Abstract
We intended to investigate the potential of several transitional zone (TZ) volume-related variables for the detection of clinically significant prostate cancer (csPCa) among lesions scored as Prostate Imaging Reporting and Data System (PI-RADS) category 3. Between September 2018 and August 2023, patients who underwent mpMRI examination and scored as PI-RADS 3 were queried from our institution. The diagnostic performances of prostate-specific antigen density (PSAD), TZ-adjusted PSAD (TZPSAD), and TZ-ratio (TZ volume/whole gland prostate volume) were analyzed. We calculated the sensitivity and specificity for each variable, the overall accuracy was evaluated with the area under the receiver operating characteristic curve (AUC). The best cutoff value was determined by the Youden index, and differences between diagnostic performances were compared with the Delong test. A total of 154 TZ lesions were included, of them 28 were diagnosed with csPCa. The AUC for PSAD, TZPSAD, and TZ-ratio were 0.644 (95% CI 0.538–0.751), 0.714 (95% CI 0.607–0.821), and 0.746 (95% CI 0.636–0.856), with corresponding optimal cutoff values of 0.11 ng/ml/ml, 0.21 ng/ml/ml, and 0.66 ng/ml/ml, respectively. PSAD was significantly inferior to either TZPSAD (P = 0.03) or TZ-ratio (P = 0.03). The combination model including TZPSAD, TZ-ratio, and age generated an AUC of 0.838 (95% CI 0.759–0.917), significantly higher than use of these variables alone, with P values of 0.001 and 0.035 for the TZPSAD and TZ-ratio, respectively. TZPSAD and TZ-ratio were found to be independent predictors for differentiating csPCa among TZ lesions categorized as PI-RADS score 3. Furthermore, by combining these two variables with others, the diagnostic performance can be improved significantly.
Similar content being viewed by others
Introduction
Prostate Cancer (PCa) is the second most common malignancy and a leading cause of cancer death among men worldwide1. It is estimated that 1.2 million new cases are diagnosed annually and 350,000 men die from it2. In the past two decades, MRI has become the most effective imaging method for detecting, diagnosing, and managing PCa3. The mpMRI-based PI-RADS has shown high diagnostic performance and is widely used for PCa risk stratification4,5,6. However, PI-RADS category 3 lesions remain challenging for doctors. While PI-RADS suggests biopsies for lesions categorized as ≥ 3, only 36% are PCa and 18.5% are csPCa, leading to many unnecessary biopsies7. These biopsies and overtreatment can cause complications such as infection or sepsis. Therefore, it is essential to explore other clinical variables or biomarkers to enhance the diagnostic accuracy for these ambiguous lesions thereby reducing unnecessary biopsies. In recent years, PSAD has been intensely studied as an independent factor or combined with other clinical variables for predicting PCa8,9,10. However, one meta-analysis revealed that when using PSAD as the independent variable, the pooled sensitivity and specificity for PI-RADS score 3 lesions were only 0.61 and 0.69 at the threshold of 0.15 ng/ml/ml11.
PSA levels can also elevate due to benign prostatic hyperplasia (BPH) and prostatitis12. In 1994, Kalish et al. proposed the use of TZPSAD (defined as dividing serum PSA by the measured TZ volume) for the prediction of PCa, as BPH is the result almost exclusively of hyperplasia of the TZ, with a usually minor contribution from hyperplasia of peripheral zone and central zone13. Subsequent studies reported that this variable is a stronger predictor for PCa than whole gland PSAD; however, in those earlier studies, the calculated volume of the prostate is based on transrectal ultrasound imaging, which has inferior soft-tissue contrast to MRI14,15,16. Several studies have shown that TZ volume (TZV), peripheral zone volume (PZV), TZ-ratio (defined as the ratio of the volume of the transition zone relative to the total prostate gland volume), and PZ-ratio (defined as the ratio of the volume of the peripheral zone relative to the total prostate gland volume) may be independent predictor for detecting csPCa in TZ or PZ14,17,18. In this study, we aimed to investigate the TZPSAD and TZ-ratio for differentiating clinically significant PCa (csPCa) among lesions scores as PI-RADS 3.
Materials and methods
Patient selection
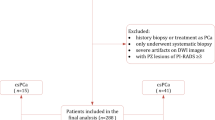
This single-center study was approved by the ethics committee of our institution (Yancheng Third People’s Hospital), and the requirement of written informed consent was waived by the institutional review board (IRB) of Yancheng Third People’s Hospital due to the retrospective nature. This study was conducted following the guiding principles of the Declaration of Helsinki, and all data were collected in compliance with the Health Insurance Portability and Accountability Act (HIPAA). Between September 2018 and August 2023, patients who underwent mpMRI examination and subsequently MRI/US guided fusion biopsy were queried from our institution’s electric medical records. We identified those lesions located in TZ and scored as category 3 based on PI-RADS, which were reassessed by two dedicated radiologists (with 5 years and 6 years of experience in urology radiology) independently, who were blinded to pathological results. Each lesion was scored strictly according to the PI-RADS v2.1 guideline again, with discrepancies resolved by discussion between two readers. Initially, 208 individuals were identified according to our inclusion criteria. Of these, 55 patients were excluded because of the following reasons: (1) diagnosed as PCa or treatment before MRI examination (n = 16); (2) only underwent systematic biopsy (n = 21); and (3) severe artifacts on MRI images (n = 18). Finally, a total of 154 patients (mean age 69.9 ± 8.0 years; median PSA 10.79, interquartile range [IQR] 7.92–16.25 ng/ml) who evaluated with PI-RADS score 3 were included in the current study. Figure 1 shows the details of the patient selection diagram.
Flowchart of the study population with the exclusion criteria. csPCa, clinically significant prostate cancer.
MRI acquisition and interpretation
All prostate MRI examinations were performed on a 3.0T scanner (Philips Ingenia, The Netherlands), and a pelvic 32-channel phased array coil was used. The mpMRI protocols used for image acquisition are as follows: axial T1-weighted imaging, axial and sagittal TSE T2-weighted imaging, single-shot echoplanar diffusion-weighted imaging (DWI), and dynamic contrast-enhanced (DCE) imaging. ADC maps were derived from DWI using b values of 0, 100, 1000, and 2000 s/mm2. DCE imaging was performed immediately after administering a contrast dose of 0.1 ml/kg gadopentetate dimeglumine (Magnevist, Bayer Schering Pharma), infused at a rate of 2–3 ml/s, followed by a saline flush of 20 ml. Details on image acquisition parameters are described in Table 1. The total prostate volume and transition zone volume were determined with the ellipsoid volume formula and the tumor size was decided on the base of T2 images, according to the ellipsoid volume formula: π/6 × transverse width × transverse length × longitudinal height. For whole gland PV, the width is the transverse dimension of the prostate, length is the craniocaudal distance from its apex to its base, and height is the anterior-posterior distance of the prostate. For TZV, the length is decided by the longest axis within the transition zone, the width is the shortest axis perpendicular to the length in the transverse plane, and the height is determined by the perpendicular dimension. PSAD was calculated by dividing the serum PSA by PV, the TZPSAD was calculated by dividing serum PSA by the measured TZV, and the TZ-ratio was decided by TZV/PV.
Prostate biopsy procedure
Lesions suspicious of PCa underwent MRI/US fusion biopsy within four weeks after MRI examination, by a urologist with 12 years of experience, using a color Doppler ultrasound device. Identified lesions were annotated on T2 images by the radiologists and segmented with real-time TRUS. For each lesion, at least two fusion-guided targeted cores (axial and sagittal planes) were obtained. Fusion biopsy was performed on a color Doppler ultrasound device, with a real-time virtual sonography imaging fusion system. An expert genitourinary pathologist evaluated the biopsy specimens and assigned each lesion a Gleason score (GS), who has an experience of 16 years and was blinded to the MRI findings. Each lesion suspicious of PCa was designated to the corresponding grade group according to the International Society of Urological Pathology (ISUP). The biopsy results were evaluated to determine the location and GS of an index tumor with the highest GS. In the case of multiple cores presented the highest GS, the core with the highest tumor-specific percentage was identified as the location of an index tumor. csPCa was defined as GS ≥ 7 (ISUP ≥ 2).
Statistical analysis
The mean ± standard deviation (SD) or median (interquartile range, IQR) is used to present descriptive statistics. For continuous variables, comparisons between two groups were evaluated using Mann-Whitney’s U test for non-normally distributed variables and independent t-test for normally distributed variables. The Shapiro-Wilks test was used to test the normal distribution of data. The sensitivity, specificity, positive likelihood ratio (LR+), and negative likelihood ratio (LR-) were calculated to evaluate the diagnostic accuracy. The optimal cutoff value that trade-off between sensitivity and specificity was determined by the Youden index. We first performed univariate analysis to identify the independent potential clinical variables associated with csPCa, including age, PSAD, PV, TZV, TZPSAD, and TZ-ratio. Subsequently, the multivariable logistic regression analysis was performed to investigate significant clinical factors for csPCa. The overall performance was assessed with the AUC and differences between models were compared with DeLong’s test19. All analysis was performed with R language (version 4.3.2), and P values ≤ 0.05 were considered statistically significant.
Results
Patient characteristics
The demographic characteristics are summarized in Table 2. At MRI/US fusion targeted biopsy, the cancer detection rate was 28/154 (18.18%). Significant differences were found between csPCa and non-csPCa in terms of age, PSAD, PV, TZV, TZPSAD, and TZ-ratio, with a P value of 0.001–0.04. However, there was no statistically significant between the two groups regarding PSA (P = 0.64).
ROC curve analysis
The ROC analysis revealed that TZ-ratio yielded the highest AUC (0.746, 95% CI 0.636–0.856) among several prostate volume-related variables, which was significantly superior to PSAD (0.644, 95% CI 0.538–0.751, P = 0.02), PV (0.634, 95% CI 0.494–0.753, P = 0.04), as shown in Fig. 2. However, it is not substantially better than TZPSAD (0.714, 95% CI 0.607–0.821, P = 0.55) or TZV (0.672, 95% CI 0.549–0.796, P = 0.09). The optimal cutoff value of PSAD for differentiating csPCa among PI-RADS score 3 lesions was 0.11 ng/ml/ml, where the sensitivity and specificity were 43.7% (95% CI 24.5%-62.8%) and 85.7% (95% CI 78.4%-91.3%), respectively. For TZPSAD, At the cutoff 0.21 ng/ml/ml, the sensitivity and specificity were 58.7% (95% CI 37.2%-75.5%) and 78.6% (95% CI 70.4%-85.4%). In the light of the TZ-ratio, at a threshold of 0.659 the calculated sensitivity and specificity were 73.0% (95% CI 51.3%-86.8%) and 75.0% (95% CI 63.5%-79.8%).
ROC Analysis for PSAD, TZPSAD, TZ-ratio, TZV, and the combination model of age, TZPSAD, and TZ-ratio. PSAD, prostate-specific antigen density; TZPSAD, transitional zone prostate-specific antigen density; TZ-ratio, transitional zone volume ratio; TZV, transitional zone volume.
Univariate and multivariate regression analyses for csPCa
Our univariable logistic regression analysis identified that age, PSAD, TZPSAD, TZV, and TZ-ratio were significant independent variables for the prediction of csPCa (Table 3). However, in the subsequent multivariable logistic regression analysis, variables of PSA, PSAD, and TZV were excluded. Therefore, only age, TZPSAD, and TZ-ratio were found to be significant factors associated with csPCa. The AUC for the combination of these 3 variables was 0.838 (95% CI 0.759–0.917), which was significantly superior to either TZPSAD (P = 0.001) or TZ-ratio alone (P = 0.03). Due to the limited positive cancer lesions, we evaluated the combination model with internal five-fold cross validation, which achieved a mean AUC of 0.814 ± 0.054, indicating stable performance. Figure 3 shows the confusion matrix of the true positive, false positive, false negative, and true negative for PSAD, TZPSAD, and TZ-ratio at the best cutoff values. Compared with PSAD, TZ-ratio can detect more csPCa (from 12 to 20), even though with a slight increase in biopsies. When combined age, TZ-ratio, and TZPSAD together, 3 more csPCa were detected.
Confusion matrix for PSAD, TZPSAD, TZ-ratio, and the combination model of age, TZPSAD, and TZ-ratio. PSAD, prostate-specific antigen density; TZPSAD, transitional zone prostate-specific antigen density; TZ-ratio, transitional zone volume ratio; TZV, transitional zone volume.
Discussion
In the current study, we investigated the potential ability of several variables for the prediction of csPCa classified as PI-RADS 3 located in TZ. Our analyses demonstrated that the two zonal-based variables of TZPSAD and TZ-ratio were significantly superior to PSAD, showing a potential capacity to differentiate csPCa in TZ. With respect to the variable of aTZPSAD, however, only exhibited moderate diagnostic performance. In our analysis, the optimal cutoff value of PSAD for differentiating csPCa was 0.11, which is slightly lower than the most reported value of 0.15 ng/ml. However, it should be noted that our study was focused on PI-RADS score 3 lesions. Regarding TZPSAD, the best threshold of 0.21 ng/ml/ml was in line with an earlier study, in which the optimal cutoff value for predicting PCa was 0.22 ng/ml/ml. When combined with 3 variables derived from multivariable logistic regression analysis, the yielded AUC demonstrated significant improvement as compared to using TZPSAD or TZ-ratio. The confusion matrix showed that with TZ-ratio, 8 more csPCa were detected as compared with PSAD, even though with slightly more biopsies. When combined age, TZ-ratio, and TZPSAD together, 3 more csPCa were detected with no more unnecessary biopsies.
The PI-RADS score 3 lesions denote an indeterminate probability of csPCa and present a clinical challenge between avoiding unnecessary biopsies and improving PCa detection. Some studies have confirmed the utility of PSAD as an independent indicator for csPCa, or combined with PI-RADS to improve the overall diagnostic performance. Nevertheless, PSAD showed moderate accuracy for PI-RADS category 3 lesions. Moreover, the optimal cutoff value for PSAD varies widely across studies. According to a recent meta-analysis, using PSAD as an independent indicator only yielded a sensitivity of 0.74 and a specificity of 0.61 at a threshold of 0.15 ng/ml/ml for PI-RADS 3 lesions11. Currently, the management of PI-RADS 3 lesions remains challenging due to the broad spectrum of associated csPCa and the limited consensus on their valuation.
The rationale behind the hypothesis of using TZPSAD and TZ-ratio as predictors for PCa is that as benign prostatic hyperplasia mainly arose from the transition zone, and vary of PSA due to benign prostatic hyperplasia should also result from the hypertrophied glands of the transition zone. Thus, the other prostate zone aside from the transition zone should be relatively constant and less influenced in benign prostatic hyperplasia. As a consequence, TZ-adjusted PSA levels that ignore PSA vary from the other prostate zones may enhance the ability of PSA to differentiate PCa. Since Kalish et al. first proposed the employment of TZPSAD as an independent predictor for PCa of lesions located in TZ, several studies have shown the promising potential of variables associated with PV13. In preliminary studies, the volume of the whole gland and transitional zone of the prostate was derived from the US modality, which is inferior to subsequent MRI in terms of precision. However, these studies still demonstrated the promising ability of TZPSAD and TZV for PCa15,16,18,20. Nevertheless, there was controversy regarding the role of zone-adjusted PSAD. In the study of Elliott et al., no advantage was seen when using transition zone density over prostate-specific antigen density for any PCa (P = 0.056); however, for csPCa TZPSAD was significantly superior to PSAD, with AUC 0.727 vs. 0.673, P < 0.00121. In a recent study, Schneider et al. compared whole gland PSAD and TZPSAD for risk stratification of men with suspected PCa. They found that TZPSAD demonstrated a stronger correlation than PSAD for differentiating PCa22. To our knowledge, our study is the first focused on PI-RADS 3 lesions. Our findings showed that when combined with age, TZPSAD, and TZ-ratio, the diagnostic performance was improved significantly as compared to using each of them alone. However, due to too few positive csPCa, it is unfeasible to test our model in a validation set. Nonetheless, some studies have demonstrated combination model or nomogram could substantially improve the overall performance18. In addition to TZ, some studies reported that PZPSAD also showed higher performance than the use of PSAD17,23. Chen et al. reported that for PZ csPCa, the AUC for PZPSAD (0.824) was significantly superior to PSAD (0.792, P < 0.01) and PZ-ratio (0.717, P < 0.01). In another study, the calculated AUC for PZPSAD was 0.81223.
Our study has limitations that should be acknowledged. First, this is a retrospective, single-center study, which may affect the generalization of our conclusion in clinical application. Thus, there is a need for prospective, multicenter studies to provide external validation. Second, because of too few positive samples, it is unfeasible to validate the model of combining age, TZPSAD, and TZ-ratio with independent test set. However, we performed internal five-fold cross validation. Third, the reference standard was fusion targeted biopsy, which may miss some potential lesions with a negative MRI but positive pathology.
Conclusions
The findings of the current study suggest that variables associated with prostate volume such as TZV, TZPSAD, and TZ-ratio all were independent predictors for the prediction of csPCa in PI-RADS score 3 lesions; moreover, we found that TZPSAD and TZ-ratio were significantly superior to PSAD. Notably, integrating TZPSAD, TZ-ratio, and age can significantly enhance the predictive accuracy for csPCa.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249. https://doi.org/10.3322/caac.21660 (2021).
Sandhu, S. et al. Prostate cancer. Lancet 398, 1075–1090. https://doi.org/10.1016/S0140-6736(21)00950-8 (2021).
Tamada, T. et al. Prostate cancer detection in patients with total serum prostate-specific antigen levels of 4–10 ng/mL: diagnostic efficacy of diffusion-weighted imaging, dynamic contrast-enhanced MRI, and T2-weighted imaging. AJR Am. J. Roentgenol. 197, 664–670. https://doi.org/10.2214/AJR.10.5923 (2011).
Barentsz, J. O. et al. ESUR prostate MR guidelines 2012. Eur. Radiol. 22, 746–757. https://doi.org/10.1007/s00330-011-2377-y (2012).
Weinreb, J. C. et al. PI-RADS prostate imaging—reporting and data system: 2015, version 2. Eur. Urol. 69, 16–40. (2016).
Turkbey, B. et al. Prostate imaging reporting and data system version 2.1: 2019 update of prostate imaging reporting and data system version 2. Eur. Urol. 76, 340–351 https://doi.org/10.1016/j.eururo.2019.02.033. (2019).
Maggi, M. et al. Prostate imaging reporting and data system 3 category cases at multiparametric magnetic resonance for prostate cancer: a systematic review and meta-analysis. Eur. Urol. Focus. 6, 463–478. https://doi.org/10.1016/j.euf.2019.06.014 (2020).
Washino, S. et al. Combination of prostate imaging reporting and data system (PI-RADS) score and prostate-specific antigen (PSA) density predicts biopsy outcome in prostate biopsy naïve patients. BJU Int. 119, 225–233. https://doi.org/10.1111/bju.13465 (2017).
Görtz, M. et al. The value of prostate-specific antigen density for prostate imaging-reporting and data system 3 lesions on multiparametric magnetic resonance imaging: a strategy to avoid unnecessary prostate biopsies. Eur. Urol. Focus. 7, 325–331. https://doi.org/10.1016/j.euf.2019.11.012 (2021).
Stevens, E. et al. Clinical utility of PSAD combined with PI-RADS category for the detection of clinically significant prostate cancer. Urol. Oncol. 38, 846.e9–846.e16 https://doi.org/10.1016/j.urolonc.2020.05.024 (2020).
Wang, S. et al. Diagnostic performance of prostate-specific Antigen density for detecting clinically significant prostate cancer in the era of magnetic resonance imaging: a systematic review and meta-analysis. Eur. Urol. Oncol. S2588-9311(23)00165-7 https://doi.org/10.1016/j.euo.2023.08.002 (2023).
Pinsky, P. F. & Parnes, H. Screening for prostate cancer. N Engl. J. Med. 388, 1405–1414. https://doi.org/10.1056/NEJMcp2209151 (2023).
Kalish, J., Cooner, W. H. & Graham, S. D. Serum PSA adjusted for volume of transition zone (PSAT) is more accurate than PSA adjusted for total gland volume (PSAD) in detecting adenocarcinoma of the prostate. Urology 43, 601–606. https://doi.org/10.1016/0090-4295(94)90170-8 (1994).
Chang, T-H. et al. Zonal adjusted PSA density improves prostate cancer detection rates compared with PSA in Taiwanese males with PSA < 20 ng/ml. BMC Urol. 20, 151. https://doi.org/10.1186/s12894-020-00717-z (2020).
Sung, D. J. et al. Comparison of prostate-specific antigen adjusted for transition zone volume versus prostate-specific antigen density in predicting prostate cancer by transrectal ultrasonography. J. Ultrasound Med. Off J. Am. Inst. Ultrasound Med. 23, 615–622. https://doi.org/10.7863/jum.2004.23.5.615 (2004).
Tang, P. et al. Transition zone PSA density improves the prostate cancer detection rate both in PSA 4.0–10.0 and 10.1–20.0 ng/ml in Chinese men. Urol. Oncol. 31, 744–748. https://doi.org/10.1016/j.urolonc.2011.06.012 (2013).
Chen, Z. et al. A novel clinically significant prostate cancer prediction system with multiparametric MRI and PSA: P.Z.A. score. BMC Cancer. 23, 1138. https://doi.org/10.1186/s12885-023-11306-2 (2023).
Wang, Y. et al. Prostate transitional zone volume-based nomogram for predicting prostate cancer and high progression prostate cancer in a real-world population. J. Cancer Res. Clin. Oncol. 143, 1157–1166. https://doi.org/10.1007/s00432-017-2389-3 (2017).
DeLong, E. R., DeLong, D. M. & Clarke-Pearson, D. L. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44, 837–845 (1988).
Djavan, B. et al. Prostate specific antigen density of the transition zone for early detection of prostate cancer. J. Urol. 160, 411–418. https://doi.org/10.1016/S0022-5347(01)62911-2 (1998).
Elliott, C. S., Shinghal, R. & Presti, J. C. The performance of prostate specific antigen, prostate specific antigen density and transition zone density in the era of extended biopsy schemes. J. Urol. 179, 1756–1761. https://doi.org/10.1016/j.juro.2008.01.033 (2008).
Schneider, A. F. et al. Comparison of PSA-density of the transition zone and whole gland for risk stratification of men with suspected prostate cancer: a retrospective MRI-cohort study. Eur. J. Radiol. 120, 108660. https://doi.org/10.1016/j.ejrad.2019.108660 (2019).
Huang, C. et al. New model of PIRADS and adjusted prostatespecific antigen density of peripheral zone improves the detection rate of initial prostate biopsy: a diagnostic study. Asian J. Androl. 25, 126. https://doi.org/10.4103/aja202218 (2023).
Author information
Authors and Affiliations
Contributions
J.Y.M. and C.F.: conceptualization and writing; X.G. and W.C.G.: software and statistical analysis. W.C.G. and D.C.S.: data collection and writing—original draft; J.Y.M and C.F.: writing—review; D.C.S.: Project development and Supervision.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Written informed consent was waived by the Institutional Review Board.
Consent for publication
Patients’ data were anonymized. Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jin, Y., Chen, F., Xu, G. et al. Transition zone-based prostate-specific antigen density for differentiating clinically significant prostate cancer in PI-RADS score 3 lesions. Sci Rep 15, 3258 (2025). https://doi.org/10.1038/s41598-025-87311-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-87311-1