Abstract
Radiation therapy is actively utilized for superficial lesions. External beam radiotherapy for cutaneous lesions utilizes electrons with low transmittance. Conventional electron beam therapy uses Cerrobend blocks for field shaping; however, the Intraoperative Radiotherapy (IORT) applicator offers superior dosimetric characteristics. The dosimetric parameters were measured using 4 and 6 MeV electron beams delivered by a Trilogy linear accelerator, and percent depth dose and lateral dose profiles were compared under the presence of the IORT applicator and Cerrobend block. The dose calculations under various IORT applicator conditions and planning studies were performed using Monte Carlo simulation. Treatment plans for three sites were evaluated in terms of coverage of the planning target volume, dose to the surrounding normal tissue, and beam-on time for two treatment modalities. The results of the measured and calculated dosimetric parameters correspond. Scattered electrons along the IORT applicators resulted in shorter dmax and R50 and sharper penumbras compared to the blocks. Oblique IORT applicators also maintained sharp penumbras. Treatment-plan analysis indicated significant reductions in normal tissue dose using the IORT applicator. Implementing IORT in clinical practice requires deliberation of extended beam-on times and associated patient safety protocols; however, the potential benefits regarding dose distribution warrant further optimization in clinical use.
Similar content being viewed by others
Introduction
Radiation therapy targeting various superficial lesions is being actively performed. Immediate postoperative radiotherapy can significantly reduce the recurrence rate of keloids, which have a high recurrence rate when only surgical resection is performed1,2,3. Palliative or definitive radiotherapy for skin lesions is also performed for locally onset cutaneous lymphoma or for the remaining lesions after total skin electron beam treatment4,5,6,7. Radiotherapy for skin lesion should be able to focus the radiation on the superficial region while minimally exposing the normal cells to radiation deep within the skin. Therefore, most external beam radiotherapies on skin lesions are performed using radiation with low transmittance, such as superficial, orthovoltage X-rays, or electron beams8,9. However, given their specialized role in treating skin lesions like melanoma, superficial or orthovoltage X-ray machines are scarce in regions with low melanoma incidence rates compared to megavoltage linear accelerators that are used for a broader spectrum of cancers.
Conventional electron beam therapy often utilizes Cerrobend blocks inserted into rectangular applicators to shape the treatment field. These applicators are typically designed with a 5-cm air-gap between the block and the skin of the patient to ensure adequate gantry clearance during treatment. However, this air-gap can contribute to increased beam penumbra. To counteract this, an additional margin is required around the target lesion10. In contrast, the Intraoperative Radiotherapy (IORT) applicators, equipped with cylindrical or beveled cones, can be positioned closer to the lesion, which minimizes the air gap and results in a sharper penumbra11. This enables smaller treatment margins and potentially improved dose conformity compared to blocks.
The primary application of the IORT applicator is literally for use in intraoperative radiation therapy. Some previous studies have investigated the dosimetric characteristics of IORT applicators using Monte Carlo simulations. For instance, Guerra et al.12 performed a Monte Carlo-based study to simulate the structure of the Varian 21EX linear accelerator with an IORT applicator, enabling dose calculations and treatment plan generation. Additionally, they evaluated PTV dose and shielding effectiveness for the rib and lung in a breast IORT treatment case. Similarly, Alhamada et al.13 utilized Monte Carlo simulations to evaluate dose distributions, including PDD and profiles, for the Mobetron 1000, a dedicated intraoperative radiation therapy device. They also extended to dose calculations on CT images of breast IORT patient.
While both studies established systems for calculating dose distributions with IORT applicators, their primary focus was on building computational models on planning CT rather than analyzing the dose distributions and comparing them with existing radiotherapy techniques. Furthermore, the applicability of IORT applicators was limited to intraoperative radiation therapy, and their suitability for skin treatments was not assessed.
Tresoldi et al.14 conducted a clinical study exploring the application of IORT applicators in the immediate postoperative radiotherapy of keloids. They treated 16 patients with 21 lesions across eight anatomical sites: the abdomen, deltoid, earlobe, sternum, neck, back, chest, and scapula. Using a mobile linear accelerator with 6 MeV electrons, they aimed to concentrate the dose on superficial lesions while minimizing deep dose delivery due to the low energy of the electron beam. While favorable outcomes were achieved in 90.5% (19/21) of cases with no recurrence observed, they did not physically evaluate the dosimetric aspects. Furthermore, in contrast to the approach used in their study, applying the IORT applicator to a conventional MV linear accelerator could expand its utility more efficiently, achieving therapeutic benefits similar to those reported in the study.
This study aims to evaluate the dosimetric advantages of IORT applicators and investigate their suitability for various skin lesion radiotherapy treatments, including keloids, using a clinical linear accelerator, thereby expanding the applicability of the IORT applicators. To assess these advantages, the dosimetric characteristics derived from phantom measurements were compared, and Monte Carlo-based treatment plans were generated for both the IORT applicator and Cerrobend block conditions.
Methods
Dosimetric parameter measurement
Radiation therapy for small skin lesions, such as those caused by mycosis fungoides or cutaneous lymphoma, and for postoperative keloids, typically targets dermal layers at depths of a few millimeters to one centimeter15,16,17. Because the electron ranges in water are approximately 2 cm and 3 cm for 4 MeV and 6 MeV beams, respectively, these energies were chosen for this study.
Electron beams were delivered using a Trilogy linear accelerator (Varian Medical Systems, CA, USA) for the dosimetric parameter measurements. The percent depth dose (PDD) and lateral dose profiles were compared using an IORT applicator and a Cerrobend block. PDD was measured using an Electron Field Diode 3G diode detector (IBA dosimetry, Schwarzenbruck, Germany) in a one-dimensional water phantom (Civco Medical Solutions, IA, USA). The lateral dose profiles were obtained using Gafchromic EBT3 films (Ashland, DE, USA) at the surface and at dmax depth of the water-equivalent solid phantoms.
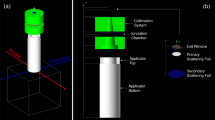
A periscopic electron cone for IORT (Radiation Products Design, MN, USA) with a 38 mm inner diameter (Fig. 1) was used in this study. The applicator tip was positioned in contact with the phantom during irradiation, and the source-to-surface distance (SSD) of 108 cm was confirmed through direct measurement performed by the authors during the experimental setup. A Cerrobend block, designed to match the 38 mm IORT applicator field size at an SSD of 100 cm, was inserted into a 10 cm × 10 cm electron applicator. That is, the setup was configured to create a field size of 38 mm in diameter on the phantom surface at each respective SSD (Fig. 1). To simulate the clinical conditions, a 5 cm air-gap was introduced between the block and the phantom surface.
(a) Acrylic IORT applicators with inner diameters of 25, 38 and 51 mm used in this study. (b) An IORT applicator mounted on a Varian Trilogy linear accelerator. (c) Setup configurations for the 38 mm field size: Cerrobend block setup and IORT applicator setup. In the Cerrobend block setup, the block is positioned within a Varian-provided electron applicator, maintaining a fixed distance of 5 cm between the block and the surface. For the IORT applicator setup, the applicator requires a physical SSD of 108 cm.
Dosimetric parameter calculation using Monte Carlo simulation
The 4 MeV and 6 MeV beams of the Trilogy were modeled in the GEANT418-based Monte Carlo (MC) simulation toolkit, Tool for Particle Simulation (TOPAS)19. For the 4 MeV beam, the energy was set to 3.9 MeV with an energy spread of 8%. The beam position distribution was modeled using a Gaussian distribution, with 1 sigma set to 0.07 cm and distributions up to 2 sigma considered. The beam angular distribution was also modeled as a Gaussian distribution, with 1 sigma set to 1.5º and a cutoff at 90º. For the 6 MeV beam, the energy was set to 6.4 MeV, while the other parameters remained the same as for the 4 MeV beam. These beam parameters were tuned to achieve agreement within 2% of the measurement under the 25 cm × 25 cm sized electron applicator20. The IORT applicator and Cerrobend block conditions were simulated in TOPAS to replicate their respective measurement conditions, and the calculated dosimetric parameters (PDD, lateral dose profile, and effective SSD) were compared with the measurement results. In addition to the measurement conditions, dose calculations were also performed for IORT applicators with diameters of 25 mm and 51 mm and end angles of 0º, 15º, 30º, and 45º. Dose calculations were performed for the Cerrobend block at various angles with a field size of 38 mm in diameter to evaluate and compare the dose distribution with that of the IORT applicator. To compare the beam-on time between the IORT applicator and the Cerrobend block conditions, the output factors for those conditions were derived relative to the reference condition of a conventional 15 × 15 cm² sized electron applicator. This selection aligns with the IAEA guidelines, which state that the reference field size should not be less than 10 × 10 cm² at the phantom surface and that a convenient choice for normalization may be used if larger. The 15 × 15 cm² field size was chosen because it is commonly used in clinical practice and ensures sufficient lateral scatter equilibrium for the energies studied.
Planning study with Monte Carlo simulation
Open-source CT data provided by the Cancer Image Archive21 was used to mimic electron radiation therapy planning for skin lesions. Based on our experience, three mainly treated sites were considered: earlobe, nose, and cheek. The nose represents the nasal bridge, and the cheek corresponds to the mid cheek area adjacent to the nose. The earlobe corresponds to the cartilage-free lobule below the ear canal and partially includes the antitragus. The planning target volume (PTV) and the volume of normal tissue surrounding PTV (surrounding normal volume; SNV), and 1 cm lateral margin with respect to the beam direction, were delineated on the open-source CT images by using Eclipse (Version 15.6, Varian, USA), which were imported into TOPAS. Figure 2 shows the segmented PTV and SNV for the three treatment cases. Considering the effect of the PTV shape on dose distribution, an irregular PTV shape was set for the earlobe treatment case, while a simple and round PTV shape was set for the nose and cheek treatment cases. The beam angle was decided based on the angle of the skin surface and the PTV shape. A Cerrobend block was created with a 7 mm margin on the PTV shape viewed from the beam direction, which was perpendicular to the skin surface. The 25 mm IORT applicator was set to include a minimum of 5 mm margin around the PTV. For the nose and cheek cases, the treatment plans were generated using a flat IORT applicator under the same beam angle conditions as the Cerrobend block plan. For the earlobe case, a beveled IORT applicator with a 45° tip was used, which enables beam delivery at an oblique angle. The prescription dose was set to 600 cGy/fx (D50%. Mean dose of PTV) for both conditions. The prescription dose was set to 600 cGy/fx, corresponding to the mean dose of the PTV (D50%), for both conditions. This approach, while deviating from ICRU 71 guidelines22, was chosen to address the challenges in achieving a uniform dose distribution within the PTV (± 5%) and to avoid hotspots. This clinical decision prioritizes the reduction of dose to surrounding normal tissues, especially in the treatment of benign conditions like keloids. The monitor unit (MU) for the treatment plan was derived through monitor unit scaling based on the results of the reference condition. Thereafter, the PTV coverage, SNV dose, and beam-on time were evaluated for the beams under presence of the IORT applicator and Cerrobend block, respectively.
Beams-eye-view diagrams and axial plane images of the planning target volumes (Magenta) and surrounding normal volumes (Yellow) for representative (a) nose, (b) cheek, and (c) earlobe treatment cases. The images were created using Eclipse version 15.6 (Varian, USA).
Gamma analysis
To evaluate the agreement between the measured and simulated dosimetric parameters, gamma analysis was performed with 2%/2 mm criteria, following the recommendations of AAPM TG-21823. The gamma index was calculated for both surface and depth dose profiles, comparing the measured values obtained using Gafchromic EBT3 films with those simulated in TOPAS. A passing rate threshold of 95% was used to determine acceptable agreement, and the gamma passing rates at the surface and dmax were specifically analyzed to validate the Monte Carlo simulations against the experimental measurements.
Results
Validation of the IORT applicator modeling with Monte Carlo simulation
The beam parameters, including the mean energy of the beams incident to the scattering foil, were tuned to the 25 cm × 25 cm sized electron applicator condition in TOPAS20. With the inclusion of the IORT applicator geometry, the mean energy was slightly more tuned at the sub-millimeter level based on the phantom measurements. Figure 3 shows the PDD and lateral dose profiles of the 4 MeV and 6 MeV electron beams with 38 mm, 0º IORT applicator from the measurements and the TOPAS MC calculation. The calculated PDD value corresponded with the measured data within 2% for the whole range. The dmax was the same as 7 mm for the 4 MeV electron beam and was 12 mm for the 6 MeV beam. Furthermore, there was no significant difference in the estimated R50 and practical range (Rp) between the two results. For the dose profile, the gamma analysis results for 2% and 2 mm criteria showed a passing rate of over 95%. The gamma passing rate at dmax was 99.9%, while the passing rate at the surface was slightly lower (99.7% and 95.3% for the 4 MeV and 6 MeV beams, respectively). The penumbra for the 20–80% dose had a difference within 0.2 mm, and the full-width half-maximum (FWHM) showed a maximum of 0.2 mm difference.
(a) Measured (symbols) and calculated (lines) percent depth dose for 4 and 6 MeV electron beams with a 38 mm IORT applicator. (b,c) Measured (black lines) and calculated (red symbols) lateral dose distributions at the surface and at depth of maximum dose (dmax).
Comparison of the dosimetric parameters between the IORT applicator and the Cerrobend block
Using the validated MC model, the dose distributions for various sized IORT applicators were calculated for the 4 and 6 MeV electron beams. Figure 4 shows the calculated PDD for the different IORT applicators and Cerrobend blocks (diameter: 25, 38, and 51 mm). The IORT applicator exhibited a shorter dmax and R50 compared to the block by approximately 1 mm and 0.7 mm, respectively. The IORT applicator increased the surface dose by 3–8% compared to the Cerrobend block, with a more pronounced effect for a larger field size. For a field size of 25 mm in diameter, the surface dose difference between the two treatment methods was 3%; however, this difference increased to 8% for a 51 mm field size. The Rp value did not exhibit a significant difference between the IORT applicator and the block. These results indicate that the IORT applicator shifts the overall dose distribution towards the water surface.
Calculated percent depth dose for the 4 MeV (black lines) and 6 MeV (red lines) electron beams with (a) 25 mm, (b) 38 mm, and (c) 51 mm IORT applicators (solid lines) compared to Cerrobend blocks (dashed lines) with equivalent field areas.
Figure 5 shows the lateral dose profile for the two conditions obtained using TOPAS. The surface and dmax dose profiles were normalized to the dose at the center of the dmax profile. The penumbras were narrower for the IORT applicator for all the conditions. The surface dose profiles for the IORT applicator had a uniform central dose, and the penumbras were reduced by approximately 5 to 7 mm compared to the Cerrobend block. Even at dmax, the IORT applicator demonstrated a 90% dose of Dmax and covered 4 mm wider on one side, and the penumbras were sharper by approximately 4 to 6 mm. The difference in the dose profile between the IORT applicator and the block was remarkable for the 4 MeV electron beam.
Radial dose distributions at the surface (dashed lines) and at dmax (solid lines) for the IORT applicator (red lines) and Cerrobend block (black lines) conditions. Field sizes of (a) 25 mm, (c) 38 mm, and (e) 51 mm for the 4 MeV electron beams, and (b) 25 mm, (d) 38 mm, and (f) 51 mm for the 6 MeV electron beams. The IORT applicator demonstrates reduced out-of-field dose compared to the Cerrobend block. For example, at the surface, the dose at 5 mm beyond the field edge is less than 3% for the IORT applicator (indicated by the red arrow), while the Cerrobend block requires an additional 5 mm distance to achieve the same dose level (indicated by the black arrow).
Dosimetric characteristics for the beveled IORT applicator
Figure 6 shows the calculated PDD for beveled applicators. Since PDD was obtained from the center of the beam on the water surface perpendicularly, the PDD graph shifts towards the surface with increasing end angle. For the 4 MeV electron beam, the dmax, R50, and Rp of the 45º IORT applicator were shortened by approximately 4 mm, 5 mm, and 2 mm, respectively. The effect was more pronounced with the 6 MeV electron beam, which had dmax, R50, and Rp reductions of approximately 7.5–8.5 mm, 8 mm, and 6 mm, respectively. Compared to the Cerrobend block at the same angle, the IORT applicator increased the surface dose by approximately 2% and shortened dmax and R50 by about 1 mm, showing differences similar to those observed at 0º for the two conditions (Fig. 6 (g, h)).
Percent depth dose (PDD) as a function of the IORT applicator angle (0º, 15º, 30º, 45º) for the field sizes of (a) 25 mm, (c) 38 mm, and (e) 51 mm for the 4 MeV electron beam, and (b) 25 mm, (d) 38 mm, and (f) 51 mm for the 6 MeV electron beam. PDD data were acquired perpendicular to the water surface at the central axis of the beam. Comparison of PDD between IORT applicators and Cerrobend blocks for (g) 4 MeV electron beams and (h) 6 MeV electron beams at 15º, 30º, and 45º.
Figure 7 presents the planar dose distribution along the longitudinal direction (major axis direction for the beveled IORT applicator) from the field center, lateral dose profiles at the surface, and dmax for the 38 mm, 45º beveled IORT applicator. As the end angle increased, the field width along the major axis direction widened, and the dose distribution was tilted according to the beam direction. This tilt translated to a shift in the dmax by approximately 3.5 mm for 4 MeV and 5.5 mm for 6 MeV electron beams. The degree of the shift did not significantly vary with the field size; however, it showed a slight dependence on the angle. Compared to the surface dose profile, the lateral dose profile at dmax was shifted by approximately 2.5 mm (4 MeV) and 4.5 mm (6 MeV) for a 30º angle and by 2 mm (4 MeV) and 3.5 mm (6 MeV) for a 15º angle (Supplementary 1). The penumbra (20–80%) at dmax did not exhibit significant differences in both directions. However, a dose below 20% of the maximum dose tended to propagate in the direction of the beam. Compared to the measurements for the 45º beveled IORT applicator, the Cerrobend block results showed a similar degree of beam shift in the depth direction. However, the penumbra at the surface and dmax for the Cerrobend block was approximately 1.5 cm for the larger value, which was about 1 cm broader than that observed under the IORT applicator condition.
Planar depth dose distribution for (a) 4 MeV and (b) 6 MeV electron beam, along the major axis of a 38 mm, 45º beveled IORT applicator. The lateral dose profiles at the surface and dmax for the IORT applicator conditions with (c) 4 MeV and (d) 6 MeV electron beams, respectively, and the results for the Cerrobend block conditions under (e) 4 MeV and (f) 6 MeV electron beams. The dmax dose profile for the 6 MeV electron beam shows a larger shift from surface dose profile, compared to the results for the 4 MeV electron beam. The planar depth dose distribution was created using MATLAB R2023b (The MathWorks, INC. USA).
Output factor
Table 1 summarizes the calculated output factors for the different IORT applicator field sizes. As expected, the output factor showed a dependence on the field size, with smaller fields having lower output factors. Interestingly, no angular dependence was observed, which can be attributed to the shallower dmax in the beveled applicators. We compared the calculated output factors between the IORT applicator and the block. The results show that the output factor for the IORT applicator was consistently lower than the Cerrobend block, with the difference ranging from approximately 15–65%. This variation is influenced by beam energy and field size. In the 4 MeV beam, which has a shorter range, and the 25 mm field size, where substantial particle loss occurs due to increased scattering along the IORT applicator wall, the difference in output factor reached as high as 65%. Conversely, higher-energy beams, such as the 6 MeV beam, or larger field sizes demonstrated smaller differences, with the output factor difference decreasing to 15% in conditions where scattering and particle loss were less pronounced.
Planning study with Monte Carlo simulation
Figure 8 shows the dose volume histograms (DVHs) of the PTV and the SNV for the three treatment plans, one using an IORT applicator and the other using a Cerrobend block. For the nose treatment plan, the PTV coverage was comparable between the two modalities, with D95% values of 533.1 cGy and 541.7 cGy for the IORT applicator and Cerrobend block, respectively. The IORT applicator increased the maximum PTV dose by 14.2% compared to the prescribed dose, while the block demonstrated a 9.0% increased dose compared to the prescription. However, the IORT plan significantly reduced the mean SNV dose by 32% (170.5 cGy for the IORT applicator versus 251.4 cGy for the Cerrobend block). While 3% of the SNV received slightly higher doses with the IORT plan (about 4% larger than the maximum dose), the majority (97%) received significantly lower doses (D5%: 497.9 cGy for IORT versus 510.0 cGy for block). Assuming a fractionation scheme of 600 cGy per treatment session, the IORT applicator plan required a substantially higher number of monitor units (3482 MU) compared to the Cerrobend block plan (1125 MU), which resulted in a three-fold increase in the beam-on time.
Dose-volume histograms (DVHs) of the PTV (red line) and margin (black line) for (a) nose, (b) cheek and (c) earlobe radiotherapy. Solid lines represent the DVH for plans using IORT applicator, and dashed lines represent the DVH for conventional plan using a Cerrobend block.
The cheek treatment plans using the IORT applicator demonstrated a 41% reduction in the mean SNV dose (236.9 cGy for IORT versus 401.7 cGy for block). The IORT plan demonstrated higher doses for only 0.12% of the SNV, with a 1.2% increase in the maximum SNV dose (622.0 cGy for IORT versus 614.7 cGy for block). The PTV coverage was slightly compromised in the IORT plan, with approximately 7% lower D95% and 1.3% higher maximum dose, which caused reduced dose homogeneity within the PTV. The output factor of the IORT plan was 3.19 times greater than that of the block plan.
For the earlobe treatment case, the IORT treatment plan slightly compromised the PTV coverage and significantly reduced the SNV dose, which is similar to the previous two cases. While the D5% of the PTV differed by approximately 4.4% (654.4 cGy for IORT versus 683.0 cGy for block), the mean SNV dose was reduced by 63% in the IORT plan (119.6 cGy for IORT versus 323.9 cGy for block). In this case, the IORT plan required a 3.35-fold increase in the beam-on time compared to the block plan.
Discussion
Dosimetric characteristics of the IORT applicator
For intraoperative radiotherapy, the IORT applicator was designed as a cylindrical tube to focus a high dose on the target area in a single fraction. In this study, the applicator was made of cylindrical polymethyl methacrylate (PMMA), with varying diameters and beveled angles to facilitate customized treatment based on the lesion size and morphology. This approach aligns with the intraoperative radiotherapy principles for skin lesion treatment.
The acrylic or metallic IORT applicator shapes the circular beam by placing the applicator end close to the lesion. As the electrons undergo continuous scattering within the applicator before reaching the target, the central dose distribution becomes more uniform with sharper field edges24. Moreover, the electrons that lose energy through scattering along the applicator wall can increase the dose at the skin surface24,25. Consequently, the IORT applicator is expected to exhibit superior dosimetric characteristics compared to the conventional treatment methods for skin lesion.
The IORT applicator reduces the mean energy of the incident electron beam, which results in shallower penetration depth. Figure 9 shows the MC simulation results of the electron energy distribution incident on the phantom surface for the 4 MeV electron beam with a 25 mm diameter circular field. The results demonstrated an increased proportion of scattered, low-energy electrons under the IORT applicator condition. While the primary electrons that did not interact with the IORT applicator or block maintained a similar Rp, the IORT applicator shifted the energy distribution of the scattered electrons to lower energies compared to the block. Consequently, under the IORT conditions, the surface dose increased and dmax was displaced closer to the surface.
Energy distribution of the particles incident on the surface of the water phantom.
The IORT applicator mitigates the effect of the dosimetric parameters based on the field size. Scattered electrons reduce the influence of the side scattering equilibrium. When the field size is smaller than the lateral range of electrons in the medium, the depth dose-distribution shifts towards the surface of the medium26. For instance, a Cerrobend block with a 25 mm diameter shortened the dmax by 3 mm and increased the surface dose by 12% compared to larger blocks. However, the IORT applicator exhibited less variation in the PDD depending on the field size. There was minimal change in the PDD for the 4 MeV electron beam, and for the 6 MeV electron beam, the dmax variation was 1 mm less and the surface dose difference was approximately 5% less than the block. Lazarus et al., using the same IORT applicator system as our study, reported that the dmax of the 19 mm IORT applicator with 6 MeV electron beams was 0.75 cm, while that of the 64 mm IORT applicator was 1.35 cm27. This discrepancy in our results occurred owing to the shorter SSD and the presence of a 1 cm air gap, which potentially affected beam spreading.
Scattered electrons along the IORT applicator contributed to a homogeneous surface dose. Figure 10 shows the calculated lateral dose distribution at the phantom surface from the primary and secondary electrons under various IORT applicator and Cerrobend block conditions. For the 25 mm IORT applicator, the dose distribution from the primary electrons exhibited a maximum at the center and decreased by approximately 20% near the field edges. Conversely, the dose distribution from the secondary electrons was approximately 20% higher at the edges compared to the center. The comparable contributions of the primary and secondary electrons at the surface resulted in a homogeneous total surface dose distribution. Similarly, for the 51 mm IORT applicator, the secondary electrons augmented the edge dose by approximately 50% relative to the center, which compensated for the reduced edge dose from the primary electrons. Essentially, the scattered secondary electrons enhanced the dose at the field edges, which resulted in a homogenous overall surface dose distribution. Furthermore, the close contact between the IORT applicator and the phantom (or skin surface) minimizes the electron scatter out of the radiation field, thereby sharpening the penumbra. The out-of-field dose was consistently below 3%, and the lateral penumbra on the surface dose remained under 1 mm in all the cases, irrespective of the applicator size and energy.
Surface radial dose distribution (4 MeV) of the primary and secondary particles for (a) 25 mm IORT applicator, (b) 51 mm IORT applicator, (c) 25 mm Cerrobend block and (d) 51 mm Cerrobend block conditions. For the results of the block, the contribution of secondary particles is much smaller compared to the primary particles. Therefore, to better represent the dose distribution on the graph, it was scaled up by a factor of 10. The results are normalized to the maximum dose of the primary particles.
In contrast, under the Cerrobend block condition, the high density (~ 9.4 g/cm3) of the material resulted in only 4% of the electrons passing through the phantom surface being classified as secondary electrons. Their dose contribution was diminished by a factor of 1/33 compared to the primary electron dose (Fig. 10). Because the primary particles dominate the dose contribution, the dose at the edge of the field on the surface was approximately 50% lower than that at the field center. The air gap between the block and the surface facilitated the scattered electrons to spread out of the field. Therefore, unlike the IORT condition, the secondary particle dose did not increase at the field boundary but tended to spread outward.
To directly compare clinically relevant conditions, this study employed a physical SSD of 108 cm for the IORT applicator and 100 cm for the Cerrobend block. In electron beam therapy using a Cerrobend block, the penumbra widens with increasing SSD due to a larger air gap, which enhances electron scattering. However, the IORT applicator showed minimal dosimetric variation with SSD changes as it operates in direct contact with the patient’s surface, eliminating the air gap. This study acknowledges that simultaneous changes in SSD and beam-shaping devices may complicate the interpretation of comparative outcomes. However, the chosen setup reflects clinical conditions, where such variations are inevitable due to the design and application requirements of beam-shaping devices in clinical practice. Therefore, this study prioritizes clinically relevant scenarios over artificially controlled conditions to better represent real-world applications. Monte Carlo simulations comparing lateral dose distributions for the IORT applicator at SSDs of 100 cm and 108 cm revealed penumbra differences of less than 1 mm across all conditions, indicating that the IORT applicator is largely unaffected by SSD changes (Supplementary Fig. 2). Similarly, no difference in PDD was observed between the two SSD conditions. According to TP O’Shea et al.28, 6–12 MeV electron beams exhibit approximately a 3% dose reduction in the build-up region when the SSD increases from 100 cm to 120 cm, with minimal changes beyond the build-up region. In contrast, this study observed that the IORT applicator setup with a longer SSD resulted in a 3–8% increase in dose in the build-up region compared to Cerrobend block setup. These findings suggest that the dosimetric differences between the IORT applicator and the Cerrobend block are primarily due to the intrinsic characteristics of the beam-shaping devices rather than SSD variations. The results highlight the IORT applicator’s ability to mitigate the effects of SSD extension by concentrating the dose more effectively, thereby enhancing dose shaping.
For skin radiotherapy, it is advantageous to enhance the surface dose, reduce the deep skin dose, and narrow the penumbra to focus the dose on the target lesion. While only minimal differences were observed between the IORT applicator and the Cerrobend block in small lesion treatments, the IORT applicator exhibited superior dose enhancement in the superficial region for larger treatment fields with diameters of 38 mm or greater. Furthermore, the applicator consistently reduced the lateral dose penumbra across all the field sizes. Consequently, the IORT applicator yielded a more favorable dose distribution in the electron beam treatment of cutaneous lesions compared to the conventional Cerrobend block techniques.
Beveled IORT applicator
The conventional electron beam radiotherapy for cutaneous lesions utilizing Cerrobend blocks typically employs a beam delivered perpendicular to the skin surface. This approach minimizes the “penumbra” effect, which is characterized by a region of decreased dose at the periphery of the treatment field. However, anatomical constraints may necessitate angled beam delivery, which can lead to several dosimetric challenges. First, an increased air gap can exacerbate the penumbra effect on the distal side of the beam. This results in a broader penumbra, as observed in the Cerrobend block condition for the 45º angle, where the penumbra at the surface and dmax was approximately 1 cm broader compared to the IORT applicator. This difference was greater than the difference observed between the two setups at 0º, highlighting the impact of angled beam delivery on penumbra broadening. Second, when non-perpendicular beam incidence is required, the larger dimensions of the conventional applicators may pose a collision risk with the patient.
Beveled IORT applicators offer several advantages by facilitating angled electron beam delivery while mitigating the penumbra effect. This is particularly beneficial when treating areas proximal to radiosensitive tissues. Angling the beam away from critical regions can achieve targeted treatment while minimizing their exposure. For example, in cases where a cutaneous lesion is located near the ocular region, angling the beam away from the eye can significantly reduce the dose to the lens. However, this also shifts the maximum dose region towards the distal side of the beam, which increases the out-of-field dose by approximately 5% in that region. Therefore, during the treatment planning and setup, the margin on the longer side of the IORT applicator wall needs to be larger than on the shorter side to ensure adequate dose coverage.
An additional advantage of the beveled applicators is their suitability for treating linear scar lesions, such as keloids formed in an elongated shape. In such cases, the radiation treatment field often needs to be aligned along the surgical incision region. Utilizing a 0º IORT applicator for these treatments results in circular beam delivery, which potentially causes unnecessary radiation exposure to normal tissues. However, the beveled applicator can deliver the beam in an elliptical shape and reduce the impact on the surrounding normal tissues. By considering the beam characteristics of the beveled applicators, including a shorter beam penetration depth (dmax reduced by 4 mm and Rp by 2 mm at a 45º angle) and the lateral shift of the dmax (3–4 mm), more effective and tissue sparing treatments can be achieved.
Dosimetric evaluation of the treatment plan and clinical considerations
The results from both the water phantom measurements and Monte Carlo simulations demonstrate the potential advantages of utilizing IORT applicators for skin lesion treatment. These findings suggest that IORT applicators could be a valuable tool in clinical settings. To successfully implement this approach, some discussions and considerations are necessary.
Currently, simulations for determining the optimum patient positioning, beam orientation, applicator size, and bolus use are often performed directly on the treatment machines or using conventional 2D simulators. The standard treatment planning systems lack the capability to calculate the dose distributions under IORT applicator conditions, which makes it difficult to verify the expected dose distribution prior to treatment. To assess the potential benefits for patients, treatment plans were created in TOPAS using open-source CT data for the IORT applicator and Cerrobend block, which were then compared. The results were consistent with the observations of the water phantom experiments, which confirmed the characteristics of IORT applicator dose distribution for the patient treatment plan.
The primary advantage of the IORT applicator is its ability to reduce the radiation dose to the normal tissues surrounding the PTV. As demonstrated in the treatment plans for all three cases, the narrow penumbra generated by the IORT applicator significantly decreased the mean SNV dose by over 30%. The elevated maximum dose to the SNV was confined to a small region adjacent to the treatment area, which would impact the treatment efficacy only minimally. Therefore, utilizing the IORT applicator can be particularly effective in minimizing the side effects when sensitive organs are located near the treatment site, and it can provide a safer and more reassuring treatment option. They are especially beneficial in addressing the concerns of keloid patients, who often worry about radiation-induced secondary cancer when applying radiotherapy to treat a benign tumor.
It is important to note that achieving irregular field shaping with the applicator alone can be challenging for irregularly shaped PTVs. Therefore, the IORT applicator might not always reduce the SNV dose. In the cases examined in this study, the SNV dose was generally lower; however, in instances where the PTV shape is highly irregular or located in close proximity to the SNV, the IORT applicator may exhibit a higher SNV dose. In such cases, additional shielding using lead and bolus can protect the healthy tissue. For example, a 4 MeV electron beam can be completely shielded using a combination of 2 mm lead and 5 mm bolus.
Additionally, as one of the methods for irregular beam shaping, 3D printing techniques can be utilized. For more precise beam shaping, imaging is essential; therefore, performing 2D X-ray or CT scans, as mentioned earlier, would also be beneficial. By utilizing such imaging data, patient-specific modules can be created and attached to the applicator’s end, enabling more precise modulation of the beam range and customized beam shaping. Incorporating 3D printing technology into IORT applicators could preserve the dosimetric advantages of IORT applicators while enabling more effective treatment of irregularly shaped lesions. This will be further explored in our future endeavors to advance personalized radiotherapy solutions.
The IORT applicator demonstrated superior dose concentration near the skin surface, which makes it particularly suitable for the treatment of superficial lesions with thin PTVs. In the IORT plan evaluated in this study, a slight suboptimal dose homogeneity within the PTV was observed, which is likely attributed to the reduced dose delivery to deeper tissues inherent in the IORT treatment planning. This effect, manifested as an approximate 1 mm shift in dmax and R50 in the measurements, was corroborated in the treatment plans. In the IORT treatment plan, the maximum dose within the PTV was localized at a depth of 4–5 mm compared to 5–6 mm in the block treatment plan. Beyond 5 mm, the PTV dose in the IORT plan decreased, with both hot spots and low dose areas evident in the DVHs. Consequently, the IORT applicator may demonstrate enhanced efficacy in the treatment of sub-centimeter lesions.
The primary factor contributing to prolonged beam-on times was the reduced output factor observed in smaller fields and lower-energy beams. While the extended SSD in the IORT cone setup resulted in a relatively modest reduction in output factor, this is reflected in the results observed for 6 MeV electron beams with 51 mm field sizes, where a 15% output factor difference was noted. In contrast, the 4 MeV beam with a 25 mm field size exhibited a substantial 65% reduction in output factor, driven by the combined effects of shorter penetration ranges and increased scattering within the IORT applicator. The block condition showed slightly higher output factor values for the 4 MeV electron beam compared to the 6 MeV condition, primarily because side scatter equilibrium is more active at lower energies, allowing scattered electrons to contribute more effectively to the dose. However, in the IORT applicator condition, where electrons are sufficiently scattered through the applicator walls, side scatter equilibrium has a negligible impact on the dose. Instead, scattering and loss within the applicator play a more significant role, contributing to the reduced output factor observed in lower-energy conditions29. As a result, the utilization of a 25 mm IORT applicator with a 4 MeV electron beam resulted in approximately 3-fold increase in the treatment time compared to conventional methodologies using a Cerrobend block.
These findings emphasize the critical importance of carefully evaluating the lesion depth and beam-on time when choosing between intraoperative radiation therapy (IORT) and conventional electron beam therapies. The trade-off between the enhanced dose concentration at superficial depths and prolonged treatment times necessitates a case-by-case assessment to optimize the therapeutic outcomes.
In clinical practice, shorter beam-on times are generally preferred owing to several factors:
-
1.
Post-operative keloid treatment: Minimizing the exposure time of the excised area to the external environment is crucial to reduce the risk of infection and promote optimal wound healing.
-
2.
Patient comfort and compliance: Extended treatment period with the IORT applicator in direct contact with the patient may be challenging, particularly for individuals who have difficulty in maintaining a specific position for prolonged periods.
-
3.
Safety considerations: Longer beam-on times increase the potential of patient movement, which could compromise treatment accuracy and efficacy.
-
4.
Resource utilization: Shorter treatment times enable more efficient use of operating room resources and personnel.
Factors such as the specific clinical scenario, patient characteristics, and institutional resources should be considered when evaluating the suitability of the IORT applicators for individual cases. Moreover, further research should focus on optimizing the IORT techniques to reduce treatment times while maintaining the dosimetric benefits, potentially through the development of applicators.
Conclusion
This study presents the potential benefits of IORT applicators in electron beam therapy for skin lesions. These applicators reduce the penumbra, which minimizes the impact on surrounding normal tissues while delivering a uniform dose to the tumor. Furthermore, they facilitate dose concentration in the superficial region and enable diverse beam delivery angles, which aid in patient positioning during treatment. Although the current treatment planning systems do not support 3D dose distribution yet using IORT applicators, future development of such systems could enhance the treatment efficiency and broaden their clinical applicability.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Akita, S. et al. Combined surgical excision and radiation therapy for keloid treatment. J. Craniofac. Surg. 18, 1164–1169 (2007).
Speranza, G., Sultanem, K. & Muanza, T. Descriptive study of patients receiving excision and radiotherapy for keloids. Int. J. Radiation Oncology* Biology* Phys. 71, 1465–1469 (2008).
Renz, P. et al. Dose effect in adjuvant radiation therapy for the treatment of resected keloids. Int. J. Radiation Oncology* Biology* Phys. 102, 149–154 (2018).
De Sanctis, V. et al. Primary cutaneous lymphoma: local control and survival in patients treated with radiotherapy. Anticancer Res. 27, 601–605 (2007).
Senff, N. J. et al. European Organization for Research and Treatment of Cancer and International Society for Cutaneous Lymphoma consensus recommendations for the management of cutaneous B-cell lymphomas. Blood J. Am. Soc. Hematol. 112, 1600–1609 (2008).
Specht, L. et al. Modern radiation therapy for primary cutaneous lymphomas: field and dose guidelines from the International Lymphoma Radiation Oncology Group. Int. J. Radiation Oncology* Biology* Phys. 92, 32–39 (2015).
Willemze, R., Hodak, E., Zinzani, P., Specht, L. & Ladetto, M. Primary cutaneous lymphomas: ESMO Clinical Practice guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 29, iv30–iv40 (2018).
Locke, J., Karimpour, S., Young, G., Lockett, M. A. & Perez, C. A. Radiotherapy for epithelial skin cancer. Int. J. Radiation Oncology* Biology* Phys. 51, 748–755 (2001).
Pashazadeh, A., Boese, A. & Friebe, M. Radiation therapy techniques in the treatment of skin cancer: an overview of the current status and outlook. J. Dermatological Treat. (2019).
Gerbi, B. J. et al. Recommendations for clinical electron beam dosimetry: supplement to the recommendations of Task Group 25. Med. Phys. 36, 3239–3279 (2009).
Baghani, H. R., Heidarloo, N., Aghamiri, S. M. R. & Mahdavi, S. R. Comparing the physical and dosimetric characteristics of cylindrical and beam shaper intraoperative radiotherapy applicators. Radiat. Phys. Chem. 158, 22–36 (2019).
Guerra, P. et al. Feasibility assessment of the interactive use of a Monte Carlo algorithm in treatment planning for intraoperative electron radiation therapy. Phys. Med. Biol. 59, 7159 (2014).
Alhamada, H. et al. 3D Monte Carlo dosimetry of intraoperative electron radiation therapy (IOERT). Physica Med. 57, 207–214 (2019).
Tresoldi, M. M. et al. Immediate postoperative treatment of keloids with intraoperative radiation therapy technology: a pilot study. Plast. Reconstr. Surgery–Global Open. 9, e3738 (2021).
Prince, H. M. et al. Management of the primary cutaneous lymphomas. Australas. J. Dermatol. 44, 227–242 (2003).
Morris, S. Skin lymphoma. Clin. Oncol. 24, 371–385 (2012).
Cheraghi, N., Cognetta Jr, A. & Goldberg, D. Radiation therapy for the adjunctive treatment of surgically excised keloids: a review. J. Clin. Aesthetic Dermatol. 10, 12 (2017).
Agostinelli, S. et al. GEANT4—a simulation toolkit. Nuclear instruments and methods in physics research section A: accelerators. Spectrometers Detectors Assoc. Equip. 506, 250–303 (2003).
Perl, J., Shin, J., Schümann, J., Faddegon, B. & Paganetti, H. TOPAS: an innovative proton Monte Carlo platform for research and clinical applications. Med. Phys. 39, 6818–6837 (2012).
Kim, S. et al. Clinical implementation of a wide-field electron arc technique with a scatterer for widespread Kaposi’s sarcoma in the distal extremities. Sci. Rep. 10, 9693 (2020).
Clark, K. et al. The Cancer Imaging Archive (TCIA): maintaining and operating a public information repository. J. Digit. Imaging 26, 1045–1057 (2013).
Gahbauer, R. et al. Prescribing, recording, and reporting electron beam therapy: contents. J. ICRU 4, 5–9 (2004).
Miften, M. et al. Tolerance limits and methodologies for IMRT measurement-based verification QA: recommendations of AAPM Task Group 218. Med. Phys. 45, e53–e83 (2018).
Beddar, A. S. et al. Intraoperative radiation therapy using mobile electron linear accelerators: report of AAPM Radiation Therapy Committee Task Group 72. Med. Phys. 33, 1476–1489 (2006).
Björk, P., Knöös, T. & Nilsson, P. Comparative dosimetry of diode and diamond detectors in electron beams for intraoperative radiation therapy. Med. Phys. 27, 2580–2588 (2000).
Hogstrom, K. R. & Almond, P. R. Review of electron beam therapy physics. Phys. Med. Biol. 51, R455 (2006).
Lazarus, G. L., van Eeden, D. & du Plessis, F. C. Validation of Monte Carlo-based calculations for megavolt electron beams for IORT and FLASH-IORT. Heliyon 8 (2022).
O’Shea, T. P., Sawkey, D. L., Foley, M. J. & Faddegon, B. A. Monte Carlo commissioning of clinical electron beams using large field measurements. Phys. Med. Biol. 55, 4083 (2010).
Nevelsky, A., Bernstein, Z., Bar-Deroma, R., Kuten, A. & Orion, I. Design and dosimetry characteristics of a commercial applicator system for intra‐operative electron beam therapy utilizing ELEKTA Precise accelerator. J. Appl. Clin. Med. Phys. 11, 57–69 (2010).
Acknowledgements
This study was supported by a grant (2024IE0003) from the Asan Institute for Life Sciences, Asan Medical Center, Seoul, Korea.
Author information
Authors and Affiliations
Contributions
U. S. Lee, S.W. Kim, C.Y. Jeong, Y. M. Goh, M. J. Park, and B.C. Cho designed the study and performed the experiments. J.W. Kwak, S.Y. Song, and B.C. Cho reviewed and guided the study and experimental design. U. S. Lee wrote the overall manuscript. U. S. Lee, J. B. Shin, and S.W. Kim analyzed the results and constructed the Monte Carlo simulation. B.C. Cho edited the manuscript. All the authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lee, US., Kim, Sw., Shin, JB. et al. Intraoperative radiotherapy IORT applicators for treatment of small skin lesions a phantom and planning study. Sci Rep 15, 5499 (2025). https://doi.org/10.1038/s41598-025-89859-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-89859-4