Abstract
This study evaluated the effects of the entomopathogenic fungi on the feeding behavior of the Neotropical brown stink bug, Euschistus heros, using electropenetrography (AC-DC) technology. Twenty females per treatment were reared under controlled conditions and exposed to soybean pods treated with fungal suspensions (2 µL on the pronotum with 5 × 106 and 6.15 × 108 conidia mL− 1 for Cordyceps javanica and Metarhizium anisopliae, respectively), chemical insecticide (Thiamethoxam + Lambda-cyhalothrin, 0.025/100 mL), and aqueous solution of Polysorbate 80 (0.01% v/v) (control). Response variables associated with count and duration of feeding behavior over 72 h of recording were modelled using GAMLSS (generalized additive models for location, scale, and shape) to assess the statistical significance of treatments and for pairwise comparisons of means (p < 0.05). The fungal treatments and chemical insecticide significantly reduced the frequency and duration of feeding events, especially stylet penetration and seed ingestion, compared to untreated controls. Additionally, insects treated with fungi spent more time in non-feeding behavior, indicating disrupted feeding behavior. Both fungi also shortened the duration of the final feeding probe by about 13 h. Furthermore, electropenetrography enables assessment of pest–plant interactions and biocontrol efficacy beyond mortality.
Similar content being viewed by others
Introduction
The Neotropical brown stink bug (NBSB), Euschistus heros (Fabricius, 1798) (Hemiptera: Pentatomidae), is an important pest in South America, causing serious damage to crops, such as soybeans [Glycine max (L.) Merr.] and maize (Zea mays L.)1. Egg-adult development can vary from 25 to 32 days (5 to 7 days in the egg stage and 20 to 25 days in the nymphal stage), and the adult phase can vary from 75 to 116 days1.
E. heros begins the infestation of soybean plants in the vegetative phase and causes damage in the reproductive phase as a pod feeder. Their feeding can have direct effects on yield and/or other parameters related to grain quality during pod development and seed filling, when the stylet penetrates the plant tissues, injects saliva, and ruptures cells2,3,4,5. Furthermore, the punctures left by the stylets can facilitate infection by pathogenic microorganisms6.
The most common method used to control the NBSB is chemical application of insecticides7. However, using only insecticides to control E. heros can lead to undesirable effects on the environment, human health, and non-target organisms, as well as favoring the selection of insecticide-resistant insects8,9. Therefore, incorporating alternative control methods compatible with Integrated Pest Management (IPM) is necessary. Biological control stands out as a key strategy for pest control as a practical and ecologically friendly tactic10.
Entomopathogenic fungi are a diverse group of organisms that have garnered significant attention for their potential in biological pest control. They have been extensively studied for their ability to infect and kill a wide range of insect species11. The fungal species Metarhizium anisopliae (Metsch.) Sorok., Beauveria bassiana (Bals.) Vuill., and Cordyceps javanica (Frieder. & Bally) have been widely applied to control E. heros12,13. The mode of action, efficacy, and virulence of the species have been studied to control E. heros14. Recently, a study verified the probing behavior of E. heros treated with M. anisopliae, demonstrating that adult insects treated with M. anisopliae exhibited an 86% reduction in probing behavior by the fourth day after exposure, and probing activity ceased entirely by the fifth day (p < 0.05)15. This behavioral disruption, monitored via electropenetrography (EPG), led to a measurable decrease in seed damage even before insect mortality occurred15.
EPG has emerged as a powerful tool to study piercing-sucking insects, offering real-time insights into their probing and non-probing activities. The insect and plant are part of the same electrical circuit to record voltage changes that represent otherwise invisible stylet penetration behaviors inside the plant, and the system generates waveforms on a computer screen15,16. This technology may be useful in the applied field of NBSB management, to evaluate, for example, the selection of insect-resistant plants and resistance inducers17, the action of insecticides2, and recently, the effect of entomopathogenic fungi18.
Evaluation of the probing behavior of Dalbulus maidis (DeLong &Wolcott) (Hemiptera: Cicadellidae) on corn, after spraying of Cordyceps javanica, indicates a significant disruption of the probing activities in 48 h19. This reduction in insect feeding activities, reported by Almeida18 and Maluta19, is important information for managing pests in the field, as it shows that, even if the insect is still alive, its feeding is compromised, and, consequently, the damage caused to the crop is mitigated. Therefore, the slower lethal effect of entomopathogenic fungi compared to synthetic insecticides can be minimized by changes in the pest’s probing behavior6.
The feeding behavior of E. heros treated with different fungal species has not been verified. We hypothesized that NBSB treated with different fungal species could exhibit different probing behaviors. Therefore, we conducted this study with the aim of ascertaining whether different fungal species can alter the probing behavior of E. heros feeding on soybean pods.
Materials and methods
Rearing of Euschistus heros
E. heros was maintained at the Integrated Pest Management Laboratory of the Instituto Federal Goiano, Urutaí, Goiás, Brazil, under controlled environmental conditions (25 ± 2 °C, relative humidity of 60 ± 10%, and a photoperiod of 14 h). Adults were housed in plastic containers (25 × 20 × 10 cm) lined with filter paper to provide a clean and stable environment. Their diet consisted of green bean pods (Phaseolus vulgaris L.), soybean seeds (Glycine max L.), and raw peanut kernels (Arachis hypogaea L.), which were replenished every 48 h. Containers were cleaned regularly to ensure hygienic rearing conditions.
A voile fabric was used as the oviposition substrate, where eggs were collected and transferred to plastic containers (Gerbox 11 × 11 × 3.5 cm) with moistened soybean pods and filter paper to maintain optimal humidity. Early instar nymphs (first and second) were fed exclusively on soybean pods, whereas from the third instar onward, their diet was expanded to include the same food sources provided to adult insects. When the nymphs reached the fifth instar, they were relocated to plastic containers to continue their development.
Soybean cultivation and plant preparation for EPG analysis
Soybean plants (cv. BRS 7470 IPRO) were cultivated in pots containing a soil-to-sand mixture at a 2:1 ratio. Seeds of a commercial cultivar were sown in 18-liter plastic pots and grown in a greenhouse (27 ± 10 °C, relative humidity of 60 ± 20%, and a photoperiod of 13 h), preventing pest infestation without the need for insecticide applications. Fertilization was performed at sowing (3 g per pot of monoammonium phosphate – MAP) and later supplemented with 5 g of urea + 5 g of potassium chloride per pot. To ensure a continuous supply of plants for the EPG trial, seeds were sown weekly in batches of five pots until a total of 75 pots was reached. Irrigation was carried out as required to support proper plant growth.
Once the plants reached the R5.5 (pod filling) reproductive stage, stem sections containing pods were excised and transferred to 0.2-liter pots with moistened soil. These plant sections were used for monitoring E. heros probing activities during the EPG recordings.
Suspension preparation
To prepare the fungal suspensions, commercial products and commercial doses were used. First, 0.65 g of Cordyceps javanica (isolate BRM 27666) and 8.13 g of Metarhizium anisopliae (isolate IBCB 425) were weighed and then mixed into 900 mL of distilled water, followed by the addition of 100 mL of an aqueous Polysorbate 80 (Tween 80) solution (99.9 mL of distilled water + 0.1 mL of Tween 80–0.01% v/v). The mixture was homogenized in a 1-L beaker to ensure even distribution of fungal propagules. The final suspension concentrations were 5 × 106 and 6.15 × 108 conidia mL− 1 for C. javanica and M. anisopliae, respectively. The conidial concentrations were enumerated by hemocytometer (Brightline Improved Neubauer, New OptikR, São Paulo, SP, Brazil) at 400× magnification. Conidial germination was verified (exceeding 98%) on potato dextrose agar (PDA) after 18 h at 26 °C. Only conidia with germ tubes greater than the conidial diameter were considered germinated.
For the chemical insecticide treatment, Thiamethoxam + Lambda-cyhalothrin was prepared by diluting 0.025 mL of the product in 100 mL of water, using a 100 mL volumetric flask for precise measurement and standardization of the solution. This dose corresponds to a quarter of the commercial dose of the product20, which was adjusted to cause only a sublethal effect on the insect and not kill the NBSB during the recording.
Electropenetrography experiment
The experiment was conducted in a controlled environment of 25 ± 2 °C, 70 ± 10% relative humidity, and a 14 h photoperiod. Adult female NBSBs were used because they exhibit a longer feeding period. They (10–12 days after the emergence of adults) were subjected to four different treatments: fungal suspensions (C. javanica and M. anisopliae), the chemical insecticide, and a control group consisting of an aqueous solution of Polysorbate 80 (0.01% v/v)15. NBSBs were topically treated (2 µL) on the pronotum. The insect activities on plant sections were assessed 4 h post-treatment using an AC-DC EPG monitor (EPG Technologies, Inc., Gainesville, FL, USA). The study was structured as a randomized block design with four treatments and twenty replicates per treatment. Each replicate consisted of a single insect, and the experiment was temporally blocked to account for variations over time.
Before the start of the recordings, insects underwent a 4-h fasting period to stimulate insect probing during the recording. They were then immobilized for gold wire attachment following the methodology proposed by Lucini and Panizzi21, using a thin strip of tape for stability. The pronotum was gently abraded with dental sandpaper to enhance adhesion of the silver glue [made with silver flake (Sigma Aldrich, St. Louis, MO, USA), white glue (Cascorez, Jundiai, SP, Brazil) and water (1:1:1 wt/vol/vol)], which was used to attach a gold wire (0.127 mm in diameter and 3.5 cm in length; Sigma Aldrich, Barueri, SP, Brazil) to a copper wire (3 cm in length) soldered to a brass nail. A small loop was made at the other end of the gold wire to increase the contact area with the insect and improve electrical conductivity22.
After the glue dried, the insects were connected to the EPG monitor, where their activities were recorded for 72 h. Twenty adults were successfully recorded (recordings without any insect probing were not used) per treatment in a completely randomized design.
All recordings were performed inside a Faraday cage to eliminate external electrical noise. Recordings were conducted using an input impedance of 107 Ohms for each of the four channels of the EPG monitor, with a voltage of 50 mV in alternating current (AC) across the plant electrode, and a gain set to 400x (actual gain of 400x with a 1x multiplier)23. An offset control was used to prevent rectifier folding and to preserve the native waveform polarity after rectification. Output voltages were amplified and captured at a sampling rate of 100 Hz per channel using the WinDaq DI-710 system (Dataq Instruments, Akron, OH, USA) connected to a computer running WinDaq Lite software version 3.11 (Dataq Instruments, Akron, OH, USA).
At the end of each recording, the NBSB treated with the fungi were separated to confirm mortality due to fungal infection. NBSB were transferred to Petri dishes (60 mm) with wet cotton and maintained at room temperature. Insects were considered infected by the fungus when mycelial and conidial growth was observed on the insect cadaver. Recordings of insects that did not exhibit fungal infection were excluded from the analysis.
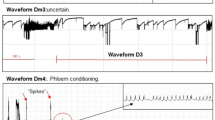
The waveform events were identified and named according to a waveform library described in the literature23: Z (non-probing), Eh1 (stylet penetration), Eh2 (xylem sap ingestion), Eh3a (cellular laceration and enzymatic maceration of seed endosperm), Eh3b (short ingestion of macerated tissues of seed endosperm), Eh4 (ingestion from unknown location – probably phloem sap), without identifying treatments to reduce any bias. Three variables were calculated to study the feeding behavior: NWEi (Number of Waveform Events per Insect), WDEi (Waveform Duration per Event per Insect), and Final Time of the Last Probe (FTLP).
Data analysis
The raw data generated by the EPG monitor was processed and analyzed using the INFEST v.2.0 software24 based on R v.4.2.1 and the gamlss package v.5.4–12. Statistical modeling was based on GAMLSS – Generalized Additive Linear Models for Location, Scale, and Shape25,26, testing fourteen distribution models for each count and duration response variable. The selection of the best-fitting model and probability distribution was based on the Akaike Information Criterion (AIC). The standardized Pearson’s residuals were graphically examined through a worm plot to check for fitting problems. The significance of the treatment effect was verified by the likelihood ratio test. Estimated marginal means were estimated for group comparisons, and pairwise tests were conducted by applying Tukey’s procedure at 5% significance.
Results
The groups presented significant (p < 0.05) differences in the means of the Number of Waveform Events per Insect (NWEi), Waveform Duration per Event per Insect (WDEi), and Final Time of the Last Probe (FTLP) as shown in Table 1. For the non-probing (Z) waveform, insects in the Control group exhibited an NWEi 3 times higher than the sublethal doses of Chemical treatment. C. javanica and M. anisopliae presented similar and intermediate values. However, the WDEi of Z was 3 times higher with the Chemical treatment than the untreated, indicating less time spent on probing activities. A similar result was observed for the NWEi associated with stylet penetration (Eh1). On average, untreated insects performed about 50 more probing events of stylet penetration than chemically treated insects. C. javanica promoted the lowest duration of Eh2 (xylem sap ingestion) and Eh3b (short ingestion of macerated tissues of seed endosperm), almost 4 times lower (p < 0.05) than the control group. However, no statistical differences were found for WDEi in Eh1, and for NWEi and WDEi in Eh3a (seed disruption), Eh3b (ingestion from seeds), as well as for NWEi in Eh3b.
The FTLP varied significantly among treatments. Insects in the Control group exhibited the highest average final probing time (about 44 h), whereas those treated with C. javanica or M. anisopliae (around 31 h) and the Chemical treatment (about 39 h) displayed markedly shorter final probing times. These findings indicate that entomopathogenic fungi reduced the overall duration of feeding.
Values are presented as mean ± standard error. Means followed by the same letter within a column are not significantly different, according to Tukey’s test at 5% significance. Waveforms: Z (non-probing), Eh1 (stylet penetration), Eh2 (xylem sap ingestion), Eh3a (seed disruption), Eh3b (ingestion from seeds), Eh4 (ingestion from unknown location – probably phloem), FTLP (The final time of the last probe).
The overall proportions of time spent probing versus non-probing waveforms are shown in Fig. 1. In the Control group, insects spent 63% of the recording time probing. However, the fungal treatments shifted this balance considerably, as C. javanica promoted 72% of non-probing time and M. anisopliae 69% of non-probing. Chemical treatment induced the most pronounced shift, with non-probing behavior accounting for 81% of the recording time. These alterations suggest that while the treated groups reduced active feeding, the entomopathogenic fungi distinctly modified the probing pattern, potentially lowering feeding damage.
Percentage of time spent in probing vs. non-probing by E. heros feeding on soybean pods under four treatments (Control, C. javanica, M. anisopliae, Chemical). Values are descriptive proportions computed from the total recorded time per treatment (aggregated data); therefore, error bars/boxplots are not shown. The number of insects (n) and total recording time (T) for each treatment: Control (n = 20; T = 72 h), C. javanica (n = 20; T = 72 h), M. anisopliae (n = 20; T = 72 h), and Chemical (n = 20; T = 72 h). Probing denotes periods with stylet penetration/feeding waveforms; non-probing accounts for all remaining time. Statistical analyses based on per-insect data are presented in Table 1.
Discussion
The present study elucidates the effects of two entomopathogenic fungi, Cordyceps javanica and Metarhizium anisopliae, on the probing behavior of Euschistus heros. Our 72 h observations capture the early infection window of entomopathogenic fungi, when conidial adhesion/germination is followed by cuticle penetration and onset of hemocoel colonization, during which sublethal, behavior-modifying effects can emerge (toxins and enzymes, such as destruxins, proteases, and chitinases, interacting with host immunity)27.
Within this period, the detected reduced probing and delayed feeding initiation are consistent with comparative reports of feeding suppression in insects exposed to entomopathogenic fungi and with the recognized suitability of EPG for resolving stylet activities and linking waveform metrics to injury and management endpoints in E. hero2,21.
The main finding was the significant reduction by fungal treatments in both the Number of Waveform Events per Insect (NWEi) and the Waveform Duration per Event per Insect (WDEi) for critical feeding behaviors, particularly stylet penetration (Eh1), xylem sap ingestion (Eh2), and seed ingestion (Eh3b). These behavioral disruptions suggest that entomopathogenic fungi impair the capacity of E. heros to initiate and maintain effective feeding. One hypothesis for these findings is that fungal infection compromises neuromuscular coordination, possibly through the production of mycotoxins such as destruxins (associated with Metarhizium spp.) or cordycepin (produced by Cordyceps spp.), which have been shown to interfere with insect physiological functions28. These compounds may reduce responsiveness or cause partial paralysis, leading to reduced probing frequency and duration29, which limits plant damage during the pod-filling (R5) stage of soybean.
Another plausible explanation is that the energy costs associated with mounting an immune response to fungal infection divert resources away from feeding activities30. Hemipteran insects, including E. heros, exhibit innate immune responses, such as melanization and antimicrobial peptide production31. Activation of these defenses is energetically costly and may lead to behavioral trade-offs, including reduced locomotion and feeding32. This phenomenon may account for the increased non-probing (Z waveform) durations observed in fungi-treated insects, as they may remain stationary or inactive to conserve energy during systemic fungal infection33.
Interestingly, while the sub-dose of chemical treatment (Thiamethoxam + Lambda-cyhalothrin) also resulted in significant increases in non-probing time, its profile was distinct. Insects exposed to the sub-dose of the chemical insecticide exhibited the highest average WDEi for the Z waveform, suggesting that while the chemical effectively induced feeding cessation, the effect could be immediate but transient. Conversely, the fungal treatments appeared to exert more gradual but sustained sublethal effects, which were reflected in the probing behavior throughout the recording time (72 h). This temporal differentiation in mode of action highlights the potential complementary roles of chemical and biological control agents within Integrated Pest Management (IPM)34,35.
The reduction in the final time of the last probe duration among the experimental groups indicates a tangible decrease in potential plant damage during the R5.5 soybean growth stage, a critical period for yield formation. Previous studies have demonstrated that feeding by E. heros during this stage leads to significant reductions in seed quality and weight36,37. The Final Time of the Last Probe (FTLP) for fungal treatments was ~ 31 h, which is biologically consistent with the infection timeline of entomopathogenic fungi. After conidial adhesion and germination (within hours), Metarhizium and Cordyceps typically penetrate the cuticle within ~ 24–48 h and begin hemocoel colonization32. During this early window, sublethal effects, e.g., destruxin-mediated immune and neuromuscular disruption as well as infection-induced metabolic stress, can reduce feeding propensity and delay the first successful stylet insertion detected by EPG38.
A 45% reduction in FTLP of E. heros treated with M. anisopliae compared to untreated insects is reported by some authors18, who state that this information is valuable for managing NBSB in the field. It shows that, even if the insect is still alive, its feeding is compromised and, consequently, the damage caused to soybean seeds is reduced. By shortening the active feeding period, entomopathogenic fungi may mitigate yield losses even in the absence of high mortality rates38. Thus, EPG data support the hypothesis that sublethal effects can substantially contribute to pest suppression, a concept increasingly recognized in biocontrol research39.
Moreover, the altered proportion of probing versus non-probing behavior in fungi-treated insects suggests a shift in behavioral priorities, possibly due to discomfort or physiological impairment. The increase in non-probing time could also reflect aversive learning or behavioral avoidance if the insects associate the feeding process with adverse physiological effects post-infection19. Though traditionally considered non-cognitive, some Hemiptera exhibit behavioral plasticity, including changes in host preference or feeding site selection following sublethal exposure to stressors40. Whether E. heros possesses such behavioral adaptability warrants further investigation, but it may be relevant in understanding long-term pest population dynamics under sustained fungal pressure.
Due to their specificity and environmental compatibility, entomopathogenic fungi offer a valuable alternative or supplement to chemical control, especially in the face of rising resistance issues and regulatory constraints on synthetic pesticides41,42. Furthermore, the compatibility of fungal treatments with other biocontrol agents, such as parasitoids or predators, presents opportunities for synergistic interactions that enhance overall pest management43.
Finally, this study highlights the value of EPG as a diagnostic tool in entomological research. By capturing real-time, fine-scale feeding behaviors, EPG allows the detection of subtle but meaningful impacts of pest management interventions. EPG data can refine our understanding of pest–host interactions and enlighten the development of more effective, targeted control strategies44.
In conclusion, the application of C. javanica and M. anisopliae significantly disrupts the probing behavior of E. heros feeding on soybean pods, reducing its potential for crop damage. The present findings support the incorporation of entomopathogenic fungi into IPM programs, with the added benefit of mitigating the drawbacks associated with exclusive chemical control. EPG may be a promising tool to evaluate the feeding behavior of NBSB treated with entomopathogenic fungi, helping in the selection of different fungal species or isolates, showing whether certain isolates inhibit insect feeding, verifying whether different concentrations of fungi affect the insect probing behavior, and strengthening biological control programs.
However, insects infected with sublethal does may resume feeding depending on the fungal isolate, dose, and abiotic conditions (notably temperature and humidity). Thus, the persistence of feeding suppression is context-dependent and warrants longer observation (≥ 7–10 d) under field-realistic conditions to determine whether effects are transient or sustained45. Within this framework, the short-term attenuation documented during reproductive stages (e.g., soybean R5.5) is agronomically meaningful because reduced stylet activity can lower injury to productive structures even before insect mortality occurs.
Data availability
The raw datasets generated during the current study are available in the Zenodo repository at: [https://doi.org/10.5281/zenodo.16572536](https:/doi.org/10.5281/zenodo.16572536).
References
Sosa-Gómez, D. R. et al. Prevalence, damage, management and insecticide resistance of stink bug populations (Hemiptera: Pentatomidae) in commodity crops. Agric. Entomol. 22, 99–118 (2020).
Lucini, T. & Panizzi, A. R. Electropenetrography (EPG): a breakthrough tool unveiling stink bug (Pentatomidae) feeding on plants. Neotropic Entomol. 47, 6–18 (2018).
Hickmann, F., Cordeiro, E. M. G., Aurelio, M. S. L., Saldanha, A. V. & Corrêa, A. S. Host preference and survivorship of Euschistus Heros strains on cotton and soybean. Entomol. Exp. Appl. 171, 564–571 (2023).
Moscardi, F. et al. Soja Manejo Integrado De Insetos E Outros Artrópodes-praga in 213–334 (Embrapa Soja, 2012).
Silva, R. A. et al. Temporal variation and Spatial distribution of Euschistus Heros (Hemiptera: Pentatomidae) during the soybean grain formation period. Res. Soc. Dev. 11, e6411931102 (2022).
Tessmer, M. A. et al. Histology of damage caused by Euschistus Heros (F.) nymphs in soybean pods and seeds. Neotrop. Entomol. 51, 112–121 (2022).
Ecco, M. et al. Stink bug control at different stages of soybean development. Arq. Inst. Biol. 87, e1132018 (2020).
Woodcock, B. A. et al. Country-specific effects of neonicotinoid pesticides on honey bees and wild bees. Science 356, 1393–1395 (2017).
Tibola, C. M., Silva, L., Sgubin, F. & Omoto, C. Monitoring resistance of Euschistus Heros (Fabricius) (Hemiptera: Pentatomidae) to insecticides by using encapsulated artificial diet bioassay. Insects 12, 559 (2021).
van Lenteren, J. C., Bolckmans, K., Köhl, J., Ravensberg, W. J. & Urbaneja, A. Biological control using invertebrates and microorganisms: plenty of new opportunities. BioControl 63, 39–59 (2018).
Gielen, R., Meister, H., Tammaru, T. & Põldmaa, K. Fungi recorded on folivorous lepidoptera: high diversity despite moderate prevalence. J. Fungi. 7, 25 (2021).
Alfina, T. & Haneda, N. F. Entomopathogenic fungi as biological agents in forest plant pest control: A systematic review. IOP Conf. Ser. Earth Environ. Sci. 959 https://doi.org/10.1088/1755-1315/959/1/012013 (2022).
Jiang, W. et al. Effects of the entomopathogenic fungus Metarhizium anisopliae on the mortality and immune response of Locusta migratoria. Insects 11, 36 (2020).
Resquín-Romero, G. et al. Virulence of Metarhizium brunneum (Ascomycota: Hypocreales) strains against stink Bugs Euschistus Heros and Dichelops furcatus (Hemiptera: Pentatomidae). J. Econ. Entomol. 113, 2540–2545 (2020).
Walker, G. P., Fereres, A. & Tjallingii, W. F. Guidelines for conducting, analyzing, and interpreting electrical penetration graph (EPG) experiments on herbivorous piercing–sucking insects. Entomol. Exp. Appl. 172, 564–580 (2024).
Backus, E. A., Cervantes, F. A., Guedes, R. N. C., Li, A. Y. & Wayadande, A. C. AC–DC electropenetrography for in-depth studies of feeding and oviposition behaviors. Ann. Entomol. Soc. Am. 112, 236–248 (2019).
Coutinho, W. B. G., Silva, F. C., Barrigossi, J. A. F., Almeida, A. C. S. & Jesus, F. G. Silicon applications in rice plants alter the stylet probing behaviors of Glyphepomis spinosa (Hemiptera: Pentatomidae). J. Insect Sci. 24, 23 (2024).
de Almeida, A. C. Can Metarhizium anisopliae reduce the feeding of the Neotropical brown stink bug, Euschistus Heros (Fabricius, 1798), and its damage to soybean seeds? J. Fungi. 11, 247 (2025).
Maluta, N., Castro, T. & Spotti Lopes, J. R. DC-electrical penetration graph waveforms for Dalbulus Maidis (Hemiptera: Cicadellidae) and the effects of entomopathogenic fungi on its probing behavior. Sci. Rep. 13, 22033 (2023).
Ministério da Agricultura e Pecuária (MAPA). AGROFIT — Sistema de Agrotóxicos Fitossanitários. Ministério da Agricultura e Pecuária (2025). https://agrofit.agricultura.gov.br/agrofit_cons/principal_agrofit_cons
Lucini, T. & Panizzi, A. R. Waveform characterization of the soybean stem feeder Edessa meditabunda: overcoming the challenge of wiring pentatomids for EPG. Entomol. Exp. Appl. 158, 118–132 (2016).
Cervantes, F. A. et al. Correlation of electropenetrography waveforms from Lygus lineolaris (Hemiptera: Miridae) feeding on cotton squares with chemical evidence of inducible tannins. J. Econ. Entomol. 110, 2068–2075 (2017).
Lucini, T. & Panizzi, A. R. Electropenetrography monitoring of the Neotropical brown-stink bug (Hemiptera: Pentatomidae) on soybean pods: an electrical penetration Graph-histology analysis. J. Insect Sci. 18, 5 (2018).
Silva, A. R., Almeida, A. C. S., Jesus, F. G. & Barrigossi, J. A. F. INFEST: an R web application to perform statistical analysis of insect feeding behavior data from EPG systems. J. Econ. Entomol. https://doi.org/10.1093/jee/toaf232 (2025).
Rigby, R. A. & Stasinopoulos, D. M. Generalized additive models for location, scale and shape. Appl. Statist. 54, 507–554 (2005).
Oliveira, G. P. et al. Alternatives for simulating and modeling simplified insect feeding eletropenetrography discrete data. Acta Sci. Agron. 47, e71591 (2025).
Arias-Chavarría, L. D. et al. Evaluation of the viability of microencapsulated Trichoderma longibrachiatum conidia as a strategy to prolong the shelf life of the fungus as a biological control agent. Front. Chem. 15, 12:1473217 (2025).
Barelli, L., Behie, S. W., Hu, S. & Bidochka, M. J. Profiling Destruxin synthesis by specialist and generalist Metarhizium insect pathogens during coculture with plants. Appl. Environ. Microbiol. 88, e0247421 (2022).
Ardia, D. R., Gantz, J. E., Schneider, B. C. & Strebel, S. Costs of immunity in insects: an induced immune response increases metabolic rate and decreases antimicrobial activity. Funct. Ecol. 26, 732–739 (2012).
Gielen, R. et al. Entomopathogenic fungi as mortality agents in insect populations: A review. Ecol. Evolut. 14, e70666 (2024).
Singh, K. S. et al. The genome sequence of the Neotropical brown stink bug, Euschistus Heros provides insights into population structure, demographic history and signatures of adaptation. Insect Biochem. Mol. Biol. 152, 103890 (2023).
Miyashita, A., Lee, T. Y. M., McMillan, L. E., Easy, R. & Adamo, S. A. Immunity for nothing and the eggs for free: apparent lack of both physiological trade-offs and terminal reproductive investment in female crickets (Gryllus texensis). PLoS ONE 14, e0209957 (2019).
Zhang, D., Qi, H. & Zhang, F. Parasitism by entomopathogenic fungi and insect host defense strategies. Microorganisms 13, 283 (2025).
Zambrano, N. D., Arteaga, W., Velasquez, J. & Chirinos, D. T. Side effects of lambda cyhalothrin and Thiamethoxam on insect pests and natural enemies associated with cotton. Sarhad J. Agric. 7, 1098–1106 (2021).
Wu, S., Youngman, R. R., Kok, L. T. & Laub, C. A. Sublethal effect of Beauveria Bassiana and Metarhizium brunneum (Hypocreales: Clavicipitaceae) on Cyclocephala lurida (Coleoptera: Scarabaeidae). J. Entomol. Sci. 51, 43–53 (2016).
Liu, D., Smagghe, G. & Liu, T. X. Interactions between entomopathogenic fungi and insects and prospects with glycans. J. Fungi. 9, 575 (2023).
Sánchez-Pérez, L., de Barranco-Florido, C., Rodríguez-Navarro, J. E. & Cervantes-Mayagoitia, S. Ramos-López, M. Á. Enzymes of entomopathogenic fungi, advances and insights. Adv. Enzyme Res. 2, 65–76 (2014).
Wang, H., Peng, H., Li, W., Cheng, P. & Gong, M. The toxins of Beauveria Bassiana and the strategies to improve their virulence to insects. Front. Microbiol. 12, 705343 (2021).
Qu, S. L., Li, S. S., Li, D. & Zhao, P. J. Metabolites and their bioactivities from the genus Cordyceps. Microorganisms 10, 1489 (2022).
Smid, H. M. & Vet, L. E. M. The complexity of learning, memory and neural processes in an evolutionary ecological context. Curr. Opin. Insect Sci. 15, 61–69 (2016).
Nanzer, S. L. L. et al. Assessment of entomopathogenic nematodes and their symbiotic bacteria to control the stink Bugs Euschistus Heros and Dichelops Melacanthus (Heteroptera: Pentatomidae) in the soybean-corn succession system. Turk. J. Zool. 45, 356–371 (2021).
Battisti, L. et al. Selectivity of Metarhizium anisopliae and Beauveria Bassiana to adults of Telenomus Podisi (Hymenoptera: Scelionidae). Semin Cienc. Agrar. 43, 727–738 (2022).
Bava, R. et al. Entomopathogenic fungi for pests and predators control in beekeeping. Vet. Sci. 9, 95 (2022).
Ebert, T. A., Backus, E. A. & Rogers, M. E. Handling artificially terminated events in electropenetrography data. J. Econ. Entomol. 111, 1987–1990 (2018).
Athanassiou, C. G., Kavallieratos, N. G., Rumbos, C. I. & Kontodimas, D. C. Influence of temperature and relative humidity on the insecticidal efficacy of Metarhizium anisopliae against larvae of Ephestia Kuehniella (Lepidoptera: Pyralidae) on wheat. J. Insect Sci. 17, 22 (2017).
Acknowledgements
The authors gratefully acknowledge the financial support provided by the National Council for Scientific and Technological Development (CNPq), [grant number: 309733/2021-9] and the Fundação de Amparo à Pesquisa do Estado de Goiás (FAPEG), [grant number: 1721]. We also thank the Instituto Federal Goiano and the Center of Excellence in Bioinputs (CEBIO) for their institutional support throughout this study.
Funding
This research was supported by the National Council for Scientific and Technological Development–CNPq [grant number: 309733/2021-9] and the Fundação de Amparo à Pesquisa do Estado de Goiás–Fapeg [grant number: 1721]. The research also received institutional support from Instituto Federal Goiano and the Center of Excellence in Bioinputs (CEBIO).
Author information
Authors and Affiliations
Contributions
Mr. Guilherme Pereira Oliveira*, ORCID: 0000-0001-7440-6773. Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Software; Supervision; Validation; Visualization; Writing – original draft; Writing – review & editing. *Corresponding Author. Professor Frederico Antonio Loureiro Soares, ORCID: 0000-0002-4152-5087. Conceptualization; Formal analysis; Investigation; Methodology; Project administration; Resources; Supervision; Validation; Writing – review & editing. Professor André Cirilo de Sousa Almeida, ORCID: 0000-0001-9786-2990. Conceptualization; Data curation; Formal analysis; Methodology; Resources; Software; Supervision; Validation; Writing – review & editing. Ms. Kaylaine Aparecida Gomes de Souza, ORCID: 0009-0008-6124-4427. Data curation; Formal analysis; Investigation; Methodology; Software; Validation; Visualization. Professor Pablo da Costa Gontijo, ORCID: 0000-0001-8173-0539. Conceptualization; Data curation; Investigation; Methodology; Resources; Software; Writing – review & editing. Professor Anderson Rodrigo da Silva, ORCID: 0000-0003-2518-542X. Conceptualization; Data curation; Formal analysis; Project administration; Resources; Supervision; Writing – review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
de Oliveira, G.P., Soares, F.A.L., Sousa Almeida, A.C.d. et al. Entomopathogenic fungi disrupt the feeding behavior of Euschistus heros in soybean. Sci Rep 16, 1261 (2026). https://doi.org/10.1038/s41598-025-31096-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-31096-w