Abstract
Colorectal cancer is the third most common cancer worldwide. Elderly patients, typically defined as those over 70 years, face a heightened risk of developing colorectal cancer as they age, and the proportion of elderly individuals affected by cancer will continue to increase. However, the current guidelines lack optimal treatment recommendations for the elderly and there is a scarcity of clinical trial evidence of clinical trial evidence addressing rectal cancer in this group. Retrospective data was extracted from patients aged 18 and who underwent curative surgery for rectal cancer. A total of 71 patients were included and divided into two groups: an elderly group (EG), comprising patients aged over 70, and a younger group (YG). Age, sex, ASA score, comorbidities, surgical treatments and post-operative morbidity and mortality were extracted from patients ‘files. The study population included 55 patients (77.46%) in the YG, and 16 patients (22.54%) in the EG. The cohort consisted of 30 men (42.3%) and 41 women (57.7%), with an average age of 57.1 ± 14.6. Comorbidities were present in 37 patients (52.1%), and 53 patients (74.6%) presented advanced-stage rectal cancer. Of the total cohort, 9 patients underwent primary surgery, while 62patients (87.3%) received neoadjuvant therapy. Laparoscopic surgery was employed in 63 patients (86.6%), with 8 cases (12.69%) of conversion to open surgery. Thirty-seven patients (52.1%) required stomas, of which 11 (16.7%) were definitive. Postoperative morbidity was higher in the EG (50%) compared to the YG (21.81%) with severe complication rate at 12.5% and 7.27%, respectively. Anastomotic leakage rate was similar in both groups (EG = 12.5% vs YG = 3.6.). However, the post-operative mortality rate was significantly higher in the EG (2.81% vs 0%). Within 90 days postoperatively, 6 readmissions were recorded, with a severe morbidity rate (Clavien-Dindo > 2) of 12.3% and a 90-day mortality of 4.2%. Curative surgery for rectal cancer in elderly patients appears to present more difficulties to manage preoperatively, and presents more postoperative mortality, requiring a tailored approach that considers the specific clinical features and functional status of this population.
Similar content being viewed by others
Introduction
Rectal cancer is a global health issue, Colorectal cancer (CRC) is the third most common cancer globally and the second leading cause of cancer death in the world, with rectal cancer accounting for one-third of these cases1. Although an increasing number of newly diagnosed colorectal cancer cases occur in individuals under 50 years old (10%)2, the median age of diagnosis remains around 70 years3. This increase coincides with a rise in life expectancy, which has grown by 5 years since 20044.
Chronologically, elderly patients start from the age of 65 years old or more5, but with increasing life expectancies, this definition now principally includes patients over 70 years old or more6. Increasing age is a well-known risk factor for several cancers, including colorectal cancer7. This segment of the population forms a heterogeneous group with different comorbidities, functional status and socio-economic aspects8. Due to population aging, as well as advancements in the control of chronic diseases and anticancer treatments, particularly in terms of survival, the proportion of elderly individuals affected by cancer or having been affected by cancer will continue to increase9,10. Additionally, this population exhibits more comorbidities (cardiovascular and endocrine diseases, other cancers, cognitive disorders, functional decline.)8,10 and tend to present more advanced stages of the disease, higher rates of comorbidities, and reduced functional status5,11 therefore practicians offered less-aggressive oncological treatment12.
The treatment of rectal cancers in elderly patients is not standardized13. Despite the fact that age is also a well known factor for perioperative morbidity and mortality12, the current guidelines do not incorporate optimal treatment recommendations for the elderly and address only partially the associated specific challenges encountered in this population13. It is also important to underline the paucity of clinical trial evidence explicitly addressing the risks and benefits of all aspects of rectal cancer in the elderly13.
This results in a wide variation and disparity in delivering a standard of care to this subset of patients3. Therefore, the decision making process for an elderly rectal cancer patient must be cautious and consider several factors such as: patient’s preferences, preoperative frailty and functional status, comorbidities, life expectancy, tumor stage, treatment toxicities and postoperative morbidity, while maintaining appropriate oncological standards and minimizing adverse effects, making understanding the impact of the surgical interventions and its outcomes in this segment of the population all the more important13.
This study aims to provide an overview of mortality, morbidity, and characteristics related to curative surgery for rectal cancer in elderly patients.
Study overview
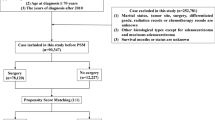
This study is a retrospective analysis derived from a prospective cancer surgery database. The surgical department specializes in general digestive surgery and has a relatively high volume of oncological procedures. The study was approved by the Mohammed V University in Rabat and all experiments were performed in accordance with relevant guidelines and regulations.
Patient population
The study included all consecutive adult patients over the age of 18 who underwent a curative intent surgical resection for rectal cancer with curative intent between January 1, 2018, and December 31, 2023.
The patient cohort was divided into two groups: an elderly group (EG), comprising patients aged over 70, and a younger group (YG), representing the remaining population. All patients included in the study were monitored for 90 days postoperatively to assess complications or mortality, with follow-up completed by March 1, 2024. There were no strict criteria for selecting the surgical approach or anastomosis type; these decisions were left to the discretion of the operating surgeon. Patients who underwent surgery with palliative intent were identified and subsequently excluded.
Definition of outcomes
The primary objectives of the study were to compare the feasibility, safety, and outcomes of oncologic rectal surgery between elderly patients and younger age groups. These comparisons focused on operative difficulty, postoperative morbidity and mortality. Morbidity and mortality were assessed using the Clavien-Dindo classification system, which categorizes postoperative complications into five grades. Grades I and II are regarded as minor complications, whereas Grades III and IV are considered severe and life-threatening. Grade V denotes patient mortality14.
Statistical analysis
Patient data were reviewed during the inclusion phase and subsequently analyzed. Qualitative variables were reported as frequencies and percentages (n,%), while quantitative variables were expressed as means and medians with interquartile ranges. A univariate analysis was performed using the chi-square test. P-values of less than 0.05 were considered statistically significant. All statistical analyses were conducted using Jamovi.
Results
The baseline characteristics of the patients are summarized in Table 1.
During the study period, 71 patients underwent surgical resection for rectal cancer. Of these, 55 patients (77.46%) were classified into the YG, and 16 patients (22.54%) were categorized into the EG.
The cohort consisted of 30 men (42.3%) and 41 women (57.7%), with an average age of 57.1 years +/- 14.6. Thirty seven patients (52.1%) had comorbidities including diabetes (13.9%), smoking (12.5%), cardiovascular disease (13.9%), a history of abdominal surgery (27.7%) and other associated medical conditions (27.8%).
The majority of tumors (83.1%) were located in the lower two-thirds of the rectum, with 53 patients (74.6%) presenting with advanced-stage rectal cancer (T3-T4; N+). All patients received surgical treatment, with 9 patients undergoing upfront surgery, and 62 patients (87.3%) receiving neoadjuvant therapy (chemoradiotherapy for 60 patients and chemotherapy for 1 patient). Laparoscopic surgery was the preferred approach, employed in 63 patients (86.6%), with 8 cases (12.69%) requiring conversion to open surgery, and 8 patients (11.3%) undergoing open surgery initially. We performed 11 Perineal Abdominal Amputation (PAA) (15.5%) (7 in the YG and 4 in the EG), 21 Partial mesorectal excision (PME) (29.6%) (17 in the YG and 4 in the EG) and 39 total mesorectal excision (TME) (54.9%) (31 in the YG and 8 in the EG). Thirty seven patients (52.1%) required stomas, of which 26 were temporary ileostomy, and 11 (16.7%) were definitive colostomy with one pseudo continent perineal colostomy.
The overall morbidity rate was 28.16%, with 6 patients (8.5%) experiencing major complications (Clavien-Dindo grade > 2). Surgical site infections occurred in 5 patients (6.94%), while postoperative hemorrhage and deep collections were each observed in 3 patients (4.22%). The anastomotic fistula rate was 6.66%, affecting 4 patients. Revision surgery was required in 5 patients (6.94%). The mortality rate was 2.8% (2 patients). Within 90 days postoperatively, 6 readmissions were recorded, with a severe morbidity rate (Clavien-Dindo grade > 2) of 12.3% and a 90-day mortality of 4.2%.
Both the elderly and the younger group were well balanced in terms of sex (P = 0.89). The EG had a higher percentage of patients with comorbidities (EG = 68.75% vs. YG = 47.27%, P = 0.13). The rates of anemia and hypoalbuminemia were similar in both groups (Hb < 10 g/dl: YG = 27.27%, EG = 31.25%, P = 0.756) (hypoalbuminemia: YG = 3.63%, EG = 6.25%, P = 0.661). Low and mid rectal cancer locations were similar in both groups, EG (68.75% vs. 87.27% P = 0.164), and most diagnosis were made at an advanced stage, with 56.25% for EG and 80% for YG, P = 0.115. Neoadjuvant treatment was administered to the majority of patients, with 89.09% in the YG and 81.25% in the EG receiving it (P = 0.857). All patients subsequently underwent surgery, with the laparoscopic approach being favored in both groups (EG = 87.5%, YG = 89.09%, P = 0.973). Two conversions were required in the EG, compared to six in the YG (EG = 12.5%, YG = 10.71%, P = 0.973). More than half of the patients in both groups required a stoma (EG = 56.25, %YG = 50.9%,, P = 0.707), with 25% of them being permanent in the YG (of which one pcc) compared to 44.44% in the EG.
Despite the slightly high morbidity rate (50% EG vs. 21.81% YG), severe complication rate (12.5% EG vs. 7.27% YG) and surgical re-interventions (12.5% EG vs. 5.54% YG) in the EG, results were not statistically significant (P = 0.07; P = 0.379). Anastomotic leakage rate was also similar in both groups (EG = 12.5% vs. YG = 3.6%, P = 0.121). However, the post operative mortality rate was significantly higher in the EG (2.81%% vs. 0% p = 0.008).
At the 3-month follow-up, all readmissions (6 Patients; 8.45%) were observed in the YG group. Furthermore, 90 days’ severe complications rate was similar in both groups (12.5% vs. 10.9%; P = 0537). Lastly, the 90 days’ mortality rate was similar in both groups (2.81% in EG vs. 1.4% in YG; P = 0.06).
Discussion
Chronologically, elderly patients start from the age of 65 years old or more5, but with increasing life expectancies, this definition now includes patients over 70 years old or more6. This segment of the population forms a heterogeneous group with different comorbidities, functional status and socio economic aspects8. In Morocco, life expectancy at birth was estimated at 76.8 years in 2022, with women having a higher life expectancy of 78.6 years compared to 75.2 years for men (which represents an increase in life expectancy of 5 years compared to 2004)4.
CRC is the third most prevalent cancer worldwide1. In our setting, its incidence is on the rise, as demonstrated by the increase in cases in the economic capital region, where rates have grown from 7.3 per 100,000 inhabitants during the period 2004–2007 to 9 per 100,000 inhabitants in 2013–2017 for both sexes9,15. In 2020, a total of 4558 new cases of colorectal cancer were registered, accounting for approximately 7.7% of all new cancer diagnoses nationwide11.
Despite an increasing incidence among younger individuals, the median age at diagnosis remains approximately 70 years3. The literature indicates that the Moroccan population most affected by this cancer comprises individuals aged over 70 in 2017 (Fig. 1)16.
Distribution of incident cases of colorectal cancer by age for the period in Morocco from 2013 to 2017 (Blue = Men, Pink = Women)19.
This age group also presents increased comorbidities, including cardiovascular disease, diabetes, higher ASA scores, and malnourishment8. Our cohort demonstrated these trends, with a higher prevalence of patients with comorbidities (EG = 68.75% vs. YG = 47.27%) and malnutrition, as reflected by hypoalbuminemia rates (YG = 3.63%, EG = 6.25%). Furthemore, an advanced TNM classification (stage III or IV) is often associated with higher postoperative morbidity and decreased overall survival, making it mandatory to adapt therapeutic strategies based on TNM staging to optimizing outcomes and minimizing postoperative complications17. Notably, our findings indicate a greater proportion of patients with advanced colorectal cancer (CRC) within the younger group (80% in YG vs. 56.25% in EG). This trend can be attributed to the demographic composition of our study population, wherein individuals aged 65 years and older represented only 6.95%, while those aged 15–64 years constituted 67.57%18. In contrast, existing literature suggests that elderly patients tend to present with more advanced stages of CRC19, which may be explained by age-related differences in seeking medical attention, symptom recognition, or referral patterns to primary care20.
This observation suggests that colorectal cancer screening in elderly individuals for earlier diagnosis could be advantageous21. However, the relevance of such screening in this demographic has not yet been proven, and there is a scarcity of data on its impact21. In our context, no organized national colorectal cancer screening program has been implemented16. In France, organized screening systematically ceases at age 7522. Beyond this age, tests are no longer centrally analyzed, and only patients at high risk of colorectal cancer or with a history of adenoma continue to be monitored by colonoscopy22. The low life expectancy of this population raises doubts about the value of such screening. Furthermore, excessive screening can cause anxiety, discomfort, and expose individuals to complications specific to screening techniques, which are more frequent in the elderly21.
A more complex risk of screening is overdiagnosis, defined as the diagnosis of a disease that would not have been detected in the absence of screening during the individual’s lifetime and would have no impact on their life expectancy. This can lead to overtreatment without any benefit to their health or life23. It is interesting to report however, the observation of Van Harten et al.24 who noted a decreased proportion of elderly patients by almost 25%, from 45.3% in 2007 to 20.4% in 2018. He suggests that this observation could be attributed, among other factors, to the increased adaptation to the Australian National Bowel Cancer Screening Program and improved general public education, potentially leading to an earlier cancer detection24.
Only 53–90% of elderly patients with colorectal cancer are treated according to national guidelines, with the most commonly reported reasons for treatment adjustments being patient preference (27%) and functional status (20%)25. In general, a patient’s status is initially assessed based on their general condition and nutritional status. For example, obesity has a complex relationship with CRC prognosis, with a high Body Mass index often associated with poorer prognosis and higher recurrence rates26.
While chronological age alone is not a reliable predictor of treatment-related outcomes and toxicities, elderly patients frequently present with significant physiological impairments and extensive medical histories8. For instance, thyroid disorders, particularly hypothyroidism, have been shown to negatively impact wound healing and contribute to increased postoperative morbidity27. Additionally, prolonged corticosteroid use is associated with an elevated risk of infections and delayed tissue repair, further complicating postoperative recovery17.
Consequently, several easily integrated screening tools have been proposed, including the G8 score, Mini Nutritional Assessment (MNA), Mini-Cog, and Adult Comorbidity Evaluation-27 (ACE-27)8. These tools classify elderly patients into three categories: fit, vulnerable, and frail (Fig. 2). “Fit patients” receive standard care, “vulnerable patients” require tailored treatment approaches, and “frail patients” are suitable only for supportive care28. A particularly noteworthy prognostic tool is the Onco-MPI, an easily calculated prognostic score designed to predict cancer-specific mortality. It has proven effective in forecasting 1-year mortality in older cancer patients, thereby aiding in clinical decision-making. However, its validation in more diverse and longer-term settings is still needed29. Additionally, emerging biomarkers such as butyrylcholinesterase have been proposed as predictive indicators of postoperative complications. A decrease in this serum enzyme has been associated with an increased risk of complications, including infections and postoperative organ failure, which could allow for better preoperative stratification of high-risk patients30.
Treatment algorithm for locally advanced rectal cancer in elderly patients. c: clinical; T: tumor; N: node; MNA: mini-nutritional assessment; ACE-27, adult comorbidity evaluation-27; RT: radiation therapy; Gy: Gray; fr: fraction; MRF: mesorectal6.
The multidisciplinary approach has significantly improved the surgical outcomes and prognosis of rectal cancer over the past two decades. The combination of neoadjuvant treatment and surgery has increased the rates of conservative surgery, reduced local recurrence rates, and prolonged survival compared to surgery alone31. However, several studies have shown that elderly patients can experience poor physical condition following neoadjuvant treatment32,33. These treatments are associated with grade 3–4 acute toxicity (27%, with 12% of diarrhea ) and long-term toxicity (14%: with 4% of strictures, 2% of bladder problems, and 9% of gastrointestinal effects), making them intolerable for older individuals34. For patients who can tolerate neoadjuvant treatment, Wu et al. found that delaying the surgery by more than 4 weeks after neoadjuvant treatment may benefit elderly patients35. This delay allows for recovery from acute radiation toxicity, improvement in overall health and treatment of comorbidities (control blood pressure and diabetes, optimizing nutritional support)35. This delay also favors better complete oncological response, and fewer postoperative complications compared to immediate surgery35.
The neoadjuvant treatments can be followed by TME or PME36. Open surgery can still be necessary for patients with advanced stages of the disease but includes a higher rate of permanent stomas and more postoperative complications24. Laparoscopic surgery represents a significant advancement in the surgical management of colorectal cancer and studies have that advanced age only should not be a contraindication to perform mini invasive approach, as their outcomes are at least equivalent, if not better, compared to an open approach12,22,37,38, with reduced analgesic intake, shorter hospital stays, and a faster return to activity, with morbidity and mortality comparable to those undergoing open surgery and a 5 year survival rates going up to 49%12,28,39. This leads to an increasing number of teams preferring the laparoscopic approach for colorectal cancer in this demographic (including ours with 81.81% of laparoscopic surgeries in elderly patients).
Surgery may be associated with significant morbidity, mortality, and functional consequences36. Notable disturbances in urinary and sexual function have been documented, but the most critical consequences are related to bowel function post-surgery36. Depending on the tumor’s location in the rectum, the anastomosis level, and the necessity for partial or total intersphincteric resection, patients may experience varying degrees of symptoms associated with low anterior resection syndrome36. It is important to note that many patients undergoing a low anastomosis may require a temporary stoma, which can ultimately become permanent in cases of failed anastomosis40 which is shown in our study where more than half of the patients in both groups required a stoma (EG = 56.25. %YG = 50.9%,, P = 0.707), with 25% of them being permanent in the YG (of which one pcc) compared to 44.44% in the EG. A stoma can significantly disrupt the psychological and social well-being of elderly patients, often resulting in psychic shock, a sense of disability, activity restrictions, reduced socialization, loss of control and intimacy, and feelings of malaise. Patients may experience a sense of being dirty, disgust, and a lack of acceptance41. Consequently, the requirement for a temporary or permanent stoma is a significant concern for many patients, with the avoidance of a stoma often being a primary expectation from rectal cancer treatment36.
In this study, postoperative mortality and morbidity rates were analyzed for both the YG and EG, along with postoperative outcomes. In both groups, the rates of morbidity, severe complications, anastomotic leakage and surgical re-interventions were slightly higher in the EG but without reaching statistical significance. This finding aligns with the work of Devoto et al., which reported an overall complication rate for elderly patients of 35.6% compared to 31.2% in younger patients, though this difference was also not statistically significant42. Earlier studies have suggested that the frequency of postoperative morbidity increases progressively with age 20. Stephan et al. supported this view, finding significantly higher rates of overall complications in the elderly group (p < 0.01), with both studies defining elderly as patients aged 85 years or older43.
These results have led some surgeons to reconsider immediate resection and its associated complications for patients who achieve a complete oncological response following neoadjuvant treatment, which occurs in 10–25% of cases44. Instead, an organ preservation strategy known as “Watch and Wait” may be proposed45. However, since the achievement of a complete response remains uncertain, W&W should be regarded as a cautious approach rather than a definitive treatment strategy36.
Our results also revealed a mortality rate of 2.81% in the EG group, which is significantly lower than the rates reported in the literature, ranging from 6.8 to 14.1%46. However, this rate remains higher than the 0% mortality observed in the YG group.
Consistent with our findings, previous studies have demonstrated that elderly patients experience elevated postoperative mortality rates, underscoring the critical importance of the immediate postoperative period47. Kessler et al. observed an increase in mortality from 0.5 to 13% in patients over the age of 8048. Conversely, Yang et al., in their meta-analysis, found no statistically significant difference in mortality rates between elderly subjects (20.2%) and younger subjects (25.2%)42.
In addition to the risks associated with the surgical procedure itself, mortality in these patients may be due to other complications associated with the terrain, such as acute renal failure, cardiac complications, respiratory failure, urinary tract infection and pneumonia were more commonly present in elderly people49. This high mortality justifies a patient-centered treatment approach, incorporating a thorough preoperative evaluation of comorbidities and nutritional status8,26 and also including elderly patients with rectal cancer in the decision making if they express their desire to be involved in the process13.
A promising innovation in this field is the Internet of Things (IoT), an interconnected network of physical objects, machines, and other devices that enables the exchange of data to support intelligent applications and services50. The integration of IoT in the management of CRC represents a major advancement in connected medicine. By enabling continuous patient monitoring through wearable devices and implantable sensors, IOT allows real-time tracking of vital parameters, physical activity, and treatment adherence. This approach enhances the early detection of postoperative complications and optimizes the personalization of therapeutic plans50.
Furthermore, the use of artificial intelligence and deep learning in the analysis of histopathological images of CRC improves diagnostic accuracy. Recent studies have demonstrated that these algorithms can reliably differentiate cancerous tissues from healthy tissues and stratify patients based on tumor risk, thereby facilitating therapeutic decision-making51,52.
This study has several limitations. It is a single-center study, which may reduce the external validity of the data. The retrospective collection of data from patients’ medical records is another limitation, as the accuracy of the information depends heavily on the precision of these records. Additionally, the size of the cohort is relatively small, compared to foreign centers. The study period is also limited. An extension of this investigation is planned to further analyze the morbidity and mortality associated with each surgical procedure.
The strengths of this study stem from its single-center design, allowing the examination of recent consecutive cases with detailed documentation of complications and relatively consistent management strategies. This study is the first within our context to evaluate rectal cancer surgery in elderly patients. The evaluation considered different surgical approaches and the specific characteristics of this patient group to propose recommendations for the safe, effective, and efficient management of rectal cancers in the elderly.
Conclusion
Curative rectal cancer surgery in elderly patients appears to present more difficulties to manage preoperatively, and more mortality postoperatively, requiring a tailored approach that takes into account the unique clinical features, comorbidities and functional status of this population. Further research is needed to better understand this population and refine treatment strategies to meet their specific needs.
Data availability
The data that support the findings of this article are available from the corresponding author upon reasonable request.
References
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 68 (1), 7–30. https://doi.org/10.3322/caac.21442 (2018).
Venugopal, A. & Stoffel, E. M. Colorectal Cancer in young adults. Curr. Treat. Options Gastro. 17, 89–98. https://doi.org/10.1007/s11938-019-00219-4 (2019).
Huang, C. K., Shih, C. H. & Kao, Y. S. Elderly rectal cancer: An updated review. Curr. Oncol. Rep. 26, 181–190 (2024).
RCRC. Registre des Cancers de la Région de Grand Casablanca, données 2008–2012. Edition 2016.
Papamichael, D. et al. Treatment of the elderly colorectal cancer patient: SIOG expert recommendations. Ann. Oncol. 20 (1), 5–16. https://doi.org/10.1093/annonc/mdn532 (2009).
Papamichael, D. et al. Treatment of colorectal cancer in older patients: International society of geriatric oncology (SIOG) consensus recommendations 2013. Ann. Oncol. 26 (3), 463–476. https://doi.org/10.1093/annonc/mdu253 (2015).
Nutter, D. W. M. The AAMC Project on the Clinical Education of Medical Students (Association of American Medical Colleges, 2002).
De Felice, F. et al. Locally advanced rectal cancer: Treatment approach in elderly patients. Curr. Treat. Options Oncol. 21 (1), 1. https://doi.org/10.1007/s11864-019-0692-8 (2020).
Australian Institute of Health and Welfare. Deaths in Australia. Life expectancy & death. [Cited 23 May 2020.] (2019). https://www.aihw.gov.au/reports/life-expectancy-death/deaths-in-australia/contents/life-expectancy
Galvin, A., Bertrand, N., Boulahssass, R., De Decker, L., Dorval, É., Clairaz, B., Castaignede, M., Mourey, L., Baldini, C., Bauvin, E. & Jacques, B. Repenser la prise en charge des sujets âgés atteints d’un cancer: propositions du groupe Priorités Âge Cancer. Bulletin du Cancer 109 (6), 714–721. https://doi.org/10.1016/j.bulcan.2022.03.013 (2022).
El Badisy, I. et al. Risk factors affecting patients survival with colorectal cancer in Morocco: Survival analysis using an interpretable machine learning approach. Res Sq [Preprint]. rs.3.rs-2435106. https://doi.org/10.21203/rs.3.rs-2435106/v1 (2023). Update in: Sci. Rep. 14 (1), 3556. https://doi.org/10.1038/s41598-024-51304-3 (2024).
Yap, R. et al. Factors affecting the post-operative outcomes in patients aged over 80 following colorectal cancer surgery. Int. J. Colorectal Dis. 38 (1), 11. https://doi.org/10.1007/s00384-022-04291-8 (2023).
Podda, M. et al. Multidisciplinary management of elderly patients with rectal cancer: Recommendations from the SICG (Italian society of geriatric Surgery), SIFIPAC (Italian society of Surgical Pathophysiology), SICE (Italian society of Endoscopic Surgery and new technologies), and the WSES (World society of Emergency Surgery) International Consensus Project. World J. Emerg. Surg. 16, 35. https://doi.org/10.1186/s13017-021-00378-9 (2021).
Dindo, D., Demartines, N. & Clavien, P. A. Classification of surgical complications: A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann. Surg. 240, 205–213 (2004).
Lahoucine Amsdar, A. et al. Risk factors for colorectal cancer in Morocco: A systematic review, clinical epidemiology and global health, 28, 101661. (2024). https://doi.org/10.1016/j.cegh.2024.101661
RCRC. Registre des Cancers de la Région de Grand Casablanca, données 2013–2017.
Wang, A. S., Armstrong, E. J. & Armstrong, A. W. Corticosteroids and wound healing: Clinical considerations in the perioperative period. Am. J. Surg. 206 (3), 410–417. https://doi.org/10.1016/j.amjsurg.2012.11.018 (2013).
« Population Morocco - evolution population. Morocco - Pyramide des âges - age median - demographie - chiffres », sur en.humandatas.com.
Ahmed, S., Howel, D., Debrah, S. & NORCCAG (Northern Region Colorectal Cancer Audit Group). The influence of age on the outcome of treatment of elderly patients with colorectal cancer. J. Geriatr. Oncol. 5 (2), 133–140. https://doi.org/10.1016/j.jgo.2013.12.005 (2014).
Surgery for colorectal. cancer in elderly patients: A systematic review. Colorectal Cancer Collab. Group. Lancet. 356 (9234), 968–974 (2000).
Guittet, L. et al. Should we screen for colorectal cancer in people aged 75 and over? A systematic review - collaborative work of the French geriatric oncology society (SOFOG) and the French federation of digestive oncology (FFCD). BMC Cancer. 23, 17. https://doi.org/10.1186/s12885-022-10418-5 (2023).
Maratt, J. K. & Calderwood, A. H. Colorectal Cancer screening andsurveillance colonoscopy in older adults. Curr. Treat. Options Gastroenterol. 17 (2), 292–302. https://doi.org/10.1007/s11938-019-00230-9 (2019).
Welch, H. G. & Black, W. C. Overdiagnosis in cancer. J. Natl. Cancer Inst. 102 (9), 605–613. https://doi.org/10.1093/jnci/djq099 (2010).
van Harten, M. J. et al. Minimally invasive surgery in elderly patients with rectal cancer: An analysis of the Bi-National colorectal Cancer audit (BCCA). Eur. J. Surg. Oncol. 46 (9), 1649–1655. https://doi.org/10.1016/j.ejso.2020.03.224 (2020).
Scheepers, E. R. M., Schiphorst, A. H., van Huis-Tanja, L. H., Emmelot-Vonk, M. H. & Hamaker, M. E. Treatment patterns and primary reasons for adjusted treatment in older and younger patients with stage II or III colorectal cancer. Eur. J. Surg. Oncol. S0748-7983(21):00059-7. https://doi.org/10.1016/j.ejso.2021.01.029 (2021).
Zhao, F. Q., Jiang, Y. J., Xing, W., Pei, W. & Liang, J. W. The safety and prognosis of radical surgery in colorectal cancer patients over 80 years old. BMC Surg. 23 (1), 45. https://doi.org/10.1186/s12893-023-01938-3 (2023). Erratum in: BMC Surg.;23 (1), 72. https://doi.org/10.1186/s12893-023-01965-0 (2023).
Rosko, A. J. et al. Hypothyroidism and wound healing after salvage laryngectomy. Ann. Surg. Oncol. 25 (5), 1288–1295. https://doi.org/10.1245/s10434-017-6278-4 (2018).
Hathout, L. et al. Management of locally advanced rectal cancer in the elderly: A critical review and algorithm. J. Gastrointest. Oncol. 9 (2), 363–376. https://doi.org/10.21037/jgo.2017.10.10 (2018).
Brunello, A. et al. Development of an oncological-multidimensional prognostic index (Onco-MPI) for mortality prediction in older cancer patients. J. Cancer Res. Clin. Oncol. 142 (5), 1069–1077. https://doi.org/10.1007/s00432-015-2088-x (2016).
Francesk, et al. Butyrylcholinesterase (BChE) levels correlate with surgical site infection risk and severity after colorectal surgery mulita. Eur. J. Surg. Oncol. 50, 109427. https://doi.org/10.1016/j.ejso.2024.109427
Dodaro, C. A., Calogero, A., Tammaro, V., Pellegrino, T., Lionetti, R., Campanile, S., Menkulazi, M., Ciccozzi, M., Iannicelli, A. M., Giallauria, F. & Sagnelli, C. Colorectal cancer in the elderly patient: the role of neo-adjuvant therapy. Open Med. 14 (1), 607–612. https://doi.org/10.1515/med-2019-0068 (2019).
Liu, W. Y. et al. Preoperative chemoradiotherapy in older patients with rectal cancer guided by comprehensive geriatric assessment within a multidisciplinary team-a multicenter phase II trial. BMC Geriatr. 24 (1), 442. https://doi.org/10.1186/s12877-024-05046-6 (2024).
François, E. et al. Comparison of short course radiotherapy with chemoradiotherapy for locally advanced rectal cancers in the elderly: A multicentre, randomised, non-blinded, phase 3 trial. Eur. J. Cancer. 180, 62–70. https://doi.org/10.1016/j.ejca.2022.11.020 (2023).
O’Donnell, C. D. J., Hubbard, J. & Jin, Z. Updates on the management of colorectal Cancer in older adults. Cancers (Basel). 16 (10), 1820. https://doi.org/10.3390/cancers16101820 (2024).
Wu, H. et al. Short-course radiotherapy with immediate or delayed surgery in rectal cancer: A metaanalysis. Int. J. Surg. 56, 195–202. https://doi.org/10.1016/j.ijsu.2018.05.031 (2018).
Cerdan-Santacruz, C. et al. Watch and wait approach for rectal Cancer. J. Clin. Med. 12 (8), 2873. https://doi.org/10.3390/jcm12082873 (2023).
Roscio, F. et al. Is laparoscopic surgery really effective for the treatment of colon and rectal cancer in very elderly over 80 years old? A prospective multicentric case-control assessment. Surg. Endosc. 30, 4372e82. https://doi.org/10.1007/s00464-016-4755-7 (2016).
Landi, F. et al. Morbidity after laparoscopic and open rectal cancer surgery: A comparative analysis of morbidity in octogenarians and younger patients. Colorectal Dis. 18, 459e67. https://doi.org/10.1111/codi.13136 (2016).
van Leeuwen, B. L., Pahlman, L., Gunnarsson, U., Sjovall, A. & Martling, A. The effect of age and gender on outcome after treatment for colon carcinoma. A population-based study in the Uppsala and Stockholm region. Crit. Rev. Oncol. Hematol. 67, 229–236 (2008).
Back, E. et al. Permanent stoma rates after anterior resection for rectal cancer: Risk prediction scoring using preoperative variables. Br. J. Surg. 108 (11), 1388–1395. https://doi.org/10.1093/bjs/znab260 (2021).
Sounga, D. et al. Vécu psychologique de la stomie digestive chez le sujet âgé: évaluation de l’anxiété, de l’image du corps et de la perception de la maladie,NPG Neurologie - Psychiatrie - Gériatrie 23 (136), 280–287 https://doi.org/10.1016/j.npg.2023.02.004 (2023).
Devoto, L. et al. Colorectal cancer surgery in the very elderly patient: A systematic review of laparoscopic versus open colorectal resection. Int. J. Colorectal Dis. 32, 1237–1242. https://doi.org/10.1007/s00384-017-2848-y (2017).
Stępień, R., Głuszek, S., Kozieł, D. & Kaczmarczyk, M. The risk of surgical treatment in patients aged 85+, with special consideration of colorectal cancer. Pol. Przegl Chir. 86 (3), 132–140. https://doi.org/10.2478/pjs-2014-0025 (2014).
Capirci, C. et al. Prognostic value of pathologic complete response after neoadjuvant therapy in locally advanced rectal cancer: Long-term analysis of 566 YpCR patients. Int. J. Radiat. Oncol. Biol. Phys. 72 (1), 99–107. https://doi.org/10.1016/j.ijrobp.2007.12.019 (2008).
Habr-Gama, A. et al. Increasing the rates of complete response to neoadjuvant chemoradiotherapy for distal rectal cancer: Results of a prospective study using additional chemotherapy during the resting period. Dis. Colon Rectum. 52 (12), 1927–1934. https://doi.org/10.1007/DCR.0b013e3181ba14ed (2009).
Park, H., Parys, S., Tan, J., Entriken, F. & Hodder, R. Post-operative outcomes in the elderly following colorectal cancer surgery. ANZ J. Surg. 91 (3), 387–391. https://doi.org/10.1111/ans.16394 (2021).
Normann, M. et al. Mortality and morbidity after colorectal cancer resection surgery in elderly patients: A retrospective population-based study in Sweden. World J. Surg. Oncol. 22, 23. https://doi.org/10.1186/s12957-024-03316-6 (2024).
Kessler, H., Hermanek, P. Jr & Wiebelt, H. Operative mortality in carcinoma of the rectum. Results of the German multicentre study. Int. J. Colorectal Dis. 8 (3), 158–166. https://doi.org/10.1007/BF00341191 (1993).
Yang, R. et al. Laparoscopic surgery after neoadjuvant therapy in elderly patients with rectal cancer. J. BUON. 22 (4), 869–874 (2017).
Muhsen, I. N. et al. Current status and future perspectives on the internet of things in oncology. Hematol. Oncol. Stem Cell Ther. 16 (2), 102–109. https://doi.org/10.1016/j.hemonc.2021.09.003 (2023).
Bousis, D. et al. The role of deep learning in diagnosing colorectal cancer. Prz Gastroenterol. 18, 266–273. https://doi.org/10.5114/pg.2023.129494 (2023).
Chlorogiannis, D. D. et al. Tissue classification and diagnosis of colorectal cancer histopathology images using deep learning algorithms. Is the time ripe for clinical practice implementation? Prz Gastroenterol. https://doi.org/10.5114/pg.2023.130337 (2023).
Funding
The authors have no source of funding or financial support except themselves.
Author information
Authors and Affiliations
Contributions
AH designed the study, YA, SEF and HS performed statistical analysis and participated in drafting the manuscript. YA, HS, and JEL collected the data, YA, HS, AM wrote the first draft of the manuscript. AH participated in the study design and critically reviewed the manuscript. BY, RM, MMA, FS and AH critically reviewed the manuscript. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent
Informed consent was obtained from all subjects and/or their legal guardian(s). The study was approved by the Ethics committee of the Mohammed V University in Rabat.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Achour, Y., Sekkat, H., Moufid, A. et al. Retrospective analysis of curative rectal cancer surgery outcomes in elderly patients. Sci Rep 15, 19917 (2025). https://doi.org/10.1038/s41598-025-91088-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-91088-8
Keywords
This article is cited by
-
Preoperative social frailty and short-term postoperative outcomes in gastrointestinal cancer surgery: a multicentre prospective cohort study in China
World Journal of Surgical Oncology (2026)