Abstract
Wild edible fruits are rich in micronutrients and serve as an essential source of nutrition for the poor in developing countries, where malnutrition is widespread. The morphological and nutritional compositions of Bauhinia thonningii pod and seed were evaluated using samples collected from two distinct agroecological zones, the warm moist lowlands (WMLL) and tepid sub-moist mid-highlands (TSMMHL), in the Tselemti district, Ethiopia. Data were analyzed using independent sample t-test, ANOVA with a general linear model, and Principal component analysis (PCA) for morphological traits, proximate composition, and mineral content to determine their association with agroecological zones. The results showed that morphological traits such as the mean pod length (p < 0.000), pod width (p = 0.036), pod thickness (p = 0.005), pericarp weight (p = 0.006), total seed weight (p = 0.003), individual seed weight (p = 0.005), and number of seeds per pod (p < 0.000) differed significantly between the two agroecological zones (p < 0.05). Higher mean values of pod length (18.34 cm), width (2.87 cm), thickness (0.85 cm), total pod weight (11.56 g), total seed weight (8.93 g), and number of seeds per pod (51 ns) were recorded in the warm moist lowlands compared to the tepid sub-moist mid-highlands. The moisture content of the B. thonningii pod (9.02%) and seed (7.02%) was higher in tepid sub-moist mid-highlands than in the warm moist lowlands. The crude protein (9.74 and 30.73%), crude fat (0.96 and 2.48%), crude fiber (26.03 and 32.86%), total carbohydrates (56.30 and 33.70%), and energy values (1106.36 and 1103.87 kJ/100 g) of the pod and seed, respectively, were higher in the WMLL compared to the TSMMHL. All proximate compositions of the B. thonningii pod and seed varied significantly between the two agroecological zones (p < 0.05), except for ash content. Most mineral concentrations in the pod and seeds, such as calcium (152.26 and 36.77 mg/100 g), magnesium (129.59 and 8.04 mg/100 g), potassium (1325.44 and 130.61 mg/100 g), and sodium (8.99 and 16.90 mg/100 g), were significantly higher in the warm moist lowland agroecology. This may be attributed to higher humidity, soil mineralization, evaporative concentration, and increased soil nutrient movement under warm lowland environmental conditions. Significant differences were observed in the concentrations of all minerals in the pods and seeds between the agroecologies, except for magnesium and zinc in the seed analysis. Overall, the findings indicate that understanding the morphological, proximate, and mineral compositions of B. thonningii is valuable for its sustainable utilization, conservation, domestication, and breeding. The pods and seeds of B. thonningii possess high nutritional potential and could be used for both human and animal nutrition following further detailed investigation.
Similar content being viewed by others
Introduction
Globally, wild plants are an important primary food source for daily survival, and livelihoods are directly dependent on them in most rural areas1,2. Wild edible plants play an essential role in providing sustenance, income, medicine, and food security to millions of individuals worldwide. People have a long-standing tradition of consuming foods obtained from trees grown in agroforestry systems or gathered from the wild3. Various types of wild plants play a key role in the diets and coping mechanisms of underprivileged rural communities residing in marginal lands across developing countries, helping them withstand harsh seasonal conditions4,5,6. These plants are essential in combating food insecurity in rural areas, serving as supplementary or emergency nourishment5. Their edible fruits often have high nutritional value and antioxidant properties, contributing to their global recognition for their health benefits7. Therefore, wild edible fruits offer a useful and inexpensive means of ensuring that people in the tropical regions have enough sources of fat, protein, carbohydrates and minerals8. Wild and domesticated fruits are important sources of vitamins and minerals, and some are rich in protein, fat, and crude fibre, particularly in rural areas where exotic fruit species are scarce8,9. Studies conducted by Biswas et al.10 and Rymbai et al.11 reported that fruits and vegetables with high fiber and antioxidant content can benefit in managing and treating diseases, such as cardiovascular problems, diabetes, inflammation, and digestive and urinary tract disorders. Nevertheless, the management practice and species diversity of these plants have deteriorated, especially in urban areas, due to the loss of traditional knowledge among local people12.
Bauhinia thonningii (Schumach.) is a leguminous plant species belonging to the family Fabaceae, sub-family Caesalpinioideae, and a tree species belonging to the monotypic genus Bauhinia13. The common English names are "Camel’s foot tree" and “Monkey Bread”, locally known as “Yekolla wanza” in Amargna and "Amam-gemel" in Tigrigna14. B. thonningii has a wide distribution range in tropical Africa, extending from West Africa to Sudan and southwards to East and central southern Africa. It is a small, dense tree found all over sub-humid Africa in wooded grasslands on different soils, from west to south. In Ethiopia, it grows at medium to low altitudes and performs well in the Moist and Wet Weyna Dega agroclimatic zones. It is distributed widely across various regions, typically ranging from 500 to 2000 m in altitude14. The local people in sub-Saharan Africa highly and widely valued the species. The wood is commonly used for poles, firewood, and charcoal. In addition, the pods serve as an essential fodder source for livestock and wild animals15. The B. thonningii tree species is used for firewood, charcoal, poles, timber, food (pods), drink (leaves, pods), medicine, fodder, bee forage, ornamental purposes, mulch, soil conservation, nitrogen fixation, tannin, dye, and rope14. The leaves of B. thonningii have been used in herbal medicine to treat inflammations, bacterial infections, worm infestations, and arrest bleeding16.
Highlighted the fruits and vegetables with better nutritional and phytochemical compounds can positively impact and benefit human health with regular consumption17,18. Many studies have also investigated the components of the B. thonningii tree leaves, bark, root, seed, and fruits have medicinal benefits. For instance, a comparison of the activity of the bark and leaves shows that the bark exhibits better antioxidant properties as reported by Ighodaro et al.19 and Dieng et al.20. Ibewuike et al.21 reported that this plant is used for treating infections and wounds and for reducing fever as well as inflammatory conditions like gingivitis. The B. thonningii plant contains flavonoids that have both anti-inflammatory and antimicrobial properties, which may explain its effectiveness in these conditions. The antibacterial activity of stem bark22; the phytochemical and proximate analysis of the leaves23; and the chemical, nutritional, antimicrobial, and antioxidant-vitamin profiles of the leaves were assessed by Ighodaro et al.19 and Ayenew et al.24.
Agroecology is a holistic and interdisciplinary approach that integrates ecological principles and practices into agricultural systems25. This approach significantly influences the morphological characteristics of plants, leading to more resilient and adaptive traits in response to harsh climatic conditions26. It focuses on sustainable farming methods that improve biodiversity, soil quality, root growth, stem thickness, leaf area, and overall ecosystem health and services25,27,28. Agroecological practices and strategies, such as using indigenous and drought-tolerant crop varieties, implementing diverse crop rotation and intercropping, utilizing organic inputs, adopting agricultural conservation practices, agroforestry, and effective water management techniques, promote the growth of plants with stronger morphological traits, including large pods and well-developed seeds25,29. The aforementioned practices enhance soil structure and microorganism activity, leading to greater nutrient availability and uptake28. As a result, pods and seeds produced in the agroecological systems tend to have a higher concentration of essential macro and micronutrients which are critical for human and animal health. Reguera et al.30 stated that several nutritional variables depend on agroecological location and cultivar, while some variables were stable across agroecological location. In addition, diverse agroecological situations could significantly impact the agronomic and nutritional properties of wild edible fruits. Okello et al.8,31 reported that there were significant differences in the physicochemical and mineral composition of the pulp and seeds of T. indica fruit across various agroecologies in Uganda. The nutritional variation among agroecologies is likely due to different climatic and environmental factors, the plant parts used, management practices, and the length of storage before analysis8.
Several studies have been conducted and documented in different parts of Ethiopia and elsewhere regarding the proximate and mineral composition of the leaves, bark, roots, and stems of the B. thonningii tree species. However, there are limited studies that explore the variations in the proximate and mineral composition of pods and seeds across different agroecological zones in semi-arid areas. Hence, the objectives of this study were (i) to describe the morphological characterization of B. thonningii fruit parts within and between agroecological zones; and (ii) to evaluate the proximate and mineral composition of pods and seeds collected from different agroecological zones in the Tselemti district, Northwestern Tigray, Ethiopia.
Materials and methods
Study area
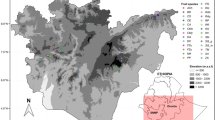
The study was carried out in the Tselemti district of Northwestern Tigray in the warm moist lowlands and tepid sub-moist mid-highlands agroecological zones situated between the latitude of 13° 05′ N and a longitude of 38° 08′ E in the semi-arid areas (Fig. 1). The region is characterized by distinct seasonal variations in Temperature and precipitation. The mean maximum temperatures vary from 33.0 °C in April to 41.7 °C in May. The mean minimum temperatures range from 15.8 °C in December and 21.7 °C in May. The annual precipitation has a unimodal distribution, with a dry season occurring between November and May and a rainy season between June and September. The mean annual rainfall is 1141.5 mm (Fig. 2). The topography is diverse, characterized by a combination of undulating hills, flat plateaus, mountains, valleys, and some immediate breaks in slope. The elevation ranges from a moderate 800 m to a higher elevation of 2870 m above sea level32. The Tselemti district agroecological zones comprise hot to warm-moist lowlands and tepid to cool-moist mid-highlands33.
Location of the study area based on agroecological classification and sub-districts in Tselemti, Northwestern Tigray, Ethiopia.
Climate diagram of Tselemti district showing rainfall distribution and temperature variation from 1981 to 2021 (Data source: Climate data from NASA’s archives).
The dominant soil types of the study area were vertisols, cambisols, and nitosols34. The farming system of the study area is a mixed agricultural system, production of crops and livestock. The types of crops cultivated comprise finger millet, sorghum, maize, teff, sesame, and chickpeas. The primary livestock include cattle, goats, donkeys, sheep, and chickens.
The natural vegetation comprises Combretum–Terminalia and Vachellia–Commiphora woodlands, characterized by small to moderate-sized drought-resistant trees and shrubs with large deciduous leaves. The study district supports several native woody tree species such as Anogeissus leiocarpus, Dichrostachys cinerea, Dovyalis abyssinica, Oxytenanthera abyssinica, Boswellia papyrifera, Erytherina abyssinica and Balanites aegyptiaca32.
Methods
Sampling design
The study district was located along an ecological gradient, considering differences in agroecological zones. Accordingly, the study district was stratified into two agroecologies: the warm moist lowlands (WMLL) and the tepid sub-moist mid-highlands (TSMMHL). This stratification facilitated comparison of B. thonningii tree morphological and nutritional composition under contrasting climatic and environmental conditions. In each agroecology, three representative sample sub-districts (a total of six sub-districts) were purposively selected based on the availability of the B. thonningii tree species in the area.
A total of 72 naturally growing adult B. thonningii trees (up to 36 trees per agroecology, and 12 trees per sub-district), were randomly selected and sampled across the two agroecologies (2 agroecologies × 3 sub-districts × 12 trees = 72 trees). The selection criteria included ease of access, absence of visible signs of pests, diseases, or fire damage, and the presence of healthy, mature trees bearing pods. Samples of individual trees and their pods were collected for morphological measurements as well as for proximate and mineral analysis. From each tree, 15 mature and fresh pods were collected, ensuring that pods were sampled from the lower, middle, and upper canopy positions to represent the target maturity stage. All 15 sample pods collected from each tree were pooled and treated as a single composite sample. Laboratory analyses were conducted for each sampled B. thonningii tree, and all assays were performed in technical triplicate to ensure repeatability of the analysis. For proximate compositional analysis, each B. thonningii tree sample was considered as a biological replicate (n = 36 trees per agroecology). In total, 1080 pods were collected from the two agroecologies (72 trees), corresponding to 540 pods per agroecology. For morphological measurements, 72 pods per agroecology were randomly sub-sampled from the 540 pods collected (Table 1). To minimize the likelihood of sampling genetically related individuals, B. thonningii trees were selected at least 200 meters31.
Sample collection methods
Mature and fresh pods were harvested during the dry season, between December and February, from the warm moist lowlands (WMLL) and tepid sub-moist mid highlands (TSMMHL) agroecologies of the Tselemti district (Fig. 3). Pods were collected from the lower, middle, and upper canopy levels of individual B. thonningii sample trees in each agroecology and sub-district. All sample paper bags were clearly labeled according to their agroecology, sub-district, and tree identification. The identities of biological replicates were coded and maintained throughout the study. Ladders were used to climb and access the canopy and collect mature pods, which were identified by their color change. Pod maturity is crucial for accurately determining mineral content, ensuring seed health, maintaining physiological development and viability, and preserving, storage properties of seeds, thereby enhancing the accuracy of research outcomes. Accordingly, B. thonningii mature pods were defined as those that were dark brown to blackish-brown, woody, and indehiscent, containing fully developed seeds (immature pods were characterized by green or light green coloration)14,35. From each sampled B. thonningii tree, five pods were collected from each of the three canopy levels, yielding a total of 15 pods per tree. Pods collected from each tree were pooled to form a composite sample. The B. thonningii pods was manually separated from the seeds and combined to create composite pod and seed samples according to agroecological classification. Pods were carefully cracked open using a hammer to split them gently along the edges and extract the seeds without causing damage. The seeds and pods were then placed in clean, dry, and properly labelled paper bags. Any decomposed or damaged pods and seeds were discarded. For fresh and dry weight determination, the composite pod and seed samples were weighed to record the fresh weight (FWt) using an analytical balance with a precision of ± 0.01 g. Sub-samples were oven-dried at 60–65 °C to a constant weight and reweighed to determine the dry weight (DWt) of the pod and seeds. The dried pod and seed samples were ground separately into fine powder using a grinder and stored at room temperature until laboratory analysis. After all preparatory processes were completed, the samples were transported to the laboratory for analysis.
Morphological characteristics of Bauhinia thonningii: (a) Tree, (b) Leave, (c) Young pods (d) Mature pods, (e) Dehisced pods, and (f) Matures seeds.
Identification of the plant
The plant species Bauhinia thonningii (Piliostigma thonningii) (Voucher No. DTK60) was recently identified and authenticated by Daniel Tadesse from the Department of Biology, College of Natural and Computational Sciences, University of Gondar, Ethiopia. This identification was conducted under the guidance of botanical taxonomy experts at the National Herbarium of the University of Addis Ababa. Identification was verified based on herbarium records and established taxonomic references. The material was deposited in the herbarium of the Department of Biology, University of Gondar, Gondar, Ethiopia for future reference, public access and study36. As this study relied on recently verified material collected from the same study areas, no new sample voucher specimens were prepared for plant identification.
Fruit morphological measurements
Morphological traits were measured from pods and seeds collected from individual B. thonningii trees in each agroecology (warm moist lowlands and tepid sub-moist mid-highlands). Each sampled pod and its corresponding seeds were analyzed separately to determine length, width, weight, and seed count. Morphological characteristics were assessed following established protocols, including measurements of pod length, width, thickness, and weight. Pods were carefully dissected to record seed weight and the number of seeds per pod. Pod weight (g) and seed weight (g) were determined using a precision analytical balance. Pod length (cm), pod width (cm), and pod thickness (cm) were measured with a measuring tape as the distance from the pod tip to the base of the pedicel. For curved pods, measurements were taken along the outer curvature. The number of seeds (ns) per pod was counted manually (Fig. 3).
Proximate analysis of pods and seeds
The analysis of proximate composition was conducted following the standard procedures recommended by the Association of Official Analytical Chemists (AOAC)37. All analyses were performed using dry (60–65 °C) and finely ground samples, with each parameter determined in technical triplicates to ensure analytical accuracy. Moisture content was determined by heating 2 g of each sample to a constant weight in a crucible placed in an oven maintained at 105 °C. Ash content was determined by incinerating 2 g of the sample in a muffle furnace at 550 °C for 4 h. Crude protein content was determined using the Kjeldahl method with 2 g of sample. Crude fat was quantified using Soxhlet extraction with petroleum ether (40–60 °C). Crude fibre content was obtained by digesting 2 g of sample sequentially with H2SO4 and NaOH, followed by incineration of the residue in a muffle furnace at 550 °C for 4 h, for both pods and seeds of B. thonningii. Total carbohydrates were calculated by difference. The energy value (kJ/100 g) was estimated based on the Atwater conversion factors: 4 kcal/g for protein and carbohydrates, and 9 kcal/g for fat, with the result converted to kilojoules (1 kcal = 4.184 kJ). The energy value in kilocalories (kcal) was converted to kilojoules (kJ) using the equation: Energy (kJ/100 g) = Energy (kcal/100 g) × 4.184. The energy value (kcal/100 g) was computed using the following equation38:
All proximate composition results were expressed on a dry weight basis (DW), and moisture content values were used to compute fresh weight equivalents where applicable.
Mineral analysis
The same biological and technical replicates were analyzed for their mineral composition, such as calcium (Ca), magnesium (Mg), phosphorus (P), potassium (K), sodium (Na), iron (Fe), manganese (Mn), copper (Cu), and zinc (Zn). For each sample, 0.5 g of dried pod and seed material was placed in digestion tubes, to which 7 mL of digestion mixture—comprising sulphuric acid (H2SO4), hydrogen peroxide (H2O2), and lithium sulphate as a catalyst was added, and thoroughly mixed. The mixture was heated in a block digester until the digest became clear, after which heating continued for an additional 30 min to ensure complete digestion of organic matter. The digest was then cooled to room temperature. All digested samples were transferred into 100 mL volumetric flasks and diluted to volume with distilled water. A 5 mL aliquot (a precisely measured portion or sub-sample) of each digested solution was taken for mineral analysis according to the respective agroecological origin. Detailed analytical procedures followed the methods described in AOAC37. The concentrations of Ca, K, and Na were determined using a Jenway flame photometer (Model PFP 7). Phosphorus concentration was determined spectrophotometrically using a Jenway UV/V Spectrophotometer (6405 model). The concentrations of Mg, Zn, Fe, Cu, and Mn were analyzed using an atomic absorption spectrophotometer (Perkin-Elmer, 2380 Model), following the AOAC protocol37. All mineral concentrations were expressed as milligrams per 100 g (mg/100 g) of dry weight. Biological replicates consisted of 36 trees per agroecology, and the resulting data were used to compute the mean and standard deviation or standard error. All the mineral analyses were conducted in triplicate, and the results were averaged.
Data analysis
The collected data were first checked for outliers, normality (Shapiro–Wilk test), and homogeneity of variances before data analysis. The differences in proximate and mineral composition of B. thonningii pod and seeds across different agroecologies were determined through ANOVA in a General Linear Model, using SPSS statistical software for Windows version 20.0. When significant variations were noticed (p < 0.05), mean separation was conducted using Tukey’s Honestly Significant Difference (HSD) test. For morphological characteristics, an independent sample t-test was performed to compare the mean values among the two agroecological zones. Principal component analysis was used to examine patterns of association among morphological measurements and their correlation with agroecological zones, using XLSTAT statistical software for Excel 2025. PCA was used to visualize the morphological characters and the variables that had the strongest and positive impact on the variation observed across agroecologies.
Results
Variation in fruit morphology
The B. thonningii pods exhibited variation in shape, ranging from curved to straight in both agroecologies. Significant differences (p < 0.05) were observed between the two agroecological zones for mean pod length (t = 4.063, p < 0.000), pod width (t = 2.120, p = 0.036), pod thickness (t = 2.827, p = 0.005), pericarp weight (t = − 2.818, p = 0.006), total seed weight (t = 3.058, p = 0.003), individual seed weight (t = − 2.823, p = 0.005), and the number of seeds per pod (t = 4.573, p < 0.000) (Table 2). Only total dry pod weight did not differ significantly across the agroecologies (t = 1.376, p = 0.171). The mean pod length (18.34 cm), pod width (2.87 cm), pod thickness (0.85 cm), total dry pod weight (11.56 g), total seed weight (8.93 g), and number of seeds per pod (51.0 ns) were all higher in the warm moist lowland compared to the tepid sub-moist mid-highland agroecology. In contrast, pericarp weight (3.47 g), and individual seed weight (0.196 g) were higher in the tepid sub-moist mid-highland than in the warm moist lowland (Table 2). Overall, most of morphological traits were higher in the warmmoist lowland, indicating that optimal temperature and moisture conditions in this agroecology favour larger pod size, higher pod weight, and better seed development.
Proximate compositions of B. thonningii pods and seeds
The moisture content of B. thonningii pods (9.02%) and seeds (7.02%) was higher in the tepid sub-moist mid-highland than in the warm moist lowland agroecology. The ash content of pods was higher than that of seeds across both agroecologies, likely due to the mineral deposition in the pod wall during maturation and soil mineral uptake. The crude protein, crude fat, crude fiber, total carbohydrates, and energy value of B. thonningii pods and seeds were higher in the WMLL compared to the TSMMHL agroecology. All proximate compositions of B. thonningii pods and seeds differed significantly between the two agroecologies (p < 0.05), except for seed. Ash content, which did not differ significantly (t = 0.907, p = 0.371) (Table 3).
Mineral composition of B. thonningii pods and seeds
In the present study, the mineral composition of B. thonningii pods and seeds varied significantly between the two agroecologies, except for magnesium and zinc concentrations in seeds, which showed no significant differences (p < 0.05, Table 4). Most mineral concentrations in pods and seeds, such as calcium (152.26 and 36.77 mg/100 g), magnesium (129.59 and 8.04 mg/100 g), potassium (1325.44 and 130.61 mg/100 g), and sodium (8.99 and 16.90 mg/100 g), respectively, were significantly higher in the WMLL compared to the TSMMHL. In contrast, the TSMMHL agroecology showed higher concentrations of phosphorus (90.71 and 20.47 mg/100 g), iron (13.82 and 69.23 mg/100 g), copper (1.27 and 0.85 mg/100 g), manganese (0.78 and 0.81 mg/100 g), and zinc (2.97 and 0.80 mg/100 g) concentration, probably due to favourable soil fertility, moderate temperature, and moisture conditions that enhance nutrients accumulation and seed maturation. Among all minerals, potassium was the most abundant, particularly in seeds, reflecting its critical role in carbohydrate transport and enzyme activation during seed development. On the other hand, phosphorus was slightly higher in TSMMHL compared to that of WMLL agroecology, which may be related to better soil pH and phosphorus availability in TSMMHL agroecology. Furthermore, the micronutrients were higher in the TSMMHL agroecology, possibly due to improved retention and availability of these minerals in the relatively cooler and less leached mid-highland soils. Overall, the results of the study revealed that most mineral compositions of pod and seed of B. thonningii are influenced by agroecological situations, whereas magnesium concentration in the seed analysis seems relatively stable across the agroecological zones (Table 4).
Variations in fruit morphology across agroecological zones
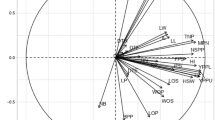
The screen plot (Fig. 4) showed that the “elbow” or the shape of the curve (red line) is definitely after the second principal component (PC2) where the curve is becoming flattened. Therefore, the PC1 and PC2 were able to explain a significant level of total variation. The PC1 eigenvalue (4.33) is the highest and accounts the largest proportion of variance (54.13%) of morphological characters. The PC1 also had a high loaded on the overall pod and seed size (length of pod, total dry pod weight, and total seed weight per pod). The second component (PC2) had a moderate eigenvalue (1.63) and the variance (20.42%). The two main principal components (PC1 and PC2) had an eigenvalue greater than one had taken, and explained 74.55% of the majority of the total morphological variation among B. thonningii pods and seeds. The PC2 also showed the pod shape variation like the pod width and the pod thickness. The third principal component (PC3) has a smaller eigenvalue (1.08); however, it still contributes significantly to the total variance. Between PC4–PC7, the morphological traits contribute little unique information, there eigenvalues are less than 1 (Fig. 4). The biplot (Fig. 5) further demonstrates the association between morphological traits and the sample agroecological zones.
Scree plot showing component eigenvalues for B. thonningii morphological traits.
The PCA biplot indicating morphological attributes of B. thonningii tree across two agroecological zones in Tselemti district, Northern Ethiopia.
Pricipal Component Analysis (PCA) was conducted using eight major morphological characteristics of B. thonningii tree pods and seeds to examaine the key characterstics that contributing to variations among the two agroecological zone. As indicated in Table 5 different morphological traits had different loading patterns. The first principal component (PC1) had a high positive loadings on total dry pod weight (0.944), total seed weight per pod (0.931), number of seeds per pod (0.890), pod pericarp weight (0.809), pod length (0.650), and pod width (0.606) which implies that PC1 mainly reflects the characteristics of pod yield and seed productivity. The second principal component (PC2) had a strong and positive association with pod width (0.668) and pod thickness (0.683), which are the pod dimensional characterstics. Overall, the PC1 records the differences in variability of the pod and seed yield, whereas the PC2 is the reflectionof the pod size attributes. Therefore, the two principal components (PC1 and PC2) have been effective in summarizing the major morphological variability of B. thonningii tree population among the two agroecologies, these implying that both pod productivity and pod dimentional characters play a significant role in determining the observed environmental variation.
Discussion
Morphological characteristics
The current study indicated that environmental difference among agroecologies had a significant effect on most morphological characteristics, except for total dry pod weight. In line with this finding, Fadel et al.39 reported that environmental factors have significant influence on morphometric attributes of pods and seeds. The environmental factors like rainfall appear to enhance the mass, length, width and thickness of the pods than with low rainfall geographical regions of Morocco. In legume trees like Nesphostylis bracteata, there was significant variation in pod length, pod width, pod weight, and number of developing seeds across all the locations40. Girmay et al.41 reported that the morphological attributes were significantly different among the morphotypes of Tamarind. Van den Bilcke et al.42 and Nasar‐Abbas et al.43 found that the pods were significantly different in the morphological characteristics for instance size, dimension, shape, weight, density, color, and seed-to-pulp ratio due to land use, climate conditions, and farming practices. According to Nasar‐Abbas et al.43, the morphological attributes of Ceratonia siliqua L. pods indicated that they vary according to their origin, and strongly depend on the biogeographical areas of the tree and the conditions of its production. The variability observed in pod characteristics and seeds (Table 2) in B. thonningii from the two agroecologies could be due to environmental factors, edaphic and geographical locations. Patchiness in terms of pod morphological attributes was noted in several accessions of Lablab purpureus44 and Sphenostylis stenocarpa45,46 collected across different agroecologies. The morphological attributes of pods are essential traits to be considered in genetic improvements and breeding programs for underexplored and underutilized legume species40,46. Thus, morphological diversity observed in pod attributes across different agroecology suggests that longer pod length, higher pod weight, and the number of seeds/pods can be utilized in germplasm conservation and breeding programs for the B. thonningii tree.
Proximate and mineral compositions
Proximate composition
Moisture content indicates the presence of water-soluble vitamins47 and was higher in pods than the seeds of the B. thonningii in both agroecologies. The higher moisture content of the B. thonningii pods was observed in the TSMMHL (9.02%) compared to the WMLL agroecology (7.87%) (Table 3). The higher moisture content of the B. thonningii seeds was also recorded in the TSMMHL (7.02%) compared to the WMLL agroecology (6.76%). In comparison to legumes, Animashahun et al.48 observed that the moisture content of Parkia biglobosa pods has a lower amount of moisture than the B. thonningii pods in both agroecologies. The moisture content of B. thonningii seeds obtained in this study was high compared with 6.71% as reported by Jimoh and Oladiji14, however, lower compared with some underutilized legumes seeds such as Canavalia ensiformis (8.5%), C. gladiate (8.5%), M. pruriens var. pruriens (8.4%)49, Parkia biglobosa (10.8%)48 and Tamarindus indica47. In contrast, the amount of moisture content in Millettia ferruginea50 and Parkia biglandulosa seeds51 was lower than the B. thonningii seeds in both agroecologies. Likewise, the moisture content of B. thonningii seed was found to be lower than some grain legume seeds such as Phaseolus vulgaris, Pisum sativum, and Vigna sinensis52. The variations in proximate compositions in different B. thonningii tree and agroecologies may be influenced by factors like climate, soil, and maturity stage.
The ash content serves as a measure of mineral content47. The results indicated that the ash content was higher in the pod than in the seeds of the B. thonningii in both agroecologies. The ash content of the B. thonningii pods and seeds was higher in the WMLL (4.30%) and TSMMHL (3.71%) agroecologies, respectively (Table 4). Melesse et al.53 reported that the ash content was higher in the pods of Moringa stenopetala, followed by Moringa oleifera, and Prosopis juliflora, than the pods of B. thonningii. In contrast, Toumi et al.54 and Animashahun et al.48 found that the ash content of Ceratonia siliqua and Parkia biglobosa pods was lower compared with the pods of B. thonningii in both agroecologies. The ash content of B. thonningii seeds obtained in this study was 3.71% in TSMMHL and 3.60% in WMLL agroecologies, higher compared with the 3.5% reported by Jimoh and Oladiji14. Some legume seeds, for instance, Dolichos lablab var. vulgaris55, Parkia biglandulosa51, and Trigonella foenum-graecum56 had the higher amounts of ash content than the seeds of B. thonningii. In contrast, Animashahun et al.48 observed that there was a lower amount of ash content in the Parkia biglobosa seeds (1.99%). Ebifa Othieno et al.47 also indicated that the ash content is lower in the semi-arid and higher in the sub-humid agroecological zone in the seed of Tamarindus indica than the B. thonningii seed ash content. Overall, these results suggest that ash content in B. thonningii seeds is stable and not strongly affected by agroecological zones or environmental variations. Mineral accumulation in seed ash appears to be primarily genetically regulated rather than influenced by external factors such as soil moisture, temperature, or altitude.
The crude protein content of B. thonningii pods and seeds was higher in the WMLL (9.74% and 30.73%) than the TSMMHL agroecology. The crude protein content was higher in the seeds than in the pod of B. thonningii across the two agroecologies (Table 3). The crude protein content of the B. thonningii pods was higher than that of Parkia biglobosa pods48. The pods of B. thonningii have a lower crude protein content than pods of Prosopis africana (12%) and Panicum maximum (7.94%). However, in the WMLL agroecology, the crude protein content of B. thonningii pods was higher than that of Panicum maximum pods, which can alleviate the nutritional deficiencies of livestock during the dry season57. Similarly, higher crude protein contents of several Ethiopian multipurpose tree species, including Moringa stenopetala, followed by Prosopis juliflora, Moringa oleifera and Millettia ferruginea53, and Ceratonia siliqua pods54 were reported. The crude protein content of B. thonningii seeds was higher in the WMLL (30.73%) and lower in the TSMMHL (27.40%) agroecologies than the Millettia ferruginea seeds which is 29.7%50. In legumes, for instance, Detarium microcarpum58, Parkia biglandulosa51, and Trigonella foenum-graecum56 were found to be higher amounts of crude protein, which indicates the good nutritional quality of the species. Jimoh and Oladiji14 found that the crude protein content of B. thonningii seeds was 30.33% which means higher for TSMMHL (27.40%) and lower for the WMLL (30.73%) agroecologies. In contrast to this study, Ebifa Othieno et al.47, Animashahun et al.48 and Kalpanadevi and Mohan55 reported that the crude protein contents were lower in the seeds of Dolichos lablab var. vulgaris, Tamarindus indica, and Parkia biglobosa compared to the B. thonningii seeds, respectively. Generally, legume trees are an essential source of high-quality protein-rich forage for subsistence and commercial livestock production57,59,60. Furthermore, proteins from legumes are essential components of the human diet. In conclusion, legumes are affordable, widely available, and consumed by nearly all people around the globe61.
The crude fat was higher in both pod and seeds of the B. thonningii in the WMLL agroecology. The higher crude fat content was recorded in the seeds than that of the pods of B. thonningii across the two agroecologies (Table 3). The B. thonningii pods had the lower crude fat values compared to other tree species such as Millettia ferruginea (7.78%), Moringa oleifera (3.11%), Moringa stenopetala (2.42%)53, and Ceratonia siliqua pods54. In contrast, the B. thonningii pods had higher crude fat values (0.96%) in the WMLL agroecology than Prosopis juliflora (0.87%)53 and Parkia biglobosa pods (0.89%)48. The crude fat content of B. thonningii seeds was recorded 1.76% in the TSMMHL and 2.48% in the WMLL agroecologies, which was lower than that of some legumes such as Parkia biglandulosa51, Tamarindus indica47, Trigonella foenum-graecum56, and higher than that of D. microcarpum seeds58.
The higher crude fibre content of the B. thonningii pods and seeds was observed in the WMLL compared to the TSMMHL agroecology. The crude fibre content was lower in the pod than in the seeds of B. thonningii across the two agroecologies (Table 3). The current study indicated that the B. thonningii pods had a lower crude fibre than pods of some other tree species, including Prosopis juliflora (31.7%), Millettia ferruginea (34%), Moringa oleifera (33%), and Moringa stenopetala (37%)53. In contrast to this finding, Toumi et al.54 and Animashahun et al.48 revealed that lower crude fibre content in Ceratonia siliqua and Parkia biglobosa pods compared to the B. thonningii pods. The crude fibre content of B. thonningii seeds was 26.67% in the TSMMHL and 32.86% in the WMLL agroecologies which is lower compared with 35.03% as reported by Jimoh and Oladiji14. This may be due to the origin and agroecological variations. Ebifa Othieno et al.47, Animashahun et al.48, Varkekara and Singh51 and Bakhtiar56 found that there was a lower amount of crude fibre in the seeds of Tamarindus indica, Parkia biglandulosa, and Trigonella foenum-graecum compared to the B. thonningii seeds in both agroecologies.
The total carbohydrate content of B. thonningii pods and seeds was higher in the WMLL (56.30% and 33.70%) than that of the TSMMHL (51.10% and 23.31%) agroecology, respectively (Table 3). In contrary to this study, Weber et al.62 reported that higher carbohydrate contents were recorded in the pods and seeds of Acacia farnesians (63.59% and 42.89%), and Prosopis juliflora (63.21% and 48.92%). The carbohydrate content found in this study was lower than in the seeds of other legumes, such as D. microcarpum58, Tamarindus indica47, Parkia biglandulosa51, Trigonella foenum-graecum56 and Parkia biglandulosa56. Tharanathan and Mahadevamma 59stated that legumes are considered the "poor man’s meat" because of their affordability and availability. Legumes are generally good sources of slow-release carbohydrates (i.e., dietary fibre). A study conducted by Jimoh and Oladiji14 reported that the carbohydrate content of B. thonningii seeds was lower than the carbohydrate content obtained in the current study in both agroecologies.
Energy is required to sustain the body’s various functions, including respiration, circulation, physical work, and protein synthesis63. The energy content of the pods was relatively higher than that of the B. thonningii seeds across the two agroecologies (Table 3). The energy value of B. thonningii seeds was higher in the WMLL (1103.87 kJ/100 g) than that of the TSMMHL (1010.42 kJ/100 g) agroecology. In contrast to this finding, Weber et al.62 stated higher energy values in the pods and seeds of Acacia farnesians (1217.54 kJ/100 g and 1134 kJ/100 g) and Prosopis juliflora (1167.34 kJ/100 g and 1410 kJ/100 g), respectively, compared with the B. thonningii pods and seeds. Similarly, studies on tree legume species have reported higher energy values in Parkia biglandulosa51 and Trigonella foenum-graecum seeds56, compared to the B. thonningii seeds in both agroecologies. In general, the crude protein, crude fat, crude fiber, total carbohydrates, and energy value of B. thonningii pods and seeds were higher in the WMLL agroecology. This may be attributed to the more favourable growth conditions and higher temperatures in the WMLL, which enhance metabolic processes such as photosynthesis and soil enzymatic activity, thereby increasing the synthesis of organic compounds.
Mineral composition
The calcium concentration was higher in the pods compared to the seeds of the B. thonningii and was higher in the WMLL compared to the TSMMHL agroecology (Table 4). Melesse et al.53 found that the calcium concentration was higher in some legume species such as Moringa stenopetala (508 mg/100 g), Moringa oleifera (556 mg/100 g), Millettia ferruginea (169 mg/100 g), and Prosopis juliflora (441 mg/100 g) pods than the calcium levels of B. thonningii pods. The calcium concentration of B. thonningii seeds obtained in this study were 28.23 mg/100 g in TSMMHL and 36.77 mg/100 g in WMLL agroecologies, which are higher compared with 4.31 mg/100 g reported by Jimoh and Oladiji14. This may be due to the origin and agroecological variations. Datir et al.40, Ebifa Othieno et al.47, and Kalpanadevi and Mohan55 reported that there was a higher amount of calcium value in the seeds of Dolichos lablab var. vulgaris; Tamarindus indica and Nesphostylis bracteata than in the B. thonningii seeds in both agroecologies, respectively. Climate, maturity at harvest, soil characteristics and seasonal variation could influence the nutritional and mineral compositions of the tree species53,64. Overall, the macronutrients were higher in the WMLL agroecology, this is likely due to higher humidity, soil mineralization, evaporative concentration, and increased soil nutrient mobility in the warm lowland environment.
Magnesium concentration of pods was higher than in the seeds of B. thonningii, with the higher concentration recorded in the WMLL than the TSMMHL agroecology (Table 4). Melesse et al.53 reported that the magnesium concentration was higher in some legume species, for instance, Moringa stenopetala (461 mg/100 g), Moringa oleifera (374 mg/100 g), Millettia ferruginea (143 mg/100 g), and Prosopis juliflora (215 mg/100 g) pods than the magnesium level of B. thonningii pods. The magnesium concentration of B. thonningii seeds in this study was 7.42 mg/100 g in the TSMMHL and 8.04 mg/100 g in the WMLL agroecologies. The values were lower compared to the findings of Kalpanadevi and Mohan55 for Dolichos lablab var. vulgaris, Tamarindus indica47, Parkia biglandulosa51, and Nesphostylis bracteata seeds40.
Phosphorus concentration of pods was significantly higher than in the seeds of B. thonningii, with the higher concentrations observed in the TSMMHL than the WMLL agroecology (Table 4). Melesse et al.53 stated that the phosphorus levels were higher in some legumes, for instance, Moringa stenopetala (516 mg/100 g), Moringa oleifera (536 mg/100 g), Millettia ferruginea (194 mg/100 g), and Prosopis juliflora (246 mg/100 g) pods than the phosphorus level of B. thonningii pods. The result indicated that the phosphorus concentration of B. thonningii seeds was 18.32 mg/100 g in the WMLL and 20.47 mg/100 g in the TSMMHL agroecologies, which was higher than the report indicated by Jimoh and Oladiji14. On the other hand, high amounts of phosphorus concentration were reported in seeds of Dolichos lablab var. vulgaris55 and Nesphostylis bracteata40 compared to the B. thonningii seeds.
The potassium concentration was higher in the pods than in the seeds of B. thonningii, with the higher concentrations recorded in the WMLL than in the TSMMHL agroecology (Table 4). Melesse et al.53 reported that the potassium concentration was lower in some legume species, such as Moringa stenopetala (427 mg/100 g), Moringa oleifera (371 mg/100 g), Millettia ferruginea (155 mg/100 g), and Prosopis juliflora (217 mg/100 g) pods than the potassium concentration of B. thonningii pods in TSMMHL (1207 mg/100 g) and WMLL (1325 mg/100 g) agroecologies. The potassium concentration of B. thonningii seeds was 119.23 mg/100 g in TSMMHL and 130.61 mg/100 g in WMLL agroecologies. These values were lower compared to the potassium concentrations reported for some legume seeds such as Dolichos lablab var. vulgaris55, Tamarindus indica47, Parkia biglandulosa51, and Nesphostylis bracteata40. The overall concentrations of potassium among all the minerals in the B. thonningii pods was found to be higher across the two agroecologies, followed by calcium and magnesium. This might be of nutritional importance, especially in parts of the world where a high dietary intake of potassium reduces blood pressure and protects people from several conditions affecting the cardiovascular system, kidneys, and bones65.
Sodium concentration was significantly different across the two agroecologies (p < 0.05). The variation could be the differences in the mineral content of the soils in the two agroecologies in the district. Sodium concentration was lower in the pods compared to the seeds of the B. thonningii, with the higher concentrations observed in the WMLL compared to the TSMMHL agroecology (Table 4). Melesse et al.53 found that the sodium concentration was higher in some legumes for example Moringa stenopetala (48 mg/100 g), Moringa oleifera (38 mg/100 g), and Prosopis juliflora (21 mg/100 g) pods than the sodium concentration of B. thonningii pods across the two agroecologies. In contrast, the sodium concentration of the B. thonningii pods in both agroecologies were higher than those of Millettia ferruginea pods (6 mg/100 g)53. The sodium concentration of B. thonningii seeds obtained in this study was 13.88 mg/100 g in the TSMMHL and 16.90 mg/100 g in the WMLL agroecologies. These values are lower compared with the values of Dolichos lablab var. vulgaris55 and Tamarindus indica seeds in sub-humid agroecology as reported by Ebifa Othieno et al.47. On the other hand, Ebifa Othieno et al.47 found that the sodium value in Tamarindus indica seeds was lower in semi-arid agroecology compared to B. thonningii seeds in the WMLL agroecology. In addition, Varkekara and Singh51 also reported lower sodium values in Parkia biglandulosa seeds.
Iron concentration of pods was lower than in the seeds of the B. thonningii. However, iron concentration was higher in the TSMMHL compared to the WMLL agroecology for both pods and seeds of B. thonningii (Table 4). Among all the minerals, the iron concentration in the B. thonningii pods was relatively higher in both agroecologies followed by potassium, calcium, magnesium, and phosphorus. Melesse et al.53 reported that the iron concentration was higher in some legumes for example Moringa stenopetala (64.1 mg/100 g) and Prosopis juliflora pods (24.1 mg/100 g) compared to the iron concentrations of B. thonningii pods across the two agroecologies. In contrast, the iron concentration of B. thonningii pods in the TSMMHL agroecologies was higher than that of Millettia ferruginea pods (10.8 mg/100 g); however, the iron concentration in the WMLL agroecology was relatively lower than that of Moringa oleifera pods (13.82 mg/100 g)53. The iron concentration of the B. thonningii seeds obtained in this study was 54 mg/100 g in the WMLL and 69 mg/100 g in the TSMMHL agroecologies, which was lower than the value reported by Jimoh and Oladiji14. Conversely, Datir et al.40, Ebifa Othieno et al.47, Kalpanadevi and Mohan55, Varkekara and Singh51 reported lower iron concentrations in Dolichos lablab var. vulgaris, Tamarindus indica, Parkia biglandulosa, and Nesphostylis bracteata seeds, respectively, compared with the B. thonningii seeds in both agroecologies. Generally, iron is a critical component of hemoglobin and myoglobin, which play vital roles in the metabolism of humans and animals66. When iron intake is chronically low, stores can become depleted, decreasing hemoglobin levels. The deficiency of iron minerals produces anemia67.
Manganese concentration was higher in seeds than the pods of the B. thonningii, with the higher concentration observed in the TSMMHL than the samples taken from the WMLL agroecology for both pods and seeds (Table 4). Melesse et al.53 reported that the manganese concentration was higher in some legumes, for instance, Moringa stenopetala (2.71 mg/100 g), Moringa oleifera (2.78 mg/100 g), Millettia ferruginea (5.49 mg/100 g), and Prosopis juliflora (4.07 mg/100 g) pods than the manganese concentration of B. thonningii pods. The manganese value of B. thonningii seeds obtained in this study was 0.81 mg/100 g in TSMMHL and 0.66 mg/100 g in WMLL agroecologies, which was lower than the value reported by Jimoh and Oladiji 14. In comparison, some legume seeds such as Dolichos lablab var. vulgaris (1.36 mg/100 g)55, Parkia biglandulosa (1.29 mg/100 g)51, and Nesphostylis bracteata (2.55 mg/100 g)40 have higher manganese concentrations compared to the B. thonningii seeds in both agroecologies.
Copper concentration was higher in the pod than the seeds of B. thonningii. However, copper concentration was higher in the TSMMHL than the samples taken from the WMLL agroecology for both pods and seeds (Table 4). Melesse et al.53 reported that the copper concentration was lower in some legumes, such as Moringa stenopetala (0.47 mg/100 g), Moringa oleifera (0.42 mg/100 g), Millettia ferruginea (0.39 mg/100 g), and Prosopis juliflora (0.41 mg/100 g) pods than the copper concentration of the B. thonningii pods in both agroecologies (Table 4). In this study, the copper concentrations of the seeds obtained was 0.85 mg/100 g in the TSMMHL and 0.72 mg/100 g in the WMLL agroecologies, which was higher than some wild legumes such as Canavalia ensiformis (0.4 mg/100 g), Cassia floribunda (0.3 mg/100 g), Mucuna monosperma (0.2 mg/100 g) value indicated by Vadivel et al.49 and Nesphostylis bracteata (0.47 mg/100 g)40. However, Varkekara and Singh51 and Kalpanadevi and Mohan55 found higher copper values in Dolichos lablab var. vulgaris seeds (1.36 mg/100 g) and Parkia biglandulosa (2.11 mg/100 g) compared to the B. thonningii seeds in both agroecologies.
Zinc concentrations were significantly higher in the pod of B. thonningii compared to the seeds, with the higher concentrations recorded in the TSMMHL compared to the WMLL agroecology (Table 4). Melesse et al.53 stated that the zinc concentrations were higher in some legumes, such as Moringa stenopetala (4.11 mg/100 g) and Prosopis juliflora pods (3.64 mg/100 g) compared with those of B. thonningii pods in both agroecologies (Table 4). On the other hand, the zinc concentrations of Moringa oleifera (2.75 mg/100 g) and Millettia ferruginea (2.55 mg/100 g) pods were lower than the TSMMHL agroecology, but higher than the WMLL agroecology. In this study, analysis revealed that zinc concentration of seeds was 0.80 mg/100 g in TSMMHL and 0.74 mg/100 g in WMLL agroecologies, which was higher than the report indicated by14. In comparison, high zinc concentrations were reported in the seeds of some wild legumes, for instance, Canavalia ensiformis, Cassia floribunda, and Mucuna monosperma49, Dolichos lablab var. vulgaris (2.24 mg/100 g)55, Parkia biglandulosa (6.86 mg/100 g)51, and Nesphostylis bracteata (1.59 mg/100 g)40 than that of the B. thonningii seeds in both agroecologies. In general, there was statistically significant variation of minerals among the different agroecologies. The variations could be due to differences in the mineral content of the soil, climatic conditions, and the physiological adaptability of the species to diverse ecological situations. Plant species obtain minerals from the soil, which directly reflect the geological formation of the area along with other soil-forming factors, particularly climate, mineralogy, and topography factors68,69,70.
Limitations and future research directions
Even though this study offers valuable insights and contributes to the current understanding of the morphological traits, proximate, and mineral compositions of B. thonningii pods and seeds across different agroecological zones in the Tselemti district, northern Ethiopia, some limitations are noted. The samples were collected during a single season, which may not fully capture temporal variations in pod and seed composition caused by inter-annual climatic and phenological differences. Furthermore, while a stratified design and appropriate sample size were employed to ensure representativeness across sites and agroecologies, the findings may not be entirely generalizable to all ecological zones and B. thonningii populations across broader landscapes. The study focused primarily on major proximate and mineral components, excluding phytochemicals, genetic variability, and soil-related factors that may influence the observed composition. In addition, interactions between soil properties and plant characteristics, as well as genetic factors affecting morphological, proximate, and mineral traits, were not investigated. Further research should consider the seasonal dynamics, genetic diversity, and soil–plant interactions, and employ advanced analytical techniques to develop a more comprehensive understanding of the species. Such studies would enhance the wider utilization of this tree for food, feed, and agroforestry applications and enable rapid assessment of its nutrient quality. In general, by addressing these gaps, subsequent research can help transform B. thonningii from an underutilized wild resource into a scientifically validated, sustainable, and integral component of food and nutritional security strategies in Africa, particularly in Ethiopia.
Conclusions
The study revealed significant variations in most morphological traits, proximate compositions, and mineral concentrations of B. thonningii pod and seeds between the two agroecological zones. B. thonningii trees growing in the warm moist lowlands exhibited higher values for pod dimensions, seed weight, crude protein, crude fat, crude fiber, total carbohydrates, and energy content, indicating more suitable environmental conditions for nutrient accumulation. The fruit components of B. thonningii from both agroecologies demonstrate considerable potential as supplementary food and nutritious feed sources for humans and livestock, given their nutrient content. However, the proximate and nutritional compositions may vary across agroecosystems, land use types, tree ages, and environmental conditions; hence, consistent and broader datasets are still lacking. Like many leguminous species, B. thonningii fruits could serve as an affordable and sustainable source of proximate and mineral nutrients, helping to alleviate macro-and micronutrient deficiencies in rural and urban communities. Variations in pod and seed morphometric traits likely reflect differences in genetic origin, geographic location, and climatic conditions. Overall, the findings highlight the potential of B. thonningii as a valuable nutritional resource, while underlining the need for further research on its anti-nutritional factors, nutrient bioavailability, and processing methods to support its safe and effective use in food and feed systems.
Data availability
Data are available from the corresponding author upon reasonable request. Requests should include a detailed rationale for data access and a commitment to using the data solely for the stated purpose, in compliance with ethical guidelines.
References
Chauke, S., Shelembe, B. G., Otang-Mbeng, W. & Ndhlovu, P. T. Ethnobotanical appraisal of wild fruit species used in Mpumalanga Province, South Africa: A systematic review. S. Afr. J. Bot. 171, 602–633. https://doi.org/10.1016/j.sajb.2024.06.047 (2024).
Khan, M. N. et al. An in-depth investigation of the nutraceutical value and medicinal perspectives of wild medicinal plants in Ojhor Valley, Hindukush Range, Chitral Pakistan. Genet. Resour. Crop Evol. https://doi.org/10.1007/s10722-024-01996-3 (2024).
Asprilla-Perea, J. & Díaz-Puente, J. M. Importance of wild foods to household food security in tropical forest areas. Food Sec. 11, 15–22. https://doi.org/10.1007/s12571-018-0846-8 (2019).
Suwardi, A. B. & Navia, Z. I. Sustainable use and management of wild edible fruit plants: A Case Study in the Ulu Masen Protected Forest, West Aceh. Indonesia. J. Sustain. For. 42(8), 811–830. https://doi.org/10.1080/10549811.2022.2123355 (2023).
Anbessa, B., Lulekal, E., Getachew, P. & Hymete, A. Ethnobotanical study of wild edible plants in Dibatie district, Metekel zone, Benishangul Gumuz Regional State, western Ethiopia. J. Ethnobiol. Ethnomed. 20(27). https://doi.org/10.1186/s13002-024-00671-2 (2024).
Priyadarshini, S., Tudu, S., Dash, S. S., Biswal, A. K. & Sahu, S. C. Wild edible plants: diversity, use pattern and livelihood linkage in Eastern India. Genet. Resour. Crop Evol. https://doi.org/10.1007/s10722-023-01833-z (2024).
Angami, T. et al. Exploring the nutritional potential and anti-nutritional components of wild edible fruits of the Eastern Himalayas. Food Measure 18, 150–167. https://doi.org/10.1007/s11694-023-02147-5 (2024).
Okello, J., Okullo, J. B., Eilu, G., Nyeko, P. & Obua, J. Mineral composition of Tamarindus indica LINN (Tamarind) pulp and seeds from different agro-ecological zones of Uganda. Food Sci. Nutr. 5(5), 959–966. https://doi.org/10.1002/fsn3.490 (2017).
Amarteifio, J. O. & Mosase, M. O. The chemical composition of selected indigenous fruits of Botswana. J. Appl. Sci. Environ. Manag. 10(2), 43–47. https://doi.org/10.4314/jasem.v10i2.43659 (2006).
Biswas, S. C. et al. Nutritional composition and antioxidant properties of the wild edible fruits of Tripura Northeast India. Sustainability 14(19), 12194. https://doi.org/10.3390/su141912194 (2022).
Rymbai, H., Verma, V. K., Talang, H., Assumi, S. R., Devi, M. B., Vanlalruati, Sangma, R. H. C. H., Biam, K. P., Chanu, L. J., Makdoh, B., Singh, A. R., Mawleiñ, J., Hazarika, S. & Mishra, V. K. Biochemical and antioxidant activity of wild edible fruits of the eastern Himalaya, India. Front Nutr. 10, 1039965. https://doi.org/10.3389/fnut.2023.1039965 (2023).
Beleta, K. G., Jiru, D. B. & Tolera, K. D. Assessment of edible woody plants’ diversity, their threats, and local people’s perception in Borecha Woreda of Buno Bedele Zone Southwestern Ethiopia. Int. J. For. Res. 2024, 7269154. https://doi.org/10.1155/2024/7269154 (2024).
Jimoh, F. O. & Oladiji, A. T. Preliminary studies on Piliostigma thonningii seeds: proximate analysis, mineral composition and phytochemical screening. Afr. J. Biotechnol. 4(12), 1439–1442 (2005).
Bekele-Tesemma, A. Useful trees of Ethiopia: identification, propagation and management in 17 agroecological zones. Nairobi: RELMA in ICRAF Project, 552 (2007).
Chidumayo, E. N. Growth responses of an African savanna tree, Bauhinia thonningii Schumacher, to defoliation, fire and climate. Trees 21(2), 231–238. https://doi.org/10.1007/s00468-006-0115-x (2007).
Ozolua, R. I., Alonge, P. & Igbe, I. Effects of leaf extracts of Piliostigma thonningii Schum on Aortic ring contractility and bleeding time in Rats. J. Herbs Spices Med. plants 15(4), 326–333. https://doi.org/10.1080/10496470903507874 (2010).
Yahia, E. M., García-Solís, P. & Celis, M. E. M. Chapter 2-Contribution of fruits and vegetables to human nutrition and health, Editor(s): Elhadi M. Yahia, Postharvest Physiology and Biochemistry of Fruits and Vegetables, Woodhead Publishing, 19–45, ISBN 9780128132784. https://doi.org/10.1016/B978-0-12-813278-4.00002-6 (2019).
Kaparapu, J., Pragada, P. M. & Geddada, M. N. R. Fruits and vegetables and its nutritional benefits. In: Egbuna, C., Dable Tupas, G. (eds) Functional Foods and Nutraceuticals. Springer, Cham., pages 241–260. https://doi.org/10.1007/978-3-030-42319-3_14 (2020).
Ighodaro, O. M., Agunbiade, S. O., Omole, J. O. & Kuti, O. A. Evaluation of the chemical, nutritional, antimicrobial and antioxidant-vitamin profiles of Piliostigma thonningii leaves (Nigerian species). Res. J. Med. Plant 6(7), 537–543. https://doi.org/10.3923/rjmp.2012.537.543 (2012).
Dieng, S. I. M. et al. Evaluation of the antioxidant activity of hydro-ethanolic leaf and bark extracts of Piliostigma thonningii Schumach. Int. J. Biol. Chem. Sci. 11(2), 768–776. https://doi.org/10.4314/ijbcs.v11i2.19 (2017).
Ibewuike, J. C., Ogungbamila, F. O., Ogundaini, A. O., Okeke, I. N. & Bohlin, L. Antiinflammatory and antibacterial activities of c-methylflavonols from Piliostigma thonningii. Phytother. Res. 11(4), 281–284 (1997).
Akinpelua, D. A. & Obuotor, E. M. Antibacterial activity of Piliostigma thonningii stem bark. Fitoterapia 71, 442–443. https://doi.org/10.1016/S0367-326X(00)00136-2 (2000).
Egharevba, H. O. & Kunle, F. O. Preliminary phytochemical and proximate analysis of the leaves of Piliostigma thonningii (Schumach) Milne-Redhead. Ethnobot Leaflets 14(5), 570–577 (2010).
Ayenew, A., Tolera, A., Nurfeta, A. & Assefa, G. Utilization and nutritive value of Piliostigma thonningii as ruminant feed in North Western Ethiopia. Ethiop. J. Appl. Sci. Technol. 10(2), 1–10 (2019).
Zenda, M. & Rudolph, M. A Systematic review of agroecology strategies for adapting to climate change impacts on smallholder crop farmers’ livelihoods in South Africa. Climate 12(3), 33. https://doi.org/10.3390/cli12030033 (2024).
Munialo, S. et al. Systematic review of the agro-ecological, nutritional, and medicinal properties of the neglected and underutilized plant species Tylosema fassoglense. Sustainability 16(14), 6046. https://doi.org/10.3390/su16146046 (2024).
Brym, Z. T. & Reeve, J. R. Agroecological principles from a bibliographic analysis of the term agroecology. In Lichtfouse, E. (eds) Sustainable Agriculture Reviews. Sustainable Agriculture Reviews, Vol 19. Springer, Cham. https://doi.org/10.1007/978-3-319-26777-7_5 (2016).
Sinclair, F., Wezel, A., Mbow, C., Chomba, S., Robiglio, V. & Harrison, R. The contribution of agroecological approaches to realizing climate-resilient agriculture. Rotterdam and Washington, DC. Available online at www.gca.org (2019).
Kerr, R. B. et al. Can agroecology improve food security and nutrition?. A review. Glob. Food Secur. 29, 100540. https://doi.org/10.1016/j.gfs.2021.100540 (2021).
Reguera, M. et al. The impact of different agroecological conditions on the nutritional composition of quinoa seeds. Peer J. 6, e4442. https://doi.org/10.7717/peerj.444 (2018).
Okello, J., Okullo, J. B. L., Eilu, G., Nyeko, P. & Obua, J. Physicochemical composition of Tamarindus indica L. (Tamarind) Fruits in the agro-ecological zones of Uganda. Food Sci. Nutr. 6, 1179–1189. https://doi.org/10.1002/fsn3.627 (2018).
Gebregerges, T., Tessema, Z. K. & Birhane, E. Effect of exclosure ages on woody plant structure, diversity and regeneration potential in the western Tigray region of Ethiopia. J. For. Res. 29(3), 697–707. https://doi.org/10.1007/s11676-017-0512-6 (2018).
EIAR. New agroecological map of Ethiopia. Coordination of national agricultural research system, Ethiopia. Ethiopian Institute of Agricultural Research, Addis Ababa: EIAR. (2011).
Gebramlak, G. D., Abadi, N. & Hizikias, E. B. Socioeconomic contribution of Oxytenanthera abyssinica (A.Rich) Munro and determinants of growing in homestead agroforestry system in Northern Ethiopia. Ethnobot. Res. Appl. 14, 479–490. https://doi.org/10.17348/era.14.0.479-490 (2016).
Hernadez-Unzon, H. Y. & Laksminarayana, S. Biochemical changes during development and ripening of Tamarind Fruit (Tamarindus indica L.). Hort. Science 17(6), 940–942. https://doi.org/10.21273/HORTSCI.17.6.940 (1982).
Tadesse, D., Masresha, G., Lulekal, E. & Alemu, A. Ethnobotanical study of wild edible plants in Metema and Quara districts Northwestern Ethiopia. J. Ethnobiol. Ethnomed. 21, 7. https://doi.org/10.1186/s13002-025-00761-9 (2025).
AOAC. Official Methods of Analysis. 17th ed. Washington, DC, USA: Association of Official Analytical Chemists. (2000).
FAO, Food energy–methods of analysis and conversion factors. Report of a Technical Workshop. Food Nutrition 77 (2003).
Fadel, F. et al. Morphometric and physicochemical characteristics of carob pods in three geographical regions of Morocco. SN Appl. Sci. 2, 2173. https://doi.org/10.1007/s42452-020-03963-w (2020).
Datir, S., Tetali, P. & Kumatkar, P. Nutritional evaluation from Nesphostylis bracteata (Syn: Sphenostylis bracteata): potentially important underutilized wild legume of the Northern Western Ghats of India. Genet. Resour. Crop Evol. https://doi.org/10.1007/s10722-024-02101-4 (2024).
Girmay, H., Tewolde-Berhan, S., Hishe, H., Asfaw, Z., Morgan, R. & Alison, P. Use and management of tamarind (Tamarindus indica L., Fabaceae) local morphotypes by communities in Tigray, Northern Ethiopia. For. Trees Livelihoods 29(2), 81–98. https://doi.org/10.1080/14728028.2020.1737582 (2020).
Van den Bilcke, N., Alaerts, K., Ghaffaripour, S., Simbo, D. J. & Samson, R. Physico-chemical properties of tamarind (Tamarindus indica L.) fruits from Mali: selection of elite trees for domestication. Genet. Resour. Crop Evol. 61, 537–553. https://doi.org/10.1007/s10722-014-0080-y (2014)
Nasar‐Abbas, S. M., e‐Huma, Z., Vu, T. H., Khan, M. K., Esbenshade, H. & Jayasena, V. Carob kibble: A bioactive‐rich food ingredient. Compr. Rev. Food Sci. Food Saf. 15(1), 63–72. https://doi.org/10.1111/1541-4337.12177 (2016)
Pandey, D. K., Singh, S., Dubey, S. K., Mehra, T. S., Mounika, V., Dixit, S. & Sawargaonkar, G. Nutrient profiling of Lablab bean [(Lablab purpureus L.) Sweet] accessions from Northeast India: Diversity exploration leading to mitigation of nutritional vulnerability. https://doi.org/10.21203/rs.3.rs-2237030/v1 (2022).
Aina, A. I. et al. Morphological characterization and variability analysis of African yam bean (Sphenostylis stenocarpa Hochst ex. A. Rich) Harms Int. J. Plant Res. 10(3), 45–52. https://doi.org/10.5923/j.plant.20201003.01 (2020).
Shitta, N. S. et al. Morphological characterization and genotypic identity of African yam bean (Sphenostylis stenocarpa Hochst ex. A. Rich. Harms) germplasm from diverse ecological zones. Plant Genet. Res. 19(1), 58–66. https://doi.org/10.1017/S1479262121000095 (2021).
Ebifa Othieno, E., Kabasa, J. D., Nyeko, P., Nakimbugwe, D. & Mugisha, A. Nutritional potential of tamarind (Tamarindus indica L.) from semi-arid and sub humid zones of Uganda. J. Food Meas. Charact. 14, 1125–1134. https://doi.org/10.1007/s11694-019-00362-7 (2020).
Animashahun, R., Idowu, A., Alabi, O., Olawoye, S., Animashahun, A., Okeniyi, F. & Oluwafemi, P. Comparative study on proximate, phytochemicals and mineral components of different parts of Parkia biglobosa (pod, seed, and leaf). J. Xi’an Shiyou Univ. Nat. Sci. Ed. 20(02), 48–52. http://xisdxjxsu.asia (2024).
Vadivel, V. & Janardhanan, K. Nutritional and antinutritional characteristics of seven south Indian Wild legumes. Plant Foods Hum. Nutr. 60, 69–75. https://doi.org/10.1007/s11130-005-5102-y (2005).
Andualem, B. & Gessesse, A. Proximate composition, mineral content and antinutritional factors of Brebra (Millettia ferruginea) seed flour as well as physicochemical characterization of its seed oil. Springerplus 3, 298. https://doi.org/10.1186/2193-1801-3-298 (2014).
Varkekara, N. J. & Singh, N. I. Assessment of physical, nutritional and functional attributes of Parkia biglandulosa W & A: An underexplored semi-wild tree legume species of Parkia genera. Indian J. Tradit. Know. 19(4), 736–743. https://doi.org/10.56042/ijtk.v19i4.44516 (2020).
Altuntas, E. & Demirtola, H. Effect of moisture content on physical properties of some grain legume seeds. N. Z. J. Crop Hortic. Sci. 35(4), 423–433. https://doi.org/10.1080/01140670709510210 (2007).
Melesse, A., Steingass, H., Schollenberger, M., Holstein, J. & Rodehutscord, M. Nutrient compositions and in vitro methane production profiles of leaves and whole pods of twelve tropical multipurpose tree species cultivated in Ethiopia. Agroforest Syst 93, 135–147. https://doi.org/10.1007/s10457-017-0110-9 (2019).
Toumi, S. et al. Morphobiometric characterisation of carob tree pods cultivated in Algeria and evaluation of physicochemical, nutritional, and sensory properties of their powders. Proc. Latvian Acad. Sci. Section. B. 78(2), 153–163. https://doi.org/10.2478/prolas-2024-0023 (2024).
Kalpanadevi, V. & Mohan, V. R. Nutritional and anti-nutritional assessment of underutilized legume D. lablab var. vulgaris L.. Bangladesh J. Sci. Ind. Res. 48(2), 119–130 (2013).
Bakhtiar, Z., Hassandokht, M., Naghavi, M. R. & Mirjalili, M. H. Variability in proximate composition, phytochemical traits and antioxidant properties of Iranian agro-ecotypic populations of fenugreek (Trigonella foenum-graecum L.). Sci. Rep. 14(1), 87. https://doi.org/10.1038/s41598-023-50699-9 (2024).
Ouedraogo, S., Sanou, L., Savadogo, P. & Kabore-Zoungrana, C. Y. Comparative nutritional evaluation of the two leguminous fodder trees Prosopis africana and Piliostigma thonningii: Effects of different levels of pod-based supplementation on the growth performance of Djallonke sheep. Int. J. Biol. Chem. Sci. 16(5), 1831–1846. https://doi.org/10.4314/ijbcs.v16i5.2 (2022).
Akpata, M. I. & Miachi, O. E. Proximate composition and selected functional properties of Detarium microcarpum. Plant Foods Hum. Nutr. 56(4), 297–302. https://doi.org/10.1023/A:1011836332105 (2001).
Tharanathan, R. N. & Mahadevamma, S. Grain legumes-a boon to human nutrition. Trends Food Sci. Technol. 14(12), 507–518. https://doi.org/10.1016/j.tifs.2003.07.002 (2003).
Yaméogo, G., Yelemou, B. & Traoré, D. Farmer’s practices and perception in agroforestry park creating in Vipalogo area (Burkina Faso). Biotechnol. Agron. Soc. Environ. 9(4), 241–248 (2005).
Semba, R. D., Ramsing, R., Rahman, N., Kraemer, K. & Bloem, M. W. Legumes as a sustainable source of protein in human diets. Glob. Food Secur. 28, 100520. https://doi.org/10.1016/j.gfs.2021.100520 (2021).
Weber, C. W., Kohlhepp, P., Idouraine, A. & Osman, M. Chemical and nutritional composition of tree legume seeds and pods from Southwestern United States and Sonora Mexico. Ecol. Food. Nutr. 37, 57–72. https://doi.org/10.1080/03670244.1998.9991537 (1998).
N. A. P. Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein, and amino acids (2005). http://www.nal.usda.gov/fnic/DRI/DRI_Energ y/energy_full_repor t.pdf (2005).
Termote, C. et al. Nutrient composition of Parkia biglobosa pulp, raw and fermented seeds: A systematic review. Crit. Rev. Food Sci. Nutr. 62(1), 119–144. https://doi.org/10.1080/10408398.2020.1813072 (2022).
Anavi, S. Nutrition and Health-the Importance of Potassium. International Potash Institute (IPI), (Switzerland, 2013). https://doi.org/10.3235/978-3-905887-08-2.
Pajarillo, E. A. B., Lee, E. & Kang, D. K. Trace metals and animal health: Interplay of the gut microbiota with iron, manganese, zinc, and copper. Anim. Nutr. 7(3), 750–761. https://doi.org/10.1016/j.aninu.2021.03.005 (2021).
Abbaspour, N., Hurrell, R. & Kelishadi, R. Review on iron and its importance for human health. J. Res. Med. Sci. 19(2), 164–174 (2014).
Towett, E. K. et al. Total elemental composition of soils in Sub-Saharan Africa and relationship with soil forming factors. Geoderma Reg. 5, 157–168 (2015).
Lambers, H. & Oliveira, R. S. Mineral nutrition. In Plant Physiological Ecology. (Springer, Cham, 2019). https://doi.org/10.1007/978-3-030-29639-1_9.
Butler, B. M. et al. Mineral–nutrient relationships in African soils assessed using cluster analysis of x-ray powder diffraction patterns and compositional methods. Geoderma 375, 114474. https://doi.org/10.1016/j.geoderma.2020.114474 (2020).
Acknowledgements
The authors are most grateful for financial support from the MU-NMBU Phase IV Project through Mekelle University and the McKnight Foundation’s Collaborative Crop Research Program. We extend our gratitude to Dr. Zenebe Girmay, the field assistants, the technical and laboratory staff, and the farmers whose fields we visited during data collection. Their invaluable contributions were essential to compiling this manuscript. Additionally, we acknowledge the Institute of International Education-Scholars Rescue Fund (IIE-SRF), and Nord University, Faculty of Bioscience and Aquaculture (FBA), and the NORGLOBAL 2 project “Towards a climate-smart policy and management framework for conservation and use of dry forest ecosystem services and resources in Ethiopia [grant number: 303600]” for supporting the research stay of Emiru Birhane at NMBU.
Funding
The research fund for this study was obtained from the MU-HU-NMBU collaborative project through Mekelle University and the Ethiopian Ministry of Education.
Author information
Authors and Affiliations
Contributions
T.G. data collection, investigation, data curation, methodology, formal analysis, writing—original draft, writing—review and editing. M.H. supervised, conceptualization, review, and editing the original draft and manuscript. S.T.B. conceptualization, conceived and designed the experiments, contributed materials and training, and review and editing. E.B. supervised, conceptualization, writing—original draft, review and editing the manuscript critically.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The study was conducted with formal approval from the Vice President for Research and Technology Transfer at Mekelle University. An official permission letter from the department of land resources management and environmental protection was submitted to the administrative offices of Tselemti district, and the Tselemti Agricultural and Rural Development Office. Verbal consent was obtained from all relevant authorities and farmers in the district. This was done after the main objectives of the study were clearly explained with the assistance of local language translators. No endangered or threatened species were collected or included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gebre, T., Haile, M., Tewolde-Berhan, S. et al. Morphological and nutritional composition of Bauhinia thonningii pods and seeds in Northern Ethiopia. Sci Rep 16, 2359 (2026). https://doi.org/10.1038/s41598-025-32054-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-32054-2