Abstract
Obesity can mediate the occurrence of many cardiovascular metabolic diseases, such as coronary heart disease and diabetes, which will lead to a significant increase in all-cause mortality in obese people. Visceral fat refers to the excessive accumulation of adipose tissue in abdominal organs, which can lead to impaired normal function of organs. In recent years, it has been reported that probiotics can reduce obesity and visceral fat content, and some studies have put forward the opposite conclusion. In order to guide the practice of nutrition, we use Meta-analysis method to analyze these conclusions. The authors searched randomized controlled trials on the effects of oral probiotics on body weight and visceral fat content in obese people published before January 2024 in several public databases, such as PubMed, Web of Science, EMBASE, Cochrane Library and CNKI. The primary outcome was body weight, and the secondary outcomes included BMI, waist circumference, visceral fat content, and low-density lipoprotein levels. This trial has been registered in PROSPERO (CRD42024521353). A total of 8 randomized controlled trials involving 412 patients were included in this meta-analysis. Meta-analysis showed that the weight loss of the experimental group taking probiotics was significantly higher than that of the control group (P < 0.00001). The reduction of waist circumference in the experimental group taking probiotics was significantly different from that in the control group (P = 0.01). The decrease of visceral fat content in the experimental group taking probiotics was significantly different from that in the control group (P < 0.00001). There was no significant difference in BMI and LDL-C levels between the experimental group and the control group. Oral probiotics can reduce weight loss, waist circumference and visceral fat content in obese patients, which may be a potential nutritional treatment for obese patients in the future. However, whether oral probiotics can reduce BMI and LDL-C levels in obese patients needs to be included in more RCTs and further discussed in the future.
Similar content being viewed by others
Introduction
Probiotics have developed rapidly and become one of the most commonly used food supplements in the world since they were proposed in 19741. Probiotics are often used to improve the dynamic balance of internal microflora to maintain human intestinal health2,3. In addition, human gut microbes may play a role in the development of metabolic diseases such as obesity, diabetes and inflammatory bowel disease4. Current studies have shown that probiotics have potential health benefits, including preventing and treating diarrhea, improving intestinal diseases, reducing the risk of neonatal infections, reducing respiratory infections and cardiac metabolic problems, and significantly reducing total cholesterol and LDL-C in people with mild to moderate hypercholesterolemia, the exact effects of probiotics on these conditions have not yet been determined5. However, the exact effect of probiotics on these conditions has not yet been determined due to inconsistent research6. At present, most studies suggest that probiotics exert their benefits through a variety of mechanisms, such as immune regulation, regulation of microbiota and one of the most important mechanisms of probiotics in obesity and promoting weight loss is to reduce oxidative stress and inflammation7,8. However, these studies have limitations. For example, they cannot fully explain the complex interactions between microorganisms, and are difficult to replicate in vivo experiments. In particular, the colonization ability of probiotics in the gastrointestinal tract of mice is limited9,10. Besides, khavari-Daneshvar et al. reported that, unlike Bifidobacterium bifidum, the number of Lactobacillus acidophilus did not change in the intestinal tract of mice for a long time, indicating that the stability of Lactobacillus acidophilus in the intestinal flora of mice was higher than that of Bifidobacterium bifidum11.
Obesity makes its all-cause mortality significantly higher than other populations by increasing the risk of cardiovascular disease and metabolic disease12. The pathogenesis of obesity involves energy utilization, food intake and body‘s self-regulation and feedback, but also has a complex interaction mechanism with medical and health conditions, socio-economic conditions and potential genetic and environmental factors13. Gene expression is one of the important factors affecting obesity and excessive visceral fat. The study of Friedman JM et al.suggested that mouse obesity is caused by mutations in at least five identified genes, namely the OB gene, DB gene, Agouti Yellow gene, Tubby gene and Fat gene encoding leptin. These mutations lead to overeating, reduced energy expenditure, and extreme obesity in mice, accompanied by metabolic abnormalities such as hyperglycemia, hyperinsulinemia, decreased thyroid hormones, and reproductive dysfunction14.
The causes of visceral fat include dysfunctional subcutaneous adipose tissue expansion and ectopic storage of triglycerides. Age, gender and genetics are the factors affecting visceral adipose tissue accumulation15. In addition, peptide hormones also have important effects on obesity and visceral fat. The most critical hormone is leptin, which is a peptide hormone mainly secreted by adipose tissue and provides signals about fat storage to the brain16. In addition, the activation of the PI3K/AKT pathway and the downstream mTOR pathway are also involved in the control of leptin on appetite and weight loss17. Another peptide hormone called ' Ghrelin ' was originally predicted to exist according to bioinformatics methods. It was later discovered and artificially isolated. It is a hormone mainly produced by endocrine cells of the gastrointestinal tract. Ghrelin mainly activate cells in the anterior pituitary and hypothalamic arcuate nucleus, including neuropeptide Y neurons that initiate appetite, which may help prepare food intake by increasing gastric peristalsis and stimulating gastric acid secretion18,19. Gastrin is a peptide hormone that stimulates gastric parietal cells to secrete gastric acid ( HCl ) and promotes gastric motility. Injection of gastrin promotes food intake and gastric emptying in mice20. Long-term intake of gastrin may increase food intake and body weight. Therefore, gastrin and ghrelin may have the same biological effects.
Continuous weight loss to normal or slightly higher than normal is the main goal and challenge of non-nutritive treatment. Long-term, multi-modal and personalized comprehensive methods are needed to control obesity and visceral fat21. Anti-obesity drugs and bariatric surgery can help most patients continue to lose weight to normal levels. These drugs act on the central nervous system to inhibit hunger and appetite22. In the past five years, targeted therapy and new anti-obesity drugs for single-gene obesity have been developed in medicine, effectively reducing weight by more than 10% in 65% of subjects. Due to the short time to market of these drugs, the data on the long-term safety, efficacy and cardiovascular effects of these drugs are still unclear23. Nutritional therapy for obesity and excessive visceral fat mainly relies on lifestyle interventions. Through alternative interventions based on dietary patterns, food quality and mindfulness, it may be better to support long-term adherence to behavioral change24. Also, the latest research results suggest that intermittent fasting and meal replacement can be used as obesity treatment options25,26. What’ s more, some previous basic studies have put forward the opposite conclusion on whether probiotics can improve obesity and visceral fat3,6,10. The purpose of this study is to explore whether oral supplementation of probiotics can significantly improve the body weight and visceral fat level of obese patients.
Method
This Meta-analysis methodology is based on the systematic review and Meta-analysis Preferred Reporting Items (PRISMA)28. This work has been evaluated in accordance with the PRISMA and Assessment of Systematic Reviews Methodology Quality Guidelines (AMSTAR). The protocol of this meta-analysis has been registered in PROSPERO and approved for implementation (CRD42024521353).
Search strategy
The search strategy used the PICOS (Population, Intervention, Comparison, Results and Research Design) criteria recommended in the Cochrane Handbook for Systematic Reviews13. We searched the PubMed, Web of Science, EMBASE, Cochrane Library and CNKI databases, and the retrieval date was until January 2024. We use the same keywords to search in the above database. The search terms were set using the MeSH thesaurus developed by the NIH. The English search terms included ‘Fatness’, ‘Obesity’, ‘Probiotics’, 'Probiotic yogurt’, 'Adiposis’, 'Obese subjects‘, 'Overweight’ et al.
In order to search for as many RCTs as possible and improve the quality of retrieval, we did not set language restrictions for inclusion in the study. At the same time, in order to ensure the high quality of retrieval, we carried out artificial secondary screening. Two reviewers (Manhou Guo and Zhe-an Shen) trained in Meta-analysis independently conducted a second screening, and the selected literature was formally included. If there is ambiguity within the two reviewers, the decision of the two reviewers will be arbitrated by the third reviewer(Yongchun Wei).
Inclusion criteria
We used the following inclusion criteria: (1)The type of study was RCT; (2) Adults with oral intake of probiotics (age ≥ 18 years); (3) The control group used a placebo; (4) Clear indications for obesity intervention (patients with BMI > 28 or BMI > 24 but with metabolic syndrome); (5) The allocation of the experimental group and the control group used a strict blind method and randomization method; (6) The experimental group and the control group were comparable in basic data.
Exclusion criteria
We also have strict data reliability exclusion criteria: (1) Trials without ethical approval; (2) Animal and in vitro experiments; (3) Patients had organ failure; (4) People with metabolic diseases other than obesity-related diseases; (5) Cancer population; (6) Special diet is being used; (7) Research on potential conflicts of interest.
Data cleaning and extraction
According to the recommended requirements of the Cochrane guidelines, the data extraction process was completed independently by two independent reviewers. The data extraction included the first author‘s name, publication year, experimental design, number of patients in the experimental group and the control group, baseline data of patients, types of oral probiotics, number of patients with improved obesity, number of patients with improved visceral fat level, number of other complications, frequency and single number of probiotics.
The primary outcome measure was the number of patients with reduced obesity. Secondary outcome measures included visceral fat improvement, correlation between baseline data and improvement, correlation between probiotic species and treatment effect, correlation between frequency and single dose of probiotics and treatment effect, and incidence of other complications.
Bias assessment
We used the Cochran Risk Bias Evaluation Tool to evaluate the risk bias of the RCTs included. The evaluation points were as follows: random sequence generation (selection bias), allocation concealment (selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective reporting (reporting bias) and other biases not mentioned above. The authors carefully assessed the risks of various types of bias and choose one of the three options.
The assessment rules were as follows: high risk (the authors believe that the risk may or will affect the accuracy of subsequent data analysis), unclear risk (the authors were unable to objectively or correctly assess the risk of the bias for various reasons or the risk level of the bias was between high and low risk) and low risk (the authors believe that this bias does not affect the accuracy of subsequent data processing or is unlikely to affect it).
Statistical methods
Revman Manager V.5.3 was used for statistical analysis. The risk ratio (RR) of the binary classification results was calculated, and the confidence interval (CI) was set to 95%. In calculating the continuous variables, this study used the mean ± standard deviation measurement, and the CI was set to 95%. The Cochran Q statistic was used to evaluate the heterogeneity, where P ≤ 0.10, indicating that the heterogeneity was statistically significant. I2 statistic was used to evaluate the effect of heterogeneity. I2 = 0% indicates no heterogeneity, 25-49%, 50-74% and > 75% indicate low, medium and high heterogeneity, respectively. When I2 ≥ 50%, random effect model (RE) and Laird weight are used. When I2 < 50%, the fixed effect model was used for testing. P < 0.05 was considered statistically significant, and P < 0.01 was considered statistically significant.
Result
Study selection and characteristics
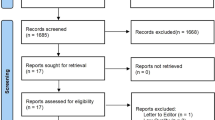
We searched databases and found a total of 381 data. After removing duplicates, 98 were retained, and 88 of them were removed after reviewing the title and abstract. Subsequently, we conducted a full-text review and raw data review of 10 articles, and eliminated 3 articles due to suspicious publication bias and possible experimental defects. Finally, we included 7 articles. Figure 1 shows our screening strategy and screening process results. Seven studies that met the inclusion and exclusion criteria were published between 2014 and 2020, with a total sample size of 412 people. All the included studies were RCTs, and the intervention strains included Bacillus subtilis, Bacillus coagulans, Lactobacillus curvatus, Lactobacillus plantarum, Lactobacillus casei, Lactobacillus rhamnosus, etc. Three studies did not give data on the gender of the enrolled subjects27,28,29. Two studies did not give data on the age of the enrolled subjects27,30. In another study, the subjects were all female36. Except for one study, the number of subjects included in other studies was more than 4031. Table 1 lists the details of the included studies and the general baseline data of the subjects.
Screening strategy, screening process and result flow chart.
Quality assessment
After bias risk assessment, two of the included studies were of high quality, and all bias risks were assessed as ‘low risk’28,29. One study did not clearly explain the blinding method used in the study. We believe that the risk of potential bias caused by blinding defects is high31. In the study of Sergeev et al., the number of subjects included was small, and there may be no universality in statistics31. One study was assessed as having a high risk of selective allocation bias because it did not provide allocation principles or allocation randomization for the experimental and control groups30. The specific bias assessment results are shown in Figs. 2, 3, and the methodological evaluation of the included RCTs is shown in Table 2.
Comprehensive table of risk of bias assessment of included studies.
List of risk of bias assessment checklists included in the study.
Primary outcome: body weight
We set body weight as the main outcome indicator of this study. 7 RCTs included in this study all provided body weight as an outcome indicator27,28,29,30,31,32,33. The heterogeneity of this outcome index was extremely low (I2 = 0%), so this outcome measure was tested using a fixed effect model. Compared with the control group, the weight loss of the experimental group taking probiotics was significantly higher (95%CI= -1.18 to -0.92, P < 0.00001) (Fig. 4). This shows that taking probiotics has a certain effect on weight loss.
Forest map showing the effect of taking probiotics on weight loss.
Secondary outcomes
Waist circumference
5 RCTs provided the secondary outcomes indicator of waist circumference changes before and after intervention28,29,31,32,33. All data are continuous variables, so Mean ± SD is used to express. The outcome index had high heterogeneity (I2 = 78%), so the outcome index was tested using RE, and sensitivity analysis was performed in part 3.5. Compared with the control group, there was a statistically significant difference in the waist circumference reduction of the experimental group taking probiotics (P = 0.01) (Supplementary Fig. 1). This shows that taking probiotics has certain significance for reducing waist circumference.
Visceral fat
4 RCTs provided the secondary outcomes indicator of changes in visceral fat content before and after intervention with probiotics27,30,31,33. The heterogeneity of the outcome index was low (I2 = 0%), so the fixed effect model was used for data analysis. Compared with the control group, there was a statistically significant difference in the reduction of visceral fat content in the experimental group taking probiotics (P < 0.00001) (Supplementary Fig. 2). This shows that taking probiotics has a certain significance in reducing visceral fat content.
Low density lipoprotein cholesterol content
3 RCTs provided the secondary outcomes indicator of changes in LDL-C levels before and after intervention with probiotics30,32,33. The heterogeneity of the outcome index was extremely high (I2 = 94%), so RE was selected to analyze the index. Compared with the control group, there was no significant statistical difference in the decrease of LDL-C level in the experimental group taking probiotics (P = 0.62) (Supplementary Fig. 3). This indicates that taking probiotics has no significant effect on reducing LDL-C Cholesterol Content.
BMI
6 RCTs included in this study provided pre-intervention and post-intervention BMI data as secondary outcome indicators27,28,29,31,32,33. The outcome index had high heterogeneity (I2 = 78%), so this outcome indicator was tested using RE, and sensitivity analysis was performed in part 3.5. Compared with the control group, there was no significant difference in BMI reduction in the experimental group taking probiotics (P = 0.56) (Supplementary Fig. 4). This shows that taking probiotics has no significant effect on reducing BMI in overweight people.
Sensitivity analysis of high heterogeneity results
Because of the high heterogeneity obtained in the statistical analysis of BMI and waist circumference data, we will conduct sensitivity analysis on the 2 secondary outcome indicators of BMI and waist circumference.
In the sensitivity analysis of BMI, we excluded one RCT32, and found that the heterogeneity decreased ( from I2 = 78% to I2 = 30% ), and the results changed (from P = 0.56 to P = 0.0002 ), which represented that there were too many confounding factors in some RCTs when analyzing BMI data. Then we analyzed the funnel plot of BMI value analysis. The more uniform the distribution of the funnel plot, the lower the risk of potential bias and unreliable results. The two-sided distribution uniformity in the funnel plot is poor, which indicates that there is a certain risk of potential bias (Fig. 5).
Funnel plot for assessing the potential bias of BMI values.
In the sensitivity analysis of the waist circumference, we excluded one RCT28, it was found that the heterogeneity decreased (from I2 = 78% to I2 = 0%), which means that there were too many confounding factors in some RCTs when analyzing the outcome index of waist circumference, but the results did not change significantly after excluding the RCT (from P = 0.01 to P = 0.02). Therefore, we believe that the high heterogeneity in this outcome index does not need special treatment, nor does it need to use funnel plot for bias analysis.
Discussion
It is reported that more than 1.9 billion adults worldwide are estimated to be overweight, of which more than 650 million adults are classified as obese. This data has risen by 2.5 times from 1975 to the present34. Obesity is usually an important risk factor for many chronic diseases, especially type 2 diabetes, cardiovascular disease, nonalcoholic fatty liver disease and a variety of malignant tumors35. As a new type of intestinal flora regulator in recent years, the effect of probiotics on weight loss is also often valued by researchers. At the same time, many nutritionists like to add probiotics to weight loss prescriptions6. This meta-analysis retrieved RCTs on the effect of oral supplementation of probiotics on weight loss and health improvement in overweight people in recent years, and concluded that oral supplementation of probiotics can effectively reduce weight, which means that oral probiotics may become an important part of nutritional therapy for overweight people in the future. However, oral probiotics do not have complete advantages, it also has certain defects. In this study, it was concluded that oral probiotics may have no significant effect on reducing BMI in overweight people. Although this outcome indicator showed high heterogeneity in the analysis, we analyzed this result in sensitivity analysis and believed that the outcome indicator still had potential bias. At the same time, some previously published meta-analyses have positive results on the issue that probiotics can improve BMI values36,37. However, the subjects of these meta-analyses usually include metabolic diseases other than obesity, such as type II diabetes. Therefore, their confounding factors need to be considered when referring to past reports.
In recent years, waist circumference has been shown to be used as an additional data of BMI to predict the incidence of cardiovascular risk events in obese people38. It is reported that the all-cause mortality of people with abdominal obesity is significantly higher than that of other types of obese people39,40, At the same time, people with abdominal obesity usually have some social and psychological problems due to changes in body appearance. This phenomenon is especially common in children41. The nutritional treatment that has been shown to be effective in reducing waist circumference is moderate-intensity exercise plus a low-salt, low-fat diet38. However, because obese people have low adherence to moderate-intensity exercise and low-salt, low-fat diets, it is necessary to develop a new therapeutic agent or method to reduce the waist circumference of this population. In this study, we found that oral probiotics have a significant statistical significance in reducing waist circumference, which brings an innovation in nutritional treatment for abdominal obese people. In the future, nutrition doctors can consider giving some probiotics to abdominal obese people on the basis of ensuring basic energy consumption, so as to help them improve treatment compliance and improve the success rate of reducing waist circumference.
Visceral fat is usually accompanied by metabolic abnormalities, including hypertriglyceridemia, elevated C-reactive protein, and hepatic insulin resistance42. Visceral fat and high LDL-C are inseparable, usually a positive correlation between the two15. Visceral fat and high LDL-C are usually controlled clinically mainly by means of drug therapy, supplemented by nutritional therapy. Intestinal flora has been shown to be closely related to biomarkers and inflammatory immune responses in vivo, and probiotics can regulate intestinal flora, thereby indirectly reducing visceral fat content and LDL-C43,44,45. This study also confirmed that oral probiotics can effectively reduce visceral fat content. However, the results of this study suggest that oral probiotics have no significant significance in reducing LDL-C. This outcome indicator is accompanied by high heterogeneity, but we did not perform sensitivity analysis on this outcome indicator, mainly because there are only 3 RCTs providing LDL-C data, and there is no indication of sensitivity analysis. Similarly, because the number of RCTs providing LDL-C data is small, the results are more likely to have potential bias.
Our study also found that probiotics had no significant effect on BMI, but there were significant differences in waist circumference and visceral fat compared with the control group, which is an interesting phenomenon. This may be due to the fact that probiotics preferentially regulate the optimization of visceral fat and subcutaneous fat through the gut-liver axis pathway, but for the general condition, the improvement of probiotics is not particularly advantageous. Shidfar and Shahraki’s studies have shown that waist circumference and visceral fat have an advantage in predicting the risk of cardiovascular disease in obese women compared with BMI46,47. At the same time, our study also found some interference of heterogeneity, which needs to be corrected by more high-quality RCTs in the future.
Similarly, there are some shortcomings in this study. For example, the analysis of inflammatory indicators of obesity was missing in this study, and there was no subgroup analysis of each probiotic, and the number of RCTs that met the inclusion and exclusion criteria was too small. In the process of weight loss, an important factor is dietary intake, but in our included 7 randomized controlled trials, are conducted under the conventional calorie diet test. At the same time, the heterogeneity of some secondary outcome indicators in this study was too high, and we explained this phenomenon in sensitivity analysis and discussion. The above problems need to be further improved by nutrition scholars in the future.
Conclusion
This study suggests that oral probiotics have certain statistical significance for weight loss, waist circumference and visceral fat content in obese patients, and can be used as a potential nutritional treatment for obese patients in the future. However, oral probiotics have no statistical significance in reducing BMI and LDL-C levels in obese patients, which may also be the outcome bias caused by high heterogeneity, and more RCTs need to be included in the future for further discussion.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Clarke, T. C., Black, L. I., Stussman, B. J., Barnes, P. M. & Nahin, R. L. Trends in the use of complementary health approaches among adults: United States, 2002–2012. Natl. Health Stat. Rep. 79, 1–16 (2015).
Sanders, M. E. et al. Health claims substantiation for probiotic and prebiotic products. Gut. Microbes 2(3), 127–133. https://doi.org/10.4161/gmic.2.3.16174 (2011).
Choi, S., Hwang, Y. J., Shin, M. J. & Yi, H. Difference in the gut microbiome between ovariectomy-induced obesity and diet-induced obesity. J. Microbiol. Biotechnol. 27(12), 2228–2236. https://doi.org/10.4014/jmb.1710.10001 (2017).
Qin, J. et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464(7285), 59–65. https://doi.org/10.1038/nature08821 (2010).
Pourrajab, B., Fatahi, S., Dehnad, A., Kord Varkaneh, H. & Shidfar, F. The impact of probiotic yogurt consumption on lipid profiles in subjects with mild to moderate hypercholesterolemia: A systematic review and meta-analysis of randomized controlled trials. Nutr. Metabol. Cardiovasc. Dis. 30(1), 11–22. https://doi.org/10.1016/j.numecd.2019.10.001 (2020).
Suez, J., Zmora, N., Segal, E. & Elinav, E. The pros, cons, and many unknowns of probiotics. Nat. Med. 25(5), 716–729. https://doi.org/10.1038/s41591-019-0439-x (2019).
Pourrajab, B., Fatahi, S., Sohouli, M. H., Găman, M. A. & Shidfar, F. The effects of probiotic/synbiotic supplementation compared to placebo on biomarkers of oxidative stress in adults: A systematic review and meta-analysis of randomized controlled trials. Crit. Rev. Food Sci. Nutr. 62(2), 490–507. https://doi.org/10.1080/10408398.2020.1821166 (2022).
Raji Lahiji, M. et al. Effects of synbiotic supplementation on serum adiponectin and inflammation status of overweight and obese breast cancer survivors: A randomized, triple-blind, placebo-controlled trial. Support Care Cancer 29(7), 4147–4157. https://doi.org/10.1007/s00520-020-05926-8 (2021).
Suez, J. et al. Post-antibiotic gut mucosal microbiome reconstitution is impaired by probiotics and improved by autologous FMT. Cell 174(6), 1406-1423.e16. https://doi.org/10.1016/j.cell.2018.08.047 (2018).
Zmora, N. et al. Personalized gut mucosal colonization resistance to empiric probiotics is associated with unique host and microbiome features. Cell 174(6), 1388-1405.e21. https://doi.org/10.1016/j.cell.2018.08.041 (2018).
Khavari-Daneshvar, H. et al. Modifications of mice gut microflora following oral consumption ofLactobacillus acidophilus and Bifidobacterium bifidum probiotics. Turk. J. Med. Sci. 47(2), 689–694. https://doi.org/10.3906/sag-1504-28 (2017).
Piché, M. E., Tchernof, A. & Després, J. P. Obesity phenotypes, diabetes, and cardiovascular diseases. Circ. Res. 126(11), 1477–1500. https://doi.org/10.1161/CIRCRESAHA.120.316101 (2020).
Lin, X. & Li, H. Obesity: Epidemiology, pathophysiology, and therapeutics. Front. Endocrinol. (Lausanne) 12, 706978. https://doi.org/10.3389/fendo.2021.706978 (2021).
Friedman, J. M. The alphabet of weight control. Nature 385(6612), 119–120. https://doi.org/10.1038/385119a0 (1997).
Tchernof, A. & Després, J. P. Pathophysiology of human visceral obesity: An update. Physiol. Rev. 93(1), 359–404. https://doi.org/10.1152/physrev.00033.2011 (2013).
Strosberg, A. D. & Issad, T. The involvement of leptin in humans revealed by mutations in leptin and leptin receptor genes. Trends Pharmacol. Sci. 20(6), 227–230. https://doi.org/10.1016/s0165-6147(99)01313-9 (1999).
Gurevich-Panigrahi, T., Panigrahi, S., Wiechec, E. & Los, M. Obesity: pathophysiology and clinical management. Curr. Med. Chem. 16(4), 506–521. https://doi.org/10.2174/092986709787315568 (2009).
Davies, J. S. Ghrelin mediated hippocampal neurogenesis. Vitam. Horm. 118, 337–367. https://doi.org/10.1016/bs.vh.2021.12.003 (2022).
Kulkarni, S. S., Singh, O. & Zigman, J. M. The intersection between ghrelin, metabolism and circadian rhythms. Nat. Rev. Endocrinol. 20(4), 228–238. https://doi.org/10.1038/s41574-023-00927-z (2024).
van der Lely, A. J., Tschöp, M., Heiman, M. L. & Ghigo, E. Biological, physiological, pathophysiological, and pharmacological aspects of ghrelin. Endocr. Rev. 25(3), 426–457. https://doi.org/10.1210/er.2002-0029 (2004).
Lauby-Secretan, B. et al. Body fatness and cancer-viewpoint of the IARC working group. N. Engl. J. Med. 375(8), 794–798. https://doi.org/10.1056/NEJMsr1606602 (2016).
Yumuk, V. et al. European guidelines for obesity management in adults. Obes. Facts. 8(6), 402–424. https://doi.org/10.1159/000442721 (2015).
Saeed, S., Arslan, M. & Froguel, P. Genetics of obesity in consanguineous populations: Toward precision medicine and the discovery of novel obesity genes. Obesity (Silver Spring) 26(3), 474–484. https://doi.org/10.1002/oby.22064 (2018).
Hassapidou, M. et al. European association for the study of obesity position statement on medical nutrition therapy for the management of overweight and obesity in adults developed in collaboration with the European federation of the associations of dietitians. Obes. Facts. 16(1), 11–28. https://doi.org/10.1159/000528083 (2023).
Cai, R. et al. Effect of community-based lifestyle interventions on weight loss and cardiometabolic risk factors in obese elderly in China: A randomized controlled trial. Exp. Gerontol. 128, 110749. https://doi.org/10.1016/j.exger.2019.110749 (2019).
Höchsmann, C. et al. Effects of a 2-year primary care lifestyle intervention on cardiometabolic risk factors: A cluster-randomized trial. Circulation 143(12), 1202–1214. https://doi.org/10.1161/CIRCULATIONAHA.120.051328 (2021).
Kim, M. et al. Effects of weight loss using supplementation with Lactobacillus strains on body fat and medium-chain acylcarnitines in overweight individuals. Food Funct. 8(1), 250–261. https://doi.org/10.1039/c6fo00993j (2017).
Nasiri, G. et al. Effects of probiotic and alpha-lipoic acid supplements, separately or in combination on the anthropometric indicators and maintenance of weight in overweight individuals. Clin. Nutr. ESPEN 41, 242–248. https://doi.org/10.1016/j.clnesp.2020.12.007 (2021).
Zarrati, M. et al. Effects of probiotic yogurt on fat distribution and gene expression of proinflammatory factors in peripheral blood mononuclear cells in overweight and obese people with or without weight-loss diet. J. Am. Coll. Nutr. 33(6), 417–425. https://doi.org/10.1080/07315724.2013.874937 (2014).
Sanchez, M. et al. Effect of Lactobacillus rhamnosus CGMCC1.3724 supplementation on weight loss and maintenance in obese men and women. Br. J. Nutr. 111(8), 1507–1519. https://doi.org/10.1017/S0007114513003875 (2014).
Sergeev, I. N., Aljutaily, T., Walton, G. & Huarte, E. Effects of synbiotic supplement on human gut microbiota, body composition and weight loss in obesity. Nutrients 12(1), 222. https://doi.org/10.3390/nu12010222 (2020).
Danielsson, P., Putri, R. R., Marcus, C. & Hagman, E. Evaluating probiotic efficacy on weight loss in adults with overweight through a double-blind, placebo-controlled randomized trial. Sci. Rep. 13(1), 18200. https://doi.org/10.1038/s41598-023-45395-7 (2023).
Razmpoosh, E., Zare, S., Fallahzadeh, H., Safi, S. & Nadjarzadeh, A. Effect of a low energy diet, containing a high protein, probiotic condensed yogurt, on biochemical and anthropometric measurements among women with overweight/obesity: A randomised controlled trial. Clin. Nutr. ESPEN 35, 194–200. https://doi.org/10.1016/j.clnesp.2019.10.001 (2020).
Larsson, S. C., Spyrou, N. & Mantzoros, C. S. Body fatness associations with cancer: Evidence from recent epidemiological studies and future directions. Metabolism 137, 155326. https://doi.org/10.1016/j.metabol.2022.155326 (2022).
Guh, D. P. et al. The incidence of co-morbidities related to obesity and overweight: A systematic review and meta-analysis. BMC Public Health 9, 88. https://doi.org/10.1186/1471-2458-9-88 (2009).
Tao, Y. W., Gu, Y. L., Mao, X. Q., Zhang, L. & Pei, Y. F. Effects of probiotics on type II diabetes mellitus: A meta-analysis. J. Transl. Med. 18(1), 30. https://doi.org/10.1186/s12967-020-02213-2 (2020).
Park, S. & Bae, J. H. Probiotics for weight loss: A systematic review and meta-analysis. Nutr. Res. 35(7), 566–575. https://doi.org/10.1016/j.nutres.2015.05.008 (2015).
Ross, R. et al. Waist circumference as a vital sign in clinical practice: A consensus statement from the IAS and ICCR working group on visceral obesity. Nat. Rev. Endocrinol. 16(3), 177–189. https://doi.org/10.1038/s41574-019-0310-7 (2020).
O’Donovan, G. et al. Changes in cardiorespiratory fitness and coronary heart disease risk factors following 24 wk of moderate- or high-intensity exercise of equal energy cost. J. Appl. Physiol. (1985) 98(5), 1619–1625. https://doi.org/10.1152/japplphysiol.01310.2004 (2005).
Church, T. S., Earnest, C. P., Skinner, J. S. & Blair, S. N. Effects of different doses of physical activity on cardiorespiratory fitness among sedentary, overweight or obese postmenopausal women with elevated blood pressure: A randomized controlled trial. JAMA 297(19), 2081–2091. https://doi.org/10.1001/jama.297.19.2081 (2007).
Aranha, L. N. & de Oliveira, G. M. M. Waist circumference: A simple measure for childhood obesity?. Arq. Bras. Cardiol. 114(3), 538–539. https://doi.org/10.36660/abc.20200031 (2020).
Jensen, M. D. Visceral fat: Culprit or canary?. Endocrinol. Metab. Clin. North Am. 49(2), 229–237. https://doi.org/10.1016/j.ecl.2020.02.002 (2020).
Zhou, B. et al. Intestinal flora and disease mutually shape the regional immune system in the intestinal tract. Front. Immunol. 11, 575. https://doi.org/10.3389/fimmu.2020.00575 (2020).
Ma, Q. et al. Research progress in the relationship between type 2 diabetes mellitus and intestinal flora. Biomed. Pharmacother. 117, 109138. https://doi.org/10.1016/j.biopha.2019.109138 (2019).
Jin, M., Qian, Z., Yin, J., Xu, W. & Zhou, X. The role of intestinal microbiota in cardiovascular disease. J. Cell Mol. Med. 23(4), 2343–2350. https://doi.org/10.1111/jcmm.14195 (2019).
Shidfar, F., Alborzi, F., Salehi, M. & Nojomi, M. Association of waist circumference, body mass index and conicity index with cardiovascular risk factors in postmenopausal women. Cardiovasc. J. Afr. 23(8), 442–445. https://doi.org/10.5830/CVJA-2012-038 (2012).
Shahraki, M., Shahraki, T., Shidfar, F. & Ansari, H. Which modifiable, non-modifiable, and socioeconomic factors have more effect on cardiovascular risk factors in overweight and obese women?. J. Res. Med. Sci. 17(7), 676–680 (2012).
Acknowledgements
The authors thank Prof. Longying Pei and Dr. Xiaoyan Zhang of Xinjiang Institute of Technology for their support for this manuscript.
Author information
Authors and Affiliations
Contributions
G Hou and ZA Shen conceived and designed this research. G Hou, J Li and L Zhang developed the search strategies, searched the databases, and conducted inspections based on eligibility and exclusion criteria. ZA Shen and C Wei extracted and analyzed the quantitative data. All authors contributed to writing, reviewing or revising this paper. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.




Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Guo, M., Li, J., Zhang, L. et al. Effects of oral supplementation of probiotics on body weight and visceral fat in obese patients: a meta-analysis and systematic review. Sci Rep 15, 6355 (2025). https://doi.org/10.1038/s41598-025-90820-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-90820-8
Keywords
This article is cited by
-
Gut allies against metabolic disorders: the therapeutic promise of probiotics
Folia Microbiologica (2025)