Abstract
The role of sodium supplements and sex in the occurrence of exercise-associated hyponatremia (EAH) remains controversial. This study investigated hydration status in ultrarunners (19 males and 9 females) who completed seven marathons over seven consecutive days. Due to the limited number of female participants, no statistical comparison between sexes was performed. Plasma sodium concentration ([Na+]) and multiple hydration markers were assessed before, during, and after the race. Reported sodium supplement consumption showed no association with plasma [Na+]. An overall decline in plasma [Na+] was observed in females (regression slope = -1.278, p = 0.02) across the event, whereas no significant change was detected in males (slope = -0.325, p = 0.57). Additionally, no significant associations were found between plasma [Na+] and other monitored variables, including sodium supplement intake, pre-race hydration strategy, body mass, total body water, plasma osmolality, hematocrit, hemoglobin, urine specific gravity, urinary [Na+], thirst rating, or fluid intake reported pre-, during, and post-stage. No cases of symptomatic or asymptomatic hyponatremia were identified, suggesting that total fluid and sodium intake were adequate to maintain fluid-electrolyte balance and prevent EAH in both sexes.
Similar content being viewed by others
Introduction
Exercise-associated hyponatremia (EAH), which can occur during or up to 24 h after physical activity, is frequently observed in endurance athletes but may also manifest in shorter sports activities1. Serum sodium concentration ([Na+]) thresholds are categorized as follows: hypernatremia (> 145 mmol/L), normonatremia (135–144.9 mmol/L), biochemical hyponatremia (129–134.9 mmol/L), and clinically significant hyponatremia (< 128.9 mmol/L)2. The terms biochemical (usually asymptomatic) hyponatremia and symptomatic hyponatremia are widely used to distinguish different levels of EAH3. According to Hew-Butler et al., EAH is best diagnosed based on clinical symptoms rather than absolute [Na+] values, as this approach provides more effective guidance for medical treatment strategies3. Although the clinical significance of asymptomatic EAH remains debated, its primary concern lies in the potential for progression to symptomatic EAH if asymptomatic athletes continue ingesting hypotonic fluids4. EAH can be categorized into hypervolemic, euvolemic, and hypovolemic types1, with its pathogenesis attributed to multiple mechanisms that involve states of volume excess or volume depletion alongside arginine vasopressin (AVP) secretion1. Symptomatic EAH may lead to serious complications such as encephalopathy, seizures, cerebral edema, and pulmonary edema1,2. Awareness of water intoxication and life-threatening dysnatremia is essential, as severe EAH cases can result in coma or death5. Athletes with asymptomatic EAH should seek immediate medical attention if any neurologic signs or symptoms associated with EAH develop within 24 h post-race, as delayed-onset symptomatic EAH is a recognized occurrence3. Despite its frequency and clinical importance, the precise etiology of EAH remains uncertain. While hydration strategies and inappropriate hydration recommendations have been identified as key contributors to excessive fluid intake5, drinking beyond thirst is widely considered the primary cause of reduced plasma [Na+] and a central pathophysiological factor in EAH development1,2,5. Risk factors include overhydration, weight gain during exercise, elevated ambient temperatures, abundant fluid availability, prolonged endurance exercise and race duration, slow pace, and high or low body mass index3, rather than previously suggested factors such as race experience1. However, other mechanisms, such as non-osmotic AVP secretion, sweat sodium loss, sodium mobilization from non-osmotically active internal stores, and additional factors, may also contribute to the development of hyponatremia5.
Despite extensive research on endurance athletes, studies examining the relationship between sodium supplementation and the occurrence of biochemical or symptomatic EAH in stage-race ultrarunners remain limited. Existing studies have produced variable results regarding the impact of sodium supplementation on plasma [Na+]5. For example, a weak positive association between sodium supplementation and post-race plasma [Na+] was identified in 156 participants of a 161-km race. The authors concluded that sodium supplementation had only a minimal effect on preventing EAH6. Conversely, the use of sodium supplements or salt tablets may stimulate thirst, potentially leading to excessive fluid intake and increasing the risk of EAH7. Koenders et al.8 reported that plasma [Na+] remained lower in individuals adhering to a low-sodium diet both before and during exercise. The role of sodium and/or sodium supplementation in mitigating EAH risk remains contentious9, with sodium requirements differing based on the type and duration of exercise. For shorter events such as marathons, sodium supplementation may be unnecessary, whereas in prolonged ultraruns of 160 km, it becomes critical due to greater fluid and sweat losses10. Despite the ongoing debate, sodium supplementation during exercise is widely recommended as a strategy to maintain hydration6. Many runners believe that sodium supplementation helps prevent muscle cramps, nausea, and gastrointestinal symptoms during exercise, making it a common practice among ultra-endurance athletes seeking to avoid EAH and sustain optimal hydration levels6.
In addition to the factors mentioned, there are other poorly understood elements that variably contribute to the occurrence of EAH. Evidence regarding potential secondary risk factors, such as female sex, and the differing risks of decreased plasma [Na+] and distinct EAH incidence rates between female and male ultrarunners remains limited and conflicting11,12. A comprehensive review of hydration and fluid balance in females underscores the lack of studies focused on sex differences in hydration and the relationship between female sex hormones and fluid regulation11. Despite these knowledge gaps, female sex is considered a potential secondary risk factor for the development of EAH1. Recent analyses of clinical presentations of EAH among male and female Ironman triathletes spanning three decades reveal that females are more likely than males to develop hyponatremia during ultra-endurance events13. Some authors propose that this increased risk of EAH in females is associated with higher fluid consumption compared to males14, while others argue that EAH is more closely associated with body mass index and race time rather than sex alone15. Notably, the female sex hormones estrogen and progesterone are involved in fluid balance and regulation16,17, particularly at specific points of the menstrual cycle18. Giersch et al.11 propose a theoretical framework, pending confirmation from high-quality studies, suggesting that fluctuations in reproductive hormones across the menstrual cycle significantly influence thirst and volume regulation in females. These hormones may impair cell volume regulation, affect thirst threshold, and stimulate AVP secretion, resulting in greater water turnover depending on the menstrual cycle phase11,16,19. However, a recent laboratory study in females suggests that while AVP may regulate fluid balance at low dehydration levels, it might have a limited impact on fluid balance, fluid regulatory responses, and volume regulation, and during more significant dehydration, it did not influence body mass decrease from dehydration during exercise in heat20. Similarly, in a study by Stachenfeld and Taylor17, the manipulation of sex hormones did not impact plasma [Na+] reduction in females, although the authors suggest that fluid retention increased and sodium decreased more in females susceptible to EAH. Additionally, a study by Eijsvogel et al.14 on responses during prolonged exercise found no differences in body mass, plasma volume, or plasma [Na+] between premenopausal and postmenopausal females. Furthermore, Giersch et al.20 suggest that the effects of exercise load and dehydration on fluid balance may be so substantial that they can override any hormonal influence. In summary, further systematic research is needed in this area, as existing results are inconsistent.
It is well established that knowledge gaps exist regarding sex as a biological variable in sports performance, and a crucial step toward addressing these gaps is to include more women in research to better understand sex differences in acute and chronic exercise responses21. However, recruiting a sufficient number of female finishers in ultra-endurance events, such as a seven-day stage race covering marathon distances each day in natural terrain, as in the current study, poses significant challenges. Consequently, many studies are conducted primarily on male athletes, often with the assumption that similar effects apply to female athletes21. To help bridge these gaps, we present a case study of nine female and nineteen male ultrarunners who completed seven marathons over seven consecutive days. Ultramarathon running events, which impose substantial physiological demands, provide a unique opportunity to assess potential health risks and physiological responses. In this study, we investigated the hydration status of the participants, including the influence of reported sodium supplement consumption on changes in plasma [Na+]. Given the inconsistencies in previous research, this study also aimed to explore the potential impact of biological sex differences on hydration markers during a seven-day multistage ultramarathon, with plasma [Na+] as the primary marker of both tonicity and water balance22,23. We hypothesized that multiple factors could differentially affect hydration status in male and female ultrarunners across the seven stages of the event. However, due to the small number of female participants, no statistical comparisons between sexes were performed.
Methods
Race
The Moravian Ultra Marathon was an international, multistage running event consisting of seven marathons held over seven consecutive days. It was the longest multistage race in the Czech Republic, taking place from June 28 to July 4, 2015. Each stage covered approximately the distance of a standard marathon (42.2–43.0 km) and traversed rugged terrain with an average ascent of 900 m, featuring a unique route each day. Each stage of the race began and ended at the same location. A general description of the individual race stages is provided in Table 1.
Participants
All race participants were invited to join the study. Out of fifty-three amateur ultrarunners who completed all seven stages, twenty-eight volunteers (53%) participated, comprising nineteen males (43% of all male finishers) and nine females (90% of female finishers). Although the proportion of female ultrarunners has increased recently, females still represent only about 10–20% of participants in such events24.
Procedures and calculations
Participant background and experience
Participants were invited to complete a web-based questionnaire before the race, focusing on pre-race running experience, training status, and typical fluid intake during similar endurance events. Data on participants’ sex, age, and average racing speed were obtained from the official race website25.
Thirst perception, reported sodium supplement consumption, and fluid intake during the race
Thirst rating (TR) was recorded on a 10-point scale, as described by Hoffman et al.26. Participants rated their thirst by responding to the question, “How thirsty do I feel at this exact moment?” with the following possible answers: not thirsty at all(TR = 0), mildly thirsty(TR = 1–3), moderately thirsty(TR = 4–6), very thirsty(TR = 7–8), and extremely thirsty(TR = 9–10). At each stage, participants recorded their sodium supplement intake from the end of the previous stage until the start of the next day’s race. During the race and immediately after each stage, data on sodium supplement consumption was also collected by our assistants at each fluid station, where they recorded sodium supplement use. Similarly, participants documented their fluid intake starting from midnight until the race start for each stage. Post-stage fluid intake from the finish line until midnight was also recorded, with pre-stage and post-stage records handed over to assistants each morning before the race start. To support accuracy and recall, we met with each athlete upon recruitment to explain recording procedures and provided daily reminders at the pre-stage point. Assistants collected data on fluid intake and thirst rating before, during, and immediately after each stage. At each fluid station, assistants recorded the number of accurately measured cups consumed, as well as other fluids ingested. Although fluids at stations could be used for other purposes (e.g., cooling), we assume that such uses mainly occurred near the fluid station. Fluids consumed as part of a meal or snack were not recorded. The race organizers did not provide specific advice regarding hydration on the website, and the ultrarunners did not carry personal fluid bottles or hydration bladders. After each stage, during post-race measurements, we conducted interviews with each runner at the sample collection point. Total fluid intake was estimated based on the data reported by athletes and assistants, representing an average value from fluid intake recorded before and during the respective stage. Each stage featured six refreshment stations offering food and beverages, including water, sports drinks, tea, soup, caffeinated drinks, mineral water, fresh and dried fruit, cheese, salty and sweet biscuits, chips, peanuts, and chocolate. Runners could also use their own refreshments.
Hydration status measurements
Body mass was measured to the nearest 0.1 kg at multiple time points: during registration on the race day, within 1.5 h before the start of each stage (pre-stage 1–7), and immediately after completing each stage (post-stage 1–7). Participants, barefoot and wearing sports clothing, were advised to void their bladders and sit for 10 to 15 min before measurements. A direct segmental multi-frequency bioelectrical impedance analysis device (InBody 720, Inbody Co. Ltd) was used to assess body mass and total body water changes. All measurements were conducted in a temperature-controlled environment indoors. Touch-type electrodes were placed on participants’ feet near the ankles and on their hands near the wrists. A current frequency of 50, 100, 500, and 1000 kHz was applied across five segments (right arm, left arm, right leg, left leg, trunk) for a total duration of approximately 90 s, until the measurements were completed.
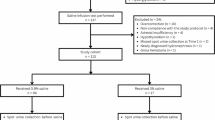
Due to the challenging conditions of the seven-stage running race, biochemical, hematological, and urinary samples were collected only before the race and immediately after completing stages one, four, and seven, corresponding to the beginning, middle, and end of the multistage race, respectively. Participants remained seated for 15 min before blood collection to standardize conditions. Blood was drawn from the antecubital vein using two Sarstedt S-Monovettes (plasma gel, 7.5 mL) for biochemical analysis and one Sarstedt S-Monovette (EDTA, 2.7 mL) (Sarstedt, Nümbrecht) for hematological analysis. Blood samples were immediately transported to the laboratory and analyzed within six hours. Plasma [Na+] was measured using the modular SWA biochemical analyzer, Module P + ISE (Hitachi High Technologies Corporation, Japan, Roche Diagnostics), while plasma osmolality was assessed with the Arkray Osmo Station (Arkray Factory, Inc., Japan). Hematocrit (Hct) was measured with the Sysmex XE 2100 (Sysmex Corporation, Japan), and hemoglobin (Hb) levels were analyzed using the Diagon D-Cell 60 automated hematology analyzer (Diagon Ltd., Budapest, Hungary). Relative plasma volume (PV) changes between pre- and post-stage measurements were calculated using the Strauss formula, which incorporates changes in Hct and Hb levels between two time points: ΔPV = 100 × [Hb (pre-stage)/Hb (post-stage)] × [1-Hct (post-stage)/1-Hct (pre-stage)]27. Urine samples were collected in Sarstedt Urine Monovettes (10 mL) and transported to the laboratory. Urinary [Na+] was measured using the Modular SWA biochemical analyzer, Module P + ISE (Hitachi High Technologies Corporation, Japan, Roche Diagnostics), and urine specific gravity (Usg) was measured with the Au Max-4030 (Arkray Factory, Inc., Japan).
Statistical analysis
Data were analyzed using MATLAB R2021a (The MathWorks, Inc., United States). In addition to the ‘raw’ parameter values (e.g., plasma [Na+], body mass), we calculated changes as the difference between pre- and post-stage values, expressing percentage changes relative to pre-stage values. The Shapiro-Wilk test was used to verify normality for parameters measured only once (e.g., age). Since normality was not confirmed for all parameters, non-parametric tests were applied to compare different data groups, with results presented as medians and interquartile ranges (IQRs). The sign test was used to assess the significance of percentage changes (compared to zero). The Wilcoxon signed-rank test was applied for comparisons within two dependent groups (e.g., pre- and post-stage body mass in males), while the Friedman test was used for comparisons across more than two dependent groups (e.g., body mass measured over seven consecutive stages in males). Pairwise comparisons for significant Friedman tests were corrected using the Bonferroni method. To assess overall trends in parameters across the race (e.g., reduction in post-stage plasma [Na+]), we used linear regression models to compare with the corresponding constant-based (no change) alternative. From our previous pilot study, which measured only demographic and select plasma parameters in runners at the race’s end, the effect size d(Cohen’s d) for plasma [Na+] in males and females was approximately 0.8. With a statistical power of 0.8 and a significance level (alpha) of 0.05, comparing independent groups using the Mann-Whitney U test (e.g., plasma [Na+] in males vs. females) would require at least 19 samples. In our study, the female group had only nine participants, resulting in a power of 0.6. Consequently, we present only within-group statistical analyses for measured parameters, without conducting statistical comparisons between males and females.
Spearman’s coefficient (ρ) was used to assess correlations between two continuous parameters (e.g., body mass and plasma [Na+]). For associations between binary and continuous parameters (e.g., sex and plasma [Na+]) measured at different moments of the race, we used the point-biserial correlation coefficient (r). The Kendall’s coefficient (τ) was applied to assess associations between ordinal and continuous parameters (e.g., post-stage thirst rating and post-stage plasma [Na+]). The Fisher’s exact test was used to compare male and female athletes’ pre-race questionnaire responses on hydration strategy (e.g., “Thirst” vs. “Pre-schedule”). Additionally, the Cochran’s Q test was utilized to examine differences in planned drinking strategies (more than two options) reported in the pre-race questionnaire. Finally, McNemar’s tests with Holm correction were applied to compare responses across different questionnaire categories.
Results
Pre-race characteristics and race Pace
Pre-race parameters and the mean race pace of the runners are summarized in Table 2.
Reported fluid intake and thirst rating
Fluid intake reported by male and female athletes at various time points during the race is summarized in Table 3. Pre-stage fluid intake values (from midnight until the stage start, pre-stage two to pre-stage seven) were similar to pre-race values for both males and females. The combined fluid intake reported during and after each stage, along with the following pre-stage fluid intake, amounted to approximately 3.6 L for stages two through seven. Additionally, fluid intake showed no correlation with any parameters representing the ultrarunners’ experience, such as years of active running, number of completed ultramarathons, mean weekly training volume in running, or longest weekly run. After stages one, four, and seven, body mass, total body water values, and race pace were not associated with fluid intake (pre-stage, during the stage, or total intake) in either female or male ultrarunners.
Thirst ratings before and after each stage are shown in Fig. 1. No differences were observed in pre-stage thirst ratings between individual stages. In male ultrarunners, reported thirst ratings significantly increased by 3–6 points on a 10-point scale after each stage compared to pre-stage values (p < 0.005). However, in female ultrarunners, the corresponding changes in thirst rating (approximately 1–2 points) were not significant. No significant association was observed between thirst ratings and fluid intake, nor between thirst ratings and post-stage plasma [Na+] at different time points (pre-stage and during the stages) in either male or female ultrarunners. Changes in plasma [Na+] were not associated with reported sodium supplement consumption. Additionally, no significant association was found between thirst rating, sodium supplement consumption, and fluid intake at different time points (before and during the stages) in male or female ultrarunners.
Thirst ratings recorded in ultrarunners during the race. F and M represent female and male ultrarunners, respectively. Boxplots depict the median (center line), interquartile range (IQR; bounds of the box), values within 1.5 times the IQR from the 25th and 75th percentiles (whiskers), and outliers, defined as values more than 1.5 times the IQR away from the 25th and 75th percentiles (‘+’). * p < 0.05 post-stage vs. corresponding pre-stage in M and F groups separately (Wilcoxon signed-rank test).
Reported race hydration strategy
Responses from the pre-race questionnaire on hydration strategy are presented in Table 4, indicating that fluid intake would primarily be influenced by thirst.
Reported sodium supplement consumption
Responses from the pre-race questionnaire on sodium supplement use are presented in Table 5.
Sodium supplements were used by 35% of male ultrarunners and 20% of female ultrarunners (see Fig. 2, left and middle panels). No association was observed between reported sodium supplement consumption and fluid intake in either male or female ultrarunners. Furthermore, no significant differences were found in post-stage plasma [Na+] (after stages one, four, and seven) or in the percentage change in plasma [Na+] (after stages one, four, and seven relative to pre-race levels) between sodium supplement users and non-users (both male and female). This was consistent across various comparisons, including sodium supplement consumption prior to specific stages (pre-stage one, four, or seven), consumption either before or during specific stages, and anytime during the race (where a runner was classified as a supplement user if they consumed at least one supplement before or during any stage). Additionally, no significant differences were observed in selected characteristics – such as age, training background, post-race body mass, total body water, blood parameters, mean fluid intake, or thirst rating throughout the race – between ultrarunners (both male and female) who used sodium supplements and those who did not (either before or during the stages).
Sodium supplement use before, during, and both before and during each stage (left, middle, and right panels, respectively). F and M indicate female and male ultrarunners, respectively.
Blood, urinary, body mass, and total body water variables
The results for ‘raw’ parameters and their percentage changes are summarized in Table 6. Post-stage plasma [Na+] did not fall below the diagnostic threshold of 135 mmol/L2, indicating that no ultrarunner had plasma [Na+] < 135 mmol/L after stages one, four, and seven. None of the participants were symptomatic before, during, or after the race. A significant overall decrease in plasma [Na+] was observed in female runners (regression slope = -1.278, p = 0.02) throughout the event, while no significant trend was found in males (slope = -0.325, p = 0.57) (see Fig. 3A). In females, plasma [Na+] percentage changes were significantly different when comparing the beginning of the race to the middle (post-stage one vs. post-stage four) and the end (post-stage one vs. post-stage seven) (see Fig. 3B; Table 6).
Plasma sodium concentration ([Na+]) in ultrarunners: (A) Plasma [Na+] measured pre-race and post-stage one, four, and seven, with median values shown as colored circles and linear regression lines fitted through post-stage values (dashed lines); (B) Plasma [Na+] changes measured after the stages relative to pre-race levels. F and M represent female and male ultrarunners, respectively. Boxplots depict the median (center line), interquartile range (IQR; box bounds), values within 1.5 times the IQR from the 25th and 75th percentiles (whiskers), and outliers, defined as values > 1.5 times the IQR from the 25th and 75th percentiles (‘+’). * p < 0.01 for significant differences compared to stage one (Wilcoxon signed-rank test with Bonferroni correction).
Plasma osmolality significantly increased in males, whereas in females, it increased significantly only after stage one and remained stable in subsequent stages. In both sexes, a significant positive correlation (ρ = 0.52–0.88) was observed between post-stage plasma [Na+] and plasma osmolality (post-stages one, four, and seven), as well as between their percentage changes relative to pre-race levels.
Among female participants in our study, five (56%) had pre-race Usg values even < 1.012 g/mL. According to the position statement by the American College of Sports Medicine (ACSM), a Usg ≤ 1.020 g/mL is considered a biomarker for euhydration28,29,30. Conversely, any Usg value below 1.012 was indicative of extreme hyperhydration in the study by Armstrong et al., which analyzed morning urine samples from 59 active males31. Hypovolemic EAH is suggested by urinary [Na+] levels < 30 mmol/L32 when accompanied by plasma [Na+] < 135 mmol/L. While no cases of biochemical or clinical hyponatremia were observed in this study, six female participants (67%) exhibited post-race urinary [Na+] < 30 mmol/L.
Body mass and total body water parameters are shown in Fig. 4. Percentage changes in post-race body mass and total body water were not associated with percentage changes in plasma [Na+] in either sex. Plasma volume decreased by 1.5% in both males and females post-stage one. It decreased by 0.9% in males and increased by 0.2% in females post-stage four, with no significant differences between post-stage four and post-stage one values. Plasma volume decreased by 1.7% in males and by 0.8% in females post-stage seven, also without significant differences from post-stage one values.
Body mass (A) and total body water (B) measured in ultrarunners pre-race, pre-stage (2–7), and post-stage (1–7). F and M indicate female and male ultrarunners, respectively. Boxplots depict the median (center line), interquartile range (IQR; box bounds), values within 1.5 times the IQR from the 25th and 75th percentiles (whiskers), and outliers, defined as values > 1.5 times the IQR from the 25th and 75th percentiles (‘+’).
Discussion
The key finding of this study is the absence of biochemical or clinical hyponatremia among ultrarunners who completed seven marathons over seven consecutive days, despite a continuous, though clinically insignificant, decrease in plasma [Na+] in females. We investigated hydration-related parameters in both male and female ultrarunners, and an additional finding was that reported sodium supplement consumption was not associated with plasma [Na+] in these participants.
Plasma osmolality and serum sodium are considered the primary markers of water balance and plasma tonicity33, and maintaining plasma osmolality is essential for preventing the adverse effects of EAH5. In females, post-stage plasma [Na+] exhibited a gradual, though non-clinical, decline throughout and after the race compared to pre-race levels, whereas these changes were not statistically significant in males. Plasma osmolality was associated with plasma [Na+] changes in both sexes. However, a significant increase in plasma osmolality was observed only in males, where it rose consistently across the entire race. In contrast, females exhibited a significant increase in plasma osmolality only after the first stage, with levels stabilizing thereafter. However, a review evaluating hydration assessment techniques29 indicates that no concentration measurement, including plasma osmolality, reliably represents total body water changes. It is possible that some osmotically active particles may have raised plasma osmolality, partially masking an actual decrease during and at the end of the race, as suggested by the decline in plasma [Na+] in female ultrarunners. Changes in plasma osmolality could also reflect elevated plasma urea concentrations during prolonged running, as suggested by Knechtle et al.33 in a study on a similar seven-day multistage ultramarathon. Based on these findings, we assume that plasma osmolality returned to its homeostatically regulated level by the day following each stage, preventing EAH even in female runners. The body’s protection and no EAH occurrence may have been due to the increase in plasma osmolality and activation of thirst mechanisms to regulate adequate fluid intake.
Based on a pre-race questionnaire, 65% of males and 56% of females in this study planned to use sodium supplements during the race, primarily to prevent cramps and EAH. Similar findings were observed among 1,152 ultrarunners, with nearly 67% indicating that sodium supplements should be available throughout the race to prevent EAH and muscle cramps7. Endurance athletes from six countries generally believed in the benefits of daily sodium intake (before or during competition), with negligible differences in sodium intake beliefs and pre-planned strategies for sodium intake between males and females34. Recent research confirms that ultrarunners continue to believe in the need for sodium intake during ultramarathons7. However, it is notable that the actual sodium supplement consumption throughout the race – before and during stages – was 30% lower in males and 36% lower in females compared to their pre-race intentions stated in the questionnaires. In fact, only one-third of.
all ultrarunners reported using sodium supplements throughout the race. Despite this, pre-stage thirst ratings remained nearly constant among males and females, suggesting full recovery and stable fluid balance between stages. Fluid balance appeared comparable between the group of ultrarunners who used sodium supplements and those who did not, with no differences in reported fluid intake amounts, body mass changes, or thirst ratings between these groups. Low dietary sodium intake for ten days prior to a race may decrease plasma [Na+], and in combination with fluid losses and fluid overconsumption, this can lead to adverse effects during the race1. However, it appears that low sodium intake is less impactful on hyponatremia risk than excessive fluid intake9. A recent study on modeling sodium requirements suggests that targeted sodium replacement may be necessary for a 160-km ultramarathon10. In contrast, a recent study examining the 80 km stage of the 250 km 6-stage RacingThePlanet ultramarathon concluded that sodium supplementation did not prevent EAH35. In our study, there was no indication that sodium supplement consumption had any impact on fluid balance, regardless of supplement use. Moreover, sodium supplement consumption was not associated with other measured variables in either group. Both groups maintained comparable fluid balance, and no cases of biochemical or clinical EAH were observed at any stage of the race. While personalized sodium replacement strategies might benefit individual athletes in such events, our findings provide no evidence to support a relationship between electrolyte-water balance and reported sodium supplement consumption during this specific seven-day multistage race, which comprised seven consecutive marathons and represents an extreme and atypical race format. However, it is important to note that we did not measure 24-hour sodium and fluid intakes, which limits our ability to draw definitive conclusions about the contribution of sodium intake to sodium changes in the current ultrarunners. Nevertheless, given the nature of the race and the results presented, it appears that sodium supplementation had no observable impact on fluid balance among the participants.
The hypothesis that female ultrarunners would adopt different pre-race and race hydration strategies compared to males was not supported. Female ultrarunners reported the same hydration strategy and similar fluid intake during each stage as their male counterparts. Moreover, no significant association was found between post-stage thirst ratings and post-stage plasma [Na+]. Ultrarunners in our study indicated that their feeling of thirst ultimately determined and influenced their hydration strategy, regardless of sex. Thirst is highly sensitive to increases in plasma osmolality, requiring only a 2–3% rise to trigger thirst22. This was evident in male ultrarunners, whose plasma osmolality increased by 2.4 to 3.1%, resulting in a significantly elevated thirst rating after each stage compared to pre-stage values. In contrast, since females maintained stable plasma osmolality after stage four and at the end of the race, their corresponding changes in thirst rating were, as expected, not significant after any measured stage. Hoffman et al.26, who observed only a weak relationship between thirst rating and plasma [Na+], supported the theory that thirst is influenced by a complex set of stimuli beyond plasma sodium. In their study, hyponatremic and non-hyponatremic 161-km ultramarathoners did not show statistically different post-race thirst ratings, leading the authors to conclude that subjectively measured thirst rating may not accurately reflect an athlete’s hydration status. Only changes in serum [Na+] reliably indicate true clinical fluid imbalance and hydration status22. In our study, no significant association was found between reported fluid intake and post-stage plasma [Na+] in either males or females. Nevertheless, Knechtle et al.36 reported that hyponatremic 100-km ultrarunners did not consume more fluids than athletes without EAH, and their reported fluid intake was not associated with post-race plasma [Na+] or plasma [Na+] changes. Since no cases of hyponatremia were identified in our study, the total dietary intake of ultrarunners was likely sufficient to maintain fluid balance.
An intriguing finding in this study was the significant decrease in body mass observed after each stage, contrasting with the stability of total body water, which remained consistent throughout the race for both males and females. The body has various regulatory mechanisms to manage volume fluctuations and osmolality changes, though it is recognized that no single method is infallible for assessing hydration status29, especially under field conditions. According to Armstrong et al.29, when fluid compartments fluctuate during exercise, body mass change is the simplest and most accurate indicator of hydration status. Monitoring body weight can be used to assess hydration changes3, as weight gain often suggests overhydration in hyponatremic cases. However, it is important to note that some athletes do not consume excessive fluids, and volume depletion and weight loss are observed, with additional factors likely contributing to EAH onset5. Nevertheless, neither variable showed any significant association with plasma [Na+] in either males or females. Despite the body mass decreases observed during each stage, body mass returned to pre-race values by the following morning. This recovery suggests that the brain likely responded effectively to the physiological stress of the previous day, maintaining stable plasma osmolality through regulatory mechanisms. It is also worth considering that females’ fluid intake may have been comparable in absolute volume to that of males. Given the differences in body mass and total body water pre-race, this could imply that females actually consumed more fluids than males, potentially explaining that an association between fluid intake and reduced [Na+] has not been conclusively demonstrated. Stachenfeld and Taylor17 suggest that excessive water consumption in females, considering their smaller body size, is the primary factor for water retention and plasma [Na+] reduction in females prone to EAH. Thus, although female ultrarunners reported the same hydration strategy as males, since they did not experience significant changes in thirst sensation during the race, unlike males, females may have consumed more fluid than their bodies required, leading to a more pronounced decrease in plasma [Na+] after each measured stage. Additionally, females may have experienced lower fluid losses due to reduced total sweat production37 compared to males and lower metabolic heat generation from their smaller body mass37, which could potentially contribute to a disturbance in thermal balance and a state of hyperhydration. However, it appears that for fluid and thermoregulatory responses and body mass loss, females may not necessarily require different fluid intake based on sex hormone concentration or menstrual cycle phase after exercise in the heat38,39. Adequate fluid intake likely compensated for any potential fluid loss during the race. While body mass decrease in ultra-athletes is often attributed to dehydration and fluid loss29, the intense physical demands of the race may also play a role. Body mass changes during prolonged exercise may result from various factors, including mobilization of fat stores, utilization of solid substrates, water release from glycogen stores to maintain body water40, skeletal muscle mass reduction41, respiratory water loss, and metabolic water production42. Given the daily distance covered by the ultrarunners (one marathon), these factors likely had a smaller impact on body mass changes than they would in longer races, such as 100-km events, where these processes contribute more to body mass changes, reflecting net fluid balance. However, the challenging, off-road conditions over seven consecutive days made this ultrarace one of the most demanding in Central Europe. Despite these conditions, the stability of total body water throughout the race suggests an effort to maintain optimal fluid balance in both sexes. We must also acknowledge that when assessing hydration status in real-time, caution is needed, as no concentration measurement during exercise, even the gold standard, accurately reflects the dynamic fluid matrix during exercise29. Potential errors in measuring fluid balance and body mass changes – particularly difficult to control in field-based research – may explain the lack of a relationship between reported fluid intake and body mass changes, not only in female ultrarunners.
The mean hematocrit and hemoglobin concentrations significantly decreased during and after the race in both sexes. Changes in plasma osmolality affect erythrocyte volume, thereby influencing hematocrit levels43. To further assess fluid balance in these ultrarunners, plasma volume was calculated using hematocrit and hemoglobin. Hemoglobin was included due to its circulation stability, reflecting plasma volume changes43. Similar to our findings, Reid et al.44 observed reduced post-race hematocrit concentrations in marathon runners, albeit without cases of EAH, and found significantly lower post-race plasma [Na+] in female runners, consistent with our results. Reid et al.44 attribute hematocrit differences between sexes to higher fluid intake among females during the marathon. Conversely, male athletes generally have greater blood volume and hemoglobin concentrations than females45, exhibiting notable hematological adaptations compared to females. Given the significant reductions in both hematocrit and hemoglobin, hyperhydration seemed to be a greater concern than dehydration for these athletes. Furthermore, hemodilution could be attributed to plasma [Na+] retention due to increased AVP46. Osmoregulation through the hypothalamus triggers the release of AVP in response to hypovolemia, with plasma volume contraction serving as a unifying non-osmotic stimulus for AVP secretion during endurance exercise5. It is important to note that plasma volume decreased after the first stage, with no significant changes throughout the remainder of the race in either sex. Some findings suggest that volume depletion may contribute to hypovolemic EAH, commonly observed in ultramarathons32. Conversely, the decreases in hematocrit and hemoglobin could also result from frequent intravascular hemolysis47 during prolonged running, which contributes to sports anemia48, including reductions in hemoglobin and hematocrit levels.
Urine biomarkers may serve as additional indicators of hydration status in female and male ultrarunners. Usg below 1.200 g/mL is considered a biomarker of hyperhydration31. Based on urinary parameters, it appears that female ultrarunners in our study started the first marathon hyperhydrated, as indicated by pre-race Usg concentrations below 1.012 g/mL in nearly 56% of them.
However, Hew-Butler et al.5 advise against using Usg as a surrogate for blood hydration markers at rest. According to the American College of Sports Medicine’s position statement on exercise and fluid replacement, a Usg value ≤ 1.020 g/mL is considered indicative of euhydration28,29,30. Therefore, we suggest that female ultrarunners entered the race in a euhydrated rather than hyperhydrated state. Conversely, Usg significantly increased after the stages in both sexes. However, Usg above 1.020 g/mL reflects urinary water conservation rather than clinically significant intracellular dehydration49. Furthermore, no significant association was found between plasma [Na+] and Usg. The increase in Usg was anticipated, as this is a common phenomenon during ultrarunning races, as seen in a study by Knechtle et al.33 on a seven-day ultrarunning event. Additionally, both female and male ultrarunners in our study returned to pre-race body mass values before each subsequent stage. Although no hyponatremic cases were observed in this study, we included urinary [Na+] as an additional indicator of water balance to provide a comprehensive view of the hydration status in female ultrarunners. No significant changes were found in urinary [Na+] after measured stages in female participants. Hew-Butler et al.5 stated that a rare but notable exception connected with EAH is associated with volume depletion, with symptoms presenting as urinary [Na+] < 30 mmol/L. Hypovolemic EAH is supported by urinary [Na+] below 30 mmol/L32 along with plasma [Na+] < 135 mmol/L. Interesting finding was that post-race urinary [Na+] was below 30 mmol/L in nearly 67% of the female ultrarunners, though their plasma [Na+] remained above 135 mmol/L. Sodium serves as the primary osmotic determinant for assessing extracellular fluid volume50 and is regulated by the kidneys. When the body conserves sodium, urinary [Na+] are typically reduced32, a phenomenon that can also result from acute water overload49. A significant decline in urinary [Na+] after the race was reported in a study comparing male and female 100-km ultramarathoners51. Similarly, in a study of seven-day multistage ultrarunners, urinary [Na+] decreased post-stage but increased again overnight before the next stage33. The authors proposed that this could result from reduced aldosterone activity, potentially leading to EAH. Since post-stage urinary [Na+] was not associated with plasma [Na+] in the ultrarunners in this study, it is plausible that lower resting urinary [Na+] values may also reflect reduced sodium intake in the days leading up to the race. Urinary indices should be interpreted with caution, as they may lag behind plasma indices measured post-exercise, given that the kidneys serve as the primary organ for rapid plasma tonicity regulation29,30. The renal response to exercise and the influence of race diet may thus limit the utility of urine indices in assessing hydration in these ultrarunners.
Field studies on EAH rarely include measurements of sweat volume or concentration due to the logistical challenges involved. Currently, no standardized instrumentation exists for estimating sweat rate, which further complicates its monitoring. Consequently, we did not assess sweat rate in the ultrarunners in this study. However, it is important to acknowledge that profuse sweating and electrolyte loss can disrupt fluid balance, particularly during exercise in heat stress, potentially increasing the risk of EAH52 or heat-related illnesses3. The primary focus is on sweat [Na+] because it represents the most significant electrolyte loss and has a substantial impact on hydration status53. For instance, Morgan et al.54 reported elevated sweat [Na+] in cases of dehydration54. Females may be more sensitive to the adverse thermoregulatory effects of dehydration during the early stages of exercise in heat40. A study investigating the impact of sex during prolonged exercise found that female athletes exhibited significantly higher core body temperatures compared to their male counterparts14. Additionally, females tend to have lower sweat rates than males, which may influence their ability to maintain optimal fluid balance55. Furthermore, females lose less sodium in sweat compared to males56. Contrary to these findings, Taylor et al.57 suggested that sweat [Na+] losses are generally minimal and do not contribute significantly to EAH risk, except in the context of prolonged endurance activities. Pahnke et al.56 found no significant differences in plasma [Na+] changes or post-race sweat [Na+] loss between male and female triathletes. In their study, plasma [Na+] changes were negatively correlated with body mass changes rather than sweat rate or sodium loss in sweat. Additionally, only a limited number of studies have explored the potential connections between AVP, sweat, urine, and [Na+] concentrations. Hew-Butler et al.53 proposed that sweat [Na+] changes are associated with water reabsorption and AVP during exercise, though sweat losses alone are not the sole regulators of fluid and sodium balance. Conversely, a subsequent study by the same author concluded that vasopressin V2 receptors, key neuroendocrine regulators of water metabolism, do not influence sweat [Na+] or sweat water content during moderate to high-intensity exercise58. Baker et al.59, in their recent review on the physiological mechanisms influencing sweat composition, stated that there are no correlations between blood and sweat variables, and that sweat electrolyte concentrations do not reliably predict hydration status, sweat rate, or other physiological parameters. Despite these findings, fluid metabolism disorders are frequently reported in female endurance athletes21,60. Therefore, we cannot entirely rule out the possibility that other unobserved factors contributed to the gradual, though non-clinical, decline in plasma [Na+] observed among the female ultrarunners in this study.
Potential sex differences may influence fluid regulation in female athletes during prolonged multistage endurance events, either dependently or independently of the menstrual cycle phase or postmenopausal status. In the present study, factors such as menstrual status, hormonal contraceptive use, and self-reported menstrual cycle phase were not recorded prior to the investigation. Field settings, especially in multi-day endurance events, make it challenging to collect data from a statistically significant number of females in the same cycle phase. Although an artificial menstrual cycle can be induced under controlled laboratory conditions17, this approach is not feasible in the context of real-world, multi-day endurance events. Additionally, given the seven-day duration of the race, some participants likely experienced overlapping phases of their menstrual cycles, further complicating the prediction of hormonal variations18. Wagner et al.61 hypothesized that higher rates of EAH in females may be related to the syndrome of inappropriate antidiuretic hormone secretion (SIADH), which is observed more frequently in females. Female diuretic responses to water stress may be affected by estrogen and progesterone, which can disrupt renal antidiuretic hormone activity and result in increased water turnover, with variations depending on the menstrual cycle phase61. A recent 2023 study analyzing serum [Na+] from medical records at the IRONMAN® World Championships (1989–2019) reported lower serum [Na+] levels in females (137.4 mmol/L) compared to males (139.7 mmol/L)13. The authors concluded that females are significantly more likely to develop EAH and present with lower serum [Na+] when visiting the medical tent during IRONMAN® triathlons. Results of other recent study during a high-altitude 161-km ultramarathon also identified female sex as risk factors for the development of EAH in their asymptomatic cohort62. Research indicated that mean plasma [Na+] in male marathon runners is significantly higher post-race than in in their female counterparts44, though no cases of EAH were reported after completing a standard marathon. According to Stachenfeld and Taylor17, females susceptible to EAH tend to retain more water and experience higher sodium loss compared to those less susceptible17. In open-water ultra-endurance swimming, females did not consume more fluids than males but displayed a higher prevalence of EAH, which the authors attributed to greater AVP secretion in females, leading to excess water retention61. Additionally, a recent laboratory study in females indicated that while AVP may regulate fluid balance under low dehydration levels20, its impact on fluid balance decreases with higher dehydration4. However, Eijsvogels et al.14 found no difference in plasma [Na+] between pre- and postmenopausal females, and Giersch et al.39 reported that sex hormones did not alter fluid response during exercise in heat conditions. While some studies identify female sex as an important risk factor for hyponatremia63, according to study of 100 km ultrarunners, the higher incidence of EAH among females may be due to their greater propensity for over-drinking rather than inherent sex differences60. Similarly, no statistical association between female sex and EAH was found in marathon runners15, and sex alone does not appear to influence AVP levels64. This indicates the need for further research to fully understand the relationship between EAH, overhydration, and sex.
Although the decline in plasma [Na+] observed in female ultrarunners during this seven-stage race, despite maintaining normonatremia, may hypothetically suggest sex as a potential factor predisposing females to electrolyte-water disorders, the absence of either asymptomatic or symptomatic EAH among participants does not conclusively support this hypothesis. Furthermore, the observed plasma [Na+] decrease appears to have minimal clinical significance, and no definitive mechanisms were identified to explain it. In conclusion, despite covering an approximate cumulative distance of 300 km across challenging terrain, participants likely consumed sufficient food and fluids with adequate sodium and water content during non-race meals between stages, enabling them to restore normal plasma [Na+] and avoid both biochemical and clinical EAH. Although we were unable to quantify total 24-hour fluid and food intake over the seven-day race period, these findings may hold relevance for ultra-endurance events36,60.
The novelty of this study lies in its multi-day design, encompassing seven marathons over seven consecutive days, and in its unique execution in a real-world field setting rather than a controlled laboratory environment. Each stage of the race constituted a standalone marathon, offering a rare opportunity to examine short-term physiological changes within the same cohort of participants: immediately following a single marathon, after completing four marathons (midway through the race), and after seven consecutive days of rigorous physical activity (after seven consecutive marathons). Future studies should focus on investigating female hormone levels to elucidate the underlying causes of plasma [Na+] decline during prolonged and demanding ultra-endurance events.
Limitations and strengths
We acknowledge certain limitations, largely due to the constraints of conducting research during a seven-day endurance competition. First, the small number of female athletes must be considered. Obtaining a sufficient number of female finishers for robust statistical analysis in this type of event is highly challenging. Consequently, many studies focus on male athletes, assuming similar effects in female athletes21. Nonetheless, we believe the physiological responses of females under such demanding conditions are valuable to document, despite the inherent difficulty in recruiting more female participants for field research of this nature. Although the proportion of females among ultrarunners has grown in recent years, it remains at only about 10–20%24. Given these factors, the statistical power for sex-based comparisons was limited in the female group, and we therefore did not perform such analyses. Similarly, without sex hormone measurements, the results contribute only partially to the existing literature. However, the recognized gap in understanding sex differences in sports performance underscores the importance of including more females in future research studies21. Secondly, our ability to accurately quantify total fluid intake and adjust body mass changes for the mass of ingested food was limited, as such calculations require comprehensive dietary analysis over the seven-day race period. Similarly, we were unable to measure sweat losses, which could have provided additional insights into fluid balance. Sodium intake was assessed through daily self-reports and interviews with each runner, with a specific focus on sodium supplement use. Participants were informed in advance that they would be asked to provide this information. These potential measurement inaccuracies, which are inherently challenging to control in field-based research, may help explain the absence of a clear relationship between reported fluid intake and body mass changes. Despite these limitations, the unique value of this study lies in its capacity to document short-term physiological changes across multiple stages of exertion. Specifically, the research examines the effects of completing a single marathon (first stage), the cumulative impact of four marathons (midpoint, fourth stage), and the physiological outcomes of seven consecutive days of intense physical activity (race conclusion), all within the same cohort of participants. This design offers a rare perspective on the progressive effects of multi-day endurance events.
Data availability
The datasets analysed in the study are available upon reasonable request from the corresponding author.
References
Hew-Butler, T. Exercise-associated hyponatremia Front. Horm. Res. 52:178–189. https://doi.org/10.1159/000493247 (2019).
Noakes, T. D. et al. Three independent biological mechanisms cause exercise-associated hyponatremia: evidence from 2,135 weighed competitive athletic performances. Proc. Natl. Acad. Sci. USA. 102, 18550–18555. https://doi.org/10.1073/pnas.0509096102 (2005).
Hew-Butler, T. et al. Statement of the Third International Exercise-Associated Hyponatremia Consensus Development Conference, Carlsbad, California, 2015. Clin. J. Sport. Med. 25(4), 303–320. https://doi.org/10.1097/JSM.0000000000000221 (2015).
Bissram, M., Scott, F. D., Liu, L. & Rosner, M. H. Risk factors for symptomatic hyponatraemia: the role of pre-existing asymptomatic hyponatraemia. Intern. Med. J. 37(3), 149–155. https://doi.org/10.1111/j.1445-5994.2006.01294.x (2007).
Hew-Butler, T., Loi, V., Pani, A. & Rosner, M. H. Exercise-Associated hyponatremia 2017 update. Front. Med. 4(21), 1–10. https://doi.org/10.3389/fmed.2017 (2017).
Hoffman, M. D. & Stuempfle, K. J. Sodium supplementation and exercise- associated hyponatremia during prolonged exercise. Med. Sci. Sports Exerc. 47(9), 1781–1787 (2015).
Hoffman, M. D. & White, M. D. Belief in the need for sodium supplementation during ultramarathons remains strong: findings from the ultrarunners longitudinal tracking (ULTRA) study. Appl. Physiol. Nutr. Metab. 45(2), 118–122. https://doi.org/10.1139/apnm-2019-0238 (2020).
Koenders, E. E., Franken, C. P., Cotter, J. D., Thornton, S. N. & Rehrer, N. J. Restricting dietary sodium reduces plasma sodium response to exercise in the heat. Scand. J. Med. Sci. Sports. 27, 1213–1220. https://doi.org/10.1111/sms.12748 (2017).
Hoffman, M. D. & Myers, T. M. Case study: symptomatic exercise-associated hyponatremia in an endurance runner despite sodium supplementation. Int. J. Sport Nutr. Exerc. Metab. 25(6), 603–606. https://doi.org/10.1123/ijsnem.2014-0241 (2015).
McCubbin, A. J. Modelling sodium requirements of athletes across a variety of exercise scenarios - Identifying when to test and target, or season to taste. Eur. J. Sport Sci. 23(6), 992–1000. https://doi.org/10.1080/17461391.2022.2083526 (2023).
Giersch, G. E. W., Charkoudian, N., Stearns, R. L. & Casa, D. J. Fluid balance and hydration considerations for Womales: review and future directions. Sports Med. 50(2), 253–261. https://doi.org/10.1007/s40279-019-01206-6 (2019).
Bailowitz, Z., Grams, R., Teeple, D. & Hew-Butler, T. Exercise-associated hyponatremia in a lactating female. Clin. J. Sport. 27, e55–e57. https://doi.org/10.1097/JSM.0000000000000344 (2017).
Johnson, K. B. et al. Clinical presentation of exercise-associated hyponatremia in male and female IRONMAN® triathletes over three decades. Scand. J. Med. Sci. Sports. 33(9), 1841–1849. https://doi.org/10.1111/sms.14401 (2023).
Eijsvogels, T. M., Scholten, R. R., van Duijnhoven, N. T., Thijssen, D. H. & Hopman, M. T. Sex difference in fluid balance responses during prolonged exercise. Scand. J. Med. Sci. Sports. 23(2), 198–206. https://doi.org/10.1111/j.1600-0838.2011.01371.x (2013).
Almond, C. S. et al. Hyponatremia among runners in the Boston marathon. N Engl. J. Med. 352(15), 1550–1556. https://doi.org/10.1056/NEJMoa043901 (2005).
Stachenfeld, N. S. Sex hormone effects on body fluid regulation. Exerc. Sport Sci. Rev. 36(3), 152–159. https://doi.org/10.1097/JES.0b013e31817be928 (2008).
Stachenfeld, N. S. & Taylor, H. S. Sex hormone effects on body fluid and sodium regulation in women with and without exercise-associated hyponatremia. J. Appl. Physiol. 107(3), 864–872. https://doi.org/10.1152/japplphysiol.91211.2008 (2009).
Owen, J. A. Physiology of the menstrual cycle. Am. J. Clin. Nutr. 28, 333–338. https://doi.org/10.1093/ajcn/28.4.333 (1975).
Claybaugh, J. R., Sato, A. K., Crosswhite, L. K. & Hassell, L. H. Effects of time of day, gender, and menstrual cycle phase on the human response to a water load. Am. J. Physiol. Regul. Integr. Comp. Physiol. 279(3), R966–973. https://doi.org/10.1152/ajpregu.2000.279.3.R966 (2000).
Giersch, G. E. W. et al. Estrogen to progesterone ratio and fluid regulatory responses to varying degrees and methods of dehydration. Front. Sports Act. Living. 3, 722305. https://doi.org/10.3389/fspor.2021.722305 (2021).
Hunter, S. K. et al. The biological basis of sex differences in athletic performance: consensus statement for the American college of sports medicine. Med. Sci. Sports Exerc. 55(12), 2328–2360. https://doi.org/10.1249/MSS.0000000000003300 (2023).
Verbalis, J. G. Disorders of body water homeostasis. Best Pract. Res. Clin. Endocrinol. Metab. 17(4), 471–503. https://doi.org/10.1016/s1521-690x (2003). (03)00049 – 6.
Sterns, R. H. Disorders of plasma sodium. N. Engl. J. Med. 372(13), 1267–1269. https://doi.org/10.1056/NEJMc1501342 (2015).
Fonseca-Engelhardt, K. et al. Participation and performance trends in ultra-endurance running races under extreme conditions - ‘Spartathlon’ versus ‘badwater’. Extr. Physiol. Med. 2(1), 15. https://doi.org/10.1186/2046-7648-2-15 (2013).
MUM. – Results. http://ultracau.ron.cz/racelog.php?menuid=event&record=52&action=results. Accessed 17 May 2021 (2015).
Hoffman, M. D., Fogard, K., Winger, J., Hew-Butler, T. & Stuempfle, K. J. Characteristics of 161-km ultramarathon finishers developing exercise-associated hyponatremia. Res. Sports Med. 21(2), 164–175. https://doi.org/10.1080/15438627.2012.757230 (2013).
Strauss, M. B., Davis, R. K., Rosenbaum, J. D. & Rossmeisl, E. C. Water diuresis produced during recumbency by the intravenous infusion of isotonic saline solution. J. Clin. Invest. 30(8), 862–868. https://doi.org/10.1172/JCI102501 (1951).
American College of Sports Medicine. American college of sports medicine position stand. Exercise and fluid replacement. Med. Sci. Sports Exerc. 39(2), 377–390. https://doi.org/10.1249/mss.0b013e31802ca597 (2007).
Armstrong, L. E. Assessing hydration status: the elusive gold standard. J. Am. Coll. Nutr. 26(5 Suppl), 575S–584S. https://doi.org/10.1080/07315724.2007.10719661 (2007).
Popowski, L. A. et al. Blood and urinary measures of hydration status during progressive acute dehydration. Med. Sci. Sports Exerc. 33(5), 747–753. https://doi.org/10.1097/00005768-200105000-00011 (2001).
Armstrong, L. E. et al. Human hydration indices: acute and longitudinal reference values. Int. J. Sport Nutr. Exerc. Metab. 20(2), 145–153. https://doi.org/10.1123/ijsnem.20.2.145 (2010).
Cairns, R. S. & Hew-Butler, T. Proof of concept: hypovolemic hyponatremia May precede and augment creatine kinase elevations during an ultramarathon. Eur. J. Appl. Physiol. 116, 647–655 (2016).
Knechtle, B. et al. Regulation of electrolyte and fluid metabolism in multi-stage ultra-marathoners. Horm. Metab. Res. 44(12), 919–926. https://doi.org/10.1055/s-0032-1312647 (2012).
McCubbin, A. J., Cox, G. R. & Costa, R. J. S. Sodium intake beliefs information sources, and intended practices of endurance athletes before and during exercise. Int. J. Sport Nutr. Exerc. Metab. 29(4), 371–381 (2019).
Lipman, G. S. et al. Effect of sodium supplements and climate on dysnatremia during ultramarathon running. Clin. J. Sport Med. 31(6), e327–e334. https://doi.org/10.1097/JSM.0000000000000832 (2021).
Knechtle, B., Knechtle, P. & Rosemann, T. Low prevalence of exercise-associated hypothermia in male 100km ultra-marathon in Switzerland. Eur. J. Appl. Physiol. 111(6), 1007–1016. https://doi.org/10.1007/s00421-010-1729-7 (2011).
Glickman, E., Cheatham, C. C., Caine, N., Blegen, M. & Marcinkiewicz, J. L. Influence of gender and menstrual cycle on a cold air tolerance test and its relationship to thermosensitivity. Undersea Hyperb Med. 27(2), 75–81 (2000).
Cheuvront, S. N. & Haymes, E. M. Thermoregulation and marathon running: biological and environmental influences. Sports Med. 31(10), 743–762. https://doi.org/10.2165/00007256-200131100-00004 (2001).
Giersch, G. E. W. et al. Sex difference in initial thermoregulatory response to dehydrated exercise in the heat. Physiol. Rep. 9(14), e14947. https://doi.org/10.14814/phy2.14947uti (2021).
Maughan, R. J., Shirreffs, S. M. & Leiper, J. B. Errors in the Estimation of hydration status from changes in body mass. J. Sports Sci. 25(7), 797–804. https://doi.org/10.1080/02640410600875143 (2007).
Nolte, H. W., Noakes, T. D. & van Vuuren, B. Protection of total body water content and absence of hyperthermia despite 2% body mass loss (‘voluntary dehydration’) in soldiers drinking ad libitum during prolonged exercise in cool environmental conditions. Br. J. Sports Med. 45(14), 1106–1112. https://doi.org/10.1136/bjsm.2010.075507 (2011).
Cheuvront, S. N. & Montain, S. J. Myths and methodologies: making sense of exercise mass and water balance. Exp. Physiol. 102(9), 1047–1053. https://doi.org/10.1113/EP086284 (2017).
Dill, D. B. & Costill, D. L. Calculation of percentage changes in volumes of blood, plasma, and red cells in dehydration. J. Appl. Physiol. 37(2), 247–248. https://doi.org/10.1152/jappl.1974.37.2.247 (1974).
Reid, S. A. et al. Study of haematological and biochemical parameters in runners completing a standard marathon. Clin. J. Sport Med. 14(6), 344–353. https://doi.org/10.1097/00042752-200411000-00004 (2004).
Schmidt, W. & Prommer, N. Impact of alterations in total hemoglobin mass on VO 2max. Exerc. Sport Sci. Rev. 38(2), 68–75. https://doi.org/10.1097/JES.0b013e3181d4957a (2010).
Fellmann, N. et al. Intracellular hyperhydration induced by a 7-day endurance race. Eur. J. Appl. Physiol. Occup. Physiol. 80(4), 353–359. https://doi.org/10.1007/s004210050603 (1999).
Lippi, G. & Sanchis-Gomar, F. Epidemiological, biological and clinical update on exercise-induced hemolysis. Ann. Transl Med. 7(12), 270. https://doi.org/10.21037/atm.2019.05.41 (2019).
Hu, M. & Lin, W. Effects of exercise training on red blood cell production: implications for anemia. Acta Haematol. 127(3), 156–164. https://doi.org/10.1159/000335620 (2012).
Hew-Butler, T. D., Eskin, C., Bickham, J., Rusnak, M. & VanderMeulen, M. Dehydration is how you define it: comparison of 318 blood and urine athlete spot checks. BMJ Open. Sport Exerc. Med. 4(1), e000297. https://doi.org/10.1136/bmjsem-2017-000297 (2018).
Polychronopoulou, E., Braconnier, P. & Burnier, M. New insights on the role of sodium in the physiological regulation of blood pressure and development of hypertension. Front. Cardiovasc. Med. 6, 136. https://doi.org/10.3389/fcvm.2019.00136 (2019).
Rüst, C. A. et al. Is the prevalence of Exercise-Associated hyponatremia higher in female than in male 100-KM Ultra-Marathoners? Hum. Mov. 13, 94–101 (2012).
Institute of Medicine of the National Academies. Dietary reference intakes for water, potassium, sodium, chloride, and sulfate. (The National Academies Press, 2004). ISBN: 0-309-53049-0.
Hew-Butler, T., Noakes, T. D., Soldin, S. J. & Verbalis, J. G. Acute changes in arginine vasopressin, sweat, urine and serum sodium concentrations in exercising humans: does acoordinated homeostatic relationship exist? Br. J. Sports Med. 44(10), 710–715. https://doi.org/10.1136/bjsm.2008.051771 (2010).
Morgan, R. M., Patterson, M. J. & Nimmo, M. A. Acute effects of dehydration on sweat composition in men during prolonged exercise in the heat. Acta Physiol. Scand. 182(1), 37–43. https://doi.org/10.1111/j.1365-201X.2004.01305.x (2004).
Lara, B. et al. Interindividual variability in sweat electrolyte concentration in marathoners. J. Int. Soc. Sports Nutr. 13(31). https://doi.org/10.1186/s12970-016-0141-z (2016).
Pahnke, M. D. et al. Serum sodium concentration changes are related to fluid balance and sweat sodium loss. Med. Sci. Sports Exerc. 42(9), 1669–1674. https://doi.org/10.1249/MSS.0b013e3181d6c72a (2010).
Taylor, N. A. S. & Machado-Moreira, C. A. Regional variations in transepidermal water loss, eccrine sweat gland density, sweat secretion rates and electrolyte composition in resting and exercising humans. Extrem Physiol. Med. 2(1), 4. https://doi.org/10.1186/2046-7648-2-4 (2013).
Hew-Butler, T., Hummel, J., Rider, B. C. & Verbalis, J. G. Characterization of the effects of the vasopressin V2 receptor on sweating, fluid balance, and performance during exercise. Am. J. Physiol. Regul. Integr. Comp. Physiol. 307(4), R366–R375. https://doi.org/10.1152/ajpregu.00120.2014 (2014).
Baker, L. B. & Wolfe, A. S. Physiological mechanisms determining eccrine sweat composition. Eur. J. Appl. Physiol. 120(4), 719–752. https://doi.org/10.1007/s00421-020-04323-7 (2020).
Knechtle, B. et al. Maintained total body water content and serum sodium concentrations despite body mass loss in female ultra-runners drinking ad libitum during a 100 Km race. Asia Pac. J. Clin. Nutr. 19(1), 83–90 (2010).
Wagner, S., Knechtle, B., Knechtle, P., Rüst, C. A. & Rosemann, T. Higher prevalence of exercise- associated hyponatremia in female than in male open-water ultra-endurance swimmers: the ‘Marathon-Swim’ in lake Zurich. Eur. J. Appl. Physiol. 112(3), 1095–1106. https://doi.org/10.1007/s00421-011-2070-5 (2012).
Khodaee, M., Saeedi, A., Harris-Spinks, C. & Hew-Butler, T. Incidence of exercise-assosciated hyponatremia during a high-altitude 161-km ultramarathon. Phys. Act. Nutr. 25(3), 16–22. https://doi.org/10.20463/pan.2021.0016 (2021).
Hawkins, R. C. Age and gender as risk factors for hyponatremia and hypernatremia. Clin. Chim. Acta. 337(1–2), 169–172. https://doi.org/10.1016/j.cccn.2003.08.001 (2003).
Hew-Butler, T. Arginine vasopressin, fluid balance and exercise: is exercise-associated hyponatraemia a disorder of arginine vasopressin secretion? Sports Med. 40(6), 459–479. https://doi.org/10.2165/11532070-000000000-00000 (2010).
Acknowledgements
The authors gratefully acknowledge the athletes for their excellent cooperation without which this study could not have been conducted. We thank the organizers of the ‘Moravian Ultra Marathonʼ for their generous support.
Author information
Authors and Affiliations
Contributions
DCH designed the study and collected the data. MF contributed to data processing and statistical analysis. DCH and MF wrote the manuscript. DCH, BK, KW, TR, MT and SR contributed to critical reading and editing of the manuscript. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study was approved by the local institutional ethics committee of the Centre of Sports Activities, Brno University of Technology, Czech Republic (ethical approval number EK 5/2015). The study was conducted in accordance with the ethical standards outlined in the Declaration of Helsinki, and all volunteers provided written informed consent.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chlibková, D., Filipenska, M., Knechtle, B. et al. No hyponatremia despite continuous plasma sodium decline in female runners during a seven stage ultramarathon. Sci Rep 15, 11400 (2025). https://doi.org/10.1038/s41598-025-90987-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-90987-0