Abstract
Autumn–Winter weather fluctuations, especially higher wind speed and lower temperature, may trigger acute coronary syndrome (ACS). However, evidence on seasonal exposure–response curves and lag structures remains scarce. We analyzed 2445 ACS admissions from a Beijing tertiary hospital (August 2016–July 2021). Daily meteorological data were linked to admission dates. We applied season-specific analyses—including Spearman correlations, multivariable logistic regression, restricted cubic spline (RCS) curves, and lag-response models—to the autumn and winter data. Wind speed showed a linear positive association with ACS risk: autumn odds ratio (OR) 1.32 (95% confidence interval [CI]: 1.05–1.61, P < 0.05) and winter OR 1.41 (95% CI: 1.13–1.87, P < 0.05). Temperature exhibited non-linear inverse relations with thresholds at ≈ 7.5°C in autumn and − 5°C in winter; below these thresholds, the risk decreased significantly per 1 °C rise (Autumn: OR = 0.76, 95% CI: 0.25–0.96, P < 0.05; Winter: OR = 0.74, 95% CI: 0.47–0.99, P < 0.05). Lag analysis revealed the significant effects at lag 2 for wind speed (Winter OR = 1.40, 95% CI: 1.19–1.67, P < 0.05) and at lag 2 for minimum temperature (Winter OR = 0.87, 95% CI: 0.745–0.996, P < 0.05). Associations were absent in spring and summer. Elevated wind speed and extremely low temperatures are significant, lagged drivers of ACS during the cold season in Beijing. Integrating these variables into seasonal early-warning systems could improve prevention for high-risk populations.
Similar content being viewed by others
Introduction
Cardiovascular diseases (CVDs) have consistently ranked among the primary contributors to global mortality and morbidity1. Despite a general decrease in mortality rates due to advancements in emergency procedures and secondary prevention measures for conditions like coronary heart disease (CHD), ischemic heart disease (IHD), notably acute coronary syndrome (ACS), remains a significant public health concern2. ACS, encompassing unstable angina pectoris (UA) and acute myocardial infarction (AMI), is defined by its abrupt onset, critical nature, elevated fatality rates, numerous associated complications, and heightened likelihood of relapse, commonly characterized by high incidence, high fatality, high risk of complications, high recurrence rate, and multiple pathogenic factors3. In recent years, China has exhibited a rising incidence and earlier onset age of ACS, posing a significant health burden to the population4.
Factors like temperature, wind speed, atmospheric pressure, humidity, and precipitation are acknowledged for their capacity to trigger or worsen cardiovascular incidents5,6. Suboptimal temperatures, particularly extreme cold conditions, are closely linked to increased morbidity and mortality from AMI7. Cold temperatures can trigger acute cardiovascular events by heightening sympathetic excitation, promoting vasoconstriction, and elevating blood viscosity8,9, Notably, a notable rise in the incidence of AMI is observed during cold weather, particularly in regions with harsh winter climates10,11.
Nonetheless, current research exhibits limitations. Primarily, the predominant focus of studies has been on a singular meteorological parameter, namely temperature, while overlooking other meteorological elements like wind speed12, the impact of which on AMI occurrences remains ambiguous13. Furthermore, systematic investigations into the nonlinear exposure–response relationship and lag effect of meteorological factors on the onset of ACS are lacking. Notably, the relationship between meteorological factors and ACS exhibits distinct geographical and seasonal patterns14,15. Variations in study outcomes across diverse climatic regions can be attributed to differences in the intensity and variation of meteorological factors, as well as population-specific sensitivity thresholds. These variations are likely influenced by geographic features, local infrastructure such as heating systems, the adaptive capacity of populations, and prevailing lifestyles.
Being the capital of China, Beijing experiences a temperate, semi-humid continental climate affected by monsoons, resulting in pronounced seasonal changes16. This climatic pattern is attributed to Beijing’s situation in the warm temperate zone, leading to four well-defined seasons characterized by cool temperatures in autumn and winter, notable fluctuations in wind velocity, and substantial diurnal temperature variations. These distinctive meteorological conditions could potentially influence the development of ACS. Research conducted in Beijing revealed a notable positive correlation between cold, dry, and windy weather conditions and the likelihood of hospitalization due to AMI12,17. This implies that the concurrent presence of these weather factors exerts a synergistic impact on cardiovascular well-being17. These Beijing-based findings consistently report 20–46% increases in ACS/AMI risk during cold or high-wind conditions 8,12,17.Nevertheless, their joint non-linear thresholds, autumn-versus-winter lag structures and dose–response curves for ACS as a whole remain unquantified; the present study addresses these gaps by integrating RCS and distributed-lag models with seasonal-specific Beijing data.
By integrating meteorological and clinical data for time-series analyses, the study aims to systematically evaluate the correlation and lag effects of wind speed and low temperature on ACS incidence during these seasons. The study seeks to elucidate dose–response relationships and explore seasonal variations. The findings will offer empirical insights into the intricate interplay between meteorological factors and acute cardiovascular events. Moreover, the study will inform urban public health policies, enhance clinical risk assessment strategies, and support the development of meteorological health services.
Information and methodology
Study environment and geographic area
The research was conducted in Beijing, China, situated in the northern region of the North China Plain (latitude 39.4° to 41.6° N, longitude 115.7° to 117.4° E), characterized by a temperate continental climate featuring hot, humid summers, and cold, dry winters. Meteorological data were collected daily from August 2016 to July 2021, encompassing seven parameters: wind speed, air pressure, air temperature, daily maximum and minimum temperatures, daily temperature range, and daily precipitation. Seasons were categorized as spring (March–May), summer (June–August), fall (September–November), and winter (December-February), with daily temperature range denoting the variance between the day’s maximum and minimum temperatures.
Case data
This study included 2,445 patients initially hospitalized in a tertiary hospital in Beijing between August 2016 and July 2021, presenting with ACS as per the Guidelines for Rapid Diagnosis and Treatment of Acute Coronary Syndromes in Emergency Care (2016 edition). Patients were aged ≥ 18 years and exclusions comprised individuals with trauma, malignant tumors, severe valvular disease, or other significant underlying conditions, as well as pregnant or lactating women, and those lacking complete clinical data.
Ethical approval
The study received approval from the Ethics Committee of Dongzhimen Hospital, Beijing University of Chinese Medicine (Approval No: 2023DZMEC-159–02). All procedures adhered to the Declaration of Helsinki and pertinent national protocols. Informed consent was not required as per the ethics committee’s decision, given the utilization of de-identified historical hospitalization data.
Data acquisition and processing
The hospital case system was utilized to extract case data, which were then inputted into Excel 2016 to document essential patient information, including hospitalization number, gender, age, admission date, diagnosis, and area of residence. Concurrently, a database of ACS patients was compiled by aggregating daily incidence data from August 2016 to July 2021. Meteorological data sourced from the National Meteorological Information Center of the China Meteorological Administration were aligned with the patients’ admission dates to establish a database encompassing daily meteorological variables within the specified timeframe. Due to the absence of hourly relative humidity records, daily precipitation and calculated mean vapor pressure were used as proxy moisture variables. Although seasonal headings are used for clarity, all inferential analyses (logistic, RCS, distributed-lag) were performed on daily exposure and daily ACS counts, capturing day-to-day meteorological fluctuations rather than monthly or seasonal averages.
Statistical analysis
Descriptive statistics
Quantitative variables are presented as mean ± standard deviation (X ± S) for normally distributed data, and as median and interquartile range (IQR) for non-normally distributed data. Qualitative variables are reported as frequency (n) and percentage (%).
Time series analysis
Time series analysis involved generating a smoothed time series plot depicting the daily occurrences of ACS episodes. This visualization aimed to elucidate the overarching trend and seasonal patterns in the distribution of ACS events.
Correlation analysis
Spearman’s correlation analysis was employed to evaluate the relationship between meteorological factors and the incidence of ACS, as well as to identify potential influencing variables.
Exposure–response relationship
-
(1)
Initially, independent-samples t-tests were used to screen for seasonal differences in meteorological variables between ACS-event days and non-event days. Variables showing P < 0.10 were then entered into a multivariable logistic regression model to estimate the adjusted odds ratio (OR) and 95% confidence interval (CI) of ACS occurrence while controlling for the remaining six meteorological factors. Stratified sensitivity analyses were further conducted for autumn and winter by categorizing each meteorological factor into low (T1), medium (T2) and high (T3) tertiles.
Due to the single-center design, data regarding city-wide daily denominators, air pollution, and influenza surveillance were unavailable. As these are critical inputs for establishing robust Poisson or DLNM time-series models, we adopted a multivariable logistic regression approach. We acknowledge that the present analysis is exploratory in nature; future multi-center studies utilizing distributed-lag non-linear models that adjust for pollution and holiday effects are needed to verify the thresholds and lag structures reported here.
-
(2)
RCS curve fitting: Curve fitting using restricted cubic spline (RCS) curves was employed to analyze the dose–response relationship between meteorological factors identified as statistically significant in the logistic model and the risk of developing ACS. A nonlinear dose–response relationship was considered present if both P total and P non-linear were less than 0.05.
Regarding collinearity among the predictors included in the final model, simultaneous adjustment and stable coefficients across season-stratified models suggested the absence of serious multicollinearity; the largest inter-meteorological r (wind vs. pressure) was 0.27, well below the conventional “weak” threshold of 0.30.
Lagged effect analysis
Lagged effect analysis was conducted to evaluate the impact of individual meteorological factors by aligning the day of ACS events in the autumn and winter seasons with meteorological data from 1 to 5 days prior, employing a case cross-matching approach.
Results
Demographic information
Seasonal and monthly admission patterns
Figures 1 and 2 illustrate the seasonal and monthly variations in inpatient admissions. Figure 1 displays the number of male and female inpatients across the four seasons—spring, summer, autumn, and winter—and reveals that winter exhibits a significantly higher inpatient volume compared to other seasons, particularly among male patients. Figure 2 further delineates the monthly trend in inpatient numbers, highlighting fluctuations throughout the year. Admissions among male patients peak during winter and early spring, and the overall inpatient count demonstrates a gradual upward trajectory.
Seasonal Distribution of Daily ACS Hospitalizations.
Monthly Distribution of ACS Hospitalizations.
Demographics of ACS patients and admission time series charts
Table 1 reveals a significant predominance of male patients over female patients, with an average patient age of around 70 years, suggesting a higher incidence of ACS in males and older individuals. Gender and age variances were noted across different ACS subtypes, particularly in patients with non-ST-elevation myocardial infarction (NSTEMI), who displayed a higher prevalence of ACS in males and older individuals. Specifically, patients with NSTEMI showed a higher average age compared to individuals with other ACS subtypes.
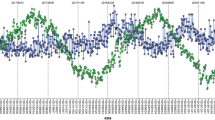
Figure 3 illustrates the temporal dynamics of admissions for ACS patients from August 2016 to June 2021. The figure reveals notable fluctuations in ACS patient admissions, with pronounced peaks observed in certain periods, notably in 2019 and early 2020. These variations may be attributed to factors such as seasonal patterns, holidays, and public health occurrences.
Temporal Trends of ACS Incidence Cases.
Meteorological information
Monthly characteristics of meteorological factors
Table S1 presents detailed meteorological data from August 2016 to July 2021 on a monthly basis. The data reveals a pattern where barometric pressure and wind speeds are higher during winter, while temperatures notably rise in summer. In July, the average temperature peaked at 27.1 °C, the highest recorded for the year, whereas the highest barometric pressure occurred in January. These seasonal variations may be linked to fluctuations in the ACS. See Table S1 in the supplementary material for additional information.
Correlation analysis among meteorological factors
Table S2 presents Spearman correlation coefficients among meteorological factors. The findings reveal a strong positive correlation (r = 0.965, P < 0.01) between daily average air temperature and daily maximum air temperature, as well as daily temperature difference, suggesting a close association between air temperature variations, maximum air temperature, and daily temperature fluctuation. Furthermore, wind speed exhibits correlations with air temperature and barometric pressure, indicating interrelationships among different meteorological variables. See Table S2 in the supplementary material for additional information.
Seasonal correlation analysis
Supplementary Table S3 summarizes the weak but statistically significant Spearman correlations between daily meteorological variables and ACS counts by season. In contrast to spring and summer, associations between ACS and both wind speed (positive) and temperature (negative) were more pronounced in autumn and winter (autumn wind speed r = 0.129, P < 0.01; winter wind speed r = 0.089, P < 0.05; mean air temperature in autumn and minimum temperature in winter both r ≈ –0.10, P < 0.05). Given that the absolute values of these coefficients are small (|r|< 0.2), they should be interpreted in conjunction with the adjusted effect estimates below. No significant associations were found during spring or summer (all P > 0.10), underscoring that the meteorological influence of low temperatures and high wind speeds is largely confined to the cold season.
Pairwise correlations (Table S2) showed that meteorological variables co-vary tightly (e.g., r (T max–T mean) ≈ 0.965). This high collinearity underscores the complexity of the environmental exposure and supports the use of season-stratified exposure–response models. Turning to the exposure–outcome associations, the bivariate correlations in Table S3 are exploratory and should be evaluated together with the adjusted effect estimates below.
T-test analysis of meteorological factors between ACS incidence and non-incidence groups by season
To further clarify whether meteorological factors were significantly different from ACS occurrence in different seasons, we performed t-test analyses on the onset and non-onset groups. The Table2 showed that wind speed was significantly higher in patients who developed ACS in the fall than in the non-developed group (P = 0.0345), while the mean air temperature (P = 0.0328) and the daily maximum air temperature (P = 0.0470) were significantly lower than in the non-developed group, suggesting that lower temperatures and higher wind speeds may be associated with ACS events. A similar trend was observed in winter, where wind speed (P = 0.0145), mean air temperature (P = 0.0273), daily maximum temperature (P = 0.0232), and daily minimum temperature (P = 0.0198) were significantly lower in patients with ACS than in the non-incident group. In comparison, the differences in all the meteorological indicators in spring and summer were not statistically significant (P > 0.05) between the onset and non-onset groups. The above results suggest that low temperatures and higher wind speeds may be important influences on the development of ACS in the fall and winter seasons. Table S4 in the supplementary material provides further details on this analysis.
Multifactorial analysis of seasonal meteorological factors and ACS risk
To further quantify the influence of meteorological factors on the risk of ACS development, we explored the predictive role of seasonal meteorological factors on the occurrence of ACS by logistic regression analysis and after controlling for confounding variables. Model2 in Table 3, after adjusting for other meteorological variables, found that meteorological factors in fall and winter were significantly associated with ACS events. In the fall, wind speed (OR = 1.213, 95% CI: 1.014–1.422, P = 0.0319), mean temperature (OR = 0.861, 95% CI: 0.589–0.989, P = 0.0387), and daily maximum temperature (OR = 0.899, 95% CI: 0.715–0.974, P = 0.0316) had a significant effect on ACS occurrence, suggesting that both increased wind speed and decreased temperature are associated with increased ACS risk. Similarly, winter wind speed (OR = 1.274, 95% CI: 1.038–1.645, P = 0.0305), mean temperature (OR = 0.871, 95% CI: 0.663–0.915, P = 0.0299) and daily minimum temperature (OR = 0.914, 95% CI: 0.840–0.951, P = 0.0243) were significantly associated with the risk of ACS occurrence. In contrast, neither spring nor summer meteorological factors showed significance (P > 0.05). These results suggest that changes in wind speed and temperature during the fall and winter seasons are significant predictors of ACS occurrence. Table S5 in the supplementary material provides further details on this analysis.
Sensitivity analysis of meteorological factors in autumn and winter
Meteorological factors may have varying impacts on cardiovascular events across different time periods. To investigate this, each meteorological factor was divided into low (T1), medium (T2), and high (T3) categories, and sensitivity analyses were conducted. High wind speed and average daily temperature were found to be significant during the fall and winter seasons, as shown in Table 4. Specifically, the high wind speed group in fall (OR = 1.32, 95% CI: 1.05–1.61, P < 0.05) and winter (OR = 1.41, 95% CI: 1.13–1.87, P < 0.05) significantly increased the risk of ACS. Conversely, higher daily mean temperatures in fall (OR = 0.76, 95% CI: 0.25–0.96, P < 0.05) and winter (OR = 0.74, 95% CI: 0.47–0.99, P < 0.05) were negatively associated with ACS risk. Moreover, elevated daily maximum temperatures in fall (OR = 0.79, 95% CI: 0.54–0.97, P < 0.05) and daily minimum temperatures in winter (OR = 0.62, 95% CI: 0.37–0.97, P < 0.05) significantly decreased the risk of ACS (P < 0.05). No statistical significance was observed for other meteorological factors or for low and medium meteorological categories (P > 0.05). These findings suggest that extreme meteorological conditions, such as high wind speed and low temperatures, may exhibit a threshold effect on ACS. Table S6 in the supplementary material provides further details on this analysis.
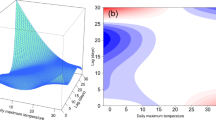
Dose–response relationship between meteorological factors and ACS risk in fall and winter
To investigate the nonlinear correlation between seasonal meteorological variables and the likelihood of ACS-related hospitalizations, RCS analyses revealed a predominantly linear positive relationship between wind speed and ACS risk in both seasons (P total < 0.05, P non-linear > 0.05; Fig. 4), with no statistically significant turning points. In contrast, temperature showed pronounced non-linear threshold effects. Autumn: average daily temperature peaked near 0 °C; risk declined above ≈ 7.5°C (bootstrap 95% CI 6 to 9°C). In winter, we identified a key epidemiological threshold for the minimum temperature at approximately − 5 °C (bootstrap 95% CI − 6 to − 4 °C), defined by the segment of the curve with the steepest slope. The risk of ACS hospitalization escalated markedly once temperatures fell below this threshold. It is important to note that this public-health-relevant threshold is distinct from the mathematical minimum of the U-shaped curve, which occurred at approximately − 0.5 °C.
Dose–Response Relationships of Meteorological Elements in Fall and Winter. Note: Left: Fall; Right: Winter ;The U-shaped peak (− 0.5 °C) is the mathematical extremum; the epidemiological threshold is defined by the steepest slope at ≈ − 5 °C (shaded band).
These findings underscore the need to incorporate linear wind-speed accumulation and temperature-threshold warnings into autumn–winter health interventions.
Analysis of lagged effects of meteorological factors on ACS risk in autumn and winter
To further elucidate the temporal relationship between meteorological conditions and ACS, this study analyzed the lagged effects of weather variables during autumn and winter. As detailed in Table 5, a 2-day lag in wind speed was significantly associated with an increased likelihood of ACS (OR = 1.11, 95% CI: 1.08–1.27, P < 0.05). This pattern was consistent across both autumn (OR = 1.24) and winter (OR = 1.40), suggesting that the cardiovascular strain imposed by elevated wind speed exerts a cumulative or delayed impact.
In terms of temperature, a significant lagged inverse association was observed. Specifically, higher average daily winter temperatures at a 5-day lag (OR = 0.956, 95% CI: 0.904–0.991, P < 0.05) and higher daily minimum temperatures at a 2-day lag (OR = 0.866, 95% CI: 0.745–0.996, P < 0.05) were associated with reduced ACS risk. Conversely, this indicates that cold exposure confers a delayed risk, potentially due to the time required for cold-induced physiological responses—such as sympathetic activation and vasoconstriction—to trigger acute clinical events.
Other variables, including maximum temperature, temperature fluctuation, and precipitation, did not show significant lagged effects. Collectively, these findings reveal a critical time window of susceptibility following exposure to high winds and low temperatures. This underscores the importance of extending health warnings and preventive interventions for several days after the onset of cold spells and high winds.Table S7 in the supplementary material provides further details on this analysis.
Discussion
A substantial body of evidence indicates a close correlation between meteorological fluctuations and the incidence of cardiovascular diseases18,19. However, the season-specific thresholds, lag structures, and dose–response relationships for wind speed and temperature on ACS risk in Beijing remained unquantified. Addressing these gaps, our study found that lower temperatures and higher wind speeds were independently associated with a heightened risk of ACS hospitalization during autumn and winter. Specifically, we identified linear positive associations for wind speed, non-linear thresholds for temperature (≈ 7.5℃ in autumn; -5℃ in winter), and significant lag effects (peak risk at lag-2 days for wind, lag-2–5 days for cold). These findings provide novel, high-resolution insights into the environmental triggers of ACS.
Although two recent Beijing mega-studies—Li et al. (8006 ACS)12 and Chen et al. (124760 AMI)5—confirmed temperature-related risk, they either used yearly linear estimates or focused on same-day effects; consequently, season-specific thresholds, wind-speed independence and lag patterns for the cold season remain quantitatively undefined. Our 5-year, day-level series precisely estimates these neglected parameters and therefore complements city-wide mega-cohorts with higher-resolution exposure–response curves.
Established research commonly depicts the correlation between temperature and cardiovascular events through U-, V-, or J-shaped curves 20. Cold temperatures are recognized as a significant risk factor for acute cardiovascular events, typically falling on the left side of U- or V-shaped curves21,22. For instance, large-scale studies conducted in Chinese urban areas have demonstrated an increase in the incidence and mortality of AMI as ambient temperatures decrease8,20. Additionally, a case-crossover study encompassing 324 cities observed a notable positive association between very low temperatures occurring within -21 days and the incidence of AMIs in unheated regions during winter (OR = 1.46, 95% CI: 1.20–1.76, P < 0.05)8. The outcomes of our study align with these findings, affirming the positive relationship between low temperatures and ACS events, while also uncovering the seasonal variability of this association with threshold characteristics.
The analysis of the dose–response relationship by the RCS method revealed an inverted U-shaped correlation between temperature and the risk of ACS, indicating that temperature’s impact on ACS is not solely dictated by low temperatures but is also influenced by seasonal variations. In autumn, when the average daily temperature fell below 7.5°C, the risk of ACS significantly increased (OR = 0.861, 95% CI:0.589–0.989, P < 0.05). In winter, we observed a notable escalation in ACS hospitalization risk particularly when the minimum temperature fell below the epidemiological threshold of − 5 °C, a point that marks the beginning of a steep increase in risk. Claeys et al.'s research supports the notion that cold environments impact the autonomic nervous system, leading to peripheral vasoconstriction, elevated arterial pressure, heightened cardiac workload, reduced coronary blood flow, and consequent myocardial ischemia5,8.Furthermore, in colder temperatures, there is an increase in platelet viscosity, plasma fibrinogen levels, and various inflammatory markers, all of which may contribute to the pathological mechanisms underlying thrombosis23.Additionally, cold-induced coronary artery spasms can exacerbate unstable angina symptoms and potentially trigger acute myocardial infarction24.
Extreme meteorological conditions such as high wind speed and low temperatures are known to be significant factors in the onset of ACS17. The combination of large diurnal temperature fluctuations, cold climates, and strong winds in the fall and winter months can impose additional stress on both the autonomic nervous system and the cardiovascular system, potentially leading to the triggering of ACS25. Despite the recognized impact of these seasonal variations and threshold effects of meteorological factors, systematic analyses of wind speed remain limited.
In addition to temperature, this study also found that higher wind speed was positively associated with the risk of ACS hospitalization, especially in fall and winter. Meta-analysis confirmed a statistically significant association between higher wind speed and the risk of coronary heart disease, especially in the occurrence of acute myocardial infarction (RR = 1.0217, 95% CI: 1.0060–1.0375, P < 0.05)26. The present study observed a positive association between higher wind speed and the risk of ACS hospitalization in autumn (OR = 1.324, 95% CI: 1.051–1.609, P < 0.05) and winter (OR = 1.414, 95% CI: 1.127–1.871, P < 0.05), indicating that high wind speed significantly increased the risk of ACS hospitalization, further validating the role of wind speed as an independent risk factor.
The impact of wind speed on ACS also exhibits a threshold effect, whereby the risk of ACS markedly rises when wind speed surpasses a specific threshold. This effect is thought to involve wind speed hastening heat dissipation from the body surface, necessitating increased energy expenditure to maintain body temperature. This process triggers vasoconstriction, heightened blood pressure, sympathetic excitation, and an augmented stress response, placing additional strain on the heart and culminating in acute cardiovascular events. This finding is consistent with prior research conducted in Beijing. For instance, an investigation into weather patterns and rates of AMI hospitalizations in Beijing demonstrated that cold, dry, and windy conditions substantially amplified the likelihood of AMI hospitalization, particularly among men17.Our current study substantiates the synergistic impact of the combined wind-cold effect and underscores the importance of investigating the collective influence of meteorological variables. Future research should prioritize examining the combined effects of multiple factors rather than focusing solely on individual influences.
Adverse health effects manifest gradually over time rather than instantaneously. This study elucidates the delayed impact of meteorological variables on ACS development, referred to as the “lag effect”. Through a comprehensive time-stratified case-crossover analysis, it was observed that the correlation between cold weather and AMI typically surfaced one day after exposure, peaked at three days, and diminished after around five days10. Consistent findings were reported by Lin et al., demonstrating a significant rise in cardiovascular event risk 3–7 days post cold exposure27. In this study, Wind speed was identified as a factor associated with increased ACS hospitalization risk, with the most pronounced effect observed on the second lag day. Cold temperature, on the other hand, exhibited its peak impact on the fifth lag day. Severe cold and windy conditions may deter outdoor activities. Following cold exposure, initial sympathetic excitation may induce vasoconstriction and heightened blood pressure, subsequently triggering an inflammatory response and endothelial dysfunction. These processes may increase blood viscosity and platelet activity, thereby promoting thrombosis and potentially triggering an ACS episode 28.
These findings indicate that meteorological factors have delayed and persistent health effects, potentially leading to an underestimation of the true burden of meteorological changes on cardiovascular health if delayed effects are disregarded. Consequently, adjustments to public health early warning systems and health management strategies are warranted. For instance, continuous monitoring of vulnerable populations for at least a week following cold spells and windy conditions is recommended to promptly detect potential spikes in morbidity. Moreover, clinical preventive and control measures should consider the lagged effects of weather, emphasizing enhanced medication management, symptom surveillance, and medical guidance in the days following extreme weather events, with a specific focus on the elderly and individuals with preexisting cardiovascular conditions.
The thresholds identified here (− 5°C for winter minimum temperature and 7.5°C for autumn mean temperature) were derived from a temperate, monsoon-influenced city (Beijing). Populations living in arctic, tropical or equatorial climates undergo long-term physiological and behavioral adaptation to their respective thermal environments. This consensus is supported by large-scale multi-city studies demonstrating that cold-related cardiovascular mortality thresholds shift by ≥ 5℃ across Köppen climate zones20,29. Consequently, the present estimates should not be extrapolated to regions with markedly different baseline temperatures or wind-speed regimes. We recommend that public-health agencies in climatologically similar temperate cities (Köppen Dwa/Dwb, latitude 35–45° N, seasonal temperature amplitude > 25°C) adopt these thresholds for cold-season early warnings, while regions outside this belt should establish their own exposure–response curves through multi-city replications.
Our findings also prompt a critical reconsideration of conventional meteorological risk models. The significant age gradient observed in our cohort—where NSTEMI patients were on average 6.7 years older than STEMI patients (74.2 vs. 67.5 years, P < 0.01)—suggests that the cold-season thresholds and lag patterns we identified may be disproportionately driven by the elderly subset. This is biologically plausible, as a growing body of evidence indicates that thermoregulatory and baroreflex functions decline with age, which amplifies cardiovascular responses to cold or wind stimuli8,30,31. Crucially, this individual-level vulnerability is not captured by traditional, aggregate metrics like the wind-chill index (WCI). Indeed, recent multi-country evidence demonstrates that modeling temperature and wind speed as independent terms outperforms the classical WCI in predicting cardiovascular outcomes 32,33.
Precisely to address this methodological gap, our study provides—for the first time in a Beijing single-center cohort—the season-specific exposure thresholds and distributed-lag curves that are absent from the previous WCI literature. More importantly, the age gradient in our data signals that older adults likely bear a disproportionate share of the excess risk from cold and wind—an age × meteorology interaction that the WCI framework is inherently unable to quantify. Therefore, in our upcoming hourly, individual-level multi-city dataset, we will explicitly test these interactions to move beyond the classical wind-chill index and towards a more personalized, physiologically-grounded risk assessment model.
Nevertheless, this study is subject to certain limitations.
First, the single-center design may constrain the generalizability of the findings, and thus validation across multiple centers and diverse climatic regions is warranted in future research.
Second, regarding environmental confounders, we were unable to include air pollution data, specifically PM2.5, or specific temporal factors such as day-of-the-week effects and public holidays in our multivariate model due to data unavailability. We acknowledge that air pollution is a potential risk factor and confounder, as suggested in recent studies12,17. However, it is worth noting that in Beijing’s autumn and winter, strong winds are typically associated with the dispersion of pollutants and improved air quality; thus, the positive association between wind speed and ACS observed in our study is unlikely to be solely driven by pollution.
Third, the study period covered the COVID-19 pandemic. As observed in our results, particularly around February 2020, there were fluctuations in hospital admissions likely due to social restrictions rather than meteorological factors. Our current model did not employ specific time-series adjustments, such as splines, to control for these long-term trends or the pandemic effect, which is a limitation of the retrospective design.
Fourth, detailed individual-level exposure data were unavailable. The absence of hourly relative humidity, wind gusts, or individual GPS tracking data prevented the reconstruction of minute-level exposure trajectories. Future studies utilizing hourly meteorological grids and individual mobility data are needed to refine personal exposure maps and clarify moisture-related pathways.
Despite these limitations, our daily-level analysis reveals critical thresholds and lag windows that can provide valuable evidence for early warning systems until more comprehensive multi-center collaborations with integrated environmental data become available.
This study systematically assessed the relationship between temperature, wind speed, and the onset of ACS through time series analysis. The findings enrich the understanding of how meteorological factors contribute to acute cardiovascular events in northern Chinese cities. The study highlights the significance of wind speed as a notable risk factor and elucidates the exposure–response relationship using the RCS method. These insights enhance the scientific and practical implications of the study’s conclusions.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
GBD 2019 Risk Factors Collaborators. Global burden of 87 risk factors in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396, 1223–1249 (2020).
GBD 2021 Forecasting Collaborators. Burden of disease scenarios for 204 countries and territories, 2022–2050: a forecasting analysis for the Global Burden of Disease Study 2021. Lancet 403, 2204–2256 (2024).
Rao, S. V. et al. 2025 ACC/AHA/ACEP/NAEMSP/SCAI guideline for the management of patients with acute coronary syndromes: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 85, 2135–2237 (2025).
Ulvenstam, A., Graipe, A., Irewall, A.-L., Söderström, L. & Mooe, T. Incidence and predictors of cardiovascular outcomes after acute coronary syndrome in a population-based cohort study. Sci. Rep. 13, 3447 (2023).
Chen, Y. et al. Association of extreme precipitation with hospitalizations for acute myocardial infarction in Beijing, China: a time-series study. Front. Public Health 10, 1024816 (2022).
Liu, T. et al. The interactive and joint associations of ambient PM2.5 and temperature on the onset of acute coronary syndrome: findings from the Chinese Cardiovascular Association (CCA) Database-Chest Pain Center Registry. Environ. Sci. Technol. 58, 21978–21988 (2024).
Mohammad, M. A. et al. Association of weather with day-to-day incidence of myocardial infarction: a SWEDEHEART nationwide observational study. JAMA Cardiol. 3, 1081–1089 (2018).
Jiang, Y. et al. Non-optimum temperature increases risk and burden of acute myocardial infarction onset: a nationwide case-crossover study at hourly level in 324 Chinese cities. EClinicalMedicine 50, 101501 (2022).
Tang, S. et al. Temperature fluctuation and acute myocardial infarction in Beijing: an extended analysis of temperature ranges and differences. Front. Public Health 11, 1287821 (2023).
Jiang, Y. et al. Cold spells and the onset of acute myocardial infarction: a nationwide case-crossover study in 323 Chinese cities. Environ. Health Perspect. 131, 87016 (2023).
Miao, H. et al. Relationship between temperature and acute myocardial infarction: a time series study in Xuzhou, China, from 2018 to 2020. BMC Public Health 24, 2645 (2024).
Li, N. et al. Associations of apparent temperature with acute cardiac events and subtypes of acute coronary syndromes in Beijing. China. Sci. Rep. 11, 15229 (2021).
Li, C.-Y. et al. Weather impact on acute myocardial infarction hospital admissions with a new model for prediction: a nationwide study. Front. Cardiovasc. Med. 8, 715629 (2021).
Huang, J. et al. Low ambient temperature and incident myocardial infarction with or without obstructive coronary arteries: a Chinese nationwide study. Eur. Heart J. 46, 439–450 (2025).
Bäck, M. & Hofmann, R. Myocardial infarction and low ambient temperature: does presence or absence of stenoses make a difference?. Eur. Heart J. 46, 451–453 (2025).
Tian, L. et al. The effects of interaction between particulate matter and temperature on mortality in Beijing. China. Environ. Sci. Process. Impacts 20, 395–405 (2018).
Han, Y. et al. Association between synoptic types in Beijing and acute myocardial infarction hospitalizations: A comprehensive analysis of environmental factors. Sci. Total Environ. 934, 173278 (2024).
Bhaskaran, K. et al. Effects of ambient temperature on the incidence of myocardial infarction. Heart 95, 1760–1769 (2009).
Analitis, A. et al. Effects of cold weather on mortality: results from 15 European cities within the PHEWE project. Am. J. Epidemiol. 168, 1397–1408 (2008).
Chen, R. et al. Association between ambient temperature and mortality risk and burden: time series study in 272 main Chinese cities. BMJ 363, k4306 (2018).
Curriero, F. C. et al. Temperature and mortality in 11 cities of the eastern United States. Am. J. Epidemiol. 155, 80–87 (2002).
Chen, K. et al. Temporal variations in the triggering of myocardial infarction by air temperature in Augsburg, Germany, 1987–2014. Eur. Heart J. 40, 1600–1608 (2019).
Cai, J. et al. The cold effects on circulatory inflammation, thrombosis and vasoconstriction in type 2 diabetic patients. Sci. Total Environ. 568, 271–277 (2016).
Shea, D. J., Ockene, I. S. & Greene, H. L. Acute myocardial infarction provoked by a cold pressor test. Chest 80, 649–651 (1981).
Mohammad, K. N., Chan, E. Y. Y., Wong, M. C. S., Goggins, W. B. & Chong, K. C. Ambient temperature, seasonal influenza and risk of cardiovascular disease in a subtropical area in southern China. Environ. Res. 186, 109546 (2020).
Takagi, H. et al. Wind and coronary artery disease. Eur. J. Prev. Cardiol. 27, 2259–2263 (2020).
Lin, S. et al. Are the current thresholds, indicators, and time window for cold warning effective enough to protect cardiovascular health?. Sci. Total Environ. 639, 860–867 (2018).
Zhang, Y. et al. The association between short-term apparent temperature exposure and human coagulation: a time-series study from Beijing, 2014–2023. Environ. Int. 195, 109262 (2025).
Gasparrini, A. et al. Mortality risk attributable to high and low ambient temperature: a multicountry observational study. Lancet 386, 369–375 (2015).
Noelke, C. et al. Increasing ambient temperature reduces emotional well-being. Environ. Res. 151, 124–129 (2016).
Saeki, K. et al. Stronger association of indoor temperature than outdoor temperature with blood pressure in colder months. J. Hypertens. 32, 1582–1589 (2014).
Kunst, A. E., Groenhof, F. & Mackenbach, J. P. The association between two windchill indices and daily mortality variation in the Netherlands. Am. J. Public Health 84, 1738–1742 (1994).
Guo, Y. et al. Temperature variability and mortality: a multi-country study. Environ. Health Perspect. 124, 1554–1559 (2016).
Author information
Authors and Affiliations
Contributions
Conceptualization: LQ, WQ; Methodology: LQ, WQ, JZ; Formal Analysis: LQ, WQ, JZ; Investigation: LQ, WQ, JF; Data Curation: LQ, WQ, QZ; Writing – Original Draft Preparation: LQ, WQ, DW, JZ; Writing – Review and Editing: LQ, WQ, XZ, XW; Supervision: XW; Project Administration: LQ, WQ, XZ, XW.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Qiu, L., Qiu, W., Zhang, J. et al. Impact of autumn–winter wind speed and low temperatures on acute coronary syndrome: a 5-year single-center study in Beijing. Sci Rep 16, 4427 (2026). https://doi.org/10.1038/s41598-025-34432-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-34432-2