Abstract
Carnivores must navigate the complexities of human modifications to their environment. Natural resources and biodiversity decline in urban areas, while people in rural areas often pose greater direct risk through actions such as hunting. To evaluate if carnivore populations adapt their behavior to local risks in rural and urban environments, we compared behavioral responses to novel objects in coyotes (Canis latrans). We placed an attractant at arrays of 30 camera-trap stations at 16 pairs of urban and rural field sites across the USA, with a novel object placed at half of the stations. Coyotes exhibited more cautious behavior and remained farther from the attractant at all sites with the novel object; however, urban coyotes got closer to the attractant than rural coyotes. There were few behavioral differences between urban and rural coyotes and none between eastern and western coyotes. Coyotes across the USA exhibit neophobic behavior but urban coyotes, especially western coyotes, are willing to take more risk (i.e., be closer to the attractant). The consistency in most metrics of coyote behavior suggest that solutions developed in one area could be universally useful. This study also demonstrates the effectiveness of a large, collaborative approach to studying broad-scale patterns in behavioral traits.
Similar content being viewed by others
Introduction
Wildlife inhabiting rapidly changing human-dominated environments must adapt to persist, and these adaptations may affect any number of life history traits, such as physiology, animal movement, reproduction, foraging, and behavior1,2. Strong selection for adaptive behavior may cause rapid evolutionary changes3, or reflect plasticity in traits or learning. Comparisons among populations that thrive in contrasting environments can advance our understanding of the adaptive potential of wildlife species.
For some wildlife species, urban landscapes offer suitable habitat with abundant natural and anthropogenic food sources and adequate cover. Urban wildlife often experience decreased exposure to predation by humans and other predators4 but increased exposure to environmental contaminants5. Thus, urban areas can offer refuge for species that are heavily hunted or trapped in rural areas since municipal regulations typically restrict or forbid recreational harvest of wildlife. Furthermore, rural-urban differences in attitudes towards wildlife affect how people respond to and interact with wildlife (i.e., whether they persecute them or not). The increase in abundance and diversity of some wildlife in urban areas is known as the urban wildlife paradox4.
Behavior of individuals within populations that have colonized highly developed urban areas6 are often different from their rural counterparts7. Studies on animals from diverse taxonomic groups – from beetles to bears – have found behavior to be more aggressive, exploratory, and bolder in urban environments compared to rural or natural systems8,9,10,11,12,13. In the case of carnivores, the development of bold or aggressive behaviors could create harmful human-wildlife interactions that reduce tolerance for wildlife and willingness to coexist. This concern is real, urban coyotes (Canis latrans) have been found to be more bold than rural populations in Colorado and North Carolina, United States of America (USA)9,14. Understanding how behavior change in different environments is important to explain how animals may adapt to urbanization and how wildlife management might reduce the risk for conflict14.
Our understanding of urban carnivore behavior commonly stems from studies in single urban areas or comparison of one rural to one urban area, with limited information on trends across regions or species distributions16. Behavioral adaptations that allow carnivores to persist in urban areas arise at the population level due to interactions with humans, which can differ across a species’ range15,16. Thus, how individuals within a population behave in one urban or rural area may differ dramatically from other areas17. Behavioral differences can also emerge for species that have inhabited urban areas for different durations, with more specialization expected for populations that have been living with people for longer18.
We experimentally compare behavioral responses to a novel object by coyotes living in cities and adjacent rural outskirts across the USA and make comparisons between coyotes in rural versus urban and east versus west areas of the USA. We used coyotes as our model species because they have adapted to live in cities across North and Central America19, with some evidence of rural and urban populations exhibiting genetic differences20. We hypothesized that this highly adaptable wildlife species increases boldness in urban areas in response to the intensity and duration of human activity the population experiences. Specifically, we predicted that coyotes exhibit more risk-taking (i.e., visit treatments sites more, get closer to the novel object) and exploratory behavior (i.e., spend more time at treatment sites and in comfortable behavior – defined as shaking, rolling, urinating, defecating, digging, pawing, scratching, stretching, eating, or taking the bait) in urban sites, given that urban coyotes are less commonly persecuted by humans than rural coyotes and that coyotes habituate to human presence; pups born to captive parents with more experience interacting with humans are more willing to feed in the presence of human observers21. We also predicted that western coyotes would exhibit more comfortable behavior at the novel objects because western coyotes have had longer to acclimate to urban areas compared to eastern coyotes that more recently settled via range expansion22. Our results are important to inform national management of coyotes and to help understand how carnivores coexist with humans, given the spread of humans across the landscape and the ability of species to survive and even thrive in human-modified areas6.
Methods
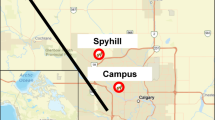
Our network of researchers completed the same experiments at 16 pairs of rural and urban field sites across the USA (Fig. 1) to evaluate coyote spatial and behavioral responses to a novel object. Coyotes can be hunted in rural areas at all our field sites. Coyotes are not hunted in urban areas but may be lethally removed in response to human-wildlife conflicts or concerns about the spread of disease. At each field site, a local team deployed an array of up to 30 camera-trap stations in adjacent rural and urban areas (for a total of up to 60 cameras per field site; Fig. 1), following instructions posted online and previously published9. Each research team identified their own urban and rural field sites for their arrays of camera-trap stations, which resulted in variation in the amount of impervious surface cover. Even so, the average (± SE) percentage of urban surface within a 1.25-km radius of each camera trap station was only 3.5 ± 0.4% for rural sites and 55.2 ± 1.4% for urban sites (Supplemental Information Table S1). To avoid spatial autocorrelation, camera-trap stations within the same array were spaced apart using the diameter of the average home-range size of coyotes as a minimum distance (3.8 km for urban areas, 6.7 km for rural areas)23,24. We used locations that were likely to be visited by coyotes to place each camera-trap station, such as green spaces, game trails, or other features that may be used by coyotes. We did not deploy camera-trap stations in areas with high levels of human disturbance that might be avoided by coyotes and supplies would be prone to theft or damage by humans.
Map of field sites across the United States of America (USA), with color coding representing each of the adjacent urban and rural areas. (a) Adjacent sites are exemplified with close-up maps for Chicago (b), Cleveland (c), and New York City (d), USA, with red circles for urban areas and green squares for rural areas.
Each camera trap was deployed ~ 1 m above ground, attached to a tree, fence, or t-post. We put t-posts in the ground ≥ 3-weeks before trials started so coyotes could habituate to the posts. We used motion-triggered cameras set to take videos that were 20–60 s in length (dependent on camera trap model) when triggered, with a 1-second delay (i.e., shortest delay possible) after being triggered to maximize information collected from each coyote visit. Camera-trap stations were randomly assigned to be either treatment (i.e., with a novel object) or control sites (i.e., without a novel object).
At both treatment and control camera-trap stations, we dug a small hole in the ground (~ 50 mm in diameter and 50 mm deep) approximately 3.5 m from the camera trap, placed a heaping tablespoon of meat bait (Sweet Meat Predator Bait, Russ Carman, New Milford, Pennsylvania) in the hole, stuffed vegetation into the hole, and placed a fatty acid tab (plaster disc ~ 25 mm diameter that is impregnated with a fatty acid scent, Pocatello Supply Depot, Idaho) on top, which is known to attract a variety of carnivores including coyotes. We refer to the combination of meat bait and fatty acid tab as the attractant. At treatment camera-trap stations only, we also established a visual novel object that surrounded the attractant: four wooden stakes with rope tied to the stakes to surround the attractant9. We placed the stakes in a square, 1 m from one another, and tied the rope at the top of the stakes about 1 m above the ground. Research teams recorded data for 3–14 weeks, with camera-trap stations revisited to replace attractants, batteries, and SD cards about every two weeks.
We placed 623 camera-trap stations across the rural and urban field sites. There were 174 control sites and 171 treatment sites in the eastern USA, while 139 control sites were equally matched with treatment sites in the western USA. The majority of camera-trap stations within these field sites were deployed between 14 June 2019 and 1 January 2021, although we also used existing data from Denver, Colorado, and a rural site in Utah9 and one paired site in Los Angeles, California, that was set between 4 February − 20 July 2021. The other site in the Los Angeles area was run at the same time as the majority of sites. Camera-trap stations were active for an average of 37.13 days (SD = 34.90, range = 2–398).
Research complied with ARRIVE guidelines and approvals include USDA-National Wildlife Research Center (QA-3074), New York State Office of Parks, Recreation and Historic Preservation permit #2019-MP8-005, Washington DNR land use license # 60-WS1103, Seattle Parks and Recreation (permission granted but no permit #s issued), Institutional Animal Care and Use Committee at the University of Nebraska-Lincoln (Protocol ID #2006), Institutional Animal Care and Use Committee at The Ohio State University (2013A00000012), Institutional Animal Care and Use Committee at University of Wyoming (no. 20180813 SB00321-02), and the Wyoming Game and Fish Department (Chap. 33 permit ID: 1019). All methods were performed in accordance with the relevant guidelines and regulations.
Because we could not identify individual coyotes from most videos, we used only the first detection of a coyote at each camera-trap station to avoid repeated measures of the same animal. We sometimes recorded multiple videos of the first coyote to be detected at a camera-trap station because the video could only record for a limited time. Thus, we considered a string of multiple videos as one event if < 10 min elapsed between videos9.
We extracted data from each video to score four separate response variables: detection of a coyote (i.e., visitation), time that the coyote spent at the station, the proportion of time at different distances from the attractant, and the proportion of time that the coyote spent exhibiting different behaviors. Detection was coded as the presence/absence of at least one coyote at each camera-trap station. For each initial presence, we quantified the amount of time the coyote visited the camera-trap station, and the proportion of time the coyote spent within one of three distance classes from the attractant at the camera-trap station: (1) far (the coyote stayed at least one body length from the attractant), (2) close (the coyote was within one body length of the attractant), and (3) on (the coyote contacted the attractant with their nose, paw, or body).
We followed Breck et al.9 for behavioral coding and quantified the proportion of time the coyote spent in one of three behavioral states: (1) vigilant (i.e., uncomfortable in Breck et al.9), (2) investigating, and (3) comfortable. We used the same definitions as Breck et al.9 for naming and coding these behaviors. Vigilant behavior indicated that the coyote was cautious or apprehensive and included tucked tail, crouched position, walking hesitantly toward or pacing around the object, and flinching. Investigating indicated the coyote was not concerned with the attractant or novel object and included a tall posture, erect tail, directed travel toward the object without pausing, and a relaxed stature. Comfortable behavior was defined as performing any of the following behavior: shaking, rolling, urinating, defecating, digging, pawing, scratching, stretching, eating, or taking the bait. To avoid interobserver errors, one person coded all videos for distance data and another person coded all videos for behavioral data. However, the research team worked together to define categories and ensure coding was consistent by each coder. Videos were scored continuously to obtain the proportion of time spent in each behavioral or distance category.
We modeled the four response variables described above according to control versus treatment camera-trap stations, urban versus rural field sites, and eastern versus western USA with the regression techniques described below. We used the Mississippi River as the boundary between eastern and western sites22 (Fig. 1). We initially attempted to further compare land cover data from across 12 clusters of field sites (Supplemental Information Table S1), but models using all 12 clusters did not converge.
All models were analyzed within a Bayesian framework using Markov Chain Monte Carlo (MCMC) simulations implemented through JAGS using Program R25 and the ‘jagsUI’ package23. We used logistic regression to evaluate the probability of detection of coyotes by treatment versus control, urban versus rural, and eastern versus western, as well as interactions between these variables. The logistic regression was run across three unthinned MCMC chains27 with 15,000 iterations each, discarding the initial 2,000 draws as burn-in following a 10,000-iteration adaptation phase.
We used a Poisson regression model to analyze the length of visit, given a coyote detection, using the same categories in the logistic regression and their interactions. Because visit times had a large range of values, we included an overdispersion parameter in this model, which resulted in an excellent fit to the data (see results). This Poisson regression model was run across three unthinned MCMC chains with 65,000 iterations each, discarding the initial 15,000 draws as burn-in following a 10,000-iteration adaptation phase.
We used Dirichlet-multinomial regressions to assess behavioral reactions of coyotes to the treatment because these models are designed to evaluate differences in proportions when there are three or more categories, as in this study. We included treatment versus control, urban versus rural, and eastern versus western in this model. Since the Dirichlet regression could only be conducted on the truncated dataset where coyotes were detected, we did not include any interactions because initial attempts led to a lack of parameter convergence.
We ran two behavioral analyses: one on the proportion of time coyotes spent at different distances from the attractant at each site (i.e., distance response), and one on the proportion of time coyotes spent exhibiting distinct behaviors (i.e., behavioral response). For the distance response model, we set the close distance, control sites, rural sites, and eastern sites as the reference categories. In the behavioral response model, we used the same reference categories but replaced close with comfortable. Both Dirichlet-multinomial regressions were run across three unthinned MCMC chains with 10,000 iterations each, discarding the initial 5,000 draws as burn-in following a 5,000-iteration adaptation phase.
We used diffuse, normal priors (mean = 0, variance = 1,000) for the Dirichlet coefficients, and we used flat, uniform priors from − 5 to 5 for coefficient estimates in the logistic and overdispersed Poisson regressions. For all models, we assessed parameter convergence by visually inspecting traceplots and by calculating the Gelman-Rubin statistic, where values < 1.1 indicate convergence across MCMC chains24. We tested model fit using Bayesian p-values, which compare test statistics calculated from observed and expected (i.e., model-generated) data25,26. Finally, we extracted the mean, median, standard deviation, and Bayesian 95% Credible Intervals to assess the strength of effects within each model. We considered predictor variables to significantly influence the response variable when 95% Credible Intervals did not include zero.
Results
Coyote detections
We recorded 635 coyote occurrences. Of the camera-trap sites, 193 sites recorded at least one coyote detection (46 eastern rural, 49 eastern urban, 47 western rural, and 51 western urban). Coyote relative abundance (i.e., the number of coyotes detected per days monitored) averaged 0.046 per site (SE = 0.006) but ranged from 0 to 1.50.
The Bayesian logistic regression for coyote detection (i.e., presence-absence) fit the data well (Bayesian p-value = 0.34). Coyote detection probability was highest at urban control sites in the west (posterior mean = 0.63, 95% credible interval [CrI] = 0.49–0.75) and lowest at rural treatment sites in the west (posterior mean = 0.17, 95% CrI = 0.10–0.25; Fig. 2; Supplemental Information Table S2). Overall detection probability (i.e., control and treatment sites combined) was higher at urban sites than at rural sites in the west but not in the east (Fig. 2; Supplemental Information Table S2). Detection probability was significantly lower at treatment sites than at controls in the west at both rural and urban sites and in the east at rural sites only (Fig. 2; Supplemental Information Table S2).
Model-predicted probabilities of coyote detection at 16 urban and 16 adjacent rural sites surveyed across the USA. Boxes show the median, upper 75%ile, and lower 25%ile; violin plots behind boxes show the full posterior distribution. Treatment sites were those with a novel object. Asterisks indicate instances where 95% credible intervals failed to overlap, indicating a difference between groups.
Coyote visitation time
There were 186 video sequences in which we could code visitation time (44 eastern rural, 48 eastern urban, 43 western rural, and 51 western urban), which averaged 29.3 s (range 1–230 s). The duration of visits was similar across all categories (Table 1). The overdispersed Poisson model had an excellent fit (Bayesian p-value = 0.51). Coyotes exhibited the shortest duration visits at eastern rural control sites (posterior mean = 11.88 s, 95% CrI = 7.91–17.13) and the longest duration visits at western urban control sites (posterior mean = 28.92, 95% CrI = 19.80–40.46; Fig. 3; Supplemental Information Table S3). There were no statistically significant differences in duration between the east and west, urban and rural, or control and treatments (Fig. 3; Supplemental Information Table S3). The largest differences in visitation times between control and treatment sites were at western urban sites (control visits were on average 9.90 s longer) and eastern rural sites (treatment visits were on average 9.80 s longer) but neither of these differences was statistically significant (95% credible intervals for both differences contained zero; Fig. 3; Supplemental Information Table S3).
Model-predicted visit durations in seconds of coyotes detected at sites surveyed across the United States. Boxes show the median, upper 75%ile, and lower 25%ile; violin plots behind boxes show the full posterior distribution. Treatment sites were those with a novel object.
Coyote behavioral distance response
There were 186 video sequences in which the distance from the attractant could be determined (44 eastern rural, 48 eastern urban, 43 western rural, and 51 western urban). The Bayesian Dirichlet model fit was good (Bayesian p-value = 0.65). Coyotes spent more time farther from the attractant at treatment sites than at control sites (posterior mean = 0.86, 95% CrI = 0.47–1.25) but were closer to the attractant at urban sites than at rural sites (posterior mean = −0.41, 95% CrI − 0.77 - −0.04). There was no significant effect of eastern versus western USA (Table 2; Fig. 4).
The average proportion of time coyotes spent at different distances from an attractant placed in front of a camera trap at 16 field sites surveyed across the USA. Panel a depicts rural versus urban sites; panel b depicts control versus treatment sites, which had a novel object.
Coyote behavioral response
There were 161 video sequences in which the behavioral reaction of coyotes to the treatment could be determined. Video sequences of coyotes on camera ranged from 2 to 229 s. The Bayesian Dirichlet model fit was adequate (Bayesian p-value = 0.06). Coyotes spent significantly more time investigating and vigilant and less time comfortable at treatment sites than at control sites (posterior mean = 0.43, 95% CrI = 0.06–0.80; Table 3; Fig. 5). At control sites, coyotes spent an average of 29.0% of the time comfortable, 51.2% of the time investigating, and 19.9% vigilant; at treatment sites these values were 12.5%, 63.2%, and 24.3%, respectively (Fig. 5). There was no effect of urban versus rural or west versus east on these behaviors (Table 3).
The average proportion of time coyotes spent in different behavioral states at 16 field sites surveyed across the USA. Treatment sites were those with a novel object.
Discussion
Our sampling of coyotes in urban and rural areas at several field sites across the USA to assess behavioral responses to a novel object is one of the largest scale carnivore behavior experiments to date. We found clear differences in coyote behavior between control and treatment sites with coyotes found farther from the attractant and exhibiting more cautious behavior (i.e., decreased time spent comfortable and increased time spent investigating and vigilant) at treatment sites across all rural and urban areas (Table 1). These findings highlight the species’ neophobic behavior27. Urban coyotes showed some risky behavior relative to rural coyotes; urban coyotes were observed closer to the attractant and western urban coyotes were more likely to be detected at sites (Table 1). However, the same pattern did not emerge for eastern urban coyotes. Finally, our results suggest western coyotes are more wary; the probability of detection was very different between treatment and control sites. We found no significant differences in visitation duration by coyotes between control and treatment, urban and rural, or eastern and western sites. Despite the large differences across our study in the size of urban areas, the ecology of rural areas, and the time since coyotes have colonized eastern versus western parts of the USA22, we found a relatively uniform behavioral responses by coyotes to our experiment (Table 1). There were few differences between eastern and western coyotes and only a few differences between urban and rural coyotes, with most differences observed in behavior at treatment and control sites. These results suggest coyotes use similar behavioral strategies when encountering novelty in their environment and have adapted similarly to urban and rural environments across the USA.
Many of our results comparing treatment to control sites follow expectations around neophobic behavior. Coyotes were only detected at less than one-third of our sites, with more detections at control sites than treatment sites. When coyotes were detected, they were observed farther from the attractant at treatment sites and showed more cautious behaviors (i.e., spent less time comfortable and more time investigating and vigilant) than at control sites. The consistency in object neophobia by coyotes observed across our field sites suggests this is an ingrained behavior resulting from strong selective pressures over evolutionary time. Similar consistencies between disparate rural and urban populations have been found in small mammals28 and corvids29, although other studies have reported lower levels of neophobia in other taxa living in urban areas30. Our results provide another example of how urbanization impacts behavior of different taxa based on environmental and life history traits36.
Although coyotes across all urban and rural sites showed neophobia, coyotes spent more time close to the attractant at camera-trap stations in urban areas across all field sites and western coyotes were more likely to be detected at urban versus rural sites. We attribute these differences to how people interact with coyotes in rural versus urban areas. In rural areas, those coyotes without a high degree of vigilance, neophobia, or avoidance of human presence are usually killed, trapped, or otherwise persecuted36 thereby removing or conditioning against neophilic or bold behaviors in the population. In urban areas, curiosity or indifference towards novel objects, scents, or situations may assist coyotes in finding new (i.e., anthropogenic) food sources or finding new habitat and, therefore, may be adaptive generally.
Behavioral adaptation to urban areas may occur rapidly (i.e., < 25 years22, resulting in behavior that is consistent across urban areas and that confers advantages to living in urban environments. However, there is likely an upper limit to adaptive bold behavior in urban environments driven by management actions (i.e., lethal removal) of overly bold animals that are removed after they attack pets or people, and also create more calls for removal actions37. Indeed, even in urban areas, overly curious or habituated coyotes will end up as roadkill, poisoned, or lethally removed if they get too comfortable around humans, eating garbage and other anthropogenic food38 or interacting negatively with humans. We hypothesize these adaptive limits explain why we only found a difference in the distance that coyotes would be from the attractant in urban versus rural areas and not in detection, behavioral states, or the duration of visits.
This difference in neophobic behavior between urban and rural areas and within western coyotes also suggests that individuals can adapt to exhibit more risky or exploratory behavior towards a novel object. Urban coyotes likely have behavioral flexibility and learning adaptations7, and our observed behavioral differences may have emerged for several reasons. First, and in accordance with our hypothesis, urban coyotes may be more willing to exhibit risk-taking behavior (i.e., approach a novel object) because they experience less persecution by humans than rural coyotes, with western rural coyotes likely experiencing higher levels of persecution31. Alternatively, urban coyotes may take greater risks due to mesopredator release39; coyote behavior may be altered because they are no longer at risk of predation by large carnivores. This could also explain the differences between urban and rural coyotes observed only in western coyotes, where rural coyotes may still interact with gray wolves (Canis lupus). mountain lions (Puma concolor), and grizzly bears (Ursus americanus). A third explanation is that urban coyotes are also likely to encounter more landscape heterogeneity than rural coyotes. Past research has shown that animals in more heterogenous landscapes make more behavioral adjustments, such as faster habituation rates32. Thus, urban coyotes may habituate more rapidly to novel objects because they consistently encounter novelty in their environments. However, this explanation does not elucidate the difference we observed between western and eastern coyotes. Finally, the increased detection rate we found at urban sites in western coyotes could be unrelated to the experiment and simply relate to higher population densities in urban areas33, but this does not explain the differences in distance response to the novel object. These reasons are not mutually exclusive and several may explain why our results of behavioral adaptations to urbanization in coyotes are similar to other urban wildlife34.
The behavioral differences we found between urban and rural areas is similar to our initial work9,14, but the consistency across sites for most of our behavioral metrics is remarkable and an important finding. Contrary to some of our hypotheses, we found no differences in the other two metrics comparing urban and rural coyote behavior: duration of visits or behavior. We also found no differences for any of the behavioral metrics when comparing eastern versus western coyotes despite differences in the time coyotes colonized these areas22. These results suggest coyotes are inherently well suited for a variety of environments, including urban areas7. The varied diet, habitat use, and social structure of coyotes could provide the needed traits to urban and rural areas throughout the USA. This idea is supported by their rapid range expansion22.
While these results support the theoretical underpinnings of neophobia, the potential emergence of behavioral modifications related to urban habituation, and behavioral consistency within a species, there are some caveats to our study. Because we only coded behavior data for the first coyote detected, this may introduce a bias. Coyotes have consistent personalities35, and it could be that a certain personality type is most likely to be the first coyote to approach an attractant. However, because most coyotes are territorial, it is also likely that a territorial resident was the first detection. Even so, we likely captured videos of different individuals but could not use these data here since we could not distinguish individuals sufficiently. Working with marked populations through hands-on research or with the assistance of artificial intelligence36 offers potential opportunities to increase data acquisition and therefore the ability to investigate finer-scale questions in future research. We also were unable to account for variables that may have influenced coyote behavior at each camera-trap site, such as the time of day or the fine-scale habitat of each site. While we tried to use a more detailed gradient of urban development level, as described in our methods, most models could not converge with more variables, so we maintained binary urban-rural and eastern-western covariates for our comparisons. Attaining deeper resolution about time of day, urbanization gradient, and other factors that we could not include in our models will require more data or more integration in the field methodology. Finally, although urban and rural coyotes may represent discrete populations20, some coyotes along the urban-rural interface likely move between both landscapes. Thus, we may have detected the same coyote at both urban and rural camera stations nearest to one another. While we tried to avoid this by spacing all camera stations greater than a home-range distance apart, a coyote that spends time in both environments may show behavioral plasticity based on their environment. We found behavioral variation previously in flight-initiation distance responses of rural coyotes based on the amount of habitat cover9, and suspect that coyotes could show behavioral flexibility for other circumstances, such as whether to approach a novel object.
The ability to rapidly assess animal behavior to improve conservation and management of species that interact with humans could reduce conflicts and increase coexistence36. Here, we implemented a relatively easy method to compare animal behavior across a broad section of a species’ entire range and found consistent behavioral responses of coyote across the USA in urban and rural areas with only a few exceptions. The consistency of coyote behavior suggests that solutions to conflicts between humans and coyotes may be transferable across urban and rural locations if they are found to work anywhere. Further, our results illustrate how an experimental approach for comparative data at widespread level is possible, but only via a network of researchers (e.g., Borer et al.34).
As urban human populations grow, our results, showing that coyotes are highly neophobic but may be more willing to approach novel objects in some urban environments, have important implications not only for managing coyotes but also for a variety of other carnivore species across the world. Many carnivore species are demonstrating an ability to adapt to urban development, which undoubtedly involves changes in behavior17,37,38. If behavioral adaptations are pervasive, then we need more understanding about the mechanisms causing changes in behavior and how this relates to conflict with humans. Furthermore, we see great potential to expand the type of experiment we conducted; although we placed a novel object at treatment sites, future experiments may be able to simply use camera traps, to attain a relevant behavioral metric because the new placement of a camera trap can serve as a novel object itself39 and could serve as a way to monitor for changes to behavior over time by adding novel objects in subsequent trials. Indeed, the presence of camera traps may explain why visitation rates to control sites in our study were not higher. This progression would help us to further understand carnivore adaptations to anthropogenic environments and help develop management solutions to reduce conflicts. Finally, we think it is important to point out that we do not know if there is a connection between the behavioral metrics we measured with a novel object and the likelihood of a coyote coming into conflict with urbanites by attacking people or pets. More comprehensive behavioral assessments, paired with conflict data, could test this idea. This kind of expanded knowledge of coevolution of carnivore and human behaviors will be necessary to enhance conservation of vulnerable carnivore species and coexistence with those that are common.
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Salamin, N., Wüest, R. O., Lavergne, S., Thuiller, W. & Pearman, P. B. Assessing rapid evolution in a changing environment. Trends Ecol. Evol. 25, 692–698 (2010).
Alberti, M. et al. The complexity of urban Eco-evolutionary dynamics. BioScience 70, 772–793 (2020).
Caspi, T., Johnson, J. R., Lambert, M. R., Schell, C. J. & Sih, A. Behavioral plasticity can facilitate evolution in urban environments. Trends Ecol. Evol. https://doi.org/10.1016/j.tree.2022.08.002 (2022).
Fischer, J. D., Cleeton, S. H., Lyons, T. P. & Miller, J. R. Urbanization and the predation paradox: the role of trophic dynamics in structuring vertebrate communities. BioScience 62, 809–818 (2012).
Levin, R. et al. The urban lead (Pb) burden in humans, animals and the natural environment. Environ. Res. 193, 110377 (2021).
Essl, F. et al. A conceptual framework for Range-Expanding species that track Human-Induced environmental change. BioScience 69, 908–919 (2019).
Lowry, H., Lill, A. & Wong, B. B. M. Behavioural responses of wildlife to urban environments. Biol. Rev. 88, 537–549 (2013).
Baxter-Gilbert, J., Riley, J. L. & Measey, J. Fortune favors the bold toad: urban-derived behavioral traits May provide advantages for invasive amphibian populations. Behav. Ecol. Sociobiol. 75, 130 (2021).
Breck, S. W., Poessel, S. A., Mahoney, P. & Young, J. K. The intrepid urban coyote: a comparison of bold and exploratory behavior in Coyotes from urban and rural environments. Sci. Rep. 9, 2104 (2019).
Šlipogor, V., Graf, C., Massen, J. J. M. & Bugnyar, T. Personality and social environment predict cognitive performance in common marmosets (Callithrix jacchus). Sci. Rep. 12, 6702 (2022).
Isaksson, C., Rodewald, A. D. & Gil, D. Behavioural and ecological consequences of urban life in birds. Front Ecol. Evol. 6, 50 (2018).
Magura, T. et al. Are there personality differences between rural vs. Urban-Living individuals of a specialist ground beetle. Carabus convexus? Insects. 12, 646 (2021).
Beckmann, J. P. & Berger, J. Rapid ecological and behavioural changes in carnivores: the responses of black bears (Ursus americanus) to altered food. J. Zool. 261, 207–212 (2003).
Schell, C. J. et al. The evolutionary consequences of human–wildlife conflict in cities. Evol. Appl. 14, 178–197 (2021).
Kellert, S. R., Black, M., Rush, C. R. & Bath, A. J. Human culture and large carnivore conservation in North America. Conserv. Biol. 10, 977–990 (1996).
Young, J. K., Ma, Z., Laudati, A. & Berger, J. Human–carnivore interactions: lessons learned from communities in the American West. Hum. Dimens Wildl. 20, 349–366 (2015).
Young, J. K., Coppock, D. L., Baggio, J. A., Rood, K. A. & Yirga, G. Linking human perceptions and spotted hyena behavior in urban areas of Ethiopia. Animals 10, 2400 (2020).
Hahs, A. K. et al. Urbanisation generates multiple trait syndromes for terrestrial animal taxa worldwide. Nat. Commun. 14, 4751 (2023).
Poessel, S. A., Gese, E. M. & Young, J. K. Environmental factors influencing the occurrence of Coyotes and conflicts in urban areas. Landsc. Urban Plan. 157, 259–269 (2017).
Adducci, A. et al. Urban Coyotes are genetically distinct from Coyotes in natural habitats. J. Urban Ecol. 6, juaa010 (2020).
Schell, C. J., Young, J. K., Lonsdorf, E. V., Santymire, R. M. & Mateo, J. M. Parental habituation to human disturbance over time reduces fear of humans in Coyote offspring. Ecol. Evol. 8, 12965–12980 (2018).
Hody, J. W. & Kays, R. Mapping the expansion of coyotes (Canis latrans) across North and Central America. ZooKeys 81–97 (2018). https://doi.org/10.3897/zookeys.759.15149
Yu-Sung, S. & Yajima, M. R2jags: Using R to Run ‘JAGS’. (2024).
Gelman, A. Parameterization and bayesian modeling. J. Am. Stat. Assoc. 99, 537–545 (2004).
Suraci, J. P., Nickel, B. A. & Wilmers, C. C. Fine-scale movement decisions by a large carnivore inform conservation planning in human-dominated landscapes. Landsc. Ecol. 35, 1635–1649 (2020).
Hobbs, N. T. & Hooten, M. B. Bayesian Models: A Statistical Primer for Ecologists (Princeton University Press, 2015).
Greggor, A. L., Thornton, A. & Clayton, N. S. Neophobia is not only avoidance: improving neophobia tests by combining cognition and ecology. Curr. Opin. Behav. Sci. 6, 82–89 (2015).
Mazza, V. & Guenther, A. City mice and country mice: innovative problem solving in rural and urban noncommensal rodents. Anim. Behav. 172, 197–210 (2021).
Greggor, A. L., Clayton, N. S., Fulford, A. J. C. & Thornton, A. Street smart: faster approach towards litter in urban areas by highly neophobic Corvids and less fearful birds. Anim. Behav. 117, 123–133 (2016).
Jarjour, C., Evans, J. C., Routh, M. & Morand-Ferron, J. Does City life reduce neophobia? A study on wild black-capped chickadees. Behav. Ecol. 31, 123–131 (2020).
Berger, K. M., Carnivore-Livestock & Conflicts Effects of subsidized predator control and economic correlates on the sheep industry. Conserv. Biol. 20, 751–761 (2006).
Sherratt, T. N., Dewan, I. & Skelhorn, J. The optimal time to approach an unfamiliar object: a bayesian model. Behav. Ecol. 34, 840–849 (2023).
Atwood, T. C., Weeks, H. P. & Gehring, T. M. Spatial ecology of Coyotes along a Suburban-to-Rural gradient. J. Wildl. Manag. 68, 1000–1009 (2004).
Miller, R. et al. Socio-ecological correlates of neophobia in Corvids. Curr. Biol. https://doi.org/10.1016/j.cub.2021.10.045 (2021).
Young, J. K., Mahe, M. & Breck, S. Evaluating behavioral syndromes in Coyotes (Canis latrans). J. Ethol. 33, 137–144 (2015).
Guo, S. et al. Automatic identification of individual primates with deep learning techniques. iScience 23, (2020).
Bateman, P. W. & Fleming, P. A. Big City life: carnivores in urban environments. J. Zool. 287, 1–23 (2012).
Šálek, M., Drahníková, L. & Tkadlec, E. Changes in home range sizes and population densities of carnivore species along the natural to urban habitat gradient. Mammal Rev. 45, 1–14 (2015).
Brooks, J., Kays, R. & Hare, B. Coyotes living near cities are bolder: implications for dog evolution and human-wildlife conflict. Behaviour 157, 289–313 (2020).
Acknowledgements
We thank Catherine Escamilla, Ashley Kimmel, Azana Cochran, Patricia Monzon, Sofia Monzon, Antonio Pepe, Nathan Folkerts, students of the Conservation Biology class at the University of Utah, and many others for their help in the field. Funding was provided by Utah State University, USDA-National Wildlife Research Center, Sageland Collaborative, University of Utah’s Global Change and Sustainability Center, the National Science Foundation (awards 1950350 and 1835410), the School of Natural Resources, University of Nebraska-Lincoln, Max McGraw Wildlife Foundation, Cook County Animal and Rabies Control, University of Wyoming, and University of Georgia Warnell School of Forestry and Natural Resources.
Funding
Funding was provided by Utah State University, USDA-National Wildlife Research Center, Sageland Collaborative, University of Utah’s Global Change and Sustainability Center, the National Science Foundation (awards 1950350 and 1835410), the School of Natural Resources, University of Nebraska-Lincoln, Max McGraw Wildlife Foundation, Cook County Animal and Rabies Control, University of Wyoming, and University of Georgia Warnell School of Forestry and Natural Resources.
Author information
Authors and Affiliations
Contributions
JY, SB, and RK conceived of the idea. JY, RK, SB, and JS wrote the manuscript text. JS and SK coded data, RM, AG, and JY conducted data analysis and made statistical figures. RK made the map. JB, SBA, JB, GC, BC, JC, KD, TG, SG, KG, TG, MH, LH, MK, SL, JM, CN, EP, KR, SR, CS, CS, LS, JY, SB, JS, RK, AG, and RM collected field data and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Young, J.K., Kays, R., Green, A.M. et al. Large-scale experimental assessment of coyote behavior across urban and rural landscapes. Sci Rep 16, 3247 (2026). https://doi.org/10.1038/s41598-025-33189-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-33189-y