Abstract
This study aimed to investigate the efficacy of intermittent theta burst stimulation (iTBS) combined with neuromuscular electrical stimulation (NMES) in the treatment of dysphagia after stroke and the changes of brain function.55 participants were randomly assigned to iTBS combined with NMES group (n = 18),iTBS group (n = 18) or NMES group(n = 19). All groups received two-week conventional swallowing therapy.On this basis, the iTBS combined with NMES group received a two-week iTBS combined with NMES treatment, the iTBS group received iTBS combined with sham NMES treatment, the NMES group received sham iTBS combined with NMES treatment. Before and after treatment, Standard Swallowing Function Assessment Scale (SSA), Penetration-Aspiration Scale(PAS), Functional Oral Intake Scale(FOIS), Yale Pharyngeal Residual Severity Scale(YPR-SRS) and swallowing quality of life questionnaire (SWAL-QOL) were used to assess swallowing function. In addition, functional near-infrared spectroscopy was used to investigate participants’ brain function.After treatment, compared with NMES or iTBS group, iTBS combined with NMES group showed significant improvement in SSA, SWAL-QOL, FOIS, PAS, and YPR-SRS scores, increased the activation of Broca (P = 0.033, with FDR corrected) and temporopolar cortex (P = 0.009, with FDR corrected), and improved the functional connectivity between rois.
Similar content being viewed by others
Introduction
Dysphagia is a common complication after stroke with an incidence ranging from 37% to 78%1, which may lead to aspiration pneumonia, malnutrition and other complications, affecting the quality of life2.The conventional treatment of dysphagia includes swallowing function training, catheter balloon dilatation, acupuncture and so on. These methods have a long intervention period, slow effect, and uncertain clinical efficacy. Therefore, the search for effective treatment is the current research hotspot.
Intermittent theta burst stimulation (iTBS) is a special transcranial magnetic stimulation mode emerging in recent years. Its stimulation pulse frequency usually matches the natural activity frequency of cerebral cortical neurons, which is easy to be accepted and responded to by the cerebral cortex3.On the one hand, iTBS can not only change the excitability of the stimulated site and its surrounding and even distant regions4, but also regulate the connectivity of the swallowing network. On the other hand, iTBS also activated corticobulbar pathways involved in the motor control and coordination of swallowing muscles, thereby contributing to the improvement of swallowing function5.However, the effect of iTBS on neural plasticity induction is different in different stages of stroke6. Moreover, the effect may not last for a long time, and relevant training is needed to further consolidate the plasticity effect of the central nervous system.Neuromuscular electrical stimulation (NMES) can directly promote oropharyngeal muscle contraction, and generate sensory stimulation that can also feed back to the central nervous system. Long-term application of NMES is beneficial to the recovery of swallowing related cortical neural plasticity in stroke patients7.However, NMES cannot promote motor learning and coordination of complex movements8.In addition, with aging, muscle plasticity decreases. The regulation of muscle by a single NMES becomes more difficult9.Swallowing requires the coordination of related muscle groups and nerves, and it is difficult to comprehensively improve dysphagia with a single intervention technique. Therefore, the combination of the two can target different aspects of the swallowing process to achieve functional complementarisation and promote central remodeling and peripheral control. At present, combination therapy is widely used in the recovery of limb motor function after stroke, while there are few reports on the treatment of patients with dysphagia after stroke, and its efficacy needs to be further clarified.
Functional near-infrared spectroscopy (fNIRS) is an emerging neuroimaging technique in the field of neuroscience in recent years. It has good temporal and spatial resolution, and is easier to carry and manipulate, which can better understand the neural control of swallowing, so as to construct better intervention programs. At present, a number of studies have applied fNIRS to the study of swallowing function10,11,12. Therefore, This study aims to investigate the efficacy of iTBS combined with NMES in patients with post-stroke dysphagia (PSD) and explored the underlying clinical brain mechanisms by fNIRS.
Results
Baseline characteristics of the patients
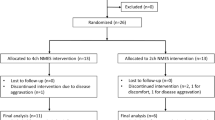
Of the 61 patients with PSD initially recruited,55 were eligible for inclusion in this study (we excluded 4 patients who did not meet inclusion criteria and 2 patients who refused participate in the trial).No one dropped out halfway through enrollment (Fig. 1).The overall characteristics of participants’ are shown in Table 1. No significant differences in age, disease duration, sex, stroke type, lesion location, and stimulation side were observed between the three groups. In addition, clinical outcomes and brain function at baseline did not differ significantly between the three groups.
Participant flow diagram.
Clinical outcomes
Effect on the swallowing function
Significant time and intervention interaction effects were observed in the scores of SSA (F = 16.852, p < 0.001), SWAL-QOL (F = 11.065, p < 0.001), FOIS (F = 16.888, p < 0.001), PAS(F = 9.043, p < 0.001) and YPR-SRS(F = 5.986, p = 0.005).Simple effect analysis showed that there were significant differences in SSA scores, SWA-QOL scores, FOIS scores, PAS scores, and YPR-SRS scores between the three groups of patients after the intervention (P < 0.05, after Bonferroni correction) (see Fig. 2).
The post hoc power analysis for the primary endpoint, the SSA score, revealed a statistical power of 0.91.
The improvement in SSA (A), SWAL-QOL (B), FOIS(C), PAS(D) and YPR-SRS(E) scores before and after treatment in the iTBS+NMES, iTBS and NMES groups.SSA: Standard Swallowing Function Assessment Scale;SWAL-QOL:swallowing quality of life questionnaire;FOIS:Functional Oral Intake scale;PAS:Penetration-Aspiration Scale;YPR-SRS:Yale Pharyngeal Residual Severity.T0:Baseline before intervention;T1:After intervention.
Brain activation
There was no significant difference in the concentration of HbO2 in each channel between the three groups before intervention (P > 0.05, with FDR corrected).The activation levels in the brain regions of all three groups of patients improved compared to before the intervention.
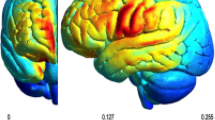
The comparison between the groups after intervention showed that significant activation in channel 12(LTPC, P = 0.009, with FDR corrected) in the iTBS combined with NMES group compared to the NMES group.The comparison between the groups after intervention showed that significant activation in channel 27(Left Broca, P = 0.033, with FDR corrected) in the iTBS combined with NMES group compared to the iTBS group.The comparison between the groups after intervention showed that significant activation in channel 12(LTPC, P = 0.018, with FDR corrected), channel 13(Left Supramarginal gyrus, P = 0.020,with FDR corrected) and channel 19(Right dorsolateral prefrontal cortex, P = 0.021,with FDR corrected) in the iTBS group compared to the NMES group.(Fig. 3).
The cortical activation maps based on analyzed HbO2 beta values:A: Brain activation between the iTBS combined with NMES group and the NMES group after intervention.B:Brain activation between the iTBS combined with NMES group and the iTBS group after intervention.C:Brain activation between the iTBS group and the NMES group after intervention.Only the areas corresponding to the significantly activated channels were shown.Shown in the figure is the P value change.the smaller the P value, the closer the color is to red, and the more intense the activation of the brain region.HbO2: oxygenated hemoglobin. The images were generated by NirSpark software (http://www.hcmedx.cn/en/NewsDetail/3770329.html).
Based on FC analysis between channels
The average FC intensities of all channels before and after the intervention in the three groups are shown in Fig. 4. There was no statistically significant difference in the average intensity of FC across all channels before the intervention in the three groups. (P < 0.05, with FDR corrected).After 2 weeks of intervention, the average FC intensity of all channels in the three groups was increased compared with that before intervention, and further study found that compared with the iTBS or NMES group, the average FC intensity of all channels in the iTBS combined with NMES group was significantly increased, and the difference was statistically significant (P < 0.05, with FDR corrected).
The average of functional connectivity (FC) intensity of all channels in the three groups before and after intervention. (a)the FC matrix of the NMES group before intervention.(b)the FC matrix of the iTBS group before intervention.(c)the FC matrix of the iTBS+NMES group before intervention.(d)the FC matrix of the NMES group after intervention.(e)the FC matrix of the iTBS group after intervention.(f)the FC matrix of the iTBS+NMES group after intervention.
Roi-based FC analysis
Before intervention, there was no significant difference in FC between rois between the three groups (P > 0.05, with FDR corrected).the FC between rois in the three groups was increased compared with that before intervention.
Additionally, Comparison of the brain functional connection intensity post-treatment between the groups revealed that compared to the NMES group, the iTBS combined with NMES group showed significant strengthening of functional connections between LPFC ~ RPFC(P = 0.004,with FDR corrected)、LPFC ~ RPMC/SMA(P = 0.009,with FDR corrected)、LPFC ~ RTPC(P = 0.049,with FDR corrected)、RPFC ~ RS1(P = 0.038,with FDR corrected)、RPFC ~ LM1(P = 0.020,with FDR corrected)、RPFC ~ RM1(P = 0.048,with FDR corrected)、RPFC ~ LPMC/SMA(P = 0.021,with FDR corrected)、RPFC ~ RPMC/SMA(P = 0.004,with FDR corrected)、RPFC ~ LTPC(P = 0.040,with FDR corrected)、RPFC ~ RTPC(P = 0.023,with FDR corrected)、LM1 ~ RPMC/SMA(P = 0.036,with FDR corrected)、RM1 ~ RPMC/SMA(P = 0.046,with FDR corrected).Compared to the iTBS group, the iTBS combined with NMES group showed significant strengthening of functional connections between LPFC ~ RPFC(P = 0.008,with FDR corrected)、LPFC ~ LS1(P = 0.001,with FDR corrected)、LPFC ~ LM1(P = 0.014,with FDR corrected)、LPFC ~ RM1(P = 0.004,with FDR corrected)、LPFC ~ LPMC/SMA(P = 0.003,with FDR corrected)、LPFC ~ RPMC/SMA(P = 0.001,with FDR corrected)、RPFC ~ LS1(P = 0.041,with FDR corrected)、RPFC ~ LM1(P = 0.043,with FDR corrected)、RPFC ~ RM1(P = 0.010,with FDR corrected)、RPFC ~ LPMC/SMA(P = 0.006,with FDR corrected)、RPFC ~ RPMC/SMA(P = 0.001,with FDR corrected)、RPFC ~ LTPC(P = 0.004,with FDR corrected)、LS1 ~ RPMC/SMA(P = 0.020,with FDR corrected)、RPMC/SMA ~ LTPC(P = 0.025,with FDR corrected).Compared to the NMES group, the iTBS group showed significant strengthening of functional connections between LS1 ~ RTPC(P = 0.014,with FDR corrected)、LM1 ~ RTPC(P = 0.011,with FDR corrected).
Safety
No significant adverse events occurred during the clinical research.
Discussion
In this randomized controlled trial, our results showed that iTBS combined with NMES effectively improved SSA scale scores, SWAL-QOL scale scores, PAS scores, FOIS scores and YPR-SRS scores in patients with dysphagia after stroke.In addition, compared with either iTBS or NMES alone, iTBS combined with NMES could significantly activate Broca and TPC, and significantly enhance FC between rois.
Compared with iTBS or NMES, iTBS combined with NMES could significantly improve the scores of SSA, SWAL-QOL, PAS, FOIS and YPR-SRS, and promote the improvement of swallowing function.It has been confirmed that the combined approach is more effective in improving PSD than single nerve stimulation therapy or traditional dysphagia therapy13, which can better promote and induce cortical plasticity and reorganization of the central nervous system to improve swallowing function14.Michou et al.15 found that combining peripheral electrical stimulation with magnetic stimulation could reduce the PAS score, improve the biomechanics of swallowing, increase the excitability of the pharyngeal cortex, and lower the PAS score.Song et al.16 found that the combined treatment of NMES and rTMS could reduce the SSA score and the grade of the drinking test, increase the FOIS score, effectively improve the swallowing function of patients, alleviate swallowing disorders, and enhance the quality of life of patients.A previous study17 have confirmed that, compared with a single neural regulation technique, the combined therapy using transcranial magnetic stimulation can significantly reduce the amount of post-swallowing pharyngeal secretions and food residues.In addition to seriously affecting the daily eating of patients with cerebral infarction, leading to a significant increase in the risk of swallowing and asphyxia, long-term dysphagia also seriously affects the quality of life of patients18. Xu et al.19 used HF-rTMS combined with electrical stimulation to treat 72 patients with PSD, and found that it could significantly improve the SWAL-QOL score and promote the improvement of the prognosis of patients.Fan et al.20found that compared with NMES treatment alone, rTMS combined with NMES treatment in patients with cerebral infarction can significantly improve SWAL-QOL scores, and it is speculated that this combined therapy can improve the quality of life by improving cerebral arterial blood flow, promoting the recovery of neurological function and improving swallowing function.The results of the above studies are consistent with those of the present study.
Compared with either iTBS or NMES alone, iTBS combined with NMES could significantly activate Broca and TPC.TPC, is located in the anterior part of the superior temporal gyrus and the middle temporal gyrus, which is involved in the default mode network and limbic system. It participates in the initiation of swallowing21.Previous studies have observed significant activation of the middle temporal gyrus, inferior frontal gyrus, pre- and postcentral gyrus, and SMA during swallowing in healthy subjects using functional magnetic resonance imaging (fMRI)22,which indicate that TPC is involved in multiple processes such as the preparation and execution of swallowing, and works in coordination with other brain regions to ensure that swallowing proceeds safely and smoothly.Broca is involved in forming the mirror neuron system.Previous studys23 have found that there is a strong activation in this area of Broca’s when performing the swallowing task.Mosier et al.24 were the first to conduct a functional magnetic resonance imaging study on the swallowing function of 8 healthy adults. The participants underwent an autonomous swallowing test involving the intake of water and saliva to identify the brain regions involved in swallowing. The results showed activation in multiple brain regions, including Broca’s area and superior temporal gyrus.Although no studies have explored the effects of iTBS combined with NMES on brain regions related to swallowing, previous studies have explored the effects of central combined with peripheral treatments on functional recovery after stroke.Through fMRI, Luo et al.25observed that rTMS combined with repetitive peripheralmagnetic stimulation could affect the remodeling of brain function by promoting the functional reorganization of important brain regions in the sensorimotor network and default mode network of patients with cerebral hemorrhage. The above studies show that the central combined with peripheral treatment can more effectively activate the relevant brain areas, which is consistent with the present study.
The results of this study also indicate that the average brain functional connectivity values of PSD patients who received iTBS combined with NMES treatment were significantly higher than those of patients who only received NMES or iTBS treatment. Moreover, the functional connection values between LPFC ~ RPMC/SMA、LPFC ~ RTPC、RPFC ~ RS1、LPFC ~ LS1、RPFC ~ LM1、RPFC ~ RM1、RPFC ~ LPMC/SMA、RPFC ~ RPMC/SMA、RPFC ~ LTPC、RPFC ~ RTPC、LM1 ~ RPMC/SMA、RM1 ~ RPMC/SMA、LPFC ~ RPFC、LPFC ~ LM1、LPFC ~ RM1、LPFC ~ LPMC/SMA、RPFC ~ LS1、LS1 ~ RPMC/SMA、RPMC/SMA ~ LTPC were significantly higher than those in the NMES or iTBS treatment.Previous studies26 have confirmed that various bilateral cortical and subcortical structures such as the precentral gyrus, postcentral gyrus, SMA, TPC and insular lobe are intertwined with each other to form a complete neural network, which together play an extremely important role in swallowing.Mihai et al.27used a dynamic causal model to demonstrate that after stimulation is delivered to one or both of the SMA and M1 S1 simultaneously, the two regions are likely to have bidirectional connections for swallowing.The PFC is involved in processing the initial sensory input and regulates the execution of relevant actions through FC between it and the motor cortex28,A study29 has shown that when swallowing in a manner close to the natural state, more subregions of the prefrontal cortex are activated.So far, no study has shown that iTBS combined with NMES can improve the FC of brain regions related to swallowing, but preliminary studies have explored the improvement of FC of brain regions in patients with dysphagia after iTBS or NMES treatment alone. Therefore, the combined treatment may enhance the FC of brain regions related to swallowing to improve swallowing function.Previous studies have confirmed30 that iTBS on the surahyoid muscle motor cortex can promote the FC increase of sensorimotor brain networks (bilateral primary sensorimotor cortex and paracentcentral lobule) and non-sensorimotor brain networks (frontal lobe, temporopolar and occipital lobe) in patients with PSD.Choi et al.31used 5 Hz rTMS to treat the SMA of a patient with severe functional dysphagia, and the connection recovery between the SMA and the precentral gyrus was observed by fMRI in the swallowing task 3 months after the treatment.The research conducted by Jakub et al.32 indicates that the peripheral sensory input information triggered by electrical stimulation can regulate the dynamic network connections between the sensory-motor cortex.
This study has confirmed that the combined treatment of iTBS and NMES can effectively promote the recovery of swallowing function in stroke patients. For the first time, we utilized fNIRS technology to reveal that this combined therapy can significantly enhance the activation level and functional connectivity of the swallowing-related cortex, providing a mechanistic explanation at the neurophysiological level for the effectiveness of this combined strategy.In terms of clinical outcomes, the combined treatment group showed significantly greater improvement in scores on standardized scales (such as SSA, FOIS) compared to the single NMES group. These behavioral improvements were significantly correlated with the recovery of pharyngeal muscle function33,34, collectively supporting the enhancement of peripheral muscle group function.Based on the above findings, the results of this study provide strong preliminary evidence for the hypothesis that “iTBS combined with NMES promotes functional recovery through a central-peripheral synergy mechanism”.
Limitations
This study has certain limitations, Firstly, this is a randomized controlled trial conducted in a single center. Although the post-hoc test power for our primary outcome was relatively high (0.91), the results still need to be treated with caution when generalized to a broader population. Therefore, In the future, multi-center, large-sample, prospective randomized controlled trials are needed to further explore the best treatment for patients with PSD.
Second, fNIRS is limited in probing deep brain activity, in the future, fNIRS can be used in combination with other neuroimaging techniques to obtain a more accurate and comprehensive understanding of brain activity.In addition, patients will need to be followed and evaluated to explore the subsequent efficacy of this combination therapy.Thirdly, this study did not measure muscle function indicators, making it impossible to distinguish the independent effects of central remodeling and peripheral muscle strengthening. Therefore, future research should focus on combining neuroimaging with peripheral physiological measurements, in order to systematically reveal the mechanism of action of different components in the combined therapy.In summary, This study demonstrates that 2 weeks of iTBS combined with NMES can improve swallowing function and reshape brain areas in patients with dysphagia after stroke. This closed-loop rehabilitation model deserves further clinical research and implementation.
Materials and methods
Ethical considerations
The study protocol was approved by the Ethics Committee of the First Affiliated Hospital of Zhengzhou University (2024-KY-0579-002).In addition, the trial has been registered on June 11, 2024 at ClinicalTrials.gov (NCT06464835) and was conducted in accordance with Good Clinical Practice and the regulatory standards of the Helsinki Declaration.All patients received a full explanation of the experimental procedures, and they acknowledged their right to quit at any time during the study.
Study design and participants
This study was designed as a two-arm, randomized, prospective clinical trial conducted in accordance with Consolidated Standards of Reporting Trials.The participants in this study were recruited from inpatients after stroke who were admitted to the Rehabilitation Medicine Department of the First Affiliated Hospital of Zhengzhou University from August 2024 to May 2025.The inclusion criteria:1)age between 18 and 80 years;2)First-episode stroke confirmed by magnetic resonance imaging or computed tomography;3)dysphagia confirmed by videofluoroscopic swallowing study;4)dysphagia lasts from 2 weeks to 6 months;5)no cognitive impairment;6)cranial integrity without craniotomy and/or craniectomy;7)signing informed consent.The exclusion criteria:1)dysphagia caused by other diseases;2)iTBS or NMES contraindications;3)the condition of the participants was unstable or worsened.The shedding criteria: (1) patients self-withdraw during the program; (2) patient develops a worsening condition or other complications during the trial that prevents continuation of this study.
Sample size
The sample size was estimated by PASS software, and a minimum sample size of 28 (14 individuals per group) was needed with the following parameters: 1-β = 0.90, α = 0.05.Since there was no study examining the efficacy of this combined therapy to patients with PSD, the effect size was based on the between-group difference in SSA scores from previous similar studies35. We increased the sample size to 18 individuals per group to take into account the 20% possible attrition.
Randomization and masking
Participants were randomly allocated to NMES group, iTBS group and iTBS combined with NMES group according to the block randomization sequence created by an investigator not involved in the study, with assignment concealed by sealed opaque envelopes. Participants and evaluators were unaware of the group assignments.
Interventions
Three groups received conventional swallowing rehabilitation training performed by a qualified therapist for 30 min per day, 5 days a week and last for 2 weeks.On this basis, the NMES group received sham iTBS combined with NMES stimulation, the iTBS group received iTBS combined with sham NMES stimulation, the iTBS combined with NMES group received iTBS combined with NMES stimulation.Two rehabilitation physicians were responsible for TMS treatment and NMES treatment, respectively.
Conventional swallowing therapy included tongue resistance training, oral-facial muscle training, Oral sensory training, airway protection training, and breathing training for 30 min each time once a day for 2 weeks.
Intermittent theta burst stimulation
We conducted iTBS treatment with 8-shaped coil connected to a YD-MT600 stimulator (Henan Xiangyu Medical Company, China).The coil was placed 2–4 cm anterior to the skull, 4–6 cm lateral to the skull, and a single pulse stimulation was delivered at 80% resting motor threshold (RMT) stimulation intensity to find the representative area of the motor cortex of the mylohyoid muscle, which was the maximum motor evoked potential (MEP).In addition, the RMT was further determined by applying single pulse stimulation to this area.If the healthy side and the affected side could not be determined, the hemisphere requiring a higher intensity evoked MEP was selected as the affected side36.Stimulus protocal was delivered at an intensity of 80% RMT,3 pulses of 50 Hz bursts repeated at 5 Hz (2 s on and 8 s off) for a total of 189 s (600 pulses) on the mylohyoid muscle motor cortex of the affected side.The sham iTBS used exactly the same equipment parameters and scalp positioning points as the real stimulation, only rotating the stimulation coil by 90°. To mask the sound produced by the coil’s operation, all participants wore professional noise-cancelling earplugs with a rated noise reduction value of 33 dB, and at the same time, wide-band white noise with a sound pressure level of 75 dB was played through the indoor speakers. This auditory masking scheme was verified through pre-experiments and was found to be effective in masking the “clicking sound” of the coil.The treatment was given once a day, 5 times a week for 2 weeks.
Neuromuscular electrical stimulation
The electrical stimulator used was neuromuscular electrical stimulator XY-K-TY-I (Henan Xiangyu Medical Company, China) with two-way square wave, a wave width of 700 ms, and the stimulation frequency was 50 Hz.The output intensity was 0 to 25 mA according to the patient’s tolerance, and the stimulating electrodes set on the movement points of the mandibular hyoid muscles on both sides.The intensity of the real stimulation was set by asking the participants to provide continuous feedback and perform continuous swallowing. The current was adjusted until a clear and visible contraction of the target muscle was observed, indicating that the current was at an appropriate level. During the sham stimulation, the frequency was set at 1 Hz, and the intensity was gradually increased from zero until the patient reported a mild, irregular skin tingling or slight tremor, and the researcher confirmed that this intensity did not cause any muscle contractions.The treatment lasted for 30 min. The treatment was given once a day, 5 times a week for 2 weeks.
Outcome measurements
Patients’ swallowing function were evaluated before and after two-week intervention.The primary outcome measure was standardized swallowing assessment (SSA).The SSA scale consists of three parts. The total score on this scale ranges from 18 to 46, with higher scores indicating worse swallowing function37.In addition, fNIRS was used to measure cortical activation and functional connectivity (FC) changes during repeated saliva swallowing before and after two-week intervention.
The secondary outcome measures were Rosenbek Penetration-aspiration Scale (PAS), Functional Oral Intake Scale (FOIS), Yale Pharyngeal Residual Severity Scale (YPR-SRS) and swallowing quality of life questionnaire (SWAL-QOL).Scores on the PAS scale range from 1 to 8, with higher scores indicating more severe aspiration and less safe swallowing38.The FOIS scale is divided into 7 points, each point is scored, which can assess the patient’s oral feeding function39.YPR-SRS scores range from 1 to 5, with higher scores indicating more severe residual disease40.The SWAL-QOL scale has 44 items, with higher scores indicating better quality of life41.
Functional near-infrared spectroscopy
Task-state data acquisition was performed using NirSmart (Huichuang. China), a 39-channel fNIRS system.The acquisition headcap was designed based on the 10/20 international standard lead system and consists of 39 channels with 16 sources and 16 detectors (Fig. 5 A).Based on Brodmann’s system, the regions of interest (ROI) in this study were the prefrontal cortex (PFC)(The left included channels 8, 9, 10, 24, 25, and 26, and the right included channels 5, 6, 7, 19, 21, and 23)、primary somatosensory cortex (S1)(The left included channels 14 and 36, and the right included channels 17 and 16)、primary mortor cortex (M1)(The left included channels 34 and 37, and the right included channels 30 and 33)、temporopolar cortex (TPC) (The left included channels 11 and 12, and the right included channels 3 and 4)、pre-motor/supplementary motor area (PMC/SMA)(The left included channels 35、39、38、29 and 28, and the right included channels 15、18、31、32 and 1).The information of the corresponding channels of each ROI was extracted for subsequent analysis.
fNIRS 36 channels distribution map and test task paradigm.A: distribution of fNIRS 39 channels. B: testing procedure of fNIRS in patients with dysphagia after stroke. fNIRS: functional near-infrared spectroscopy; RSST: repeated swallowing of saliva.
In a quiet treatment room, the subjects were rested in a seated position for 5 min. The fNIRS head cap was then worn. The subjects were instructed to sit quietly and relax for 30 s before performing the swallowing task (Figure. 5B).
fNIRS data processing
NirSpark software was used to analyze fNIRS data.The data of patients with right-side lesion were mirrored to align with the corresponding fNIRS channels on the opposite side.After inversion, the unaffected side of all patients were located on the right side, and the affected side were located on the left side.
The pre-processing was performed in the Preprocess module of NirSpark, and the spline interpolation method was used to correct motion artifacts. The standard deviation threshold of the signal was set as 6, and the peak threshold was set as 0.5. 0.01–0.1 Hz bandpass filtering to remove physiological fluctuations noise; The Beer-Lamber law was modified to convert light intensity data into oxygenated hemoglobin (HbO2) concentration relative changes.Block averaging analysis and linear correction were performed in BlockAvg module. The initial 30 s resting state of the task was removed, and the block averaging analysis was performed on the three repeated task blocks to obtain the 40 s task block (the first 20 s was the swallowing task, and the last 20 s was the rest).The general linear model was used to analyze the correlation between blood oxygen changes and timing tasks, and the beta value reflecting the activation of cerebral cortex when subjects performed swallowing tasks was calculated.In addition, in order to evaluate the brain FC, the Network module was used to extract the changes in HbO2 concentration at each time point during the swallowing task, and the Pearson correlation coefficient of HbO2 concentration on the time series was analyzed, which was defined as FC strength after Fisher-Z transformation.The t test was used to compare the differences of brain activation and FC between the three groups, and p < 0.05 was considered statistically significant.The p-values corresponding to all fNIRS-related results have been adjusted according to the FDR method and are reported.
Statistical analysis
Statistical analysis was performed using SPSS 26.0 software. The measurement data were normally distributed and were expressed as mean ± standard deviation. The comparison between the three groups was conducted using the one-way ANOVA. Counting data are presented as cases (%), and comparisons between groups are conducted using the χ2 test.The comparison between the three sets of repeated measurement data was conducted using repeated measures analysis of variance. If the interaction effect was significant, we used the Bonferroni correction method to perform multiple comparison corrections for the pairwise comparisons in the simple effect analysis, with a significance level set at 0.05. In addition, we conducted a post hoc power calculation based on the actual sample size, using the G*Power software (version 3.1.9.7).
Data availability
Relevant data are reasonably available from the corresponding author.
References
Martino, R. et al. Dysphagia after stroke: incidence, diagnosis, and pulmonary complications[J]. Stroke 36 (12), 2756–2763. https://doi.org/10.1161/01.STR.0000190056.76543.eb (2005).
Kocica, J. et al. Screening for dysphagia in patients with relapsing-remitting multiple sclerosis[J]. Mult Scler. Relat. Disord. 83, 105418. https://doi.org/10.1016/j.msard.2023.105418 (2024).
Hoy, K. E. et al. Enhancement of working memory and Task-Related oscillatory activity following intermittent theta burst stimulation in healthy Controls[J]. Cereb. Cortex. 26 (12), 4563–4573. https://doi.org/10.1093/cercor/bhv193 (2016).
Li, Y. et al. Cerebral functional manipulation of repetitive transcranial magnetic stimulation in cognitive impairment patients after stroke: an fMRI Study[J]. Front. Neurol. 11, 977. https://doi.org/10.3389/fneur.2020.00977 (2020).
Mistry, S., Michou, E., Rothwell, J. & Hamdy, S. Remote effects of intermittent theta burst stimulation of the human pharyngeal motor system[J]. Eur. J. Neurosci. 36 (4), 2493–2499. https://doi.org/10.1111/j.1460-9568.2012.08157.x (2012).
Grefkes, C. & Fink, G. R. Disruption of motor network connectivity post-stroke and its noninvasive neuromodulation[J]. Curr. Opin. Neurol. 25 (6), 670–675. https://doi.org/10.1097/WCO.0b013e3283598473 (2012).
Zhang, Y. et al. Neuromuscular electrical stimulation improves swallowing initiation in patients with post-stroke dysphagia[J]. Front. Neurosci. 16, 1011824. https://doi.org/10.3389/fnins.2022.1011824 (2022).
Paillard, T. Combined application of neuromuscular electrical stimulation and voluntary muscular contractions[J]. Sports Med. 38 (2), 161–177. https://doi.org/10.2165/00007256-200838020-00005 (2008).
Carnaby, G. D., LaGorio, L., Silliman, S. & Crary, M. Exercise-based swallowing intervention (McNeill dysphagia Therapy) with adjunctive NMES to treat dysphagia post-stroke: A double-blind placebo-controlled trial[J]. J. Oral Rehabil. 47 (4), 501–510. https://doi.org/10.1111/joor.12928 (2020).
Kober, S. E. & Wood, G. Hemodynamic signal changes during saliva and water swallowing: a near-infrared spectroscopy study[J]. J. Biomed. Opt. 23 (1), 1–7. https://doi.org/10.1117/1.JBO.23.1.015009 (2018).
Zhang, X. et al. Modulating swallowing-related functional connectivity and behavior via modified pharyngeal electrical stimulation: A functional near-infrared spectroscopy evidence[J]. Front. Neurol. 13, 1006013. https://doi.org/10.3389/fneur.2022.1006013 (2022).
Kober, S. E. et al. Hemodynamic signal changes accompanying execution and imagery of swallowing in patients with dysphagia: A multiple Single-Case Near-Infrared spectroscopy Study[J]. Front. Neurol. 6, 151. https://doi.org/10.3389/fneur.2015.00151 (2015).
Banda, K. J. et al. Comparative effectiveness of combined and single neurostimulation and traditional dysphagia therapies for Post-Stroke dysphagia: A network Meta-Analysis[J]. Neurorehabil Neural Repair. 37 (4), 194–204. https://doi.org/10.1177/15459683231166940 (2023).
Kusumaningsih, W., Lestari, N. I., Harris, S., Tamin, S. & Werdhani, R. A. The effectivity of pharyngeal strengthening exercise, hyolaryngeal complex range of motion exercise, and swallowing practice in swallowing function of ischemic stroke patients with neurogenic dysphagia[J]. J. Exerc. Rehabil. 15 (6), 769–774. https://doi.org/10.12965/jer.19.38652.326 (2019).
Michou, E. et al. Targeting unlesioned pharyngeal motor cortex improves swallowing in healthy individuals and after dysphagic stroke[J]. Gastroenterology 142 (1), 29–38. https://doi.org/10.1053/j.gastro.2011.09.040 (2012).
Song, C., Li, G. & Zhang, J. Observation on the effect of High-frequency rTMS combined with NMES in treating dysphagia after Stroke[J]. Med. J. Natl. Defending Forces Southwest. China. 28 (11), 1066–1068. https://doi.org/10.3969/j.issn.1004-0188.2018.11.025 (2018).
Zhang, Y. et al. Effectiveness of repeated peripheral magnetic stimulation combined with repeated transcranial magnetic stimulation for swallowing disorders after stroke[J]. Chin. J. Rehabilitation. 40 (6), 344–348. https://doi.org/10.3870/zgkf.2025.06.004 (2025).
Takizawa, C., Gemmell, E., Kenworthy, J. & Speyer, R. A systematic review of the prevalence of oropharyngeal dysphagia in Stroke, parkinson’s Disease, alzheimer’s Disease, head Injury, and Pneumonia[J]. Dysphagia 31 (3), 434–441. https://doi.org/10.1007/s00455-016-9695-9 (2016).
Xu, J., Zhao, W., Wang, B. & Chen, M. Effects of high-frequency repetitive transcranial magnetic stimulation combined with electrical stimulation on dysphagia after stroke[J]. Chin. J. Prim. Med. Pharm. 29 (1), 51–55. https://doi.org/10.3760/cma.issn1008-6706.2022.01.011 (2022).
Fan, Y. & Niu, G. The effect of repeated transcranial magnetic stimulation combined with neuromuscular electrical stimulation in the treatment of cerebral infarction[J]. Mod. Instruments Med. Treat. 30 (2). https://doi.org/10.11876/mimt202402015 (2024).
Wu, G. et al. Blood oxygen level-dependent functional magnetic resonance imaging study on the activation mechanism of brain functional areas related to swallowing function[J]. Neural Injury Funct. Reconstruction. 17 (07), 413–415. https://doi.org/10.16780/j.cnki.sjssgncj.20210555 (2022).
Jing, Y. et al. Comparison of activation patterns in mirror neurons and the swallowing network during action observation and execution: A Task-Based fMRI Study[J]. Front. Neurosci. 14, 867. https://doi.org/10.3389/fnins.2020.00867 (2020).
Wang, Z. et al. Effects of different mylohyoid muscle stimulations on swallowing cortex excitability in healthy subjects[J]. Behav. Brain Res. 470, 115055. https://doi.org/10.1016/j.bbr.2024.115055 (2024).
Mosier, K. M., Liu, W. C., Maldjian, J. A., Shah, R. & Modi, B. Lateralization of cortical function in swallowing: a functional MR imaging study[J]. AJNR Am. J. Neuroradiol. 20 (8), 1520–1526 (1999).
Luo, H. & Xu, L. Effect of repetitive transcranial magnetic stimulation combined with repetitive peripheral magnetic stimulation on upper extremities motor function in patients with cerebral hemorrhage:a randomized controlled trial based on resting state-functional Magenetic resonance imaging[J]. Chin. J. Rehabilitation Theory Pract. 30 (9), 1060–1068. https://doi.org/10.3969/j.issn.1006-9771.2024.09.009 (2024).
Martin, R. E., Goodyear, B. G., Gati, J. S. & Menon, R. S. Cerebral cortical representation of automatic and volitional swallowing in humans[J]. J. Neurophysiol. 85 (2), 938–950. https://doi.org/10.1152/jn.2001.85.2.938 (2001).
Mihai, P. G., Otto, M., Platz, T., Eickhoff, S. B. & Lotze, M. Sequential evolution of cortical activity and effective connectivity of swallowing using fMRI[J]. Hum. Brain Mapp. 35 (12), 5962–5973. https://doi.org/10.1002/hbm.22597 (2014).
Hikosaka, O., Nakamura, K., Sakai, K. & Nakahara, H. Central mechanisms of motor skill learning[J]. Curr. Opin. Neurobiol. 12 (2), 217–222. https://doi.org/10.1016/s0959-4388(02)00307-0 (2002).
Zhang, C., Chen, X., Zheng, J., Jiang, Z. & Wu, X. The study of prefrontal cortex function in different swallowing task based on functional near-infrared spectroscopy[J]. Chin. J. Rehabilitation Med. 38 (10), 1372–1378. https://doi.org/10.3969/j.issn.1001-1242.2023.10.007 (2023).
Zhang, G. et al. Intermittent Theta-Burst stimulation reverses the After-Effects of contralateral virtual lesion on the suprahyoid muscle cortex: evidence from dynamic functional connectivity Analysis[J]. Front. Neurosci. 13, 309. https://doi.org/10.3389/fnins.2019.00309 (2019).
Choi, S. & Pyun, S. Repetitive transcranial magnetic stimulation on the supplementary motor area changes brain connectivity in functional Dysphagia[J]. Brain Connect. 11 (5), 368–379. https://doi.org/10.1089/brain.2020.0818 (2021).
Limanowski, J. et al. Action-Dependent processing of touch in the human parietal operculum and posterior Insula[J]. Cereb. Cortex. 30 (2), 607–617. https://doi.org/10.1093/cercor/bhz111 (2020).
Zheng, H. et al. Efficacy of action observation therapy on patients with dysphagia after stroke: A randomized controlled Trial[J]. Dysphagia https://doi.org/10.1007/s00455-025-10866-8 (2025).
Yanagawa, K. et al. Tongue shear wave elastography for bulbar dysfunction in amyotrophic lateral sclerosis[J]. Clin. Neurophysiol. 179, 2111367. https://doi.org/10.1016/j.clinph.2025.2111367 (2025).
Rao, J. et al. Bilateral cerebellar intermittent theta burst stimulation combined with swallowing speech therapy for dysphagia after stroke: A Randomized, Double-Blind, Sham-Controlled, clinical Trial[J]. Neurorehabil Neural Repair. 36 (7), 437–448. https://doi.org/10.1177/15459683221092995 (2022).
Sun, G., Yang, L. & Sun, X. Clinical study of high-frequency repetitive transcranial magnetic stimulation on the representative area of the healthy hemisphere swallowing cortex combined with swallowing rehabilitation training in the treatment of dysphagia after cerebral apoplexy[J]. Chin. J. Rehabilitation. 37 (01), 7–11. https://doi.org/10.3870/zgkf.2022.01.002 (2022).
Christensen, M. & Trapl, M. Development of a modified swallowing screening tool to manage post-extubation dysphagia[J]. Nurs. Crit. Care. 23 (2), 102–107. https://doi.org/10.1111/nicc.12333 (2018).
Rosenbek, J. C., Robbins, J. A., Roecker, E. B., Coyle, J. L. & Wood, J. L. A penetration-aspiration scale[J]. Dysphagia 11 (2), 93–98. https://doi.org/10.1007/BF00417897 (1996).
Crary, M. A., Mann, G. D. C. & Groher, M. E. Initial psychometric assessment of a functional oral intake scale for dysphagia in stroke patients[J]. Arch. Phys. Med. Rehabil. 86 (8), 1516–1520. https://doi.org/10.1016/j.apmr.2004.11.049 (2005).
Rocca, S., Pizzorni, N., Valenza, N., Negri, L. & Schindler, A. Reliability and construct validity of the Yale pharyngeal residue severity rating scale: performance on videos and effect of bolus Consistency[J]. Diagnostics (Basel). 12 (8), 1897. https://doi.org/10.3390/diagnostics12081897 (2022).
Chan, H. F., Ng, M. L., Kim, H. & Kim, D. Y. Swallowing-related quality of life among oral-feeding Chinese patients with parkinson’s disease - a preliminary study using Chinese SWAL-QOL[J]. Disabil. Rehabil. 44 (7), 1077–1083. https://doi.org/10.1080/09638288.2020.1791979 (2022).
Acknowledgements
The authors thank all participants in the study.
Funding
This research was funded by the NHC Key Laboratory of Prevention and Treatment of Cerebrovascular Disease, Medical technologies R & D Program of Henan province (Grant No.LHGJ20220348), and Henan Provincial Department of Science and Technology Key R&D Special Project (241111310600).
Author information
Authors and Affiliations
Contributions
X.Z., H-p. L. and L-g. W. have given substantial contributions to the conception or the design of the manuscript. X.L. were involved in the research conducting and data collection. J.Z. and Y-y. S. performed data sorting and analysis. X.L. and Y.L. have participated to drafting the manuscript, X.Z. supervised the study and revised it critically. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study protocol was approved by the Ethics Committee of the First Affiliated Hospital of Zhengzhou University (2024-KY-0579-002).In addition, the trial has been registered on June 11, 2024 at ClinicalTrials.gov (NCT06464835) and was conducted in accordance with Good Clinical Practice and the regulatory standards of the Declaration of Helsinki.Participants gave informed consent to participate in the study before taking part.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Luo, X., Li, Y., Li, H. et al. Effects of combined intermittent theta burst stimulation with neuromuscular electrical stimulation in the treatment of dysphagia after stroke. Sci Rep 16, 4614 (2026). https://doi.org/10.1038/s41598-025-34668-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-34668-y