Abstract
Few studies have investigated the efficacy of immuno-oncology (IO) combinations at different metastatic sites in renal cell carcinoma (RCC). We evaluated the differential efficacy of IO–IO and IO–tyrosine kinase inhibitor (TKI) combinations by metastatic site in metastatic RCC (mRCC). This retrospective multicenter study by the JK-FOOT Study Group included 579 patients with intermediate- or poor-risk mRCC (per International Metastatic RCC Database Consortium criteria) treated with first-line IO combinations between September 2018 and December 2024. Metastatic sites were lymph nodes, lungs, bones, liver, brain, and others. The primary endpoints were progression-free survival (PFS) and overall survival (OS); the secondary endpoint was objective response rate. Efficacy was compared between IO–IO and IO–TKI for each site. For lymph node (n = 36), lung (n = 132), or brain (n = 16) metastases, OS or PFS was not significantly different between IO–IO and IO–TKI. In bone metastases (n = 80), OS tended to favor IO–TKI (P = 0.053). In liver metastases (n = 22), OS was significantly longer with IO–TKI (P = 0.011). IO–TKI may be a more appropriate first-line option than IO–IO for mRCC with bone or liver metastases, while efficacy is similar for other sites.
Similar content being viewed by others
Introduction
Kidney cancer accounts for approximately 5% of all malignancies and ranks as the sixth most common cancer in men and the tenth in women worldwide1. Renal cell carcinoma (RCC) predominantly occurs in Western countries, whereas its incidence is relatively lower in Asian populations. According to Global Cancer Statistics 2020, there were 431,288 newly diagnosed cases of kidney cancer and 179,368 related deaths globally. Approximately 30% of patients with RCC present with metastatic disease at the time of diagnosis2.
Among patients with clear cell RCC, the most frequent metastatic sites include the lungs (70%), lymph nodes (45%), bone (32%), liver (18%), adrenal glands (10%), and brain (8%)3. Other less common metastatic sites include the thyroid, pancreas, breast, skin, and skeletal muscle. The prognosis of metastatic RCC (mRCC) has markedly improved following the introduction of targeted therapies, particularly vascular endothelial growth factor (VEGF) pathway inhibitors and, more recently, immune checkpoint inhibitors (ICIs)4.
Currently, several ICI-based combination regimens—including nivolumab plus ipilimumab, pembrolizumab plus axitinib, avelumab plus axitinib, nivolumab plus cabozantinib, and pembrolizumab plus lenvatinib—are widely used as first-line systemic treatments for mRCC5,6,7,8,9. Immuno-oncology plus tyrosine kinase inhibitor (IO–TKI) combinations offer robust antitumor activity with higher objective response rates (ORRs), longer progression-free survival (PFS), and lower rates of progressive disease (PD), compared with nivolumab plus ipilimumab. Conversely, nivolumab plus ipilimumab provides durable responses and superior overall survival (OS) in selected patients10.
Despite these advances, some patients are ineligible for ICI-based therapy or relapse soon after adjuvant ICI treatment, making VEGF-targeted TKI monotherapy an important alternative strategy11. Emerging evidence suggests that treatment efficacy may vary by metastatic organ site. For instance, an extended analysis of the CheckMate 9ER trial demonstrated organ-specific differences in target lesion responses with nivolumab plus cabozantinib compared with sunitinib12. Nevertheless, the prognostic implications of organ-specific metastases and the comparative efficacy of IO–IO versus IO–TKI therapy remain insufficiently defined3,13. Therefore, in this multicenter retrospective study, we evaluated the therapeutic outcomes of IO–IO and IO–TKI combination therapies according to metastatic sites in patients with mRCC.
Materials and methods
Patient selection
This study was approved by the institutional review board of Osaka Medical and Pharmaceutical University, Osaka, Japan (approval number: RIN750–2571). Due to its retrospective design, the need for written informed consent was waived; instead, opt-out information was publicly provided. All study procedures adhered to the principles of the Declaration of Helsinki and applicable national regulations.
We retrospectively reviewed 579 patients with mRCC who received first-line ICI-based combination therapy between January 2018 and December 2024 at multiple Japanese institutions (The Jikei University School of Medicine, Minato-ku, Tokyo, Japan; Okayama University Graduate School of Medicine, Dentistry and Pharmaceutical Sciences, Okayama, Japan; Osaka Medical and Pharmaceutical University, Takatsuki, Osaka, Japan; Kindai University Faculty of Medicine, Osaka-Sayama, Osaka, Japan; Fujita Health University School of Medicine, Toyoake, Aichi, Japan; and 15 affiliated hospitals across Japan). Patients classified as having intermediate or poor risk disease according to the International Metastatic Renal Cell Carcinoma Database Consortium (IMDC) criteria were eligible for inclusion. Metastatic site categories included lymph nodes, lung, bone, liver, brain, and other distant organs.
Outcome measurements
The primary endpoints were PFS and OS, while the secondary endpoint was ORR. Treatment responses were evaluated using the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1, and categorized as complete response (CR), partial response (PR), stable disease (SD), or PD.
Disease evaluation and follow-up
Tumor assessments were performed by the treating physicians at screening and subsequently every 2–3 months after the initiation of combination immunotherapy, using imaging modalities such as computed tomography (CT) or magnetic resonance imaging (MRI). Tumor response was evaluated by the treating physicians in accordance with RECIST version 1.1. For each patient, demographic and clinical data were collected at baseline, including age, sex, Karnofsky Performance Status (KPS), IMDC risk group, histological subtype, history of nephrectomy, neutrophil-to-lymphocyte ratio, and metastatic sites. The best response during treatment (CR, PR, SD, or PD) was recorded for each patient. Decisions regarding treatment discontinuation due to disease progression or adverse events, as well as resumption of therapy after discontinuation, were made at the treating physician’s discretion in consultation with the patient and in accordance with regimen-specific guidelines.
Statistical analysis
Patients were categorized into six groups according to the sites of metastasis:
-
1)
lymph node metastases only;
-
2)
lung metastases with or without lymph node metastases;
-
3)
bone metastases with or without lung and/or lymph node metastases;
-
4)
liver metastases with or without lung and/or lymph node metastases;
-
5)
liver and/or bone metastases with or without lung, lymph node, or other distant metastases; and
-
6)
brain metastases.
Metastases to organs other than the lymph nodes, lungs, bones, liver, or brain were classified as “other distant metastases.” Only patients classified as intermediate or poor risk according to the International Metastatic RCC Database Consortium (IMDC) criteria were included.
Patients received either IO–IO or IO–TKI combination therapy as first-line systemic treatment. IO–IO therapy consisted of nivolumab plus ipilimumab. IO–TKI therapy included one of the following regimens: pembrolizumab plus axitinib, nivolumab plus cabozantinib, avelumab plus axitinib, or pembrolizumab plus lenvatinib. Treatment regimen selection was at the discretion of the treating physician based on clinical judgment and patient condition.
PFS, OS, ORR, and prognostic factors were evaluated within each metastasis group by comparing patients treated with IO–IO versus IO–TKI combination therapy.
Statistical analyses were performed using EZR (Saitama Medical Center, Jichi Medical University), a graphical user interface for R (R Foundation for Statistical Computing). ORR was compared using Fisher’s exact test. OS and PFS were estimated using the Kaplan–Meier method, with between-group differences assessed using the log-rank test. Hazard ratios for OS and PFS were calculated using Cox proportional hazards regression models. A two-sided P value < 0.05 was considered statistically significant. Multivariate analyses included age, IMDC risk category, first-line therapy, and KPS.
Results
Patient characteristics
A total of 579 patients with mRCC were included in the analysis. Baseline characteristics are summarized in Table 1. The distribution of metastatic sites is shown in Fig. 1. Kaplan–Meier curves for OS by metastatic site are presented in Fig. 2.
Flowchart of patient selection for the study. A total of 579 patients with metastatic renal cell carcinoma (mRCC) treated with immune checkpoint inhibitor (ICI)-based combination therapy were screened. Patients were excluded if they had favorable IMDC risk (n=89), received first-line TKI monotherapy (n=28), or lacked a pathological diagnosis (n=35). The final cohort was categorized by metastatic site for subsequent efficacy analyses.mRCC, metastatic renal cell carcinoma; ICI, immune checkpoint inhibitor; IO, immuno-oncology; TKI, tyrosine kinase inhibitor; IMDC, International Metastatic RCC Database Consortium.
Kaplan–Meier curves for PFS and OS stratified by metastatic site in patients with mRCC. (a) PFS in patients treated with IO–IO combination therapy. (b) OS in patients treated with IO–IO combination therapy. (c) PFS in patients treated with IO–TKI combination therapy. (d) OS in patients treated with IO–TKI combination therapy. IO, immuno-oncology; TKI, tyrosine kinase inhibitor; OS, overall survival; PFS, progression-free survival; mRCC, metastatic renal cell carcinoma.
Among patients treated with IO–IO, PFS and OS differed significantly among metastatic sites (P = 0.04 and P = 0.002, respectively). Patients with liver metastases treated with IO–IO had the poorest prognosis. By contrast, among patients treated with IO–TKI, PFS and OS did not significantly differ across metastatic sites (P = 0.21 and P = 0.52, respectively).
Lymph node metastasis
A total of 36 patients had lymph node metastases only (IO–IO, n = 21; IO–TKI, n = 15).
Median PFS was 42.4 months in the IO–IO group and 24.5 months in the IO–TKI group (P = 0.95) (Fig. 3a) Median OS was not reached in the IO–IO group and was 33.8 months in the IO–TKI group (P = 0.45) (Fig. 3b). Adjusted for age, Karnofsky Performance Status (KPS), and IMDC risk, the type of first-line treatment (IO–IO vs IO–TKI) was not significantly associated with OS (Table 2). The ORR was 47.6% in the IO–IO group and 46.7% in the IO–TKI group, with no significant difference between the groups (P = 1.00) (Table 3). Among IO–TKI regimens, pembrolizumab plus lenvatinib was associated with significantly longer OS than other IO–TKI combinations (P = 0.04) (Supplementary Fig. S1a, b).
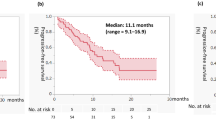
Kaplan–Meier curves for PFS and OS by metastatic site in patients with mRCC treated with IO–IO or IO–TKI combination therapy. PFS (a) and OS (b) in patients with lymph node-only metastases. PFS (c) and OS (d) in patients with lung metastases with or without lymph node metastases. PFS (e) and OS (f) in patients with bone metastases with or without lung and/or lymph node metastases. PFS (g) and OS (h) in patients with liver metastases with or without lung and/or lymph node metastases. PFS (i) and OS (j) in patients with liver and/or bone metastases with or without lung, lymph node, or other distant metastases. PFS (k) and OS (l) in patients with brain metastases. IO, immuno-oncology; TKI, tyrosine kinase inhibitor; OS, overall survival; PFS, progression-free survival; mRCC, metastatic renal cell carcinoma.
Lung metastasis
A total of 132 patients had lung metastases with or without lymph node metastases (IO–IO, n = 62; IO–TKI, n = 70). Median PFS was 15.0 months in the IO–IO group and 14.5 months in the IO–TKI group (P = 0.37) (Fig. 3c). Median OS was 42.7 months in the IO–IO group and was not reached in the IO–TKI group (P = 0.80) (Fig. 3d). After adjustment for age, KPS, and IMDC risk, the type of first-line treatment was not significantly associated with OS (Table 2). The ORR was 51.6% in the IO–IO group and 64.3% in the IO–TKI group, and this difference was not statistically significant (P = 0.14) (Table 3). Among IO–TKI regimens, avelumab plus axitinib was associated with significantly worse OS than other IO–TKI combinations (P = 0.003), whereas PFS did not differ significantly (P = 0.46) (Supplementary Fig. S1c, d).
Bone metastasis
A total of 80 patients had bone metastases with or without lung and/or lymph node metastases (IO–IO, n = 34; IO–TKI, n = 46). Median PFS was 6.3 months in the IO–IO group and 35.2 months in the IO–TKI group (P = 0.01) (Fig. 3e). Median OS was 56.8 months in the IO–IO group and was not reached in the IO–TKI group (P = 0.054) (Fig. 3f). After adjustment for age, KPS, and IMDC risk, the type of first-line treatment was not significantly associated with OS, although a trend favoring IO–TKI was observed (P = 0.055) (Table 2). The ORR was 35.3% in the IO–IO group and 41.3% in the IO–TKI group, with no significant difference between the groups (P = 0.63) (Table 3). Among IO–TKI regimens, no significant differences in PFS (P = 0.52) or OS (P = 0.94) were observed, although all IO–TKI combinations showed relatively favorable outcomes (Supplementary Fig. S1e, f).
In an extended cohort of 89 patients with bone metastases with or without lung, lymph node, and/or other distant metastases (IO–IO, n = 37; IO–TKI, n = 52), no significant differences in PFS (P = 0.37) or OS (P = 0.60) were found among IO–TKI regimens, although nivolumab plus cabozantinib demonstrated numerically better outcomes (Supplementary Fig. S2a, b).
Liver metastases
A total of 22 patients had liver metastases with or without lung and/or lymph node metastases (IO–IO, n = 11; IO–TKI, n = 11). Median PFS was 2.3 months in the IO–IO group and 12.5 months in the IO–TKI group (P = 0.015) (Fig. 3g). Median OS was 5.8 months in the IO–IO group and was not reached in the IO–TKI group (P = 0.01) (Fig. 3h). After adjustment for age, KPS, and IMDC risk, IO–TKI therapy remained significantly associated with improved OS (P = 0.033) (Table 2). The ORR was 27.2% in the IO–IO group and 36.4% in the IO–TKI group (P = 0.64) (Table 3). Among IO–TKI regimens, no significant differences in PFS (P = 0.25) or OS (P = 0.28) were observed, although nivolumab plus cabozantinib showed numerically better PFS, and both nivolumab plus cabozantinib and pembrolizumab plus axitinib showed numerically better OS (Supplementary Fig. S1g, h).
In an extended cohort of 33 patients with liver metastases, with or without lung, lymph node, and/or other distant metastases (IO–IO, n = 16; IO–TKI, n = 17), a significant difference in PFS was observed among IO–TKI regimens (P = 0.006), whereas OS was not significantly different (P = 0.112) (Supplementary Fig. S2c, d).
Liver and/or bone metastases
A total of 145 patients had liver and/or bone metastases with or without lung, lymph node, and/or other distant metastases (IO–IO, n = 63; IO–TKI, n = 82). Median PFS was 4.5 months in the IO–IO group and 35.2 months in the IO–TKI group (P = 0.00002) (Fig. 3i). Median OS was 13.3 months in the IO–IO group and was not reached in the IO–TKI group (P = 0.002) (Fig. 3j). After adjustment for age, KPS, and IMDC risk, IO–TKI therapy remained significantly associated with improved OS (P = 0.003) (Table 2). The ORR was 31.7% in the IO–IO group and 41.0% in the IO–TKI group; this difference was not statistically significant (P = 0.15) (Table 3). Among IO–TKI regimens, no significant differences in PFS (P = 0.92) or OS (P = 0.87) were observed, although all IO–TKI combinations demonstrated relatively favorable outcomes (Supplementary Fig. S1i, j).
Brain metastases
A total of 16 patients had brain metastases, irrespective of other distant metastatic sites (IO–IO, n = 10; IO–TKI, n = 6). Median PFS was 9.1 months in the IO–IO group and 7.5 months in the IO–TKI group (P = 0.069) (Fig. 3k). Median OS was 23.5 months in the IO–IO group and 25.8 months in the IO–TKI group (P = 0.76) (Fig. 3l). After adjustment for age, KPS, and IMDC risk, the type of first-line treatment was not significantly associated with OS (Table 2). The ORR was 50.0% in the IO–IO group and 66.7% in the IO–TKI group, with no significant difference between the groups (P = 1.00) (Table 3).
Among IO–TKI regimens, no significant differences in PFS or OS were observed (Supplementary Fig. S1k, l).
Treatment-related adverse events
Treatment-related adverse events (AEs) are summarized in Supplementary Table 1. Any-grade adverse events were generally more frequent in the IO–TKI group, particularly among patients with lung, liver, or liver and/or bone metastases. However, there were no significant differences between the IO–IO and IO–TKI groups in the incidence of grade ≥3 adverse events across any metastatic site. Dose reductions were more common in the IO–TKI group, especially in patients with lung, bone, or liver and/or bone metastases. In contrast, the frequency of treatment discontinuation due to adverse events did not differ significantly between the two groups for any metastatic site.
Discussion
The therapeutic landscape for metastatic clear cell RCC has undergone significant advancements over the past decade14. Currently, ICI-based combination therapies are the standard first-line treatment for mRCC. However, the efficacy of ICIs according to metastatic site and the comparative effectiveness of different therapeutic regimens remain unclear. Patients with bone, liver, or brain metastases from mRCC have poorer prognoses than those with lung or lymph node metastases3. In this study, we compared the therapeutic outcomes of two combination regimens, IO–IO and IO–TKI, to determine if the efficacy varies by metastatic site. Our findings showed that IO–TKI combinations were more effective in patients with liver or bone metastases. However, no significant difference in efficacy was observed between IO–IO and IO–TKI regimens for patients with lymph node, lung, or brain metastases.
Dudani et al. analyzed data from 10,105 patients with mRCC to evaluate the association between metastatic sites and OS3. Among 9,252 patients with clear cell RCC, the most common metastatic sites were the lung (70%), lymph nodes (45%), bone (32%), liver (18%), adrenal gland (10%), and brain (8%). 88% of patients received TKIs, 4.5% received mTOR inhibitors, and only 6.1% received ICI-based regimens. The median OS was 25.1 months for patients with clear cell RCC with lung metastases, 21.4 months for those with lymph node metastases, 19.4 months for those with bone metastases, 17.6 months for those with liver metastases, 16.5 months for those with brain metastases, and 15.6 months for those with pleural metastases. Metastases to the brain, liver, and bone were consistently associated with poorer survival outcomes15. In contrast, metastases to endocrine organs such as the pancreas, thyroid, and adrenal glands were associated with relatively favorable OS.
Several studies have reported the clinical benefit of nivolumab for patients with lymph node metastases from RCC. Jajodia et al. reported organ-specific response rates to nivolumab in a cohort of 30 patients with mRCC16. The metastatic sites included the lung (n = 26), lymph nodes (n = 20), liver (n = 10), soft tissue (n = 7), brain (n = 5), adrenal glands (n = 4), and peritoneum (n = 4). Based on iRECIST criteria, the best ORRs for each site were 19% (lung), 35% (lymph nodes), 10% (liver), 0% (soft tissue), 20% (brain), 25% (adrenal glands), and 25% (peritoneum). Although limited by a small sample size, these findings suggest that lymph node metastases are more responsive to ICIs, while liver and soft tissue metastases demonstrate the poorest responses. Our results also showed that the patients with lymph node or lung metastasis treated with IO combination therapy had a better prognosis.
In this study, no significant difference was observed in PFS or OS between IO–IO and IO–TKI combination therapies in patients with lung metastases. Among the IO–TKI regimens, only the avelumab plus axitinib combination was associated with poorer OS (P = 0.003).
Bone metastases are associated with poor prognosis among patients with mRCC3. In our study, patients with bone metastases showed a trend toward improved OS with IO–TKI combination therapy compared with IO–IO therapy. Recent studies have shown the potential benefit of cabozantinib for patients with bone metastases. The phase III METEOR trial, which compared cabozantinib with everolimus as a second-line treatment, demonstrated a PFS benefit in patients with bone metastases17. Furthermore, recent subgroup analyses from the CheckMate 9ER trial, which evaluated the combination of nivolumab and cabozantinib, confirmed OS and PFS benefits in patients with bone metastases18.
In the CLEAR trial, which compared lenvatinib plus pembrolizumab with sunitinib for advanced RCC, subgroup analyses showed that patients with poor prognostic factors, such as bone or liver metastases, experienced improved OS and PFS with combination therapy compared with sunitinib19. An exploratory post-hoc subgroup analysis of the CLEAR trial, which evaluated treatment efficacy by metastatic site in patients with mRCC, demonstrated that the combination of lenvatinib plus pembrolizumab provided superior PFS, ORR, and duration of response compared with sunitinib across all assessable subgroups, irrespective of metastatic site (lung, lymph nodes, bone, or liver). Moreover, OS tended to favor lenvatinib plus pembrolizumab in most metastatic sites.20.
Cabozantinib targets multiple tyrosine kinase receptors, including VEGFR2, MET, AXL, RET, and KIT, which are important mediators of tumor cell survival, metastasis, and tumor angiogenesis21. Cabozantinib inhibits VEGFR and MET, affecting the function of osteoclasts and osteoblasts, thereby possibly altering the bone microenvironment. In this study, among treatments for bone and liver metastases, the IO–TKI regimen of nivolumab plus cabozantinib demonstrated better PFS. In patients with bone metastases, in addition to the better OS observed with nivolumab plus cabozantinib, the combinations of pembrolizumab plus lenvatinib and pembrolizumab plus axitinib also demonstrated better outcomes.
Patients with liver metastases showed a systemic decrease in tumor-specific T cells, which limits the efficacy of immunotherapy22. Various hepatic-resident cell types, including Kupffer cells, liver sinusoidal endothelial cells, hepatocytes, plasmacytoid dendritic cells, NKT cells, and hepatic stellate cells, influence T cell fate and survival within the hepatic microenvironment. Through unbiased single-cell analysis, liver-derived CD11b⁺F4/80⁺ macrophages were identified as critical mediators that induce apoptosis of antigen-specific CD8⁺ T cells via the Fas–FasL pathway in the liver metastatic microenvironment. These findings suggest that for patients with liver metastases, IO–TKI combination therapy is a more suitable treatment strategy.
In this study, no significant differences in OS or PFS were observed between IO–IO and IO–TKI combination therapies in patients with brain metastases. Sunitinib has not demonstrated substantial efficacy against brain metastases23. In a phase II trial involving 16 patients with RCC and untreated brain metastases, no intracranial objective responses were reported. SD was observed as the best intracranial response in only five patients. The median PFS was 2.3 months, and the median OS was 6.3 months, indicating poor outcomes. In contrast, cabozantinib might be effective for the brain metastasis. The efficacy of cabozantinib was evaluated in 33 patients with advanced RCC and brain metastases who received the drug in second-line or later settings, none of whom had undergone brain radiotherapy or surgery within 2 months prior to treatment initiation24. The ORR was 55%, including 3 CRs, 14 PRs, and 10 SDs, suggesting the potential effectiveness of cabozantinib in treating brain metastases from RCC. Cabozantinib crosses the blood-brain barrier and may have clinical activity in patients with brain metastases25. In whole-brain lysates of non-tumor-bearing mice, cabozantinib attained 20% of peak plasma levels, suggesting its ability to penetrate the blood-brain barrier. This finding has not been confirmed in human trials. Although the efficacy of cabozantinib for advanced RCC was confirmed by two randomized trials26,27, these studies largely excluded patients with active intracranial disease, and only a small number of patients with adequately treated and stable brain metastases were enrolled.
There is no available data regarding the intracranial efficacy of other TKIs, such as axitinib or pazopanib, in this specific subgroup of patients. To date, the only clinical trial evaluating ICI in patients with mRCC and brain metastases is the GETUG-AFU 26 NIVOREN8 trial28. This was a phase II, single-arm trial assessing the safety and efficacy of nivolumab monotherapy in patients who had progressed after VEGF-targeted therapy. The study included 73 patients with brain metastases, of whom 39 had untreated intracranial target lesions and had not received prior local therapies such as radiotherapy or surgery. The intracranial ORR in this subgroup was 12%, indicating that nivolumab monotherapy has limited activity in untreated brain metastases. They emphasized the importance of local therapy prior to initiating systemic treatment. Our results also showed poor prognosis of patients with brain metastasis compared with other metastases, and no difference in efficacy between IO–IO and IO–TKI combination therapy. The optimal strategy for brain metastasis should be developed.
A study analyzing 657 tumor samples investigated differences in molecular subtypes, genomic features, and the tumor microenvironment (TME) between primary and metastatic sites in RCC29. Although no significant difference in PD-L1 positivity was observed between primary and metastatic tumors overall, the expression rate in bone metastases was notably lower than that in primary tumors. While several clinical trials have reported that PD-L1 expression is not a reliable predictor of response to ICIs, the reduced expression in bone metastases may partly explain the limited efficacy of IO–IO combination therapy in such cases30. This study also estimated the abundance of various cell populations within the TME and identified significant differences between metastatic sites compared with primary renal tumors. Endothelial and monocytic lineage cells were more abundant in bone metastases, whereas B cell-rich microenvironments were observed in lung and skin metastases. Although not statistically significant, T cell abundance appeared higher in lymph node metastases than in the kidney, and CD8⁺ and cytotoxic T cells were enriched in endocrine metastases. Fibroblast populations were notably more abundant across metastatic sites, particularly in the liver, lung, bone, pleura, and soft tissues.
Regarding immune checkpoint gene expression, RNA levels of CTLA4, TIM3, LAG3, or PD1 were not significantly different between primary and metastatic sites. However, PD-L1 expression was higher in pleural metastases, and PD-L2 expression was elevated in lung and bone metastases compared with that in the kidney. These findings suggest that site-specific differences in the TME contribute to the variation in treatment outcomes observed across different metastatic sites. Recent evidence also suggests that primary renal tumors may exhibit distinct sensitivities to immunotherapy. In a subgroup analysis of the CheckMate 214 trial, patients who had not undergone nephrectomy and retained an evaluable primary tumor showed greater tumor shrinkage with first-line nivolumab plus ipilimumab compared with sunitinib, further supporting the concept of organ-specific differences in treatment response31. In patients with lymph node, lung, and bone metastases treated with IO–IO combination therapy, the 36-month PFS and OS rates were approximately 30–40% and 50–70%, respectively, comparable to the results reported in the CheckMate 214 trial. In this study, we further compared treatment outcomes by metastatic site in patients receiving IO–IO or IO–TKI combination therapy (Fig. 2). Among the patients treated with IO–IO therapy, PFS and OS were significantly different across metastatic sites, indicating heterogeneous efficacy depending on the organ involved. In contrast, among those treated with IO–TKI therapy, no significant inter-site differences were observed, suggesting consistent therapeutic efficacy across metastatic sites.
Notably, patients with liver or bone metastases, typically considered to have poor prognoses, demonstrated longer OS than those with lymph node or lung metastases in the IO–TKI group. These findings support the notion that the efficacy of combination immunotherapy may vary depending on the metastatic site.
When comparing treatment-related adverse events between the IO–IO and IO–TKI combination therapy groups, the incidence of any-grade AEs tended to be higher in the IO–TKI group; however, there was no significant difference in the incidence of grade ≥3 adverse events between the two groups. Dose reductions were more frequently observed in the IO–TKI group across all metastatic sites except for brain metastases. Treatment discontinuation was significantly more common in the IO–IO group only among patients with lung metastases, whereas no significant differences were observed at other metastatic sites. Despite these findings, as shown in Figure 3, patients with bone or liver metastases in the IO–TKI group demonstrated better OS.
Limitations
This study has several limitations. First, it was a retrospective analysis, and unknown confounding factors may have influenced the selection of first-line therapy. Second, the IO–TKI group consisted of four different regimens with relatively small sample sizes, limiting the strength of regimen-specific comparisons. Third, several metastatic sites—especially the brain—had a limited number of cases, reducing the statistical power to detect inter-site differences. Moreover, the mechanisms underlying the differential efficacy of IO–IO and IO–TKI therapies according to metastatic sites remain speculative and have not yet been fully elucidated. Given these small sample sizes, the findings should be interpreted with caution. Finally, longer follow-up and the accumulation of additional cases will be required to validate the site-specific differences in therapeutic efficacy suggested by this study.
Conclusion
The effectiveness of first-line ICI-based combination therapy in mRCC varies by metastatic site. IO–TKI therapy may be more appropriate for patients with bone or liver metastases, whereas IO–IO therapy remains a reasonable option for patients with lymph node or lung metastases. These findings may support personalized treatment selection and optimize clinical outcomes in patients with mRCC.
Data availability
Data will be made available by the corresponding author for reasonable requests.
Abbreviations
- ORR:
-
Objective response rate
- PFS:
-
Progression-free survival
- mRCC:
-
Metastatic renal cell carcinoma
- IO:
-
Immuno-oncology
- OS:
-
Overall survival
- TKI:
-
Tyrosine kinase inhibitor
- CR:
-
Complete response
- PR:
-
Partial response
- SD:
-
Stable disease
- PD:
-
Progressive disease
- RECIST:
-
Response evaluation criteria in solid tumors
- iRECIST:
-
Immune-related response evaluation criteria in solid tumors
- KPS:
-
Karnofsky performance status
References
Bray, F. et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 68, 394–424 (2018).
Bukavina, L. et al. Epidemiology of renal cell carcinoma: 2022 update. Eur. Urol. 82, 529–542 (2022).
Dudani, S. et al. Evaluation of clear cell, papillary, and chromophobe renal cell carcinoma metastasis sites and association with survival. JAMA Netw. Open 4, e2021869 (2021).
Ivanyi, P. et al. The treatment of metastatic renal cell carcinoma. Dtsch. Arztebl. Int. 121, 576–586 (2024).
Motzer, R. J. et al. Nivolumab plus ipilimumab versus sunitinib in first-line treatment for advanced renal cell carcinoma: extended follow-up of efficacy and safety results from a randomised, controlled, phase 3 trial. Lancet Oncol. 20, 1370–1385 (2019).
Powles, T. et al. Pembrolizumab plus axitinib versus sunitinib monotherapy as first-line treatment of advanced renal cell carcinoma (KEYNOTE-426): extended follow-up from a randomised, open-label, phase 3 trial. Lancet Oncol. 21, 1563–1573 (2020).
Choueiri, T. K. et al. Updated efficacy results from the JAVELIN Renal 101 trial: first-line avelumab plus axitinib versus sunitinib in patients with advanced renal cell carcinoma. Ann. Oncol. 31, 1030–1039 (2020).
Motzer, R. J. et al. Nivolumab plus cabozantinib versus sunitinib in first-line treatment for advanced renal cell carcinoma (CheckMate 9ER): long-term follow-up results from an open-label, randomized, phase 3 trial. Lancet Oncol. 23, 888–898 (2022).
Motzer, R. et al. Lenvatinib plus pembrolizumab or everolimus for advanced renal cell carcinoma. N. Engl. J. Med. 384, 1289–1300 (2021).
Chen, Y. W., Panian, J., Rose, B., Bagrodia, A. & McKay, R. R. Recent developments in the management of renal cell cancer JCO Oncol. Pract. 6, 875 (2025).
El Zarif, T. et al. First-line systemic therapy following adjuvant immunotherapy in renal cell carcinoma: an international multicenter study. Eur. Urol. 86, 503–512 (2024).
Powles, T. et al. Final overall survival analysis and organ-specific target lesion assessments with two-year follow-up in CheckMate 9ER: nivolumab plus cabozantinib versus sunitinib for patients with advanced renal cell carcinoma. J. Clin. Oncol. 40(suppl 6), 350 (2022).
Wei, H. et al. The prognosis and clinicopathological features of different distant metastases patterns in renal cell carcinoma: analysis based on the SEER database. Sci. Rep. 11, 17822 (2021).
Motzer, R. J. et al. NCCN Guidelines® insights: kidney cancer, version 2.2024. J. Natl Compr. Canc. Netw. 22, 4-16 (2024).
Gong, J., Maia, M. C., Dizman, N., Govindarajan, A. & Pal, S. K. Metastasis in renal cell carcinoma: biology and implications for therapy. Asian J. Urol. 3, 286–292 (2016).
Jajodia, A. et al. Analysis of spatial heterogeneity of responses in metastatic sites in renal cell carcinoma patients treated with nivolumab. Tomography 8, 1363–1373 (2022).
Choueiri, T. K. et al. Cabozantinib versus everolimus in advanced renal-cell carcinoma. N. Engl. J. Med. 373, 1814–1823 (2015).
Choueiri, T. K. et al. Nivolumab plus cabozantinib versus sunitinib for advanced renal-cell carcinoma. N. Engl. J. Med. 384, 829–841 (2021).
Grünwald, V. et al. Phase 3 CLEAR study in patients with advanced renal cell carcinoma: outcomes in subgroups for the lenvatinib-plus-pembrolizumab and sunitinib arms. Front. Oncol. 13, 1223282 (2023).
Grünwald, V. et al. Clinical outcomes by baseline metastases in patients with renal cell carcinoma treated with lenvatinib plus pembrolizumab versus sunitinib: Post hoc analysis of the CLEAR trial. Int. J. Cancer. 156, 1326–1335 (2024).
Yakes, F. M. et al. Cabozantinib (XL184), a novel MET and VEGFR2 inhibitor, simultaneously suppresses metastasis, angiogenesis, and tumor growth. Mol. Cancer Ther. 10, 2298–2308 (2011).
Yu, J. et al. Liver metastasis restrains immunotherapy efficacy via macrophage-mediated T cell elimination. Nat. Med. 22, 152–164 (2021).
Chevreau, C. et al. A phase II trial of sunitinib in patients with renal cell cancer and untreated brain metastases. Clin. Genitourin. Cancer 12, 50–54 (2014).
Hirsch, L. et al. Clinical activity and safety of cabozantinib for brain metastases in patients with renal cell carcinoma. JAMA Oncol. 7, 1815–1823 (2021).
Zhang, Y., Guessous, F., Kofman, A., Schiff, D. & Abounader, R. XL-184, a MET, VEGFR-2 and RET kinase inhibitor for the treatment of thyroid cancer, glioblastoma multiforme and NSCLC. IDrugs 13, 112–121 (2010).
Choueiri, T. K. et al. Cabozantinib versus everolimus in advanced renal cell carcinoma (Meteor): final results from a randomised, open-label, phase 3 trial. Lancet Oncol. 17, 917–927 (2016).
Choueiri, T. K. et al. Cabozantinib versus sunitinib as initial targeted therapy for patients with metastatic renal cell carcinoma of poor or intermediate risk: the Alliance A031203 CABOSUN trial. J. Clin. Oncol. 35, 591–597 (2017).
Flippot, R. et al. Safety and efficacy of nivolumab in brain metastases from renal cell carcinoma: results of the GETUG-AFU 26 NIVOREN multicenter phase II study. J. Clin. Oncol. 37, 2008–2016 (2019).
Gulati, S. et al. Molecular analysis of primary and metastatic sites in patients with renal cell carcinoma. J. Clin. Invest. 134, e176230 (2024).
Gulati, S. & Vogelzang, N. J. Biomarkers in renal cell carcinoma: are we there yet?. Asian J. Urol. 8, 362–375 (2021).
Albiges, L. et al. First-line nivolumab plus ipilimumab versus sunitinib in patients without nephrectomy and with an evaluable primary renal tumor in the CheckMate 214 trial. Eur. Urol. 81, 266–271 (2022).
Acknowledgements
We thank Marco De Velasco, PhD. for his kind assistance with the English editing of this manuscript.
Author information
Authors and Affiliations
Consortia
Contributions
Shingo Toyoda contributed to the protocol and project development, data analysis, and manuscript writing and editing. Takafumi Yanagisawa and Keiichiro Mori contributed to the protocol and project development and data collection. Lan Inoki, Mamoru Hashimoto, Wataru Fukuokaya, Shingo Nishimura, Ryoichi Maenosono, Takehiro Iwata, Kensuke Bekku, Takuhisa Nukaya, and Takuya Tsujino contributed to data collection. Kiyoshi Takahara, Teruo Inamoto, Haruhito Azuma and Kazumasa Komura contributed to the protocol and project development. Kazutoshi Fujita contributed to the project development, manuscript editing, and supervision. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Toyoda, S., Inoki, L., Hashimoto, M. et al. Comparative efficacy of immune checkpoint inhibitor combination therapies by metastatic site in metastatic renal cell carcinoma. Sci Rep 16, 3303 (2026). https://doi.org/10.1038/s41598-025-33198-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-33198-x