Abstract
Cisplatin-based chemotherapy is the standard advanced esophageal squamous cell carcinoma (ESCC) treatment. However, the 5-fluorouracil plus L-leucovorin and oxaliplatin (FOLFOX) regimen is available in Japan as an alternative to cisplatin-based chemotherapy, but its efficacy and safety remain unclear. Hence, we aimed to evaluate patients with advanced ESCC who received FOLFOX therapy retrospectively. Patients with advanced ESCC who received FOLFOX therapy at 18 institutions between April 2019 and October 2020 were included. Fifty-two patients received first-line FOLFOX treatment, while 39 received later-line FOLFOX treatment. The median progression-free survival, median overall survival, and objective response rate were 3.8 months (95% confidence interval [CI]: 3.0–5.0), 13.9 months (95% CI: 10.3–17.9 months), and 35% in the first-line group and 2.4 months (95% CI: 2.1–3.8 months), 7.2 months (95% CI: 5.3–9.8 months), and 4% in the later-line group, respectively. The most frequent grade 3 or 4 adverse events were neutropenia (23%) and anemia (12%) in the first-line group and neutropenia (18%), anorexia (13%), anemia (10%), and nausea (10%) in the later-line group. First-line FOLFOX treatment is suitable for patients with advanced ESCC. However, FOLFOX has shown limited efficacy as a later-line treatment.
Similar content being viewed by others
Introduction
Cisplatin plus 5-fluorouracil (5-FU) (CF) treatment has been used as a first-line therapy1,2; however, no conclusive evidence substantiates the survival advantages of CF in patients with unresectable advanced or recurrent esophageal squamous cell carcinoma (ESCC). Taxanes and other agents have been used as second-line chemotherapies for patients who are unresponsive to combination therapies3,4. Recently, compared with CF therapy, CF plus immune checkpoint inhibitors (ICIs) or dual ICI combinations have been shown to prolong overall survival (OS). Therefore, CF plus ICIs or a combination of dual ICIs has become the standard treatment for patients with advanced ESCC5,6. However, although cisplatin-based treatment has demonstrated significant efficacy, cisplatin itself can pose challenges for certain patients, such as elderly individuals or those with renal dysfunction, owing to the requirement for hydration and the high risk of toxicity1,2,5,6,7. Consequently, there is an unmet medical need to establish a standard therapy suitable for patients who cannot tolerate cisplatin. Oxaliplatin is a platinum-based cytotoxic agent used as an alternative to cisplatin in treating gastric cancer8. In a previous study of patients with ESCC, 5-FU plus l-leucovorin and oxaliplatin (FOLFOX) therapy showed efficacy comparable to that of CF therapy9,10. Although FOLFOX therapy can be utilized for advanced ESCC, no previous report demonstrates its efficacy and safety in patients with advanced ESCC who are unsuitable for cisplatin therapy.
Another challenge in treating advanced ESCC is the limited availability of effective drugs. Oxaliplatin is effective in patients with cisplatin-resistant gastric cancer11,12; however, no study has evaluated the efficacy and safety of FOLFOX therapy in heavily treated patients with advanced ESCC.
Hence, elucidating the efficacy and safety of FOLFOX therapy in first-line settings with advanced ESCC that are unsuitable for cisplatin therapy and later-line settings for advanced ESCC is very important, and a Japanese multicenter retrospective study serves as a potential solution to address these unmet medical needs.
Materials and methods
Patients
Between April 2019 and October 2020, patients with histologically confirmed esophageal squamous cell carcinoma, clinical stage IVB (Union for International Cancer Control tumor-node-metastasis classification in 8th edition) or recurrence, an Eastern Cooperative Oncology Group (ECOG) performance status (PS) score of 0–2, an age ≥ 20 years, and an evaluable lesion treated with FOLFOX as a first-line or later-line treatment were considered eligible for this study. The patients with cisplatin intolerance were eligible for first-line treatment. In the present study, cisplatin intolerance was defined as. (i) renal dysfunction (creatinine clearance (CCr) < 60 ml/min), (ii) elderly (> 75 years), (iii) ECOG, PS2, (iv) pleural or ascites effusion, (v) cardiac dysfunction (Ejection fraction < 60%), (vi) cisplatin use as perioperative chemotherapy and intolerance. First-line chemotherapy was defined as the absence of previous chemotherapy for unresectable or recurrent ESCC or chemotherapy administered more than 6 months after the last dose of perioperative chemotherapy. Later-line chemotherapy was defined as previous chemotherapy for unresectable or recurrent ESCC or chemotherapy administered as a treatment for recurrence that occurred within 6 months after the last dose of perioperative chemotherapy. Patients for whom FOLFOX therapy was combined with radiation or immune checkpoint inhibitors were excluded. Patients with two cancers were excluded. This study was conducted at 18 different institutions in Japan, and the clinical data were analyzed retrospectively. Due to the retrospective nature of the study, it was approved by the review board of the National Hospital Organization of Nagoya Medical Center that the need to obtain informed consent was waived and that the opt-out methodology was acceptable. Patient participation was obtained through an opt-put methodology. This study followed the principles of the Helsinki Declaration and was approved by the review board of the National Hospital Organization of Nagoya Medical Center (2021-019).
FOLFOX regimen
The FOLFOX regimen consisted of 85 mg/m2 oxaliplatin (L-OHP), 200 mg/m2 leucovorin, and a bolus injection of 5-fluorouracil 400 mg/m2 intravenously on day 1, followed by a 46-h continuous infusion of 5-FU 2,400 mg/m2 every 2 weeks. The criteria for dose reduction of each drug and the amount of dose reduction were determined by each physician. Treatment was continued until obvious disease progression occurred, unacceptable toxicity developed, or the patient refused to continue treatment.
Assessments and statistical criteria
OS was defined as the period from the date of treatment initiation to the date of death. Progression-free survival (PFS) was defined as the period from the date of treatment initiation to the date of confirmed progression, recurrence, or death. Moreover, the date of progression may also be defined as the clinical progression of the disease (PD) when no apparent progression was detected on imaging examination. Still, the patient was considered to have progressed as judged by each treating physician. The following analyses were performed to assess OS and PFS. Additional analyses of OS and PFS above and below the CCr cutoff value of 60 ml/min, which is one of the criteria for determining cisplatin dosage, and OS and PFS in patients aged ≥ 70 years and ≤ 70 years were performed. CCr was estimated using the Cockcroft–Gault equation.
The response rate was calculated using Response Evaluation Criteria in Solid Tumors version 1.1 and endoscopic evaluation of the primary tumor. The best response to FOLFOX treatment was detected in patients with target lesions. The timing of the evaluations was not specified. The Glasgow prognostic score was calculated based on a score of 2 for patients with a serum C-reactive protein level > 10 mg/L and hypoalbuminemia ≤ 3.5 g/L.
The Japanese version of the Common Terminology Criteria for Adverse Events (version 5.0) developed by the Japan Clinical Oncology Group was used to evaluate adverse events (AEs).
The median OS and PFS were evaluated using Kaplan–Meier curves. Univariate and multivariate Cox regression analyses were performed to determine the factors that affected OS and PFS in patients who received later-line FOLFOX treatment and the variables with a p-value ≤ 0.01 in the univariate analyses included in the multivariate analyses. A two-sided p-value of 0.05 or less was considered significant. All the data were analyzed using EZR version 4.2.213.
Results
Patient characteristics
Fifty-two patients received first-line treatment, while 39 patients received later-line treatment. The median ages were 72 years (46–85 years) in the first-line group and 68 years (47–85 years) in the later-line group. The median CCr was 50 ml/min (21–100 mL/min) in the first-line group. The numbers of patients resistant to and intolerant to cisplatin in the later-line group were 35 and 4, respectively (Table 1). Moreover, FOLFOX was used in the first-line group for the following reasons: renal dysfunction (n = 27), old age (n = 11), ECOG PS performance status score of 2 (n = 5), pleural effusion or ascites (n = 5), cardiac dysfunction (n = 5), and cisplatin intolerance, based on the physicians’ judgment. Duplicate responses were noted, and no clear numerical criteria were established for each reason.
Efficacy
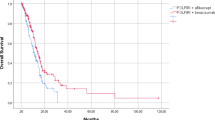
The median follow-up times were 14.7 months in the first-line group and 19.9 months in the later-line group. In the first-line group, the median OS and median PFS were 13.9 months (95% confidence interval [CI]: 10.3–17.9 months) and 3.8 months (95% CI: 3.0–5.0 months), respectively (Fig. 1. A and B). The median OS and median PFS by age group (≥ 70 years and < 70 years) were 13.9 months and 16.6 months (hazard ratio [HR]: 0.87, p = 0.93) and 3.8 months and 5.0 months (HR: 1.19, p = 0.59), respectively. The median OS and PFS according to the CCr level (≥ 50 ml/min and <50 ml/min) were 13.9 months and 14.4 months (HR: 1.16, p = 0.66), and 3.5 months and 5.1 months (HR: 1.36, p = 0.31), respectively. Univariate analysis of several factors was performed; however, neither renal function nor advanced age significantly impacted OS or PFS (Table 2A).
Kaplan–Meier analysis. (a) OS in the first-line group. (b) Progression-free survival in the first-line group. CI, confidence interval; OS, overall survival; PFS, progression-free survival.
In the later-line group, the median OS and median PFS were 7.4 months (95% CI: 5.3–10.1 months) and 2.3 months (95% CI: 2.1–3.6 months), respectively (Fig. 2. A and B). In cases where long-term survival was achieved in patients with a history of prior ICI, we examined the effect of treatment with a history of ICI administration. Eleven patients (28%) received prior ICI treatment, and no differences were found in the median OS (7.2 vs. 7.1 months) or median PFS (2.3 vs. 2.1 months) between patients with and without a history of ICI use. One patient was in prior treatment with S-1 monotherapy. The best tumor response was stable disease; PFS and OS were 3.5 months and 8.7 months, respectively. In addition, uni-multivariate analysis revealed that GPS was the only predictor of longer PFS. (HR = 2.30, p = 0.04) (Table 2B).
Kaplan–Meier analysis. (a) OS in the later-line group. (b) Progression-free survival in the later-line group. CI, confidence interval; OS, overall survival; PFS, progression-free survival.
With respect to tumor response, 48 patients in the first-line treatment group and 25 patients in the later-line treatment group with target lesions were evaluated. In the first-line group, 1 patient (2%) achieved a complete response (CR), while 16 patients (33%) achieved a partial response (PR); the ORR was 35% (95% CI: 23–50%), while the disease control rate (DCR) was 67% (95% CI: 52–78%). In the later-line group, one patient (4%) achieved a PR; the ORR was 4%, while the DCR was 36%. No differences were found in the ORR (0% vs. 5%) or DCR (43% vs. 33%) between patients with and without a history of ICI use.
Toxicity
The treatment-related AEs are listed in Table 3. In the first-line treatment group, the most frequent grade 3 or higher AEs were neutropenia (23%), anemia (12%), and anorexia (10%). No treatment-related deaths occurred. In the later-line treatment group, the most frequent grade 3 or higher AEs were neutropenia (18%), anorexia (13%), anemia (10%), and nausea (10%); similarly, no treatment-related deaths occurred (Table 3).
The reasons for treatment discontinuation were progression (n = 43, 83%), patient desire (n = 5, 10%), toxicity (n = 3, 6%), achieved CR (n = 1, 2%) in first-line group and progression (n = 38, 97%), toxicity (n = 1, 3%) in later-line group.
The number of dose reductions from the initial treatment was 11 patients (21%) in the first-line group and 7 patients (18%) in the later-line group.
Subsequent treatment
At the end of follow-up, 5 patients in the first-line group continued FOLFOX treatment; 37 received subsequent treatment, while 10 received best supportive care (BSC). Furthermore, 27 patients (73%) received second-line treatment, 20 of whom received nivolumab and 7 of whom received paclitaxel.
At the end of the follow-up, all patients in the later-line group discontinued FOLFOX treatment; 16 received subsequent treatment, while 23 received BSC. The subsequent treatments administered included nivolumab (n = 8, 30%), paclitaxel (n = 3, 8%), docetaxel in two (5%) patients, S-1 in two (5%) patients, and other treatments in three (8%) patients.
Discussion
This study is the first to report the efficacy and safety of FOLFOX therapy as a first-line chemotherapy for patients with advanced ESCC who were unsuitable for cisplatin and as a later-line chemotherapy for heavily treated patients. The median OS, median PFS, and ORR in patients who received CF as first-line therapy were 10.4–11.7 months, 4.3–6.1 months, and 24.1–40.7%, respectively7,14,15. The median PFS and ORR were consistent with those reported in previous studies. Despite having greater median age than the CF therapy group in the KEYNOTE-590 trial (68 years)14, CheckMate 648 trial (67 years)15, and a previous retrospective study (62 years)7, as well as obtaining a median CCr of 50 ml/min, encompassing 63% of the patients with a CCr < 60 mL/min, the first-line group in the current study exhibited longer OS than did the first-line group in previous studies. Univariate analysis indicated that age and renal function did not influence OS or PFS. Furthermore, there were some cases in which a patient with ICI as second-line therapy achieved long-term survival, and the rate of subsequent chemotherapy administration reached 79%, surpassing the figures reported in previous studies (68–75%)7,14,15, potentially contributing to the extension of OS.
In the present study, the incidence rates of nausea (52.6–62.7%)14,15, anorexia (58.2–60.0%)14,15, and diarrhea (17.8–26.9%) of any grade14,15 were lower than those reported in previous CF therapy studies. The incidence of hematologic toxicities increased, although all events were manageable, and no treatment-related fatalities were reported in either the first- or later-line groups.
However, L-OHP showed a partial lack of cross-resistance to cisplatin both in vitro and in vivo. Previous studies in patients with advanced gastric cancer showed modest efficacy of FOLFOX therapy in patients who were heavily treated and had a history of cisplatin administration11,12. Therefore, FOLFOX therapy can demonstrate antitumor activity in heavily treated patients, including those who have received cisplatin. Although some previous studies have reported the efficacy and safety of oxaliplatin in heavily pretreated patients with other types of carcinomas11,12,16,17, its efficacy and safety in treating ESCC remain uncertain. In previous studies, S-1, an oral fluoropyrimidine drug, demonstrated modest efficacy in pretreated advanced ESCC patients18,19,20,21,22 (Table 4). The present study showed that FOLFOX was less effective against ESCC than previous studies. If the patient can take oral medication, S-1 should be used in the fourth-line treatment, followed by FOLFOX therapy. If the patient is uncertain about oral medication, it should be used in the fourth-line treatment. However, the results S-1 may have been obtained in patients who could take oral medication and whose condition was maintained to some extent, even though they were in the later-line setting, so caution is needed in interpreting the comparison of efficacy with FOLFOX.
When the ORR, PFS, and OS reported in the current studies about taxanes were 7.7–29.6%, 2.1–4.9 months, and 7.3–9.9 months23,24. Although direct comparisons cannot be made because of differences in patient backgrounds, such as subjects and treatment lines, the results are generally considered in the same range as previously reported results and may be an option for backward treatment.
However, this study has several limitations. First, the sample size was relatively small. Second, cisplatin intolerance has not been defined, and the indications for FOLFOX use have not been established. Third, the doses were adjusted at the discretion of the individual physicians, which may have affected the efficacy.
Conclusions
FOLFOX therapy may be used as a first-line treatment for patients with advanced ESCC who are unsuitable for cisplatin, such as those with renal dysfunction or older patients; however, FOLFOX therapy has shown limited efficacy as a later-line treatment. A prospective observational study is currently underway, and results are warranted.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Iizuka, T. et al. Phase II evaluation of cisplatin and 5-fluorouracil in advanced squamous cell carcinoma of the esophagus: A Japanese esophageal oncology group trial. Jpn. J. Clin. Oncol. 22, 172–176 (1992).
Hayashi, K. et al. Phase II evaluation of protracted infusion of cisplatin and 5-fluorouracil in advanced squamous cell carcinoma of the esophagus: A Japan esophageal oncology group (JEOG) trial (JCOG9407). Jpn J. Clin. Oncol. 31, 419–423. https://doi.org/10.1093/jjco/hye090 (2001).
Kato, K. et al. A phase II study of Paclitaxel by weekly 1-h infusion for advanced or recurrent esophageal cancer in patients who had previously received platinum-based chemotherapy. Cancer Chemother. Pharmacol. 67, 1265–1272. https://doi.org/10.1007/s00280-010-1422-x (2011).
Muro, K. et al. A phase II study of single-agent docetaxel in patients with metastatic esophageal cancer. Ann. Oncol. 15, 955–959. https://doi.org/10.1093/annonc/mdh231 (2004).
Sun, J. M. et al. Pembrolizumab plus chemotherapy versus chemotherapy alone for first-line treatment of advanced esophageal cancer (KEYNOTE-590): A randomized, placebo-controlled, phase 3 study. Lancet 398, 759–771. https://doi.org/10.1016/S0140-6736(21)01234-4 (2021).
Doki, Y. et al. Nivolumab combination therapy in advanced esophageal squamous-cell carcinoma. N Engl. J. Med. 386, 449–462. https://doi.org/10.1056/NEJMoa2111380 (2022).
Hiramoto, S. et al. A retrospective analysis of 5-fluorouracil plus cisplatin as first-line chemotherapy in the recent treatment strategy for patients with metastatic or recurrent esophageal squamous cell carcinoma. Int. J. Clin. Oncol. 23, 466–472. https://doi.org/10.1007/s10147-018-1239-x (2018).
Yamada, Y. et al. Phase III study comparing oxaliplatin plus S-1 with cisplatin plus S-1 in chemotherapy-naïve patients with advanced gastric cancer. Ann. Oncol. 26, 141–148. https://doi.org/10.1093/annonc/mdu472 (2015).
Adenis, A. et al. Continuation versus discontinuation of First-Line chemotherapy in patients with metastatic squamous cell esophageal cancer: A randomized phase II trial (E-DIS). Eur. J. Cancer. 111, 12–20. https://doi.org/10.1016/j.ejca.2019.01.016 (2019).
Conroy, T. et al. Definitive chemoradiotherapy with FOLFOX versus fluorouracil and cisplatin in patients with esophageal Cancer (PRODIGE5/ACCORD17): final results of a randomized, phase 2/3 trial. Lancet Oncol. 15, 305–314. https://doi.org/10.1016/S1470-2045(14)70028-2 (2014).
Kondoh, C. et al. Salvage chemotherapy with the combination of oxaliplatin, leucovorin, and 5-fluorouracil in advanced gastric cancer refractory or intolerant to fluoropyrimidines, platinum, taxanes, and Irinotecan. Gastric Cancer 21, 1050–1057. https://doi.org/10.1007/s10120-018-0825-y (2018).
Mitani, S. et al. A phase II study of modified FOLFOX6 for advanced gastric cancer refractory to standard therapies. Adv. Ther. 37, 2853–2864. https://doi.org/10.1007/s12325-020-01358-2 (2020).
Kanda, Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transpl. 48, 452–458. https://doi.org/10.1038/bmt.2012.244 (2013).
Kojima, T. et al. First-line pembrolizumab + chemotherapy in Japanese patients with advanced/metastatic esophageal cancer from KEYNOTE-590. Esophagus 19, 683–692. https://doi.org/10.1007/s10388-022-00920-x (2022).
Kato, K. et al. First-Line nivolumab plus ipilimumab or chemotherapy versus chemotherapy alone in advanced esophageal squamous cell carcinoma: A Japanese subgroup analysis of Open-Label, phase 3 trial (CheckMate 648/ONO-4538-50). Esophagus 20, 291–301. https://doi.org/10.1007/s10388-022-00970-1 (2023).
Fracasso, P. M. et al. Phase II study of oxaliplatin in platinum-resistant and refractory ovarian cancer: A gynecologic group study. J. Clin. Oncol. 21, 2856–2859. https://doi.org/10.1200/JCO.2003.03.077 (2003).
Chollet, P. et al. Single agent activity of oxaliplatin in heavily pretreated advanced epithelial ovarian cancer. Ann. Oncol. 7, 1065–1070. https://doi.org/10.1093/oxfordjournals.annonc.a010500 (1996).
Nomura, M. et al. Active salvage chemotherapy versus best supportive care for patients with recurrent or metastatic squamous cell carcinoma of the esophagus refractory or intolerable to fluorouracil, platinum, and taxane. Cancer Chemother. Pharmacol. 78, 1209–1216. https://doi.org/10.1007/s00280-016-3179-3 (2016).
Akutsu, Y. et al. S-1 monotherapy as second- or third-line chemotherapy for unresectable and recurrent esophageal squamous cell carcinoma. Oncology 84, 305–310. https://doi.org/10.1159/000348294 (2013).
Tamaoki, M. et al. S-1 salvage chemotherapy for esophageal squamous cell carcinoma refractory to the standard chemotherapy. J. Cancer Sci. Ther. 09, 353–357. https://doi.org/10.4172/1948-5956.1000441 (2017).
Ito, T. et al. S-1 monotherapy after failure of platinum plus 5-fluorouracil chemotherapy in recurrent or metastatic esophageal carcinoma. Anticancer Res. 39, 3931–3936. https://doi.org/10.21873/anticanres.13545 (2019).
Nomura, M. et al. A phase II study of S-1 therapy for patients with advanced and recurrent esophageal cancer resistant or intolerable to fluorouracil, platinum, and taxane therapy (OGSG 1404). Esophagus 19, 711–716. https://doi.org/10.1007/s10388-022-00931-8 (2022).
Chin, K. et al. Effectiveness of taxanes following nivolumab in patients with advanced esophageal squamous cell carcinoma: A retrospective chart review of patients in ATTRACTION-3. Esophagus 20, 302–308. https://doi.org/10.1007/s10388-022-00972-z (2023).
Yamamoto, S. et al. Randomized phase II study of docetaxel versus Paclitaxel in patients with esophageal squamous cell carcinoma refractory to fluoropyrimidine- and platinum-based chemotherapy: OGSG1201. Eur. J. Cancer 154, 307–315. https://doi.org/10.1016/j.ejca.2021.06.035 (2021).
Acknowledgements
We would like to thank Drs. Sugiyama, Tsuchihashi, and Hirata for their invaluable guidance and insightful discussions pertaining to this study. I would like to express my gratitude to all the doctors who registered their valuable cases and had active discussions with us.
Author information
Authors and Affiliations
Contributions
All the authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Kazuhiro Shiraishi. The first draft of the manuscript was written by Kazuhiro Shiraishi, and all the authors commented on previous versions of the manuscript. All the authors have read the final manuscript and have approved this submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Shiraishi, K., Okada, M., Yamamoto, S. et al. The efficacy and safety of FOLFOX therapy for advanced esophageal squamous cell carcinoma. Sci Rep 15, 8031 (2025). https://doi.org/10.1038/s41598-025-92657-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-92657-7