Abstract
To explore the relationship between the Body Roundness Index (BRI) and the prevalence of osteoarthritis (OA) and rheumatoid arthritis (RA) among American adults, providing new insights for identifying OA and RA in adults. We analyzed data from the National Health and Nutrition Examination Survey (NHANES) 2015–2023 and conducted a large cross-sectional study. BRI was calculated based on body measurements, while OA and RA cases were identified through questionnaires. Participants under 20 years of age and those with incomplete data were excluded. Weighted multivariate logistic regression models, restricted cubic spline (RCS) functions, and stratified analyses were used to assess the relationship between BRI levels and the prevalence of OA and RA in American adults. To further evaluate BRI’s diagnostic potential for OA and RA, receiver operating characteristic (ROC) curves were employed to analyze and calculate the area under the curve (AUC). After screening, 17,544 participants were included, with 2,382 cases of OA (13.58%) and 987 cases of RA (5.63%). Multivariate logistic regression analyses showed a positive correlation between BRI and OA prevalence in American adults in both the unadjusted and adjusted models. A similar correlation was observed for RA in the unadjusted and partially adjusted models (P < 0.001), but the fully adjusted model showed no significant association between BRI and RA (P > 0.05). In the unadjusted model, the prevalence of OA in the highest BRI quartile was 3.47 times than that of the lowest quartile (95% CI: 2.84, 4.24, P < 0.001). Even in the fully adjusted model, the prevalence of OA in the highest BRI quartile remained 1.46 times higher than that of the lowest quartile (95% CI: 1.02, 2.08, P < 0.05). RCS curves demonstrated a non-linear relationship between BRI and both OA and RA, with a significant increase in prevalence as BRI levels rose (P < 0.001). Subgroup analyses and forest plots indicated a positive correlation between BRI and OA and RA in most subgroups (P < 0.05). ROC curves showed that BRI had a better predictive ability for OA and RA risk compared to BMI. There is a significant positive correlation between BRI and the prevalence of OA and RA in American adults, especially OA. Maintaining a lower BRI may help prevent the onset of OA and RA.
Similar content being viewed by others
Introduction
Osteoarthritis (OA) and rheumatoid arthritis (RA) are the two most common chronic joint diseases, significantly impacting patients’ quality of life and posing a heavy burden on global public health systems1,2. According to the World Health Organization, OA is the most common form of arthritis worldwide, and its incidence increases with age. A study by Safiri et al.3reported that the global prevalence of OA exceeds 20%, with the condition being especially prevalent among the elderly population3,4,5. RA, on the other hand, is a chronic, systemic autoimmune disease characterized by a high disability rate and is more common among middle-aged women6,7. A global, regional, and national study on the burden of RA estimated that in 2020, 17.6 million people worldwide were affected by RA, with projections suggesting that by 2050, this number will rise to 31.7 million8. As the global population continues to age, the incidence of both OA and RA is increasing, making it crucial to understand the risk factors for these diseases in order to inform prevention and treatment strategies.
Obesity is considered a major risk factor for both OA and RA9,10. Obesity not only accelerates joint wear through mechanical pressure11 but also exacerbates systemic inflammation through the secretion of pro-inflammatory cytokines by visceral fat tissue12. However, the traditional body mass index (BMI), commonly used to assess obesity, has certain limitations9. BMI merely reflects the ratio of weight to height and cannot accurately distinguish between fat and muscle mass, nor can it assess the distribution of visceral fat13. Recent research by Xue et al.14 found that there may be a non-linear positive correlation between visceral fat metabolic score and OA risk, and that visceral fat metabolic score could serve as a more accurate indicator for diagnosing OA. Therefore, identifying more precise anthropometric measurements to better predict the impact of obesity on joint diseases is of significant research and clinical importance.
The Body Roundness Index (BRI) is a novel anthropometric tool that has garnered increasing attention in recent years. By combining waist circumference and height, BRI more accurately reflects body roundness and the distribution of visceral fat, effectively evaluating obesity-related health risks15. Previous studies have shown that BRI is closely associated with various chronic diseases, such as diabetes, cardiovascular disease, and metabolic syndrome16,17,18. However, research on the relationship between BRI and OA and RA remains limited, especially when using large, representative datasets. Exploring the potential link between BRI and arthritis could provide new perspectives for the early screening and prevention of joint diseases.
This study aims to assess the association between BRI and OA/RA using data from the National Health and Nutrition Examination Survey (NHANES) and to compare BRI with other traditional obesity measures, such as BMI, in predicting arthritis risk. The findings will provide theoretical support for the prevention and early intervention of arthritis. Additionally, this study may offer new insights for clinical practice and public health strategies. If BRI proves effective in early screening and intervention for arthritis, future healthcare providers and policymakers will be better equipped to identify high-risk individuals, reduce the prevalence and disability rates of arthritis, and ultimately alleviate the economic burden on society.
Materials and methods
Study population
This large cross-sectional study utilized data from the 2015–2023 cycles of the NHANES, conducted by the National Center for Health Statistics (NCHS). NHANES employs a multistage, stratified sampling method to collect data on various aspects of health and nutrition in the U.S. population through interviews, physical examinations, questionnaires, and laboratory testing. The NHANES study protocol was approved by the NCHS Institutional Review Board, and all participants provided informed consent. NHANES data are publicly available on its official website.
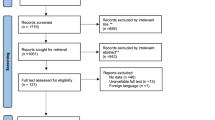
In this study, we included 37,464 individuals who participated in the 2015–2023 NHANES cycles. We excluded participants younger than 20 years of age and those with incomplete data for OA, RA, high-sensitivity C-reactive protein (HSCRP), BMI, waist circumference, height, white blood cell count (WBC), systemic inflammatory index (SII), physical activity, hypertension, smoking, alcohol consumption, or other covariates. After exclusions, 17,544 participants remained in the final analysis (Fig. 1).
Flowchart of Sample Selection. The study population consists of adults aged 20 and above.
Study variables
Exposure and outcomes
The main exposure of interest in this study was BRI, which was derived from the Body Measures under Examination Data. This dataset includes waist circumference (WC), height, and BMI measurements, and BRI was calculated using the following formula:

Outcome variables were the presence of OA and RA. These outcomes were determined from self-reported data in questionnaires. Participants were asked, “Has a doctor or other health professional ever told {you/SP} that {you/she/he} had arthritis (ar-thry-tis)?” If the response was “Yes,” they were further asked, “What type of arthritis?” Participants who responded “Rheumatoid arthritis” were classified as having RA, while those who responded “Osteoarthritis” were classified as having OA. Those who did not report these conditions were placed in the non-OA or non-RA groups, respectively.
Covariates
Several potential covariates that may influence the relationship between BRI and OA or RA were included in the analysis, such as age (y), gender, race, BMI (kg/m2), serum HSCRP levels (mg/L), serum WBC levels (x 103 cells/uL), SII, hypertension, physical activity, smoking, and alcohol consumption. Age, gender, race, physical activity, smoking, and alcohol data were obtained from questionnaires. Race was categorized as Mexican American, Other Hispanic, Non-Hispanic White, Non-Hispanic Black, and Other Races (including multi-racial individuals). BMI was categorized into three groups based on obesity status (< 25, 25–30, and > 30). Smoking status was based on the question: Smoked at least 100 cigarettes in life, divided into smoking and non-smoking groups. Physical activity was classified as heavy, moderate, or insufficient based on the questionnaire responses regarding vigorous recreational or work-related activities. Hypertension was defined based on self-reported physician diagnosis or average systolic blood pressure ≥ 130 mmHg or diastolic blood pressure ≥ 80 mmHg, following established guidelines19. Alcohol consumption was categorized as heavy (≥ 3 drinks/day for men or ≥ 2 drinks/day for women), moderate (1–2 drinks/day for men or 1 drink/day for women), or non-drinker20. Serum HSCRP and WBC levels were obtained from laboratory data, while the SII was calculated using the formula: SII = (platelet count × neutrophil count) / lymphocyte count.
Statistical analysis
All analyses were weighted according to NHANES guidelines. Continuous variables that were normally distributed were presented as weighted means ± standard deviation (SD) and compared using weighted linear regression. Categorical variables were presented as weighted percentages and compared using the weighted χ² test. To evaluate the relationship between BRI and OA and RA, we used weighted multivariate logistic regression models, adjusting for covariates in three models:
-
Model 1: No covariate adjustment.
-
Model 2: Adjusted for age, gender, and race.
-
Model 3: Adjusted for model 2 variables plus HSCRP, WBC, SII, BMI, hypertension, physical activity, smoking, and alcohol consumption.
To assess the non-linear relationship between BRI and OA and RA, we used restricted cubic splines (RCS). The diagnostic performance of BRI versus BMI for OA and RA was compared using ROC curves, and the area under the curve (AUC) was used to assess the predictive ability. Weighted subgroup analyses and forest plots were used to evaluate the relationship between BRI and OA/RA in different subgroups, and P values were calculated in age-stratified analyses to assess the effect of age on the relationship between BRI and arthritis. Statistical analyses were performed using R software (version 4.2.0), with statistical significance defined as P < 0.05.
Results
Participant characteristics
A total of 17,544 participants met the inclusion criteria, with 2,382 individuals diagnosed with OA (prevalence 13.58%) and 987 individuals diagnosed with RA (prevalence 5.63%). Significant differences were found between OA and non-OA participants in terms of gender, age, race, BRI, BMI, HSCRP, SII, hypertension, physical activity, smoking, and alcohol consumption (P < 0.01), while no significant differences were found in WBC levels between the two groups (Table 1). Significant differences were also found between RA and non-RA participants in age, race, BRI, BMI, HSCRP, SII, WBC levels, hypertension, physical activity, smoking, and alcohol consumption (P < 0.05) (Table 2).
Association between BRI and OA/RA
Weighted multivariate logistic regression analyses indicated a positive correlation between BRI and OA in all three models (P < 0.001). In the unadjusted model, the prevalence of OA in the highest BRI quartile was 3.47 times than that of the lowest quartile (95% CI: 2.84, 4.24, P < 0.001). Even in the fully adjusted model, the prevalence of OA in the highest BRI quartile remained 1.46 times higher than that of the lowest quartile (95% CI: 1.02, 2.08, P < 0.05) (Table 3). The unadjusted and partially adjusted models showed a positive correlation between BRI and RA (P < 0.001), but this association became non-significant in the fully adjusted model (P > 0.05) (Table 4).
BRI and OA/RA: non-linear relationships
RCS analyses demonstrated a non-linear relationship between BRI and both OA and RA in all three models (P < 0.05). As BRI increased, the risk of both OA and RA also increased (Fig. 2).
Restricted cubic spline curve (RCS) plot of the relationship between BRI and OA/RA risk under different models. The solid blue line represents the odds ratio (OR) of OA/RA. The shaded area represents the 95% confidence interval.
Comparison of BRI and BMI for OA/RA diagnosis
ROC curve analyses revealed that BRI had a better AUC for diagnosing OA than BMI. Similarly, BRI outperformed BMI in diagnosing RA, indicating that BRI is a more accurate predictor of OA and RA risk (Fig. 3; Tables 5 and 6).
ROC plot of BRI and BMI for diagnosis of OA/RA, blue shaded area represents area under the curve. ROC: receiver operating characteristic, BRI: body roundness index, BMI: body mass index.
Subgroup analysis of BRI and OA/RA
The subgroup analysis of BRI and OA demonstrated a significant positive correlation across all subgroups for gender, race, smoking status, hypertension, alcohol consumption, and physical activity, as well as in the 41–60 and > 60 age groups and the BMI > 30 group (Fig. 4). Interaction tests showed significant interactions for gender, race, smoking status, and hypertension (P < 0.05). Similarly, in the subgroup analysis of BRI and RA, a positive correlation was observed in all subgroups except for the Insufficient Physical Activity group and the BMI < 25 group (P < 0.05). However, interaction tests indicated no significant interactions between subgroups for RA (P > 0.05) (Fig. 5).
Subgroup analysis of the relationship between BRI and OA. OR: Odds Ratio, CI: Confidence Interval, BMI: body mass index.
Subgroup analysis of the relationship between BRI and RA. OR: Odds Ratio, CI: Confidence Interval, BMI: body mass index.
Discussion
This study is one of the first large-scale cross-sectional analyses exploring the association between BRI and OA/RA, providing new evidence for the application of BRI in arthritis risk assessment. Our findings demonstrate that BRI outperforms BMI in predicting OA risk, supporting its potential use as a future clinical screening tool. We identify that BMI may underestimate OA risk in certain individuals, particularly those with normal BMI but elevated BRI, emphasizing the need for further research on the role of BRI. The results of this study revealed a significant positive association between BRI and the prevalence of OA and, to a lesser extent, RA in American adults. These findings highlight the potential of BRI as a novel tool for assessing obesity-related health risks, particularly in joint diseases like OA. Compared to traditional BMI, BRI offers a more accurate reflection of visceral fat distribution, making it a better predictor of disease risk. Our results showed that gender, race, smoking, hypertension, drinking status and age 41–60, > 60 group and BMI > 30 group, BRI was positively associated with OA and RA. And the results of age subgroup analysis showed that the associations between BRI and OA and RA were more consistent in different age groups, but P for interaction > 0.05, indicating that the interaction effect of age on the relationship between BRI and OA and RA was not significant. These results highlight the potential clinical applications of the BRI in different populations and support the need for personalized health assessment.
Osteoarthritis and BRI: a strong correlation
One of the main findings of this study is the significant correlation between higher BRI and OA. Specifically, participants in the highest BRI quartile had a 2.01 times higher risk of developing OA compared to those in the lowest quartile (95% CI: 1.58–2.55, P < 0.001), which is consistent with previous studies showing that obesity, especially visceral obesity, is a major risk factor for OA14,21,22. The pathogenesis of OA is multifactorial, but mechanical loading on the joints and the influence of metabolic and inflammatory processes are thought to be primary factors11,23,24. Obesity, as reflected by BRI, exacerbates both mechanical and systemic inflammatory factors, leading to the worsening of OA. Excess weight places excessive stress on weight-bearing joints, such as the knees and hips, accelerating cartilage wear and degeneration. This mechanical damage is one of the main pathways through which obesity influences OA risk11. Studies have shown that for every 1 kg reduction in body weight, the load on the knee joint is reduced by more than twofold, significantly decreasing the risk of joint damage25.
In addition to mechanical stress, the role of adipose tissue in systemic inflammation has gained increasing recognition. Obesity, particularly central or visceral obesity, promotes the release of pro-inflammatory cytokines such as interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), and adipokines from adipose tissue. These cytokines not only exacerbate low-grade systemic inflammation but also directly contribute to cartilage destruction and joint inflammation23,24. Research has shown that the accumulation of visceral fat is positively correlated with the development of OA14, reflecting the strong impact of visceral fat and related inflammatory processes on joint health. BRI, which better reflects these factors, has emerged as a new tool for studying the relationship between obesity and OA.
Rheumatoid arthritis and BRI: a more complex relationship
In contrast, the association between BRI and RA became non-significant after adjustment, suggesting that the pathophysiology of RA may be more complex, involving multiple immune system factors26,27. In our study, although a positive correlation between BRI and RA was observed in the unadjusted model, this association disappeared after multivariate adjustment. This finding aligns with other studies that emphasize the complicated relationship between RA and body weight28,29,30,31. Among RA patients, obesity is closely associated with decreased joint function and lower quality of life. Several studies have shown that overweight RA patients typically require more aggressive treatment and respond less effectively to therapies10,32,33. Marchand et al.34, in a large prospective cohort study involving 2,583,266 individuals, examined the relationship between long-term weight changes and RA risk, finding that weight gain over time significantly increased RA risk in women, with a threefold increase in RA risk for those gaining ≥ 20 kg. Similarly, a large cross-sectional study by Ferguson et al.35, involving 502,417 participants, demonstrated a positive association between central obesity, as measured by waist circumference, and the incidence of RA. However, compared to OA, the relationship between RA and obesity remains less understood. It likely involves a combination of factors, including inflammatory responses, immune system dysregulation, and metabolic abnormalities. Some studies suggest that obesity may exacerbate disease severity by increasing systemic inflammation31. Additionally, obesity may impair immune tolerance by promoting inflammation and reducing regulatory B (Breg) and regulatory T (Treg) cells, which leads to an increase in Th17 and Th1 cells, creating an environment conducive to the development of autoimmune diseases36.
Clinical implications and preventive strategies
Traditionally, BMI has been widely used to assess obesity. However, because it does not account for fat distribution and composition, many researchers have sought more precise measurement tools. As a novel obesity assessment index, the BRI offers the advantage of more accurately reflecting visceral fat distribution, providing a more effective evaluation of health risks compared to BMI. Recent research by Xue et al.14 indicates a potential non-linear positive correlation between visceral fat metabolic score and OA risk, suggesting that the metabolic score of visceral fat could serve as a more accurate diagnostic indicator for OA. As the prevalence of OA and RA continues to rise, adopting BRI for risk assessment could present new opportunities for early screening and intervention. Our ROC analysis results demonstrated that BRI is more accurate than BMI in predicting the risks of OA and RA. This finding suggests that clinicians could integrate BRI into routine examinations to better identify high-risk individuals.
Recent research by Zhang et al.37 revealed a U-shaped relationship between BRI and all-cause mortality, indicating that both excessively low and high BRI levels are associated with increased mortality risk. This further suggests that BRI is not only a predictor of joint health issues but also an important metric for evaluating overall health and mortality risk. Since BRI is highly sensitive to individual fat distribution, it could be used in the future for clinical health assessments and large-scale public health initiatives as a key tool for preventing and managing obesity-related diseases38,39,40.
Moreover, the widespread application of BRI could advance the practice of personalized medicine. By providing more accurate assessments of individual obesity risk, physicians can offer more targeted treatment recommendations based on BRI values, such as visceral fat reduction strategies or other lifestyle interventions, to mitigate the development of arthritis and other metabolic diseases.
Limitations and future directions
Although this study provides valuable insights, several limitations should be considered. First, the cross-sectional nature of the study prevents us from establishing a causal relationship between BRI and arthritis. Longitudinal studies are needed to confirm the temporal association between BRI and the development of OA and RA. Second, the reliance on self-reported data for OA and RA diagnosis may introduce misclassification bias. Future studies should incorporate objective diagnostic measures, such as radiographic confirmation, to improve the accuracy of findings. Finally, some participants had a combination of both OA and RA, but given the relatively low prevalence of RA compared with OA and the small number of overlapping cases, separate subgroup analyses were less statistically reliable. Future studies with larger sample sizes are needed to further explore the implications of dual diagnosis. Additionally, while BRI shows promise as a risk assessment tool for joint diseases, further research is needed to compare its predictive ability with other anthropometric indices, such as waist-to-hip ratio and visceral fat area.
Conclusion
In conclusion, this study demonstrates a significant positive association between BRI and the prevalence of OA and, to a lesser extent, RA in American adults. BRI may serve as a valuable tool for assessing arthritis risk, particularly in populations with high levels of central obesity. Given the rising global burden of arthritis, incorporating BRI into clinical practice could help identify high-risk individuals and implement early interventions aimed at reducing obesity and inflammation. Future studies should focus on validating these findings in diverse populations and exploring the potential for BRI to guide personalized prevention and treatment strategies for arthritis.
Data availability
All data are available in the NHANES database (www.cdc.gov/nchs/nhanes).
Abbreviations
- BRI:
-
Body Roundness Index
- OA:
-
Osteoarthritis
- RA:
-
Rheumatoid arthritis
- NHANES:
-
National Health and Nutrition Examination Survey
- RCS:
-
Restricted cubic spline
- ROC:
-
Receiver operating characteristic
- AUC:
-
Area under the curve
- OR:
-
Odds ratios
- CI:
-
Confidence intervals
- BMI:
-
Body mass index
- HSCRP:
-
High-sensitivity C-reactive protein
- WBC:
-
White blood cell count
- SII:
-
Systemic inflammatory index
- WC:
-
Waist circumference
References
Pincus, T. et al. Osteoarthritis is as severe as rheumatoid arthritis: evidence over 40 years according to the same measure in each disease. Clin. Exp. Rheumatol. 37 (Suppl 120(5), 7–17 (2019).
Reginster, J. Y. The prevalence and burden of arthritis. Rheumatology 41 (Supp 1), 3–6 (2002).
Safiri, S. et al. Global, regional and National burden of osteoarthritis 1990–2017: a systematic analysis of the global burden of disease study 2017. Ann. Rheum. Dis. 79 (6), 819–828 (2020).
Sun, X. et al. Osteoarthritis in the Middle-Aged and elderly in China: prevalence and influencing factors. Int. J. Env Res. Pub He. 16 (23), 4701 (2019).
Abramoff, B., Caldera, F. E. & Osteoarthritis Pathology, diagnosis, and treatment options. Med. Clin. 104 (2), 293–311 (2020).
Almutairi, K., Nossent, J., Preen, D., Keen, H. & Inderjeeth, C. SAT0576 THE PREVALENCE OF RHEUMATOID ARTHRITIS: A SYSTEMATIC REVIEW OF POPULATION-BASED STUDIES. Ann. Rheum. Dis. 79 (Suppl 1), 1246 (2020).
Rudan, I. et al. Prevalence of rheumatoid arthritis in low- and middle-income countries: A systematic review and analysis. J. Glob Health. 5 (1), 10409 (2015).
Black, R. J. et al. Global, regional, and National burden of rheumatoid arthritis, 1990–2020, and projections to 2050: a systematic analysis of the global burden of disease study 2021. Lancet Rheumatol. 5 (10), e594–e610 (2023).
Godziuk, K. & Hawker, G. A. Obesity and body mass index: past and future considerations in osteoarthritis research. Osteoarthr. Cartil. 32 (4), 452–459 (2024).
Dubovyk, V. et al. Obesity is a risk factor for poor response to treatment in early rheumatoid arthritis: a NORD-STAR study. Rmd Open. 10 (2), e004227 (2024).
Batushansky, A. et al. Fundamentals of OA. An initiative of osteoarthritis and cartilage. Obesity and metabolic factors in OA. Osteoarthr. Cartil. 30 (4), 501–515 (2022).
Wang, T., He, C. & Yu, X. Pro-Inflammatory cytokines: new potential therapeutic targets for Obesity-Related bone disorders. Curr. Drug Targets. 18 (14), 1664–1675 (2017).
Gutin, I. Body mass index is just a number: conflating riskiness and unhealthiness in discourse on body size. Sociol. Health Ill. 43 (6), 1437–1453 (2021).
Xue, H. et al. Association of the visceral fat metabolic score with osteoarthritis risk: a cross-sectional study from NHANES 2009–2018. Bmc Public. Health. 24 (1), 2269 (2024).
Schweitzer, K. Could the body roundness index one day replace the BMI? Jama-J Am. Med. Assoc. 16 (332), 1317–1318 (2024).
Yang, M. et al. Body roundness index trajectories and the incidence of cardiovascular disease: evidence from the China health and retirement longitudinal study. J. Am. Heart Assoc. 13 (19), e034768 (2024).
Qiu, L., Xiao, Z., Fan, B., Li, L. & Sun, G. Association of body roundness index with diabetes and prediabetes in US adults from NHANES 2007–2018: a cross-sectional study. Lipids Health Dis. 23 (1), 252 (2024).
Konstantinova, E. D., Maslakova, T. A. & Ogorodnikova, S. Y. The predictive capability of several anthropometric indices for identifying the risk of metabolic syndrome and its components among industrial workers. Sci. Rep-Uk. 14 (1), 15327 (2024).
Whelton, P. K. et al. /ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018; 71(19):e127-e248. (2017).
Willenbring, M. L., Massey, S. H. & Gardner, M. B. Helping patients who drink too much: an evidence-based guide for primary care clinicians. Am. Fam Physician. 80 (1), 44–50 (2009).
Sampath, S., Venkatesan, V., Ghosh, S., Kotikalapudi, N. & Obesity Metabolic syndrome, and Osteoarthritis-An updated review. Curr. Obes. Rep. 12 (3), 308–331 (2023).
Zhang, Y. et al. Visceral adipose tissue and osteoarthritis, a two-sample Mendelian randomized study. Front. Med-Lausanne. 10, 1324449 (2023).
Kraus, V. B. et al. Direct in vivo evidence of activated macrophages in human osteoarthritis. Osteoarthr. Cartil. 24 (9), 1613–1621 (2016).
De Roover, A., Escribano-Núñez, A., Monteagudo, S. & Lories, R. Fundamentals of osteoarthritis: inflammatory mediators in osteoarthritis. Osteoarthr. Cartil. 31 (10), 1303–1311 (2023).
Aaboe, J., Bliddal, H., Messier, S. P., Alkjær, T. & Henriksen, M. Effects of an intensive weight loss program on knee joint loading in obese adults with knee osteoarthritis. Osteoarthr. Cartil. 19 (7), 822–828 (2011).
Brown, P., Pratt, A. G. & Hyrich, K. L. Therapeutic advances in rheumatoid arthritis. Bmj-Brit Med. J. 384, e070856 (2024).
Li, R. et al. FTO-mediated RNA m(6)A methylation regulates synovial aggression and inflammation in rheumatoid arthritis. Bba-Mol Basis Dis. 1870 (7), 167341 (2024).
Qin, B. et al. Body mass index and the risk of rheumatoid arthritis: a systematic review and dose-response meta-analysis. Arthritis Res. Ther. 17 (1), 86 (2015).
Ohno, T., Aune, D. & Heath, A. K. Adiposity and the risk of rheumatoid arthritis: a systematic review and meta-analysis of cohort studies. Sci. Rep-Uk. 10 (1), 16006 (2020).
Chung, M. K. et al. DATA FROM A NATIONWIDE COHORT. Ann. Rheum. Dis. 82 (Suppl 1), 601 (2023).
Jalal, A. M., Mawlood, Z. K., Aref, S. F. & Al-Nimer, M. S. Association between the quality of life domains with anthropometric and adiposity indices in rheumatoid arthritis: A Cross-Sectional study from a single centre in Erbil-Iraq. Mediterr. J. Rheumatol. 35 (2), 247–254 (2024).
Tsoi, M. F. et al. Association between body weight and tocilizumab effectiveness in rheumatoid arthritis: results from the BSRBR-RA. Rheumatology. :keae500. (2024).
Baker, J. F., Reed, G., Poudel, D. R., Harrold, L. R. & Kremer, J. M. Obesity and response to advanced therapies in rheumatoid arthritis. Arthrit Care Res. 74 (11), 1909–1916 (2022).
Marchand, N. E. et al. Long-term weight changes and risk of rheumatoid arthritis among women in a prospective cohort: a marginal structural model approach. Rheumatology 61 (4), 1430–1439 (2022).
Ferguson, L. D. et al. Association of central adiposity with psoriasis, psoriatic arthritis and rheumatoid arthritis: a cross-sectional study of the UK biobank. Rheumatology 58 (12), 2137–2142 (2019).
Tsigalou, C., Vallianou, N. & Dalamaga, M. Autoantibody production in obesity: is there evidence for a link between obesity and autoimmunity?? Curr. Obes. Rep. 9 (3), 245–254 (2020).
Zhang, X. et al. Body roundness index and All-Cause mortality among US adults. Jama Netw. Open. 7 (6), e2415051 (2024).
Rico-Martín, S. et al. Effectiveness of body roundness index in predicting metabolic syndrome: A systematic review and meta-analysis. Obes. Rev. 21 (7), e13023 (2020).
Calderón-García, J. F. et al. Effectiveness of body roundness index (BRI) and a body shape index (ABSI) in predicting hypertension: A systematic review and Meta-Analysis of observational studies. Int. J. Env Res. Pub He. 18 (21), 11607 (2021).
Li, C. I. et al. Association of body indices and risk of mortality in patients with type 2 diabetes. Bmj Open. Diab Res. Ca. 11 (4), e003474 (2023).
Acknowledgements
Thanks to NHANES database participants and staff.
Author information
Authors and Affiliations
Contributions
Tao Gao and Chao Wu analyzed and interpreted the patient data regarding the OA. Tao Gao were major contributors in writing the manuscript. Zhi-Yu Chen, Tao Li, Xu Lin, Hai-Gang Hu and Jian-Dong Tang assisted with data collection. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study protocol was approved by the NCHS Ethics Review Board, and all participants provided informed consent.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gao, T., Chen, ZY., Li, T. et al. Association between body roundness index and osteoarthritis/rheumatoid arthritis: a cross-sectional study. Sci Rep 15, 9682 (2025). https://doi.org/10.1038/s41598-025-94606-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-94606-w
Keywords
This article is cited by
-
Body roundness index and mortality risk in chronic diseases: a national prospective longitudinal study in China
Aging Clinical and Experimental Research (2025)