Abstract
In this study, a statistical estimation is done for an epidemic model of cryptosporidiosis by changing it into a fractional order system. The disease-free equilibrium point, and the endemic equilibrium point are the two equilibrium points and Jacobian matrix theory is used to determine stability. The basic reproductive number \({R}_{0}\) is calculated and examined for its role in disease dynamics and stability analysis. The numerical technique named Grunwald Letnikov non-standard finite difference (GL-NSFD) scheme is designed for solving the fractional epidemic model. To investigate the characteristics and properties of numerical design, a test problem is considered for the simulation. For the underlying system, a non-classical numerical approach is suggested. The state variables cannot be negative because they describe the number of people. The suggested numerical scheme must have the properties of positivity and boundedness. The positivity and boundedness of the fractional order cryptosporidiosis epidemic model are investigated with the help of Laplace and inverse Laplace transformation. Finally, the conclusions of the study are elaborated.
Similar content being viewed by others
Introduction
A micro parasite called cryptosporidium that can survive in the intestines of both humans and animals and passes through the faces of an infected person or animal causes the diarrheal condition known as cryptosporidiosis. Common names for both the illness and the parasite are “crypto”. The parasite has a strong outer skin that makes it particularly resistant to chlorine-based chemicals and allows it to live outside the body for long periods of time. Crypto has gained attention as one of the most common causes of waterborne illness in humans in the United States during the past 20 years, both in recreational and drinking water. An infected person or animal’s stool results could contain millions of crypto viruses. When the symptoms commence, crypto begins to shed in the stool, and it can persist for weeks after the symptoms stop 1.
Ernest Edward Tyzzer discovered Cryptosporidium in the intestine tissue of healthy mice in 1907 2, but the first human instances of cryptosporidiosis weren’t discovered until 1976 3,4. But until 1983, only a few additional occurrences of cryptosporidium-related severe, persistent diarrhea in humans were observed in the United States among urban males with AIDS and immunodeficient people, particularly calf handlers 5. Global medical and scientific communities have taken notice of the link between AIDS and cryptosporidium and subsequent outbreaks of cryptosporidiosis in immunologically healthy people 6.
The study of every facet of the disease increased as knowledge of the parasite’s pollution and the development of diagnostic screening procedures increased. By the middle of the 1980s, many people became aware of cryptosporidium as a recently discovered and possibly dangerous human intestinal virus 7,8,9. This paper aims to propose a fractional-order age-structured model for the analysis of smoking epidemic with a focus on the influence of the age factor on the smoking behaviour and its dynamicity 10. Therefore, the present paper aims to develop a fractional model for co-infection of Marburg and Monkeypox virus, which gives a clearer understanding of the possibility of the transmission of the two diseases 11. The study conducted on a fractional-order model of Ebola and Malaria co-infection focuses on the impact of detection and treatment parameters to the diseases 12. This research aims at discussing the mechanisms of smoking behaviour by developing Age-Scale Models with Fractal-Fractional Derivatives, concentrated on the impact of the government interventions 13. Zarin et al. studied haar wavelet collocation methods of the fractional-order antidotal computer virus model 14. Jitsinchayakul et al. studied fractional modeling of the COVID-19 epidemic model with harmonic mean type incidence rate 15. Chu et al. studied a vigorous study of fractional order mathematical model for the SARS-CoV-2 epidemic with Mittag–Leffler kernel 16. Zarin studied a numerical study of a nonlinear COVID-19 pandemic model by finite difference and meshless methods 17. Zarin et al. studied fractional modeling of the COVID-19 pandemic model with real data from Pakistan under the ABC operator 18.
Applications of fractional-order differential models in the context of infectious diseases present unique benefits in the modeling of real-world infectious disease transmission. These models employ non-integer derivatives with a fractional-boundary condition to model memory effects and long-range temporal dependencies that appear in actual epidemiological data but can be described only approximately with the help of integer-order derivatives. For example, the fractional order models can be more effective in capturing long-term dependencies of disease epidemics in a population, which involve past infection rates in formulating the current behavior. This capability is particularly useful where diseases and incubation periods are lengthy or where diseases are cyclic in nature. Furthermore, fractional order models can be used at different scales of transmission rates ranging from small outbreaks to world spreading and hence they are helpful in public health. In concrete situations, they enrich the accuracy of predicting the development of diseases, evaluating the efficiency of measures to limit them, and distributing significant resources during a pandemic. In this sense, the improvement in the complexity of the model, through a fractional order approach, yields a system response that is closer to the actual behavior of the disease, allowing for better strategy conception for its prevention and control.
Adjusting the fractional order of equations that describe the dynamics of a disease can affect the behavior and results of the model, making it much more practical in comprehending the spread and control of various diseases. The fractional only in these models signifies the memory and hereditary nature of the models, which shows how past states are effective in the current behavior of the system. In summary, enhancing disease dynamic models with fractional orders enhances realism, and gets researchers closer to capturing key aspects of infectious disease epidemiology, thereby improving the development of public health intervention strategies.
Definitions and preliminaries
Some fundamental definitions for the fractional derivatives are given in this section.
Definition 1
The Gamma function is defined as,
Definition 2
Let \(\text{y}(\uptau )\) satisfies some smoothness conditions in every finite interval \((0,t)\) with \(t\le \tau\). Then
Definition 3
Single parameter Mittag-Laffler (M-L) function is defined as,
Moreover, two parameter form of (M-L) function can be written as,
Definition 4
Let \(\text{Z}(\uptau )\) satisfy some smoothness condition in every finite interval (0, t) with \(\text{t}\le\uptau .\) Then Caputo fractional derivative is defined as,
where the norm is defined as, \({\Vert \text{Z}\Vert }_{\infty }=\text{sup}\{\left|Z\left(t\right)\right|:t\in I\}\). By \(C(I,R)\) we mean the Banach space of all continuous functions from I into R 19.
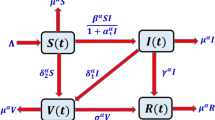
Unraveling of the model
The state variables in the model are \(S(t)\), \(I(t)\), \(R(t)\) and \(E(t)\) which describe the susceptible, infected, recovered individuals and microbial population respectively the parameters are enlisted below.
Parameter symbol | Parameter rates |
|---|---|
\(\wedge\) | Recruitment |
\(\omega\) | Lose of immunity |
\(\sigma\) | Recovery |
\(\mu\) | Natural |
\(\psi\) | Mortality |
\(\upsilon\) | Contact of microbe population |
\(\kappa\) | Concentration of microbe population in the environment |
\(\pi\) | Cryptosporidiosis infected to the environment |
\({\mu }_{b}\) | Mortality of microbes |
\(\rho\) | Contact with the environment |
By replacing Caputo derivatives instead of ordinary derivative, the fractional delayed model will adopt the form as explained in system.
Model analysis
In this section, we investigate the positivity, boundedness, unique existence of solution to the model (1).
Positivity
The compartmental epidemic model have some key traits such as positivity, boundedness and convergence toward the steady states. Here, we will establish the result with proof for the positivity of the system (1).
Theorem 1
For any initial positive values, then (1) is positive invariant in \({\mathbb{R}}_{+}^{4}\).
Proof
Consider 1st equation system (1),
Let \({M}_{1}={\mu }^{\lambda }S\left(t\right)+\left(\frac{{\upsilon }^{\lambda }I\left(t\right)}{{K}^{\lambda }+I\left(t\right)}+{\rho }^{\lambda }E\left(t\right)\right)\).
So, the above expressions becomes as,
\({}_{0}^{\text{c}}{\text{D}}_{\text{t}}^{\uplambda }\text{ S}(\text{t})\ge -\left({\text{M}}_{1}\right)\text{S}\), this implies that \({}_{0}^{\text{c}}{\text{D}}_{\text{t}}^{\uplambda }\text{ S}(\text{t})+{\text{M}}_{1}\text{S}\ge 0\).
By taking Laplace transformation
Now, applying the inverse Laplace transformation
Similarly doing for the rest of the equations. We conclude that the model holds the positivity.□
Boundedness
In this segment, we prove another paramount feature of the model (1) i.e. the boundedness.
Lemma 1
For positive initial conditions, the system (1) is bounded for all t ∈ [0, tm).
Proof
Consider the system (1) in the following form,
By taking Laplace transformation on both sides
Now by taking inverse Laplace transformation on both sides
Let,
By using, \({E}_{\alpha ,\beta }\left(z\right)=z{E}_{\alpha ,\alpha +\beta }\left(z\right)+\frac{1}{\Gamma (\beta )}\).
We can write above inequality as
\(N\left(t\right)\le M\), Which is required (\(\Gamma \left(1\right)=1)\). □
Existence and uniqueness
In this section, we present the unique existence of the solution of system (1),
Lemma 2
For positive initial conditions, the solution of (1) will exist and unique.
Proof
Consider,
Therefore, X(S) satisfies the Lipchitz condition, for contraction mapping
Similarly, for the rest of the equations we have,
Also, \(\text{F}=\text{max}\left\{ {\text{F}}_{1},{\text{F}}_{2},{\text{F}}_{3},{\text{F}}_{4}\right\}\).
Therefore,
For F < 1, K(S), M(E), N(I) and L(R) are contraction mappings. □
Basic reproductive number (R0)
In this section, next generation matrix method is used for the calculation of R0 and it is formed as,\({R}_{0}=\frac{{\upsilon }^{\lambda }{\wedge }^{\lambda }{{\mu }^{\lambda }}_{b}+{\rho }^{\lambda }{\wedge }^{\lambda }{\pi }^{\lambda }{\mu }^{\lambda }{K}^{\lambda }}{{{\mu }^{\lambda }}_{b}{\mu }^{\lambda }{K}^{\lambda }\left({\mu }^{\lambda }+{\psi }^{\lambda }+{\sigma }^{\lambda }\right)}\).
In this section, the sensitivity index of the parameters involved in the reproduction number and its graphical representation are presented. All parameters are sensitive, but most are more sensitive, showing a positive ratio at given data, and others are less sensitive.
Steady states
In this section, two equilibrium points are of the model (1) are presented in this section.
Definition 5
A point \({x}^{*}\) is said to be an equilibrium point of the system \({}_{0}^{\text{c}}{\text{D}}_{\text{t}}^{\uplambda }\) = \(f\left(t,x\left(t\right)\right),x\left({t}_{0}\right)>0\), \(iff f\left(t,{x}^{*}\left(t\right)\right)=0\).
The disease-free equilibrium point of system (1) is,
To find the endemic equilibrium point first consider,
where,
Thus,
Local stability
In this section, local stability of the epidemic model is investigated at the disease free equilibrium point. In this connection the following result are established.
Definition 6
An equilibrium point \({x}^{*}\) of the system \({}_{0}^{\text{c}}{\text{D}}_{\text{t}}^{\uplambda }=f\left(t,x\left(t\right)\right),x\left({t}_{0}\right)>0,\) is said to be asymptotically stable if all the eigenvalues of the Jacobian matrix (J) evaluated at \({x}^{*}\) satisfies \(\left|\text{arg}({\lambda }_{i})\right|>\frac{\alpha \pi }{2}\), where \({\lambda }_{i}\) are eigenvalue of J.
Theorem 2
The disease-free equilibrium \({E}_{0}\) is locally asymptotically stable if all the eigen values of the Jacobian matrix are negative.
Proof
The Jacobian matrix of the system (1) and its elements are given below
Consider Jacobian at \({E}_{0}\),
Since \(det\left({J}_{{E}_{0}}-\lambda I\right)=0\),
root is negative and real,
where \({\uplambda }_{2}\),\({\lambda }_{3}\) and \({\uplambda }_{4}\) belongs to
by using the Routh-Hurwitz criterion for \({3}^{rd}\) order polynomials we have
Here
All the three roots are positive and must be satisfied the condition
Hence, by Routh-Hurwitz criterion \({E}_{0}\) is stable. □
Theorem
The existing equilibrium (\(EE\)) is locally asymptotically stable, if, \({\mathcal{R}}_{0}>1\).
Proof
The Jacobian matrix of the system (1) and its elements are given below.
The Jacobian matrix at existing equilibrium (\(\text{EE}\)) is as follows,
where,
All the three roots are positive and must be satisfied the condition
Hence, by Routh-Hurwitz criterion system is locally stable at EE. □
Numerical scheme and results
This section is devoted to present the numerical scheme for the solution of underlying model.
Definition 7
Let \(y(\tau )\) satisfies some smoothness conditions in every finite interval (0,t) with \(t\le \tau .\) The nonstandard Grunwald–Letnikov Approximation for \(y(\tau )\) is
where \({e}_{v}^{\lambda }=-{\left(1\right)}^{v-1}\left(\begin{array}{c}\lambda \\ v\end{array}\right), {r}_{n+1}^{\lambda }={h}^{\lambda }{r}_{0}^{\lambda }\left({\tau }_{n+1}\right)={\gamma }_{0,-1}^{\lambda }{\left(n+1\right)}^{-\lambda }\)
and the coefficient
The Grunwald–Letnikov Approximation is the extension of the Euler method, so its order of convergence coincides with order Euler method by taking \(\lambda\)=1. For more details, the reference 20 is helpful.
Using above approximation for system (1), we have
similarly, we have the following results
Now the following results ensure the ability of proposed technique to retain the positive and bounded behavior of solution.
Lemma 3
Assume that all of the variables and control parameters are positive i.e.,
\({S}_{0}>0, {I}_{0}>0, {R}_{0}>0, {E}_{0}>0\) and \({\wedge }^{\lambda }>0\), \({\omega }^{\lambda }>0\), \({\mu }^{\lambda }>0\), \({\psi }^{\lambda }>0\), \({\sigma }^{\lambda }>0\), \({\rho }^{\lambda }>0, {\upsilon }^{\lambda }>0, {\kappa }^{\lambda }>0, {\pi }^{\lambda }>0, {\mu }_{b}^{\lambda }>0,\phi {\left(h\right)}^{\lambda }>0,\) are all positive, then \({S}_{n}>0,{I}_{n}>0,{R}_{n}>0\) and \({E}_{n}>0\) is satisfied for all \(n=0,1,2,3\ldots \epsilon {Z}^{+}\).
Proof
Since
For \(n=0\),
since, \({S}_{0}\) and all the parameters are positive.
Then \({S}_{1}\ge 0\). Similarly it can easily be proved that \({I}_{1},{R}_{1} and {E}_{1}\ge 0.\) Next we suppose that the result holds for \(n=\left\{\text{1,2},\text{3,4},\dots ,n-1\right\} i.e {S}_{n},{I}_{n},{R}_{n}\text{and }{E}_{\text{n}} \ge 0, \forall n=\left\{\text{1,2},\text{3,4},\dots ,n-1\right\}\).
Moreover for \(n\in {Z}^{+}\) we have
since, all the discretize state variables and parameters are positive. Therefore \({S}_{n+1}\ge 0.\) Similarly \({I}_{n+1},{R}_{n+1},{E}_{n+1}\ge 0.\) Hence the proposed numerical scheme preserved the positivity for \(n\in {Z}^{+}\). □
Lemma 4
Let \({S}_{0},{I}_{0},{R}_{0}\) are finite so, \({S}_{0}+{I}_{0}+{R}_{0}\le {L}_{0}.\) All the parameters \({\wedge }^{\lambda },{\omega }^{\lambda },{\mu }^{\lambda },{\psi }^{\lambda },{\sigma }^{\lambda },{\rho }^{\lambda },{\pi }^{\lambda },{\upsilon }^{\lambda },{\kappa }^{\lambda }\),\({\mu }_{b}^{\lambda }\) and \(\phi {\left(h\right)}^{\lambda }\) are positive.
Then there is a constant \({M}_{n+1}\) such that \({S}_{n+1}\le {M}_{n+1}\),\({I}_{n+1}\le {M}_{n+1}\), and \({R}_{n+1}\le {M}_{n+1}\forall n\in {Z}^{+}0<{E}_{n+1}<1\) for microbe papulation.
Proof
Since all the parameters and state variables are positive then there exists a constant \({M}_{n+1} ,\) such that \({S}_{n+1},{I}_{n+1},{R}_{n+1}\text{and }{E}_{n+1}\le {M}_{n+1}\).
This result is proved by mathematical induction. Firstly, consider the case when \(n=0\) in the above expression.
similarly, it is easy to check that this constant is such that \({I}_{1}\le {M}_{1}\), \({R}_{1}\le {M}_{1}\) and \({E}_{1}\le {M}_{1}\). We define \({M}_{1}\) as the maximum. Now for \(n=1\) we can see that
similarly, \({I}_{2}\le {M}_{2}\), \({R}_{2}\le {M}_{2}\) and \({E}_{2}\le {M}_{2}\).
Now for some positive \(n\in {Z}^{+}\)
similarly, \({I}_{n+1}\le {M}_{n+1}\), \({R}_{n+1}\le {M}_{n+1}\) and \({E}_{n+1}\le {M}_{n+1}\).
Hence, scheme preserve the boundedness. □
Numerical simulations
In this segment, graphs are plotted for different values of the parameters given in the above Table 1 to support our claimed feature of the scheme. Figure 1 shows the sensitivity indices of parameter’s involved in reproduction number.
Sensitivity indices of parameter’s involved in reproduction number.
All the plots in Fig. 2 show the dynamics of susceptible individuals due the cryptosporidiosis disease toward the disease-free equilibrium point. Four different graphs are plotted against the different values of fractional order parameter \(\lambda\). The values of \(\lambda\) are described in the Fig. 2. Every graph converges towards the disease-free equilibrium point with a different rate of convergence depending upon the value of \(\lambda\).It can be observed that the graph having the higher value of \(\lambda\) converges fastly to the fixed point as compared to the graph with a smaller value of \(\lambda\). So, it can be concluded that the rate of convergence towards the disease-free equilibrium point is directly proportional to the value of \(\lambda\).
The graphs of susceptible population with different values of \(\lambda\).
All the graphs in Fig. 3 show the progress of infected individuals due the cryptosporidiosis toward the disease-free equilibrium. Four different graphs are plotted against the different values of fractional order parameter \(\lambda\) and these values are 0.75 0.8 0.85 0.9. The values of \(\lambda\) are described in the Fig. 3. Every graph converges towards the disease-free equilibrium point with a different rate of convergence depending upon the value of \(\lambda\).It can be observed that the graph against the higher value of \(\lambda\) converges fastly to the fixed point as compared to the graph with smaller value of \(\lambda\). So, it can be concluded that rate of convergence towards the disease-free equilibrium point is directly proportional to the value of \(\lambda\).
The graphs of infected population with different values of \(\lambda\).
All the sketches in Fig. 4 reflect the progress of infected individuals due the cryptosporidiosis toward the disease-free equilibrium point. Four different graphs are plotted against the different values of fractional order parameter \(\lambda\). The values of \(\lambda\) are described in the Fig. 4. Every graph converges towards the disease-free equilibrium point with a different ratio of convergence depending upon the value of \(\lambda\).It can be noticed that the graph with the higher value of \(\lambda\) converges fastly to the fixed point as compared to the graph with smaller value of \(\lambda\). So, it can be concluded that rate of convergence towards the disease-free equilibrium point is directly proportional to the value of \(\lambda\).
The graphs of recovered population with different values of \(\lambda\).
All the patterns in Fig. 5 reflect the progress of infected individuals due the cryptosporidiosis toward the disease-free equilibrium point. Four different graphs are plotted against the different values of fractional order parameter \(\lambda\) and these values are 0.75 0.8 0.85 0.9. The values of \(\lambda\) are mentioned in the Fig. 5. Every graph converges towards the disease-free equilibrium point with a different ratio of convergence depending upon the value of \(\lambda\).It can be observed that the graph with the higher value of \(\lambda\) converges fastly to the fixed point as compared to the graph with smaller value of \(\lambda\). So, it can be concluded that rate of convergence towards the disease-free equilibrium point is directly proportional to the value of \(\lambda\).
The graphs of microbial population with different values of \(\lambda\).
All the sketch in Fig. 6 reflect the progress of infected individuals due the cryptosporidiosis toward the endemic equilibrium point. Four different graphs are plotted against the different values of fractional order parameter \(\lambda\). The values of \(\lambda\) are mentioned in the Fig. 6. Every graph converges towards the endemic equilibrium point with a different ratio of convergence depending upon the value of \(\lambda\). It can be noticed that the graph with the higher value of \(\lambda\) converges fastly to the fixed point as compared to the graph with smaller value of \(\lambda\). So, it can be concluded that rate of convergence towards the endemic equilibrium point is directly proportional to the value of \(\lambda\).
The graphs of susceptible population with different values of \(\lambda\).
All the plots in Fig. 7 reflect the progress of infected individuals due the cryptosporidiosis toward the endemic equilibrium point. Four different graphs are plotted against the different values of fractional order parameter \(\lambda\) and these values are 0.75 0.8 0.85 0.9. The values of \(\lambda\) are mentioned in Fig. 7. Every graph converges towards the endemic equilibrium point with a different ratio of convergence depending upon the value of \(\lambda\).It can be observed that the graph with the higher value of \(\lambda\) converges fastly to the fixed point as compared to the graph with a smaller value of \(\lambda\). So, it can be concluded that the rate of convergence towards the endemic equilibrium point is directly proportional to the value of \(\lambda\).
The graphs of infected population with different values of \(\lambda\).
All the sketches in Fig. 8 reflect the progress of recovered individuals due the cryptosporidiosis toward the endemic equilibrium point. Four different graphs are plotted against the different values of fractional order parameter \(\lambda\). The values of \(\lambda\) are mentioned in Fig. 8. Every graph converges towards the endemic equilibrium point with a different ratio of convergence depending upon the value of \(\lambda\). It can be noticed that the graph with the higher value of \(\lambda\) converges fastly to the fixed point as compared to the graph with a smaller value of \(\lambda\). So, it can be concluded that the rate of convergence towards the endemic equilibrium point is directly proportional to the value of \(\lambda\).
The graphs of recovered population with different values of \(\lambda\).
All the graphs in Fig. 9 reflect the progress of recovered individuals due the cryptosporidiosis toward the endemic equilibrium point. Four different graphs are plotted against the different values of fractional order parameter \(\lambda\) and these values are 0.75 0.8 0.85 0.9. The values of \(\lambda\) are mentioned in Fig. 9. Every graph converges towards the endemic equilibrium point with a different ratio of convergence depending upon the value of \(\lambda\).It can be noticed that the graph with the higher value of \(\lambda\) converges fastly to the fixed point as compared to the graph with a smaller value of \(\lambda\). So, it can be concluded that the rate of convergence towards the endemic equilibrium point is directly proportional to the value of \(\lambda\).
The graphs of microbial population with different values of \(\lambda\).
GL NSFD simulations are more flexible and render high accuracy results which makes it possible to predict the dynamics of disease by considering the biological systems accurately. As compared to the standard finite difference methods, GL NSFD can handle the nonlinearities and it is capable to preserve crucial properties such as positivity as well as the boundedness which is essential in modelling the spread of diseases in the real world. These models enable one introduce complexities such as transmission rates, recovery rates and spatial distribution of a disease hence offering a more realistic account of how diseases originate and develop. Due to the capabilities of GL NSFD simulations in accurately depicting the dynamics of disease transmission, its output can be used in development of public health principles, prediction of possible scenarios of outbreaks and the assess the likely effects of measures including vaccination, quarantine and social distancing. It also improves the accuracy of epidemiological models, and helps implement disease control measures that are more efficient and relevant.
Covariance of cryptosporidiosis model
The correlation coefficient is a statistical value that shows how closely two sets of values are related, both positively, negatively and the coefficient ranges from + 1 and − 1 respectively. Specifically, a value of + 1 on the coefficient signifies a perfect positive linear relationship and it means, that as the values of one variable go up, the other variable also goes up in a perfectly linear fashion. On the other hand, a value of -1 signifies a negative linear correlation which means a perfect negative correlation in which the increase of one is related to the perfect linear decrease of the other. A positive value indicates that there is a positive linear relationship and if the value is equal to zero then there is no linear relationship in the pair of variables. Co-efficient of correlation shows how one variable might influence another; yet, it does not express cause and cause-and-effect relationship. We have discussed the covariance of the model for Cryptosporidiosis transmission epidemic between compartments in this section. In order to address these, we calculated correlation coefficients and described the results in Table 2. The susceptible class has an inverse relationship with other compartments as shown by solutions in Table 2. If the susceptible class increases eventually, it will be possible to decrease other compartments thus resulting into a cryptosporidiosis-free equilibrium of the model.
Conclusions
A cryptosporidiosis model is taken into consideration for the study in this article. The state variables in the mathematical model are S, I, R, and E. For the model, two steady equilibrium states endemic and disease-free are defined. The next-generation matrix calculates a basic reproduction number. At DFE, the model’s stability and the numerical scheme’s stability are both examined. The part of analyzing disease stability and transmission is also looked at. For validating the preliminary findings, a numerical example and simulations are also presented. The technique may be used to model non-linear integer order epidemic models with delay factors in the future. The numerical scheme illustrates the dynamics of the disease in the model’s many compartments. Every equation verifies the convergence to the real steady state and the positive, bounded solutions. So, for the solution of non-linear epidemic models, the NSFD scheme is a trustworthy and effective numerical design. The study findings provide a predictive tool for incident patterns of outbreaks to public health authorities and prioritize the interventions for cryptosporidiosis control. Stressing major transmission drivers underlines the need for improved water quality, public awareness campaigns, and better surveillance systems. These insights support preferential strategies for mitigating disease spread and protecting vulnerable populations.
Data availability
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
References
Riggs, M. W. Recent advances in cryptosporidiosis: The immune response. Microbes Infect. 4(10), 1067–1080 (2002).
Tyzzer, E. E. A sporozoan found in the peptic glands of the common mouse. Proc. Soc. Exp. Biol. Med. 5(1), 12–13 (1907).
Nime, F. A., Burek, J. D., Page, D. L., Holscher, M. A. & Yardley, J. H. Acute enterocolitis in a human being infected with the protozoan Cryptosporidium. Gastroenterology 70(4), 592–598 (1976).
Meisel, J. L., Perera, D. R., Meligro, C. & Rubin, C. E. Overwhelming watery diarrhea associated with a Cryptosporidium in an immunosuppressed patient. Gastroenterology 70(6), 1156–1160 (1976).
Current, W. L. et al. Human cryptosporidiosis in immunocompetent and immunodeficient persons: Studies of an outbreak and experimental transmission. N. Engl. J. Med. 308(21), 1252–1257 (1983).
Fayer, R. et al. Cryptosporidium parvum infection in bovine neonates: Dynamic clinical, parasitic and immunologic patterns. Int. J. Parasitol. 28(1), 49–56 (1998).
Ongerth, J. E. & Stibbs, H. H. Identification of Cryptosporidium oocysts in river water. Appl. Environ. Microbiol. 53(4), 672–676 (1987).
Navin, T. R. & Juranek, D. D. Cryptosporidiosis: A clinical, epidemiologic, and parasitologic review. Rev. Infect. Dis. 6(3), 313–327 (1984).
Casemore, D. P., Sands, R. L. & Curry, A. Cryptosporidium species is a “new” human pathogen. J. Clin. Pathol. 38(12), 1321–1336 (1985).
Addai, E., Zhang, L., Asamoah, J. K. K. & Essel, J. F. A fractional order age-specific smoke epidemic model. Appl. Math. Model. 119, 99–118. https://doi.org/10.1016/j.apm.2023.02.019 (2023).
Zhang, N. et al. Fractional modeling and numerical simulation for unfolding Marburg-Monkeypox virus co-infection transmission. Fractals https://doi.org/10.1142/s0218348x2350086x (2023).
Zhang, L., Addai, E., Ackora-Prah, J., Arthur, Y. D. & Asamoah, J. K. K. Fractional-order Ebola-malaria coinfection model with a focus on detection and treatment rate. Comput. Math. Methods Med. 2022, 1–19. https://doi.org/10.1155/2022/6502598 (2022).
Addai, E., Adeniji, A., Peter, O. J., Agbaje, J. O. & Oshinubi, K. Dynamics of age-structure smoking models with government intervention coverage under fractal-fractional order derivatives. Fractal Fractional 7(5), 370. https://doi.org/10.3390/fractalfract7050370 (2023).
Zarin, R., Khaliq, H., Khan, A., Ahmed, I. & Humphries, U. W. A numerical study based on haar wavelet collocation methods of fractional-order antidotal computer virus model. Symmetry 15(3), 621 (2023).
Jitsinchayakul, S. et al. Fractional modeling of COVID-19 epidemic model with harmonic mean type incidence rate. Open Phys. 19(1), 693–709 (2021).
Chu, Y. M., Zarin, R., Khan, A. & Murtaza, S. A vigorous study of fractional order mathematical model for SARS-CoV-2 epidemic with Mittag-Leffler kernel. Alex. Eng. J. 71, 565–579 (2023).
Zarin, R. Numerical study of a nonlinear COVID-19 pandemic model by finite difference and meshless methods. Partial Differ. Equ. Appl. Math. 6, 100460 (2022).
Zarin, R., Khan, A., Akgül, A. & Akgül, E. K. Fractional modeling of COVID-19 pandemic model with real data from Pakistan under the ABC operator. AIMS Math. 7(9), 15939–15964 (2022).
Hallaci, A., Boulares, H. & Ardjouni, A. Existence and uniqueness for delay fractional differential equations with mixed fractional derivatives. Open J. Math. Anal. 4(2), 26–31. https://doi.org/10.30538/psrp-oma2020.0059 (2020).
Scherer, R., Kalla, S. L., Tang, Y. & Huang, J. The Grünwald-Letnikov method for fractional differential equations. Comput. Math. Appl. 62(3), 902–917 (2011).
Acknowledgements
The authors express their gratitude to Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2025R913), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Funding
Not available.
Author information
Authors and Affiliations
Contributions
N.A.: Model and analysis, problem solution; W.A.A.: Problem formulation; H.A.Z.A.A.: results and discussion; M.E.A.E.: plotting and numerical simulations; M.T.: analysis; software; O.A.A: methodology; simulations, plotting; Z.I.: writing; analysis; conclusion, editing; A.R.: Results, plotting, simulations; B.C.: numerical analysis; software; M.R: methodology, writing, analysis; I.K.: methodology; simulations, plotting.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Not available (No human/animal data is used here).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ahmed, N., Alhilfi, W.A., AlMansury, H.A.Z. et al. A statistical estimation of fractional order cryptosporidiosis epidemic model. Sci Rep 15, 14002 (2025). https://doi.org/10.1038/s41598-025-92144-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-92144-z