Abstract
In this case-control study, the main risk factors for atrophic gastritis (AG) were comprehensively analyzed in a real-world environment to identify potential risk factors associated with garlic intake and its effects on AG. Design Upper gastrointestinal endoscopy and pathological examination were performed as part of a gastric cancer screening and health check-up program. The detailed characteristics of both the case group and healthy control group were recorded and analyzed. All participants were fasted for at least 4 h and a urea breath test13C-UBT) was performed in all participants at rest. Both univariate and multivariate logistic regression analyses were performed and presented as the odds ratio (OR) and 95% confidential interval (CI), with additional subgroup analysis stratified by infectious state based on the presence, eradication or absence of H. pylori. Setting Gansu Province in China. Participants 10,035 people from Gansu Province in China were included. Among 7,058 participants, 4,712 (66.8%) had AG. Garlic intake was a significant risk factor for AG in participants currently (infected state) or previously (eradicated state) infected with H. pylori (OR = 1.39, 95% CI: 1.06–1.83; OR = 1.16, 95% CI = 1.01–1.32). Garlic intake was not significantly associated with AG in participants without H. pylori’ s infection (OR = 1.14, 95% CI: 0.88–1.46). The association between garlic intake and AG differed by H. pylori infectious state. People in the infected or eradicated states are at a higher risk for AG associated with garlic intake. Diet may regulate the pathogenic role of H. pylori and intestinal flora.
Similar content being viewed by others
Introduction
Gastric cancer (GC) is the world’s third and China’s second leading cause of cancer-related death1,2,3. According to the latest incidence and mortality data of GC from 185 countries and regions were extracted from the GLOBOCAN 2022 database, there was an estimated global 968,000 new GC cases and 660,000 deaths in 20224. The incidence of GC has wide geographic variation, east Asia had the highest burden, with 53.8% of cases and 48.2% of deaths among all geographic regions. The incidence of GC is 15–20-fold higher in East Asia and East Europe compared to the incidence in the African continent. Because GC is often diagnosed in an advanced stage with metastases, the prognosis is poor, even in high-income countries4,5. The formation of carcinomas in the stomach is believed to involve progressive processes: chronic gastritis, atrophic gastritis (AG), intestinal metaplasia (IM), gastric intraepithelial neoplasia (GIN), and finally GC6. Histological changes in AG, IM, or GIN are regarded as precancerous lesions of GC6. AG is a critical precancerous lesion in these cumulative processes of developing GC7,8.
Poor eating habits and H. pylori infection are considered important risk factors for AG, and H. pylorialso causes peptic ulcers and other digestive tract-related illnesses9,10,11.
A recent systematic review and meta-analysis, which included 224 studies from 71 countries or regions across all six WHO regions, reported that the estimated global prevalence of H pylori infection decreased from 58.2% in 1980-90 to 43.1% in 2011-22. Prevalence was relatively stable between 1991 and 2010, but fell sharply between 2011 and 2022, with the greatest decline in the WHO African Region. In addition, a lower prevalence of H pyloriinfection has been reported in younger people, in high-income countries or countries with high levels of universal health coverage, and in retrospective studies12.
H. pyloricolonized humans about five thousand years, seeming to build a symbiotic relationship with the human beings13. However, H. pylori is by no means the only dominant intestinal microbiota species in the stomach. A growing body of evidence suggests that H. pylori contributes to the development and progression of GI disease through a variety of pathways. Types and proportions of microbial flora in the feces may be different in the H. pylori-infected populations compared to normal populations, leading to gastric inflammation and cancers in the gastrointestinal (GI) tract (including esophageal, gastric, colorectal, liver, and pancreatic cancers)14. Research shows that there is no significant difference in the prevalence of H. pyloribetween patients with AG and GC in Japan9. So far, few studies have indicated that the human gastric microbiota is different in healthy and diseased individuals13. The predominant phyla of the gastric environment include Actinobacteria, Bacteroidetes, Firmicutes and Proteobacteria, which include H. pylori15,16. Furthermore, recent advances have shown that in the healthy population, the gastric microbiota does not have significant differences according to geographical area and ethnicity, and it is quite similar at both the genera level and the phyla level. Meanwhile, the major driver of gastric microbiota diversity is the presence of H. pyloriinfection17,18. More and more research has focused on the ability of hard foods and probiotics to inhibit the pathogenicity of H. pyloriwhile maintaining the normal microbial environment in the GI tract to prevent GC19,20,21.
Garlic (Allium sativum L) is one of the most popular vegetables in Asia, especially in China. Consumption of garlic and its products prevents the occurrence of AG, and this prevention may be related to H. pyloriinhibition22,23, although some studies have found that garlic had no protective effects against the development of AG and GC24,25. To date, it remains unclear the effect of garlic intake on AG risk in different infectious states of H. pylori. In the present study, we comprehensively analyzed the main risk factors for AG in a real-world environment, and further explored the effects of associated risk factors (including garlic intake) in different infectious states of H. pylori (uninfected state, infected state and eradicated state) on AG.
Methods
Study population
The study was conducted within the framework of the National Upper Gastrointestinal Cancer Early Detection Project in Wuwei City, Gansu Province of China, one of the high-risk areas of upper gastrointestinal cancer. 10,035 study participants with GI symptoms were screened from a high-risk population from 21 administrative villages in Jinyang Town, Siba Town and Qingyuan Town in Liangzhou District, Wuwei City, Gansu Province from November 2015 to November 2016 using the Survey of Health Factors of Upper Gastrointestinal Cancer (Early Diagnosis and Treatment Technical Scheme of China Cancer Screening in 2014 Edition). Upper GI endoscopy and pathological examination were performed as a part of a gastric cancer screening and health check-up program at Wuwei Cancer Hospital of Gansu Province. This study was also approved by the Ethics Committee of National Cancer Center, Chinese Academy of Medical Sciences (2015SQ00223), and all subjects signed the written informed consent before participating this study.
The inclusion criteria of the case group were set as follows: (1) 40–70 years old, and (2) pathologically diagnosed with AG by 2 independent, experienced pathologists. The exclusion criteria of the case group were as follows: (1) pathologically diagnosed with IM, GIN, GC, chronic non-atrophic gastritis and other diet-related diseases, (2) patients who had undergone surgery for GC, and (3) patients who had not stop taking antibiotics, proton pump inhibitors and other medications for at least one week before the13C-Urea Breath Test.
The inclusion criteria of the control group were set as follows: (1) 40–70 years old and (2) pathologically diagnosed with normal glandular epithelium after ESD/EMR by 2 independent, experienced pathologists. The exclusion criteria of the control group were as follows: (1) presence of other diet-related diseases, (2) patients who had undergone surgery for GC, and (3) patients who had not stop taking antibiotics, proton pump inhibitors and other medications for at least one week before the13C-Urea Breath Test.
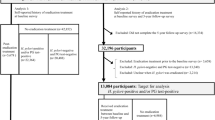
After screening the above inclusion and exclusion criteria, 7,058 people were enrolled. The detailed selection criteria are demonstrated in Fig. 1.
Study flow chart. Notes: A total of 7058 patients with gastrointestinal symptoms received endoscopic and pathological examinations, and all people underwent Helicobacter pylori testing.
Survey content and project
The study was supported by Gansu Province Wuwei Tumor Hospital and Joint project of Chinese Academy of Medical Sciences. The project was conducted in Gansu Province Wuwei Tumor Hospital, and relevant staff were trained before implementation. All subjects were asked to complete a self-administered and structured questionnaire including basic personal information, dietary habits, smoking and alcohol history, garlic intake, family history of gastric cancer, history of disease and results of H. pylori detection. The specific contents of the investigation were strictly abided by using the “Technical Program for Screening and Early Diagnosis and Treatment of Upper Gastrointestinal Cancer (Early Diagnosis and Treatment Technical Scheme of Chinese Cancer Screening in 2014 Edition)”. Eliminated potential recall bias and investigate all patients before diagnosis.
13C-urea breath test (UBT)
The 13C-UBT is a commonly used test for H. pylori infection. The test uses the urease produced by H. pylorito break down urea containing the radioactive 13C marker into carbon dioxide and urea acid, and the change in carbon dioxide levels in the exhaled air determines whether or not be infected with H. pylori.
Patients are required to stop taking antibiotics, proton pump inhibitors and other medications for at least one week before the test and to fasting for at least 4 h prior to the test to ensure that there is no food in the stomach to interfere with the test results. A urea solution containing the radioactive 13C marker is administered orally. After oral administration, patients were waited about 30 min for the urea to be broken down by H. pylori into carbon dioxide and urea acid. Patients exhaled through a respirator into a sample tube containing an absorbent. The laboratory measured the change in carbon dioxide levels in the sample tube to determine whether the patient was infected with H. pylori. The H. pylori infected state was determined if the test value was ≥ 100 dpm/mmol, the H. pylori uninfected state if the test value was < 100 dpm/mmol and there was no previous H. pylori infection, and the H. pylori eradicated state if the test value was < 100 dpm/mmol and there was a previous H. pylori infection.
Endoscopic and pathological examinations
According to the new Sydney standard, 5 tissue specimens were collected from each patient for pathological examination. The location and requirements for tissue collection were as follows: A1: small curvature of the gastric antrum (2–3 cm from pylorus); A2: large curvature of the gastric antrum (2–3 cm from pylorus); B1: small curvature of gastric body (4 cm from gastric angle); B2: large curvature of the gastric body (8 cm from cardia); IA: gastric angle. Biopsy tissue was fixed in formalin and sent to pathology. Routine paraffin sections were prepared and stained with hematoxylin-eosin (HE). The diagnosis of AG in this study was based on pathological diagnosis.
Quality control and personnel training
All investigators were divided into different groups according to the survey items, including digestive physicians, specialist nurses and graduate students who had been strictly trained. After qualified training, investigators participated in the survey. Data were entered into EpiData Software3.1, and a duplicate record was used. During the survey, project reviewers reviewed each questionnaire each survey day, and uncompleted questionnaires were required to be completed and corrected in time.
Statistical analysis
Continuous variables are expressed as mean ± SD following the normal distribution analysis and were compared using Student’s t-test or a 1-way analysis of variance test. Categorical variables were expressed by counts and percentages and compared with χ2 statistics or Fisher’s exact test, as appropriate. To identify potential risk factors for AG, univariate and multivariate logistic regression analysis of binary variables were carried out with negative/positive AG as the dependent variable and other parameters, including basic information, living habits, disease history and HP, as independent variables. Regression analysis was performed, and results are presented as the odds ratio (OR) with the corresponding 95% confidential interval (CI). Statistical analyses were performed using SPSS (version 21.0; IBM Corp., Armonk, NY, USA), with 0.05 used as the significance level.
Results
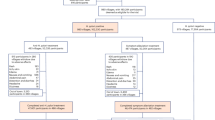
A total number of 7,058 participants were analyzed in this study. Among them, 4,712 (66.8%) had AG and served as the case group, and 2,346 (33.2%) had normal glandular epithelium and served as the control group. The average age of case group was 52.94 years old (SD = 7.47), whereas the average age of the control group was 51.21years old (SD = 7.48). The mean age of the case group was higher than that of control group (P < 0.001). There were 2,501 (53.1%) males and 2,211 (46.9%) females within the case group, and there were 1,399 (59.6%) males and 947 (40.4%) females within the control group. There was a statistically significant difference between the two groups in gender (P < 0.001). About 72.1% of cases and 76.1% of controls were literate. After multivariate logistic regression analysis, risk estimation of AG based on different dietary habits was obtained after controlling for body mass index (BMI), smoking, drinking, marital status, fresh vegetables intake and family history of tumors. Older, male, larger family size, unclean drinking water, H. pylori infection and excessive intake of meat, egg, milk and garlic were significant risk factors for AG. Education, bean food (such as tofu, soybean milk, soymilk etc.) and hard food (such as hard pancake, fried prawn etc.) intake were significant protective factors. Alcohol consumption also did not show significant association with AG. The general characteristics of the study subjects at baseline and the association of dietary habits with AG are shown in Table 1; Fig. 2.
Analysis of risk factors for AG by forest plots with odds ratios.
In the H. pylori uninfected state, after the multivariate adjustment, garlic intake (OR = 1.14, 95% CI = 0.88–1.46, P = 0.32) was not associated with AG (Table 2). However, older age (OR for 50~=1.38, 95% CI = 1.09–1.86, P = 0.02; OR for 60~70=1.61, 95% CI = 1.20–2.38, P = 0.01) and alcohol consumption (OR = 1.50, 95% CI = 1.01–2.23, P = 0.04) were significant risk factors (Table 2; Fig. 3).
Analysis of risk factors for AG in H. pylori’ s uninfected state by forest plots with odds ratios.
In the H. pylori’ s infected state, Table 3; Fig. 4 shows that older age (OR for 50~=1.40, 95% CI = 1.03–1.90, P = 0.03; OR for 60~70=1.66, 95% CI = 1.10–2.52, P = 0.02) and garlic intake (OR = 1.39, 95% CI = 1.06–1.83, P = 0.02) were significant risk factors on AG after the multivariate adjustment.
Analysis of risk factors for AG in H. pylori’ s infected state by forest plots with odds ratios.
In the H. pylori’ s eradicated state, after the multivariate adjustment, the factors associated with AG were older age (OR for 50~=1.46, 95% CI = 1.25–1.69, P < 0.001; OR for 60~70=1.60, 95% CI = 1.30–1.98, P < 0.001), male (OR = 1.39, 95% CI = 1.11–1.73, P < 0.001), garlic intake (OR = 1.16, 95% CI = 1.01–1.32, P = 0.04), family history of tumors (OR = 1.18, 95% CI = 1.01–1.38, P = 0.04) and unclean drinking water (OR for mantle water~=1.50, 95% CI = 1.24–1.82, P < 0.001; OR for deep well water=1.31, 95% CI = 1.11–1.55, P < 0.001; Table 4; Fig. 5). Suitable hard food intake (OR = 0.82, 95% CI = 0.71–0.94, P = 0.01) was a significant protective factor for AG (Table 4). Alcohol consumption (OR = 1.03, 95% CI = 0.83–1.28, P = 0.77) was not significantly associated with AG (Table 4).
Analysis of risk factors for AG in H. pylori’ s eradicated state by forest plots with odds ratios.
Discussion
AG is characterized by the loss of gastric glands, which are replaced by connective tissue or glandular structures that are inappropriate for the location, giving rise to non-metaplastic or metaplastic atrophy26. It is also one of the most common digestive system diseases worldwide and has been defined by WHO as the initial step of cancer27. The human microbiome is an important driver in the etiopathogenesis of AG and cancer28. The occurrence and development of AG are influenced by an interplay between host genetic and environmental factors. The causes of gastritis include H. pyloriinfection, viruses and other pathogenic microbes26. GI microflora and its related metabolites are also closely related to the occurrence and development of precancerous lesions, such as AG, by the induction of inflammation and immune disorders14. H. pyloriis by no means the only predominant species of gut microbiota in the stomach. Accumulating evidence shows that specific bacteria and bacterial dysbiosis in the GI tract can potentiate the development and progression of GI tract neoplasms by damaging DNA, activating oncogenic signaling pathways, producing tumor-promoting metabolites, such as secondary bile acids, and suppressing antitumor immunity29. Host metabolism and inter-microbial interactions in the cancer microbiota have been recognized gradually, but the questions on whether H. pylori interacts with the gut microbiota to regulate host metabolism and whether the presence of H. pyloridirectly shapes the gastric microbial composition of the gut microbiome remain unknown30.
Few studies have directly examined the effect of garlic intake on AG, while the effect of garlic intake on gastrointestinal physiology was controversial. The present study found that the association between garlic intake and AG differed by H. pyloriinfectious state. People in the infected or eradicated states are at a higher risk for AG associated with garlic intake. Garlic has been studied for its antimicrobial, antifungal, antihypertensive, hypoglycemic, and antitumor properties31. In addition to amino acids, vitamins, and micronutrients, garlic contains various organosulfur compounds including diallyl sulfide32. Experimental and human studies suggested that these organosulfur compounds in garlic have chemopreventive and other beneficial effects31,32,33, affecting each stage of carcinogenesis and influencing physiologic processes that modify cancer risk34. A gastroscopic screening survey in Cangshan County have found that infection with H. pyloriis a risk factor and garlic may be protective, in the development and progression of advanced precancerous gastric lesions in an area of China at relatively low risk of GC35. A randomized intervention trial conducted in Linqu County, Shandong province, China, have reported that H pyloritreatment for two weeks and vitamin or garlic supplementation for seven years were associated with a statistically significant reduced risk of death due to gastric cancer for more than 22 years36. However, The findings from two large prospective US cohort studies (the Nurses’ Health Study and the Health Professionals Follow-up Study) do not support the hypothesis that high garlic intake reduces risk of gastric cancer37. Our findings suggests that diet may regulate the pathogenic role of H. pylori and intestinal flora, and individualized dietary prescriptions for populations with different H. pylori infection state could provide direction for future studies. For example, dietary prescriptions for people in the H. pylori infected or eradicated states could be explored in clinical practice, with recommendations to reduce garlic intake and thereby reduce the risk of AG. Future research deserves to continue to explore how these results can be translated into clinical practice, specifically regarding the integration of dietary interventions into the prevention and management of AG.
Despite the high prevalence of H. pylori, Indian individuals have a low gastric cancer incidence of less than 10 per 100,000 per year38. The phenomenon of “Indian Enigma” seemed to suggest that diet might play a significant role. Dietary interventions enabled a decrease in H. pyloricolonization and resulted in a decrease in gastritis prevalence, thus potentially reducing the risk of gastric adenocarcinoma development39. Among these, lifestyle factors such as diet were not in the limelight as principal factors that could modulate H. pylori-linked gastric diseases until recently. There are various factors behind this paradoxical outcome, including virulence of the infecting H. pyloristrains, GI microflora, genetic background of the host and factors related to lifestyle, such as dietary habits40. Based on the efficacy and safety of H. pylori eradication and patient dependence, there are three infectious states of H. pylori: uninfected, infected and eradicated states. Environmental factors, particularly dietary habits, may play a critical role in the formation of AG. Therefore, it is necessary to find new non-antimicrobial ways to prevent microbe-associated AG and to control H. pylori-related chronic inflammatory processes and mediators responsible for carcinogenesis41. Dietary interventions decrease AG prevalence when the stomach microenvironment is colonized by H. pylori, and it also potentially lowers the risk for gastric adenocarcinoma development40,42. Available evidence strongly supports the notion that diet may play a critical role in defining the ultimate outcome of H. pyloriinfection, particularly if certain dietary components are taken on a regular basis for a long period of time41. We found that alcohol consumption was a significant risk factor for AG in the absence of H. pylori infection. However, a population-based study conducted in Saarland, Germany, reported an inverse association with AG for moderate alcohol consumption of both beer and wine, partly by facilitating the elimination of H. pylori43. In addition, alcohol consumption was not associated with an increased risk of gastric cardia adenocarcinoma or gastric non-cardia adenocarcinoma in a large US cohort, the NIH-AARP Diet and Health Study44. This suggests that the associations between alcohol consumption and H. pylori as well as atrophic gastritis and gastric cancer remain still controversial and need to be investigated further in the future.
There are many chemical constituents in garlic, such as sulfur-containing organic compounds and saponins, and sulfur-containing organic compounds are important active substances. These sulfur-containing organic compounds include garlic amine and allicin, which has hypolipidemic40, antioxidant, free radical scavenging45and anti-tumor actions46. Researchers have also found that garlic and its products have anti-bacterial effects against H. pyloriresiding in the stomach47,48. Allitridi-induced protein alterations, indicating that the antibacterial mechanism of allicin on H. pylori, may be related to its multi-target inhibition in energy metabolism and biosynthesis as assessed by proteomic analysis. It was also found that allitridi decreased production of virulence factors49.
The effects of garlic and its products on H. pyloriand other gastric microorganisms in the GI microenvironment remains unclear. Due to the controversial reports on the effects of garlic and its products on AG, a case-control study in Cangshan County, Shandong Province, China, was conducted and showed that garlic had a protective effect on AG in a low-incidence area of gastric cancer35. Another Japanese scholar found that garlic had no protective effect on AG25, and garlic supplementation had no beneficial effects on the prevalence of precancerous gastric lesions or on gastric cancer incidence50. A study based on the Taskopru population found that garlic intake for long durations did not appear to have an effect on the prevalence of H. pylori infection, and garlic consuming subjects had a significantly lower average antibody titer than non garlic consuming groups, which might suggest an indirect inhibitory effect on the reproduction of H. pyloriand possibly progression to more serious peptic ulcer diseases51. Therefore, the long-term effectiveness and safety of AG prevention via H. pylorieradication alone needs to be further verified1,52,53,54. There was also insufficient evidence that garlic consumption reduced H. pyloriinfection55. However, dietary intervention based on the microbial status inside the body is essential56.
In the present study, it was found that the micro-ecological environment in the GI tract is different under the different infection states of H. pylori, so different individualized dietary patterns should be adjusted based on the infection state. In different H. pylori infection states, and in the primary prevention of AG, different dietary effects are also different. In this study, it was found that there was no significant association between AG and garlic intake in the H. pylori uninfected state; however, garlic intake was a significant risk factor in the infected and eradicated states. We also found that alcohol consumption was a significant risk factor for AG in the H. pylori uninfected state but not in the infected and eradicated states.
The principal risk factors for AG in the real-world environment were comprehensively analyzed and relevant conclusions were drawn in this study. However, there were some limitations in the study that should be acknowledged. First, the absence of direct measurements of gastric microbiota composition in the different H. pylori infection states affects the experimental support of this study, which also may weaken the interpretation that garlic intake affects AG progression through microbiota modulation. Because this is an observational study, the research hypothesis should be confirmed in further studies. In the future, the study of GI microflora macrogenomes and dietary regulation with respect to AG will be explored. In addition, the effect of garlic intake on H. pylori infection was not analyzed as H. pylori infection was the basis for the population grouping in this study. Further research is needed to clarify how garlic intake alters the effects of H. pylori infection. Furthermore, limitations of the sample source may limit the extrapolation of our findings. Future studies are warranted to further investigate this issue in the whole population rather than in the high-risk population. Finally, the retrospective nature of this study could introduce information bias, despite the strict design and conduct of the study.
Conclusion
The association between garlic intake and AG differed by H. pylori infectious state. People in the infected or eradicated states are at a higher risk for AG associated with garlic intake. Diet may regulate the pathogenic role of H. pylori and intestinal flora, and individualized dietary prescriptions for populations with different H. pylori infection state could provide direction for future studies. Future research deserves to continue to explore how these results can be translated into clinical practice, specifically regarding the integration of dietary interventions into the prevention and management of AG.
Data availability
All data and materials generated and analyzed during the present study are available from the corresponding author upon reasonable request.
References
World Health Organization Internal Association of Cancer Registries. Available online: http://globocan.iarc.fr/Pages/age-specific_table_sel.aspx (accessed on 15 February 2019).
Liou, J. M., Lee, Y. C., El-Omar, E. M. & Wu, M. S. Efficacy and Long-Term Safety of H. pylori Eradication for Gastric Cancer Prevention. Cancers 11, (2019). https://doi.org/10.3390/cancers11050593
Yu, Y. et al. Risk factors for gastric intraepithelial neoplasia in Chinese adults: a case-control study. Cancer Manage. Res. 10, 2605–2613. https://doi.org/10.2147/cmar.S166472 (2018).
Tan, N. et al. Global, regional, and National burden of early-onset gastric cancer. Cancer Biology Med. 21, 667–678. https://doi.org/10.20892/j.issn.2095-3941.2024.0159 (2024).
Danpanichkul, P. et al. Epidemiology of Gastrointestinal cancers: a systematic analysis from the global burden of disease study 2021. Gut https://doi.org/10.1136/gutjnl-2024-333227 (2024).
Whiting, J. L., Sigurdsson, A., Rowlands, D. C., Hallissey, M. T. & Fielding, J. W. The long term results of endoscopic surveillance of premalignant gastric lesions. Gut 50, 378–381. https://doi.org/10.1136/gut.50.3.378 (2002).
Kato, I. et al. A comparative case-control analysis of stomach cancer and atrophic gastritis. Cancer Res. 50, 6559–6564 (1990).
Vannella, L. et al. Risk factors for progression to gastric neoplastic lesions in patients with atrophic gastritis. Aliment. Pharmacol. Ther. 31, 1042–1050. https://doi.org/10.1111/j.1365-2036.2010.04268.x (2010).
Takahashi, S. Long-term Helicobacter pylori infection and the development of atrophic gastritis and gastric cancer in Japan. J. Gastroenterol. 37 Suppl 13, 24–27. https://doi.org/10.1007/bf02990095 (2002).
Ohkuma, K. et al. Association of Helicobacter pylori infection with atrophic gastritis and intestinal metaplasia. J. Gastroenterol. Hepatol. 15, 1105–1112. https://doi.org/10.1046/j.1440-1746.2000.02305.x (2000).
Kuipers, E. J. & Appelmelk, B. J. Helicobacter pylori and atrophic gastritis. Biomed. Pharmacotherapy = Biomedecine Pharmacotherapie. 51, 150–155. https://doi.org/10.1016/s0753-3322(97)85582-5 (1997).
Li, Y. et al. Global prevalence of Helicobacter pylori infection between 1980 and 2022: a systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 8, 553–564. https://doi.org/10.1016/s2468-1253(23)00070-5 (2023).
Lam, S. Y., Yu, J., Wong, S. H., Peppelenbosch, M. P. & Fuhler, G. M. The Gastrointestinal microbiota and its role in oncogenesis. Best Pract. Res. Clin. Gastroenterol. 31, 607–618. https://doi.org/10.1016/j.bpg.2017.09.010 (2017).
Meng, C., Bai, C., Brown, T. D., Hood, L. E. & Tian, Q. Human gut microbiota and Gastrointestinal Cancer. Genom. Proteom. Bioinform. 16, 33–49. https://doi.org/10.1016/j.gpb.2017.06.002 (2018).
Bik, E. M. et al. Molecular analysis of the bacterial microbiota in the human stomach. Proc. Natl. Acad. Sci. U.S.A. 103, 732–737. https://doi.org/10.1073/pnas.0506655103 (2006).
Tohumcu, E. et al. Helicobacter pylori and the human Gastrointestinal microbiota: A multifaceted relationship. Antibiot. (Basel Switzerland). 13 https://doi.org/10.3390/antibiotics13070584 (2024).
Delgado, S., Cabrera-Rubio, R., Mira, A., Suárez, A. & Mayo, B. Microbiological survey of the human gastric ecosystem using culturing and pyrosequencing methods. Microb. Ecol. 65, 763–772. https://doi.org/10.1007/s00248-013-0192-5 (2013).
Engstrand, L. & Lindberg, M. Helicobacter pylori and the gastric microbiota. Best Pract. Res. Clin. Gastroenterol. 27, 39–45. https://doi.org/10.1016/j.bpg.2013.03.016 (2013).
Tsugane, S. & Sasazuki, S. Diet and the risk of gastric cancer: review of epidemiological evidence. Gastric Cancer: Official J. Int. Gastric Cancer Association Japanese Gastric Cancer Association. 10, 75–83. https://doi.org/10.1007/s10120-007-0420-0 (2007).
Han, Y. M. et al. Dietary, non-microbial intervention to prevent Helicobacter pylori-associated gastric diseases. Annals Translational Med. 3 https://doi.org/10.3978/j.issn.2305-5839.2015.03.50 (2015).
Rezaeimanesh, N., Farzi, N., Pirmanesh, S., Emami, S. & Yadegar, A. Management of multi-drug resistant Helicobacter pylori infection by supplementary, complementary and alternative medicine; a review. Gastroenterology and hepatology from bed to bench 10, S8-s14 (2017).
O’Gara, E. A., Maslin, D. J., Nevill, A. M. & Hill, D. J. The effect of simulated gastric environments on the anti-Helicobacter activity of Garlic oil. J. Appl. Microbiol. 104, 1324–1331. https://doi.org/10.1111/j.1365-2672.2007.03637.x (2008).
Hasna, B. et al. In vitro and in vivo study of combined effect of some Algerian medicinal plants and probiotics against Helicobacter pylori. Microorganisms 11 https://doi.org/10.3390/microorganisms11051242 (2023).
Ma, J. L. et al. Fifteen-year effects of Helicobacter pylori, Garlic, and vitamin treatments on gastric cancer incidence and mortality. J. Natl Cancer Inst. 104, 488–492. https://doi.org/10.1093/jnci/djs003 (2012).
Kuwahara, Y. et al. Relationship between serologically diagnosed chronic atrophic gastritis, Helicobacter pylori, and environmental factors in Japanese men. Scand. J. Gastroenterol. 35, 476–481. https://doi.org/10.1080/003655200750023723 (2000).
Tong, Q. Y. et al. Gastric intestinal metaplasia: progress and remaining challenges. J. Gastroenterol. 59, 285–301. https://doi.org/10.1007/s00535-023-02073-9 (2024).
Wu, J. Y., Lee, Y. C. & Graham, D. Y. The eradication of Helicobacter pylori to prevent gastric cancer: a critical appraisal. Expert Rev. Gastroenterol. Hepatol. 13, 17–24. https://doi.org/10.1080/17474124.2019.1542299 (2019).
Scott, A. J. et al. International Cancer Microbiome consortium consensus statement on the role of the human Microbiome in carcinogenesis. Gut 68, 1624–1632. https://doi.org/10.1136/gutjnl-2019-318556 (2019).
Mima, K. et al. The role of intestinal bacteria in the development and progression of Gastrointestinal tract neoplasms. Surg. Oncol. 26, 368–376. https://doi.org/10.1016/j.suronc.2017.07.011 (2017).
Shiotani, A., Cen, P. & Graham, D. Y. Eradication of gastric cancer is now both possible and practical. Sem. Cancer Biol. 23, 492–501. https://doi.org/10.1016/j.semcancer.2013.07.004 (2013).
Ali, M., Thomson, M. & Afzal, M. Garlic and onions: their effect on eicosanoid metabolism and its clinical relevance. Prostaglandins Leukot. Essent. Fat. Acids. 62, 55–73. https://doi.org/10.1054/plef.1999.0124 (2000).
Galeone, C. et al. Onion and Garlic use and human cancer. Am. J. Clin. Nutr. 84, 1027–1032. https://doi.org/10.1093/ajcn/84.5.1027 (2006).
Guercio, V., Galeone, C., Turati, F. & La Vecchia, C. Gastric cancer and allium vegetable intake: a critical review of the experimental and epidemiologic evidence. Nutr. Cancer. 66, 757–773. https://doi.org/10.1080/01635581.2014.904911 (2014).
Nicastro, H. L., Ross, S. A. & Milner, J. A. Garlic and onions: their cancer prevention properties. Cancer Prev. Res. (Philadelphia Pa). 8, 181–189. https://doi.org/10.1158/1940-6207.Capr-14-0172 (2015).
You, W. C. et al. Helicobacter pylori infection, Garlic intake and precancerous lesions in a Chinese population at low risk of gastric cancer. Int. J. Epidemiol. 27, 941–944. https://doi.org/10.1093/ije/27.6.941 (1998).
Li, W. Q. et al. Effects of Helicobacter pylori treatment and vitamin and Garlic supplementation on gastric cancer incidence and mortality: follow-up of a randomized intervention trial. BMJ (Clinical Res. ed.). 366, l5016. https://doi.org/10.1136/bmj.l5016 (2019).
Kim, H., Keum, N., Giovannucci, E. L., Fuchs, C. S. & Bao, Y. Garlic intake and gastric cancer risk: results from two large prospective US cohort studies. Int. J. Cancer. 143, 1047–1053. https://doi.org/10.1002/ijc.31396 (2018).
Goh, K. L. Lessons learnt from the epidemiology of Helicobacter pylori infection in Malaysia: JGHF Marshall and Warren lecture 2017. J. Gastroenterol. Hepatol. 33, 1177–1184. https://doi.org/10.1111/jgh.14131 (2018).
Hołubiuk, Ł. & Imiela, J. Diet and Helicobacter pylori infection. Przeglad Gastroenterologiczny. 11, 150–154. https://doi.org/10.5114/pg.2016.61487 (2016).
Zaidi, S. F., Ahmed, K., Saeed, S. A., Khan, U. & Sugiyama, T. Can diet modulate Helicobacter pylori-associated gastric pathogenesis?? An Evidence-Based analysis. Nutr. Cancer. 69, 979–989. https://doi.org/10.1080/01635581.2017.1359310 (2017).
Lee, S. Y., Shin, Y. W. & Hahm, K. B. Phytoceuticals: mighty but ignored weapons against Helicobacter pylori infection. J. Dig. Dis. 9, 129–139. https://doi.org/10.1111/j.1751-2980.2008.00334.x (2008).
Elkayam, A., Peleg, E., Grossman, E., Shabtay, Z. & Sharabi, Y. Effects of allicin on cardiovascular risk factors in spontaneously hypertensive rats. Isr. Med. Association Journal: IMAJ. 15, 170–173 (2013).
Gao, L., Weck, M. N., Stegmaier, C., Rothenbacher, D. & Brenner, H. Alcohol consumption and chronic atrophic gastritis: population-based study among 9,444 older adults from Germany. Int. J. Cancer. 125, 2918–2922. https://doi.org/10.1002/ijc.24618 (2009).
Wang, S., Freedman, N. D., Loftfield, E., Hua, X. & Abnet, C. C. Alcohol consumption and risk of gastric cardia adenocarcinoma and gastric noncardia adenocarcinoma: A 16-year prospective analysis from the NIH-AARP diet and health cohort. Int. J. Cancer. 143, 2749–2757. https://doi.org/10.1002/ijc.31740 (2018).
Hui, C. et al. S-allyl-L-cysteine sulfoxide inhibits tumor necrosis factor-alpha induced monocyte adhesion and intercellular cell adhesion molecule-1 expression in human umbilical vein endothelial cells. Anat. Record (Hoboken N J. : 2007). 293, 421–430. https://doi.org/10.1002/ar.21070 (2010).
Zhang, W. et al. Allicin induces apoptosis in gastric cancer cells through activation of both extrinsic and intrinsic pathways. Oncol. Rep. 24, 1585–1592. https://doi.org/10.3892/or_00001021 (2010).
Rana, S. V., Pal, R., Vaiphei, K. & Sharma, S. K. Ola, R. P. Garlic in health and disease. Nutr. Res. Rev. 24, 60–71. https://doi.org/10.1017/s0954422410000338 (2011).
Zardast, M., Namakin, K., Esmaelian Kaho, J. & Hashemi, S. S. Assessment of antibacterial effect of Garlic in patients infected with Helicobacter pylori using urease breath test. Avicenna J. Phytomedicine. 6, 495–501 (2016).
Liu, S. et al. The antibacterial mode of action of allitridi for its potential use as a therapeutic agent against Helicobacter pylori infection. FEMS Microbiol. Lett. 303, 183–189. https://doi.org/10.1111/j.1574-6968.2009.01877.x (2010).
You, W. C. et al. Randomized double-blind factorial trial of three treatments to reduce the prevalence of precancerous gastric lesions. J. Natl Cancer Inst. 98, 974–983. https://doi.org/10.1093/jnci/djj264 (2006).
Salih, B. A. & Abasiyanik, F. M. Does regular Garlic intake affect the prevalence of Helicobacter pylori in asymptomatic subjects? Saudi Med. J. 24, 842–845 (2003).
Adachi, K., Notsu, T., Mishiro, T. & Kinoshita, Y. Long-term effect of Helicobacter pylori eradication on prevalence of reflux esophagitis. J. Gastroenterol. Hepatol. 34, 1963–1967. https://doi.org/10.1111/jgh.14730 (2019).
Curado, M. P., de Oliveira, M. M. & de Araújo Fagundes, M. Prevalence of Helicobacter pylori infection in Latin America and the Caribbean populations: A systematic review and meta-analysis. Cancer Epidemiol. 60, 141–148. https://doi.org/10.1016/j.canep.2019.04.003 (2019).
Matsuno, K. et al. Time trends in the incidence of esophageal adenocarcinoma, gastric adenocarcinoma, and superficial esophagogastric junction adenocarcinoma. J. Gastroenterol. 54, 784–791. https://doi.org/10.1007/s00535-019-01577-7 (2019).
Li, Z., Ying, X., Shan, F. & Ji, J. The association of Garlic with Helicobacter pylori infection and gastric cancer risk: A systematic review and meta-analysis. Helicobacter 23, e12532. https://doi.org/10.1111/hel.12532 (2018).
Singh, R. K. et al. Influence of diet on the gut Microbiome and implications for human health. J. Translational Med. 15, 73. https://doi.org/10.1186/s12967-017-1175-y (2017).
Acknowledgements
We thank the participatory patients and their supportive families, as well as our colleagues and Tumor Registration Area of Gansu Province.
Funding
The study was supported by Regional Science Foundation Project of National Natural Science Foundation of China (NO. 82160900), the National Public Welfare Industry Research Project, the Project of Industrial Support Program for colleges and universities in Gansu province (No. 2020 C-36).
Author information
Authors and Affiliations
Contributions
WJ, ZJ, JL, WX and LY contributed to the study design and data analyses and drafted the paper. WJ and LY confirm the authenticity of all the raw data, all authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was conducted according to the guidelines laid down in the Declaration of Helsinki and all procedures involving research study participants were approved by the Ethics Committee of National Cancer Center, Chinese Academy of Medical Sciences (2015SQ00223), and all subjects signed the written informed consent before participating this study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wu, J., Zhang, J., Jin, L. et al. Analysis of garlic intake on atrophic gastritis risk in different infectious states of Helicobacter pylori in a case-control study. Sci Rep 15, 8481 (2025). https://doi.org/10.1038/s41598-025-92376-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-92376-z