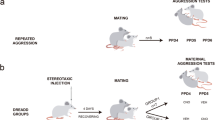
Abstract
During lactation, mammalian pups rely on the dam for survival and associative learning related to maternal stimuli. This study investigates how maternal preference shifts during the late lactation period in mouse pups. Pups conditioned with odorized dams during this period exhibited distinct preferences: those exposed to 4-methylthiazole (4MT) preferred it, while controls avoided it, and eugenol (EG)-conditioned pups showed no response. Interestingly, dams painted with 4MT displayed reduced maternal care compared to EG- or oil-painted dams. To separate maternal presence from odor experience, pups were exposed to 4MT or EG without the dam. Odor-preference learning occurred only when pups remained with their siblings but failed when separated from both the dam and all of their siblings. These findings suggest that dam separation in the presence of siblings facilitates odor-preference learning, indicating a developmental shift towards reduced dam reliance and preparation for independence.
Similar content being viewed by others
Introduction
In mammals, the transition from weaning to psychological independence represents a critical phase in developmental trajectories. This process is particularly prolonged and complex in humans, distinguishing it from patterns observed in many other animal species. These differences arise from a combination of biological, social, and cognitive factors that influence the pace and nature of development.
Human children experience an extended period of dependence following weaning, both emotionally and physically. In contrast, many non-human mammals undergo a rapid transition to independence after weaning, acquiring survival skills and dispersing from their parents relatively quickly. For instance, species such as ungulates and carnivores exhibit brief parental dependency followed by accelerated learning, enabling swift adaptation to environmental demand1,2.
In rodents, developmental transitions occur during lactation. During the early lactation period (postnatal days (P)0–9), pups exhibit an odor preference when paired with a neutral odor and electrical stimulation, facilitated by low stress sensitivity3,4. This associative learning changes in the mid-lactation period (P10–15), as stress sensitivity develops and disrupts odor preference learning unless the dam is present to reduce stress. By the late lactation period (P16–weaning), the dam’s stress-reducing effects diminish, and associative learning does not occur even in her presence. Although the physical transition from maternal milk to solid food begins at P17 and concludes around P285, the timing of psychological independence remains unclear. The early survival capacity of rodents complicates observation under standard captive conditions.
During this developmental period, sensory systems mature through both genetic programming and neuronal activity6,7,8,9,10,11. In the mouse olfactory system, odor-evoked neuronal activity during the neonatal critical period drives olfactory imprinting, which increases sensitivity to conditioned odors. Sema7A in olfactory sensory neurons interacts with PlxnC1 in mitral/tufted (M/T) cell dendrites, facilitating synapse formation and glomerular growth12. This odor imprinting requires the dam’s presence as a reward and oxytocin in pups for attractive odor memory and social bonding13. However, while neonatal imprinting mechanisms are well understood, little is known about odor-preference learning and the signaling systems involved during the late lactation period.
This study focuses on odor-preference learning during the late lactation period. Using specific odorants, 4-methylthiazole (4MT, an aversive odorant) and eugenol (EG, a neutral odorant), we investigated the developmental shift in odor-associated learning and its relationship to changes in maternal and sibling interactions. Our findings reveal a shift in the dam’s value from positive to negative in the presence of siblings. This raises the possibility that psychological weaning begins before the end of the physical weaning period, facilitating the transition to independence and the acquisition of social behaviors.
Results
Odor-preference shift by 4MT-nip conditioning during the lactation period
In previous studies, rat pups nursed by dams whose bellies and nipples were odorized with aversive odorants, such as peppermint or isoamyl acetate, exhibited an approach behavior towards the odors14,15. To investigate whether a similar odor preference could be conditioned in mouse pups, we odorized dams’ nipples with either sunflower seed oil (oil-nip conditioning group, serving as a control) or oil-diluted 4MT (4MT-nip conditioning group) twice daily from P12 to P18. This conditioning was performed outside the olfactory imprinting period12. We subsequently conducted an odor preference test on male pups at P19. During the preference test, filter paper pieces containing either oil or 4MT were placed on each short side of a test cage, and we calculated the preference index (PI) based on the average relative position of the pups within the test cage. Notably, oil-nip-conditioned pups exhibited avoidance behavior towards 4MT (Fig. 1A). In contrast, 4MT-nip-conditioned pups displayed clear approach behavior for 4MT under the same conditions. The female siblings cohabitating with them also displayed approach behavior for 4MT (Supplementary Fig. S1A). To ensure that oil-nip-conditioned pups did not form a preference for the oil itself, we conducted a preference test using filter paper pieces soaked in oil or nothing. The results indicated that oil-nip-conditioned pups exhibited no preference for oil (Fig. 1A), confirming they did not develop a preference for the oil. These findings suggest that 4MT-nip conditioning effectively switched the pups’ behavior from avoidance to approach, highlighting the conditioning’s impact on odor preference.
Odor preference acquisition in pups by nipple odorization of dams. (A) Locomotion of pups conditioned by oil-nip or 4MT-nip during P12-18 was traced in odor preference test at P19. Oil-nip- (n = 30) and 4MT-nip-conditioned pups (n = 23) displayed significant avoidance and approach for 4MT, respectively. Oil-nip-conditioned pups (n = 13) displayed no significant bias for oil. (B) The temporal observation of the preference index for 4MT during P15-29 in pups conditioned by oil-nip (n = 6) or 4MT-nip (n = 6) during P12-28 displayed a gradual loss of acquired 4MT preference. (C) The effects of commencing time between regular (P12-18) and late-onset 4MT-nip conditioning (P14-21) were analyzed. Although the regularly conditioned pups showed significant preference at P17 (n = 15) and P19 (n = 8), the late-onset-conditioned pups showed no preference at P19, 21 (n = 13). Welch’s unpaired t-test (A–C): **, p < 0.01; ****, p < 0.0001. One sample t-test (OST), theoretical mean = 0 (A–C): $, p < 0.05; $$, p < 0.01; $$$, p < 0.001; $$$$, p < 0.0001.
Time limit of acquisition and maintenance of odor-preference memory
As mouse pups age, they exhibit a decrease in nipple suckling behavior and an increase in pellet-eating behavior16. This transition suggests that physical weaning may lead to the termination of an acquired odor preference. To explore whether an acquired odor preference can be maintained when the odorization period extends beyond P21, we continued 4MT nipple odorization until P28 and conducted odor preference tests for 4MT every other day from P15 to P29. Notably, while the oil-nip conditioning group continued to show avoidance behavior after P17, the 4MT-nip conditioning group demonstrated a gradual decline in the preference index for 4MT, with a significant shift towards avoidance observed at P29 (Fig. 1B). These findings suggest that an acquired 4MT preference gradually diminishes and transforms into avoidance as lactation becomes less essential, indicating a correlation between lactation period and odor-preference memory retention.
To determine whether the timing of 4MT-nip conditioning initiation is critical, we delayed the start of conditioning. When conditioning began at P12 (conditioning day 1, CD1), the conditioned pups displayed a preference for 4MT at P17 (CD6) and P19 (CD8) (Fig. 1C). In contrast, when conditioning commenced at P14 (CD1), the conditioned pups no longer exhibited a preference at P19 (CD6) and P21 (CD8). These findings suggest that initiating 4MT-nip conditioning at P14 falls outside the critical period for 4MT-preference acquisition.
Suppression of stress responses and stress-related brain regions by 4MT-nip conditioning
Previous research has shown that aversive odorants, including 4MT, can induce stress responses17. To investigate the impact of 4MT exposure on stress response, we analyzed plasma corticosterone concentration. The results revealed a significant increase in corticosterone concentration in 4MT-exposed oil-nip-conditioned pups compared to oil-exposed oil-nip-conditioned pups (Fig. 2A). Next, we assessed whether 4MT conditioning during lactation could mitigate the stress response to 4MT. Our findings showed no significant difference in corticosterone concentration between oil-exposed 4MT-nip-conditioned and 4MT-exposed 4MT-nip-conditioned pups, indicating that 4MT-nip conditioning during the lactation period effectively suppressed the stress response to 4MT.
Suppression of stress response and neuronal firing of stress-related brain regions in 4MT-nip-conditioned pups for 4MT exposure. (A) The amount of plasma corticosterone was compared between oil- (n = 9) or 4MT-exposure groups (n = 9) in oil-nip conditioned pups and between oil- (n = 7) or 4MT-exposure groups (n = 7) in 4MT-nip conditioned pups. In the amount of plasma corticosterone, oil-nip-conditioned pups showed a significant increase in association with 4MT exposure, but 4MT-nip-conditioned pups showed no increase. Welch’s unpaired t-test: **, p < 0.01. (B) Expression of c-fos gene was analyzed by in situ hybridization in oil-exposed pups including oil-nip (n = 4) and 4MT-nip conditioned pups (n = 2), 4MT-exposed oil-conditioned pups (n = 6), and 4MT-exposed 4MT-conditioned pups (n = 6) in brain regions: the paraventricular nucleus of the hypothalamus (PVN), the oval bed nucleus of the stria terminalis (BNSTov), capsular part, lateral division, and medial division of the central amygdaloid nucleus (CeC, CeL, CeM). In regions of interest, c-fos( +) cells were counted. Scale bar, 250 µm. Dunn’s multiple comparison tests: **, p < 0.01; ***, p < 0.001.
Under stressful condition, corticotropin-releasing factor (CRF)-producing neurons in the paraventricular hypothalamic nucleus (PVN) secrete CRF. This CRF induces adrenocorticotropic hormone (ACTH) secretion in the pituitary gland, which in turn triggers corticosterone release from the adrenal cortex18. Corticosterone secretion was suppressed in 4MT-nip conditioned pup mice (Fig. 2A). Therefore, we examined neuronal firing in PVN by c-fos gene expression with in situ hybridization. The number of c-fos( +) cells was significantly increased in the 4MT-exposed oil-nip group, compared with that in oil-exposed oil-nip group (Fig. 2B). In contrast, the number of c-fos( +) cells in 4MT-exposed 4MT-nip group was significantly decreased, compared with that in 4MT-exposed oil-nip group. These were consistent with the result of the corticosterone assay.
Activation of PVN neurons is regulated by many brain regions including the bed nucleus of the stria terminalis (BNST) and central amygdala (CeA)18,19. These regions were activated by 4MT exposure in oil-conditioned mice (Fig. 2B). However, activation of these regions was suppressed in 4MT-conditioned pup mice. These results suggest that 4MT conditioning suppresses the activation of the brain regions for stress and pain.
Irrelevance of oxytocin in odor-preference acquisition
Oxytocin, a neuropeptide, plays a significant role in dam-pup bonding20. Previous studies have demonstrated that intracerebral administration of an oxytocin antagonist, atosiban, to rat pups at P15 inhibits the acquisition of a maternally associated odor preference21. Therefore, we investigated whether oxytocin receptor (OTR) is required for 4MT-preference acquisition by conducting 4MT-nip conditioning on OTR knock-out (KO) pups. Surprisingly, OTR homozygous KO pups exhibited a normal 4MT preference (Fig. 3A), suggesting that OTR is not essential for the acquisition of the 4MT preference.
Requirements for 4MT-preference acquisition. (A) Preference index for 4MT was analyzed in oil-nip-conditioned (n = 8) and 4MT-nip-conditioned pups (n = 7) in oxytocin receptor (OTR) heterozygous knockout (left), and in oil-nip- (n = 6) and 4MT-nip-conditioned pups (n = 9) in OTR homozygous knockout (right). OTR homozygous knockout mice showed normal 4MT-preference acquisition. (B) Frequencies of dams’ lactation for three or more pups (left) and immobility without lactation (right) during 1–30 min (upper) and 31–60 min (lower) after oil (n = 21), 4MT (n = 21), or EG-nip painting (n = 18) during P13-15 were counted. During 1–30 min dams painted with 4MT displayed a significant decrease in the frequency of lactation and a significant increase in immobility. (C) Preference index for 4MT in oil-dam-unpaired (Oil-DUP, n = 19), 4MT-dam-unpaired (4MT-DUP, n = 32), 4MT-dam-paired (4MT-DP, n = 17), and 4MT-dam/sibling-unpaired conditionings (4MT-DSUP, n = 16). Pups in 4MT-DUP and 4MT-DP conditionings showed a significant preference for 4MT while those in 4MT-DSUP conditioning showed no preference. (D) Preference index for 4MT at P11–18 in oil-DUP- and 4MT-DUP-conditioned pups conditioned during P12-18. Pups in 4MT-DUP conditioning displayed significant aversion at P13 and significant preference at P17 and P19, while those in oil-DUP conditioning displayed significant aversion at P17 and P19. Welch’s unpaired t-test (A, D), Dunn’s multiple comparison test (B), Tukey’s multiple comparison test (C): *, p < 0.05; **, p < 0.01; ***, p < 0.001; ****, p < 0.0001. OST (A, C, D): $, p < 0.05; $$, p < 0.01; $$$, p < 0.001.
Loss of maternal care in 4MT-nip conditioning
The above result in OTR KO pups raised the possibility that dams are not involved in 4MT-preference acquisition. Therefore, we examined dams’ behavior during 4MT-nip conditioning. During the first 30 min after the onset of 4MT-nip conditioning, the frequency of breastfeeding in 4MT-nip conditioning was reduced, and the frequency of immobility without breastfeeding was increased, compared with that in oil-nip conditionings (Fig. 3B). During the second 30 min, there was no significant difference in the frequency of those behaviors in Kruskal–Wallis tests. These results indicate that 4MT-nip conditioning reduces maternal care, raising the possibility that maternal care is not necessary for pups’ 4MT-preference acquisition.
Acquisition of 4MT preference by maternal separation
To explore the necessity of maternal presence during 4MT exposure, we isolated a group of sibling pups from their dams and exposed them to 4MT during a 30-min isolation period, following the same schedule as 4MT-nip conditioning. This method, named 4MT-dam-unpaired (DUP) conditioning, was compared to oil-DUP conditioning, where 4MT in 4MT-DUP conditioning was replaced with oil as a negative control. Additionally, we included a 4MT-dam-paired (DP)-conditioned group, where pups were exposed to 4MT with their dam after a 30-min isolation period. The results revealed that oil-DUP-conditioned and 4MT-DP-conditioned pups exhibited avoidance and preference for 4MT, respectively (Fig. 3C). The female siblings cohabitating with them also exhibited similar behavioral changes (Supplementary Fig. S1B). Interestingly, 4MT-DUP-conditioned pups also displayed a preference for 4MT. This outcome suggests that the cohabitation of dams during 4MT exposure is not a prerequisite for 4MT-preference acquisition and that 4MT-DUP conditioning is an effective substitute for 4MT-nip conditioning.
To assess the temporal change of 4MT preference in 4MT-DUP conditioning, we conducted DUP conditioning on the same schedule as 4MT-nip conditioning (P12 to P18) and assessed the temporal change of 4MT preference from P11 to P19. The findings revealed that oil-DUP-conditioned pups displayed no behavioral response for 4MT at P13 and P15 but began to exhibit avoidance at P17 (Fig. 3D). In contrast, 4MT-DUP-conditioned pups displayed significant 4MT avoidance at P13, which shifted to significant 4MT preference at P17 and P19. These results indicate that the 4MT avoidance observed at P13 in 4MT-DUP conditioning is not an innate instinct but rather acquired through isolation from their dams. Furthermore, the 4MT avoidance observed at P13 diminishes by P15, transitioning into 4MT preference at P17 and P19. This shift suggests a noteworthy change in valence associated with dam isolation, evolving from negative to positive over the course of these early postnatal days.
Odor-preference learning induced by maternal separation in the presence of siblings
Next, we used eugenol (EG) as a neutral odorant for nipple odorization conditioning. EG-nip-conditioned pups showed no behavioral response for EG as the oil-nip conditioning group did (Fig. 4A). This result indicates that nipple odorization conditioning with EG is insufficient for odor preference acquisition.
An odor preference is formed by odor experience with siblings during maternal separation. (A) Locomotion of oil-nip-conditioned (n = 13) and EG-nip-conditioned pups (n = 15) conditioned during P12-19 was traced in an odor preference test for EG at P19 (left). EG-nip-conditioned pups showed no odor preference (right). Welch’s unpaired t-test. (B) Preference index for EG in oil-dam-unpaired (Oil-DUP, n = 12), EG-dam-unpaired (EG-DUP, n = 14), EG-dam-paired conditionings (EG-DP, n = 8), EG-simple exposure-conditioned (EG-SE, n = 13) and EG-dam/sibling-unpairing-conditioned pups (4EG-DSUP, n = 14). Pups in EG-DUP and EG-DP conditionings displayed significant preference while those in EG-SE and EG-DSUP conditionings displayed no significant preference. Tukey’s multiple comparison test: **, p < 0.01; ***, p < 0.001. OST (A, B): $, p < 0.05; $$$$, p < 0.0001.
An odor preference was observed in 4MT-nip conditioning, but not in EG-nip conditioning. Maternal care may be different between 4MT-nip and EG-nip conditioning. Therefore, we examined maternal behavior in EG-nip conditioning. During 30 min after the onset of EUG-nip conditioning, the frequencies of breastfeeding and immobility without breastfeeding were normal (Fig. 3B). To examine whether maternal separation affects odor learning, we performed EG-DUP and EG-DP conditioning. Interestingly, pups in both conditioning groups showed a preference for EG (Fig. 4B). In EG-simple-exposure (SE) conditioning, pups exposed to EG for 30 min in their home cage with a dam displayed neutral behavior for EG. These results indicate that maternal separation is effective for odor-preference learning.
In EG-DUP conditioning, pups were exposed to EG in a sibling group and were able to interact with each other by huddling. Maternal deprivation, in which both a dam and all of their siblings are deprived, is more stressful for pups than maternal separation, in which only a dam is separated and their littermates are cohabitated22. Siblings may assist with EG-preference acquisition. We performed odor conditioning in solitude to know whether other siblings are required for EG-preference acquisition. Solitude conditioning inhibited EG-preference acquisition significantly (Fig. 4B). Solitude conditioning with 4MT also inhibited 4MT-preference acquisition (Fig. 3C). These results indicate that the presence of siblings is essential for odor-preference acquisition during maternal separation.
Discussion
In the present study, we demonstrated that 4MT-nip conditioning during P12-18 altered pups’ behavior towards 4MT, shifting from avoidance to approach by P19 (Fig. 1A). Previous studies have shown that certain mildly aversive odorants (e.g., peppermint, isoamyl acetate) effectively induce odor-preference acquisition during lactation, whereas strongly aversive odorants (e.g., 2,4,5-trimethylthiazoline, TMT) are not effective14,15,23. Consistent with these findings, we found that 4MT, which is less aversive than TMT24, effectively induced odor-preference acquisition. In 4MT-nip-conditioned pups, stress hormone release and neuronal activity in key stress-related regions, including the paraventricular nucleus (PVN), bed nucleus of the stria terminalis (BNST), and central amygdala (CeA), were suppressed during 4MT exposure (Fig. 2A,B). The PVN, a central hub of the stress response, receives input from the CeA and BNST19. The CeA is implicated in both innate and learned fear behaviors25,26, while BNST activity correlates with increased anxiety27,28, and its inhibition produces anxiolytic effects29. These results suggest that 4MT-nip conditioning effectively reduces fear and anxiety responses associated with 4MT exposure.
While oxytocin is known to play a crucial role in dam-pup bonding20, our results indicate that the knockout of the oxytocin receptor did not affect 4MT-preference acquisition (Fig. 3A). This suggests that the dam-pup bond may not be directly involved in odor-preference acquisition. Arginine vasopressin (AVP), a neuropeptide involved in pair-bonding behavior, may be related to the odor-preference acquisition. Behavioral analysis of dams during 4MT-nip conditioning revealed maternal neglect (Fig. 3B), and further experiments showed that exposure to 4MT during maternal separation (4MT-DUP conditioning), which mimics maternal neglect, was sufficient to induce odor-preference learning (Fig. 3C). Interestingly, while EG-nip conditioning failed to induce odor-preference acquisition, EG-DUP conditioning was effective (Fig. 4A,B), suggesting that maternal separation during late lactation has a positive valence. In addition, maternal separation may increase attentional levels, thereby enhancing memory formation.
Extended 4MT-nip conditioning revealed that the acquired 4MT preference gradually diminished between P17-19 and was completely lost by P29 (Fig. 1B). This decline may correspond to the end of the physical weaning period, which is completed by P27 in mice16. Furthermore, late-start 4MT-nip conditioning during P14 to P20 was ineffective for odor-preference acquisition (Fig. 1C), highlighting a sensitive period for this learning. Intriguingly, 4MT-DUP conditioning induced odor aversion at P13 but resulted in odor preference by P17 (Fig. 3D). This finding aligns with prior work showing that early postnatal rat pups (P0-9) form positive odor memories despite aversive stimuli, due to the stress-hyporesponsive nature of this period3,30. During the mid-lactation period (P10-15), maternal presence buffers pups’ stress responses, enabling positive odor-memory formation despite mature stress systems. By late lactation (P16 onwards), this maternal buffering effect diminishes. Our findings extend this model, showing that maternal separation is initially aversive (P13) but becomes positively valued by P17, marking the emergence of psychological independence.
The onset of psychological independence in mice may be associated with behaviors observed during P17-19, such as increased out-of-nest exploration and a preference for novel environments31,32,33,34,35. This transition likely facilitates the development of sociality and independence. Importantly, early weaning before P16, when psychological independence has not yet emerged, has been linked to adverse psychological outcomes in adulthood36,37. Our study raises the possibility that the psychological independence period begins by P17, emphasizing the importance of aligning weaning timing with the onset of psychological independence to mitigate potential negative effects.
Recent findings indicate that somatostatin-positive neurons in the zona incerta integrate olfactory and somatosensory inputs to establish maternal bonding in pups38. These neurons lose their function in adulthood as their projections to other brain regions retract after weaning38,39,40. This developmental shift may underlie the change in the valence of maternal separation, transforming it into a positive experience during late lactation.
Our study showed that 4MT-nip conditioning induces both approach behavior and reduced stress responses toward 4MT. However, the causal relationship between these changes remains unclear. Additionally, EUG-DUP conditioning also induces approach behavior. The specific brain regions responsible for approach behavior have not yet been identified. Once identified, causal relationships could be clarified through loss-of-function and gain-of-function experiments. Figure 1C shows that P14-start conditioning does not result in preference acquisition, whereas P12-start conditioning does. Furthermore, Fig. 3D indicates that oil-DUP conditioned pups at P11, P13, and P15 do not exhibit avoidance behavior toward 4MT. Based on these results, we speculate that 4MT is not inherently stressful for pups conditioned at either P12 or P14. Therefore, the differences in conditioning effects between P12 and P14 are likely not due to variations in stress levels. These developmental differences could be better understood by identifying and developmentally analyzing the specific brain regions responsible for approach behavior.
Finally, we found that odor-DSUP conditioning (odor exposure during separation from both the dam and all of their sibling) inhibited odor-preference acquisition, while odor-DUP conditioning (separation from only the dam) facilitated it (Figs. 3C, 4B). This suggests that siblings modulate the effects of maternal separation. Separation from both the dam and siblings likely induces a more stressful state, potentially mediated by stress hormones, which may suppress learning. Sibling interactions during maternal separation may play a crucial role in shaping the development of social behaviors after weaning.
In humans, psychological independence typically follows physical weaning. However, this period has not been analyzed in other animals. Our findings suggest that psychological independence begins to emerge during late lactation in mice, providing potential insights into the mechanisms underlying psychological development during human adolescence.
Methods
Mice
We obtained C57BL/6N mice from Japan SLC Inc., and oxytocin receptor knockout (OTR-KO) mice41 from Dr. Katsuhiko Nishimori (Fukushima Medical University). To obtain pups for odor conditioning experiments, female mice (6-week-old or older) were mated with male mice in the evening, and male mice were separated from female mice next morning after confirming the plugs. Mice were raised in the 7:00–19:00 light and 19:00–7:00 dark cycles. While male pups were analyzed except for the Supplemental Fig. S1, female pups were not excluded from rearing. To euthanize mice, they were deeply anesthetized with isoflurane. All the animal experiments were approved by the Animal Experiment Committee at University of Fukui. All methods were performed in accordance with the Guide for the Care and Use of Laboratory Animals of the Science Council of Japan and the relevant guidelines and regulations. The study is reported in accordance with ARRIVE guidelines (https://arriveguidelines.org).
Odor conditioning
Odor conditioning was performed twice a day (8:00–9:00 and 17:00–18:00), using 200µL of 100% sunflower seed oil (no odor control, Sigma), 10% (v/v) 4-methylthiazole (4MT, Tokyo Chemical Industry)/oil, or 10% (v/v) eugenol (EG, Tokyo Chemical Industry)/oil. In the odor-nip conditioning, all dam nipples were spotted with 200µL of oil containing 10% (v/v) odorant, and then dams were put back to the home cage with pups. In the conditioning experiment, pieces of odor-spotted 3 × 4 cm filter paper (3MM Chr Whatman) with 50 µl of 10%(v/v) odorant/oil or oil alone were used. In the odor-dam-unpaired (DUP) conditioning, all pups were separated from their dams and exposed to an odorant in a clean cage with their siblings for 30 min. They were then returned to the home cage. In the odor-dam-paired (DP) conditioning, pups were separated from their dams and moved to a clean cage with their siblings for 30 min. The pups were exposed to an odorant in their home cage with their dams for 30 min, and then the odorant was removed. In the dam-sibling-unpaired (DSUP) odor conditioning, the pups were separated from their dams and exposed to the odor-containing filter paper in solitude in a clean cage separated into four areas for 30 min, and then they were returned to the home cage. In the EG simple-exposure (SE) conditioning, the pups were exposed to EG in their home cage with the dam for 30 min.
Behavioral assay
Odor preference test: All behavior tests were conducted during 8:00–12:00. In experiments observing the temporal change of odor preference (Figs. 1B, 3D), odor preference tests were performed during 8:00–11:00 before morning odor conditioning (11:00–12:00). In the odor preference test, autoclaved clean test cages (17 × 27 × 12.5 cm, CLEA), surrounded by transparent acrylic walls (30 cm height), were used. Four test cages were arranged under a digital video camera GZ-E700-A (JVC), and the locomotion of a maximum of 4 mice was simultaneously recorded from the top. Before the test, a pup was habituated in the test cages for 3 min, and two pieces of filter paper (3 × 4 cm) soaking 50 µl of oil or 10%(v/v) odorant/oil were put on both sides of the cage for 3 min. In the tests, the odorants or the conditioned mice were arranged so as not to be biased to one side. In the recorded movie, the pup body was extracted by binarization, and the centroid of the body was traced with Fiji Plugin Mtrack242. From the centroid data, horizontal movement distance was measured, and pups whose movement distance was less than 400 cm in the odor preference tests for 3 min were excluded from the analyses except for the Fig. 3D experiment. The relative position in the test cage was defined as follows: the edge of the odorant side, the center, and the edge of the oil side indicate + 50, 0, and -50, respectively. The centers of round bumps in four corners of the test cage bottom were defined as cage edges. The preference index was calculated from the average of the relative position in the test cage for 3 min. Dam behavior: In odor-nip conditioning, a home cage including a dam and her pups was surrounded by the transparent acrylic wall, and the dam behavior was recorded from the top with the digital video camera, and examined every minute during 1–30 and 30–60 min just after nipple odorization when conditioned pups were at P13-15. The frequency of lactation behavior was counted when a dam breastfed 3 pups or more. The frequency of immobility without lactation was counted when a dam showed immobility without breastfeeding.
Plasma concentrations of corticosterone
Oil- and 4MT-nip conditioning was initiated at P12 and terminated at P18, as described above. At P19, the conditioned pups were exposed to 50µL of 10% 4MT/oil solution (or 100% oil as a control) applied to a piece of filter paper. Thirty minutes after exposure, the 4MT-conditioned (or oil-conditioned) mice were decapitated, and trunk blood was immediately collected into tubes containing 10µL of 0.05 M EDTA, pH 8.0. The tubes were centrifuged at 1,600 × g at 4℃ for 15 min, and the plasma corticosterone concentration of the supernatant was measured by Corticosterone Enzyme Immunoassay Kit (Arbor assays) and SpectraMax M5 Microplate Reader (Molecular Devices).
In situ hybridization
Pups were isolated from their dam and put in a clean cage for at least 4 h. A piece of filter paper immersed with 50µL of oil or 10%(v/v) 4MT/oil was put on the cage lid. After 30 min, the pups were deeply anesthetized with isoflurane, and their brains were taken out, and frozen in O.C.T compound (Sakura Finetek). Brain Sects. (10 µm thick) were prepared with a cryostat (Leica CM3050 S), and thaw-mounted on MAS-coated glass slides (Matsunami). In the sections, c-fos expression was detected with a c-fos antisense probe by in situ hybridization. The probe was synthesized from the pGEM-T vector (Promega) including the c-fos gene (583-1207 bp, NM_010234.3). In situ hybridization was performed as described previously43. After in situ hybridization, the sections were counterstained with methyl green (H-3402, VECTOR), and mounted with Mount-Quick Mounting Media (Newcomer Supply) and coverslips (Matsunami). The brain sections were photographed with a digital microscope (Keyence, BZ-9000). Brain regions were identified with the mouse brain atlas44. In the bilateral brain regions of interest, c-fos positive cells were counted and divided by area size.
Data analysis
For statistical analyses and graph depiction, GraphPad Prism version 8.4.3 for macOS was used. In Figs. 1A–C, 3A,C,D, 4A,B, and Supplementary Figs. S1A & S1B, one-sample t-tests were performed as the theoretical mean was 0. In Figs. 1A,B, 2A, 3A,D, 4A, and Supplementary Figs. S1A & S1B, Welch’s unpaired t-tests were performed. In Figs. 2B and 3B, Dunn’s multiple comparison test was performed after Kruskal–Wallis tests. In Figs. 3C and 4B, Tukey’s multiple comparison test was performed after one-way ANOVA. Graphs in Figs. 2B and 3B indicate median ± 95% confidential interval (CI), and the other graphs indicates mean ± standard deviation (SD).
Data availability
All datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request. Further information and requests for resources and reagents are also available from the corresponding author.
References
Clutton-Brock, T. H. The Evolution of Parental Care (Princeton University Press, 1991).
Holekamp, K. E. & Smale, L. Dispersal status influences hormones and behavior in the male spotted hyena. Horm. Behav. 33, 205–216. https://doi.org/10.1006/hbeh.1998.1450 (1998).
Rincon-Cortes, M. & Sullivan, R. M. Early life trauma and attachment: immediate and enduring effects on neurobehavioral and stress axis development. Front. Endocrinol. (Lausanne) 5, 33. https://doi.org/10.3389/fendo.2014.00033 (2024).
Schmidt, M. V. et al. The postnatal development of the hypothalamic-pituitary-adrenal axis in the mouse. Int. J. Dev. Neurosci. 21, 125–132. https://doi.org/10.1016/s0736-5748(03)00030-3 (2003).
Ost’adalova, I. & Babicky, A. Periodization of the early postnatal development in the rat with particular attention to the weaning period. Physiol. Res. https://doi.org/10.33549/physiolres.932385 (2012).
Espinosa, J. S. & Stryker, M. P. Development and plasticity of the primary visual cortex. Neuron 75, 230–249. https://doi.org/10.1016/j.neuron.2012.06.009 (2012).
Hensch, T. K. Critical period plasticity in local cortical circuits. Nat. Rev. Neurosci. 6, 877–888. https://doi.org/10.1038/nrn1787 (2005).
Hooks, B. M. & Chen, C. Critical periods in the visual system: changing views for a model of experience-dependent plasticity. Neuron 56, 312–326. https://doi.org/10.1016/j.neuron.2007.10.003 (2007).
Kirkby, L. A., Sack, G. S., Firl, A. & Feller, M. B. A role for correlated spontaneous activity in the assembly of neural circuits. Neuron 80, 1129–1144. https://doi.org/10.1016/j.neuron.2013.10.030 (2013).
Nakashima, A. et al. Structured spike series specify gene expression patterns for olfactory circuit formation. Science 365, eaaw5030 (2019). https://doi.org/10.1126/science.aaw5030
Nakashima, A. et al. Agonist-independent GPCR activity regulates anterior-posterior targeting of olfactory sensory neurons. Cell 154, 1314–1325. https://doi.org/10.1016/j.cell.2013.08.033 (2013).
Inoue, N., Nishizumi, H., Naritsuka, H., Kiyonari, H. & Sakano, H. Sema7A/PlxnCl signaling triggers activity-dependent olfactory synapse formation. Nat. Commun. 9, 1842. https://doi.org/10.1038/s41467-018-04239-z (2018).
Inoue, N. et al. The olfactory critical period is determined by activity-dependent Sema7A/PlxnC1 signaling within glomeruli. Elife 10, e65078. https://doi.org/10.7554/eLife.65078 (2021).
Galef, B. G. Jr. & Kaner, H. C. Establishment and maintenance of preference for natural and artificial olfactory stimuli in juvenile rats. J. Comp. Physiol. Psychol. 94, 588–595. https://doi.org/10.1037/h0077693 (1980).
Sevelinges, Y., Mouly, A. M., Levy, F. & Ferreira, G. Long-term effects of infant learning on adult conditioned odor aversion are determined by the last preweaning experience. Dev. Psychobiol. 51, 389–398. https://doi.org/10.1002/dev.20378 (2009).
Murofushi, W., Mori, K., Murata, K. & Yamaguchi, M. Functional development of olfactory tubercle domains during weaning period in mice. Sci. Rep. 8, 13204. https://doi.org/10.1038/s41598-018-31604-1 (2018).
Kobayakawa, K. et al. Innate versus learned odour processing in the mouse olfactory bulb. Nature 450, 503–508. https://doi.org/10.1038/nature06281 (2007).
Herman, J. P. et al. Regulation of the Hypothalamic-Pituitary-Adrenocortical Stress Response. Compr. Physiol. 6, 603–621. https://doi.org/10.1002/cphy.c150015 (2016).
Kondoh, K. et al. A specific area of olfactory cortex involved in stress hormone responses to predator odours. Nature 532, 103–106. https://doi.org/10.1038/nature17156 (2016).
Nagasawa, M., Okabe, S., Mogi, K. & Kikusui, T. Oxytocin and mutual communication in mother-infant bonding. Front. Hum. Neurosci. 6, 31. https://doi.org/10.3389/fnhum.2012.00031 (2012).
Nelson, E. & Panksepp, J. Oxytocin mediates acquisition of maternally associated odor preferences in preweanling rat pups. Behav. Neurosci. 110, 583–592 (1996).
Cater, M. & Majdic, G. How early maternal deprivation changes the brain and behavior?. Eur. J. Neurosci. 55, 2058–2075. https://doi.org/10.1111/ejn.15238 (2022).
Hacquemand, R., Pourie, G., Jacquot, L. & Brand, G. Postnatal exposure to synthetic predator odor (TMT) induces quantitative modification in fear-related behaviors during adulthood without change in corticosterone levels. Behav. Brain Res. 215, 58–62. https://doi.org/10.1016/j.bbr.2010.06.024 (2010).
Root, C. M., Denny, C. A., Hen, R. & Axel, R. The participation of cortical amygdala in innate, odour-driven behaviour. Nature 515, 269–273. https://doi.org/10.1038/nature13897 (2014).
Davis, M. The role of the amygdala in fear and anxiety. Annu. Rev. Neurosci. 15, 353–375. https://doi.org/10.1146/annurev.ne.15.030192.002033 (1992).
Duvarci, S. & Pare, D. Amygdala microcircuits controlling learned fear. Neuron 82, 966–980. https://doi.org/10.1016/j.neuron.2014.04.042 (2014).
Somerville, L. H., Whalen, P. J. & Kelley, W. M. Human bed nucleus of the stria terminalis indexes hypervigilant threat monitoring. Biol. Psychiatry 68, 416–424. https://doi.org/10.1016/j.biopsych.2010.04.002 (2010).
Yassa, M. A., Hazlett, R. L., Stark, C. E. & Hoehn-Saric, R. Functional MRI of the amygdala and bed nucleus of the stria terminalis during conditions of uncertainty in generalized anxiety disorder. J. Psychiatr. Res. 46, 1045–1052. https://doi.org/10.1016/j.jpsychires.2012.04.013 (2012).
Kim, S. Y. et al. Diverging neural pathways assemble a behavioural state from separable features in anxiety. Nature 496, 219–223. https://doi.org/10.1038/nature12018 (2013).
Moriceau, S. & Sullivan, R. M. Maternal presence serves as a switch between learning fear and attraction in infancy. Nat. Neurosci. 9, 1004–1006. https://doi.org/10.1038/nn1733 (2006).
Goodwin, G. A. & Yacko, H. Emergence of the exploratory motive in rats. Dev. Psychobiol. 45, 34–48. https://doi.org/10.1002/dev.20012 (2004).
Alberts, J. R. & Leimbach, M. P. The first foray: maternal influences in nest egression in the weanling rat. Dev. Psychobiol. 13, 417–429. https://doi.org/10.1002/dev.420130408 (1980).
Gerrish, C. J. & Alberts, J. R. Postsuckling behavioral arousal in weanling rats (Rattus norvegicus). J. Comp. Psychol. 111, 37–49. https://doi.org/10.1037/0735-7036.111.1.37 (1997).
Thiels, E., Alberts, J. R. & Cramer, C. P. Weaning in rats: II. Pup behavior patterns. Dev. Psychobiol. 23, 495–510. https://doi.org/10.1002/dev.420230605 (1990).
Terranova, M. L. & Laviola, G. Individual-Differences in Mouse Behavioral-Development - Effects of Precocious Weaning and Ongoing Sexual Segregation. Anim. Behav. 50, 1261–1271 (1995).
Kikusui, T. et al. Early weaning increases anxiety via brain-derived neurotrophic factor signaling in the mouse prefrontal cortex. Sci. Rep. 9, 3991. https://doi.org/10.1038/s41598-019-40530-9 (2019).
Ito, A., Kikusui, T., Takeuchi, Y. & Mori, Y. Effects of early weaning on anxiety and autonomic responses to stress in rats. Behav. Brain Res. 171, 87–93. https://doi.org/10.1016/j.bbr.2006.03.023 (2006).
Li, Y. et al. Neurons for infant social behaviors in the mouse zona incerta. Science 385, 409–416. https://doi.org/10.1126/science.adk7411 (2024).
Lin, C. S., Nicolelis, M. A., Schneider, J. S. & Chapin, J. K. A major direct GABAergic pathway from zona incerta to neocortex. Science 248, 1553–1556. https://doi.org/10.1126/science.2360049 (1990).
Nicolelis, M. A., Chapin, J. K. & Lin, R. C. Development of direct GABAergic projections from the zona incerta to the somatosensory cortex of the rat. Neuroscience 65, 609–631. https://doi.org/10.1016/0306-4522(94)00493-o (1995).
Takayanagi, Y. et al. Pervasive social deficits, but normal parturition, in oxytocin receptor-deficient mice. Proc. Natl. Acad. Sci. USA 102, 16096–16101. https://doi.org/10.1073/pnas.0505312102 (2005).
Kuhn, J. https://imagej.net/plugins/mtrack2. (2012).
Katori, S. et al. Protocadherin-alpha family is required for serotonergic projections to appropriately innervate target brain areas. J. Neurosci. 29, 9137–9147. https://doi.org/10.1523/JNEUROSCI.5478-08.2009 (2009).
Paxinos, G. & Franklin, K. B. J. Paxinos and Franklin’s the mouse brain in stereotaxic coordinates. 4th ed., Elsevier Academic Press (2013).
Acknowledgements
This work was supported by JSPS KAKENHI Grant Number JP17H0660, JP21H05684, & JP22H00433 to H.S.; JP19H04745, JP20K06909, JP23H02514, and JP23H04224 to H.N.; JP19K06760 to S.K. and by NIG-JOINT(25A2021) to S.K.. We thank Dr. Takeshi Yagi, Dr. Takuji Iwasato, Dr. Shun Hamada, and Ms. Ayaka Wada for support and comments on this manuscript.
Author information
Authors and Affiliations
Contributions
All experiments were designed by S.K. and performed by S.K. and Y.N-K. under the supervision of H.N. and H.S. The manuscript was prepared by S.K., H.N., and H.S.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Katori, S., Nishizumi, H., Noguchi-Katori, Y. et al. Separation of pups from their mother mice enhances odor associative learning at the late lactation stage. Sci Rep 15, 6700 (2025). https://doi.org/10.1038/s41598-025-90821-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-90821-7