Abstract
Saffron (Crocus sativus L.) is the most expensive spice in the World, and Italy is among the major European producers. The study aims to improve value and recognition of high quality saffron by proposing a subdivision within the first quality category, according to ISO 3632 standards. The analysis of 125 saffron samples, collected from different Italian regions in 2021–2022 harvesting seasons, revealed that 95% of the samples met the first quality criteria, following ISO guidelines. Consequently, for the first quality samples, a differentiation into “premium”, “superior” and “high-quality” subcategories was proposed. Along with traditional methods, FT-NIR spectroscopy combined with chemometrics was employed for a comprehensive quality assessment. Discriminant and class-modelling approaches were developed to predict saffron quality subcategory. Best results were obtained by linear discriminant analysis models with accuracy in calibration (92.7%), cross-validation (86.4%) and prediction (87.0%). However, SIMCA modelling resulted more appropriate for class-modelling, confirming that none of the “premium” samples were misclassified as “high-quality” and vice-versa. The results support the inclusion of subcategories within ISO 3632 standards, thus refining the classification of saffron quality. Furthermore, the study emphasises the effectiveness of FT-NIR spectroscopy as a valuable tool for saffron quality assessment, with potential implications for industry standards and practices.
Similar content being viewed by others
Introduction
Globally, the spice market is certainly growing, with a value of 7.4 billion USD in 2021 and a growth expectation of about 8.3 billion USD in 20261. Among them, saffron, which is obtained from the dried stigma of Crocus sativus L., has the highest economic value2 and it plays an important role in the culinary culture of various regions of the World due to its glycosidic constituents that impart colouring and flavouring properties3. Indeed, saffron quality is determined by its colour, aroma, and flavour, which are related to the levels of crocin and crocetins, safranal volatiles, and picrocrocin4.
Until today, saffron quality characteristics are only evaluated on a voluntary basis, according to ISO 3632 1,2:2010–20115,6. This standard requires the use of target methods for the quantification of a specific marker, indicative of a particular property, to obtain saffron classification against the established limits. The ISO 36325,6 defines three quality categories (I, II and III) according to the content of crocin, safranal, picrocrocin and humidity (Table 1).
The ISO standard establishes that for moisture and aroma strength the values assigned are the same for all three quality categories, whereas the values for colouring and flavour strength are the only parameters that discriminate among the three quality categories (Table 1)5,6.
In particular, Giupponi et al.7 demonstrated that saffron produced in Italy is mostly classified in the first ISO class5,6, with 84–93% falling into this category. This trend was confirmed by Leoni8 who studied how some good practices adopted by Italian saffron farmers, as flowers being harvested before light exposure and dark conservation, could affect the quality of the spice. In particular, they showed that the quality of saffron seems to be strongly linked to the changes in its pigments, which are responsible for its colouring strength.
The trend leads to the willingness for stricter quality standards able to valorise products which highly exceed the limits to be considered in I category according to the ISO standard, in particular saffron reaching flavour strength above 90 \(A_{{1cm}}^{{1\% }} \left( {257~nm} \right)\) and colouring strength above 240 \(A_{{1cm}}^{{1\% }} \left( {440~nm} \right)\).
Among the stakeholders need there is also the possibility of testing saffron quality by alternative approaches to ISO standards. From this side several methods have been applied for saffron quality assessment, including traditional identification (e.g., crocin, picrocrocin and safranal determination, moisture content) using UV-vis spectrophotometer9, thermostatic heater as expected by ISO 3632, and HPLC10. Traditional identification methods are sensitive but have some disadvantages which can be overcome by non-target techniques, such as NIR, that are inexpensive, timesaving, non-destructive, reagents-saving, and not requiring skilled operators. Additionally, they provide a fingerprint of the analysed food products in a holistic way11.
In recent years, the interest and the motivation to establish rapid and non-destructive methods for the evaluation of saffron have increased12 and NIR approaches have been developed to face adulteration and origin issues13,14,15,16.
However, those methods could be also useful to address the stakeholder need for the valorisation of premium products, and, thus, stimulate farmers to continuously improve the produce quality.
In this context, the present study aimed to address two main goals. Firstly, it aims at defining a subdivision within the first quality category based on ISO standard, thus responding to the need of producers to recognize premium products. Secondly, a method based on FT-NIR spectroscopy, combined with chemometric methods, is proposed for the assessment of the new quality sub-classes discrimination.
Materials and methods
Saffron collection and quality analysis
One hundred twenty-five Italian saffron samples were collected from different Italian regions: 5 Calabria, 3 Campania, 23 Emilia-Romagna, 4 Friuli Venezia Giulia, 8 Lazio, 39 Lombardy, 3 Marche, 7 Piedmont, 3 Apulia, 3 Sardinia, 6 Tuscany, 6 Trentino Alto Adige, 15 Veneto. Each sample was analysed in duplicate. Saffron harvested in 2021 and 2022 was stored in specially sealed vials to minimize degradation, in the dark and at room temperature.
Analysis of saffron followed the procedures outlined in ISO 3632 (ISO 3632 1,2:2010–2011)5,6. Initially, 500 mg of saffron sample was weighed, dried for 16 h in an oven at 105 °C and reweighed to determine moisture and volatile matter content. Subsequently, 125 mg of powdered saffron was weighed and transferred into a 250 mL volumetric flask. The flask was filled to volume with Millipore water and stirred at room temperature for 1 h using a magnetic stirrer bar (1000 r/min). After stirring, an aliquot of the solution was filtered into a volumetric cylinder, discarding the initial 40 ml and retaining the subsequent 20 ml. Finally, 10 ml of solution was transferred to a 100 ml volumetric flask, filled to volume and stirred for a few minutes.
Moisture and volatile matter content, W%, expressed as a percentage of the initial sample, was calculated according to formula (1):
where m0 represents the initial mass (g) of the test sample, and mf represents the mass (g) of the dry residue.
The resulting solution was analysed using a UV-vis spectrophotometer (Varian Cary 50). Absorbance measurements were taken at 257 nm, 330 nm and 440 nm, corresponding to picrocrocin, safranal and crocin molecules, respectively by zeroing with Millipore water and using quartz cuvettes.
The value of A1cm1% was calculated with the following formulae (2, 3, 4):
The results obtained from the UV-Vis spectrophotometer allowed the samples to be classified according to ISO 3632 standard5,6, as can be shown in the table (Table 1).
FT-NIR analysis
The MPA FT-NIR spectrophotometer (Bruker Optics, Milano, Italy), managed by the software Opus™ (v. 6.5, Bruker Optics, Milano, Italy), was used for saffron powder analysis. Measurements were performed in reflection, using an integrating sphere, and spectra collection was in the range of 12,500–3800 cm− 1, with a resolution of 8 cm− 1, and with each measurement consisting of 32 scans for both samples and background13. To carry out the analyses, special vials with 0.5 ± 0.01 g of saffron powder were used; two vials were filled for each sample and each of them was analysed in duplicate.
Statistical data analysis
Data exploration was performed in the MATLAB environment (v. 2017b, Mathworks, Inc., Natick, MA, USA) using the PLS toolbox (v. 8.5, Eigenvector Research, Inc., Aeattle, WA, USA). Classification models and variable selection were performed by the V-Parvus package17.
-
Principal component analysis.
Principal component analysis (PCA) was applied to explore sample distribution based on the data collected according to ISO analytical procedures and FT-NIR spectra. As an unsupervised exploratory procedure, PCA allows visualization, in a reduced space, of relationships between items (via the score plot), variables (via the load plot) and their relationship (via the bi-plot)18.
PCA was applied to ISO results to identify sample grouping, which were further classified into (i.e., “premium”, “superior”, “high-quality”) to be used as a priori information in classification model development.
For FT-NIR data, PCA was applied after spectral range reduction and pre-treatments. Specifically, the NIR spectral range was reduced to the region between 9000 and 3800 cm− 1, and different spectral pretreatments, such as Standard Normal Variate (SNV) and first derivate (Savitzky–Golay filter) were applied alone or in combination. Samples were colour-coded based on the quality subcategories identified after the PCA on the ISO results.
PCA was also used to identify outliers by calculating the Euclidian distance of each sample from the centre of the N-dimensional principal component space.
-
Classification model development.
The development of classification models for saffron quality prediction involved the associating the FT-NIR spectra with an a priori information. Subcategories, previously identified by the characterization of saffron using ISO methods, were used to define the classes (“premium”, “superior”, “high-quality”).
The classification models were developed by Linear Discriminant Analysis (LDA) and Soft Independent Modelling of Class Analogy (SIMCA).
As LDA, a supervised pattern recognition method, utilizes discriminant canonicals to calculate the centre of matrix covariance, requires the number of samples exceeding the number of variables a feature selection approach (fifteen wavenumbers) was implemented using the SELECT algorithm17,19.
SIMCA, a supervised classification method developed by Wold and Sjöström20, followed the same feature selection approach as LDA, then an independent PCA on the spectral variables of calibration sets for the considered subcategories (“premium”, “superior”, “high-quality”) is performed. In the PCA-reduced space, SIMCA constructs a multidimensional space for classifying external test set samples based on the distance between each sample and the models. SIMCA creates models equal to the number of classes, leading to sample assignments as follows: (1) exclusive assignment to one class; (2) no assignment to any class; (3) fitting two or more classes.
Supervised classification models were validated through both internal cross-validation, by 5 cancellation groups, and prediction, by an independent random external test set containing around 30% of the total data.
Model performance was evaluated in terms of sensitivity (TP/(TP + FN)), specificity (TN/(TN + FP)), and correct classification percentage, a.k.a. the proportion of samples correctly accepted in the external test set belonging to the modelled class.
Results and discussion
Qualitative characterization
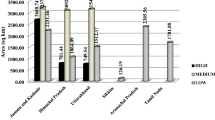
Out of the one hundred and twenty-five samples analysed, 95% (n = 117) belong to the first category of quality, whereas five samples (4%) belong to the second category and none to the third category according to the ISO standard5,6. Two saffron samples were not classified (nc) due to their high humidity content (> 12%) (Fig. 1); these results are consistent with previous literature findings7.
Result of ISO 3632 for all 125 saffron samples.
The standard deviation values of the whole dataset are higher for colour strength (246.25 ± 25.03 \(A_{{1cm}}^{{1\% }} \left( {440~nm} \right)\)) and flavour strength (99.33 ± 9.09 \(A_{{1cm}}^{{1\% }} \left( {257~nm} \right)\) than for the other parameters, i.e. aroma strength (28.87 ± 3.89 \(A_{{1cm}}^{{1\% }} \left( {330~nm} \right)\) and moisture (6.92 ± 1.81%); these findings aligned with previous works7,8. Therefore, the division of first category into three subcategories was primarily focused on colour and flavour strength, as illustrated in Fig. 2. The Box and Whisker representation of the flavour strength (Fig. 2A) allowed to identify that most of the samples (interquartile range, IQR) are characterised by 95–107 \(A_{{1cm}}^{{1\% }} \left( {257~nm} \right)\), this range was preliminary identified as “superior” subcategory.
The range included in the upper whisker, i.e. 107–117 \(A_{{1cm}}^{{1\% }} \left( {257~nm} \right)\), was preliminary identified as “premium” subcategory; whereas range included in the lower whisker, i.e. 95–78 \(A_{{1cm}}^{{1\% }} \left( {257~nm} \right)\), was preliminary identified as “high-quality” subcategory. Similarly for the Box and Whisker representation of the colour strength (Fig. 2B) it was identified a “superior” subcategory included in the IQR (235–260 \(A_{{1cm}}^{{1\% }} \left( {440~nm} \right)\), a “premium” subcategory in the upper whisker, i.e. 260–300 \(A_{{1cm}}^{{1\% }} \left( {440~nm} \right)\), and a “high-quality” subcategory included in the lower whisker, i.e. 235–201 \(A_{{1cm}}^{{1\% }} \left( {440~nm} \right)\). Samples belonging to the same subcategory for both properties were assigned to that subcategory.
Boxplots relating to flavour strength (A) and colour strength (B) showing the method of quartiles, used to divide the samples into four subcategories.
However, some samples could not be unambiguous assigned to a subcategory, so a multivariate exploration of ISO results, by PCA, was used to solve the lack of assignation.
On the resulting PC1 (43.38% of explained variance) vs. PC2 (29.52% of explained variance) bi-plot (Fig. 3A), samples with unambiguous subcategory assignation, i.e. sample resulting in the same subcategory for both flavour and colour strength, were coloured according to the specific subcategory, whereas samples with ambiguous quality category were marked with a star (*).
Focusing on the distribution along the principal component axis (PC1) for the ISO data, a general trend can be observed: “premium” samples (green squares), align with positive values, “high-quality” samples (red circles) with negative values, and “superior” samples (yellow diamonds) occupy central positions along PC1.
The distribution is explained by the variable position in the bi-plot: aroma strength (a.k.a. aromatic power) as almost no influence on PC1 distribution, having values proximate to 0; moisture confirms to have a negative effect on quality distribution, having negative PC1 values; both flavour strength (a.k.a bittering p.) and colouring strength (a.k.a. colouring p.) contributed to positive positioning of “premium” samples along PC1.
Consequently, to assign the ambiguous samples (marked with a star, *) to a subcategory, the biplot plane of PC1 vs. PC2 (Fig. 3A) was divided into three sagittal portions. Samples within each section were then appropriately assigned (Fig. 3B).
PCA biplot of ISO data for saffron samples divided into 3 quality subcategories.
This approach effectively grouped “premium”, “superior” and “high-quality” saffron samples, highlighting the importance of a multivariate approach in data exploration when more than two variables are considered.
FT-NIR
-
Spectra inspection.
The FT-NIR spectra obtained from the analysis of saffron samples belonging to the first ISO category (n = 117) presented similar trends (Fig. 4). Consequently, directly distinguishing sample quality or quality subcategories proved challenging.
NIR spectra (9000 and 3800 cm-1) of three samples representing the 3 qualitative subcategories: (A) raw NIR spectra, (B) spectra after SNV and first derivative transformation.
In any case, before conducting chemometric analysis on FT-NIR spectra, the spectral features of saffron were examined to gain insight into the functional groups responsible for the observed characteristics. The absorption bands within the FT-NIR spectra (Fig. 4A) can be described as follows13,16: (i) at approximately 8300 cm− 1 there is an absorption associated with the second overtone of C–H stretching; (ii) an absorption band related to the first overtone of O–H stretching or N–H stretching is visible in the range of 7100–6000 cm− 1; (iii) the first overtone of C–H stretching is discernible within the range of 6000–5400 cm− 1; (iv) around 5170 cm− 1 a combination of O–H stretching and the first overtone of C–O deformation is observed; (v) at 4750 cm− 1 a band corresponds to the combination of C–O stretching and O–H deformation; and (iv) at 4320 cm− 1 there is an absorption associated with the combination of C–C and C–H stretching.
This spectral region (9000 and 3800 cm− 1) was utilized for subsequent analysis.
In conclusion, the analysis of FT-NIR spectra provided valuable insights into the chemical composition of saffron samples, guiding subsequent chemometric analysis for quality assessment.
-
PCA analysis.
By examining the components PC1 (76.34% of explained variance) and PC2 (11.14% of explained variance) of the FT-NIR spectra, no discernible distribution of samples based on qualitative subcategories was revealed (data not shown). However, through an exploration of PC3 (4.81% of total variance) and PC4 (2.26% of total variance), a distribution of samples according to the previously identified subcategories was evident (Fig. 5A–C).
Score plot PC1 vs. PC4 (A), PC2 vs. PC4 (B) and PC3 vs. PC4 (C) coloured according to the 3 quality subcategories and loading plot (D). The plots are obtained from PCA applied on the NIR spectra of saffron samples, pretreated by SNV and first derivate.
This became particularly clear plotting PC4 against PC3, as shown in the scores plot in Fig. 5C, samples positioned in the IV quadrant were mainly of “premium” quality, whereas intermediate scores were typical for “superior” samples; “high-quality” samples were positioned in the II quadrant. The region most influencing the shift of samples along PC4 (Fig. 5D, PC4-black line) lay between 4700 and 4000 cm− 1, which was also identified as the most insightful region in a study by Castro et al.21. Specifically, this region is attributed to the combination band category, characterized by vibrations involving C–H paired with C–C, C–H paired with C–H, and N–H paired with C–H bonds. Chemical compounds frequently associated to these bond vibrations include carbohydrates and proteins18.
-
Quality subcategories identification.
To discriminate the samples according to the three different quality subcategories, the FT-NIR spectra (selected range 9000 and 3800 cm− 1) were used to create LDA and SIMCA models. Different spectra pre-treatments were evaluated. Approximately 70% of the samples belonging to the first quality class according to the ISO (79 samples, 13 in premium, 46 in superior, 20 in high quality classes) were allocated for model validation and cross-validation, whereas the remaining 30% (38 samples, 7 in premium, 23 in superior, 8 in high quality classes) were used for predictive performance evaluation. The fifteen variables were selected from the calibration dataset using the SELECT algorithm (Fig. 6).
NIR spectra of three saffron samples, representing the identified quality subcategories, pre-treated with SNV derived first. The vertical lines identify the 15 variables, with greater weight in the classification, used in the models.
The best performance was observed for the models after pretreatment with SNV in combination with 1th derivative. Tables 2 and 3 present the results obtained by LDA and SIMCA models, respectively. A highly accurate weighted correct classification rate was achieved with the LDA model (Table 2): 92.75% in calibration, 86.44% in cross-validation, and 87.02% in prediction.
Weighted specificity and sensitivity in calibration were both higher than 90% (specificity of 94.60% and sensitivity of 91.14%) with prediction values ranging between 82.36 and 95.49%, for sensitivity and specificity, respectively. Notably, the discriminant model demonstrated a significant quality, in that no “premium” samples were misclassified as “high-quality” and vice versa. This finding aligns with the researched conducted by Zalacain et al.14, which underscored the capabilities of NIR spectroscopy for determining the chemical composition of saffron according to ISO 3632 parameters. Indeed, the models developed by Zalacain et al.14 exhibited an error of 20.40 \(A_{{1cm}}^{{1\% }}\) for the colour strength and 6.48 \(A_{{1cm}}^{{1\% }}\) for flavour strength, comparable to the distance between the “premium” and the “high-quality” subcategories, characterised by errors of 26.10 \(A_{{1cm}}^{{1\% }}\) and 12. 29 \(A_{{1cm}}^{{1\% }}\), respectively. This also explain why some samples from “premium” and “high-quality” subcategories are confused with the “superior” ones.
For SIMCA model (Table 3), the correct classification rate was 88.59% in calibration. However, this value exhibited a decline during cross-validation and prediction, reaching 69.62% and 61.55%, respectively.
The developed model achieved a weighted specificity of 90.80% in calibration and 82.76% in prediction, while sensitivity in calibration reached 88.61% and 76.69% in prediction. Thus, the model appears to be overfitted on the calibration data, suggesting the need for better sampling considering highest variability to develop a class-modelling approach. In SIMCA model, being based on a class-modelling approach, there may be samples that are not assigned to any class or assigned to multiple classes, thus generating an ambiguous assignment. The graphical representation by Coomans’ plot highlights these cases. In Fig. 7 the Cooman’s plot of the prediction phase is represented, considering two classes at a time.
Cooman’s plots resulting from the prediction phase of SIMCA modelling: Class 1 vs. Class 2 (A), Class 1 vs. Class 3 (B), Class 2 vs. Class 3 (C).
In Fig. 7A the axes represent the distances of samples from the models of class 1 (premium) and class 2 (superior), respectively. The two dashed lines correspond to the critical acceptance thresholds for each model at the specified confidence level (in this case 95%). Samples from both classes are plotted as scatter points, with their coordinates reflecting their relative similarity to the two models, and the plot is divided into four sectors. In sector 1 (S1), samples are accepted solely by class 1 (premium), only samples belonging to “premium” class are in this section. In sector 2, samples are accepted only by class 2 (superior), here are also located two samples belonging to “high-quality” class. In sector 3 (S3), samples that are accepted by both models are located. This overlap occurs because the models for each class are built independently, and their class spaces may intersect; here one sample belonging to “Premium” class is present, together with 5 samples belonging to “Superior” class. Finally, in sector 4 (S4) are located six “high-quality” samples that are correctly rejected by both models. Furthermore, some “superior” samples are in this sector, these samples indicate that the variables used do not fully define the class boundaries, preventing forced (and potentially incorrect) classifications that might instead arise in discriminant analysis approaches. Similar consideration could be retrieved from Fig. 7B,C, where it is possible to notice that none of samples of “Premium” class is confused, not even with multiple assignments, with “high – quality” class.
Overall, LDA and SIMCA modelling of the FT-NIR spectra proved to be effective in discriminating between different subcategories of saffron quality. LDA demonstrates high accuracy and specificity, however the assignation to any quality subcategory is always forced. On the other hand the soft approach proposed by SIMCA modelling allowed for perfect discrimination between “premium” and “high-quality” saffron, even if some samples resulted unassigned or multiple assigned.
Conclusion
This work has confirmed the high quality of Italian saffron, with 95% (n = 117) of the samples belonging to the first ISO 3632 quality category. The outcome highlights the success of past efforts in promoting good agricultural practices and remarks the need of recognition of restrictive quality standard.
Thus, to encourage the quality differentiation and the continuous improvement among farmers, this study proposed a subdivision of the first ISO 3632 quality category into “premium”, “superior” and “high – quality” based on flavour and colour strength. This subdivision promises to improve the quality recognition of saffron not only in Italy but also on a global scale.
Additionally, this study has successfully developed a FT-NIR spectroscopy approach to address the need of the control bodies for a cost-effective and environmentally friendly method to assess saffron quality. The LDA classification method achieved high predictive accuracy (87.0%) in discriminating between subcategories. The SIMCA model allowed the discrimination between “premium” and “high-quality” saffron. In light of these findings, it would be reasonable to consider the integration in the future analytical standards of the subdivision of the first quality category into subcategories (premium, superior and high-quality), as well as the implementation of qualitative analysis of saffron by NIR spectroscopy.
Data availability
The datasets generated during and analysed during the current study are available from the corresponding author on reasonable request.
References
https://www.businesscoot.com/en/study/the-spice-market-italy (2023).
Kiani, S., Minaei, S. & Ghasemi-Varnamkhasti, M. Instrumental approaches and innovative systems for saffron quality assessment. J. Food Eng. 216, 1–10. https://doi.org/10.1016/j.jfoodeng.2017.06.022 (2018).
Bagur, M. et al. Saffron: an old medicinal plant and a potential novel functional food. Molecules 23 (1), 30. https://doi.org/10.3390/molecules23010030 (2017).
Heidarbeigi, K. et al. Detection of adulteration in saffron samples using electronic nose. Int. J. Food Prop. 18 (7), 1391–1401. https://doi.org/10.1080/10942912.2014.915850 (2015).
ISO 3632-1:2011. Spices:Saffron (Crocus sativus L.)—Part 1: Specification.
ISO 3632-2.:2010. Spices: Saffron (Crocus sativus L.)–Part 2: Test methods.
Giupponi, L. et al. Quality traits of saffron produced in Italy: Geographical area effect and good practices. J. Appl. Bot. Food Qual. 92, 336–342. https://doi.org/10.5073/jabfq.2019.092.045 (2019).
Leoni, V. et al. How harvest, cleaning and conservation good practices affect the quality of saffron: results of a research conducted in Italy. J. Appl. Bot. Food Qual. 113, 105–113. https://doi.org/10.5073/JABFQ.2022.095.014 (2022).
Maggi, L. et al. Rapid determination of safranal in the quality control of saffron spice (Crocus sativus L). Food Chem. 127 (1), 369–373. https://doi.org/10.1016/j.foodchem.2011.01.028 (2011).
Valle García-Rodríguez, M. et al. Determination of saffron quality by high-performance liquid chromatography. J. Agric. Food Chem. 62 (32), 8068–8074. https://doi.org/10.1021/jf5019356 (2014).
Grassi, S. & Alamprese, C. Spectroscopic non-targeted techniques in combination with linear discriminant analysis for wine vinegar authentication. Food Bioprocess. Technol. 17 (2), 479–488 https://doi.org/10.1007/s11947-023-03143-9 (2023).
Kiani, S., Minaei, S. & Ghasemi-Varnamkhasti, M. Instrumental approaches and innovative systems for saffron quality assessment. J. Food Eng. 216, 1–10. https://doi.org/10.1016/j.jfoodeng.2017.06.022 (2018).
Li, S. et al. Rapid determination of crocins in saffron by near-infrared spectroscopy combined with chemometric techniques. Spectrochim Acta A: Mol. Biomol. Spectrosc. 190, 283–289. https://doi.org/10.1016/j.saa.2017.09.030 (2018).
Zalacain, A. et al. Near-infrared spectroscopy in saffron quality control: Determination of chemical composition and geographical origin. J. Agric. Food Chem. 53 (24), 9337–9341. https://doi.org/10.1021/jf050846s (2005).
Shawky, E., El-Khair, R. M. A. & Selim, D. A. NIR spectroscopy-multivariate analysis for rapid authentication, detection and quantification of common plant adulterants in saffron (Crocus sativus L.) stigmas. LWT 122, 109032. https://doi.org/10.1016/j.lwt.2020.109032 (2020).
Amirvaresi, A., Nikounezhad, N., Amirahmadi, M., Daraei, B. & Parastar, H. Comparison of near-infrared (NIR) and mid-infrared (MIR) spectroscopy based on chemometrics for saffron authentication and adulteration detection. Food Chem. 344, 128647. https://doi.org/10.1016/j.foodchem.2020.128647 (2021).
Forina, M. et al. PARVUS: An extendable package of programs for data exploration, classification and correlation. J. Chemometr. 4 (2), 191–193 (1988). https://hdl.handle.net/11581/110109
Wold, S., Esbensen, K. & Geladi, P. Principal component analysis. Chemometri. Intell. Lab. Sys. 2 (1–3), 37–52. https://doi.org/10.1016/0169-7439(87)80084-9 (1987).
Kowalski, B. R. & Bender, C. F. An orthogonal feature selection method. Patt. Recognit. 8 (1), 1–4. https://doi.org/10.1016/0031-3203(76)90023-6 (1976).
Wold, S. & Sjöström, M. SIMCA: a method for analyzing chemical data in terms of similarity and analogy. (1977). https://doi.org/10.1021/bk-1977-0052.ch012
Castro, R. C., Ribeiro, D. S., Santos, J. L. & Pascoa, R. N. Near infrared spectroscopy coupled to MCR-ALS for the identification and quantification of saffron adulterants: Application to complex mixtures. Food Control. 123, 107776. https://doi.org/10.1016/j.foodcont.2020.107776 (2021).
Acknowledgements
This study was carried out within the Agritech National Research Center and received funding from the European Union Next-GenerationEU (PIANO NAZIONALE DI RIPRESA E RESILIENZA (PNRR) – MISSIONE 4 COMPONENTE 2, INVESTIMENTO 1.4 – D.D. 1032 17/06/2022, CN00000022). This manuscript reflects only the authors’ views and opinions, neither the European Union nor the European Commission can be considered responsible for them.
Author information
Authors and Affiliations
Contributions
I. L.: Formal analysis; Data curation; Visualization; Writing - original draft.D. P.: Formal analysis; Visualization; Writing - original draft.S. G.: Conceptualization; Investigation; Methodology; Data curation; Visualization; Writing - review & editing. S. B.: Conceptualization; Writing - original draft.A. G.: Conceptualization; Funding acquisition; Project administration; Resources; Supervision; Writing - review & editing.L. G.: Conceptualization; Investigation; Methodology; Writing - original draft; Writing - review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Locatelli, I., Pedrali, D., Grassi, S. et al. Progress in quality assessment of Italian saffron. Sci Rep 15, 2175 (2025). https://doi.org/10.1038/s41598-025-86440-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-86440-x