear that V is a non-negative function since all \(x > 0\) satisfy the condition \(x - \ln x - 1 > 0\). By using Itô’s formula, we can express V in an equivalent form as follows:
where
where \(K_1\) is a positive constant. We may obtain by substituting the previous inequality into Eq. (7).
The expectation form of Eq. (8) is then obtained by integrating with the interval \([0, T \wedge t_m]\).
Let us define \(\Omega _a = \{t_a \le t\}\), where \(P(\Omega _a) \ge \varepsilon\) and \(a \ge a_1\) by Eq. (4). It is important to note that for each \(\omega \in \Omega _a\), at least one of the values \(S(t_a, \omega ), I(t_a, \omega ), C(t_a, \omega )\), or \(R(t_a, \omega )\) must equal a or \(\frac{1}{a}\). As a result, we conclude:
Combining Eqs. (9) and (10), we can obtain
Here \(F_\Omega\) represents the indicator function, which takes the value0 or 1. If \(a \rightarrow {\infty }\), it leads to a contradictory inequality, specifically \(\infty > EV(S(0), I(0), C(0), R(0)) + KT = \infty\). Hence, we conclude that \(t_\infty = \infty\) a.s. \(\square\)
Stationary distribution
A key characteristic of stochastic systems is the absence of an endemic equilibrium, which prevents the use of standard stability analysis to examine the disease’s steady spread or the conditions for its persistence. To address this limitation, we draw extensively on the work of Khasminskii32. Specifically, we consider a regular Markov process X(t), where the diffusion matrix is represent as:
Lemma 1
Let \(U \subset \mathbb {R}^d\) represent the closure of a limited domain with a regular boundary, and suppose that X(t) admits the existence of a stationary distribution \(\pi (\cdot )\). Then the following characteristics are true:
-
(i)
For each \(x \in \mathbb {R}^{d_{+}} \setminus U\), \(\exists\) a non-negative \(C^2\) function \(V_3\) such that \(LV_{3} <0\).
-
(ii)
The diffusion matrix D(X) has a minimum eigenvalue that is limited away from zero within U.
To define the stochastic basic reproduction number \(R_0^s\), we consider the expected number of new infections under stochastic perturbations. The white noise terms introduce variance-based correction terms that influence both the susceptible and infected populations. When the SDEs are linearized around the DFE and Itô’s formula is applied, the effective death rates of the compartments are increased by half the variance of the noise terms (due to Itô correction terms). Specifically:
-
For \(S(t)\), the effective death rate becomes \(\alpha + \frac{\eta _1^2}{2}\),
-
For \(I(t)\), the effective removal rate becomes \(\alpha + \gamma + \frac{\eta _2^2}{2}\),
-
For \(C(t)\), the effective removal rate becomes \(\alpha + \mu + \frac{\eta _3^2}{2}\).
This expression reflects how stochastic fluctuations effectively dampen transmission potential, thus lowering \(R_0^s\) compared to the deterministic \(R_0\). The noise intensities \(\eta _i\) capture environmental and demographic uncertainties, and their incorporation improves the realism and robustness of the model.4,33,34
Theorem 3
The solution to system (2) is ergodic and has a distinct stationary distribution \(\pi (.)\) if \(R_0^s > 1\).
Proof
In order to confirm condition (i) of Lemma 1, we first create a nonnegative \(C^2-\) function \(V_4\) for this we define
where \(b_1, b_2, b_3\) are two positive constants defined in the latter, and \(\theta \in (1, 2)\) satisfies
using Itô’s lemma and model (2), we get drift term
Let
then
Consequently,
Finally,
where
Following that, we get
where
Then, we assume
where the constant \(b_5 > 0\) satisfies \(F_1(S) + F_3(C) + F_4(R) - b_4 b_5 \le -2\). Likewise, we may also get that
where
Now, we consider
where the constants \(\epsilon > 0\) in the set \(\mathbb {R}_+^4 D\) are suitably small and satisfy the following criteria :
where
Next, we will certify that \(LV_3 < 0\) on \(\mathbb {R}_+^4 D\). To keep things simple, we shall split \(\mathbb {R}_+^4\) into eight sections.
If \((S, I, C, R) \in D_1\) for case 1, we have
hence using Eq. (13), we may obtain that
If \((S, I, C, R) \in D_2\) for case 2, we have
hence using Eq. (14), we may obtain that
If \((S, I, C, R) \in D_3\) for case 3, we have
hence using Eq. (15), we may obtain that
If \((S, I, C, R) \in D_4\) for case 4, we have
hence using Eq. (16), we may obtain that
If \((S, I, C, R) \in D_5\) for case 5, we have
hence using Eq. (18), we may obtain that
If \((S, I, C, R) \in D_6\) for case 6, we have
hence using Eq. (19), we may obtain that
If \((S, I, C, R) \in D_7\) for case 7, we have
hence using Eq. (17), we may obtain that
If \((S, I, C, R) \in D_8\) for case 8, we have
hence using Eq. (20), we may obtain that
Thus, we get from Eqs (21)-(28) the following:
As of right now, Lemma 1 of condition (i) has been demonstrated. Additionally, the diffusion matrix of model (2) may be computed by the following,
Denote
we can get that
Consequently, condition (ii) of lemma 1 is also demonstrated. Therefore, system (2) has just one stationary distribution, according to lemma 1. \(\square\)
Optimal control problem
In the preceding sections, we examined the stability of the stochastic model (1). However, beyond stability analysis, it is crucial to focus on strategies for controlling the rate of elimination of diseases. In this section, deterministic and stochastic systems are the two different contexts in which we tackle the optimum control issue. We begin by analyzing the deterministic model (29), which serves as the counterpart to the stochastic system (2).
We modify models (2) and (29) by introducing two control variables: \(u_1(t)\), which represents efforts to reduce disease transmission (e.g., through vaccination, awareness campaigns, or outreach programs), and \(u_2(t)\), which represents treatment interventions aimed at reducing I(t) and preventing progression to C(t). The control set is defined as follows:
In this case, \(t_f\) stands for the control policy’s endpoint. Fully effective vaccinations are indicated by \(u_1(t) = 1\), whereas ineffective vaccinations are indicated by \(u_1(t) = 0\). Similarly, \(u_2(t) = 1\) denotes that treatment is entirely effective, whereas \(u_2(t) = 0\) indicates complete ineffectiveness of the treatment.
Optimal control strategies provide a systematic framework for reducing the disease’s impact, particularly chronic infections C(t), while accounting for the delayed dynamics of the system. By integrating prevention and treatment interventions, the model aims to promote a healthier population and develop a cost-efficient plan for combating the disease.
Without loss of generality, the primary objective for controlling and minimizing the given objective or cost function is the aim of the deterministic system (29):
subject to the following system:
with the initial conditions given in (3), Eqs. (30) and (31) are referred to as optimal control systems. In Eq. (30), the term \(\omega _1 I(t) + \omega _2 C(t)\) represents the cost associated with the infected population. The costs of therapy interventions and vaccinations are quantified by the positive constants \(\omega _4\) and \(\omega _3\), respectively. Eq. (30) highlights that both vaccination and treatment are resource-intensive, and the control system is designed to balance these costs effectively. The integrand function
shows the true cost at that moment t. In model (31), it is clear that four distinct types of people will always be positive and bound by any beginning value in the way that follows:
For simplicity, we abridge \(u_1 = u_1(t), u_2 = u_2(t)\).
Theorem 4
The optimum control \(u_1^*, u_2^* \in U\) for the optimal control systems (30) and (31) is to attain the least objective value, that is, \(J(u_1^*, u_2^*) = \underset{0\le u_i\le 1}{\min }\ J(u_1, u_2).\)
Proof
The theorem may be proven if the three requirements listed below are fulfilled:
-
(i)
The model (31) cannot have an empty set of solutions.
-
(ii)
The control variables may represent the state variables linearly, and U is both convex and closed.
-
(iii)
The integrand function \(L_1\) is convex within the control set, and \(L_1 \ge Z(u_1, u_2)\), where \(Z(u_1, u_2)\) is a continuous function. Furthermore, \(\Vert (u_1, u_2)^{-1} Z(u_1, u_2) \Vert \rightarrow \infty\) as \(\Vert (u_1, u_2) \Vert \rightarrow \infty\), with \(\Vert \cdot \Vert\) representing the \(L^2(0, T)\) norm.
In model (31), the whole population N satisfies the equation \(\frac{dN}{dt} = \Lambda - \alpha N - \mu C\), indicating that N is bounded, such that \(S + I + C + R \le \frac{\Lambda }{\alpha }\). The system (31) clearly satisfies the Lipschitz condition. Consequently, by applying the Picard-Lindelöf theorem (35), the (i) condition is fulfilled. Additionally, the definition of U ensures that it is both convex and closed. Moreover, the state variables are linearly dependent on the control variables, which satisfies condition (ii).
The function \(L_1\) is also convex. Specifically, \(L_{1} (S,I,C,R,u_{1} ,u_{2} ) =\) \(\omega _{1} I + \omega _{2} C +\) \(\frac{1}{2}(\omega _{3} u_{1}^{2} + \omega _{4} u_{2}^{2} ) \ge\) \(\frac{1}{2}(\omega _{3} u_{1}^{2} + \omega _{4} u_{2}^{2} )\).
Defining \(\omega _0 = \min (\omega _3, \omega _4) > 0\) and \(Z(u_1, u_2) =\) \(\frac{1}{2}\omega _0(u_1^2 + u_2^2)\), it follows that \(L_1(S, I, C, R, u_1, u_2) \ge Z(u_1, u_2)\). It is evident that \(Z(u_1, u_2)\) is continuous and satisfies \(\Vert (u_1, u_2)\Vert ^{-1}Z(u_1, u_2) \rightarrow \infty\) as \(\Vert (u_1, u_2)\Vert \rightarrow \infty\), thereby meeting condition (iii). Based on these results (35,36,37), the proof is complete. \(\square\)
We start by constructing the Hamilton function. \(H(S, I, C, R, u_1, u_2)\) in order to determine the optimality condition of the optimal control systems (30) and (31) as,
\(\lambda _i(i=S, I, C, R)\) is an adjoint variable in the function mentioned above. Here we develop the major theorems based on the classical PMP.
Theorem 5
Since the optimum control \(u_1^*, u_2^*\) is present in the optimal control systems (30) and (31), there are adjoint variables \(\lambda _S, \lambda _I, \lambda _C, \lambda _R\) that fulfill the following canonical equations:
with transversality conditions \(\lambda _i(t_f) = 0 \,(i=S, I, C, R)\). The matching optimum control \(u_1^*, u_2^*\) is provided as follows:
and
Proof
It is assumed that the optimal control systems (30) and (31) include the optimal controls \(u_1^*, u_2^*\). According to PMP, there are associated adjoint variables \(\lambda _S, \lambda _I, \lambda _C, \lambda _R\) that satisfy the canonical equations.
with transversality conditions \(\lambda _i(t_f) = 0\,(i=S, I, C, R)\).
According to classical calculus, the optimality requirement is satisfies \(\frac{\partial H}{\partial u_1} = 0\), at \(u_1 = u_1^*\) and \(\frac{\partial H}{\partial u_2} = 0\), at \(u_2 = u_2^*\). Thus, we obtain
when we combine the control set U definition, we obtain
and
Eqs.(32) and (33) are two possible ways to express it. This concludes the proof. \(\square\)
Stochastic optimal control
The following stochastic control system is obtained by taking into consideration the stochastic system (2) for efficiently regulating the cost function with the identical set of control variables.
with the initial value (3). Another way to represent System (34) in vector form is as
and
here, it is assumed that the components \(r_i\) and \(u_i\) are functions of t. Likewise, the original data may be redefined as:
The components of the functions f and q in vector form have the following structure
and
Our cost functional is provided by taking into consideration the characteristics of the quadratic control variables.
Here, \(k_i\) for \(i=1,\ldots ,4\) represent positive constants. With the goal of reducing infection rates while minimizing expenses or negative consequences, these functions are essential in assessing the effectiveness of interventions.
This study’s primary objective is to determine the ideal collection of control variables, \(u^*(t)=(u_2^*(t),u_1^*(t))\), that satisfies the following criteria:
in this case, the controls u are part of an acceptable set, which is represented as
the maximum permitted values of the corresponding control variables, which are positive constants that must be greater than one, are denoted by \(u_i^{\text {max}}\) in this case. It is crucial to specify the Hamiltonian function \(\text {H}(r,u,c,d)\) for the specified control issue as follows when using the stochastic maximum principle:
where the notation \(\langle \cdot ,\cdot \rangle\) denotes the Euclidean inner product and two sets of adjoint variables are represented by the variables \(c=[c_1,c_2,c_3,c_4]'\) and \(d=[d_1,d_2,d_3,d_4]'\). The suggested stochastic system is then taken into account by extending the classical maximum principle.
The symbol \(r^*(t)\) here represents the function r(t)’s optimum route. Relations (44) and (45) are connected with the following conditions:
these are known as the initial and terminal conditions, respectively. The relationship (46) makes it clear that the ideal control value is dependent on \(c(t)\), \(d(t)\), and \(r^*(t)\), which may be written as follows:
where the continuous function \(\phi\) may be calculated using the relation (46). It is possible to express the Hamiltonian from Eq.(43) as
Additionally, by applying the stochastic maximum principle that follows
one set of the adjoint equations is readily obtained a
Likewise, the initial and transversality conditions are eq. (3) and
and
where \(c_1(t_f) = -k_5S\), \(c_2(t_f) = -k_6I\), \(c_3(t_f) = -k_7C\), \(c_4(t_f) = -k_8R\). By differentiating the Hamiltonian equation with regards \(u_1\) and \(u_2\), the optimal control expressions \(u_1^*\) and \(u_2^*\) are obtained as follows:
Numerical simulation
In this study, we looked at how noise level and time delay affected the HCV model’s dynamic behavior and control schemes. Numerical simulations will also be used to validate the theoretical findings. In these simulations, the effects of the parameters on the stochastic optimal system and disease extinction, the stability of the system, and the use of the control technique will all be examined. For Python simulations, the stochastic Runge-Kutta fourth-order approach (RK4) is used. The following are the discretized equations that relate to model (2):
Comparison of the acute and chronic infected populations in the context of disease extinction is conducted for systems (2) and (29). The results are presented as follows: (a) The deterministic HCV model (29) for the acute infected population with different time delays. (b) The deterministic HCV model (29) for the chronically infected population with different time delays. (c) The stochastic HCV model (2) for the acute infected population with different time delays. (d) The stochastic HCV model (2) for the chronically infected population with different time delays.
In the system (2) \(\xi _{i,k}, i = 1, 2, 3, 4\) are independent and equally distributed random variables with a Gaussian distribution N(0, 1). Table 2 contains a list of the system’s parameter values.
To begin, we confirm that the stochastic model (2) has a stationary distribution over the time interval \(t \in [0, 200]\). Assuming the parameters from Table 2. we calculate the basic reproduction number as \(R^s_0 = 2.847805 > 1\). This suggests that the disease will continue, as reflected in the simulation results shown in Fig. 2b . For comparison, the deterministic model (29) was subjected to the identical parameters and initial conditions, producing the numerical simulation depicted in Fig. 2a . The trajectories of the four solid lines in the figure suggest that the model stabilizes into a steady state over time.
The simulation in Fig. 3a,b,c,d demonstrates how the dynamics of acute and chronic infected people are greatly impacted by the time delay \(\tau\). For the acute infected population, smaller time delays (\(\tau = 1\)) result in a rapid rise and higher peaks. In stochastic models, this leads to frequent fluctuations around the steady state, while deterministic models exhibit faster dynamics, with acute infections peaking earlier and stabilizing at slightly higher levels. Conversely, larger time delays (\(\tau = 6, 12\)) delay the peak of the acute population, which occurs at a lower magnitude. Stochastic models demonstrate a damping effect, with reduced fluctuations, while deterministic models show slower rises and prolonged stabilization.
For the chronic infected population, smaller time delays (\(\tau = 1\)) lead to earlier peaks and lower steady-state levels. In stochastic models, the chronic population declines quickly after peaking, while deterministic models show a faster stabilization at lower levels. Larger time delays (\(\tau = 6, 12\)) result in a more gradual rise in the chronic population, with delayed peaks and slower declines. Stabilization occurs later, and the steady-state levels are slightly higher compared to smaller delays. The fluctuations in stochastic models are smaller, indicating reduced variability in chronic infections as the delay increases.
The population dynamics for the deterministic model (29) with and without control along time (days). (a) for S(t) individuals, (b) for I(t) individuals, (c) for C(t) individuals, and (d) for R(t) individuals.
In summary, smaller delays amplify infection rates, leading to quicker peaks and faster stabilization for both acute and chronic infections. On the other hand, larger delays slow down the progression of infections, delay peaks, and result in more prolonged stabilization periods, with lower acute peaks and slightly higher steady-state levels for chronic infections. These findings highlight the crucial role of time delays in shaping the infection dynamics, influencing both the timing and intensity of infection peaks as well as the stability of chronic infection levels.
The graphical results in Fig. 4a to 4d demonstrate the stark contrast between the controlled and uncontrolled scenarios. For the susceptible population S(t), the dynamics in Fig. 4a show that optimal control significantly reduces the number of susceptible individuals, helping to curtail the spread of the infection more effectively. For the acutely infected population I(t), as shown in Fig. 4b , optimal control leads to a much faster decline in infections, with the disease nearing extinction earlier compared to the uncontrolled case. Similarly, for the chronically infected population C(t), displayed in Fig. 4 , optimal control measures result in a more pronounced reduction in chronic infections, reflecting the effectiveness of the interventions in managing long-term cases. Lastly, the recovered population R(t) in Fig. 4d exhibits significant differences, where the controlled scenario stabilizes at a lower value, indicating that fewer individuals transition to recovery due to the prevention of new infections. These findings clearly illustrate that under controlled conditions, the number of infected individuals-both acute and chronic-declines more rapidly, leading to the faster extinction of the disease. The results highlight the importance of vaccination and treatment interventions as optimal control measures to achieve better health outcomes and efficient disease management.
Optimal control variable profiles for the deterministic model (29) are presented: (a) \(u_1^*(t)\) along time (days) and (b) \(u_2^*(t)\) along time (days).
The results presented in Fig. 5 illustrate the population dynamics of the deterministic model (29) with and without the implementation of optimal control strategies. The focus is on understanding how vaccination and treatment interventions impact the dynamics of four key groups: S(t), I(t), C(t), and R(t). These simulations also examine the optimal control problem for systems (30) and (31), comparing the outcomes with the uncontrolled model (29). The simulations covered a time span of \(0-500\) units, assuming that the costs of vaccination and treatment were not prohibitively high. The initial population values were set at \(S(0) = 100\), \(I(0) = 20\), \(C(0) = 10\), and \(R(0) = 20\). In Fig. 5a , the vaccination control \(u_1^*(t)\) starts at its maximum value (1.0), indicating an aggressive initial vaccination effort to reduce the susceptible population and prevent new infections. Over time, as the susceptible population decreases and the infection is brought under control, the vaccination effort gradually diminishes, with \(u_1^*(t)\) approaching zero. This indicates that as the disease nears eradication, further vaccination is no longer necessary. Similarly, Fig. 5b shows the treatment control \(u_2^*(t)\), which also begins at its maximum value (1.0) to address the infected population robustly. As the infection declines due to effective treatment, the value of \(u_2^*(t)\) steadily decreases, eventually reaching near-zero levels as the disease is controlled and eliminated. These results highlight the effectiveness of the optimal control strategy, which dynamically adjusts vaccination and treatment efforts over time. The strategy emphasizes an intense initial response to combat the infection, followed by a gradual reduction in interventions, ensuring efficient resource utilization while effectively managing and eradicating the disease. The profiles represent the time-dependent application of vaccination \(u_1^*(t)\) and treatment \(u_2^*(t)\) aimed at minimizing the spread of infection and reducing associated costs.
The results in Fig. 6 use the stochastic RK4 method with initial values in Table 2 to show the population dynamics of the stochastic model (34) with and without the use of optimal control. The adjoint equations and the stochastic system were successively assessed under the transversality requirement in order to determine the best control procedure. Initially, the RK4 method was used to solve the stochastic system (34). Next, the adjoint system (52) was solved using a backward iterative method while adhering to the transversality conditions (53) and (54). The control variables were then updated based on the relationship outlined in (55), and the iteration process was terminated once the error between successive iterations fell below a specified tolerance.
The dynamics of the susceptible population, \(S(t)\), displayed in Fig. 6a , indicate a sharp decline under optimal control compared to the uncontrolled scenario, demonstrating the effectiveness of vaccination in reducing the susceptible individuals. Figure 6b highlights the dynamics of the infected individuals, \(I(t)\), which shows a significant decrease when control strategies are applied, signifying the role of optimal vaccination and treatment in mitigating the spread of infection. For the chronically infected population, \(C(t)\), Fig. 6c reveals a notable reduction under control, further emphasizing the benefits of treatment interventions. Finally, Fig. 6d shows the dynamics of the recovered population, \(R(t)\), which increase more rapidly with control, reflecting improved recovery rates facilitated by effective interventions. Overall, these results highlight the critical role of optimal control strategies, such as vaccination and treatment, in reducing infection levels, enhancing recovery, and efficiently managing the disease.
The population dynamics for the stochastic model (34) with and without control along time (days) (a) for S(t) individuals, (b) for I(t) individuals, (c) for C(t) individuals, and (d) for R(t) individuals.
Optimal control variable profiles for the stochastic model (34) are presented: (a) \(u_1^*(t)\) along time (days) and (b) \(u_2^*(t)\) along time (days).
Figure 7 illustrates the optimal control variable profiles \(u_1^*(t)\) and \(u_2^*(t)\) for the stochastic model (34) and (52) Subplot 7a shows the profile of \(u_1^*(t)\), which represents the vaccination control strategy. The control variable fluctuates dynamically between its lower and upper bounds, reflecting intermittent vaccination efforts aimed at minimizing the susceptible population while balancing effectiveness with resource constraints. Subplot 7b depicts the profile of \(u_2^*(t)\), corresponding to the treatment control strategy for the infected population. The control variable rises sharply at the beginning, indicating an aggressive treatment approach to rapidly reduce the infection, followed by stabilization at lower levels as the number of diseased individuals declines. These results demonstrate the efficiency of optimal control strategies in managing disease progression by reducing the susceptible and infected individuals while ensuring the efficient use of resources.
Conclusion
In this study, a SDDE model was developed to better understand the complex transmission dynamics of the HCV under the influence of environmental fluctuations and time delays. The model captures essential disease processes, including natural progression from acute to chronic stages, disease-induced mortality, and control strategies, while incorporating stochastic perturbations that represent random environmental and demographic influences. A new Lyapunov functional incorporating integral terms for delays was constructed to analyze the stability of the system. This approach facilitated the establishment of key theoretical properties such as the existence and uniqueness of global positive solutions, ergodicity, and the existence of stationary distributions. Numerical simulations validated the theoretical findings and revealed that both time delay and noise intensity are critical factors influencing HCV dynamics, and longer time delays were shown to prolong the disease outbreak, emphasizing the necessity of timely interventions. A major contribution of this work lies in the formulation and analysis of optimal and stochastic optimal control strategies. By applying PMP in the stochastic setting, we derived necessary conditions for optimal interventions. The resulting forward-backward stochastic differential system was solved using the forward-backward sweep method. The controls, representing vaccination and treatment efforts, were optimized to minimize the number of infected individuals while also reducing the cost of interventions. The simulations demonstrated that optimal control significantly reduces the disease burden. Moreover, the incorporation of stochasticity into the control design enables more robust decision-making in uncertain environments. The framework allows for adaptive interventions that account for variability in disease dynamics, making it more suitable for real-world public health applications where uncertainty is unavoidable. Hence, this study provides a comprehensive framework that integrates stochastic analysis, time-delay dynamics, and optimal control strategies to guide effective HCV management.
Future Directions: To enhance the model’s applicability and realism, future research could explore multi-strain HCV dynamics, include spatial heterogeneity, and investigate the effects of different vaccine deployment strategies. Additionally, coupling this framework with real-time data and machine learning techniques could improve prediction accuracy and inform more adaptive public health interventions. Interactions with co-infections and socioeconomic factors may also be integrated for broader applicability
Data availability
All data generated or analysed during this study are included in this article.
References
World Health Organization. Hepatitis C https://www.who.int/news-room/factsheets/detail/hepatitis-c (2024).
Misra, A. K., Maurya, J. & Sajid, M. Modeling the effect of time delay in the increment of number of hospital beds to control an infectious disease. Math. Biosci. Eng. 19, 11628–11656. https://doi.org/10.3934/mbe.2022541 (2022).
Kuang, Y. Delay Differential Equations (Academic Press, New York, 1993).
Mao, X. Stochastic Differential Equations and Applications (Horwood Publishing, New York, 1997).
Zhang, S. & Xu, X. Dynamic analysis and optimal control for a model of hepatitis c with treatment. Commun. Nonlinear Sci. Numer. Simul. 46, 14–25. https://doi.org/10.1016/j.cnsns.2016.10.017 (2017).
Martcheva, J. & Castillo-Chavez, C. Diseases with chronic stage in a population with varying size. Math. Biosci. 182, 1–25. https://doi.org/10.1016/S0025-5564(02)00184-0 (2005).
Yuan, J. & Yang, Z. Global dynamics of an sei model with acute and chronic stages. J. Comput. Appl. Math. 213, 465–476. https://doi.org/10.1016/j.cam.2007.01.042 (2008).
Zhang, S. & Zhou, Y. Dynamics and application of an epidemiological model for hepatitis c. Math. Comput. Model. 56, 36–42. https://doi.org/10.1016/j.mcm.2011.11.081 (2012).
Hu, X. & Sun, F. Threshold dynamics for an epidemic model with acute and chronic stages. Int. J. Nonlinear Sci. 12, 105–111 (2011).
Okosun, K. O. & Makinde, O. D. Optimal control analysis of hepatitis c virus with acute and chronic stages in the presence of treatment and infected immigrants. Int. J. Biomath. 7, 1–23. https://doi.org/10.1142/S1793524514500193 (2014).
Khan, A. Q., Yaqoob, A. & Alsaadi, A. Discrete hepatitis c virus model with local dynamics, chaos and bifurcations. AIMS Mathematics 9, 28643–28670. https://doi.org/10.3934/math.20241390 (2024).
Din, Anwarud. Bifurcation analysis of a delayed stochastic HBV epidemic model: Cell-to-cell transmission. Chaos Solit. Fractals 181, 114714. https://doi.org/10.1016/j.chaos.2024.114714 (2024).
Din, A. & Li, Y. Optimizing HIV/AIDS dynamics: stochastic control strategies with education and treatment. Eur. Phys. J. Plus 139, 812. https://doi.org/10.1140/epjp/s13360-024-05605-1 (2024).
Khan, W.A., Zarin, R., Zeb, A., Khan, Y.,& Khan, A. Navigating food allergy dynamics via a novel fractional mathematical model for antacid-induced allergies. J. Math. Tech. Model. 1, 25–51. https://doi.org/10.56868/jmtm.v1i1.3 (2024).
Ullah, S. Investigating a coupled system of Mittag-Leffler type fractional differential equations with coupled integral boundary conditions. J. Math. Tech. Model. 1, 16–28 (2024).
Tul Ain, Q. Nonlinear stochastic cholera epidemic model under the influence of noise. J. Math. Tech. Model. 1, 52–74 (2024).
Shah, S.M.A., Tahir, H., Khan, A., Khan, W.A. & Arshad, A. Stochastic model on the transmission of worms in wireless sensor network. J. Math. Tech. Model. 1, 75–88 (2024).
Khan, A., Ikram, R., Din, A., Humphries, U. W. & Akgul, A. Stochastic covid-19 seiq epidemic model with time delay. Results Phys. 30, 104775. https://doi.org/10.1016/j.rinp.2021.104775 (2021).
Ji, C. & Jiang, D. Threshold behaviour of a stochastic sir model. Appl. Math. Model. 39, 5067–5076. https://doi.org/10.1016/j.apm.2014.03.037 (2014).
Zhao, Y. & Jiang, D. The threshold of a stochastic sirs epidemic model with saturated incidence. Appl. Math. Lett. 34, 90–93. https://doi.org/10.1016/j.aml.2013.11.002 (2014).
Din, A. & Li, Y. Controlling heroin addiction via age-structured modeling. Adv. Differ. Equations 2020, 521. https://doi.org/10.1186/s13662-020-02983-5 (2020).
Din, A. & Li, Y. Stationary distribution extinction and optimal control for the stochastic hepatitis b epidemic model with partial immunity. Physica Scripta 96, 074005. https://doi.org/10.1088/1402-4896/abfacc (2021).
Din, A., Li, Y., Khan, T. & Zaman, G. Mathematical analysis of spread and control of the novel coronavirus (covid-19) in china. Chaos Solit. Fractals 141, 110286. https://doi.org/10.1016/j.chaos.2020.110286 (2020).
Yusuf, A., Acay, B., Mustapha, U. T., Inc, M. & Baleanu, D. Mathematical modeling of pine wilt disease with caputo fractional operator. Chaos Solit. Fractals 143, 110569. https://doi.org/10.1016/j.chaos.2020.110569 (2021).
Liu, Q., Jiang, D., Shi, N., Hayat, T. & Alsaedi, A. Stationary distribution and extinction of a stochastic sirs epidemic model with standard incidence. Phys. A: Stat. Mech. Appl. 469, 510–517. https://doi.org/10.1016/j.physa.2016.11.077 (2017).
Li, F., Zhang, S. & Meng, X. Dynamics analysis and numerical simulations of a delayed stochastic epidemic model subject to a general response function. Comput. Appl. Math. 38, 95. https://doi.org/10.1007/s40314-019-0857-x (2019).
Liu, Rong & Guo, Ke. Insights into unexpected relapse and recovery in HCV-infected patients by studying a stochastic within-host HCV model. Appl. Math. Lett. 149, 108937. https://doi.org/10.1016/j.aml.2023.108937 (2024).
Faris, J. W. Stochastic differential equations model for the interaction of HCV with liver cells. Acad. Sci. J. 2, 241–257 https://doi.org/10.24237/ASJ.02.04.794B (2024).
Cui, J.-A. et al. Global dynamics of an epidemiological model with acute and chronic hcv infections. Appl. Math. Lett. 103, 106203. https://doi.org/10.1016/j.aml.2019.106203 (2020).
Mao, X., Marion, G. & Renshaw, E. Environmental brownian noise suppresses explosions in population dynamics. Stoch. Process. their Appl. 97, 95–110. https://doi.org/10.1016/S0304-4149(01)00126-0 (2002).
Zhang, X.-B., Wang, X.-D. & Huo, H.-F. Extinction and stationary distribution of a stochastic sirs epidemic model with standard incidence rate and partial immunity. Phys. A: Stat. Mech. Appl. 531, 121548. https://doi.org/10.1016/j.physa.2019.121548 (2019).
Khasminskii, R. Stochastic Stability of Differential Equations (Springer Science & Business Media, Berlin, 2011).
Allen, L. J. S. An introduction to stochastic epidemic models. In Mathematical Epidemiology, vol. 1945 of Lecture Notes in Mathematics, 81–130, https://doi.org/10.1007/978-3-540-78911-6_3 (Springer, 2008).
Gray, A., Greenhalgh, D., Hu, L., Mao, X. & Pan, J. A stochastic differential equation SIS epidemic model. SIAM J. on Appl. Math. 71, 876–902. https://doi.org/10.1137/100789766 (2011).
Coddington, E. & Levinson, N. Theory of Ordinary Differential Equations (Tata McGraw-Hill Education, New York, 1955).
Russell, S. The economic burden of illness for households in developing countries: a review of studies focusing on malaria, tuberculosis, and human immunodeficiency virus/acquired immunodeficiency syndrome. Am. J. Trop. Medicine Hyg. 71, 147–155 (2004).
Gaff, H. D., Schaefer, E. & Lenhart, S. Use of optimal control models to predict treatment time for managing tick-borne disease. J. Biol. Dyn. 5, 517–530. https://doi.org/10.1080/17513758.2010.535910 (2011).
Acknowledgements
The Researchers would like to thank the Deanship of Graduate Studies and Scientific Research at Qassim University for financial support (QU-APC-2025).
Author information
Authors and Affiliations
Contributions
N.K. wrote the main manuscript text. T.S.C. has done conceptualization and supervision. T.S.C., I.S.C. and M.S. have provided methodology. N.K., T.S.C. and I.S.C. have done investigation. N.K. has done formal analysis and used software. M.S. has initiated funding. N.K., T.S.C., I.S.C. and M.S. have done validation. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kumar, N., Sajid, M., Chauhan, T.S. et al. Simulation and optimal control of stochastic delay differential models for hepatitis C virus epidemics. Sci Rep 16, 813 (2026). https://doi.org/10.1038/s41598-025-30528-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-30528-x