Abstract
Low lean body mass increases fall risk. Some diabetes medications, specifically SGLT2 inhibitors and GLP-1RAs, can cause muscle and body mass loss. This study assessed their association on falls in type 2 diabetes patients. An annual fall survey was conducted for up to 5 years on individuals with type 2 diabetes admitted to our department. Fall risk factors were identified using discrete-time survival analysis. The study observed 471 participants over a median period of 2 years. The participants had a median age of 64 years, with a fall incidence rate of 17.1 per 100 person-years. Independent fall predictors identified were fall history, SGLT2 inhibitor use, and age. The odds ratios (95% confidence intervals) for using SGLT2 inhibitors only, GLP-1RAs only, and both combined were 1.80 (1.10–2.92), 1.61 (0.88–2.84), and 2.89 (1.27–6.56), respectively. SGLT2 inhibitor use was an independent risk factor for falls, while GLP-1RAs’ effects were not statistically significant. However, the combined use of SGLT2 inhibitors and GLP-1RAs significantly increased the risk of falls. Therefore, it is important to consider this risk when prescribing these medications to people with type 2 diabetes.
Similar content being viewed by others
Introduction
Falls and related injuries can occur during walking or other physical activities at any age and are a significant health and socioeconomic burden, causing distress, physical disability and other medical conditions, hospitalization, institutionalization, and even death1,2. In particular, ~ 17 million person-years of life were lost because of falls in 20171, and fall-related costs account for ~ 1% of healthcare spending in developed countries3. Major fall risk factors include a history of falls, balance and visual impairment, muscle weakness, certain medications, gait problems, cognitive decline, and diabetes4,5.
Elderly individuals with diabetes are 1.5–3 times more likely to experience falls than those without diabetes5. Previously reported risk factors for falls in persons with diabetes include diabetic neuropathy, diabetic retinopathy, elevated cystatin C levels, insulin or sulfonylurea use, and decreased grip strength, knee extension, and ankle dorsiflexor muscle strength6,7,8,9,10. A prospective cohort study on risk factors for falls in persons with type 2 diabetes revealed a positive correlation between severe hypoglycemia and fall prevalence (hazard ratio = 2.23) and suggested that the increased fall incidence related to insulin or sulfonylureas use can be attributed to the ability of these medications to induce hypoglycemia11. Previously, we used machine learning to identify significant fall risk factors in functionally independent elderly and nonelderly individuals with type 2 diabetes as grip strength, fasting serum C-peptide level, knee extension muscle strength, ankle dorsiflexor muscle strength, and proliferative diabetic retinopathy; however, insulin and sulfonylurea treatments were not identified as significant fall risk factors12. The emergence of numerous blood glucose–lowering medications has led to a decrease in the use of sulfonylureas and an increase in the use of dipeptidyl peptidase-4 inhibitors, glucagon-like peptide-1 receptor agonists (GLP-1RAs), and sodium-glucose cotransporter 2 (SGLT2) inhibitors in combination therapies13. Compared with insulin therapy alone, insulin therapy coupled with the use of these medications was reported to achieve better blood glucose control without increasing the risk of hypoglycemia14,15,16. The coadministration of basal insulin with oral glucose-lowering medications or GLP-1RAs poses a lower risk of hypoglycemia than basal-bolus therapy while retaining the efficiency of the latter in managing blood glucose levels17,18. Given the decrease in the risk of hypoglycemia with the advances in diabetes pharmacotherapy, insulin and sulfonylurea use may no more be an important fall risk factor. However, GLP-1RAs and SGLT2 inhibitors are known to reduce body weight, affecting both fat and lean body masses19, and may therefore lead to muscle weakness and increase the risk of falls. To examine the hitherto unexplored correlation between the administration of these drugs and fall incidence, we herein examined the effects of GLP-1RAs and SGLT2 inhibitors on falls in elderly and nonelderly persons with type 2 diabetes, and assessed the corresponding fall risks.
Results
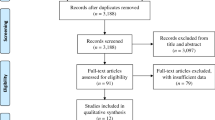
In total, 678 persons were enrolled in the study. Of these, 471 were included in the analysis after excluding participants that did not return questionnaires or provide data on weight at the first year postdischarge (Fig. 1). The participants (272 males and 199 females: 471 in total) had a median age of 63 (51–71) years. The median follow-up period was 2 (1–3) years, corresponding to a total of 1,013 person-years of observation. The number of individuals who reported at least one fall after discharge was 173, corresponding to a fall incidence rate (number of falls/total person-years of observation) of 17.1 per 100 person-years. Fifteen individuals experienced fractures due to falls. The follow-up rates of the fall survey (respondents/survey recipients) at the first, second, third, fourth, and fifth years after discharge were 69% (471/678), 55% (265/482), 44% (161/367), 39% (107/274), and 29% (62/192), respectively. The corresponding follow-up rates for the weight survey were 69% (465/678), 53% (256/482), 41% (152/367), 38% (104/274), and 29% (56/192), respectively. The changes in body weight in the first, second, third, fourth, and fifth years after discharge were − 0.7 (− 3.2 to 1.7), 0.2 (− 3.0 to 3.1), 0.0 (− 3.2 to 2.5), − 0.3 (− 4.0 to 2.9), and − 0.2 (− 3.6 to 3.5) kg, respectively.
Flowchart of selection of study participants.
Tables 1 and 2 present the characteristics of participants who responded to the survey one 1 year after discharge. Among the participants taking GLP-1RAs, 42 were on dulaglutide, 20 were on liraglutide, 6 were on semaglutide, 3 were on exenatide extended-release (exenatide XR), and 3 were on lixisenatide. To examine the risk factors for postdischarge falls, we carried out discrete-time survival analysis using fall history at admission, gender, age, height, BMI, grip strength, fasting serum CPR level, knee extension muscle strength, ankle dorsiflexion muscle strength, presence of proliferative retinopathy, glucose-lowering drugs used at discharge, and postdischarge weight change as covariates. Fall history at admission, SGLT2 inhibitor intake, and age were identified as independent predictors for falls in all models, and gender was additionally identified as such for Model 1. In all models, GLP-1RA intake was not a significant risk factor, and no significant interaction between SGLT2 inhibitor and GLP-1RA intakes was observed. The C-indices of Models 1, 2, 3, and 4 were 0.682, 0.606, 0.593, and 0.550. The odds ratios (95% confidence intervals) for falls in the best-performing model (Model 1) were as follows: Fall history = 2.26 (1.57–3.26), SGLT2 inhibitor intake = 1.80 (1.10–2.92), gender (female) = 1.73 (1.03–2.89), age = 1.02 (1.01–1.04), and GLP-1RA intake = 1.61 (0.88–2.84) (Table 3).
Figure 2A,B show the unadjusted and adjusted (based on Model 1) survival curves, respectively, for four groups, namely SGLT2 inhibitors only, GLP-1RAs only, SGLT2 inhibitors + GLP-1RAs, and neither of the two types of drugs. The odds ratio for falls in the case of combined drug use, i.e., 2.89 (1.27–6.56) significantly exceeded that observed for the use of neither drug.
Five-year (A) nonadjusted and (B) Model 1–adjusted survival curves for four groups, namely sodium-glucose cotransporter 2 (SGLT2) inhibitors only, glucagon-like peptide-1 receptor agonists (GLP-1RAs) only, SGLT2 inhibitors + GLP-1RAs, and neither of the two types of drug.
Discussion
A prospective survey on falls in persons with type 2 diabetes was conducted at 1-year intervals for up to 5 years after discharge to investigate the association of SGLT2 inhibitor and GLP-1RA intake on fall incidence. The intake of SGLT2 inhibitors was identified as an independent risk factor for falls, and the combined intake of SGLT2 inhibitors and GLP-1RAs resulted in a higher fall incidence rate than the use of either drug type on its own. The association between SGLT2 inhibitor use and falls remained an independent risk factor for falls even after adjusting for known risk factors for falls, such as history of falls, skeletal muscle mass, muscle strength, and retinopathy. This finding may provide important implications for the treatment strategy of diabetes in the elderly.
SGLT2 inhibitors induce weight loss through energy loss due to urinary glucose excretion, leading to a reduction in body fat, lean body, and skeletal muscle masses19. Decreases in skeletal muscle mass and strength are associated with falls12,20. In addition, SGLT2 inhibitors increase urine volume through osmotic diuresis, thus decreasing fluid volume and increasing urinary frequency21,22. Frequent urination, especially nocturia, increases the risk of falls22. A retrospective study showed that 19% of participants who started taking SGLT2 inhibitors discontinued their intake because of frequent urination23. Fluid volume depletion is a risk factor for (orthostatic) hypotension24, thus increasing the risk of falls25,26. The odds ratio for orthostatic hypotension due to SGLT2 inhibitor intake was reported as 1.24 (95% confidence interval: 1.08–1.43) compared with the control group27. Thus, the increased risk of falls due to the intake of SGLT2 inhibitors was ascribed to the concomitant decrease in skeletal muscle mass, increase in urinary frequency, and decrease in blood volume.
GLP-1RA intake was not identified as a significant fall predictor, possibly because most participants used GLP-1RAs with a low weight-reducing effect, such as dulaglutide, liraglutide, exenatide XR, and lixisenatide, which did not strongly influence the risk of falls. Only seven participants used semaglutide, which has a strong weight-reducing effect. However, no interaction between SGLT2 inhibitor and GLP-1RA intakes was observed. The odds ratio of falls observed for the combined use of these drugs exceeded those observed for each drug individually. Given that the same trend was previously reported for weight loss28, the increased risk of falls observed for the combined use of the above drugs may be due to the concomitant increase in muscle weakness. Semaglutide and tirzepatide have strong weight loss effects and may further increase the risk of falls, which is a topic of future investigations. Herein, no significant relationship was observed between the occurrence of falls and weight changes. As the current study did not specify the timing of weight measurements, e.g., time of day or before/after meals, and relied on self-reported values, the weight changes could be inaccurate.
SGLT2 inhibitors and GLP-1RAs are recommended and often administered together in persons with type 2 diabetes to prevent cardiovascular and renal events, and promote weight loss29. The increased risk of falls associated with the intake of SGLT2 inhibitors alone, and in combination with GLP-1RAs, suggests that caution should be exercised when these drugs are administered to sarcopenia-affected persons with type 2 diabetes. However, the administration of dapagliflozin to patients with heart failure or chronic kidney disease was reported to be beneficial regardless of frailty30,31. In this context, the increased risk of falls should not prevent the use of SGLT2 inhibitors in persons with sarcopenia or frailty. However, when administering SGLT2 inhibitors or GLP-1RAs to these patients, one should implement nutritional and exercise therapies to minimize muscle mass loss.
Sulfonylurea and insulin administrations were not significantly associated with falls, possibly because intensive inpatient education and optimization of diabetes management, including the introduction of continuous glucose monitoring for patients receiving insulin therapy, effectively reduced the risk of hypoglycemia.
In summary, the effects of SGLT2 inhibitors and GLP-1RAs on falls in persons with type 2 diabetes were investigated by prospectively following up hospitalized elderly and nonelderly individuals with type 2 diabetes for up to 5 years after discharge. The use of SGLT2 inhibitors significantly increased the risk of falls, whereas that of GLP-1RAs had an insignificant effect. Furthermore, no interaction between the two drugs was observed, and the risk of falls further increased upon their combined administration. Our findings suggest the need to (i) consider the increased risk of falls in the case of combined therapy and (ii) provide fall-prone persons receiving such therapy with an appropriate diet and exercise.
The limitations of our study include the follow-up rate: for cohort studies, an 80% follow-up rate is desirable, whereas our first follow-up rate was 69%. Therefore, a selection bias could not be ruled out. Moreover, as questionnaires were mailed every year after discharge, a recall bias is possible. Previous studies found that 13% of participants who confirmed experiencing falls could not recall having fallen at the end of the study (i.e., after 12 months)32. Therefore, the fall rate could have been underestimated. Furthermore, the participants were hospitalized with poorly controlled blood glucose levels, i.e., our results may not necessarily apply to persons with type 2 diabetes with stable glycemic control in an outpatient setting. Additionally, although this research commenced in February 2014, SGLT2 inhibitors were not introduced in Japan until April 2014. Over the course of the study period, the prescription trends for antidiabetic medications shifted due to accumulating evidence supporting the efficacy of SGLT2 inhibitors and GLP-1RAs. Consequently, participants’ treatment regimens may have changed during follow-up. However, as our study did not track medication changes throughout the observation period, potential treatment-related biases cannot be excluded. Finally, this study presents results from a single center, and the validity of our findings should therefore be confirmed for larger and more diverse populations, including those with different racial backgrounds.
Methods
Study design and participants
A questionnaire surveying falls over a duration of 5 years was carried out on 678 persons with type 2 diabetes who were totally independent in walking and daily living activities but had been admitted to the University of Tsukuba Hospital because of poor glycemic control between February 2014 and December 2021. The exclusion criteria were (i) vitreous hemorrhage or retinal detachment, (ii) New York Heart Association functional class of II or higher, (iii) malignancy under treatment, (iv) intake of glucocorticoids, Cushing’s syndrome, or acromegaly, (v) postgastrectomy, (vi) inability to walk independently without assistive devices, (vii) nondiabetic neuropathy, and (viii) difficulty in understanding instructions.
This study was approved by the Ethics Committee of the University of Tsukuba Hospital (approval number: H27-31) and conducted according to the Declaration of Helsinki. Written informed consent was obtained from all participants.
Follow-up of falls and body weight
Fall was defined as “coming in contact with the ground (or floor) from a standing or sitting position with a body part other than the foot in contact with the ground (floor surface) against the patient’s intention”33. The reliability of surveying the occurrence of falls over a previous 1-year period using the recall method was confirmed previously32. A fall history for the previous year was obtained on hospital admission. Each year after their discharge, the study participants were mailed a questionnaire asking about the number of falls (never, once, twice, or more) experienced within the previous 1-year period and their current weight.
Clinical data acquisition, laboratory tests, and physical function assessment
Clinical data acquisition, laboratory tests, and physical function assessments were performed as described elsewhere12. Sociodemographic information, history of falls for the previous year at admission, medical history, anthropometric data, and information on medications administered at discharge were collected. Body composition was assessed by bioelectrical impedance analysis (InBody 720, InBody Japan, Tokyo, Japan). Body mass index (BMI) was calculated by dividing weight (kg) by the square of height (m2). Skeletal muscle mass index was calculated by dividing limb skeletal muscle mass (kg) by the square of height (m2).
Diabetic retinopathy was assessed by ophthalmologists. Diabetic polyneuropathy was diagnosed based on two or more of the following four criteria from The Diabetic Neuropathy Study Group in Japan and Michigan Neuropathy Screening Instrument: (i) decreased vibration perception using a 128-Hz tuning fork at the bilateral medial malleoli (< 10 s), (ii) loss of tactile sensation using a 10-g monofilament at the bilateral foot, (iii) decreased or absent bilateral Achilles tendon reflex, and (iv) numbness, pain, paresthesia, or hypoesthesia in the bilateral lower limbs or feet. Peripheral artery disease was diagnosed when the ankle-brachial index for either of the lower limbs was less than 0.9. Cardiac autonomic neuropathy was diagnosed when the variation coefficient of RR intervals at rest was below 2%. Diabetic nephropathy corresponded to urinary albumin excretion rates of ≥ 30 mg/day.
Blood samples were collected in the morning after an overnight fast within 3 days after admission to measure the plasma levels of glucose and serum levels of total cholesterol, high-density lipoprotein cholesterol, triglycerides, low-density lipoprotein-cholesterol, creatinine, C-peptide, and HbA1c. The glomerular filtration rate was estimated using an equation modified for the Japanese population34.
Physical activity during hospitalization was assessed using an accelerometer (Mediwalk, Terumo, Tokyo, Japan). Knee extension strength and knee extension endurance on the dominant foot side were measured using an isokinetic dynamometer (Biodex System 3, Sakai Medical, Tokyo, Japan). The dorsiflexion strength of the ankle joint was determined using a hand-held dynamometer (µTAS F-1, Anima, Tokyo, Japan). Toe pinch force was assessed using a pinch force dynamometer (Checker-kun, Nissin Sangyo, Saitama, Japan). The grip strength of the dominant hand was assessed using a Smedley analog grip strength tester (ST100T-1780, Toei Light, Tokyo, Japan). Truncal flexibility was assessed by measuring the finger–floor distance.
Balance capability was assessed using the one-leg standing time with eyes open and index of postural stability35. The latter index was determined using a gravicorder (GP-6000, Anima, Tokyo, Japan), and the modified index of postural stability was measured while the participant stood with eyes closed on a gravicorder covered with foam rubber35.
Statistical analysis
The sample size was determined by considering the number of cases in prior studies on falls and number of participants that could be annually enrolled in the present study. Participants with missing data (31 persons) were excluded from the analysis.
Continuous variables were checked for normal distribution using the Shapiro–Wilk test and expressed as means ± standard deviations or medians (25th and 75th percentiles) based on the distribution.
Four discrete–time logistic models36,37 were constructed to extract fall risk factors. Model 1 considered gender, age, height, BMI, SGLT2 inhibitors, GLP-1RAs, interaction of SGLT2 inhibitors and GLP-1RAs, and fall history at admission. Model 2 considered the factors of Model 1 plus insulin, sulfonylureas/glinides, metformin, grip strength, fasting serum C-peptide level, knee extension strength, dorsiflexion strength, and proliferative diabetic retinopathy. Model 3 considered the factors of Model 2 plus weight change. Model 4 considered the factors of Model 3 plus dipeptidyl peptidase-4 inhibitors, α-glucosidase inhibitors, and thiazolidinediones.
All statistical analyses were conducted using R software (version 4.3.2; R Core Team, Vienna) and SPSS software (version 24.0; IBM Japan). Significance levels for all statistical tests and confidence levels for all confidence intervals were set at 0.05 and 95%, respectively.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
James, S. L. et al. The global burden of falls: global, regional and National estimates of morbidity and mortality from the global burden of disease study 2017. Inj. Prev. 26, i3–i11. https://doi.org/10.1136/injuryprev-2019-043286 (2020).
Kwan, M. M., Close, J. C., Wong, A. K. & Lord, S. R. Falls incidence, risk factors, and consequences in Chinese older people: a systematic review. J. Am. Geriatr. Soc. 59, 536–543. https://doi.org/10.1111/j.1532-5415.2010.03286.x (2011).
Seppala, L. J. et al. STOPPFall (Screening tool of older persons prescriptions in older adults with high fall risk): a Delphi study by the EuGMS task and finish group on Fall-Risk-Increasing drugs. Age Ageing 50, 1189–1199. https://doi.org/10.1093/ageing/afaa249 (2021).
Tinetti, M. E. & Kumar, C. The patient who falls: it’s always a trade-off. JAMA 303, 258–266. https://doi.org/10.1001/jama.2009.2024 (2010).
Yang, Y., Hu, X., Zhang, Q. & Zou, R. Diabetes mellitus and risk of falls in older adults: a systematic review and meta-analysis. Age Ageing 45, 761–767. https://doi.org/10.1093/ageing/afw140 (2016).
Schwartz, A. V. et al. Older women with diabetes have a higher risk of falls: a prospective study. Diabetes Care 25, 1749–1754. https://doi.org/10.2337/diacare.25.10.1749 (2002).
Macgilchrist, C. et al. Lower-limb risk factors for falls in people with diabetes mellitus. Diabetes Med. 27, 162–168. https://doi.org/10.1111/j.1464-5491.2009.02914.x (2010).
Schwartz, A. V. et al. Diabetes-related complications, glycemic control, and falls in older adults. Diabetes Care 31, 391–396. https://doi.org/10.2337/dc07-1152 (2008).
de Roman, T., Cambier, D., Calders, P., Van Den Noortgate, N. & Delbaere, K. Understanding the relationship between type 2 diabetes mellitus and falls in older adults: a prospective cohort study. PLoS One 8, e67055. https://doi.org/10.1371/journal.pone.0067055 (2013).
Vinik, A. I. et al. Aging, diabetes, and falls. Endocr. Pract. 23, 1117–1139. https://doi.org/10.4158/EP171794.RA (2017).
Lee, A. K. et al. Severe hypoglycemia and risk of falls in type 2 diabetes: the atherosclerosis risk in communities (ARIC) study. Diabetes Care 43, 2060–2065. https://doi.org/10.2337/dc20-0316 (2020).
Suzuki, Y. et al. Exploratory analysis using machine learning of predictive factors for falls in type 2 diabetes. Sci. Rep. 12, 11965. https://doi.org/10.1038/s41598-022-15224-4 (2022).
Yokoyama, H. et al. Trends in glycemic control in patients with insulin therapy compared with non-insulin or no drugs in type 2 diabetes in Japan: a long-term view of real-world treatment between 2002 and 2018 (JDDM 66). BMJ Open Diabetes Res. Care 10. https://doi.org/10.1136/bmjdrc-2021-002727 (2022).
Yki-Jarvinen, H. et al. Effects of adding linagliptin to basal insulin regimen for inadequately controlled type 2 diabetes: a >/=52-week randomized, double-blind study. Diabetes Care 36, 3875–3881. https://doi.org/10.2337/dc12-2718 (2013).
Shin, Y., Choi, H. & Lim, S. Comparison betweeen Dapagliflozin add-on therapy and insulin dose escalation in patients with uncontrolled type 2 diabetes treated with insulin: DVI study. Diabetes Res. Clin. Pract. 175, 108843. https://doi.org/10.1016/j.diabres.2021.108843 (2021).
Pozzilli, P. et al. Placebo-controlled, randomized trial of the addition of once-weekly glucagon-like peptide-1 receptor agonist dulaglutide to titrated daily insulin glargine in patients with type 2 diabetes (AWARD-9). Diabetes Obes. Metab. 19, 1024–1031. https://doi.org/10.1111/dom.12937 (2017).
Giugliano, D. et al. Feasibility of simplification from a basal-bolus insulin regimen to a fixed-ratio formulation of basal insulin plus a GLP-1RA or to basal insulin plus an SGLT2 inhibitor: BEYOND, a randomized, pragmatic trial. Diabetes Care 44, 1353–1360. https://doi.org/10.2337/dc20-2623 (2021).
Rosenstock, J. et al. Impact of a weekly glucagon-like peptide 1 receptor agonist, albiglutide, on glycemic control and on reducing prandial insulin use in type 2 diabetes inadequately controlled on multiple insulin therapy: a randomized trial. Diabetes Care 43, 2509–2518. https://doi.org/10.2337/dc19-2316 (2020).
Sargeant, J. A. et al. A review of the effects of glucagon-like peptide-1 receptor agonists and sodium-glucose cotransporter 2 inhibitors on lean body mass in humans. Endocrinol. Metab. (Seoul) 34, 247–262. https://doi.org/10.3803/EnM.2019.34.3.247 (2019).
Reijnierse, E. M. et al. Lower skeletal muscle mass at admission independently predicts falls and mortality 3 months post-discharge in hospitalized older patients. J. Gerontol. Biol. Sci. Med. Sci. 74, 1650–1656. https://doi.org/10.1093/gerona/gly281 (2019).
Scholtes, R. A. et al. Natriuretic effect of two weeks of Dapagliflozin treatment in patients with type 2 diabetes and preserved kidney function during standardized sodium intake: results of the DAPASALT trial. Diabetes Care 44, 440–447. https://doi.org/10.2337/dc20-2604 (2021).
Li, C. X. et al. Comparative safety of different sodium-glucose transporter 2 inhibitors in patients with type 2 diabetes: a systematic review and network meta-analysis of randomized controlled trials. Front. Endocrinol. (Lausanne) 14, 1238399. https://doi.org/10.3389/fendo.2023.1238399 (2023).
Saijo, Y. et al. Reasons for discontinuing treatment with sodium-glucose cotransporter 2 inhibitors in patients with diabetes in real-world settings: the KAMOGAWA-A study. J. Clin. Med. 12 https://doi.org/10.3390/jcm12226993 (2023).
Maule, S. et al. Orthostatic hypotension: evaluation and treatment. Cardiovasc. Hematol. Disord. Drug Targets 7, 63–70. https://doi.org/10.2174/187152907780059029 (2007).
Poon, I. O. & Braun, U. High prevalence of orthostatic hypotension and its correlation with potentially causative medications among elderly veterans. J. Clin. Pharm. Ther. 30, 173–178. https://doi.org/10.1111/j.1365-2710.2005.00629.x (2005).
Gupta, V. & Lipsitz, L. A. Orthostatic hypotension in the elderly: diagnosis and treatment. Am. J. Med. 120, 841–847. https://doi.org/10.1016/j.amjmed.2007.02.023 (2007).
Bhanu, C. et al. Drug-induced orthostatic hypotension: A systematic review and meta-analysis of randomised controlled trials. PLoS Med. 18, e1003821. https://doi.org/10.1371/journal.pmed.1003821 (2021).
Mantsiou, C. et al. Glucagon-like peptide-1 receptor agonists and sodium-glucose co-transporter-2 inhibitors as combination therapy for type 2 diabetes: A systematic review and meta-analysis. Diabetes Obes. Metab. 22, 1857–1868. https://doi.org/10.1111/dom.14108 (2020).
American Diabetes Association Professional Practice Committee. Pharmacologic approaches to glycemic treatment: standards of care in diabetes-2024. Diabetes Care 47, S158–S178. https://doi.org/10.2337/dc24-S009 (2024).
Butt, J. H. et al. Efficacy and safety of Dapagliflozin according to frailty in heart failure with reduced ejection fraction: a post hoc analysis of the DAPA-HF trial. Ann. Intern. Med. 175, 820–830. https://doi.org/10.7326/M21-4776 (2022).
Vart, P. et al. Efficacy and safety of Dapagliflozin in patients with chronic kidney disease across the spectrum of frailty. J. Gerontol. Biol. Sci. Med. Sci. https://doi.org/10.1093/gerona/glad181 (2023).
Cummings, S. R., Nevitt, M. C. & Kidd, S. Forgetting falls. The limited accuracy of recall of falls in the elderly. J. Am. Geriatr. Soc. 36, 613–616. https://doi.org/10.1111/j.1532-5415.1988.tb06155.x (1988).
Gill, T. M., Williams, C. S. & Tinetti, M. E. Environmental hazards and the risk of nonsyncopal falls in the homes of community-living older persons. Med. Care 38, 1174–1183. https://doi.org/10.1097/00005650-200012000-00004 (2000).
Matsuo, S. et al. Revised equations for estimated GFR from serum creatinine in Japan. Am. J. Kidney Dis. 53, 982–992. https://doi.org/10.1053/j.ajkd.2008.12.034 (2009).
Suzuki, Y. et al. Age-dependent changes in dynamic standing-balance ability evaluated quantitatively using a stabilometer. J. Phys. Ther. Sci. 30, 86–91. https://doi.org/10.1589/jpts.30.86 (2018).
Thomas, W., Moritz, B., David, K. & Matthias, S. CRAN—Package discSurv. https://cran.r-project.org/web/packages/discSurv/index.html (2022).
Tutz, G. & Schmid, M. Modeling Discrete Time-to-Event Data (Springer, 2016).
Acknowledgements
This work was supported by JSPS KAKENHI Grant-in-Aid for Early-Career Scientists JP24K20536.
Funding
This research was funded by the “Grant for Research on Medical Safety” from University of Tsukuba Hospital.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by YS, HS and KM. The first draft of the manuscript was written by YS and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Suzuki, Y., Suzuki, H., Maruo, K. et al. Longitudinal association of SGLT2 inhibitors and GLP-1RAs on falls in persons with type 2 diabetes. Sci Rep 15, 9178 (2025). https://doi.org/10.1038/s41598-025-91101-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-91101-0
This article is cited by
-
The use of SGLT2 inhibitors in older people: What is important?
Aging Clinical and Experimental Research (2025)