Abstract
As a novel inflammatory marker, Systemic Immune-Inflammation Index (SII) has recently been recognized as a prognostic indicator for a variety of diseases including malignant cancers, coronary artery disease, hyperlipidemia, and hepatic steatosis. Carotenoids are a group of abundant lipid-soluble phytochemicals, and studies have suggested that they have antioxidant, antiapoptotic, and anti-inflammatory properties. However, a systematic analysis of the association between serum carotenoids and SII is still lacking. The purpose of this investigation was to explore the association between serum carotenoid concentration and SII. The cross-sectional investigation included general people (age ≥ 20) with complete information on SII and five different serum carotenoids (Trans-lycopene, β-carotene, α-carotene, lutein/zeaxanthin, and β-cryptoxanthin). Multivariate linear regression analyses were used to evaluate the association between serum carotenoids and SII among general people. The potential non-linear relationship was determined using threshold effect analysis and fitted smoothing curves. Subgroup analysis was performed to explore the potential stratified factors. 15903 participants were enrolled in our investigation. Based on multivariate linear regressions, the highest quartiles of serum carotenoids were found significantly associated with SII compared with the lowest quartiles. The results showed the negative association between SII and the concentration of five different serum carotenoids. According to the non-linear analysis, we found that there are non-linear relationships between β-carotene and trans-lycopene and SII in general people with an inflection point of 6.90 (log2-transformed, ug/dL) and 4.01 (log2-transformed, ug/dL), respectively. The results from subgroup analysis provide several potential moderating effects, such as race, current drinker, and age. This study revealed the relationship between the concentration of several serum carotenoids and SII across the general American population. Further prospective and longitude investigations are needed.
Similar content being viewed by others
Introduction
Systemic Immune-Inflammation Index (SII) is a novel inflammatory marker introduced by Hu et al. in 2014. It reflects systemic inflammation throughout the human body1. SII, a stable inflammatory biomarker, is calculated using the formula: platelet count × neutrophil count / lymphocyte count1. Unlike traditional inflammatory indices, numerous studies have demonstrated that SII not only reflects the balance between inflammatory and immune responses2,3 but has also recently been recognized as a prognostic biomarker for a variety of diseases, including malignant cancers1, coronary artery disease4, hyperlipidemia3, and hepatic steatosis5. For example, Hu et al.’s initial study on SII analyzed a cohort of 133 patients and further validated their findings in a prospective study involving 123 patients. Univariate and multivariate analyses demonstrated that SII was a significant predictor of poor outcomes in patients with hepatocellular carcinoma1. Similarly, a retrospective analysis indicated that SII is a valuable indicator for predicting recurrence in colorectal cancer patients6. More recently, Huang et al. analyzed a retrospective dataset of 324 participants, applying single-variable tests, binary logistic regression analysis, ROC curve analysis, restricted cubic spline tests, and curve fitting. Their findings identified SII as a risk factor for coronary artery lesions, suggesting its utility as an auxiliary laboratory biomarker for predicting coronary artery lesions7.
Carotenoids, are a group of abundant lipid-soluble phytochemicals, with representative conjugated polyene chain containing 8 units of C5 like isoprenoid8, consisting of 9–11 double bonds. They can be classified into hydrocarbon carotenoids (such as lycopene, β-carotene, and α-carotene) and oxygen-containing derivatives of hydrocarbon carotenoids (including xanthophylls, lutein, zeaxanthin, and β-cryptoxanthin)9. In the human body, over 95% of the circulating carotenoids are among these six types10. Studies suggest that carotenoids possess antioxidant, anti-apoptotic, and anti-inflammatory properties due to their distinctive structures11,12. For example, research has shown that carotenoids interact with the nuclear factor κB pathway by inhibiting the translocation of nuclear factor κB to the nucleus, thereby reducing the production of inflammatory cytokines such as interleukin-8 and prostaglandin E213. Several cellular models for studying potential anti-inflammatory effects of carotenoids have been established. Jung et al. found that monocytes/macrophages, which play a significant role in many inflammatory processes, are important targets of carotenoids14. In vitro studies have demonstrated that β-carotene, lycopene, and lutein can reduce reactive oxygen species (ROS) production, thereby mitigating inflammation15.
Previous studies have reported the relationship between carotenoids and various inflammation indicators, such as TNF-RII, IL-6, IL-1ra, CRP, and neutrophil16,17,18. However, these studies are constrained by their experimental settings, which limit direct applicability to population-level data. Moreover, few studies have found the association between serum carotenoids and SII. Therefore, we investigated the relationship between SII and serum carotenoids through a population-based cross-sectional study utilizing data from the National Health and Nutrition Examination Survey (NHANES) adult participants.
Methods
NHANES
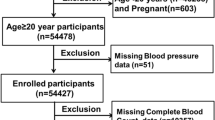
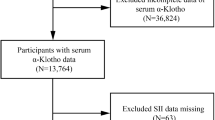
NHANES is a research program designed to assess the health and nutritional status of adults and children in the U.S, employing a complex, multistage, and probabilistic sampling technique. In this study, a total of 31,509 participants were initially considered (Fig. 1).
Flowchart of participant selection.
Exposure variable
Automated hematology analyzers was used to measure Lymphocyte, neutrophil, and platelet counts (expressed as ×103 cells/ml). SII was calculated using the formula: platelet count × neutrophil count / lymphocyte count. High-performance liquid chromatography (HPLC) was employed to measure the levels of six different serum carotenoids (trans-β-carotene, cis-β-carotene, α-carotene, β-cryptoxanthin, lutein/zeaxanthin, and trans-lycopene.) for NHANES 2001–2006. Information regarding participants was available on trans-βcarotene, cis-β-carotene, α-carotene, β-cryptoxanthin, lutein/zeaxanthin, and trans-lycopene. Information on total lycopene was not provided for NHANES 2001–2002. The analysis utilized the total β-carotene level, which was calculated by summing the concentrations of cis-β-carotene and trans-β-carotene. Total-carotene is calculated as the sum of multiple carotenoids.
Covariates
Demographic data were collected through questionnaire interviews and included age, sex, marital status, race, leisure-time physical activity, education level, and family poverty ratio. Body mass index (BMI) was calculated as weight divided by height squared. Alcohol consumption was categorized as never (< 12 drinks in lifetime), current (≥ 12 drinks and currently drinking), and former (no drinks in the past year but ≥ 12 drinks in lifetime). Smoking status was classified as former (≥ 100 cigarettes but not currently smoking), current (≥ 100 cigarettes and currently smoking), and never (< 100 cigarettes in lifetime). Hypertension was defined as systolic blood pressure ≥ 140 mmHg, diastolic blood pressure ≥ 90 mmHg, a prior diagnosis, or a history of antihypertensive medication use. Diabetes was diagnosed based on fasting glucose levels (mmol/L) ≥ 7.0, glycohemoglobin (%) ≥ 6.5, use of antidiabetic medications or insulin, or a prior physician diagnosis of diabetes mellitus (DM). Cerebrovascular disease (CVD) was defined as having a history of stroke, congestive heart failure, heart attack, angina, or coronary artery disease. All data are publicly available at www.cdc.gov/nchs/nhanes/.
Statistical analyses
Following NHANES recommendations, weighted chi-square and one-way analyses were performed to identify disparities in descriptive analyses. Multifactorial linear regression analysis was utilized to evaluate the correlation between SII and serum carotenoids. Model 1 was unadjusted, Model 2 was adjusted for age and sex, and Model 3 was further adjusted for race, education level, marital status, alcohol intake, smoking status, leisure-time physical activity, BMI, family poverty ratio, hypertension, diabetes, and CVD. The smoothed curve fits were generated to examine potential nonlinear relationships. A threshold effect analysis model was applied to calculate the relationship and inflection point. To explore the correlation between SII and the concentrations of five different serum carotenoids, subgroup analyses were conducted, stratifying by age, sex, race, education level, marital status, alcohol intake, smoking status, leisure-time physical activity, BMI, family poverty ratio, hypertension, diabetes, and CVD.
Results
Baseline characteristics of participants
In this study, 10,593 participants were enrolled, with a mean age of 46.36 years (SE: 0.35), and 49.43% of them were male. The weighted baseline characteristics of participants, stratified by the β-carotene quartiles, are presented in Table 1. Statistically significant differences were observed in age, sex, race, education level, marital status, smoking status, alcohol consumption, family poverty ratio, BMI, leisure-time physical activity, diabetes mellitus (DM), the levels of four different serum carotenoids (P < 0.05), and SII in relation to serum β-carotene quartiles. Specifically, individuals in Quartile 4 were typically older, more likely to be female, non-Hispanic White, better educated, married, alcohol consumers, non-smokers, with a family poverty ratio > 3.5, and were less likely to have diabetes, cardiovascular disease (CVD), or hypertension. Additionally, they engaged more frequently in leisure-time physical activity.
Association between SII and serum carotenoids
We conducted multivariable regression analyses to evaluate the relationship between SII and serum carotenoid concentrations. According to the quartiles of SII, there was statistically significant difference among the SII quartiles 1 in terms of 4 different serum carotenoids and serum total carotene in model 1, 2 and 3 (Table 2). When serum carotenoids were calculated as continuous variables, multivariate regression analysis revealed an inverse correlation between β-carotene, α-carotene, β-cryptoxanthin, Lutein/zeaxanthin, and trans lycopene, total carotene, and SII after full adjustments. When they were divided into quartiles, the results showed, the β values and 95% CIs were (-56.46 [95% CI, -82.10 to -30.81]) for α-carotene, (− 74.27, [95% CI, -96.61 to -51.93]) for β-carotene, (− 54.40, [95% CI, − 77.86 to − 30.94]) for β-cryptoxanthin, (− 74.31 (95% CI, -100.41 to − 48.21]) for lutein/ zeaxanthin, (− 33.23 [95% CI, -59.61 to -6.86]) for trans-lycopene, and (-72.55 [95% CI, -100.14 to -44.95]) for total carotene after adjusting all covariates, compared with the lowest quartile.
The analyses of nonlinear relationship
We explored the nonlinear relationships between serum carotenoids and SII using piecewise linear regression models (Table 3; Fig. 2A–E). The results indicated a linear relationship between SII and α-carotene, β-cryptoxanthin, and lutein/zeaxanthin. However, nonlinear relationships were observed between SII and β-carotene as well as trans-lycopene, with inflection points at 6.90 (log2-transformed, µg/dL) and 4.01 (log2-transformed, µg/dL), respectively.
The association between SII and serum carotenoids. Association between SII and serum carotenoids (A) α-carotene, (B) β-carotene, (C) β-cryptoxanthin, (D) Lutein/zeaxanthin, (E) Trans-lycopene). Adjusted for age, sex, race, education level, marital status, alcohol intake, smoking status, leisure-time physical activity, BMI, the family of poverty ratio, TC, hypertension, diabetes, and CVD; total serum β-carotene was used in the analysis.
Subgroup analysis
To validate our findings, we conducted subgroup analyses based on age (< 60/≥60 years), gender (male/female), BMI (< 30/≥30 kg/m²), race (White/non-White), current alcohol consumption (yes/no), current smoking status (yes/no), hypertension (yes/no), diabetes (yes/no), and CVD (yes/no) (Supplementary Tables 1–5). No significant interactions were found between serum carotenoids and SII across most subgroups.
Discussion
This population-based study focus on the relationship between serum carotenoids, including α-carotene, β-carotene, β-cryptoxanthin, lutein/zeaxanthin, and trans-lycopene and SII. Our findings indicate that higher levels of these five serum carotenoids are associated with lower SII values. Additionally, nonlinear relationships were observed between certain serum carotenoids and SII.
According to previous research, inflammatory markers are associated with the incidence of various chronic diseases including diabetes, CVDs, neurodegenerative disorders, and malignancies19,20. It has been reported that a healthy life style such as regular consumption of fruits and vegetables can reduce the incidence of those chronic conditions21,22,23,24. With the global health burden of chronic diseases escalating due to an aging population, there is an urgent need to explore key factors influencing systemic inflammation and their impact on general health.
Researches showed that as an important inflammatory marker, the role of SII in various diseases have widely been observed3,5,25. For example, Yang et al. examined the predictive value of SII in patients with coronary artery disease (CAD). Interestingly, compare to traditional risk factors, they found that SII had a better prediction of major cardiovascular events in CAD patients after coronary intervention26. In individuals with elevated SII levels, the immune system remains persistently activated, resulting in chronic inflammation of the joints and other tissues27. Satis et al. reported that SII levels were significantly higher in patients with rheumatoid arthritis compared to healthy controls and were positively correlated with disease severity28. In 2017, study suggested that SII may be an independent prognostic indicator for advanced lung cancer patients and is better than other inflammation-based factors in terms of prognostic ability29. The role of SII as a risk factor in patients with diabetic depression have also been described30. However, the specific mechanisms through which SII affects these diseases remain unclear.
Carotenoids, known for their antioxidant and anti-inflammatory properties31,32, are believed to offer protective effects against various chronic diseases33. For instance, carotenoids are thought to be beneficial in preventing CVDs34. A meta-analysis of longitudinal studies showed that the concentrations of total carotene, αcarotene, β-carotene, and lycopene were inversely associated with the risk of all-cause mortality35. The Age-Related Eye Disease Study (AREDS) demonstrated that β-carotene-rich diets are beneficial for age-related macular degeneration (AMD)36. In animal studies, carotenoids have shown cardioprotective properties by enhancing glutathione peroxidase levels and superoxide dismutase (SOD) activity in norepinephrine-induced cardiac hypertrophy37. Their antioxidant and anti-inflammatory effects are attributed to their functional groups and the number of conjugated double bonds, which enable them to neutralize reactive oxygen species (ROS) and free radicals, thereby protecting lipid membranes from peroxidation and reducing cellular inflammation38,39,40,41. In our study, the adverse connection between the concentration of five serum carotenoids and SII may also due to the aforementioned underlying mechanism.
Previous studies have explored the relationship between carotenoids and various inflammation indicators, such as TNF-RII, IL-6, IL-1ra, CRP, and neutrophil. For example, research conducted in breast cancer patients examined the relationship between carotenoids and serum CRP, sTNF-RII, IL-6, and IL-1ra, suggesting that serum carotenoid concentrations may confer anti-inflammatory benefits. However, the small sample sizes in these studies limited their outcomes. Also, those Traditional inflammatory indices do not capture the potential role of serum carotenoids as effectively as SII, a novel and promising inflammatory marker. In our study, we investigated the inverse relationship between five different serum carotenoids and SII. Nonlinear analysis revealed that β-carotene and trans-lycopene have nonlinear relationships with SII, with inflection points at 6.90 (log₂-transformed, µg/dL) and 4.01 (log₂-transformed, µg/dL), respectively. Our findings suggest that the five different serum carotenoids are negatively associated with SII, indicating their potential role in modulating systemic inflammation. To our knowledge, this is the first study to investigate the relationship between SII and the concentrations of these five serum carotenoids.
The present study has multiple strengths. This research was from a substantial, nationally representative sample, including 10,593 participants, allowing for the weighted outcomes that reflect the U.S. population at the national level. Also, we considered a broad range of potential confounding factors and conducted subgroup analyses to make sure the results are consistent. However, there are several limitations. For example, as a cross-sectional survey, this study cannot build a cause-and-effect relationship between five serum carotenoids and SII in general people. Moreover, the relationship between total serum carotenoids and SII could not be established due to the incomplete total lycopene. Lastly, considering the complex metabolism of serum carotenoids in vivo, additional research should be taken into consideration.
Conclusion
In conclusion, our study from a substantial, national sample representative U.S. population showed an inverse relationship between five serum carotenoids and SII in general people. SII, an evaluation index of systemic inflammatory response can be used to predict the severity of certain diseases and monitor treatment effects. The findings have potential public health implications and support the metabolic benefits of serum carotenoids on inflammation. However, the causal relationship and the underlying mechanism still need further investigations.
Data availability
The survey data are publicly available on the internet for data users and researchers throughout the world (https://www.cdc.gov/nchs/nhanes/ ).
References
Hu, B. et al. Systemic immune-inflammation index predicts prognosis of patients after curative resection for hepatocellular carcinoma. Clin. Cancer Res. 20, 6212–6222. https://doi.org/10.1158/1078-0432.CCR-14-0442 (2014).
Chen, J. H. et al. Systemic immune-inflammation index for predicting prognosis of colorectal cancer. World J. Gastroenterol. 23, 6261–6272. https://doi.org/10.3748/wjg.v23.i34.6261 (2017).
Mahemuti, N. et al. Association between systemic Immunity-Inflammation index and hyperlipidemia: A population-based study from the NHANES (2015–2020). Nutrients 15 https://doi.org/10.3390/nu15051177 (2023).
Miao, Y., Yan, Q., Li, S., Li, B. & Feng, Y. Neutrophil to lymphocyte ratio and platelet to lymphocyte ratio are predictive of chemotherapeutic response and prognosis in epithelial ovarian cancer patients treated with platinum-based chemotherapy. Cancer Biomark. 17, 33–40. https://doi.org/10.3233/CBM-160614 (2016).
Xie, R. et al. Association between SII and hepatic steatosis and liver fibrosis: A population-based study. Front. Immunol. 13, 925690. https://doi.org/10.3389/fimmu.2022.925690 (2022).
Nakamoto, S. et al. Systemic immune-inflammation index predicts tumor recurrence after radical resection for colorectal cancer. Tohoku J. Exp. Med. 261, 229–238. https://doi.org/10.1620/tjem.2023.J074 (2023).
Huang, T., Peng, Q., Zhang, Y., Zhu, Z. & Fan, X. The systemic Immune-Inflammation index (SII) and coronary artery lesions in Kawasaki disease. Clin. Exp. Med. 24, 4. https://doi.org/10.1007/s10238-023-01265-0 (2024).
Kopsell, D. A. & Kopsell, D. E. Accumulation and bioavailability of dietary carotenoids in vegetable crops. Trends Plant. Sci. 11, 499–507 (2006).
Saini, R. K. & Keum, Y. S. Carotenoid extraction methods: A review of recent developments. Food Chem. 240 https://doi.org/10.1016/j.foodchem.2017.07.099 (2018).
Mueller, L. & Boehm, V. Antioxidant activity of beta-carotene compounds in different in vitro assays. Molecules 16, 1055–1069. https://doi.org/10.3390/molecules16021055 (2011).
Wang, M., Tang, R., Zhou, R., Qian, Y. & Di, D. The protective effect of serum carotenoids on cardiovascular disease: A cross-sectional study from the general US adult population. Front. Nutr. 10, 1154239. https://doi.org/10.3389/fnut.2023.1154239 (2023).
Milani, A., Basirnejad, M., Shahbazi, S. & Bolhassani, A. Carotenoids: Biochemistry, Pharmacology and treatment. Br. J. Pharmacol. 174, 1290–1324. https://doi.org/10.1111/bph.13625 (2017).
Bai, S. K. et al. Beta-carotene inhibits inflammatory gene expression in lipopolysaccharide-stimulated macrophages by suppressing redox-based NF-kappaB activation. Exp. Mol. Med. 37, 323–334. https://doi.org/10.1038/emm.2005.42 (2005).
Jung, W. J. & Sung, M. K. Effects of major dietary antioxidants on inflammatory markers of RAW 264.7 macrophages. Biofactors 21, 113–117. https://doi.org/10.1002/biof.552210122 (2004).
Bouayed, J. & Bohn, T. Exogenous antioxidants–double-edged swords in cellular redox State: health beneficial effects at physiologic doses versus deleterious effects at high doses. Oxid. Med. Cell. Longev. 3, 228–237. https://doi.org/10.4161/oxim.3.4.12858 (2010).
Zuniga, K. E. & Moran, N. E. Low serum carotenoids are associated with self-reported cognitive dysfunction and inflammatory markers in breast cancer survivors. Nutrients 10 (2018). https://doi.org/10.3390/nu10081111
Rossoni-Junior, J. V. et al. Annato extract and beta-carotene modulate the production of reactive oxygen species/nitric oxide in neutrophils from diabetic rats. J. Clin. Biochem. Nutr. 50, 177–183. https://doi.org/10.3164/jcbn.11-49 (2012).
Macedo, R. C., Bolin, A. P., Marin, D. P. & Otton, R. Astaxanthin addition improves human neutrophils function: In vitro study. Eur. J. Nutr. 49, 447–457. https://doi.org/10.1007/s00394-010-0103-1 (2010).
Nielsen, O. H., Rudiger, N., Gaustadnes, M. & Horn, T. Intestinal interleukin-8 concentration and gene expression in inflammatory bowel disease. Scand. J. Gastroenterol. 32, 1028–1034. https://doi.org/10.3109/00365529709011220 (1997).
Cook, E. B. Tear cytokines in acute and chronic ocular allergic inflammation. Curr. Opin. Allergy Clin. Immunol. 4, 441–445. https://doi.org/10.1097/00130832-200410000-00018 (2004).
Coyne, T. et al. Diabetes mellitus and serum carotenoids: findings of a population-based study in Queensland, Australia. Am. J. Clin. Nutr. 82, 685–693. https://doi.org/10.1093/ajcn.82.3.685 (2005).
Britton, G. Structure and properties of carotenoids in relation to function. FASEB J. 9, 1551–1558 (1995).
Wang, P., Long, F., Lin, H. & Wang, T. Dietary phytochemicals targeting Nrf2 for chemoprevention in breast cancer. Food Funct. 13, 4273–4285. https://doi.org/10.1039/d2fo00186a (2022).
Riccioni, G. et al. Carotenoids and asymptomatic carotid atherosclerosis. J. Biol. Regul. Homeost. Agents 24, 447–452 (2010).
Sylman, J. L. et al. The predictive value of inflammation-related peripheral blood measurements in cancer staging and prognosis. Front. Oncol. 8, 78. https://doi.org/10.3389/fonc.2018.00078 (2018).
Yang, Y. L. et al. Systemic immune-inflammation index (SII) predicted clinical outcome in patients with coronary artery disease. Eur. J. Clin. Invest. 50, e13230. https://doi.org/10.1111/eci.13230 (2020).
Yorulmaz, A., Hayran, Y., Akpinar, U. & Yalcin, B. Systemic immune-inflammation index (SII) predicts increased severity in psoriasis and psoriatic arthritis. Curr. Health Sci. J. 46, 352–357. https://doi.org/10.12865/CHSJ.46.04.05 (2020).
Satis, S. New inflammatory marker associated with disease activity in rheumatoid arthritis: The systemic Immune-Inflammation index. Curr. Health Sci. J. 47, 553–557. https://doi.org/10.12865/CHSJ.47.04.11 (2021).
Tong, Y. S., Tan, J., Zhou, X. L., Song, Y. Q. & Song, Y. J. Systemic immune-inflammation index predicting chemoradiation resistance and poor outcome in patients with stage III non-small cell lung cancer. J. Transl Med. 15, 221. https://doi.org/10.1186/s12967-017-1326-1 (2017).
Wang, J., Zhou, D., Dai, Z. & Li, X. Association between systemic Immune-Inflammation index and diabetic depression. Clin. Interv Aging 16, 97–105. https://doi.org/10.2147/CIA.S285000 (2021).
Kabir, M. T. et al. Therapeutic promise of carotenoids as antioxidants and anti-inflammatory agents in neurodegenerative disorders. Biomed. Pharmacother. 146, 112610. https://doi.org/10.1016/j.biopha.2021.112610 (2022).
Hajizadeh-Sharafabad, F., Zahabi, E. S., Malekahmadi, M., Zarrin, R. & Alizadeh, M. Carotenoids supplementation and inflammation: A systematic review and meta-analysis of randomized clinical trials. Crit. Rev. Food Sci. Nutr. 62, 8161–8177. https://doi.org/10.1080/10408398.2021.1925870 (2022).
Bohn, T. et al. Mechanistic aspects of carotenoid health benefits—where are we now? Nutr. Res. Rev. 34, 276–302. https://doi.org/10.1017/S0954422421000147 (2021).
Zheng, L., Yu, X., Xia, Z., Guo, Y. & Dai, Y. The associations between serum vitamins and carotenoids with chronic obstructive pulmonary disease: results from the NHANES. Int. J. Chron. Obstruct Pulmon Dis. 18, 2985–2997. https://doi.org/10.2147/COPD.S432995 (2023).
Aune, D. et al. Dietary intake and blood concentrations of antioxidants and the risk of cardiovascular disease, total cancer, and all-cause mortality: A systematic review and dose-response meta-analysis of prospective studies. Am. J. Clin. Nutr. 108, 1069–1091. https://doi.org/10.1093/ajcn/nqy097 (2018).
Group, A. R. et al. The Age-Related eye disease study 2 (AREDS2): Study design and baseline characteristics (AREDS2 report number 1). Ophthalmology 119, 2282–2289. https://doi.org/10.1016/j.ophtha.2012.05.027 (2012).
Shen, X. C. & Qian, Z. Y. Effects of crocetin on antioxidant enzymatic activities in cardiac hypertrophy induced by norepinephrine in rats. Pharmazie 61, 348–352 (2006).
Miller, N. J., Sampson, J., Candeias, L. P., Bramley, P. M. & Rice-Evans, C. A. Antioxidant activities of carotenes and xanthophylls. FEBS Lett. 384, 240–242. https://doi.org/10.1016/0014-5793(96)00323-7 (1996).
Edge, R., McGarvey, D. J. & Truscott, T. G. The carotenoids as anti-oxidants–a review. J. Photochem. Photobiol B. 41, 189–200. https://doi.org/10.1016/s1011-1344(97)00092-4 (1997).
Zhao, D., Kwon, S. H., Chun, Y. S., Gu, M. Y. & Yang, H. O. Anti-neuroinflammatory effects of fucoxanthin via inhibition of Akt/NF-kappaB and MAPKs/AP-1 pathways and activation of PKA/CREB pathway in lipopolysaccharide-activated BV-2 microglial cells. Neurochem Res. 42, 667–677. https://doi.org/10.1007/s11064-016-2123-6 (2017).
Liu, T., Liu, W. H., Zhao, J. S., Meng, F. Z. & Wang, H. Lutein protects against beta-amyloid peptide-induced oxidative stress in cerebrovascular endothelial cells through modulation of Nrf-2 and NF-kappab. Cell. Biol. Toxicol. 33, 57–67. https://doi.org/10.1007/s10565-016-9360-y (2017).
Acknowledgements
We thank the National Health and Nutrition Examination Surveys for providing the data.
Funding
This work was supported by Hunan provincial innovation foundation for postgraduate (No. CX20230328).
Author information
Authors and Affiliations
Contributions
L.P. and J.W. contributed to the conception and design ofthe study. W.N. and S.Y. performed resource analysis, andwrote the first draft of the manuscript together. All authors read andapproved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yan, SH., Nai, W., Peng, L. et al. Association of serum carotenoids and SII among general people, based on NHANES 2001–2006. Sci Rep 15, 10107 (2025). https://doi.org/10.1038/s41598-025-94594-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-94594-x