Abstract
The clinical usefulness of serum leucine-rich alpha-2 glycoprotein (LRG) levels as a surrogate marker of endoscopic activity including postoperative recurrence in patients with Crohn’s disease (CD) remains unclear, and LRG production in the small intestinal mucosa has not been explored. The present study investigated the usefulness of serum LRG to ascertain endoscopic activity, the secretion of LRG from the small intestinal mucosa, and the significance of LRG as a predictor of postoperative disease course. We included 364 patients who underwent transanal endoscopy at Osaka University Hospital. Serum LRG correlated highly with endoscopic severity (LRG, r = 0.65; CRP, r = 0.37) and reflected strictly moderate endoscopic activity better than serum CRP. Especially, serum LRG reflected mucosal healing even in patients whose inflammation was confined to the small intestine. In multivariate analyses, serum LRG was an independent factor influencing mucosal healing. LRG was more strongly expressed in the inflamed mucosa of the small intestine compared with that in uninflamed mucosa, and serum LRG was more strongly correlated with postoperative small intestinal recurrence severity than CRP (LRG, r = 0.62; CRP, r = 0.32). In conclusion, serum LRG is a useful surrogate marker of endoscopic CD severity and activity, with increased LRG expression in the small bowel predicting postoperative recurrence.
Similar content being viewed by others
Introduction
Inflammatory bowel disease (IBD) is broadly classified into Crohn’s disease (CD) and ulcerative colitis (UC) and presents with intestinal symptoms such as diarrhoea, bloody stool, and abdominal pain due to chronic intestinal inflammation of unknown aetiology. IBD impairs the quality of life of patients and its high incidence among the younger generation causes large social losses. In addition, patients can develop cancer owing to long-term chronic inflammation. Thus, it is crucial to treat IBD to improve not only the symptoms but also intestinal inflammation and mucosal healing. The treat to target (T2T) strategy recommends to set specific goals to treat patients with IBD, and improved inflammatory markers have been defined as mid-term goals in IBD treatment to improve long-term disease outcomes1. This is because clinical symptoms in patients with CD poorly correlate with mucosal inflammation severity, making it difficult to assess CD disease activity only with clinical symptoms2,3,4,5. The latest guidelines, therefore, set endoscopic mucosal improvement as a further goal in IBD treatment;1 however, endoscopies are highly invasive. Therefore, identifying non-invasive biomarkers that reflect IBD disease activity is crucial.
The T2T strategy involves setting therapeutic targets using biomarkers such as C-reactive protein (CRP) and faecal calprotectin (FC), leading to appropriate therapeutic interventions to improve the long-term disease course. Leucine-rich alpha-2 glycoprotein (LRG) is a new serum biomarker6 for monitoring IBD activity that was approved for use in June 2020 in Japan. LRG expression is induced by interleukin (IL)-6, as well as tumour necrosis factor (TNF)-α and IL-22 mediated inflammation. In contrast, CRP is produced in the liver upon stimulation with IL-6;7 as such, CRP cannot detect IL-6-independent inflammation. In addition, the serum level of CRP may not be elevated even in case of severe IBD disease activity; some patients show negative CRP results despite endoscopically active disease8. Therefore, serum LRG levels can reflect inflammation in CRP-negative UC9, and be elevated in clinically active CRP-negative CD6. The usefulness of LRG in the endoscopic evaluation of CD activity has been reported. One advantage of LRG over FC is that it can be measured in serum, making it more convenient. However, a limitation of LRG is that its levels may also be elevated in inflammatory diseases other than IBD, such as infectious diseases. Therefore, these conditions should be considered when interpreting LRG levels in clinical practice. LRG is also useful for assessing mucosal healing of small intestine10,11 and predicting endoscopic disease remission after treatment12,13. One report stated that 13.6 µg/mL as the cut-off value for LRG was useful for predicting mucosal healing defined as simple endoscopic sub-score for CD (SES-CD) < 3.13 However, utilities of LRG in more endoscopically severe cases have not been fully assessed. Additionally, the usefulness of LRG as a biomarker of postoperative recurrence remains unclear. Previous studies showed that approximately 80% of CD patients had residual lesions in the small bowel postoperatively14,15. Because clinical symptoms are unreliable in predicting the presence of inflammatory lesions in the small intestine16,17 and symptoms of recurrence are similar to those of postoperative functional obstacles18, monitoring residual lesions in the small intestine after surgery is a crucial issue that needs to be addressed. Although the secretion of LRG from the colonic mucosa of patients with UC has been reported19, whether the small intestinal mucosa secretes LRG is unelucidated. We hypothesised that the inflamed intestinal epithelium in the small bowel may potentially be able to increase serum LRG levels, as serum LRG can indicate intestinal inflammation in the small bowel.
Herein, we aimed to investigate the usefulness of serum LRG in clinical practice to ascertain endoscopic activity in patients with CD. We also aimed to explore the secretion of LRG from the small intestinal mucosa of patients with CD patients and its significance in predicting postoperative disease course.
Materials and methods
Study design and patients
This retrospective, single-centre study included patients with CD with uncomplicated infection who underwent transanal endoscopy between July 2013 and September 2022 at the Osaka University Hospital. Transanal endoscopy was performed with balloon-assisted endoscopy or colonoscopy, depending on the location of the inflammatory lesions. Transanal endoscopy were compared with serum marker results measured within 60 days. The exclusion criteria were as follows: patients who underwent total colectomy or ostomy, and those in whom mucosal inflammation could not be evaluated due to the quality of endoscopy examination or bowel stricture (Fig. 1). This study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Ethics Committee of Osaka University Hospital. Since this is a retrospective study, kindly provide a statement where it is mentioned that informed consent was waived by the Institutional Ethics Committee of Osaka University Hospital.
Study flowchart.
Endoscopic assessment of disease activity
The colon and ileum were evaluated and endoscopic disease severity was assessed using SES-CD. Mucosal healing was defined as SES-CD < 412 and moderate endoscopic disease activity was defined as SES-CD ≥ 720,21 to study the cut-off value of serum LRG in cases with endoscopically more severe disease than in previous reports13. To assess inflammation in the small bowel, patients with inflammation in the colon with SES-CD ≥ 1 were excluded and SES-CD was evaluated for the ileum of patients. For evaluating the postoperative recurrence, we included the patients who had the first postoperative transanal endoscopy from 6 months to 2 years after bowel resection. Postoperatively, endoscopic disease severity was evaluated using the Rutgeerts score (RS) and recurrence was defined as RS ≥ 222. Endoscopic bowel preparation was performed using a polyethylene glycol-based electrolyte solution according to the standard protocol of our hospital. All endoscopists who scored the endoscopic findings were blinded to the serum marker results.
Blood collection and measurement of serum LRG and CRP
Blood samples collected from the patients were promptly centrifuged and stored at -80 °C until further use. Serum LRG concentrations were measured using a NANOPIA LRG Kit based on the latex turbidimetry method (Sekisui Medical, Tokyo, Japan) in clinical practice or using stored serum samples. Serum CRP concentrations were measured in clinical practice using latex immune nephelometry. The lower limits of measurements for LRG and CRP were 5.0 µg/mL and 0.04 mg/dL, respectively. The cut-off value for normal CRP normal levels was defined as 0.2 mg/dL, as previously reported6,9.
Immunohistochemical (IHC) staining of LRG
IHC was performed as previously described23 using endoscopic biopsy samples from CD patients. The specimens were fixed in 10% formaldehyde, embedded in paraffin, cut into 20 μm sections, de-paraffinised in xylene and ethanol, and boiled at 110 °C for 20 min for antigen retrieval. IHC was performed using anti-LRG rabbit monoclonal antibody (1:200, ab178698; Abcam, Chicago, IL), incubated overnight at 4 °C. Sections were stained with the Real Envision Detection System (K5007; Dako Co, Denmark) and 3,3-diaminobenzidine and counterstained with haematoxylin. Inflammatory UC tissues were used as positive controls. LRG expression was categorised into positive and negative groups, as in a previous report24; scores of 1 and 2 were defined as positive, and a score of 0 was defined as negative. Two independent gastroenterologists evaluated the IHC results, and discrepancies were resolved by discussion among the three investigators.
Statistical analysis
Continuous variables are presented as medians and interquartile ranges (IQR). Correlations were assessed using Spearman’s rank-order correlation coefficients. To determine factors associated with endoscopic disease activity, logistic regression was applied. Receiver operating characteristic (ROC) curve analysis was performed to evaluate the cut-off value, and its sensitivity and specificity for endoscopic disease activity were calculated. The area under the curves (AUCs) and 95% confidence interval (CI) were compared using the DeLong method25. P < 0.05 was set as statistical significance. We used the available-case analysis to deal with the missing data. Statistical analyses were performed by JMP statistical software (version 17.1.0; SAS Institute, Inc., Cary, NC, USA).
Results
Patients
Figure 1 shows the study flowchart for the entire cohort and patients enrolled in each analysis. A total of 533 cases with CD underwent transanal endoscopy during the study period, and 364 consecutive cases were enrolled in the study (entire cohort) after excluding 169 cases based on the exclusion criteria. The median interval of serum marker measurements and transanal endoscopy was 6 (IQR, 2–22) days. For endoscopic evaluation of the ileum without colonic inflammation, 131 cases with the presence of inflammatory mucosa in the colon were excluded, and 233 cases were selected for analysis (ileum cohort). Postoperative endoscopic recurrence was evaluated in 45 cases who underwent transanal endoscopy within 2 years of surgery (postoperative cohort). Patient characteristics of each group are summarised in Table 1.
Usefulness of LRG in evaluating mucosal healing and moderate endoscopic activity
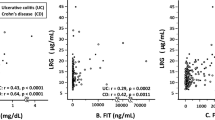
In the entire cohort, the serum LRG showed a higher correlation with SES-CD than serum CRP (Spearman rank correlation coefficient: LRG, 0.65; CRP, 0.37). The cut-off value of serum LRG for mucosal healing (SES-CD < 412) determined by the ROC curve analysis was 14.5 µg/mL; the prevalence of mucosal healing was 45.3%. At this cut-off value, the sensitivity was 0.81, specificity was 0.75, positive predictive value (PPV) was 0.73, negative predictive value (NPV) was 0.83, and accuracy was 0.78 (Table 2). The AUC for LRG was significantly higher than that for CRP (LRG, 0.84 [95% CI, 0.79–0.87]; CRP, 0.77 [95% CI, 0.72–0.82]; P-value, 0.001). In the univariate analysis of factors related to mucosal healing, LRG, CRP, disease duration, BMI, disease location, history of bowel surgery, steroid use, and Harvey–Bradshaw index (HBI) were significant factors (Supplemental Table 1). In the multivariate analysis of these factors, LRG, CRP, and steroid use were independent predictors of mucosal healing. This result suggests that 14.5 µg/mL of serum LRG can be a reasonable clinical cut-off value for evaluating mucosal healing.
We further investigated the utility of serum LRG as a predictive biomarker for moderate endoscopic activity (SES-CD ≥ 720,21). The cut-off value of serum LRG for SES-CD ≥ 7 was 20.1 µg/mL in the ROC curve analysis, and the prevalence was 39.3% (sensitivity: 0.74, specificity: 0.88, PPV: 0.80, NPV: 0.84, accuracy: 0.82). The AUC for LRG was also significantly higher than that for CRP (LRG, 0.85 [95% CI, 0.81–0.89]; CRP, 0.79 [95% CI, 0.74–0.83]; P-value, < 0.001). These results indicate that serum LRG levels can reflect not only mucosal healing but also moderate endoscopic CD activity more strictly than serum CRP levels. Alterations in the cut-off value of serum LRG were examined to evaluate moderate endoscopic activity. Each value was decreased by 2 µg/mL from 20.1 µg/mL, and the sensitivity, specificity, and predictive values of serum LRG and CRP were evaluated (Table 3). Next, the ability to exclude patients with moderate endoscopic disease activity (SES-CD ≥ 7; LRG, 10 µg/mL; CRP, 0.04 mg/dL) when the cut-off values for CRP and LRG were reduced to increase sensitivity. Assuming a prevalence of 10% in clinical remission and 50% in non-remission, the NPVs for each situation were calculated (Table 4). The NPVs of LRG and CRP for excluding moderate endoscopic disease activity in remission were 0.99 and 0.97, respectively, with both showing high exclusion ability. Conversely, the NPVs of LRG and CRP in the non-remission phase were 0.92 and 0.78, respectively, indicating a low ability of CRP to exclude moderate endoscopic disease activity in the non-remission phase. This suggests that LRG is useful for excluding moderate endoscopic disease activity in both remission and non-remission phases.
Serum biomarkers and mucosal healing in evaluating the ileum
Next, the usefulness of serum LRG levels for detecting inflammatory sites in the ileum was assessed. In the ileum cohort, serum LRG levels, but not serum CRP levels, correlated with SES-CD (Spearman rank correlation coefficient: LRG, 0.42; CRP, 0.09). ROC curve analysis using the same definition of mucosal healing (SES-CD < 412) was performed, with a prevalence of 64.4%. The cut-off value of serum LRG was 14.5 µg/mL, which was the same as that of the entire cohort. The AUC of LRG was also significantly higher than that of CRP (LRG, 0.77 [95% CI, 0.70–0.83]; CRP, 0.71 [95% CI, 0.64–0.77]; P-value, 0.024) (Table 5). The sensitivity, specificity, PPV, NPV, and accuracy were 0.82, 0.64, 0.80, 0.66, and 0.76, respectively. Factors related to mucosal healing were assessed using these cut-off values for LRG and CRP. In the univariate analysis, LRG, CRP, and steroid use were significant factors for mucosal healing in the ileum, and in the multivariate analysis, all these factors were independent factors associated with mucosal healing (Supplemental Table 2). These results suggest that the cut-off value of 14.5 µg/mL for serum LRG may be useful in evaluating mucosal healing in the ileum. Then we examined the colon cohort in the same manner as the ileum cohort, but the AUCs of LRG and CRP in the colon cohort were comparable (LRG, 0.86; CRP, 0.85) in predicting mucosal healing (SES-CD < 4), indicating that LRG is particularly useful for evaluating ileal inflammation. Additionally, we performed a subanalysis examining the correlation between serum biomarkers and SES-CD according to disease location (based on the Montreal classification26) and disease duration (Supplemental Tables 3, 4). Serum LRG levels showed stronger correlations with SES-CD than CRP across all disease locations (Spearman rank correlation coefficient: L1: LRG, 0.62; CRP, 0.25; L2: LRG, 0.55; CRP, 0.14; L3: LRG, 0.69; CRP, 0.45), as well as in both early and long-standing disease (disease duration > 2 years: LRG, 0.64; CRP, 0.33; disease duration < 2 years: LRG, 0.65; CRP, 0.43).
IHC staining of mucosal LRG in the small intestine
Next, IHC staining of LRG was performed on endoscopically collected small intestinal mucosal samples to examine whether LRG was secreted in the inflamed mucosa of the small intestine in patients with CD. Increased LRG expression was detected in the epithelial cells of the inflamed mucosa, and inflammatory cell infiltration into the mucosa was also observed (Fig. 2a i-iv). Conversely, the endoscopically-viewed uninflamed mucosa of patients with CD were scarcely stained (Fig. 2a v-viii). Next, the ratio of LRG expression in the inflamed mucosa was compared to that in the uninflamed mucosa. The percentage of LRG-positive cases in the uninflamed mucosa was 14.3% (1 of 7 cases), whereas the percentage of LRG-positive cases in the inflamed mucosa was 83.3% (5 of 6 cases) (Fig. 2b). These results suggest that the inflamed small bowel mucosa could potentially increase serum LRG levels.
Immunohistochemistry (IHC) staining to detect LRG. (a) Expression of LRG is increased in the inflamed mucosa of the ileum (i-iv), but not in the uninflamed mucosa (v-viii). Scale bar = 100 μm. (b) The percentage of LRG-positive cases was higher in the inflamed mucosa than in the uninflamed mucosa (83.3% vs. 14.3%).
Usefulness of LRG in evaluating postoperative recurrence
Finally, the utility of serum LRG levels for detecting postoperative recurrence was investigated in the postoperative cohort. Serum LRG levels showed stronger correlations with RS than CRP, and HBI did not correlate with SES-CD (Spearman rank correlation coefficient: LRG, 0.62; CRP, 0.32; HBI, 0.07) (Table 6). The cut-off value of serum LRG for postoperative recurrence (RS ≥ 222) was 13.2 µg/mL in the ROC curve analysis; the prevalence of postoperative recurrence was 49.0%. Using this cut-off value, the sensitivity was 0.86, specificity was 0.78, PPV was 0.79, NPV was 0.86 and accuracy was 0.82 for predicting endoscopic disease recurrence postoperatively. The AUC for LRG was higher than that for CRP (LRG, 0.84; CRP, 0.76). These results suggest that serum LRG levels may be valuable in identifying postoperative recurrence.
Discussion
Herein, we provide important insights into the role of LRG as a disease activity marker in patients with endoscopically moderate CD activity, and LRG as a predictive marker for postoperative recurrence. Furthermore, we demonstrated LRG secretion from the inflamed mucosa of the ileum in patients with CD, corroborating the basis of using LRG for the evaluation of mucosal healing in the ileum.
In the T2T strategy, CRP and FC are the set biomarkers of therapeutic targets1. However, they have some limitations. Some patients show negative CRP results despite endoscopically active disease8, and the intra-stool variation in FC is considerable at high concentration ranges27. As another biomarker, serum LRG has been reported to correlate with endoscopic severity12,28 and be useful as a surrogate biomarker of ileocolonoscopy to evaluate mucosal healing12,13. A previous report indicated that a serum LRG value of 13.6 µg/mL predicted mucosal healing defined as SES-CD < 3. Herein, the serum LRG value predicting mucosal healing, defined as SES-CD < 4, was 14.5 µg/mL, which is consistent with the previously reported results. To examine the efficacy of LRG as a biomarker in the T2T strategy, a serum LRG cut-off value of 20.1 µg/mL was calculated, which predicted patients with moderate endoscopic disease activity (SES-CD ≥ 7).20,21 These LRG cut-off values showed higher AUC than respective CRP cut-off values, suggesting that LRG is useful as a disease activity marker after a negative conversion of CRP. Furthermore, in the non-remission phase, a cut-off value of 10.0 µg/mL for LRG could exclude endoscopically active inflammation, defined as SES-CD ≥ 7, with an NPV of 92%, indicating its usefulness as a surrogate marker for endoscopy, unlike CRP. Yasutomi et al. reported an association between serum LRG and endoscopically active lesions in patients with CD28, using a modified SES-CD to assess endoscopic disease activity, which differed from the present study. They showed that serum LRG had the highest AUC among other biomarkers, such as CRP, faecal immunochemical test, and FC, predicting active inflammation in patients with CD, defined as a modified SES-CD > 6. Unfortunately, no cut-off value was reported. Three previous studies12,13,28 have compared LRG with FC. While our study did not directly compare LRG and FC, referring to these studies provides insight into their relationship. LRG exhibited a stronger correlation with endoscopic disease severity than FC, and in the analysis of mucosal healing and active inflammation, LRG showed AUC values equivalent to those of FC. FC may have a weaker correlation with SES-CD in small bowel evaluations than in colonic assessments28, although the sample size for these subanalyses was relatively small. In our study, by excluding the confounding effects of colonic inflammation, we were able to analyze more small bowel cases than previous studies and confirm the clinical usefulness of LRG in evaluating small bowel inflammation.
Monitoring the disease activity of small intestinal lesions in patients with CD is an important clinical issue, and the usefulness of LRG for this has been the focus of many studies. Kawamoto et al. reported that LRG is an excellent biomarker for detecting small intestinal ulcers29. An LRG cut-off value of 13.4 µg/mL showed significantly higher sensitivity and PPV than CRP or CD activity index. Omori et al. reported an LRG cut-off value of 14 µg/mL for detecting small bowel ulcerative lesions found by capsule endoscopy in CD patients with CD activity index < 150 and CRP < 0.5 mg/dL.11 Herein, serum LRG was also useful for predicting mucosal healing in the small bowel. However, its usefulness in small bowel assessment has not been directly compared with its usefulness in colon assessment. In our study, by examining the colon cohort in the same manner as the ileum cohort, we found that LRG was particularly valuable for assessing ileal inflammation. Although these reports indicate that LRG is useful in the evaluation of small intestinal lesions, the mechanism by which small intestinal lesions upregulate serum LRG levels in patients is not known. In literature, several LRG sources have been reported. In patients with IBD, LRG derived from peripheral blood mononuclear cells (PBMC) contributes to serum LRG levels30. In UC, LRG is produced by intestinal epithelial cells in addition to hepatocytes and neutrophils;19 however, this is not evident in CD. This study showed that inflammatory cells infiltrating the mucosa, as well as intestinal epithelial cells, produced more LRG than non-inflammatory lesions in the IHC staining of LRG using biopsy specimens. LRG derived from PBMCs partly contributes to increased serum LRG, and serum LRG levels reflect the endoscopic severity of IBD30, suggesting that endoscopic inflammatory mucosa in the small bowel could be a potential source of increased serum LRG, similar to that in UC.
The incidence of postoperative endoscopic recurrence within 1 year in Crohn’s disease has been reported to be 35–85%, with clinical recurrence of 10–38%17, suggesting that endoscopic disease recurrence precedes the recurrence of clinical symptoms. Early detection of postoperative recurrence may improve subsequent outcomes, and current guidelines recommend ileal endoscopy within 6–12 months postoperatively31. The usefulness of FC as a non-invasive marker for monitoring postoperative recurrence has been reported32. A meta-analysis reported an optimal FC cut-off value of 150 µg/g, with a sensitivity of 70%, specificity of 69%, and AUC of 0.73. Herein, the cut-off value for serum LRG to predict postoperative endoscopic disease recurrence was 13.2 µg/mL, with a sensitivity of 86%, specificity of 78%, and AUC of 0.84, indicating that serum LRG has a better detection accuracy than FC. Whether FC is useful in detecting small intestinal lesions remains debated. This study demonstrated the usefulness of LRG in monitoring postoperative recurrence in addition to its local production in the small intestine, making it valuable for CD management. Furthermore, based on our findings, we propose that serum LRG levels be measured postoperatively within 6–12 months, with endoscopic evaluation considered if serum LRG levels exceed 13.2 µg/mL. This approach would enable targeted endoscopic procedures for patients at higher risk of postoperative recurrence. To validate this strategy, future studies should stratify postoperative patients into high-risk and low-risk groups based on this cut-off value and evaluate its impact on clinical outcomes.
This study has some limitations. First, this was a single-center retrospective study; therefore, selection and information biases could not be eliminated. Although this is a cross-sectional study using objective data, such as serum LRG levels and endoscopic scores, its retrospective nature limits its ability to fully account for extra-intestinal complications or other inflammatory diseases that may elevate serum LRG levels. Additionally, compared to previous studies12,13, our study included more patients with lower disease activity. However, since our cohort was recruited consecutively, we expect it to reflect the clinical utility of LRG in CD patients. Second, serum inflammatory markers and endoscopic findings were not directly compared with FC, because serum LRG and FC could not be simultaneously measured owing to the Japanese clinical insurance system, and because we didn’t have stored fecal samples to measure FC at the same time as serum samples to measure LRG. Third, when both the small bowel and colon are inflamed, the contribution of LRG produced by the inflamed small intestinal mucosa to serum LRG levels remains unclear. Finally, this was a single-centre study, and low number of cases of postoperative recurrence were assessed. A large-scale prospective study is needed to externally validate our findings.
In conclusion, this study indicates that LRG produced in the inflamed mucosa of the small intestine is a valuable surrogate marker for endoscopy that can predict mucosal healing, moderate or greater endoscopic disease activity, and postoperative endoscopic disease recurrence. These findings strengthen and expand upon the existing evidence supporting the clinical usefulness of LRG.
Data availability
The data are available from the corresponding author upon reasonable request.
References
Turner, D. et al. STRIDE-II: an update on the selecting therapeutic targets in inflammatory bowel disease (STRIDE) initiative of the international organization for the study of IBD (IOIBD): determining therapeutic goals for Treat-to-Target strategies in IBD. Gastroenterology 160, 1570–1583. https://doi.org/10.1053/j.gastro.2020.12.031 (2021).
Laterza, L. et al. Multiparametric evaluation predicts different Mid-Term outcomes in Crohn’s disease. Dig. Dis. 36, 184–193. https://doi.org/10.1159/000487589 (2018).
Bernstein, C. N., Loftus, E. V. Jr., Ng, S. C., Lakatos, P. L. & Moum, B. Hospitalisations and surgery in Crohn’s disease. Gut 61, 622–629. https://doi.org/10.1136/gutjnl-2011-301397 (2012).
Ungaro, R. C. et al. Deep remission at 1 year prevents progression of early Crohn’s disease. Gastroenterology 159, 139–147. https://doi.org/10.1053/j.gastro.2020.03.039 (2020).
Neurath, M. F. & Travis, S. P. Mucosal healing in inflammatory bowel diseases: a systematic review. Gut 61, 1619–1635. https://doi.org/10.1136/gutjnl-2012-302830 (2012).
Serada, S. et al. iTRAQ-based proteomic identification of leucine-rich alpha-2 glycoprotein as a novel inflammatory biomarker in autoimmune diseases. Ann. Rheum. Dis. 69, 770–774. https://doi.org/10.1136/ard.2009.118919 (2010).
Pepys, M. B. & Hirschfield, G. M. C-reactive protein: a critical update. J. Clin. Invest. 111, 1805–1812. https://doi.org/10.1172/jci18921 (2003).
Solem, C. A. et al. Correlation of C-reactive protein with clinical, endoscopic, histologic, and radiographic activity in inflammatory bowel disease. Inflamm. Bowel Dis. 11, 707–712. https://doi.org/10.1097/01.mib.0000173271.18319.53 (2005).
Shinzaki, S. et al. Leucine-rich Alpha-2 glycoprotein is a serum biomarker of mucosal healing in ulcerative colitis. J. Crohns Colitis. 11, 84–91. https://doi.org/10.1093/ecco-jcc/jjw132 (2017).
Kawamura, T. et al. Accuracy of serum Leucine-Rich Alpha-2 glycoprotein in evaluating endoscopic disease activity in Crohn’s disease. Inflamm. Bowel Dis. https://doi.org/10.1093/ibd/izac076 (2022).
Omori, T. et al. Serum Leucine-Rich Alpha-2 Glycoprotein in Quiescent Crohn’s Disease as a Potential Surrogate Marker for Small-Bowel Ulceration detected by Capsule Endoscopy. J. Clin. Med. 11 https://doi.org/10.3390/jcm11092494 (2022).
Shinzaki, S. et al. Leucine-rich alpha-2 glycoprotein is a potential biomarker to monitor disease activity in inflammatory bowel disease receiving adalimumab: PLANET study. J. Gastroenterol. 56, 560–569. https://doi.org/10.1007/s00535-021-01793-0 (2021).
Abe, I. et al. Serum leucine-rich alpha-2 glycoprotein as a predictive factor of endoscopic remission in Crohn’s disease. J. Gastroenterol. Hepatol. https://doi.org/10.1111/jgh.15907 (2022).
Kusaka, J. et al. Residual lesions on capsule endoscopy is associated with postoperative clinical recurrence in patients with Crohn’s disease. Dig. Dis. Sci. 63, 768–774. https://doi.org/10.1007/s10620-018-4942-6 (2018).
Kono, T. et al. Prospective postsurgical capsule endoscopy in patients with Crohn’s disease. World J. Gastrointest. Endosc. 6, 88–98. https://doi.org/10.4253/wjge.v6.i3.88 (2014).
De Cruz, P., Kamm, M. A., Prideaux, L., Allen, P. B. & Moore, G. Mucosal healing in Crohn’s disease: a systematic review. Inflamm. Bowel Dis. 19, 429–444. https://doi.org/10.1002/ibd.22977 (2013).
Buisson, A., Chevaux, J. B., Allen, P. B. & Bommelaer, G. Peyrin-Biroulet, L. Review Article: the natural history of postoperative Crohn’s disease recurrence. Aliment. Pharmacol. Ther. 35, 625–633. https://doi.org/10.1111/j.1365-2036.2012.05002.x (2012).
Yamamoto, T. & Watanabe, T. Surgery for luminal Crohn’s disease. World J. Gastroenterol. 20, 78–90. https://doi.org/10.3748/wjg.v20.i1.78 (2014).
Serada, S. et al. Serum leucine-rich alpha-2 glycoprotein is a disease activity biomarker in ulcerative colitis. Inflamm. Bowel Dis. 18, 2169–2179. https://doi.org/10.1002/ibd.22936 (2012).
Feagan, B. et al. Performance of Crohn’s disease clinical trial endpoints based upon different cutoffs for patient reported outcomes or endoscopic activity: analysis of EXTEND data. Inflamm. Bowel Dis. 24, 932–942. https://doi.org/10.1093/ibd/izx082 (2018).
Khanna, R. et al. Endoscopic scoring indices for evaluation of disease activity in Crohn’s disease. Cochrane Database Syst Rev Cd010642 (2016). (2016). https://doi.org/10.1002/14651858.CD010642.pub2
Rutgeerts, P. et al. Predictability of the postoperative course of Crohn’s disease. Gastroenterology 99, 956–963. https://doi.org/10.1016/0016-5085(90)90613-6 (1990).
Jin, Z. et al. The prognostic impact of Leucine-Rich α-2-Glycoprotein-1 in cholangiocarcinoma and its association with the IL-6/TGF-β1 axis. J. Surg. Res. 252, 147–155. https://doi.org/10.1016/j.jss.2020.03.018 (2020).
Furukawa, K. et al. Clinicopathological significance of Leucine-Rich α2-Glycoprotein-1 in Sera of patients with pancreatic cancer. Pancreas 44, 93–98. https://doi.org/10.1097/mpa.0000000000000205 (2015).
DeLong, E. R., DeLong, D. M. & Clarke-Pearson, D. L. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44, 837–845 (1988).
Satsangi, J., Silverberg, M. S., Vermeire, S. & Colombel, J. F. The Montreal classification of inflammatory bowel disease: controversies, consensus, and implications. Gut 55, 749–753. https://doi.org/10.1136/gut.2005.082909 (2006).
Cremer, A. et al. Variability of faecal calprotectin in inflammatory bowel disease patients: an observational Case-control study. J. Crohns Colitis. 13, 1372–1379. https://doi.org/10.1093/ecco-jcc/jjz069 (2019).
Yasutomi, E. et al. Leucine-rich alpha-2 glycoprotein as a marker of mucosal healing in inflammatory bowel disease. Sci. Rep. 11, 11086. https://doi.org/10.1038/s41598-021-90441-x (2021).
Kawamoto, A. et al. Serum leucine-rich alpha-2 glycoprotein; a novel biomarker for small bowel mucosal activity in Crohn’s disease. Clin. Gastroenterol. Hepatol. https://doi.org/10.1016/j.cgh.2021.06.036 (2021).
Yoshimura, T. et al. Evaluation of Serum Leucine-Rich Alpha-2 Glycoprotein as a New Inflammatory Biomarker of Inflammatory Bowel Disease. Mediators Inflamm 8825374 (2021). (2021). https://doi.org/10.1155/2021/8825374
De Cruz, P. et al. Crohn’s disease management after intestinal resection: a randomised trial. Lancet 385, 1406–1417. https://doi.org/10.1016/s0140-6736(14)61908-5 (2015).
Tham, Y. S. et al. Fecal calprotectin for detection of postoperative endoscopic recurrence in Crohn’s disease: systematic review and meta-analysis. Th. Adv. Gastroenterol. 11, 1756284818785571. https://doi.org/10.1177/1756284818785571 (2018).
Author information
Authors and Affiliations
Contributions
Ta.Ta. and S.S. contributed equally. Ta.Ta., S.S. and H.I. planned and conducted the study. Ta.Ta. collected data. Ta.Ta., S.S., T.Y., Y.T., A.A., T.A., M.T., Y.O., R.U., Y.T., T.I., T.O., H.I. and Y.H. interpreted the data. Ta.Ta., S.S., T.Y. and H.I. drafted the manuscript. Te.Ta. supervised the study.
Corresponding author
Ethics declarations
Competing interests
S. Shinzaki received contract research funds from Sekisui Medical. All other authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tashiro, T., Shinzaki, S., Yoshihara, T. et al. Leucine-rich Alpha-2 glycoprotein could be clinically useful in active and postoperative Crohn’s disease. Sci Rep 15, 9031 (2025). https://doi.org/10.1038/s41598-025-93831-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-93831-7