Abstract
This study evaluated the long-term efficacy and safety of the widely used drugs entecavir (ETV) and tenofovir alafenamide (TAF), as well as the incidence of HCC.A nonrandomized, prospective, observational analysis included 77 patients with chronic hepatitis B who were assigned to continue ETV or switch TAF. After 240 weeks, the mean changes in serum hepatitis B surface antigen (− 0.365 ± 0.069 log IU/mL vs. 0.301 ± 0.039 log IU/mL, p = 0.39) and hepatitis B core-related antigen (− 0.215 ± 0.092 log IU/mL vs. − 0.195 ± 0.056 log IU/mL) were not significantly different between the ETV and TAF groups. There were also no differences between the two groups in estimated glomerular filtration rate (− 5.407 ± 1.660 vs. − 2.666 ± 1.52, p = 0.240), urinary β2-microglobulin β/creatinine (ETV: 2.330 ± 0.374 at baseline and 2.335 ± 0.257 at 240 weeks; TAF: 2.720 ± 0.073 and 2.123 ± 0.310, p = 0.996 and 0.455, respectively) or urinary N-acetyl-β-D-glucosaminidase/creatinine (ETV: 0.040 ± 0.005 at baseline and 0.044 ± 0.004 at 240 weeks; TAF: 0.049 ± 0.005 and 0.053 ± 0.005, p = 0.642 and 0.684, respectively). Finally, no significant difference was found in the incidence of HCC between the ETV and TAF groups (log-rank test, p = 0.08). In conclusion, the long-term observation of this study demonstrated that ETV and TAF have comparable efficacy and safety.
Clinical trial registration: UMIN000026465.
Similar content being viewed by others
Introduction
Hepatitis B surface antigen (HBsAg) is an important surrogate marker for assessing therapeutic efficacy and predicting prognosis in patients with chronic hepatitis B (CHB)1 and is specified in major guidelines2,3,4,5. Although interferon (IFN) is the most effective therapeutic agent for HBsAg-negative conversion6, its efficacy and tolerability are low7,8,9,10; therefore, IFN therapy is rarely used. Nucleic acid analog preparations are the mainstream treatment for hepatitis B. Although the HBsAg-negative conversion rate is low with nucleic acid analogs alone, studies have reported a good prognosis after HBsAg-negative conversion. In a study by Kim et al.11, of the 110 patients with CHB who achieved HBsAg clearance on nucleic acid analog therapy, only two had hepatocellular carcinoma (HCC), which was lower than the incidence in those with HBsAg-positive (hazard ratio [HR] = 0.09, p < 0.01). Moreover, HBsAg clearance was associated with a good prognosis, even in patients with cirrhosis. In a previous study12, 309 patients with biopsy-confirmed decompensated cirrhosis were enrolled with a mean follow-up of 5.7 years; 32 achieved HBsAg clearance, 1 (3%) of whom died of HCC. However, among patients who remained HBsAg-positive, 11% had HCC, and 20% died. Overall, patients who achieved HBsAg clearance had a lower risk of HCC (p = 0.013) and longer survival (p < 0.001) than those who remained HBsAg-positive. Therefore, in cases where HBsAg-negative conversion is achieved with nucleic acid analog therapy, a significant improvement in prognosis similar to that with IFN therapy can be expected.
It has been reported that HB core-related antigen (HBcrAg) correlates with the amount of cccDNA in liver tissue, regardless of whether nucleic acid analogs are administered or not. There is a lot of evidence regarding the association between HB core-related antigen and hepatocarcinogenesis. In the natural course of events, high HB core-related antigen levels, even with low HBs antigen levels, are a risk factor for hepatocarcinogenesis13, and hepatocarcinogenesis after HBe antigen negativity, particularly in cases with HBe antigen negativity, HBV DNA levels of 2,000–19,999 IU/ml, and normal ALT levels, is associated with hepatocarcinogenesis14. In cases of hepatocarcinogenesis in HBe antigen-negative patients undergoing nucleic acid analog therapy, the amount of HB core-related antigen before treatment and 1 year after treatment is associated with hepatocarcinogenesis15. Furthermore, it has been suggested that even in cases where HBs antigen has been negative, residual HB core-related antigen may pose a risk of cancer16.
Several comparative studies have been conducted on nucleic acid analog formulations. The short-term HBsAg-lowering effect at 48 weeks was higher with tenofovir disoproxil fumarate (TDF) than with entecavir (ETV), while that with TDF and tenofovir alafenamide (TAF) was comparable17,18. Additionally, side effects, such as renal dysfunction and decreased bone density, were significantly lower with TAF than with TDF19,20. However, clinical trials comparing ETV and TAF have not been conducted. ETV is currently used in many patients with hepatitis B due to its antiviral effect and safety; however, the effect of switching from ETV to TAF remains unclear.
In our previous study, the ETV continuation and TAF modification groups showed similar efficacy and safety at 96 weeks21. This study aimed to evaluate the long-term efficacy and safety, including HBsAg and HBcrAg levels.
Methods
Patients
This study included 80 nucleos(t)ide-naïve patients with CHB, defined as HBsAg-positive and HB virus (HBV) DNA-positive for at least 6 months before treatment, who underwent long-term ETV therapy. The inclusion criteria were as follows: (1) patients who were treated with ETV for ≥ 1 year and had an HBV-DNA level of < 2.1 log copies/mL (equivalent to 1.3 log10 IU/mL); (2) HBsAg levels of ≥ 800 IU/mL or 80–800 IU/mL, and variation before the screening test does not exceed − 0.1 log IU/mL/year; (3) those restricted to genotype C and registered.
The exclusion criteria were as follows: previous use of other nucleos(t)ide analogs; use of corticosteroids or immunomodulatory drugs; the presence of antibodies to hepatitis C virus and other likely causes of chronic liver disease, such as autoimmune or alcoholic liver diseases; clinical signs of decompensated cirrhosis or HCC; severe comorbidities, including impaired cardiac or respiratory function; renal dysfunction (estimated glomerular filtration rate [eGFR] < 60); females who were possibly pregnant, expectant mothers, or lactating mothers.
This study was approved by the Ethical Committee of the Kindai University (approval number: 28-271) and conducted per the principles outlined in the Helsinki Declaration of 1964 (2013 revision). Additionally, all patients provided written informed consent. This study was registered in the UMIN Clinical Trials Registry (registration ID number: UMIN000026465) as a prospective comparative study for up to 48 weeks after study initiation. The initial study enrollment was on April 01, 2017 (01/04/2017). In this study, we examined the subsequent long-term course.
Study design
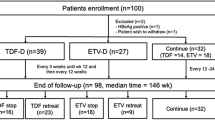
This study was a nonrandomized prospective observational study conducted at a single center. As this was a pilot study, sample size estimation was not performed. Patients were recruited in March 2017 and assigned to either continue ETV (n = 32) or switch to TAF (n = 48). All patients receiving ETV were given the option to switch to TAF, and those who agreed were switched to TAF, while those who did not wish to switch were allowed to continue ETV. During the observation period of up to 240 weeks, one patient in the ETV group and two in the TAF change group discontinued due to hospital transfer. Therefore, a comparative study was conducted between the ETV continuation (n = 31) and TAF change (n = 46) groups (Fig. 1). ETV (Baraclude) was administered orally at 0.5 mg once daily under fasted conditions, and TAF (Vemlidy) was administered orally at 25 mg once daily. The primary efficacy endpoint was the change in serum HBsAg levels from baseline at 240 weeks.
Study design. This was a prospective controlled trial conducted at a single center. Patients were recruited in March 2017 and assigned to the ETV continuation (n = 32) or TAF change (n = 48) groups according to the wishes of ETV continuation patients. During the follow-up period of up to 240 weeks, one patient in the ETV group and two in the TAF change group discontinued the follow-up due to hospital transfer. Finally, a comparative study was conducted between the ETV continuation (n = 31) and TAF change (n = 46) groups. ETV (Baraclude) was administered orally at 0.5 mg once daily under fasted conditions. TAF (Vemlidy) was administered orally at a dose of 25 mg once daily. The primary efficacy endpoint was the change in serum HBsAg levels from baseline at 48 weeks. ETV entecavir, TAF tenofovir alafenamide, HBsAg hepatitis B surface antigen.
Virological analyses and clinical evaluations
The serum levels of HBsAg, HB surface antibody, HB e-antigen (HBeAg), and HB e-antibody were measured using a chemiluminescent immunoassay (CLIA) with the ARCHITECT kit (Abbott Japan, Tokyo, Japan). HBcr antigen levels were measured by chemiluminescent enzyme immunoassay (CLEIA) using the Lumipulse HBcrAg kit (Fujirebio, Kanagawa, Japan). Additionally, HBV-DNA levels were measured using a real-time polymerase chain reaction assay (COBAS TaqMan HBV Test, version 2.0; Roche Diagnostics, Tokyo, Japan)22. HBV genotypes were identified using enzyme-linked immunosorbent assay with monoclonal antibodies against type-specific epitopes in the preS2-region (Institute of Immunology, Tokyo, Japan)23. Moreover, Mac-2 binding protein glycosylation isomer (M2BPGi) levels were measured using the CLIA method with HISCL-5000 (Sysmex, Kobe, Japan). Finally, the eGFR (mL/min/1.73 m2) was calculated using the following equation generated by the Japanese Society of Nephrology: 194 × serum creatinine – 1.094 × (age) – 0.287 × (0.739, for females).
Clinical endpoints were mean changes in HBsAg, HBcrAg, ALT, eGFR, phosphorus, and M2BPGi levels at 240 weeks. Urinary NAG/Cr and β2MG/Cr were also evaluated as markers of proximal tubular damage, and bone mineral density of the lumbar spine and femur were also evaluated. Finally, the incidence of HCC was also examined.
Statistical analyses
Distributions of continuous variables were analyzed using Student’s t-test. Additionally, differences in proportions were tested using Fisher’s exact test. The incidence rates of HCC were compared using the log-rank test with Kaplan–Meier curves. All analyses were performed using SPSS Statistics for Windows, version 11.5 (SPSS Inc., Chicago, IL). Data are expressed as mean ± standard error, and statistical significance was set at p < 0.05.
Results
Clinical characteristics
The patient’s baseline characteristics are presented in Table 1. Overall, 31 and 46 patients in the ETV continuation and TAF change groups infected with genotype C HBV, respectively, were analyzed. The proportion of patients who were HBeAg-positive was 10% (n = 3) and 9% (n = 4) in the ETV continuation and TAF change groups, respectively, with no significant difference between the two groups (p = 1.0). Moreover, the average HBsAg levels were 3.2 logIU/mL and 3.1 logIU/mL in the ETV continuation and TAF change groups, respectively, with no significant difference between the two groups (p = 0.54). Furthermore, the average reduction in HBsAg levels during the most recent year of ETV administration before the introduction of this study was − 0.022 logIU/mL/year and − 0.030 logIU/mL/year in the ETV continuation and TAF change groups, respectively, showing no significant difference (p = 0.57). HBcrAg levels were 3.7 logU/mL and 3.5 logU/mL in the ETV continuation and TAF change groups, respectively, with no significant difference between the two groups (p = 0.39). Finally, the average eGFR was 80 and 78 in the ETV continuation and TAF change groups, respectively, with no significant difference (p = 0.43).
Virological and biological evaluations
The mean changes in serum HBsAg levels at week 240 were − 0.365 ± 0.069 log IU/mL and − 0.301 ± 0.039 log IU/mL in the ETV continuation group and the TAF change group, respectively, and were not significantly different (p = 0.39) (Fig. 2a). The mean changes in HBcrAg levels were − 0.215 ± 0.092 log IU/mL and − 0.195 ± 0.056 log IU/mL in the ETV continuation group and the TAF change group, respectively, and were not significantly different (p = 0.87) (Fig. 2b). The mean changes in serum alanine transaminase (ALT) levels were 1.037 ± 1.042 U/L and − 0.463 ± 2.333 U/L in the ETV continuation group and the TAF change group, respectively, and were not significantly different (p = 0.605) (Fig. 3). Moreover, the M2BPGi level was 0.091 ± 0.035 and 0.117 ± 0.036 in the ETV continuation and TAF change groups, respectively, showing no significant difference (p = 0.610) (Fig. 4).
Comparison of mean changes in HBsAg and HBcrAg levels at 240 weeks. (a) Comparison of mean changes in serum HBsAg levels at 240 weeks after the start of the trial. The mean change in serum HBsAg levels was − 0.365 ± 0.069 log IU/mL and − 0.301 ± 0.039 log IU/mL in the ETV continuation and TAF change groups, respectively, with no significant difference (p = 0.39). (b) The mean changes in HBcrAg levels were − 0.215 ± 0.092 log IU/mL and − 0.195 ± 0.056 log IU/mL in the ETV continuation group and the TAF switch group, respectively, and were not significantly different (p = 0.87). ETV entecavir, TAF tenofovir alafenamide, HBsAg hepatitis B surface antigen, HBcrAg hepatitis B core-related antigen.
Comparison of mean changes in ALT level at 240 weeks. Comparison of mean changes in serum ALT level at 240 weeks after the start of the trial. Serum ALT levels were 1.037 ± 1.042 and − 0.463 ± 2.333 in the ETV continuation and TAF change groups, respectively, showing no significant difference (p = 0.605). ALT alanine transaminase.
Comparison of the mean change in M2BPGi at 240 weeks after the start of the trial. M2BPGi levels were 0.091 ± 0.035 and 0.117 ± 0.036 in the ETV continuation and TAF change groups, respectively, showing no significant difference (p = 0.610). ETV entecavir, TAF tenofovir alafenamide, M2BPGi Mac-2 binding protein glycosylation isomer.
Furthermore, a comparative study of renal function-related parameters was conducted. The mean change in eGFR was − 5.407 ± 1.660 and − 2.666 ± 1.529 in the ETV continuation and TAF change groups, respectively, with no significant difference (p = 0.240) (Fig. 5a). Additionally, the mean change in serum phosphorus level was − 0.085 ± 0.127 mg/dL and − 0.056 ± 0.081 mg/dL in the ETV continuation and TAF change groups, respectively, with no significant difference (p = 0.842) (Fig. 5b). The baseline urinary N-acetyl-beta-D-glucosaminidase/creatinine (NAG/Cr) was 0.040 ± 0.005 vs. 0.044 ± 0.004 at 240 weeks in the ETV continuation group and 0.049 ± 0.005 vs. 0.053 ± 0.005 in the TAF change group, with no significant difference (p = 0.642 and 0.684, respectively) (Fig. 6a). The urinary beta-2-microglobulin (β2MG)/Cr ratio was 2.330 ± 0.374 vs. 2.335 ± 0.257 at 240 weeks in the ETV continuation group and 2.720 ± 0.073 vs. 2.123 ± 0.310 in the TAF change group, with no significant difference (p = 0.996 and 0.455, respectively) (Fig. 6b).
Comparison of mean changes in eGFR and serum phosphorus at 240 weeks. (a) Comparison of mean changes in eGFR at 240 weeks after the start of the trial. The mean change in eGFR was − 5.407 ± 1.660 and − 2.666 ± 1.529 in the ETV continuation and TAF change groups, respectively, showing no significant difference (p = 0.240). (b) Comparison of mean changes in serum phosphorus (P) at 240 weeks after the start of the trial. The mean change in serum P was − 0.085 ± 0.127 and − 0.056 ± 0.081 in the ETV continuation and TAF change groups, respectively, showing no significant difference (p = 0.842). ETV entecavir, TAF tenofovir alafenamide, eGFR estimated glomerular filtration rate.
Comparison of mean values of urinary NAG/Cr and β2MG/Cr at 240 weeks. (a) Comparison of the mean rate of change in urinary NAG/Cr at 240 weeks after the start of the trial. The baseline urinary NAG/Cr levels were 0.040 ± 0.005 vs. 0.044 ± 0.004 at 240 weeks in the ETV continuation group and 0.049 ± 0.005 vs. 0.053 ± 0.005 at 240 weeks in the TAF continuation group, with no significant difference observed (p = 0.642 and 0.684, respectively). (b) Comparison of the mean rate of change in urinary β2MG/Cr at 240 weeks after the start of the trial. The baseline urinary β2MG/Cr levels were 2.330 ± 0.374 vs. 2.335 ± 0.257 at 240 weeks in the ETV continuation group and 2.720 ± 0.073 vs. 2.123 ± 0.310 at 240 weeks in the TAF continuation group, with no significant difference observed (p = 0.996 and 0.455, respectively). ETV entecavir, TAF tenofovir alafenamide, NAG/Cr N-acetyl-beta-D-glucosaminidase/creatinine, β2MG/Cr beta-2-microglobulin/creatinine.
Moreover, the mean bone mineral density was compared at 240 weeks from the beginning of the study. Regarding lumbar spine examination, the baseline value of the ETV continuation group at 240 weeks was 0.903 ± 0.035 g/cm2 vs. 0.814 ± 0.042 g/cm2, while that of the TAF change group was 0.939 ± 0.035 g/cm2 vs. 0.859 ± 0.041 g/cm2, with no significant difference (p = 0.176 and 0.216, respectively) (Fig. 7a). Regarding the femoral examination, the baseline value of the ETV continuation group at 240 weeks was 0.656 ± 0.028 g/cm2 vs. 0.561 ± 0.032 g/cm2, while that of the TAF change group was 0.690 ± 0.023 g/cm2 vs. 0.600 ± 0.033 g/cm2 (p = 0.081 and 0.047, respectively) (Fig. 7b).
Comparison of mean bone mineral density at 240 weeks. (a) Comparison of the lumbar spine: following lumbar spine examination, the baseline values were 0.903 ± 0.035 g/cm2 vs. 0.814 ± 0.042 g/cm2 at 240 weeks in the ETV continuation group and 0.939 ± 0.035 g/cm2 vs. 0.859 ± 0.041 g/cm2 at 240 weeks in the TAF continuation group, with no significant differences (p = 0.176 and 0.216, respectively). (b) Comparison of femur: following the femoral examination, the baseline values in the ETV continuation group were 0.656 ± 0.028 g/cm2 vs. 0.561 ± 0.032 g/cm2 at 240 weeks and 0.690 ± 0.023 g/cm2 vs. 0.600 ± 0.033 g/cm2 at 240 weeks in the TAF continuation group, both showing decreased symptoms (p = 0.081 and 0.047, respectively). ETV entecavir, TAF tenofovir alafenamide.
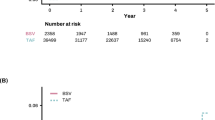
Finally, the incidence of HCC was compared using Kaplan-Meier curves (Fig. 8) (log-rank test). Two patients had HCC in the ETV continuation group; however, no significant difference was found in cumulative HCC incidence between the two groups (p = 0.08). The two patients who had HCC were both relatively young men with advanced fibrosis and low platelet (PLT) values. In both cases, HBsAg levels were lower than those at baseline during HCC onset; however, the reduction in HB core-related antigen (HBcrAg) levels was insufficient (Table 2).
Comparison of HCC incidence rates. The incidence of HCC was compared using Kaplan–Meier curves (log-rank test). Two patients had HCC in the ETV continuation group; however, no significant difference was found in cumulative cancer incidence between the two groups (p = 0.08). HCC hepatocellular carcinoma, ETV entecavir.
Discussion
Several comparative studies have been conducted on nucleic acid analog formulations. The short-term HBsAg-lowering effect at 48 weeks was higher with TDF than with ETV, while that with TDF and TAF were comparable17,18. However, side effects, such as renal dysfunction and decreased bone density, were significantly lower with TAF than with TDF, and TAF was safer19,20. Additionally, clinical trials comparing ETV and TAF have not been conducted, and ETV is currently used in many patients with hepatitis B due to its antiviral effect and safety; however, TAF changes in such patients remain unclear. Recently, various studies have reported cases where ETV was changed to TAF. For instance, Uchida et al.24 assessed HBsAg levels and renal function before and after switching to TAF in 74 patients who received ETV for ≥ 192 weeks and had HBsAg levels of ≥ 100 IU/mL. They found that the change in HBsAg levels at 144 weeks after starting ETV administration was − 0.046 log IU/mL (median value) and − 0.261 log IU/mL at 144 weeks after switching to TAF; the rate of decline was significantly accelerated in the TAF change group (p < 0.001). Moreover, the rate of change in eGFR was − 5.9% at 144 weeks after initiating ETV administration and − 3.2% at 144 weeks after switching to TAF, with no significant difference (p = 0.301). Ogawa et al.25 reported the HBsAg level, HBV-DNA negative conversion rate, and renal function at 144 weeks after switching from ETV to TAF in 174 patients. The change in HBsAg level was − 0.20 log IU/mL (median value), similar to that reported by Uchida et al.24, although the patients’ backgrounds were slightly different. Moreover, they detected HBV-DNA in 39 (22.4%) patients during TAF switching; however, HBV-DNA was negative in 37 (94.9%) patients at 144 weeks after TAF switching. Additionally, the change in eGFR was − 2.9 at 144 weeks after switching to TAF, and high safety was reported.
In this study, mean changes in HBsAg levels were similar between the ETV continuation and TAF change groups. Comparing ETV and TDF in domestic phase 3 study17, the mean change in HBsAg levels was significantly reduced with TDF. Therefore, in this study, it was expected that the HBsAg levels would decrease in the TAF change group. However, in this study, mean changes in HBsAg levels were similar between the ETV continuation and TAF change groups. This is because the domestic phase 3 study was conducted in naive patients; however, in this study, patients on ETV were switched to TAF. Furthermore, in this study, the proportion of patients who were HBeAg-positive was as low as 9%, which was considered a factor.
In this study, we also investigated the transition of HBcrAg. It has been reported that HBcr antigen is a risk factor for HCC development if HBsAg is low but HBcrAg is high13. Furthermore, it has been suggested that even in cases where HBsAg has become negative, the persistence of HBcrAg is a risk factor for HCC development16. In other words, HBcrAg is a predictor of HCC development independent of HBsAg, and in some cases may be more suitable for predicting HCC development than HBsAg. In this study, the average change in HBcrAg levels was similar in the ETV continuation group and the TAF change group. In addition, there were two cases of HCC development in this study (ETV continuation group). In these two cases, HBsAg levels had decreased compared to baseline at the time of the development of HCC, but HBcrAg levels had not decreased sufficiently (Table 2).
Regarding safety, no significant difference was observed in the mean change in eGFR between the ETV continuation and TAF change groups. Additionally, NAG/Cr and β2MG/Cr, measured as renal tubular disorder markers, were comparable between both groups. A similar result was observed for serum phosphorus levels, indicating that ETV and TAF have similar effects on renal function, ensuring long-term safety. Regarding the effect on bone density, no significant decrease was observed in bone density in the ETV continuation and TAF change groups, even at 240 weeks of observation, when examining the lumber spine. However, following the femoral examination, the ETV continuation and TAF change groups showed decreased bone density (p = 0.081 and 0.047, respectively). Notably, impaired reabsorption of phosphorus due to renal tubular disorder is reportedly a factor in the decrease in bone mineral density caused by nucleic acid analogs. However, in this study, ETV and TAF did not exacerbate renal tubular disorders or decreased serum phosphorus values, suggesting that nucleic acid analogs are unlikely to contribute to the decrease in bone mineral density. Instead, long-term observations of aging may be a contributing factor. Lastly, serum ALT level is another important safety indicator in this study; no significant difference was observed in the mean change in ALT, confirming the safety of switching from ETV to TAF.
Finally, we compared the cumulative incidence of liver cancer between the ETV continuation and TAF change groups. To date, many studies have been conducted on the carcinogenic suppression effects of different nucleic acid analog preparations using ETV and TDF. The first large-scale study to compare the difference in hepatocarcinogenic suppression effects between ETV and TDF was a study by Choi et al. using a national medical database in South Korea. In propensity score matching (PSM) analysis, the TDF and ETV groups had HRs of 0.68 and 0.68, respectively. Similar results were obtained in the hospital cohort26. However, in South Korea, Kim et al. analyzed the cumulative liver cancer incidence in a multicenter hospital cohort comprising patients who started ETV or TDF as initial treatment from 2012, when TDF was approved, to 2014. Their analysis, using PSM and inverse probability weighting, revealed no significant difference between the ETV and TDF groups27. Since these reports, several cohort studies have compared the hepatocarcinogenic suppression effects of ETV and TDF; while some reports show the superiority of TDF, many others demonstrate that the two are equivalent28. However, all their studies support the equivalence of ETV and TDF or the superiority of TDF, and no evidence of the superiority of ETV exists. Although there have been many reports on the inhibitory effects of ETV and TDF on the development of HCC, there have been few comparative studies of ETV and TAF. Lee et al. reported that the inhibitory effects of ETV and TAF on the development of HCC were equivalent29. In this study, two patients in the ETV continuation group developed HCC, but the inhibitory effect on HCC development was not significantly different between the two groups. Both the patients with HCC had advanced fibrosis with low PLT values. Furthermore, during HCC onset, the HBsAg level was reduced compared to baseline; however, the HBcrAg level was not sufficiently reduced (Table 2). It has been reported that the amount of HBcrAg is more accurate than that of HBsAg in evaluating the risk of developing HCC during the administration of nucleic acid analogs14,30. Specifically, even if there is a reduction in HBsAg levels, patients with insufficient reduction in HBcrAg are at risk of having HCC. This is thought to be because HBcrAg reflects the amount of covalently closed circular DNA in the liver more sensitively than HBsAg.
This study has several limitations. It was conducted at a single institution, and the number of cases was small, making it a preliminary study. In particular, the study on the incidence of HCC had an insufficient sample size and low event rate, which may have impaired the statistical power of the Kaplan-Meier curve and log-rank test. In addition, the observation period was only 5 years, which is not sufficient.
In conclusion, the effects on serum HBsAg levels and renal function were similar over the 240-week comparative long-term observation between the ETV continuation and TAF change groups. Therefore, future research targeting a larger patient population is necessary.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Kawanaka, M. et al. Quantitative levels of hepatitis B virus DNA and surface antigen and the risk of hepatocellular carcinoma in patients with hepatitis B receiving long-term nucleos(t)ide analogue therapy. Liver Cancer. 3, 41–52 (2014).
Terrault, N. A. et al. AASLD guidelines for treatment of chronic hepatitis B. Hepatology 63, 261–283 (2016).
European Association for the Study of the Liver. Electronic address: easloffice@easloffice.eu & European Association for the study of the liver. EASL 2017 clinical practice guidelines on the management of hepatitis B virus infection. J. Hepatol. 67, 370–398 (2017).
Sarin, S. K. et al. Asian-pacific clinical practice guidelines on the management of hepatitis B: a 2015 update. Hepatol. Int. 10, 1–98 (2016).
Tanaka, E. & Matsumoto, A. Guidelines for avoiding risks resulting from discontinuation of nucleoside/nucleotide analogs in patients with chronic hepatitis B. Hepatol. Res. 44, 1–8 (2014).
Arase, Y. et al. Long-term outcome after hepatitis B surface antigen seroclearance in patients with chronic hepatitis B. Am. J. Med. 119, 71e9–7116 (2006).
Hu, Q. et al. The efficacy and safety of adding on or switching to peginterferon α-2b in HBeAg-positive chronic hepatitis B patients with long-term entecavir treatment: a multicentre randomised controlled trial. Aliment. Pharmacol. Ther. 56, 1394–1407 (2022).
Wirth, S. et al. Efficacy and safety of peginterferon alfa-2a (40KD) in children with chronic hepatitis B: the PEG-B-ACTIVE study. Hepatology 68, 1681–1694 (2018).
Marcellin, P. et al. Telbivudine plus pegylated interferon alfa-2a in a randomized study in chronic hepatitis B is associated with an unexpected high rate of peripheral neuropathy. J. Hepatol. 62, 41–47 (2015).
Ratnam, D. et al. Efficacy and tolerability of pegylated interferon-α-2A in chronic hepatitis B: a multicenter clinical experience. J. Gastroenterol. Hepatol. 27, 1447–1453 (2012).
Kim, G. A. et al. HBsAg seroclearance after nucleoside analogue therapy in patients with chronic hepatitis B: clinical outcomes and durability. Gut 63, 1325–1332 (2014).
Fattovich, G. et al. Delayed clearance of serum HBsAg in compensated cirrhosis B: relation to interferon alpha therapy and disease prognosis. European concerted action on viral hepatitis (EUROHEP). Am. J. Gastroenterol. 93, 896–900 (1998).
Hosaka, T. et al. Impact of hepatitis B core-related antigen on the incidence of hepatocellular carcinoma in patients treated with nucleos(t)ide analogues. Aliment. Pharmacol. Ther. 49, 457–471 (2019).
Tseng, T. C. et al. High level of Hepatitis B Core-related Antigen Associated with increased risk of Hepatocellular Carcinoma in patients with chronic HBV infection of intermediate viral load. Gastroenterology 157, 1518–1529 (2019).
Kaneko, S. et al. Hepatitis B core-related antigen predicts disease progression and hepatocellular carcinoma in hepatitis B e antigen-negative chronic hepatitis B patients. J. Gastroenterol. Hepatol. 36, 2943–2951 (2021).
Inoue, T. et al. Hepatitis B core-related antigen: a novel and promising surrogate biomarker to guide anti-hepatitis B virus therapy. Clin. Mol. Hepatol. 29, 851–868 (2023).
Koike, K., Suyama, K., Ito, H., Itoh, H. & Sugiura, W. Randomized prospective study showing the non-inferiority of tenofovir to entecavir in treatment-naïve chronic hepatitis B patients. Hepatol. Res. 48, 59–68 (2018).
Buti, M. et al. Tenofovir alafenamide versus tenofovir disoproxil fumarate for the treatment of patients with HBeAg-negative chronic hepatitis B virus infection: a randomised, double-blind, phase 3, non-inferiority trial. Lancet Gastroenterol. Hepatol. 1, 196–206 (2016).
Rashidi-Alavijeh, J. et al. Safety and efficacy of tenofovir alafenamide in liver transplant recipients: a single center experience. Transpl. Infect. Dis. 23, e13522 (2021).
Kaneko, S. et al. Tenofovir alafenamide for hepatitis B virus infection including switching therapy from tenofovir disoproxil fumarate. J. Gastroenterol. Hepatol. 34, 2004–2010 (2019).
Hagiwara, S. et al. Comparison of efficacy and safety of entecavir and switching from entecavir to tenofovir alafenamide fumarate in chronic hepatitis B: long-term effects from a prospective study. Hepatol. Res. 51, 767–774 (2021).
Hochberger, S. et al. Fully automated quantitation of hepatitis B virus (HBV) DNA in human plasma by the COBAS AmpliPrep/COBAS TaqMan system. J. Clin. Virol. 35, 373–380 (2006).
Usuda, S. et al. Serological detection of hepatitis B virus genotypes by ELISA with monoclonal antibodies to type-specific epitopes in the preS2-region product. J. Virol. Methods. 80, 97–112 (1999).
Uchida, Y. et al. Superiority of tenofovir alafenamide fumarate over entecavir for serum HBsAg level reduction in patients with chronic HBV infection: a 144-week outcome study after switching of the nucleos(t)ide analog. PLOS ONE. 17, e0262764 (2022).
Ogawa, E. et al. Switching to tenofovir alafenamide for nucleos(t)ide analogue-experienced patients with chronic hepatitis B: week 144 results from a real-world, multi-centre cohort study. Aliment. Pharmacol. Ther. 56, 713–722 (2022).
Choi, J. et al. Risk of hepatocellular carcinoma in patients treated with entecavir vs tenofovir for chronic hepatitis B: a Korean nationwide cohort study. JAMA Oncol. 5, 30–36 (2019).
Kim, S. U. et al. A multicenter study of entecavir vs. tenofovir on prognosis of treatment-naïve chronic hepatitis B in South Korea. J. Hepatol. 71, 456–464 (2019).
Choi, W. M., Yip, T. C., Lim, Y. S., Wong, G. L. & Kim, W. R. Methodological challenges of performing meta-analyses to compare the risk of hepatocellular carcinoma between chronic hepatitis B treatments. J. Hepatol. 76, 186–194 (2022).
Lee, H. W. et al. Effect of tenofovir alafenamide vs. tenofovir disoproxil fumarate on hepatocellular carcinoma risk in chronic hepatitis B. J. Viral Hepat. 28, 1570–1578 (2021).
Hosaka, T. et al. Ultrasensitive assay for hepatitis B core-related antigen predicts hepatocellular carcinoma incidences during entecavir. Hepatol. Commun. 6, 36–49 (2022).
Author information
Authors and Affiliations
Contributions
T.M., S.H., N.N., N.O., A.Y., T.Y., Y.K., M.T., M.K.: Study concept and design; T.M., S.H., N.N., N.O., A.Y., T.Y., Y.K., M.T., M.K.: Patient recruitment and characterization; T.M., S.H., N.N.: Data acquisition; T.M., S.H.: Data analysis; T.M., S.H., N.N., N.O., A.Y., T.Y., Y.K., M.T., M.: Article drafting; all authors provided input and critical revision and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Matsubara, T., Hagiwara, S., Nishida, N. et al. Observational pilot study of switching from entecavir to tenofovir alafenamide in patients with chronic hepatitis B. Sci Rep 15, 869 (2025). https://doi.org/10.1038/s41598-025-85317-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-85317-3