Abstract
Acute coronary syndrome (ACS) presents sex-based differences in pathophysiology. Variations in biomarker patterns post-ACS, reflecting myocardial injury, vascular inflammation, and remodeling, may indicate critical differences in cardiovascular disease mechanisms and outcomes. We analyzed biomarker patterns in 787 patients (22% females) from the BIOMArCS study, all without re-ACS during the study period. We tracked levels of hs-cTnT, NT-proBNP, hs-CRP, GDF-15, and additional biomarkers in a subcohort of 191 patients over one year. Serial blood samples were collected to compare acute-phase (first month after ACS) and stabilized-phase (2–12 months post-ACS) biomarker trajectories between sexes, adjusting for age, BMI, and kidney function using linear mixed-effects models. Females showed significantly lower hs-cTnT levels (mean 386 pg/mL versus 559 pg/mL in males, p = 0.002 acute phase; 8.5 pg/mL versus 10.8 pg/mL, p < 0.001 at 180 days). NT-proBNP levels were higher in females (mean 70 pmol/L vs. 47 pmol/L, p < 0.001 acute phase; 30 pmol/L vs. 19 pmol/L, p < 0.001 at 180 days). Hs-CRP levels were also elevated in females (mean 1.8 mg/L vs. 1.5 mg/L, p = 0.02 at 180 days). Galectin-3 levels remained higher in females (22.8 ng/mL vs. 18.6 ng/mL, p = 0.03). This study provides the first comprehensive analysis of sex-specific biomarker trajectories following ACS. Distinct differences in hs-cTnT, NT-proBNP, and inflammatory markers suggest that sex-specific diagnostic thresholds and personalized treatment strategies after ACS may be warranted, although their clinical value still needs confirmation in larger prospective studies.
Similar content being viewed by others
Introduction
The pathogenesis of acute coronary syndrome (ACS) in females and males differs. A key factor is the difference in the anatomy of the epicardial coronary arteries, as females tend to have smaller arteries than males. Females have a higher baseline myocardial blood flow, increasing shear stress in the coronary arteries. This higher shear stress is associated with less focal lipid accumulation, lower pathologic remodeling, and less plaque instability, leading to a lower incidence and later presentation of ACS in females1. Moreover, the cardioprotective effects of oestrogen in premenopausal females contribute to reduced atherosclerosis progression and a lower risk of ACS2. Additionally,females exhibit less severe and more diffuse atherosclerosis and fewer lipid-rich atheromata3,4,5,6.
Biomarkers can reflect parts of the underlying pathophysiological processes of atherosclerosis, including lipid metabolism, vascular inflammation, endothelial dysfunction, increased thrombogenicity, myocardial injury, remodeling, and fibrosis7,8,9. Therefore, the analysis of laboratory biomarkers may help predict an increased vulnerability to developing a first or repeat ACS event and the ensuing increased risk of death. Differences in biomarker profiles between females and males may highlight potentially significant sex-based differences in the pathophysiological mechanisms contributing to cardiovascular disease (CVD) and, more specifically, ACS. For instance, previous studies on cardiac troponin T (cTnT) have shown that reference limits vary by age and sex in population-based and hospital cohorts10,11. Additionally, research has identified sex differences in cardiac biomarker measurements of cTnT, N-terminal pro-B-type natriuretic peptide (NT-proBNP), and soluble suppression of tumorigenicity 2 (sST2) in population-based cohorts and chronic diseases such as chronic kidney disease and diabetes12,13,14,15,16.
Against this background, knowledge about sex-dependent serial biomarker patterns shortly after the presentation for ACS and the following stabilized phase is needed to determine representative cut-off values in females and males to improve diagnosis of repeated ACS and, in the future, for more personalized therapy management after the acute event. However, information on sex differences in serial measurements of biomarker levels is still lacking. With the current study, we want to gain more insight into the differences in the evolution of biomarkers between females and males up to one year after ACS admission.
Methods
Study design and study cohorts
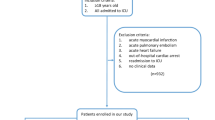
We used data from the BIOMarker Study to Identify the Acute Risk of Coronary Syndrome (BIOMArCS), a prospective observational study. Details of the study design and population have been published previously7,17. In brief, the study enrolled 844 ACS patients (187 females and 657 males) aged 40 years or older with at least one cardiovascular (CV) risk factor. Participants were recruited across 18 hospitals in The Netherlands from 2008 to 2015. Patients with myocardial ischemia caused by conditions other than atherosclerotic coronary artery disease (CAD), severe heart failure (left ventricular ejection fraction < 30% or NYHA class III or IV), or severe chronic kidney disease (eGFR < 30 ml/min/1.73 m22) were excluded. For the majority of patients, the first blood sample was taken within 4 weeks after the index-ACS (n = 569). For the other patients, the first blood sample was taken at their first outpatient visit within 6 weeks after the index-ACS. After the acute phase (first 30 days), blood was collected every 2 weeks up to month 6 after the index-ACS, followed by monthly blood collection until 1 year after the index-ACS (see Fig. S1). The primary endpoint was defined as the composite of cardiovascular death or hospital admission for non-fatal ACS. The medical ethics committees of the participating hospitals approved our study. All patients gave written informed consent. The study was performed in accordance with all relevant guidelines and regulations, as well as the Declaration of Helsinki.
For the current analysis, we excluded patients who reached the primary endpoint (n = 45) to ensure an unbiased analysis of biomarker trajectories following ACS without distortion from recurrent ACS events. Twelve participants were lost to follow-up due to death other than cardiovascular reasons or discontinuation after the acute phase of the index-ACS. This resulted in a final study cohort (full cohort) of 787 patients (173 females, 22%; see Fig. 1) in which we measured four biomarkers (see below). On average, 14.0 (median 17.0, IQR 8.0–19.0) serial samples were collected per female patient and 15.1 (median 17.0, IQR 13.0–19.0) per male patient over 1-year follow-up. Additionally, we measured a set of nine biomarkers in a subset of 191 patients (subcohort) without a recurrent AC. The subcohort consisted of 142 patients who were randomly selected from the full cohort of 844 patients, supplemented with 49 patients who underwent intensive blood sampling during the first 4 days as described in18 and which included 43 females (23%) and 148 males (77%). These patients contributed an average of 5.4 serial samples per female and 5.6 per male.
Flow chart of the study cohorts and the biomarkers measured in each cohort. The full cohort included all patients from the BIOMArCS cohort with a complete dataset without a primary endpoint; four biomarkers were measured in all samples. The subcohort is a subset of the full cohort, with nine additional biomarkers measured in this cohort. This figure was created in Biorender.com.
Biomarkers
The following biomarkers were analyzed using commercially available, validated immunoassays according to the manufacturers’ protocols, as detailed in Supplementary Table S1: high-sensitivity cardiac troponin T (hs-cTnT), high-sensitivity cardiac troponin I (hs-cTnI), high-sensitivity N-terminal pro-B-type natriuretic peptide (hs-NT-proBNP), high-sensitivity C-reactive protein (hs-CRP), Growth Differentiation Factor-15 (GDF-15), soluble suppression of tumorigenicity 2 (sST2), creatinine, HDL-cholesterol, and total cholesterol. LDL-cholesterol was calculated using the Friedewald formula. For Myeloperoxidase (MPO), Galectin-3 (Gal-3), von Willebrand Factor (vWF), and Cystatin C, in-house assays developed and validated by the laboratory at the University Medical Center (UMC) Utrecht, The Netherlands, were used. Detailed information on the composition and validation of these in-house assays is available upon request. The selected biomarkers reflect distinct pathophysiological atherosclerotic pathways or have been shown to adequately predict the risks of future CV events or long-term consequences of an ACS like heart failure7,9.
Statistical analysis
We present continuous variables as median, 25th, and 75th percentile categorical data as numbers with percentages. We used the Mann–Whitney (continuous variables) and chi-squared or Fisher’s exact test as appropriate (categorical variable) to evaluate differences between females and males. If one of the tests showed a p-value < 0.05, the effect size was calculated in the Mann–Whitney test using the Rank-Biserial Correlation, for the Chi-square test using Cramér’s V, and for Fisher’s exact test using the Phi coefficient. The measured biomarkers showed a skewed distribution and were log2-transformed for further analysis, but we display results on a linear scale for the benefit of clinical applicability.
Linear mixed-effect models were applied to describe the changes in biomarker levels after the index-ACS according to sex while accounting for the correlation between serial follow-up measurements in each patient. In our models, we adjusted for age, body mass index (BMI), time from index-ACS, and kidney function in the case of the subcohort. Additionally, we included an interaction term to assess how sex influences biomarker levels over time following the index ACS. We calculated separate linear mixed-effects models for each biomarker, focusing on the acute phase (less than 30 days after the ACS) and the stabilized phase (30 days or more after the ACS).
We used the R software version 4.4.019 to perform all analyses. Linear mixed-effect models were calculated using the lme function from the nlme package20. We calculated confidence intervals at the 95% level and considered a p-value below 0.05 (two-sided test) statistically significant.
Results
Study cohorts
Table 1 presents the baseline characteristics of the full cohort and subcohort, stratified by sex. Some baseline characteristics differed slightly between sexes, as detailed below. Among patients without a primary endpoint in the BIOMArCS study, statistically significant sex differences were observed in heart rate (median: 73 bpm in females vs. 71 bpm in males), systolic blood pressure (median: 144.5 vs. 136.0 mmHg), history of hypertension (61.3 vs. 53.6%), smoking status (42.2% vs. 39.7%), percutaneous coronary intervention (PCI) (71.1 vs. 83.4%), and discharge with ACE inhibitor medication (71.1 vs. 83.4%), though all had small effect sizes. Serum creatinine levels were significantly lower in females than in males (median: 68.0 vs. 85.0 µmol/L in the full cohort), with a medium effect size. The subcohort results showed similar trends. Unexpectedly, there were no statistically significant sex differences in ACS type, history of heart failure, or history of diabetes.
Biomarker patterns in the acute phase following the index ACS
We observed similar patterns for most biomarkers in both males and females in the full cohort immediately after the index ACS (see Table 2). As expected from the definition of an ACS, both sexes exhibited elevated cTnT levels in the full cohort, with males showing significantly higher values compared to females (mean hs-cTnT: 559 pg/mL in males vs. 386 pg/mL in females). Hs-NT-proBNP levels were also elevated in both sexes but significantly higher in females (mean hs-NT-proBNP: 70 pmol/L in females vs. 47 pmol/L in males). Hs-CRP and GDF-15 levels were slightly higher in females than in males; however, these differences did not reach statistical significance.
The results in the subcohort (see Table 3) showed elevated cTnI levels in both males and females shortly after the index ACS (mean TnI: 2445 pg/mL in females vs. 3644 pg/mL in males). HDL levels differed significantly between females and males (mean HDL: 1.08 mmol/L in females vs. 0.98 mmol/L in males, p = 0.009). Galectin-3 levels were significantly higher in females than males (mean Galectin-3: 25.4 ng/mL in females vs. 18.9 ng/mL in males). Cystatin C, sST2, and LDL levels were slightly elevated in both sexes with no statistically significant difference.
Biomarker pattern in the clinically stabilized phase after the index ACS
Biomarker patterns were similar in both sexes during the stabilized phase following the initial ACS event in the full cohort, as summarized in Table 4 and Fig. 2. The levels of hs-cTnT returned to a normal range in median, though females exhibited significantly lower hs-cTnT values than males from 30 days post-ACS (mean hs-cTnT after 30 days: 9.9 pg/mL in females vs. 12.5 pg/mL in males, p < 0.001) through six months (mean hs-cTnT after 6 months: 8.5 pg/mL in females vs. 10.8 pg/mL in males). The decline was faster in males compared to females (p = 0.01) during the stabilized phase. Although the median hs-cTnT levels were within the normal range, 21% of males and 12% of females had values above the upper reference limit of 14 pg/mL six months after the index-ACS.
cTnT, NT-proBNP, CRP, and GDF-15 levels in the full cohort. The results are based on linear mixed effect models, with log(biomarker) as the dependent variable and sex, age, BMI, and time of the venipuncture since the index acute coronary syndrome (ACS) as explanatory variables. Top row: cTnT (left) and NT-proBNP (right). Bottom row: CRP (left) and GDF-15 (right). cTnT and NT-proBNP, and CRP were significantly different among the sexes in the stabilized phase.
Meanwhile, hs-NT-proBNP levels were significantly higher in females than males at 30 days post-ACS (mean hs-NT-proBNP after 30 days: 40 pmol/L in females vs. 26 pmol/L in males, p < 0.001) and 180 days post-ACS (mean hs-NT-proBNP after 6 months: 30 pmol/L in females vs. 19 pmol/L in males). Hs-CRP levels were also notably higher in females during the clinically stabilized phase post-ACS at both time points 30 and 180 days after the index ACS (mean hs-CRP after 30 days: 1.9 mg/L in females vs. 1.5 mg/L in males; mean hs-CRP after 180 days: 1.8 mg/L in females vs. 1.5 mg/L in males, p = 0.018). Notably, the confidence intervals overlapped for all biomarkers, even when reaching statistical significance.
In the sub cohort (see Table 5 and Fig. 3) we found sST2 levels significantly lower in females than males (mean sST2 after 180 days: 26.4 ng/mL in females vs. 29.8 ng/mL in males, p = 0.022). Galectin-3 levels were significantly higher in females than males (mean Galectin-3 after 180 days: 22.8 ng/mL in females vs. 18.6 ng/mL in males, p = 0.029). The HDL levels remained significantly higher in females during the stabilized phase (mean HDL after 180 days: 1.14 mmol/L in females vs. 1.03 mmol/L in males, p = 0.012).
Biomarker levels in the subcohort. The results are based on linear mixed effect models, with log(biomarker) as the dependent variable and sex, age, BMI, kidney function, and time of the venipuncture since the index acute coronary syndrome (ACS) as explanatory variables. Top row: cTnI, cystatin C, and sST2 levels. sST2 levels differed statistically significant in the stabilized phase. Middle row: Galectin-3, vWF, and MPO levels. Levels of Galectin-3 were significantly higher in females in the stabilized phase. Bottom row: Total, HDL-, and LDL-cholesterol. Levels of HDL cholesterol were significantly higher in females in the stabilized phase.
Discussion
Here, we present the first study that describes sex differences in cardiovascular biomarker patterns up to one year after an ACS based on high-frequency serial measurements. The study highlights significant variations in cTnT, NT-proBNP, hs-CRP, HDL, and Galectin-3 levels, but with overlapping confidence intervals despite statistically significant differences.
The observed sex-specific biomarker differences may not solely reflect biological sex but may also be influenced by associated clinical factors or gender-related factors (e.g., smoking habits). In our cohort, as shown in Table 1, there were no significant sex differences in ACS type (STEMI, NSTEMI, or unstable angina), history of heart failure, or age, suggesting that these factors are unlikely to account for the observed differences. However, differences in myocardial volume, hemodynamics, or other unmeasured factors cannot be excluded, as we observed sex differences in hypertension prevalence, systolic blood pressure, and smoking history, though with small effect sizes, limiting their potential influence on the observed results.
We found lower adjusted hs-cTnT levels in females compared to males in both the acute and stabilized phases, consistent with prior findings 16. These lower troponin levels in females primarily reflect the smaller left ventricular mass in females21,22,23. Additional factors may include sex-specific CAD pathophysiology24 and the cardioprotective effects of estrogen, which attenuates myocardial remodeling and hypertrophy via receptor-mediated pathways, particularly in premenopausal females6,25,26. The lower age quantile of 53.2 years in females indicates that most participants were post-menopausal, and the protective effect of estrogens is likely reduced. A notable observation in our study is that hs-cTnT and hs-cTnI frequently remain elevated long after the index ACS event, consistent with earlier analyses of the BIOMArCS subcohort by van den Berg et al.27. These sustained elevations most likely reflect ongoing pathological processes such as microvascular dysfunction, wall stress, and cardiac remodeling, which may differ by sex28,29, underscoring the need for individualized, rather than population-based, interpretation in both sexes. Still, the general clinical relevance of lower troponin levels in females remains controversial30. Although no significant sex differences in troponin I levels were observed in our study, this finding should be interpreted with caution. Other studies showed apparent sex-related differences in TnI levels16; thus, the lack of statistical significance may not imply the absence of actual biological differences. Our findings suggest that while sex-specific patterns may exist for some cardiac biomarkers, they may not be consistently detectable across all studies or populations.
In contrast, the adjusted NT-proBNP levels were significantly higher in females than in males during both study phases. While hormonal influences, such as estrogen-mediated effects on natriuretic peptide secretion, may contribute31, the higher prevalence of diastolic dysfunction and comorbidities, including hypertension and atrial fibrillation in older females, likely represents a more substantial driver of these sex differences32. Elevated NT-proBNP levels in females have also been linked to an increased risk of heart failure and mortality33,34. These findings underscore the need to explore sex-specific reference values for NT-proBNP in post-ACS risk assessment.
We observed elevated hs-CRP levels in females during the stabilized phase, suggesting sex-specific inflammatory responses post-ACS. CRP has long been linked to vascular inflammation and cardiovascular disease manifestations such as myocardial infarction, and numerous studies have supported its role in cardiovascular risk prediction35. Evidence on sex differences in CRP, however, remains inconsistent: some European population-based cohorts report no systematic differences between females and males36, whereas other studies have shown higher hs-CRP concentrations in females in healthy populations as well as in patients with stable CAD or ACS37,38,39,40. Taken together, these findings suggest that elevated CRP levels in females may indicate subtle differences in systemic inflammatory activity that are not fully explained by adiposity or cardiac remodeling alone. Such differences could contribute to sex-specific cardiovascular risk patterns, although the underlying mechanisms remain speculative and warrant further investigation. To further assess inflammatory activity, we measured MPO levels in our subcohort and found similar MPO concentrations in females and males.
During both the acute and stabilized phases after ACS, females exhibited higher adjusted Galectin-3 concentrations compared to males. Population-based studies have consistently reported higher Galectin-3 levels in females than in males, although the underlying reasons remain incompletely understood. These sex differences likely reflect a combination of systemic inflammatory activity, sex-specific cardiac remodeling, and differences in body composition41,42,43. Although BMI was accounted for in our analyses, it does not fully capture sex-specific differences in body composition. Females and males with similar BMI can differ in fat percentage, lean mass, and fat distribution, which may influence circulating biomarkers such as Galectin-3. Therefore, the higher levels observed in females could, at least in part, reflect underlying differences in body composition, with possible contributions from systemic inflammation and, to a lesser extent, hormonal influences such as estrogen-mediated modulation.
Significantly higher values for high-density lipoprotein (HDL) cholesterol were observed in females compared to males during both investigated phases, with adjusted values falling below the generally recommended levels (> 1.2 mmol/L in females and 1.0 mmol/L in males; see44). Premenopausal females have a less atherogenic plasma lipid profile than age-matched males45. However, after menopause, lipid profiles become less distinct, but HDL levels remain persistently higher13 consistent with our findings.
While our study was not designed to develop diagnostic or prognostic models, it underscores the importance of accounting for biological sex when interpreting cardiovascular biomarkers. The observed sex-specific patterns—particularly for hs-cTnT, NT-proBNP, hs-CRP, Galectin-3, and HDL—may be relevant to future strategies for individualized risk assessment after ACS. Incorporating sex-specific reference values or differential weighting of risk factors, as seen in the SEX-SHOCK study46, may enhance cardiovascular risk prediction. However, the idea of using sex-specific thresholds or algorithms should be approached with caution, as previous studies have not shown clear improvements in management or outcomes when applying such approaches47,48. Therefore, while our results indicate biologically significant differences, their clinical value needs validation through larger, prospective studies.
Limitations
The relatively low proportion of female participants in our cohort (22%) could limit the statistical power for sex-specific analyses. Nonetheless, this underrepresentation reflects the real-world prevalence of ACS, in which females are less often affected and are typically diagnosed at an older age. Additionally, our study cohort consisted exclusively of Dutch ACS patients and thus, generalizability to populations with different ethnic compositions or healthcare systems is limited. Besides, we adjusted for key covariates such as age, BMI, time from index ACS, and kidney function; potential unmeasured confounders remain, for example, presentation type (STEMI vs. NSTEMI), time-to-treatment, and possible sex-specific referral or management biases. Our findings should be interpreted with caution, and further studies including larger numbers of females and more ethnically diverse populations are warranted to confirm our observations.
Conclusion
Our study comprehensively analyzes sex-specific differences in biomarker patterns during the acute and stabilized phases following an ACS. We found significant sex differences in Galectin-3, NT-proBNP, cTnT, and lipid profiles in patients after an ACS, but no notable differences in sST2, MPO, GDF-15, cystatin C, or TnI. Our findings suggest a complex interplay of biological factors in cardiovascular health post-ACS. Sex-specific reference thresholds for biomarkers like troponin and NT-proBNP could improve the diagnostic accuracy of recurrent ACS and risk stratification, particularly in females. Markers such as CRP and Galectin-3 reflect distinct inflammation and remodeling processes in females. Thus, different inflammation mediators could be potential targets for a more personalized treatment after an ACS.
Although many biomarkers showed comparable trends across sexes, the subtle differences emphasize the importance of more individualized approaches in clinical practice. However, as this is an observational and hypothesis-generating study, implementation of sex-specific diagnostic thresholds should be approached with caution and requires validation in independent cohorts and prospective interventional studies. Future research should aim to develop refined sex-specific diagnostic and prognostic tools and to elucidate the underlying pathomechanisms responsible for the observed sex differences. In addition, future studies should focus on prospective, sex-stratified biomarker validation, the integration of biomarker data into sex-specific risk scores, and the validation of their clinical utility in randomized controlled trials with adequate female representation. These directions will help clarify the translational relevance of our findings and guide the design of future studies in this important area.
Data availability
The dataset analysed during the current study is available from the corresponding author on reasonable request.
References
Haider, A. et al. Sex and gender in cardiovascular medicine: Presentation and outcomes of acute coronary syndrome. Eur. Heart J. 41(13), 1328–1336. https://doi.org/10.1093/eurheartj/ehz674 (2020).
Iorga, A. et al. The protective role of estrogen and estrogen receptors in cardiovascular disease and the controversial use of estrogen therapy. Biol. Sex Differ. 8, 1–16. https://doi.org/10.1186/s13293-017-0152-8 (2017).
Burke, A. P., Farb, A., Malcom, G. & Virmani, R. Effect of menopause on plaque morphologic characteristics in coronary atherosclerosis. Am. Heart J. 141(2 Suppl), S62. https://doi.org/10.1067/mhj.2001.109946 (2001).
Lansky, A. J. et al. Gender and the extent of coronary atherosclerosis, plaque composition, and clinical outcomes in acute coronary syndromes. JACC Cardiovasc. Imaging 5(3 Suppl), S72. https://doi.org/10.1016/j.jcmg.2012.02.003 (2012).
Pagidipati, N. J. & Peterson, E. D. Acute coronary syndromes in women and men. Nat. Rev. Cardiol. 13(8), 471–480. https://doi.org/10.1038/nrcardio.2016.89 (2016).
Westerman, S. & Wenger, N. K. Women and heart disease, the underrecognized burden: Sex differences, biases, and unmet clinical and research challenges. Clin. Sci. (London) 130(8), 551–563. https://doi.org/10.1042/CS20150586 (2016).
Oemrawsingh, R. M. et al. Cohort profile of BIOMArCS: The BIOMarker study to identify the Acute risk of a Coronary Syndrome—a prospective multicentre biomarker study conducted in the Netherlands. BMJ Open 6(12), e012929. https://doi.org/10.1136/bmjopen-2016-012929 (2016).
Chow, S. L. et al. Role of biomarkers for the prevention, assessment, and management of heart failure: A scientific statement from the American Heart Association. Circulation 135(22), e1054–e1091. https://doi.org/10.1161/CIR.0000000000000490 (2017).
Katsioupa, M. et al. Novel biomarkers and their role in the diagnosis and prognosis of acute coronary syndrome. Life 13(10), 1992. https://doi.org/10.3390/life13101992 (2023).
Gaertner, C. et al. Revisited upper reference limits for highly sensitive cardiac troponin T in relation to age, sex, and renal function. J. Clin. Med. 10(23), 5508. https://doi.org/10.3390/jcm10235508 (2021).
Gessner, R. et al. Higher troponin T serum concentrations in hospital patients without diagnosed cardiac diseases compared to a population-based cohort. Clin. Chem. Lab. Med. 61(11), 2046–2052. https://doi.org/10.1515/cclm-2023-0151 (2023).
Daniels, L. B. & Maisel, A. S. Cardiovascular biomarkers and sex: The case for women. Nat. Rev. Cardiol. 12(10), 588–596. https://doi.org/10.1038/nrcardio.2015.105 (2015).
Lew, J. et al. Sex-based differences in cardiometabolic biomarkers. Circulation 135(6), 544–555. https://doi.org/10.1161/CIRCULATIONAHA.116.024749 (2017).
Lau, E. S. et al. Sex differences in circulating biomarkers of cardiovascular disease. J. Am. Coll. Cardiol. 74(12), 1543–1553. https://doi.org/10.1016/j.jacc.2019.07.068 (2019).
Asrih, M. et al. Sex-specific modulation of circulating growth differentiation factor-15 in patients with type 2 diabetes and/or obesity. Endocr. Connect. 11(7), 643–653. https://doi.org/10.1530/EC-21-0273 (2021).
Eggers, K. M. et al. Temporal biomarker concentration patterns during the early course of acute coronary syndrome. Clin. Chem. Lab. Med. 62(6), 1167–1176. https://doi.org/10.1515/cclm-2024-0017 (2024).
Oemrawsingh, R. M. et al. High-frequency biomarker measurements of troponin, NT-proBNP, and C-reactive protein for prediction of new coronary events after acute coronary syndrome: BIOMArCS study. Circulation 139(1), 134–136. https://doi.org/10.1161/CIRCULATIONAHA.118.036349 (2019).
Boersma, E. et al. Details on high frequency blood collection, data analysis, available material and patient characteristics in BIOMArCS. Data Brief 27, 104750. https://doi.org/10.1016/j.dib.2019.104750 (2019).
R Core Team. R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing. (2024).
Pinheiro, J., Bates, D. & R Core Team. nlme: Linear and Nonlinear Mixed Effects Models [R package]. Version 3.1–166. CRAN.R-project.org/package=nlme (2024).
de Simone, G., Devereux, R. B., Daniels, S. R. & Meyer, R. A. Gender differences in left ventricular growth. Hypertension 26(6), 979–983. https://doi.org/10.1161/01.hyp.26.6.979 (1995).
De Lemos, J. A. et al. Association of troponin T detected with a highly sensitive assay and cardiac structure and mortality risk in the general population. JAMA 304(22), 2503–2512. https://doi.org/10.1001/jama.2010.1768 (2010).
Neeland, I. J. et al. Biomarkers of chronic cardiac injury and hemodynamic stress identify a malignant phenotype of left ventricular hypertrophy in the general population. J. Am. Coll. Cardiol. 61(2), 187–195. https://doi.org/10.1016/j.jacc.2012.10.012 (2013).
Jaffe, A. S. & Apple, F. S. High-sensitivity cardiac troponin assays: isn’t it time for equality?. Clin. Chem. 60(1), 7–9. https://doi.org/10.1373/clinchem.2013.217927 (2014).
Donaldson, C. et al. Estrogen attenuates left ventricular and cardiomyocyte hypertrophy via an estrogen receptor-dependent pathway that increases calcineurin degradation. Circ. Res. 104(2), 265–275. https://doi.org/10.1161/CIRCRESAHA.108.179903 (2009).
Piro, M., Della Bona, R., Abbate, A., Biasucci, L. M. & Crea, F. Sex-related differences in myocardial remodeling. J. Am. Coll. Cardiol. 55(11), 1057–1065. https://doi.org/10.1016/j.jacc.2009.09.065 (2010).
van den Berg, V. J. et al. Stabilization patterns and variability of hs-CRP, NT-proBNP, and ST2 during 1 year after acute coronary syndrome admission: Results of the BIOMArCS study. Clin. Chem. Lab. Med. 58(12), 2099–2106. https://doi.org/10.1515/cclm-2020-0415 (2020).
Eggers, K. M., Lagerqvist, B., Venge, P., Wallentin, L. & Lindahl, B. Persistent cardiac troponin I elevation in stabilized patients after an episode of acute coronary syndrome predicts long-term mortality. Circulation 116(17), 1907–1914. https://doi.org/10.1161/CIRCULATIONAHA.107.708529 (2007).
Park, K. C., Gaze, D. C., Collinson, P. O. & Marber, M. S. Cardiac troponins: from myocardial infarction to chronic disease. Cardiovasc. Res. 113(14), 1708–1718. https://doi.org/10.1093/cvr/cvx183 (2017).
Clerico, A. et al. Variability of cardiac troponin levels in normal subjects and in patients with cardiovascular diseases: Analytical considerations and clinical relevance. Clin. Chem. Lab. Med. 61(7), 1209–1229. https://doi.org/10.1515/cclm-2022-0510 (2023).
Suthahar, N. et al. Sex-specific associations of obesity and N-terminal pro-B-type natriuretic peptide levels in the general population. Eur. J. Heart Fail. 20(8), 1205–1214. https://doi.org/10.1002/ejhf.1161 (2018).
Redfield, M. M. et al. Plasma brain natriuretic peptide concentration: impact of age and gender. ACC Curr. J. Rev. 12(1), 44. https://doi.org/10.1016/s0735-1097(02)02059-4 (2003).
Maisel, A. S. et al. Rapid measurement of B-type natriuretic peptide in the emergency diagnosis of heart failure. N. Engl. J. Med. 347(3), 161–167. https://doi.org/10.1056/NEJMoa020233 (2002).
Troughton, R., Felker, G. M. & Januzzi, J. L. Jr. Natriuretic peptide-guided heart failure management. Eur. Heart J. 35(1), 16–24. https://doi.org/10.1093/eurheartj/eht463 (2014).
Biasucci, L. M. et al. Elevated levels of c-reactive protein at discharge in patients with unstable angina predict recurrent instability. Circulation 99(7), 855–860. https://doi.org/10.1161/01.CIR.99.7.85 (1999).
Imhof, A. et al. Distributions of C-reactive protein measured by high-sensitivity assays in apparently healthy men and women from different populations in Europe. Clin. Chem. 49(4), 669–672. https://doi.org/10.1373/49.4.669 (2003).
McConnell, J. P. et al. Gender differences in C-reactive protein concentrations-confirmation with two sensitive methods. Clin. Chem. Lab. Med. https://doi.org/10.1515/CCLM.2002.011 (2002).
Gupta, S., Gupta, V. K., Gupta, R., Arora, S. & Gupta, V. Relationship of high-sensitive C-reactive protein with cardiovascular risk factors, clinical presentation and angiographic profile in patients with acute coronary syndrome: An Indian perspective. Indian Heart J. https://doi.org/10.1016/j.ihj.2013.04.035 (2013).
Gijsberts, C. M. et al. Severity of stable coronary artery disease and its biomarkers differ between men and women undergoing angiography. Atherosclerosis https://doi.org/10.1016/j.atherosclerosis.2015.02.002 (2015).
Khera, A. et al. Race and gender differences in C-reactive protein levels. J. Am. Coll. Cardiol. https://doi.org/10.1016/j.jacc.2005.04.051 (2005).
Suthahar, N., Meems, L. M., Ho, J. E. & de Boer, R. A. Sex-related differences in contemporary biomarkers for heart failure: a review. Eur. J. Heart Fail. https://doi.org/10.1002/ejhf.1771 (2020).
Nayor, M. et al. Circulating galectin-3 is associated with cardiometabolic disease in the community. J. Am. Heart Assoc. https://doi.org/10.1161/JAHA.115.002347 (2015).
Pang, J. et al. Relationship of galectin-3 with obesity, IL-6, and CRP in women. J. Endocrinol. Invest. https://doi.org/10.1007/s40618-016-0490-3 (2016).
Catapano, A. L. et al. ESC/EAS guidelines for the management of dyslipidaemias. Eur. Heart J. https://doi.org/10.1093/eurheartj/ehw272 (2016).
Wang, X., Magkos, F. & Mittendorfer, B. Sex differences in lipid and lipoprotein metabolism: it’s not just about sex hormones. J. Clin. Endocrinol. Metab. https://doi.org/10.1210/jc.2010-2061 (2011).
Wang, Y. et al. Sex-specific prediction of cardiogenic shock after acute coronary syndromes: the SEX-SHOCK score. Eur. Heart J. https://doi.org/10.1093/eurheartj/ehae593 (2024).
Rubini Gimenez, M. et al. Sex-specific chest pain characteristics in the early diagnosis of acute myocardial infarction. JAMA Internal Med. https://doi.org/10.1001/jamainternmed.2013.12199 (2014).
Kraler, S. et al. Acute coronary syndromes: mechanisms, challenges, and new opportunities. Eur. Heart J. 46(29), 2866–2889. https://doi.org/10.1093/eurheartj/ehaf289 (2025).
Funding
The work was supported and funded by the Netherlands Heart Foundation (grant number 2007B012), the Netherlands Heart Institute-Interuniversity Cardiology Institute of the Netherlands (project number 071.01) and the Working Group on Cardiovascular Research Netherlands, all of which are non-commercial funding bodies. An unrestricted research grant was further obtained from Eli Lilly, the Netherlands.
Author information
Authors and Affiliations
Contributions
CG: Conceptualization, Formal Analysis, Methodology, Software, Visualisation, Writing—Original Draft Preparation, Writing—Review & Editing; MtH: Conceptualisation, Methodology, Writing—Original Draft Preparation, Writing—Review & Editing; KMA: Conceptualization, Data Curation, Funding Acquisition, Supervision , Writing—Review & Editing; RMO: Project Administration, Validation, Writing—Review & Editing; RHvS: Data Curation, Writing—Review & Editing; YBdR: Data Curation, Writing—Review & Editing; IK: Supervision, Validation, Writing—Review & Editing; YA: Conceptualization, Supervision, Validation, Writing—Review & Editing; EB: Conceptualization, Data Curation, Formal Analysis, Funding Acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Visualisation, Writing—Review & Editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
IK received travel reimbursement from Olink and SomaLogic. The other authors declare that they have no competing interests.
Ethics approval and consent to participate
BIOMArCS was conducted in accordance with the Declaration of Helsinki. The study protocol was approved by the institutional review board of the Erasmus MC (approval nr. MEC-2007-185), which decision was adopted by all other participating centers in accordance with Dutch law. All study participants provided written informed consent. BIOMArCS is registered in The Netherlands Trial Register under the unique identifiers NTR1106 and NTR1698.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gärtner, C., Haaf, M.E.t., Akkerhuis, K.M. et al. Sex-based differences in biomarker trajectories in acute coronary syndrome patients from the BIOMArCS study. Sci Rep 16, 1585 (2026). https://doi.org/10.1038/s41598-025-30969-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-30969-4