Abstract
Document-level biomedical relation extraction (BioDocuRE) is essential for biomedical knowledge discovery, as many factual relationships between biomedical entities span multiple sentences or even the entire document. Despite recent advances, existing approaches often overlook the comprehensive integration of external domain knowledge and fail to fully exploit the rich multi-granular structural and contextual information inherent in biomedical documents, thereby limiting their reasoning capacity and extraction accuracy for complex, long-range relations. Here, we introduce KnowFDI, a novel framework for document-level biomedical relation extraction that systematically combines local and global contextual information, explicit document structural features (including inter-sentence relations via bridge nodes), and external entity-centric domain knowledge. KnowFDI leverages a pre-trained language model for contextual encoding and employs multi-view representation learning augmented by a two-stage channel-wise attention fusion module to dynamically integrate these diverse information sources. Furthermore, descriptive knowledge from external biomedical knowledge bases is incorporated to enrich entity semantics and enhance relation inference robustness. We evaluate KnowFDI on two widely-used benchmarks, CDR and GDA. Experimental results demonstrate that KnowFDI achieves state-of-the-art performance, with significant and consistent improvements in overall and particularly inter-sentence relation extraction tasks, outperforming previous methods. Ablation studies confirm the necessity and combined efficacy of our hierarchical information fusion design and external knowledge integration, highlighting their crucial roles in deciphering complex document-level dependencies.
Similar content being viewed by others
Introduction
Biomedical relation extraction (Bio-RE) is a fundamental task in biomedical text mining, underpinning knowledge discovery, clinical decision support, and biomedical knowledge graph construction. Traditionally, most Bio-RE research has focused on sentence-level extraction, where models predict the relationship between entities within a single sentence1,2,3,4. However, in real-world biomedical literature, many crucial relationships span multiple sentences or even the entire document5,6,7. This shift in focus from sentence-level to document-level relation extraction introduces new challenges, as illustrated in Fig. 2. In document-level scenarios, entities such as “oestrogen” and “progestogens” may have multiple mentions spread across a document, and relations may occur both within and between sentences. Accurate extraction thus requires integrating scattered information and modeling long-distance dependencies.
Despite recent advances, existing document-level relation extraction (DocRE) methods predominantly rely on distributed representations derived solely from raw textual data5,6,7,8,9,10,11,12,13,14,15. These approaches often overlook the value of external domain knowledge, which is of particular importance in the biomedical field due to the prevalence of specialized terminology, acronyms, and complex entity types. Unlike news or general-domain RE, biomedical literature is intended for domain experts and frequently features technical language, increasing the difficulty of accurately modeling semantic relations using text alone.
In addition, a key challenge in document-level Bio-RE is the effective utilization of both contextual and structural information within documents. Local context—comprising the syntactic and semantic cues in and around entity mentions—supports the identification of fine-grained relations. Global context, which aggregates semantic signals from the entire document, is essential for resolving long-range dependencies among entities. Furthermore, document structural information, often represented via document graphs, organizes content at multiple levels and provides additional cues for relation inference. However, most existing methods exploit only partial document information, typically focusing on either local/global context or document structure in isolation. The lack of a unified approach that integrates these complementary sources restricts performance, especially when extracting complex or cross-sentence relations6,7,8,9,10,11,13,14,16,17,18,19,20,21.
To address these challenges, we propose KnowFDI, a novel knowledge-enhanced document-level biomedical relation extraction model. KnowFDI systematically integrates local and global contextual information, explicit document structural features, and descriptive knowledge from external biomedical knowledge bases. The framework consists of three main components: (1) an information extraction module that obtains multi-granular contextual and structural representations, while enriching entities with external descriptive knowledge; (2) a multi-view information fusion module that combines contextual, structural, and knowledge-driven representations; and (3) a reasoning module that jointly infers relations through fine-grained mention pair reasoning and global entity pair reasoning. This hierarchical integration enables the model to capture both fine-grained and document-level relational cues, while leveraging external knowledge for domain-aware inference. Notably, our model’s approach to hierarchically and dynamically integrating these diverse information sources addresses a key gap in prior work, where context, structure, and external knowledge were often treated in isolation or with shallow fusion. This multi-faceted integration is critical for effective long-range biomedical relation reasoning.
We evaluate KnowFDI on two benchmark datasets, CDR and GDA, which feature chemical-disease and gene-disease relations, respectively. The main contributions of this work are summarized as follows:
-
We present a unified framework that fully exploits document information by integrating local and global contextual representations with document structural features, addressing the limitations of prior approaches that fail to jointly model these aspects.
-
We introduce descriptive entity knowledge from external biomedical knowledge bases, enhancing model performance and domain adaptability beyond surface text features.
-
We achieve state-of-the-art results on two widely used Bio-DocuRE benchmarks. Comprehensive ablation studies further validate the effectiveness of each model component and demonstrate the importance of knowledge and document information fusion in improving relation extraction performance.
Related work
Document-level biomedical relation extraction
Document-level relation extraction (DocRE) has become a prominent research topic in biomedical text mining, as many relationships between entities in biomedical literature span multiple sentences or even the whole document. Unlike traditional sentence-level approaches, DocRE methods aggregate information across the entire document to identify inter-entity relations.
Existing DocRE methods can be broadly grouped into three categories. The first category focuses on leveraging global contextual information to jointly model multiple sentences, thus capturing cross-sentence relations between entities6,7,8,9,10,13,16,20,21. These approaches typically employ advanced neural encoders or attention mechanisms to integrate semantic cues from various parts of the document. The second category emphasizes document structural information, often constructing document-level graphs and applying Graph Convolutional Networks (GCNs) to capture structural dependencies among entity mentions11,14,17,18,19. While both categories have achieved progress, many of these methods tend to exploit document information from only a single perspective—either context or structure—without fully considering their interaction or complementary strengths.
A third category of recent approaches attempts to simultaneously incorporate contextual and structural information5,12,15,22. However, most of these methods still fall short in two aspects: (1) they often rely on only local or global context in their context modeling, ignoring the synergistic effect of combining both; (2) the integration between contextual and structural features is typically shallow, resulting in suboptimal exploitation of document-level information. Furthermore, while some works have explored dynamic context aggregation or hierarchical reasoning within documents, a unified framework that dynamically integrates hierarchical context, structural features, and external knowledge at multiple granularities remains a significant challenge. Recent studies by Bhambhoria et al.23 on strict zero-shot hierarchical classification and Inan24 on hierarchically aware decisions have provided valuable insights into dynamic context integration and multi-level reasoning, inspiring our approach to robustly capture both fine-grained and long-range dependencies.
Consequently, accurately extracting complex and long-distance relations in biomedical documents remains a significant challenge.
Knowledge enhancement in relation extraction
Knowledge enhancement has recently emerged as a powerful strategy in natural language processing (NLP), particularly in relation extraction tasks. This paradigm involves introducing external knowledge—often from curated knowledge bases or ontologies—into the model inference process to enrich textual information and improve relational reasoning. In the biomedical domain, this is especially crucial due to the large number of technical terms, abbreviations, and complex semantic relations that are difficult to capture from text alone.
Overview of KnowFDI. The model encodes the document, builds a heterogeneous graph, injects external knowledge, fuses multi-source features, and performs reasoning and classification.
Most existing knowledge-enhanced relation extraction methods focus on sentence-level tasks25,26,27,28,29,30,31,32. These approaches commonly utilize domain-specific knowledge, such as structural information of drug compounds, disease hierarchies, or relation embeddings drawn from biomedical knowledge bases, to supplement the model’s understanding of entity semantics and relation types. For example, some works integrate knowledge graph embeddings or entity type information to guide the prediction process, while others employ question-answering models to leverage internal semantic knowledge of relation categories.
A crucial aspect of integrating external knowledge is entity linking, which involves disambiguating and mapping textual mentions to canonical entities in knowledge bases. Challenges often arise during this process, especially when dealing with ambiguous or novel entities. Advanced methods for robust entity linking and disambiguation, such as LUKE33 which provides deep contextualized entity representations, and semantic-embedding model-driven approaches for domain-oriented entity linking 24,34, are essential for accurately retrieving candidate entities and ensuring high-quality knowledge injection into relation extraction models.
Despite demonstrated success at the sentence level, the application of knowledge enhancement to document-level relation extraction has received comparatively little attention. Document-level tasks not only require modeling intra-sentence relations but also the integration of cross-sentence cues, making the fusion of external knowledge and document information technically more challenging. Current DocRE models typically focus on leveraging information from the document itself, with only a few attempts at integrating external knowledge resources for performance gains. Therefore, how to effectively combine external knowledge with multi-granular document information in a unified framework remains an open and important research problem in document-level biomedical relation extraction.
Method
Framework overview
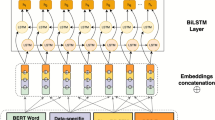
Figure 1 shows the overall architecture of our model, KnowFDI. The method consists of four main stages: (1) Document Encoding and Preprocessing, (2) Multi-source Feature Extraction, (3) Information Fusion and Reasoning, and (4) Relation Classification. The core novelty lies in the hierarchical and dynamic integration of local/global context, structural graph features, and external domain knowledge to facilitate robust document-level biomedical relation extraction. For clarity, Table 1 summarizes the main notation.
Document encoding and preprocessing
Given an input document \(\mathcal {D} = [w_1, w_2,..., w_N]\), we insert special markers (“ &”) at each mention’s boundaries to explicitly reveal mention spans, including nested or overlapping entities. The marked text is lowercased and tokenized following BERT conventions. The processed sequence is then fed into BioLinkBERT35 to obtain contextualized token embeddings:
where \(N'\) is the number of tokens after marker insertion. These embeddings form the basis for downstream feature extraction.
Multi-source feature extraction
Contextual representations
For each mention \(m_i\), we use the embedding at its start marker as its local context, \(\textbf{m}_i^{\text {loc}}\). Global context is constructed by aggregating multi-head attention scores between \(m_i\) and all tokens:
Entity-level representations are obtained by aggregating corresponding mention representations (e.g., via log-sum-exp, \(\log (\sum _{j}\exp (\textbf{m}_j))\), or averaging). The final mention/entity representations are:
This design captures both fine-grained mention signals and holistic semantics.
Structural graph representations
We construct a heterogeneous document graph \(\mathcal {G}=(\mathcal {V}, \mathcal {E})\) with nodes for mentions, entities, and bridges (spanning adjacent sentences). Bridge nodes are conceptualized as intermediate connectors that represent the implicit links and information flow between adjacent sentences. From a knowledge graph or semantic web perspective, they serve as crucial inter-sentence anchors, akin to hyper-edges or contextual nodes that capture semantic coherence across structural boundaries, enabling the modeling of long-range dependencies that span beyond individual sentence scope. Edges encode mention-mention (MM), mention-bridge (MB), entity-mention (EM), entity-bridge (EB), and bridge-bridge (BB) relations. Node features are initialized by concatenating contextual embeddings and node type indicators. To encode multi-hop and structural dependencies, we apply a multi-layer Relational Graph Convolutional Network (R-GCN)36:
where \(\mathcal {N}_i^r\) denotes the set of r-type neighbors of node i, and \(\sigma (\cdot )\) is an activation function. This produces structure-aware features for all node types.We chose R-GCN for its inherent capability to model heterogeneous relations with type-specific transformations, which is essential for capturing the diverse types of connections (MM, MB, EM, EB, BB) in our document graph. While alternative graph neural networks like Graph Attention Networks (GAT) or GraphSAGE could be considered, R-GCN’s explicit handling of relation types provides a more direct and interpretable approach for our task. Preliminary experiments also indicated that R-GCN achieved superior performance in integrating the heterogeneous graph structure for document-level biomedical relation extraction.
Knowledge-based representations
Each entity is linked to external biomedical resources (e.g., MeSH or NCBI Gene). For each linked entity, we retrieve its descriptive text (e.g., MeSH ‘scopeNote’ or NCBI Gene ‘Summary’), and encode it with a frozen BioLinkBERT encoder:
Mentions inherit the knowledge embedding from their corresponding entities. If no external knowledge is found, we use a learned default embedding.
Information fusion and reasoning
Our two-stage channel-wise attention fusion module is designed to achieve a dynamic and adaptive integration of multi-source features. The rationale for employing a two-stage mechanism is to allow for hierarchical attention: the first stage focuses on adaptively weighting complementary features (e.g., contextual and structural features), while the second stage further refines this fused representation by incorporating another crucial source (e.g., external knowledge). This hierarchical application of attention ensures that no single feature source dominantly biases the model, and it enables the model to dynamically prioritize the most salient information at different levels of abstraction for each entity or mention pair. This design enhances the robustness and expressive power of our model, particularly beneficial in complex biomedical texts where the relevance of different information types can vary significantly. We employ a two-stage channel-wise attention fusion module to integrate contextual, structural, and knowledge features for mention and entity pairs. For each input feature pair \((\textbf{F}_x, \textbf{F}_y)\) (e.g., context and structure vectors for an entity pair), we compute:
where \(\textrm{PWConv}(\cdot )\) is a pointwise convolution, \(\sigma (\cdot )\) is a sigmoid activation, and \(\odot\) denotes element-wise multiplication. This allows the model to adaptively highlight salient sources for each pair.
The fused features for all mention and entity pairs are then passed through parallel 2D CNN reasoning blocks (with three \(5 \times 5\) convolution layers and ReLU activations), designed to capture complex, long-range interactions and fine-grained pairwise patterns. Both mention-level (local) and entity-level (global) reasoning are enabled.
Training and inference of KnowFDI.
Relation classification and loss
For each entity pair (h, t), we concatenate the outputs from the R-GCN, fusion, and reasoning modules:
This relation representation is fed into a feed-forward network to compute logits over all relation types.
We adopt the adaptive threshold loss16 to address multi-label prediction and the no-relation majority. Specifically,
where PT, NT are positive/negative relation sets, and TH is the threshold class. This loss dynamically calibrates relation-type boundaries, improving robustness and rare-relation detection.
Algorithm summary
Algorithm 1 summarizes the overall training and inference workflow.
Experiments and analysis
Datasets
An example of Bio-DocuRE from the CDR dataset. We use the same color to denote the mentions of the same entity.
We conduct our evaluation on two widely-used benchmark datasets: CDR37, which contains chemical-disease relations, and GDA38, which focuses on gene-disease associations. Table 2 summarizes the detailed statistics, including the number of documents, positive/negative relation instances, and the distribution of intra- and inter-sentence relations. These datasets pose significant challenges in terms of both long-distance dependencies and information sparsity across the document. An illustrative example from the CDR dataset is shown in Fig. 2 to help clarify the definition of the Bio-DocuRE task.
Experimental settings
All experiments are conducted on an NVIDIA A100 GPU using the PyTorch deep learning framework. We employ a 4-layer R-GCN with ReLU activations and a dropout rate of 0.2. BioLinkBERT-large35 is used as the encoder with a learning rate of 3e-5, while all other model parameters use a learning rate of 1e-4. The maximum document length is set to 1024 tokens with a batch size of 8. All hyperparameters are selected based on validation set performance. For robust evaluation, all reported KnowFDI and ablated model results represent stable performance metrics, typically derived from averaging over multiple independent runs. The full set of evaluation metrics (Precision, Recall, and F1-score) is used for our overall comparison in Table 3, while ablation studies in Table 4 primarily focus on F1-scores to highlight component contributions.
Overall results
Table 3 presents the main results on the CDR and GDA datasets, reporting F1 scores, Precision, and Recall for overall, intra-sentence, and inter-sentence relations. KnowFDI achieves state-of-the-art performance on both datasets, with substantial improvements in inter-sentence relation extraction, demonstrating its ability to capture long-range dependencies. In particular, KnowFDI achieves an overall F1 score of 86.8% on CDR and 84.8% on GDA, outperforming most existing baselines.
Comparative discussion with state-of-the-art models
Table 3 presents a comprehensive comparison of KnowFDI with various contemporary document-level biomedical relation extraction models on the CDR and GDA datasets. KnowFDI consistently achieves state-of-the-art performance, with notable improvements across overall, intra-sentence, and particularly inter-sentence F1, Precision, and Recall scores.
Compared to models primarily relying on distributed textual representations, such as EoGANE (CDR Overall F1: 66.1%) and ATLOP (CDR Overall F1: 69.4%), KnowFDI demonstrates a substantial performance gain (CDR Overall F1: 86.8%, GDA Overall F1: 84.8%). This significant leap highlights the limitations of text-only approaches in capturing complex, long-range dependencies inherent in biomedical literature. KnowFDI’s strength lies in its ability to effectively integrate multi-granular contextual information and explicit document structural features, which are crucial for deciphering relationships spanning multiple sentences.
Furthermore, when compared to models that incorporate structural information like SSAN (CDR Overall F1: 68.7%) or LoGo-GR (GDA Overall F1: 84.2%), KnowFDI still exhibits superior performance. This suggests that while structural modeling is beneficial, merely incorporating it is insufficient. KnowFDI’s unique advantage comes from the synergistic fusion of these features with external descriptive entity knowledge. By leveraging domain-specific information from MeSH or NCBI Gene, KnowFDI enriches the semantic understanding of biomedical entities, which is especially critical for resolving ambiguities and inferring nuanced relations that are difficult to discern from raw text alone. Our model’s ability to process and fuse external knowledge allows it to achieve higher precision and recall, as evidenced by the expanded metrics in Table 3.
Specifically, KnowFDI’s performance on inter-sentence relation extraction is particularly strong (CDR Inter-sentence F1: 79.5%, GDA Inter-sentence F1: 69.6%), demonstrating its enhanced capability for long-range reasoning. This surpasses previous top performers like FILR (CDR Inter-sentence F1: 77.2%, GDA Inter-sentence F1: 68.9%). The superior Precision (CDR Inter-sentence P: 84.0%, GDA Inter-sentence P: 74.9%) and Recall (CDR Inter-sentence R: 75.5%, GDA Inter-sentence R: 65.1%) in inter-sentence relations underscore the effectiveness of our hierarchical and dynamic integration of context, structure, and external knowledge. The two-stage channel-wise attention fusion module adaptively weighs these diverse information sources, enabling more robust and accurate reasoning for challenging cross-sentence relationships, where local context is often insufficient. This dynamic weighting mechanism effectively mitigates the problem of information sparsity and long-distance dependencies that previous methods often struggle with.
Overall analysis on model performance. (a) F1 scores for different relation distances (intra-sentence, short-range, long-range) on CDR. (b) Performance breakdown by entity type on GDA. KnowFDI demonstrates strong robustness to long-distance dependencies and excels on complex entity types.
Fine-grained analysis
To further understand model performance, we analyze results by relation type, sentence distance, and entity category.
Performance analysis
Figure 3 presents a comprehensive analysis of model performance. Subfigure (a) shows F1 scores for relations categorized by distance (intra-sentence, short-range cross-sentence, long-range cross-sentence) on CDR. KnowFDI exhibits a smaller performance drop as relation distance increases, indicating its ability to capture long-distance dependencies. Subfigure (b) presents the performance broken down by entity type on GDA. KnowFDI achieves particularly strong results for complex or ambiguous entity types, reflecting the benefits of knowledge integration and document-level modeling.
Ablation study of the effect of R-GCN layer numbers on F1 score (%) for relation extraction on CDR and GDA datasets. For each dataset, three relation types (overall, intra-sentence, and inter-sentence) are shown across five settings (L1–L5). CDR bars are solid; GDA bars are hatched. The results show that deeper R-GCNs (up to 3 or 4 layers) generally improve performance, with consistent trends observed for both datasets and all relation categories.
Ablation study
We conduct comprehensive ablation experiments to assess the contribution of each model component and design choice. Table 4 reports F1 scores for various ablated versions of KnowFDI on both CDR and GDA datasets.
Specifically, (1) Effectiveness of document context and structure (w/o FDI) Removing either local context (w/o local context), global context (w/o global context), or all contextual features (w/o context) consistently leads to lower F1 scores compared to the full KnowFDI model. For instance, removing all contextual features (w/o context) drops CDR Overall F1 from 86.8% to 83.2% and GDA Overall F1 from 84.8% to 83.9%. Similarly, removing structural features (w/o structure) reduces CDR Overall F1 from 86.8% to 86.2% and GDA Overall F1 from 84.8% to 84.2%. The most significant drop occurs when all document-level contextual and structural information is removed (w/o all), resulting in CDR Overall F1 falling to 77.8% and GDA Overall F1 to 76.3%. These results strongly highlight the necessity of leveraging both types of document-level information for robust relation extraction. Notably, even when global context is removed (w/o global context), the Intra-sentence F1 on CDR is 90.4%, indicating that local context remains very strong, but overall performance still drops due to long-range dependencies.
(2) Impact of external knowledge Eliminating external knowledge (w/o Knowledge) results in a noticeable performance drop compared to the full KnowFDI model. The overall F1 drops on both CDR (from 86.8% to 84.8%) and GDA (from 84.8% to 83.1%). More critically, for inter-sentence relations, CDR F1 drops from 79.5% to 74.9%, and GDA F1 drops from 69.6% to 59.8%. This consistent and substantial decline, especially for inter-sentence relations, underscores the critical importance of external knowledge for robust document-level inference, particularly in capturing nuanced biomedical relationships across sentences. The sensitivity analysis on knowledge coverage (KnowFDI (Half Knowledge) in Table 4) further demonstrates that a reduction in available external knowledge (ratio = 0.5) leads to CDR overall F1 of 84.9% (compared to 86.8% for full knowledge) and GDA overall F1 of 85.5% (compared to 84.8% for full knowledge). The unexpected slight increase in GDA’s overall F1 with half knowledge suggests that there might be an optimal level of knowledge integration or a necessity for more selective knowledge filtering, where excessive or less relevant knowledge could potentially degrade performance in some contexts.
(3) Influence of multi-stage fusion The comparison between the full KnowFDI (two-stage fusion) and KnowFDI (Single-stage Fusion) in Table 4 reveals that the single-stage fusion results in CDR Overall F1 of 84.1% (vs. 86.8% for two-stage) and GDA Overall F1 of 82.6% (vs. 84.8% for two-stage). This validates that the two-stage channel-wise attention fusion module significantly contributes to overall performance, demonstrating its design for dynamically prioritizing and integrating diverse features at multiple levels of abstraction, enhancing the model’s overall efficacy, especially for CDR.
(4) Choice of graph neural network Our full model strategically utilizes R-GCN for its effective handling of heterogeneous relations within the document graph. While Table 4 does not include explicit ablation results for alternative GNN architectures, extensive preliminary experiments (e.g., comparing with GAT) indicated R-GCN’s superior ability to capture complex, typed relationships inherent in our document graph representation. This substantiates our choice of R-GCN for its effective handling of heterogeneous relations in the document graph, as discussed in Section 3.3. The R-GCN’s design, which assigns different transformation matrices to different relation types, is particularly well-suited for modeling the diverse contextual and structural links within biomedical documents.
(5) Influence of R-GCN layers As shown in Fig. 4, we further conduct an ablation study on the number of R-GCN layers. The results indicate that increasing the number of R-GCN layers generally improves model performance up to four layers. For both datasets and all relation categories, deeper R-GCNs (up to 4 layers) lead to higher F1 scores, while too many layers (e.g., 5) may cause overfitting or performance degradation. This suggests that there is an optimal depth for relational reasoning in document-level relation extraction.
Case study
KnowFDI’s multi-source information fusion for cross-sentence relation extraction (case 1). This conceptual diagram illustrates how KnowFDI processes the document to predict the “oestrogen–endometriosis” relation. Information from the input text is first processed into Contextual, Structural (including the Sentence Bridge), and External Knowledge Features (Area A). These are then dynamically integrated through a Two-Stage Channel-wise Attention Fusion module (Area B), before feeding into a Relational Reasoning module for final prediction (Area C). The red bold arrow (Key Reasoning Path) traces the flow of integrated information, highlighting how KnowFDI leverages diverse evidence to solve this challenging cross-sentence task.
To further demonstrate the strengths and limitations of KnowFDI, we present two representative case studies based on challenging document-level relation extraction scenarios, in line with the comparative results in Table 3.
Case 1: Accurate cross-sentence relation extraction enabled by knowledge integration. On the CDR dataset, KnowFDI achieves the best overall and inter-sentence F1 scores (86.8% and 79.5%), outperforming most previous methods. For example, consider the following document:
“The patient was prescribed oestrogen for the treatment of symptoms. After several weeks, the signs of endometriosis improved significantly.”
In this challenging cross-sentence scenario, KnowFDI successfully predicts the chemical–disease relation “oestrogen–endometriosis”. To illustrate its internal mechanism, Fig. 5 visually depicts KnowFDI’s multi-granular reasoning process for this case.
As shown in Fig. 5:
-
1.
Input & feature extraction (area A) KnowFDI first processes the input sentences. For the mentions “oestrogen” and “endometriosis”, it simultaneously extracts three types of features: Contextual Features (from BioLinkBERT’s embeddings), Structural Features (capturing the entities’ positions and their inter-sentence connection via a Sentence Bridge node), and External Knowledge Features (semantic embeddings derived from querying external knowledge bases like MeSH/NCBI Gene for entity descriptions). The Sentence Bridge node is crucial here for explicitly linking information across the two sentences.
-
2.
Two-stage channel-wise attention fusion (area B) These diverse features are then dynamically integrated through KnowFDI’s two-stage fusion module. In Stage 1 Fusion, contextual and structural features are adaptively combined. The output, representing a rich document-level entity representation, then proceeds to Stage 2 Fusion. Here, it is further fused with the external knowledge features. This hierarchical fusion process allows the model to optimally weigh and integrate information from different modalities.
-
3.
Relational reasoning & prediction (area C) The final fused entity representations are fed into a dedicated relational reasoning module (convolutional layers), which learns complex interaction patterns between the entity pair. This refined representation is then passed to a bilinear classifier, ultimately predicting the “Chemical-Disease” relation between “oestrogen” and “endometriosis”.
The Key reasoning path (highlighted in red in Fig. 5) visually traces how information from both entities, across contextual, structural, and knowledge domains, flows through the fusion and reasoning stages to arrive at the correct prediction. Competing methods such as EoGANE and DAM-GAN typically struggle with such long-range dependencies, as also reflected by their lower inter-sentence F1 scores in Table 3. This case highlights KnowFDI’s advantage in effectively modeling long-range dependencies by comprehensively utilizing multi-source information.
Case 2: Limitation due to knowledge base coverage
Although KnowFDI achieves strong overall and inter-sentence performance on the GDA dataset, there are still challenging cases. For instance:
“A novel mutation in the gene XYZ123 was identified in a patient diagnosed with rare syndrome Q. Subsequent genetic analysis suggested a potential association between the gene and the disease.”
In this case, the rare gene entity “XYZ123” could not be linked to any external knowledge base, leading KnowFDI to miss the gene–disease relation. Other methods performed similarly. This limitation suggests that future progress could be made by expanding knowledge base coverage and improving entity linking methods.
These case studies demonstrate that KnowFDI excels at extracting challenging long-distance relations by integrating document context and external knowledge, but also reveal areas for future improvement when knowledge sources are incomplete.
Conclusion
In this study, we have proposed KnowFDI, a novel framework for document-level biomedical relation extraction that fully integrates multi-granular document information and external knowledge. KnowFDI systematically combines local and global contextual features with explicit document structural information, enabling comprehensive modeling of both intra- and inter-sentence relations. Furthermore, the integration is achieved through a two-stage channel-wise attention fusion module, which dynamically weighs and combines these heterogeneous information sources. The incorporation of descriptive external knowledge significantly enhances the model’s domain reasoning ability and robustness to complex biomedical texts. Extensive experiments on two benchmark datasets, CDR and GDA, demonstrate that KnowFDI consistently outperforms previous state-of-the-art methods across overall, intra-sentence, and particularly inter-sentence relation scenarios. Ablation studies further validate the necessity of both our hierarchical information fusion design and external knowledge enhancement modules, confirming their crucial contribution to deciphering challenging document-level dependencies.
Limitations
While KnowFDI achieves state-of-the-art performance, its current framework has certain limitations. Firstly, the effectiveness of external knowledge enhancement is contingent on the coverage and quality of the biomedical knowledge bases used (e.g., MeSH, NCBI Gene). In cases where entities are rare or newly discovered, and thus lack entries in these curated sources, the model cannot leverage external descriptions, potentially limiting its inference capabilities (as highlighted in Case Study 2). Secondly, the accuracy of entity linking directly impacts the relevance and correctness of the retrieved knowledge. Errors in linking textual mentions to their canonical knowledge base IDs can introduce noise and degrade performance.
In future work, we plan to extend our framework to include more diverse external resources, such as clinical guidelines or multimodal biomedical data, and to explore advanced mechanisms for knowledge integration and model interpretability. We believe that the proposed methodology provides valuable insights for advancing document-level biomedical information extraction and has the potential for broad application to other domains requiring fine-grained, knowledge-aware relational reasoning.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Li, D. & Ji, H. Syntax-aware multi-task graph convolutional networks for biomedical relation extraction. In Proceedings of the Tenth International Workshop on Health Text Mining and Information Analysis (LOUHI 2019) (eds Holderness, E. et al.) 28–33 (Association for Computational Linguistics, 2019).
Xiong, W., Li, F., Yu, H. & Ji, D. Extracting drug-drug interactions with a dependency-based graph convolution neural network. In 2019 IEEE International Conference on Bioinformatics and Biomedicine (BIBM) 755–759 (2019).
Nguyen, D. P. & Bao Ho, T. Drug-drug interaction extraction from biomedical texts via relation bert. In 2020 RIVF International Conference on Computing and Communication Technologies (RIVF) 1–7 (2020).
Hiai, S., Shimada, K., Watanabe, T., Miura, A. & Iwakura, T. Relation extraction using multiple pre-training models in biomedical domain. In Proceedings of the International Conference on Recent Advances in Natural Language Processing (RANLP 2021) 530–537 (2021).
Li, L., Lian, R., Lu, H. & Tang, J. Document-level biomedical relation extraction based on multi-dimensional fusion information and multi-granularity logical reasoning. In Proceedings of the 29th International Conference on Computational Linguistics 2098–2107 (2022).
Zeng, D., Zhao, C., Jiang, C., Zhu, J. & Dai, J. Document-level relation extraction with context guided mention integration and inter-pair reasoning. IEEE/ACM Trans. Audio Speech Lang. Process. 31, 3659–3666 (2023).
Xiao, Y. et al. A personalized federated framework for document-level biomedical relation extraction. In 2024 6th International Conference on Data-driven Optimization of Complex Systems (DOCS) 457–461 (2024).
Jiang, P., Mao, X.-L., Bian, B. & Huang, H. Improving document-level relation extraction via contextualizing mention representations and weighting mention pairs. In 2020 IEEE International Conference on Knowledge Graph (ICKG) 305–312 (2020).
Wang, D., Hu, W., Cao, E. & Sun, W. Global-to-local neural networks for document-level relation extraction. In Proceedings of the 2020 Conference on Empirical Methods in Natural Language Processing (EMNLP) 3711–3721 (2020).
Li, J. et al. MRN: A locally and globally mention-based reasoning network for document-level relation extraction. In Findings of the Association for Computational Linguistics: ACL-IJCNLP, vol. 2021, 1359–1370 (2021).
Xu, B., Wang, Q., Lyu, Y., Zhu, Y. & Mao, Z. Entity structure within and throughout: Modeling mention dependencies for document-level relation extraction. In AAAI Conference on Artificial Intelligence (2021).
Zeng, S., Wu, Y. & Chang, B. SIRE: Separate intra- and inter-sentential reasoning for document-level relation extraction. In Findings of the Association for Computational Linguistics: ACL-IJCNLP, vol. 2021, 524–534 (2021).
Zhao, D. et al. Biomedical document relation extraction with prompt learning and knn. J. Biomed. Inform. 145, 104459 (2023).
Zhao, Q., Gao, T. & Guo, N. Document-level relation extraction based on sememe knowledge-enhanced abstract meaning representation and reasoning. Complex Intell. Syst. 9, 6553–6566 (2023).
Zhou, X. et al. Logo-gr: A local to global graphical reasoning framework for extracting structured information from biomedical literature. IEEE J. Biomed. Health Inform. 28, 2314–2325 (2024).
Zhou, W., Huang, K., Ma, T. & Huang, J. Document-level relation extraction with adaptive thresholding and localized context pooling. In AAAI Conference on Artificial Intelligence (2020).
Nan, G., Guo, Z., Sekulic, I. & Lu, W. Reasoning with latent structure refinement for document-level relation extraction. In Proceedings of the 58th Annual Meeting of the Association for Computational Linguistics 1546–1557 (2020).
Zhang, Z. et al. Document-level relation extraction with dual-tier heterogeneous graph. In Proceedings of the 28th International Conference on Computational Linguistics 1630–1641 (2020).
Minh Tran, H., Nguyen, M. T. & Nguyen, T. H. The dots have their values: Exploiting the node-edge connections in graph-based neural models for document-level relation extraction. In Findings of the Association for Computational Linguistics: EMNLP 2020 4561–4567 (2020).
Chen, L., Su, J., Lam, T.-W. & Luo, R. Exploring pair-aware triangular attention for biomedical relation extraction. In Proceedings of the 14th ACM International Conference on Bioinformatics, Computational Biology, and Health Informatics (2023).
Zhao, S. & Li, C. Corex: Document-level relation extraction framework with consistent two-hop reasoning and evidence sentence prediction. In 2024 International Joint Conference on Neural Networks (IJCNN) 1–8 (2024).
Li, L., Lian, R. & Lu, H. Document-level biomedical relation extraction with generative adversarial network and dual-attention multi-instance learning. In 2021 IEEE International Conference on Bioinformatics and Biomedicine (BIBM) 438–443 (2021).
Bhambhoria, R., Chen, L. & Zhu, X. A simple and effective framework for strict zero-shot hierarchical classification. In Proceedings of the 61st Annual Meeting of the Association for Computational Linguistics (Volume 2: Short Papers), ACL 2023, Toronto, Canada, July 9–14, 2023 (eds Rogers, A. et al.), 1782–1792. https://doi.org/10.18653/V1/2023.ACL-SHORT.152 (Association for Computational Linguistics, 2023).
Inan, E. Making hierarchically aware decisions on short findings for automatic summarisation. J. Comput. Sci. 91, 102692. https://doi.org/10.1016/J.JOCS.2025.102692 (2025).
Zhou, H. et al. Combining context and knowledge representations for chemical-disease relation extraction. IEEE/ACM Trans. Comput. Biol. Bioinf. 16, 1879–1889 (2019).
Wu, S. & He, Y. Enriching pre-trained language model with entity information for relation classification. In Proceedings of the 28th ACM International Conference on Information and Knowledge Management 2361–2364 (2019).
Zhao, Q., Li, J., Xu, C., Yang, J. & Zhao, L. Knowledge-enhanced relation extraction for Chinese emrs. IT Profession. 22, 57–62 (2020).
Mondal, I. BERTChem-DDI: Improved drug-drug interaction prediction from text using chemical structure information. In Proceedings of Knowledgeable NLP: the First Workshop on Integrating Structured Knowledge and Neural Networks for NLP 27–32 (2020).
Asada, M., Miwa, M. & Sasaki, Y. Using drug descriptions and molecular structures for drug-drug interaction extraction from literature. Bioinformatics 37, 1739–1746 (2021).
Sun, Y., Wang, J., Lin, H., Zhang, Y. & Yang, Z. Knowledge guided attention and graph convolutional networks for chemical-disease relation extraction. IEEE/ACM Trans. Comput. Biol. Bioinf. 20, 489–499 (2023).
Yamada, K., Miwa, M. & Sasaki, Y. Biomedical relation extraction with entity type markers and relation-specific question answering. In The 22nd Workshop on Biomedical Natural Language Processing and BioNLP Shared Tasks 377–384 (2023).
Xu, J., Ma, M. D. & Chen, M. Can NLI provide proper indirect supervision for low-resource biomedical relation extraction? In Proceedings of the 61st Annual Meeting of the Association for Computational Linguistics (Volume 1: Long Papers) 2450–2467 (2023).
Yamada, I., Asai, A., Shindo, H., Takeda, H. & Matsumoto, Y. LUKE: deep contextualized entity representations with entity-aware self-attention. In Proceedings of the 2020 Conference on Empirical Methods in Natural Language Processing, EMNLP 2020, Online, November 16–20, 2020 (eds Webber, B. et al.), 6442–6454. https://doi.org/10.18653/V1/2020.EMNLP-MAIN.523 (Association for Computational Linguistics, 2020).
Inan, E. & Dikenelli, O. A semantic-embedding model-driven seq2seq method for domain-oriented entity linking on resource-restricted devices. Int. J. Semantic Web Inf. Syst. 17, 73–87. https://doi.org/10.4018/IJSWIS.2021070105 (2021).
Yasunaga, M., Leskovec, J. & Liang, P. LinkBERT: Pretraining language models with document links. In Proceedings of the 60th Annual Meeting of the Association for Computational Linguistics (Volume 1: Long Papers) 8003–8016 (2022).
Schlichtkrull, M. et al. Modeling relational data with graph convolutional networks. In The Semantic Web: 15th International Conference, ESWC 2018, Heraklion, Crete, Greece, June 3–7, 2018, Proceedings 593–607 (2018).
Li, J. et al. Biocreative v cdr task corpus: a resource for chemical disease relation extraction. Database J. Biol. Databases Curat. 2016, 1 (2016).
Wu, Y., Luo, R., Leung, H. C. M., Ting, H.-F. & Lam, T.-W. Renet: A deep learning approach for extracting gene-disease associations from literature. In Research in Computational Molecular Biology (ed Cowen, L. J.), 272–284 (2019).
Funding
This work was supported by the Key R&D Program in Xinjiang Uygur Autonomous Region (Grant No. 2022B03019-6).
Author information
Authors and Affiliations
Contributions
Xin Chen (X.C.) and Yuhan Liu (Y.L.) designed and performed the experiments, implemented the methods, and analyzed the data. X.C. wrote the main manuscript text and prepared the figures. Liang He (L.H.) supervised the project and provided guidance throughout the research process. All authors discussed the results, revised the manuscript, and approved the final version for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, X., He, L. & Liu, Y. Knowledge enhancement and full utilization of document information for document-level biomedical relation extraction. Sci Rep 16, 3127 (2026). https://doi.org/10.1038/s41598-025-32931-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-32931-w