Abstract
Excessive application and continuous use of glyphosate herbicide negatively affect plant growth. In addition to damaging soil health in agricultural systems, it disrupts protein synthesis, nutrient cycle, chlorophyll decrease, and photosynthesis, leading to slow growth and decreased plant mass. TWBC can significantly enhance plant growth by improving soil structure, encouraging water retention, and enhancing microbial activity as a slow-release nutrient. Zeolite is a natural mineral that retains essential nutrients, prevents soil leaching, and enhances soil structure, aeration, and water-holding capacity, thereby promoting plant growth. Its buffering capacity changes soil pH, ensuring optimal nutrient acquisition. This study examines the combined effect of TWBC with ZL as a supplement to enhance maize growth under glyphosate stress. Treatments included control, 0.15%, 0.30%, 0.60%, and 1.20% TWBC, applied with 0% and 5% ZL using a completely randomized design in three replications. The combination of 0.60% TWBC with 5% ZL led to an increase in maize plant height (44.91%), root length (49.69%), shoot fresh weight (44.21%), shoot dry weight (58.47%), root fresh weight (52.55%), and root dry weight (62.50%) compared to the control. Additionally, a significant increase in leaf N (23.50%), P (29.09%), and K (17.24%) was observed, while a decrease in glyphosate concentration (88.89%) under glyphosate stress validated the effect of 0.60% TWBC with 5% ZL treatment. In conclusion, the findings suggest that a 0.60% TWBC with 5% ZL is the most effective amendment for enhancing plant growth and mitigating glyphosate stress in maize plants.
Similar content being viewed by others
Introduction
The excessive application of glyphosate and continuous use of the broad-spectrum herbicide negatively affect plant growth and damage soil health in agricultural systems. Plants ingest glyphosate primarily by blocking 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS), a vital catalyst in the shikimate pathway to produce aromatic amino acids. The inhibitory effect causes disruption of protein synthesis, a decrease in chlorophyll, and deterioration of photosynthesis, resulting in slow plant growth and a change in leaf coloration, as well as decreased plant mass1,2,3,4,5,6,7 . The formation of excessive reactive oxygen species (ROS) under glyphosate stress triggers oxidative damage, which activates antioxidant defense by upregulating peroxidase (POD), catalase (CAT), and superoxide dismutase (SOD)4. The application of glyphosate in soil causes three main detrimental effects, including modification of microbial communities, decreased beneficial microorganisms, and disturbances in nutrient cycles, particularly affecting nitrogen and phosphorus movements. The absorption process of essential micronutrients, including iron, manganese, and zinc, becomes restricted when glyphosate binds to these nutrients, making them difficult for plants to obtain. Glyphosate residues can create additional phytotoxicity because they remain in the soil, migrate to groundwater, and accumulate in plant tissues. This disruption collectively results in compromised plant health, reduced agricultural production, and lasting damage to soil fertility8.
Zeolite (ZL) is a naturally occurring aluminosilicate mineral with a porous structure and a strong cation exchange capacity, making it a superior amendment for enhancing soil quality and promoting plant growth9. It retains essential nutrients, including ammonium (NH₄⁺), potassium (K⁺), calcium (Ca2⁺), and magnesium (Mg2⁺), and prevents soil leaching, ensuring plants receive a consistent supply of nutrients10. Zeolite also improves soil structure, aeration, and water-holding capacity, which are crucial for root development and overall plant vigor, especially under drought or nutrient-deficient conditions11. The buffering capacity of zeolite changes soil pH and maintains optimal conditions for nutrient acquisition. Utilizing zeolite leads to increased plant biomass output, while also enhancing root growth and improving the physiological functioning of the plants12.
Biochar (BC) is a carbon-rich, porous material produced through the pyrolysis of organic biomass under limited oxygen conditions13. It is widely recognized for enhancing soil fertility, improving water retention, reducing greenhouse gas emissions, and immobilizing soil contaminants, making it a promising tool for sustainable agriculture14. Tea is a major agricultural product that grows in tropical and subtropical areas worldwide. The annual production of tea worldwide exceeded 5.98 million metric tons in 2017, resulting in significant waste generation from tea production15. The nutrient content of tea waste includes nitrogen (N), phosphorus (P), potassium (K), calcium (Ca), and polyphenols, which enhance soil quality and support plant development. Tea waste’s organic matter enhances microbial activity, soil structure, and nutrient availability16. Solving waste materials into biochar represents a sustainable method to minimize solid waste accumulation. The research evaluates how the transformation of tea waste into biochar helps manage waste while investigating its environmental benefits as a sustainable soil amendment for reducing stress and improving crop yield under glyphosate stress.
Glyphosate stress causes detrimental effects on maize plant growth because it disrupts the shikimic acid pathway through which plants synthesize aromatic amino acids17. The disruption in the shikimic acid pathway results in reduced protein production, stunted growth, chlorosis, and lower biomass accumulation18. Prolonged exposure to glyphosate stress causes damage to root development, blocking nutrient absorption and photosynthetic capacity, which results in reduced crop yield19. The current research investigates how tea waste biochar (TWBC) and ZL mitigate glyphosate stress and improve maize growth. This research addresses a critical knowledge gap by investigating the individual and synergistic effects of TWBC and ZL on maize performance under stress induced by glyphosate. The novelty of the study lies in demonstrating TWBC and ZL as an integrated management approach that not only mitigates the toxic effects of glyphosate but also improves maize resilience, nutrient uptake, and productivity.
Materials and methods
Experimental site
In 2024, a pot experiment was carried out at the Pesticide Quality Control Laboratory (PQCL) (30°09′17.3″N 71°26′35.0″E), situated in Multan, Punjab, Pakistan. To evaluate the soil’s physicochemical properties, soil samples were collected and then air-dried before being sieved through a 2-mm mesh. The soil characteristics were determined as follows (Table 1):
Preparation and characterization of biochar
Initially, tea waste was collected from tea stalls in the local area (30°11′31.4″N 71°28′41.6″E). After sun drying, pyrolysis was performed at 525 ± 5 °C for 94 min. The BC characterization was conducted according to standard protocols for assessing its properties. The tea waste biochar exhibited a pH of 8.13 and electrical conductivity (EC) of 3.48 dS/m. The volatile matter content was 17.5% (%), ash content 21.2% (%), and fixed carbon 61.3% (%), indicating a stable and carbon-rich composition. The biochar had a Brunauer–Emmett–Teller (BET) surface area of 142 m2/g and a total pore volume of 0.27 cm3/g. The cation exchange capacity (CEC) was measured at 38.4 cmol/kg. Elemental analysis revealed 1.60% nitrogen, 0.10% phosphorus, 0.3% potassium, 0.6% calcium, 0.10% magnesium, 0.30% sulfur, 182 mg/kg iron, and 35 mg/kg zinc. Fourier-transform infrared spectroscopy (FTIR) analysis was conducted to examine the functional groups present in the biochar by using Thermo Scientific Nicolet 6700.
Zeolite
For the present study, a natural zeolite was obtained from the local mining resources market in Pakistan (Khyber Pakhtunkhwa). The characteristics include: CEC ~ 92.1 cmol kg⁻1, Si/Al ratio 3–5, water-holding capacity (30% by weight), porosity ~ 34%, bulk density 1.15 g/cm3, and specific gravity ~ 2.25 g/cm3.
Treatment plan and glyphosate contamination
The treatments included five levels of tea waste biochar (TWBC) (0%, 0.15%, 0.30%, 0.60%, and 1.20% TWBC), applied with two levels of zeolite (ZL) (0% and 5% ZL). Total number of treatments were 10 [T1 (control = 0%TWBC + 0%ZL), T2 (0.15%TWBC + 0%ZL), T3 (0.30%TWBC + 0%ZL), T4 (0.60%TWBC + 0%ZL), T5 (1.20%TWBC + 0%ZL), T6 (0%TWBC + 5%ZL), T7 (0.15%TWBC + 5%ZL), T8 (0.30%TWBC + 5%ZL), T9 (0.60%TWBC + 5%ZL) and T10 (1.20%TWBC + 5%ZL) applied following completely randomized design (CRD) with three replications. However, a 2.5 mg/L glyphosate concentration was maintained in the irrigation water to induce glyphosate stress20. The plants were irrigated with this water to maintain the 65% field capacity (FC) throughout the experiment.
Fertilizer
To address the maize nutritional requirements, 119 kg per acre of nitrogen (N), 69 kg per acre of phosphorus (P), and 50 kg per acre of potassium (K) were applied, utilizing urea as the nitrogen source and single superphosphate for phosphorus, at the recommended rates.
Irrigation
The irrigation of each pot is measured and adjusted using a moisture gauge (ADVANCED™; 4-in-1 Soil Meter; China). The researcher checked the soil moisture level daily to verify the desired threshold, which was 65% of the field capacity, as indicated by the instrument reading scale.
Collecting, sterilization, and sowing of seeds
The maize seeds used in this study were procured from a licensed seed vendor authorized by the Government of Punjab, Pakistan. Stringent quality control procedures were implemented to ensure seed viability, with damaged or weak seeds deliberately excluded during the selection process. An extensive surface sterilization process took place before sowing the selected seeds. The seeds required three ethanol rinses at 95% concentration following treatment with 5% sodium hypochlorite solution. The seeds received an additional three rinses of sterilized deionized water to remove any remaining sterilizing agents after their exposure to the 5% sodium hypochlorite solution. Each pot contains 5 kg of soil and initially received 10 seeds for sowing. The thinning procedure followed seed germination to maintain two seedlings per pot21.
Data collection
The data was collected 65 days after harvesting. After harvesting, the fresh and dry weights of the shoots and roots were measured using a digital balance. Samples were oven-dried for 72 h at 65 °C to calculate the dry weights of the shoots and roots.
Estimation of chlorophyll (SPAD)
SPAD (SPAD-502 Portable Chlorophyll Meter) was used to measure leaf chlorophyll content in the portable leaf chlorophyll meter.
Antioxidants
The activity of superoxide dismutase (SOD) was measured by assessing the inhibition of NBT decrease at a wavelength of 560 nm22. Peroxidase (POD) activity was measured at 420 nm with the standard protocol of23. By monitoring the decomposition of hydrogen peroxide (H2O2), catalase (CAT) activity was measured. The H2O2 decomposition was measured through the decrease in the absorbance at 240 nm24. Ascorbate peroxidase (APX) activity was measured through the ascorbate oxidation reaction in the presence of H2O2 at 290 nm using method25.
Gas exchange attributes
Data were collected on a clear, sunny day between 10:30 and 11:30 AM, coinciding with the optimal light intensity for photosynthetic activity26 using an infrared gas analyzer (CI-340 Photosynthesis System; CID, Inc., USA).
Total soluble proteins (TSP)
Total soluble protein content was determined using the biuret method described by27. The absorbance was measured at 545 nm using a UV spectrophotometer. A standard curve was prepared using bovine serum albumin to quantify the protein concentration.
Free proline
The estimation of free proline was carried out following an established protocol28. Proline was extracted using sulfosalicylic acid and then reacted with ninhydrin and glacial acetic acid. The mixture was subsequently treated with toluene and heated at 100 °C, after which the absorbance of the resulting toluene phase was recorded at 520 nm.
Leaves nutrient analysis
Nitrogen content in this study was assessed using a modified version of the micro-Kjeldahl method, originally described by29. Potassium levels were measured using a flame photometer. The yellow colorimetric method was applied for phosphorus determination, with absorbance recorded at 420 nm using a spectrophotometer, following the procedure outlined by30.
Glyphosate analysis
Dried leaf samples were ground into a fine powder using a stainless-steel grinder. For each sample, 1.0 g of powdered leaf material was weighed and transferred into a 50 mL centrifuge tube. Extraction was performed by adding 10 mL of acidified methanol (methanol:water: formic acid, 15:84:1, v/v/v). The mixture was vortexed for 10 min and sonicated for 30 min at room temperature. The extracts were then centrifuged at 10,000 rpm for 10 min, and the supernatant was collected for analysis. Quantification of glyphosate was performed using High-Performance Liquid Chromatography (HPLC). A C18 reversed-phase column was used for separation. The mobile phase consisted of a mixture of phosphate buffer (0.02 M, pH 2.5) and methanol (90:10, v/v), delivered at a flow rate of 1.0 mL/min. The injection volume was 20 µL. Calibration curves were constructed using glyphosate standard solutions in the range of 0.1–10 µg/mL, and the retention time was recorded for all standards and sample extracts.
Statistical analysis
Data analysis was conducted using standard statistical methods as described by31. OriginPro, Excel 365, and MS Office were employed for data processing and visualization. A two-factorial ANOVA was applied to compute the significance of the treatments. Paired comparison graphs were generated using the Fisher’s Least Significant Difference (LSD) test at a significance level of p ≤ 0.05. Additionally, OriginPro was used to construct Pearson correlation matrices, convex hull cluster diagrams, and hierarchical cluster plots32.
Results
The broad peak at ~ 3410 cm⁻1 corresponds to O–H stretching vibrations, indicating hydroxyl groups. The band near 3190 cm⁻1 is assigned to N–H stretching, associated with amine functionalities. A peak at 1615 cm⁻1 represents C=C stretching of aromatic structures, while the band around 1510 cm⁻1 corresponds to C=O stretching vibrations. The absorption band near 1410 cm⁻1 is attributed to C–O stretching, and the peaks at ~ 1230–1260 cm⁻1 represent P–O–C linkages. Additionally, P–OH bending vibrations were evident near 1070 cm⁻1 (Fig. 1; Table 2).
FTIR for the characterization of tea waste biochar.
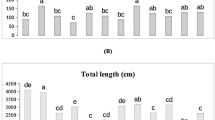
Under 0% ZL, adding 0.15%TWBC, 0.30% TWBC, 0.60% TWBC, and 1.20% TWBC treatments caused a significant increase in plant height (15.99%, 40.57%, 20.52%, and 36.78%), root length (17.95%, 40.56%, 24.24%, and 34.97%), and chlorophyll SPAD (23.54%, 48.86%, 30.36%, and 43.51%) and showed a decrease in proline (17.39%, 35.85%, 20.67%, and 29.34%) compared to control (0%TWBC). Treatments 0.15%TWBC, 0.30%TWBC, 0.60%TWBC, and 1.20%TWBC showed an increase in plant height (15.91%, 30.74%, 44.91%, and 38.64%), root length (12.73%, 27.93%, 49.69%, and 35.52%), chlorophyll SPAD (17.58%, 35.30%, 51.34%, and 40.79%) but decrease in proline (13.37%, 30.00%, 51.16%, and 35.42%) over control (0% TWBC) under 5% ZL (Fig. 2).
Effect of tea waste biochar (TBC) and Zeolite (ZL) on plant height, root length, chlorophyll (SPAD), and proline in maize. Mean values (± SD) are based on three independent replicates. Variations among treatments were statistically verified using Fisher’s LSD at a 5% probability level, where non-matching letters denote significant differences.
Adding 0.15% TWBC, 0.30% TWBC, 0.60% TWBC, and 1.20% TWBC treatments resulted increase in shoot fresh weight (16.93%, 37.37%, 20.91%, and 33.30%), shoot dry weight (8.49%, 30.66%, 12.83%, and 27.36%), root fresh weight (19.32%, 42.15%, 24.48%, and 38.53%), and root dry weight (10.05%, 31.25%, 15.76%, and 28.80%) over the control (0% TWBC) under 0%ZL. Treatment with 0.15% TWBC, 0.30% TWBC, 0.60% TWBC, and 1.20% TWBC under 5% ZL exhibit an increase in shoot fresh weight (11.45%, 29.20%, 44.21%, and 33.46%), shoot dry weight (11.26%, 36.49%, 58.47%, and 40.99%), root fresh weight (13.96%, 31.67%, 52.55%, and 39.94%), and root dry weight (12.50%, 39.29%, 62.50%, and 45.41%) compared to control (0% TWBC) (Fig. 3).
Effect of tea waste biochar (TBC) and Zeolite (ZL) on shoot fresh weight, shoot dry weight, root fresh weight, and root dry weight in maize. Mean values (± SD) are based on three independent replicates. Variations among treatments were statistically verified using Fisher’s LSD at a 5% probability level, where non-matching letters denote significant differences.
Treatments 0.15% TWBC, 0.30% TWBC, 0.60% TWBC, and 1.20% TWBC showed a rise in total soluble sugar (TSP) (12.07%, 32.18%, 16.67%, and 27.59%), free amino acid (FAA) (26.83%, 2.44%, 43.90%, and 7.32%) and caused a decrease in peroxidase (POD) (17.14%, 50.00%, 24.24%, and 41.38%), and ascorbate peroxidase (APX) (15.49%, 67.35%, 24.24%, and 46.43%) over control (0% TWBC) under 0% ZL. Treatment with 0.15% TWBC, 0.30% TWBC, 0.60% TWBC, and 1.20% TWBC under 5% ZL exhibits an increase in TSP (12.83%, 29.95%, 44.39%, and 34.22%), FAA (41.30%, 2.17%, 17.39%, and 4.35%), and demonstrates a decrease in POD (19.35%, 44.16%, 76.19%, and 54.17%), and APX (27.87%, 77.27%, 99.41%, and 95.26%) than control (0% TWBC) (Fig. 4).
Effect of tea waste biochar (TBC) and Zeolite (ZL) on total soluble sugar (TSP), free amino acid (FAA), peroxidase (POD), and ascorbate peroxidase (APX) in maize. Mean values (± SD) are based on three independent replicates. Variations among treatments were statistically verified using Fisher’s LSD at a 5% probability level, where non-matching letters denote significant differences.
Treatments 0.15% TWBC, 0.30% TWBC, 0.60% TWBC, and 1.20% TWBC under 0% ZL exhibit a decrease in CAT (8.67%, 41.74%, 13.19%, and 26.36%), SOD (10.00%, 37.50%, 15.24%, and 27.37%), and glyphosate residue (9.47%, 40.54%, 16.85%, and 31.65%) in leaf and caused increase in glyphosate residue in soil (2.00%, 29.00%, 8.00%, and 20.00%) from the control (0% TWBC). Treatment with 0.15% TWBC, 0.30% TWBC, 0.60% TWBC, and 1.20% TWBC under 5% ZL exhibits a decrease in CAT (17.04%, 43.64%, 66.32%, and 58.00%), SOD (17.00%, 40.96%, 60.27%, and 51.95%), and glyphosate residue in leaf (21.43%, 52.24%, 88.89%, and 64.52%) and caused increase in glyphosate residue in soil (22.83%, 46.74%, 69.57%, and 56.52%) from control (0% TWBC) (Fig. 5).
Effect of tea waste biochar (TBC) and Zeolite (ZL) on catalase (CAT), superoxide dismutase (SOD), glyphosate residue in soil, and glyphosate residue in leaf in maize. Mean values (± SD) are based on three independent replicates. Variations among treatments were statistically verified using Fisher’s LSD at a 5% probability level, where non-matching letters denote significant differences.
Adding 0.15% TWBC, 0.30% TWBC, 0.60% TWBC, and 1.20% TWBC treatment increased leaf N (5.01%, 18.00%, 8.35%, and 14.29%), leaf P (8.41%, 22.43%, 11.21%, and 18.69%), and leaf K (4.26%, 13.68%, 6.84%, and 11.55%) over the control (0% TWBC) under 0% ZL. Treatment with 0.15% TWBC, 0.30% TWBC, 0.60% TWBC, and 1.20% TWBC under 5% ZL exhibits an increase in leaf N (8.56%, 18.03%, 23.50%, and 20.40%), leaf P (11.82%, 22.73%, 29.09%, and 25.45%), and leaf K (7.35%, 14.09%, 17.24%, and 15.59%) than the control (0% TWBC) (Fig. 6).
Effect of tea waste biochar (TBC) and Zeolite (ZL) on leaf N, leaf P, and leaf K in maize. Mean values (± SD) are based on three independent replicates. Variations among treatments were statistically verified using Fisher’s LSD at a 5% probability level, where non-matching letters denote significant differences.
Applying 0.15% TWBC, 0.30% TWBC, 0.60% TWBC, and 1.20% TWBC treatments caused an increase in photosynthetic rate (6.17%, 18.02%, 9.14%, and 14.81%), stomatal conductance (7.41%, 21.30%, 9.26%, and 16.67%), transpiration rate (15.79%, 0%, 23.68%, and 0.26%), and crude protein (6.33%, 25.33%, 10.67%, and 20.00%) over control (0% TWBC) under 0% ZL. Under 5% ZL treatment 0.15% TWBC, 0.30% TWBC, 0.60% TWBC, and 1.20% TWBC demonstrate an increase in photosynthetic rate (7.35%, 15.64%, 20.14%, and 17.54%), stomatal conductance (8.93%, 20.54%, 26.79%, and 23.22%), transpiration rate (21.95%, 0%, 17.89%, and 12.20%), and crude protein (9.32%, 24.12%, 31.19%, and 27.33%) over control (0% TWBC) (Fig. 7).
Effect of tea waste biochar (TBC) and Zeolite (ZL) on photosynthetic rate, stomatal conductance, transpiration rate, and crude protein in maize. Mean values (± SD) are based on three independent replicates. Variations among treatments were statistically verified using Fisher’s LSD at a 5% probability level, where non-matching letters denote significant differences.
In PCA, the control group clustered on the negative side of PC1 and near the center of PC2, indicating a distinct biochemical and physiological profile. The 0.15% TWBC treatment shifted toward the center and slightly positive PC2 values, while 0.30% TWBC occupied the positive PC1 and negative PC2 region, showing greater influence on plant traits. The 0.60% TWBC treatment shifted further into the upper-right quadrant (positive PC1 and PC2), reflecting enhanced trait divergence. The 1.20% TWBC group extended along PC1 but overlapped partially with the 0.30% treatment, maintaining a unique though less pronounced effect profile (Fig. 8A).
Cluster plot convex hull for different levels of tea waste biochar (TWBC) (A), zeolite (ZL) levels (B), and hierarchical cluster plot (C) for studied attributes.
For zeolite, the 0% ZL group clustered compactly in the negative PC1 region with limited variability (PC1: − 7.6585 to + 2.4423; PC2: − 2.3133 to + 1.7532). In contrast, the 5% ZL group displayed a broader and more dispersed convex hull (PC1: − 5.7645 to + 8.0592; PC2: − 1.1314 to + 2.5873), highlighting greater variability and physiological response to treatment (Fig. 8B).
Hierarchical clustering further clarified trait associations (Fig. 8C). Shoot fresh weight clustered with chlorophyll content at the highest similarity (0.063), while shoot and root dry weights formed another close cluster (0.106). CAT activity grouped with soil glyphosate residue (0.145), and leaf N with leaf K (0.178), reflecting nutrient interactions. Plant height clustered with photosynthetic rate (0.334), and stomatal conductance with crude protein (0.350), underscoring their functional relationships. In contrast, glyphosate residue in leaves clustered separately (0.310), proline appeared at a much lower similarity (1.139), and antioxidant enzymes (POD, SOD, APx) only joined at very high dissimilarity (> 10), indicating distinct stress-related behavior. The transpiration rate formed the most distant cluster (48.84), confirming its unique regulation compared with other traits (Fig. 8C).
Plant height showed strong positive correlations with root length (r = 0.988), shoot fresh weight (r = 0.993), root fresh weight (r = 0.992), shoot dry weight (r = 0.970), and root dry weight (r = 0.966), indicating that taller plants were associated with greater biomass accumulation in both aboveground and belowground parts. Similarly, all growth parameters were highly positively correlated with chlorophyll content (r ranging from 0.966 to 0.995), photosynthetic rate (r = 0.995), and crude protein content (r = 0.990), showing close associations between plant vigor and photosynthetic performance. Chlorophyll content was positively correlated with most growth parameters and photosynthetic rate (r = 0.991). Proline content showed strong negative correlations with plant height (r = − 0.988), shoot and root biomass (r ranging from − 0.959 to − 0.992), and chlorophyll (r = − 0.997), consistent with its typical association with stress-related growth decrease. Antioxidant enzymes such as POD, SOD, CAT, and APx showed strong and significant negative correlations with growth and physiological traits (r ranging from − 0.96 to − 0.997), but strong significant positive correlations among themselves (r > 0.978), reflecting their coordinated response pattern. Soil glyphosate residue exhibited a strong positive correlation with plant height (r = 0.884), biomass accumulation (r > 0.88), and photosynthesis (r = 0.879). In contrast, glyphosate residue in leaves showed strong negative correlations with all growth traits (r < − 0.96), photosynthesis (r = − 0.971), and chlorophyll content (r = − 0.972), suggesting that higher glyphosate accumulation in tissues was associated with reduced plant physiological performance. Leaf N, P, and K contents were positively correlated with growth traits (r > 0.95), chlorophyll (r > 0.97), and crude protein (r > 0.98) while negatively correlated with antioxidant enzyme activities (r < − 0.97), showing links between nutrient status, growth, and oxidative stress. Photosynthetic rate was strongly correlated with plant height (r = 0.995), chlorophyll (r = 0.991), crude protein (r = 0.994), and stomatal conductance (r = 0.994). Stomatal conductance was positively correlated with most growth parameters (r > 0.97). Transpiration rate showed moderate to weak correlations with growth parameters but a notably strong positive correlation with FAA (r = 0.939), suggesting a close association with nitrogen metabolism (Fig. 9).
Pearson correlation analysis for the studied attributes.
Discussion
Soil residual glyphosate
Disturbance in the soil ecological balance, resulting in low crop productivity due to the persistence of glyphosate residues in the soil, has raised increasing concern. Besides its short half-life, it can become concentrated in the soil matrix due to continuous and over-application33. Glyphosate sorption in soils is largely controlled by interactions with Fe and Al oxides, clay minerals, and organic matter, with reported adsorption coefficients (Kd) ranging from 10 to 150 L kg⁻1 and organic carbon partition coefficients (Koc) between 300 and 2000, depending on soil type and environmental conditions. The presence of competing anions such as phosphate, nitrate, and sulfate further complicates this behavior, as they compete with glyphosate for binding sites and can markedly reduce its sorption, thereby enhancing its mobility34,35. Residual glyphosate can alter microbial community structure by selectively inhibiting sensitive taxa while favoring glyphosate-tolerant microorganisms, potentially reducing microbial diversity and enzymatic activity essential for nutrient cycling36. Moreover, concerns have been raised about the potential leaching of glyphosate and its metabolite aminomethylphosphonic acid (AMPA) into groundwater, especially under sandy or low pH soils, posing risks to water quality and human health37.
Tea waste biochar (TWBC)
The carbon-rich product TWBC, which originates from tea production waste created through pyrolysis, shows substantial effectiveness in enhancing the stress-resilience of soil while boosting its physical and chemical characteristics38. The porous structure of TWBC helps roots receive adequate air and water, which creates a suitable soil environment for plant root development39. The high surface area and numerous functional groups, such as hydroxyl carboxyl and carbonyl in TWBC, make it an exceptional material for adsorbing agrochemical residue, including glyphosate40. The adsorption mechanism of glyphosate into the rhizosphere decreases its availability, preventing harmful effects upon plants’ metabolic processes. The addition of TWBC in the study positively affected photosynthetic activity while boosting chlorophyll levels and raising nutrient absorption, particularly nitrogen needed for chlorophyll production and amino acid generation41,42. TWBC reduces the accumulation of reactive oxygen species (ROS) and limits glyphosate-induced oxidative stress by reducing POD and CAT activities. The dual role of TWBC emerges as a stress mediator and growth promoter through enhancing physiological stability and improving access to available resources43. Several studies have reported adsorption capacities of biochars for glyphosate ranging from approximately 44 mg g⁻1 (for woody biochar) to as high as 123 mg g⁻1 (for steam-activated rice husk biochar under optimized conditions)44. However, the sorption efficiency can be influenced by the presence of competing anions such as phosphate, nitrate, and sulfate, which compete for the same active sites on the biochar surface and thus may reduce glyphosate binding45.
Zeolite (ZL)
The natural aluminosilicate mineral zeolite (ZL) shows crucial importance in sustaining soil fertility together with plant stress tolerance against glyphosate exposure through its high cation exchange capacity (CEC)9,46. ZL crystalline structure allows cation adsorption, including NH₄⁺ K⁺ and Ca2⁺ within the soil, which promotes both nutrient retention and reduces leaching losses47. The combination of a 5% ZL solution with TWBC applied to maize fields enhanced all growth parameters, including shoot development, root length, photosynthesis rates, and protein content. The ion-absorption qualities of zeolite regulate soil pH levels while creating barriers that hinder glyphosate migration through the soil system48. This behavior decreased glyphosate accumulation in plant tissues and enhanced glyphosate storage within the soil system. Zeolites have also demonstrated direct adsorption of glyphosate, with modified forms, such as Cu-exchanged zeolite 4A, showing maximum adsorption capacities of up to 112.7 mg g⁻1. Beyond glyphosate, natural zeolites richly adsorb nutrients—clinoptilolite-amended columns reduced leaching loss by 82% compared to control in loamy sand systems9,49. ZL promotes microbial activities and root-zone oxygenation while working in combination with TWBC to enhance soil fertility9. ZL-treated plants displayed lower enzyme activities of stress-responsive POD, SOD, and CAT as a sign of successful oxidative damage prevention. ZL promotes nutrient dynamics and creates beneficial rhizosphere conditions that benefit sustainable plant growth under stressful environments50.
Although the present findings clearly demonstrate the potential of TWBC and ZL in improving plant growth and reducing stress indicators, it is important to acknowledge certain limitations. The experiment was conducted under controlled pot conditions, which may not fully capture the complexity of field environments where soil heterogeneity, rainfall, microbial interactions, and long-term nutrient dynamics can influence outcomes. Therefore, further validation through multi-season field trials is required to confirm the consistency and scalability of these results. From a practical perspective, the combined application of TWBC and ZL appears promising for farmers, as tea waste biochar is a low-cost material that can be produced locally, while zeolite is increasingly available in agricultural markets. Together, these amendments not only enhance crop growth and stress resilience but also provide a cost-effective and environmentally friendly approach to sustainable soil management.
Conclusion
In conclusion, 0.6% of TWBC and 5% of ZL increases maize growth under glyphosate stress. Additionally, the 0.6% TWBC with 5% ZL treatment can regulate antioxidant levels, increase essential nutrient concentration, and reduce the negative effects of glyphosate on maize. Beyond enhancing plant growth and physiological resilience, these amendments contribute to improved soil quality, which are central to sustainable crop production. These results suggest that integrating such low-cost and eco-friendly amendments into conventional farming practices could provide farmers with practical tools to balance productivity and environmental safety. Growers should consider using 0.6% TWBC with 5% ZL treatment to alleviate glyphosate stress in maize plants. However, recommendations require field validation under diverse conditions and cost–benefit analysis to ensure practical adoption.
Data availability
All data generated or analyzed during this study are included in this published article.
References
Rivas-Garcia, T., Espinosa-Calderón, A., Hernández-Vázquez, B. & Schwentesius-Rindermann, R. Overview of environmental and health effects related to glyphosate usage. Sustainability 14, 6868 (2022).
Van Bruggen, A. H. C. et al. Indirect effects of the herbicide glyphosate on plant, animal and human health through its effects on microbial communities. Front. Environ. Sci. 9, 763917 (2021).
Maldani, M. et al. Effect of glyphosate and paraquat on seed germination, amino acids, photosynthetic pigments and plant morphology of Vicia faba, Phaseolus vulgaris and Sorghum bicolor. Environ. Sustain. 4, 1–11 (2021).
Wang, J. et al. Effect of graphene oxide-glyphosate nanocomposite on wheat and rape seedlings: Growth, photosynthesis performance, and oxidative stress response. Environ. Technol. Innov. 27, 102527 (2022).
Shopova, E. et al. Modulation of physiological stress response of Triticum aestivum L. to glyphosate by brassinosteroid application. Life 11, 1156 (2021).
Shahid, M., Shafi, Z., Ilyas, T., Singh, U. B. & Pichtel, J. Crosstalk between phytohormones and pesticides: Insights into unravelling the crucial roles of plant growth regulators in improving crop resilience to pesticide stress. Sci. Hortic. 338, 113663 (2024).
Mohy-Ud-Din, W. et al. Glyphosate in the environment: Interactions and fate in complex soil and water settings, and (phyto) remediation strategies. Int. J. Phytoremediation 26, 816–837 (2024).
Kaur, R. et al. Pesticide residues degradation strategies in soil and water: a review. Int. J. Environ. Sci. Technol. 20, 1–24 (2021).
Mondal, M. et al. Zeolites enhance soil health, crop productivity and environmental safety. Agronomy 11, 448 (2021).
Cataldo, E. et al. Application of zeolites in agriculture and other potential uses: A review. Agronomy 11, 1547 (2021).
Kordala, N. & Wyszkowski, M. Zeolite properties, methods of synthesis, and selected applications. Molecules 29, 1069 (2024).
Belviso, C. et al. Impact of zeolite from coal fly ash on soil hydrophysical properties and plant growth. Agriculture 12, 356 (2022).
Leng, L. et al. An overview on engineering the surface area and porosity of biochar. Sci. Total Environ. 763, 144204 (2021).
Qiu, L. et al. Importance of oxygen-containing functionalities and pore structures of biochar in catalyzing pyrolysis of homologous poplar. Chin. J. Chem. Eng. 65, 200–211 (2024).
Zohora, K. F. T. & Arefin, M. R. Tea and tea product diversification: A review. Turkish J. Agric. Sci. Technol. 10, 2334–2353 (2022).
Arhin, I. et al. Looking into the future of organic tea production and sustainable farming: A systematic review. Int. J. Agric. Sustain. 20, 942–954 (2022).
Fuchs, B., Saikkonen, K. & Helander, M. Glyphosate-modulated biosynthesis driving plant defense and species interactions. Trends Plant Sci. 26, 312–323 (2021).
Fuchs, B. et al. A Glyphosate-based herbicide in soil differentially affects hormonal homeostasis and performance of non-target crop plants. Front. Plant Sci. 12, 787958 (2022).
Yu, H. et al. Effects of microplastics and glyphosate on growth rate, morphological plasticity, photosynthesis, and oxidative stress in the aquatic species Salvinia cucullata. Environ. Pollut. 279, 116900 (2021).
Sacała, E. & Roszak, M. Mitigation of glyphosate-based herbicide toxicity in maize (Zea mays L.) seedlings by ascorbic acid. Toxicol. Environ. Chem. 100, 550–559 (2018).
Ahmad, M. et al. An Ethnobotanical study of Medicinal Plants in high mountainous region of Chail valley (District Swat- Pakistan). J. Ethnobiol. Ethnomed. 10, 36 (2014).
Dhindsa, R. S., Plumb-Dhindsa, P. L. & Reid, D. M. Leaf senescence and lipid peroxidation: Effects of some phytohormones, and scavengers of free radicals and singlet oxygen. Physiol. Plant. 56, 453–457 (1982).
Hori, M. et al. Changes in the hepatic glutathione peroxidase redox system produced by coplanar polychlorinated biphenyls in Ah-responsive and-less-responsive strains of mice: mechanism and implications for toxicity. Environ. Toxicol. Pharmacol. 3, 267–275 (1997).
Aebi, H. Catalase in vitro. Methods Enzym. 105, 121–126 (1984).
Nakano, Y. & Asada, K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 22, 867–880 (1981).
Nazar, R., Khan, M. I. R., Iqbal, N., Masood, A. & Khan, N. A. Involvement of ethylene in reversal of salt-inhibited photosynthesis by sulfur in mustard. Physiol. Plant. 152, 331–344 (2014).
Racusen, D. & Johnstone, D. B. Estimation of protein in cellular material. Nature 191, 492–493 (1961).
Bates, L. S., Waldren, R. P. & Teare, I. D. Rapid determination of free proline for water-stress studies. Plant Soil 39, 205–207 (1973).
Steyermark, A. L. & McGee, B. E. Progress in elemental quantitative organic analysis: 1960. Microchem. J. 5, 389–410 (1961).
Olsen, S. R., Sommers, L. E., Page, A. L. & others. Methods of soil analysis. Part 2, 403–430 (1982).
Steel, R. G. D. & Torrie, J. H. Principles and procedures of statistics A biometrical approach 3rd ed. 334–381. (McGraw Hill Book Company Inc., 1996) https://doi.org/10.2307/2333165.
OriginLab Corporation. OriginPro. (OriginLab, Northampton, MA, USA., 2021).
Borggaard, O. K. & Gimsing, A. L. Fate of glyphosate in soil and the possibility of leaching to ground and surface waters: A review. Pest Manag. Sci. Former. Pestic. Sci. 64, 441–456 (2008).
Wang, Y., Zhou, D. M. & Sun, R. J. Effects of phosphate on the adsorption of glyphosate on three different types of Chinese soils. J. Environ. Sci. 17, 711–715 (2005).
Gimsing, A. L., Borggaard, O. K. & Sestoft, P. Modeling the kinetics of the competitive adsorption and desorption of glyphosate and phosphate on goethite and gibbsite and in soils. Environ. Sci. Technol. 38, 1718–1722 (2004).
Newman, M. M. et al. Glyphosate effects on soil rhizosphere-associated bacterial communities. Sci. Total Environ. 543, 155–160 (2016).
Carretta, L., Masin, R. & Zanin, G. Review of studies analysing glyphosate and aminomethylphosphonic acid (AMPA) occurrence in groundwater. Environ. Rev. 30, 88–109 (2022).
Wang, Z. et al. Recent advances review in tea waste: High-value applications, processing technology, and value-added products. Sci. Total Environ. 946, 174225 (2024).
Zou, Z. et al. Biochar application method influences root growth of tea (Camellia sinensis L.) by altering soil biochemical properties. Sci. Hortic. 315, 111960 (2023).
Abdolahi Arshad, M., Rangzan, N. & Nadian Ghomsheh, H. Effect of spent tea waste, compost and biochar on some growth parameters and concentration of nitrogen, phosphorus and potassium in spinach (Spinacia oleracea L.) under salinity stress. J. Plant Nutr. 47, 1029–1044 (2024).
Peiris, C. et al. Effect of acid modified tea-waste biochar on crop productivity of red onion (Allium cepa L). Chemosphere 288, 132551 (2022).
Seth, D. et al. Sustainable environmental practices of tea waste—A comprehensive review. Environ. Sci. Pollut. Res. 32, 1–19 (2023).
Ahmad, S. et al. Salt stress amelioration and nutrient strengthening in spinach (Spinacia oleracea L.) via biochar amendment and zinc fortification: Seed priming versus foliar application. Sci. Rep. 14, 15062 (2024).
Mayakaduwa, S. S. et al. Equilibrium and kinetic mechanisms of woody biochar on aqueous glyphosate removal. Chemosphere 144, 2516–2521 (2016).
Liu, Z. et al. Impacts of biochar concentration and particle size on hydraulic conductivity and DOC leaching of biochar–sand mixtures. J. Hydrol. 533, 461–472 (2016).
Soltys, L. M., Mironyuk, I. F., Tatarchuk, T. R. & Tsinurchyn, V. I. Zeolite-based composites as slow release fertilizers (review). Phys. Chem. Solid State 21, 89–104 (2020).
GirijaVeni, V., Reddy, K. S., Sharma, K. L., Shankar, K. S. & Rohit, J. Role of zeolites in improving nutrient and water storage capacity of soil and their impact on overall soil quality and crop performance. Soil Sci. Fundam. Recent Adv. 449–467 (2021).
AbdEL-Azeiz, E. H., Elsonbaty, A. & Elsherpiny, M. A. Enhancing the quantitative and qualitative traits of wheat grown with low mineral nitrogen level through zeolite, sorbitol and copper. Egypt. J. Soil Sci. 64, 1053–1067 (2024).
Zavareh, S., Farrokhzad, Z. & Darvishi, F. Modification of zeolite 4A for use as an adsorbent for glyphosate and as an antibacterial agent for water. Ecotoxicol. Environ. Saf. 155, 1–8 (2018).
Mahmoud, A. W. M., Rashad, H. M., Esmail, S. E. A., Alsamadany, H. & Abdeldaym, E. A. Application of silicon, zinc, and zeolite nanoparticles—A tool to enhance drought stress tolerance in coriander plants for better growth performance and productivity. Plants 12, 2838 (2023).
Acknowledgements
The authors express their gratitude to the Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2026R101), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Conceptualization; S.D.; S.N.; Conducted experiment; S.D.; Formal analysis; S.D.; Methodology; S.S.; S.D.; Writing—original draft; S.D.; M.O.A.; A.M.G.; E.M.; Writing—review & editing; M.O.A.; A.M.G.; E.M.;
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
I declare that manuscript reporting studies do not involve human participants, data, or tissue. So, it is not applicable. Experimental research and field studies on plants (cultivated or wild), including the collection of plant material, must comply with relevant institutional, national, and international guidelines and legislation. I confirmed that all methods were performed according to the relevant guidelines/regulations/legislation. The seeds were purchased from a local certified seed dealer of the Government of Punjab, Pakistan.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Danish, S., Alotaibi, M.O., Saima, S. et al. Effect of tea waste biochar and zeolite on maize growth and glyphosate uptake irrigated with glyphosate contaminated water. Sci Rep 16, 2198 (2026). https://doi.org/10.1038/s41598-025-34120-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-34120-1